Abstract
Multiple laminin isoforms including laminins 5 (α3 β3 γ2), 6 (α3 β1 γ1), 10 (α5 β1 γ1), and possibly laminins 7 (α3 β2 γ1) and 11 (α5 β2 γ1) are present in the epidermal basement membrane. However, only the precise epidermal ultrastructural localization of laminin 5 (α3 β3 γ2) has been elucidated. We therefore determined the precise expression and ultrastructural localization of the α5, β1, β2, and γ1 chains in the epidermis. The expression of laminin chains in skin samples was analyzed from patients with epidermolysis bullosa (EB, n=15) that harbor defects in specific hemidesmosome (HD)-associated components. The expression of the α5, β1, and γ1 chains (present in laminins 10/11) and β2 chain (laminins 7/11) was unaffected in all intact (unseparated) skin of EB patients including Herlitz junctional EB with laminin-5 defects (n=6). In the basement membrane of human epidermis, the α5, β1, β2, and γ1 chains were expressed but also localized to the dermal vessels. Immunogold electron microscopy of normal human epidermis localized the α5, β1, β2, and γ1 chains to the upper lamina densa, with between 84% and 92% of labeling restricted to beneath the HDs, similar to laminin 5 (n200 gold particles per sample, sample number n=4) but distinct from collagen IV labeling (with only 63% labeling beneath HDs, p<0.001). Taken together, the majority of the α5β1/β2γ1 laminin chains are located beneath HDs. This suggests that laminin-10-associated chains have specific functions or molecular interactions beneath HDs in the epidermal basement membrane.
Keywords
I
The distinct roles of laminin isoforms in the processes of cutaneous morphogenesis are poorly understood. Laminin 10 (α5 β1 γ1), however, has recently been implicated in several functions including hair follicle development (Li et al. 2003). In an α5 chain (laminin 10/11) knockout mouse model, the addition of exogenous laminin 10 was used to correct follicular development (Li et al. 2003). Laminin 10 is therefore implicated in hair follicle cell growth and adhesion (Gu et al. 2001; Pouliot et al. 2002; Li et al. 2003). Cell adhesion assays have demonstrated that multiple laminins (including laminins 5, 10, and 11) can act as adhesive substrates for keratinocytes and that this adhesion is mediated by the integrins α3β1 and α6β4 (Pouliot et al. 2002). However, in certain non-epithelial cells, the integrins α3β1, α6β1, and α6β4 and α dystroglycan are expressed and have been identified as possible laminin 10/11 receptors (Kikkawa et al. 1998, 2000; Yu and Talts 2003).
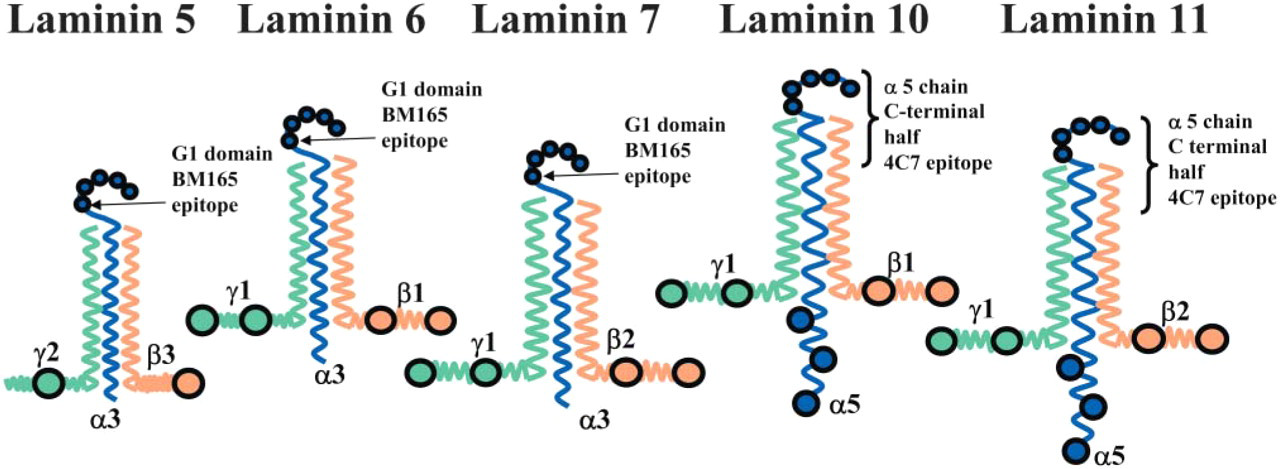
Schematic diagram showing the structure of laminin 5 and 10 chains and the position of two antibody binding sites in the α3 chain G1 domain (BM 165) and the carboxyl terminal half of the α5 chain (4C7). This schematic diagram is not drawn to scale and does not include any α3 chain splice variants of laminin 5 (α3 β3 γ2). The antibody BM165 binds to the first globular (G1) domain of the α3 chain of laminins 5/6 (McMillan et al. 2003b), whereas the antibody 4C7 binds to the carboxyl terminal half end of the α5 chain (present in both laminins 10 and 11).
Antibodies are now available that recognize specific laminin chains and provide new tools to investigate the structure of the epidermal basement membrane. These antibodies include 4C7 (Engvall et al. 1990; Tiger et al. 1997) that recognizes a carboxyl terminal domain of the human α5 chain of laminins 10 (α5 β1 γ1, see Figure 1) and 11 (α5 β2 γ1). This antibody blocks the epitope involved in neurite cell adhesion to the laminin α5 chain (Engvall et al. 1986; Makino et al. 2002). Further laminin-specific antibodies to β and γ chains include 2E8 (recognizing the β1 chain (Engvall et al. 1986), D18 (γ1) (Sanes et al. 1990), and C4 (β2) (Hunter et al. 1989).
To better understand the position and possible functions of epidermal molecules, we examined the precise localization of the α5, β1, β2, and γ1 laminin chains and collagen IV in the interfollicular and follicular epidermal basement membrane. In addition, the expression of laminin chains was also assessed in a range of epidermolysis bullosa (EB) patients’ skin harboring defects in several basement membrane components, including laminin 5. We have quantitatively analyzed and compared the localizations of laminin α5, β1, β2, and γ1 chains with that of laminin 5 (α3 β3 γ2) from our previously published data (McMillan et al. 2003b) and collagen IV. Comparison of α5, β1, β2, and γ1 chain expression with laminin 5, a well-studied HD-associated isoform, will determine a more precise localization for these laminin isoforms. Our data support the hypothesis that multiple laminin isoforms colocalize beneath HDs in normal and diseased epidermal basement membranes.
Materials and Methods
Skin Samples
Samples of adult and neonatal control skin from non-spe cialized sites (abdomen, arm, thigh, n=8; and scalp skin, n=2) were obtained from routine surgical procedures. Skin samples were frozen for cryostat sectioning or processed for post-embedding immunogold electron microscopy (IEM) as described below. In all cases, the biopsies were performed with the patient's or guardian's informed consent, with the relevant institutional approval for experiments handling human material, and in accordance with the Helsinki Declaration.
Skin samples from patients affected with a group of rare genodermatoses, EB, were included in this study (n=15, see Table 1). Details of the number of patients for each EB disease subtype, their age at biopsy, details of any identified mutations, or significant results of diagnostic antibody staining are listed, in addition to the results of their laminin antibody staining findings (see Table 1). Four Herlitz junctional (HJ) EB patients harbored laminin-5 chain mutations that were reported in the literature (Takizawa et al. 1998a-d). In one EB simplex associated with muscular dystrophy (EBS-MD) patient, genetic defects have been reported (Pulkkinen et al. 1996).
Confocal Immunofluorescence Microscopy
Indirect immunofluorescence was performed as previously described (Kennedy et al. 1985) using cryostat skin sections. Laminin chain expression was confirmed in control skin using the following antibodies: 4C7 recognizing the human α5 chain (see Figure 1) present in laminins 10 and 11 (dilution 1:25; Chemicon International, Temecula, CA) (Engvall et al. 1990; Tiger et al. 1997). The monoclonal antibody 2E8 recognizing the β1 chain (neat) (Engvall et al. 1986), the monoclonal antibody D18 that recognizes the γ1 chain (see Figure 1) (neat) (Sanes et al. 1990), and an antibody C4 to the β2 chain (see Figure 1) (used neat) (Hunter et al. 1989) were also included. The antibodies 2E8, D18, and C4 were obtained from the Developmental Studies Hybridoma Bank, University of Iowa (Iowa City, IA). The mouse monoclonal M3F7 recognizing the helical domain of the α1 and α2 chains of collagen IV (used neat) (Foellmer et al. 1983) was also obtained from the Developmental Studies Hybridoma Bank. Laminin-5 antibodies included the mouse monoclonal BM 165 directed against the laminin-5 α3 chain terminal first globular (G1) domain (see Figure 1) (diluted 1:50) (Marinkovich MP, unpublished data) (McMillan et al. 2003b); K140 directed against the laminin-5 β3 chain adjacent to domain IV; GB3 directed against the laminin-5 γ2 chain (Harlan Sera Lab; Loughborough, UK); and a rabbit polyclonal serum directed against the entire laminin-5 molecule (1:200) (McMillan et al. 2003b). The melanocyte marker antibody TMH-1 recognized the b-locus protein (rat antibody, 1:10 dilution) and was previously described by Masunaga et al. (1996).
Comparison of laminin 5 and laminin 10 expression in patients with various forms of epidermolysis bullosa

EB, epidermolysis bullosa; HJEB, Herlitz junction epidermolysis bullosa; NHJEB, non-Herlitz junctional epidermolysis bullosa; SRDEB, severe recessive dystrophic epidermolysis bullosa; JEB-PA, junctional epidermolysis bullosa associated with pyloric atresia; which is also known as EBS-PA, epidermolysis bullosa associated with pyloric atresia. +++, normal, bright staining pattern along dermal-epidermal junction; ++, reduced dermal-epidermal junction staining compared to controls; +, severely reduced dermal-epidermal junction staining compared to controls; -, absent dermal-epidermal junction staining compared to controls.
Epidermal sections were fixed in cold acetone (-20C) for 10 min and incubated with 5% normal rabbit sera in 0.1 M Dulbecco's PBS for 5 min at 37C. Sections were incubated with primary antibodies and subsequently with secondary antibodies conjugated to fluorescein isothiocyanate or Texas Red (FITC; rabbit anti-mouse IgG or goat anti-rabbit IgG, 1:200; DAKO, Tokyo, Japan; Texas Red conjugated donkey anti-rabbit; Amersham, UK). To label TMH-1, a preabsorbed cyanine (CY5)-conjugated goat anti-rat antibody was used (Jackson ImmunoResearch; West Grove, PA). All secondary antibodies were diluted in 3% BSA in 0.1 M PBS for 30 min at 37C in a darkened, humidified chamber. Sections were then labeled with a ToPro-3 nuclear counterstain (diluted 1:20,000, blue channel; Jackson ImmunoResearch) if appropriate. The sections were then mounted in Permafluor (Thermo Shandon; Pittsburgh, PA) and examined with a confocal microscope (Fluoview FV300; Olympus, Tokyo, Japan) using an inverted microscope (IX70; Olympus). Controls included normal skin cryostat sections with the primary antibody substituted by PBS, myeloma supernatant, or an irrelevant immunoglobulin isotype, as a negative control. All experiments were performed at least in duplicate.
Immunogold Electron Microscopy
Four samples of human skin were cryofixed and processed for postembedding IEM according to the previously described methods (Shimizu et al. 1989,1990). Samples were washed in PBS and cryoprotected in 20% glycerol (in PBS) for up to 1 hr at 4C. Subsequently, cryofixation was performed in liquid propane at −190C using a freeze plunge apparatus (Leica CPC; Cambridge, UK) followed by freeze substitution over 3 days at −80C in methanol using an automated freeze substitution system (AFS; Leica). Specimens were embedded in Lowicryl K11M (Ladd Research Industries; Burlington, VT) resin over 4 days at −60C. The temperature was gradually raised and the resin was polymerized under UV light and liquid nitrogen vapor at 10C. Ultrathin sections were then cut and collected on pioloform-coated nickel grids. Sections were stained with uranyl acetate only (15 min) and observed with a transmission electron microscope (Hitachi H-7100; Tokyo, Japan) at 75 kV. Blocks showing good ultrastructure were selected for immunolabeling experiments. Sections were preincubated in buffer containing PBS with 5% normal goat serum (NGS), 1% BSA, and 0.1% gelatin. Primary antibodies or human antisera were all diluted in PBS buffer containing 1% NGS, 1% BSA, and 0.1% gelatin and incubated at 37C for 2 hr. The sections were then washed in a drop of PBS buffer four times (5 min each) and placed on a drop of secondary linker antibody, again diluted in PBS buffer (for 2 hr at 37C). The secondary antiserum, rabbit anti-mouse IgG (DAKO; Ely, UK) was diluted 1:500. Sections were then incubated with a final antibody layer using 5-nm gold-conjugated labeled goat anti-rabbit or goat anti-mouse anti-bodies (Biocell; Cardiff, UK) diluted 1:500 in Tris-buffered saline (TBS) for 2 hr at 37C. For double labeling on K11M sections, the α5 chain of laminins 10/11 (4C7 and a 5-nm gold-conjugated goat anti-mouse) and rabbit anti-laminin 5 (polyclonal highlighted by 15-nm gold-conjugated goat anti-rabbit; Biocell) were used. Sections were washed twice in TBS buffer and twice in distilled water (5 min each). After staining with 15% alcoholic uranyl acetate (3 min) and lead citrate (15 min), sections were observed with a transmission electron microscope (H-7100; Hitachi). Controls included normal skin sections with the primary antibody substituted by PBS, myeloma supernatant, preimmune rabbit serum, or an irrelevant immunoglobulin isotype, as a negative control. All experiments were performed in triplicate.
Immunogold Quantitative Analysis
The techniques for ultrastructural labeling were similar to those performed by McMillan et al. (2003b). Electron micrographs were taken at a standard magnification (30K) and were enlarged by a standard factor X2.08. The final magnification (X62,500) was checked using electron micrographs taken of a carbon diffraction grating. For standardization purposes, all observations were made by one observer (JRM). At least 200 gold particles were assessed per specimen for each antibody or antiserum and four specimens from different individuals were examined (see Table 1 and Table 2). A 5-nm immunogold-conjugated final antibody layer was used. The percentage of gold particles perpendicularly beneath an observable electron-dense HD cytoplasmic outer attachment plaque as described by McMillan and Eady (1996) was scored and calculated from a large number of gold particles in skin from four individuals.
Only non-obliquely sectioned areas of dermal-epidermal junction were included with clearly defined HD plaques, lamina lucida (LL), and lamina densa (LD). The dermal-epidermal junction beneath melanocytes or in damaged areas was excluded from this study. Gold particles that appeared clumped or associated with any deposit were excluded.
For each antibody or antisera, the positions of gold particles were statistically tested by one-way ANOVA and a two-sample t-test using the Minitab statistical package (Minitab Inc; University of Pennsylvania, Philadelphia, PA). An anti-body (4C7) that recognizes a carboxyl terminal domain of the α5 chain of laminins 10/11 was used to determine the mean position of labeling directly beneath the keratinocyte plasma membrane (Engvall et al. 1986; Makino et al. 2002)(see Figure 1 for epitope position). The labeling of the α5 chain was compared with the distribution of the G1 domain of laminin-5 α3 chain (using data previously reported by McMillan et al. 2003b).
Results
Confocal Fluorescence Microscopy of Control Skin
Laminin-5 staining was restricted to the dermal-epidermal junction in control skin (data not shown). This was similar to the dermal-epidermal junction staining of α5 chain of laminins 10 (data not shown). Laminins 10 and 11 were also expressed in dermal blood vessels. Laminin 11 (as identified by the β2 chain) dermal-epidermal junction staining was present in adult control thigh and arm skin but was variable in other samples including scalp skin. Therefore, β2 chain expression appears to be distinct and independent from that of the α5 chain. Staining for the α5, β1, and γ1 chains was weaker in the adult dermal-epidermal junction than in blood vessels (data not shown), whereas dermal-epidermal junction staining was generally brighter in younger skin samples (<16 years, data not shown). This would appear to support a previous report of age-dependent expression of the laminin 10/11 chains (Pouliot et al. 2002).
Confocal fluorescence microscopy (Figures 2A–2C) showed that both laminin-5 and α5 chains are expressed in the dermal-epidermal junction of control interfollicular epidermis except for small gaps (white arrows in Figures 2A and 2B) beneath small isolated cells presumed to be melanocytes staining blue for the melanocyte marker, TMH-1 (Figures 2A and 2B) (Masunaga et al. 1996). These data suggest that laminin-10 chains are restricted to beneath keratinocytes and are not expressed beneath melanocytes.
A previous scalp skin and hair follicle immunohis-tochemical study (Akiyama et al. 1995) demonstrated a specific staining pattern for many HD and anchoring filament components, particularly laminin 5, which manifests as reduced staining around the lower hair bulb and a reemergence of staining over the dermal papilla region (Akiyama et al. 1995). We observed this characteristic pattern of staining along the majority of the follicles, for both laminin 5 (bracketed area in Figure 3A) and the laminin α5 chain (brackets and dotted line in Figures 3B and 3D). The laminin β1 and γ1 chains also showed this staining pattern (data not shown). There was no staining for the β2 chain in the hair follicle (data not shown). The dermal-epidermal junction of the bulge region stained for both α3 and the α5 chains (Figures 3A and 3B, Bu) and the α5 chain also stained the erector pili muscle (Figure 3C, EP).
Immunogold particle distribution shows the majority of laminin labeling is restricted to beneath the hemidesmosome (HD) plaque

Data from McMillan et al. 2003b and Masunaga et al. 1996.
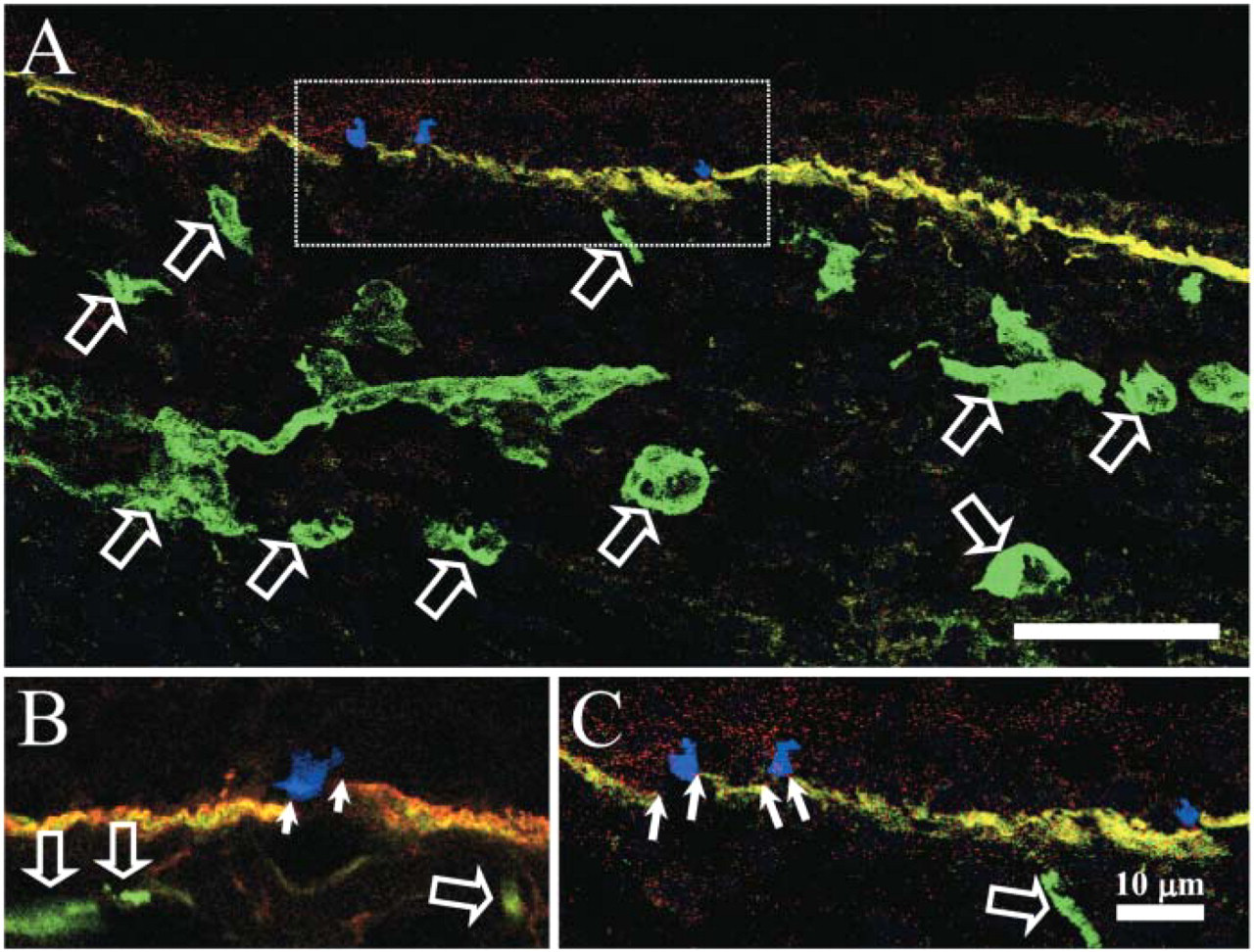
Reduced laminin 10 (green, FITC) and laminin 5 (red, Texas Red) labeling below the melanocyte marker TMH-1 (blue, CY-5) of normal adult control skin. Both laminin 5 and the α5 chain colocalize (orange color) within the dermal-epidermal junction (
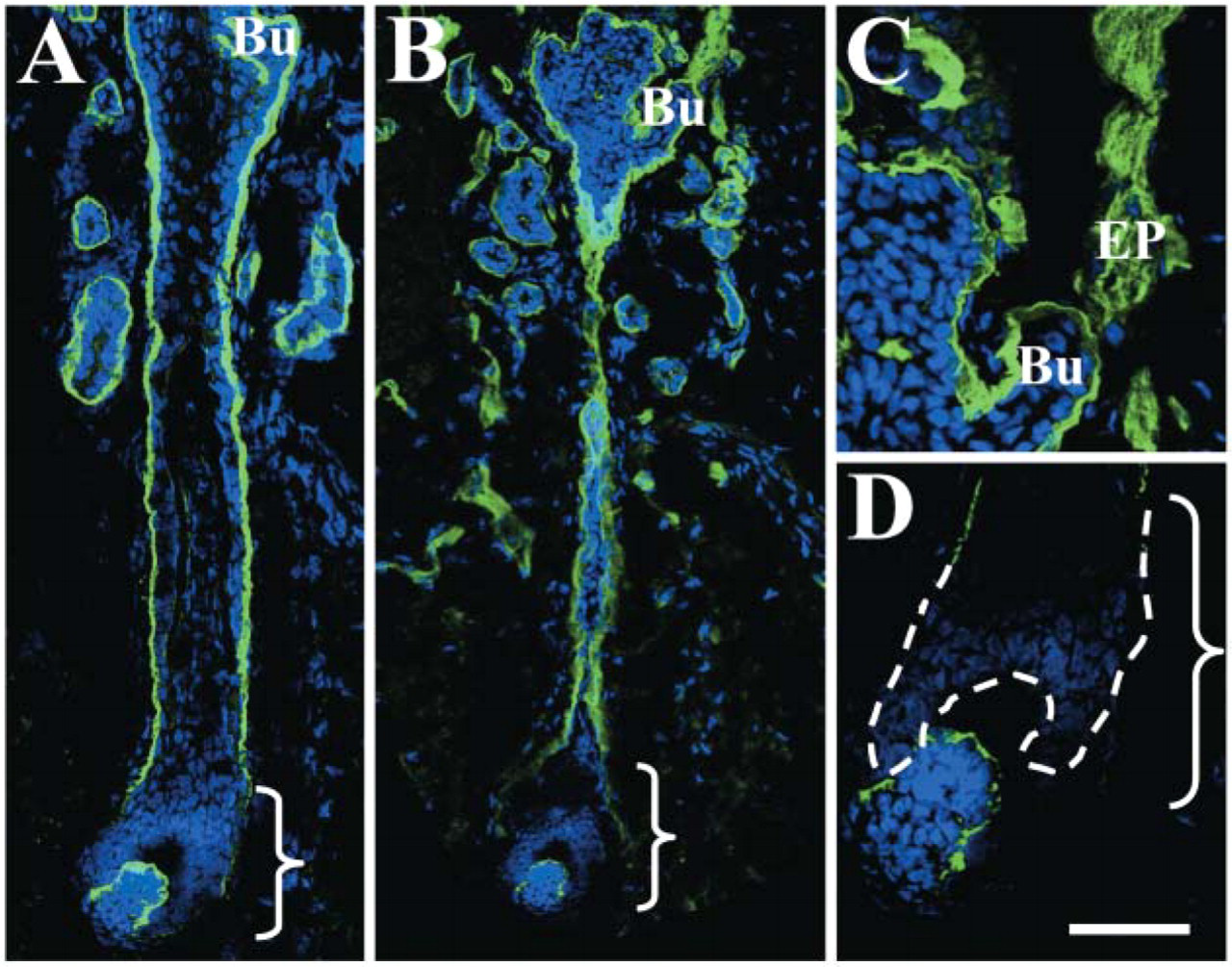
Indirect immunofluorescence shows a typical hemidesmosomal (HD) component-like expression pattern of the laminin α5 chain in late anagen human hair follicles. A previous scalp hair follicle immunohistochemical study (Akiyama et al. 1995) demonstrated a specific staining pattern for many HD-anchoring filament-associated components including laminin 5. In our study, both laminin-5 polyclonal staining (
Confocal Fluorescence Microscopy of Epidermolysis Bullosa Skin
The expression of laminins 5 (γ2 chain), 10 (all chains), and 11 (β2 chain) in patients with different forms of EB was compared. In both control (Figure 4A) and all of the EB subtypes (Figures 4B–4H), α5, β1, β2, and γ1 chain expression was detectable. Laminin expression was weak in areas of split skin particularly in HJEB with defects in laminin 5 (α5 chain, Figures 4B and 4C, respectively, asterisks show the split area) (Uitto and Pulkkinen 2001). The reduction in α5 and β2 chain expression in HJEB patients, particularly over split skin, suggested that this effect might be due to antigen degradation in vivo in the split areas. The presence of α5 and β2 chains in intact EB skin, however, confirms that these chains are capable of being independently synthesized and assembled even in the total absence or in the presence of defective laminin 5. All other EB cases also showed normal staining for other laminins including junctional EB associated with pyloric atresia (JEB-PA) (with defects in α6β4 integrin, Figure 4E), EB simplex associated with muscular dystrophy (EBS-MD) (with defects in plectin, Figure 4F), non-lethal junctional (NLJEB) (with defects in BP180, Figure 4G), and severe recessive dystrophic epidermolysis bullosa (SRDEB) (Figure 4H). In control and SRDEB patients’ skin there was normal staining for laminin 5 (γ2 chain using the antibody GB3, see Figure 4L). This was in contrast to laminin-5 chain staining that was severely reduced or absent in both the NLJEB (Figure 4J) and HJEB cases (Figure 4K), harboring severe defects in laminin-5 expression.
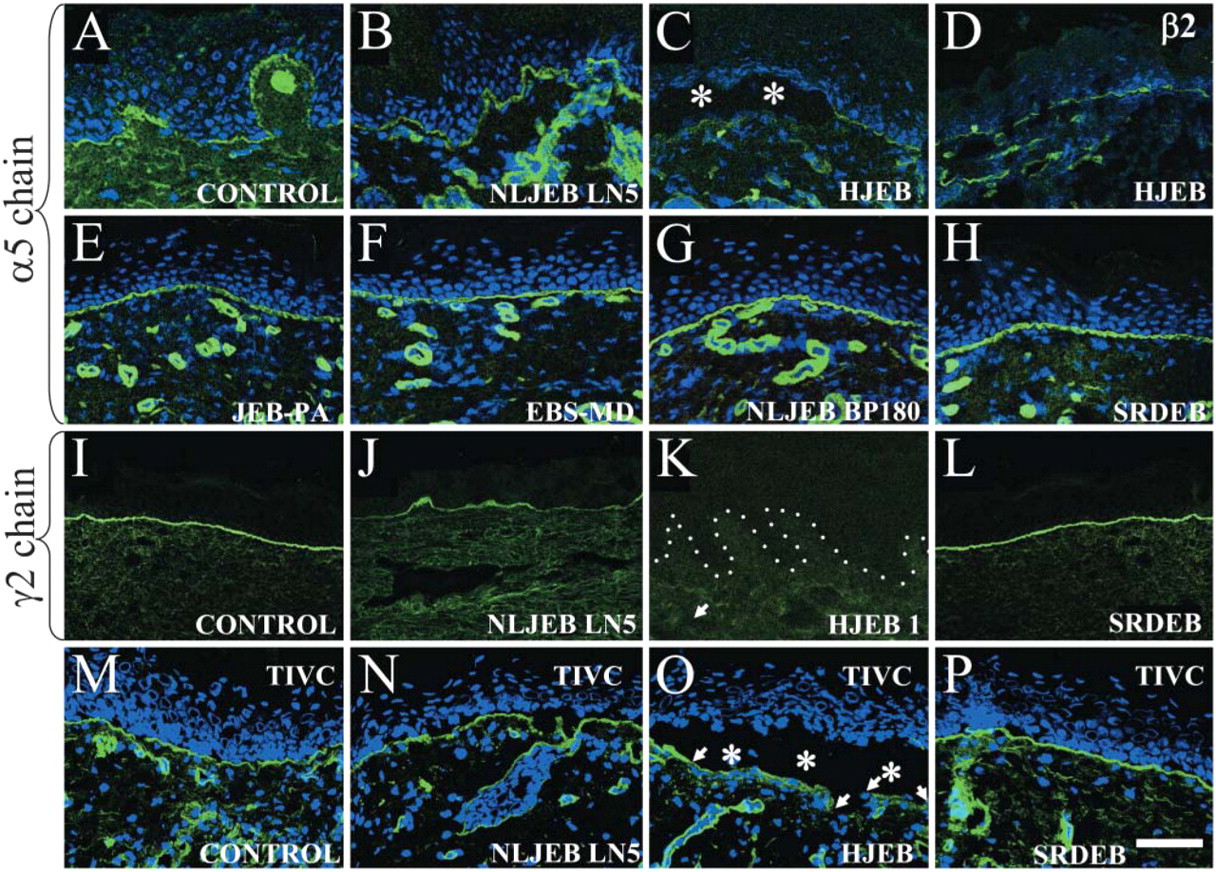
Laminin α5 and β2 chains are expressed beneath the epidermis in epidermolysis bullosa (EB) skin but are focally reduced in split lethal Herlitz junctional EB (HJEB) skin. The laminin α5 chain in control skin showed linear fluorescence along the dermal-epidermal junction and in dermal blood vessels (
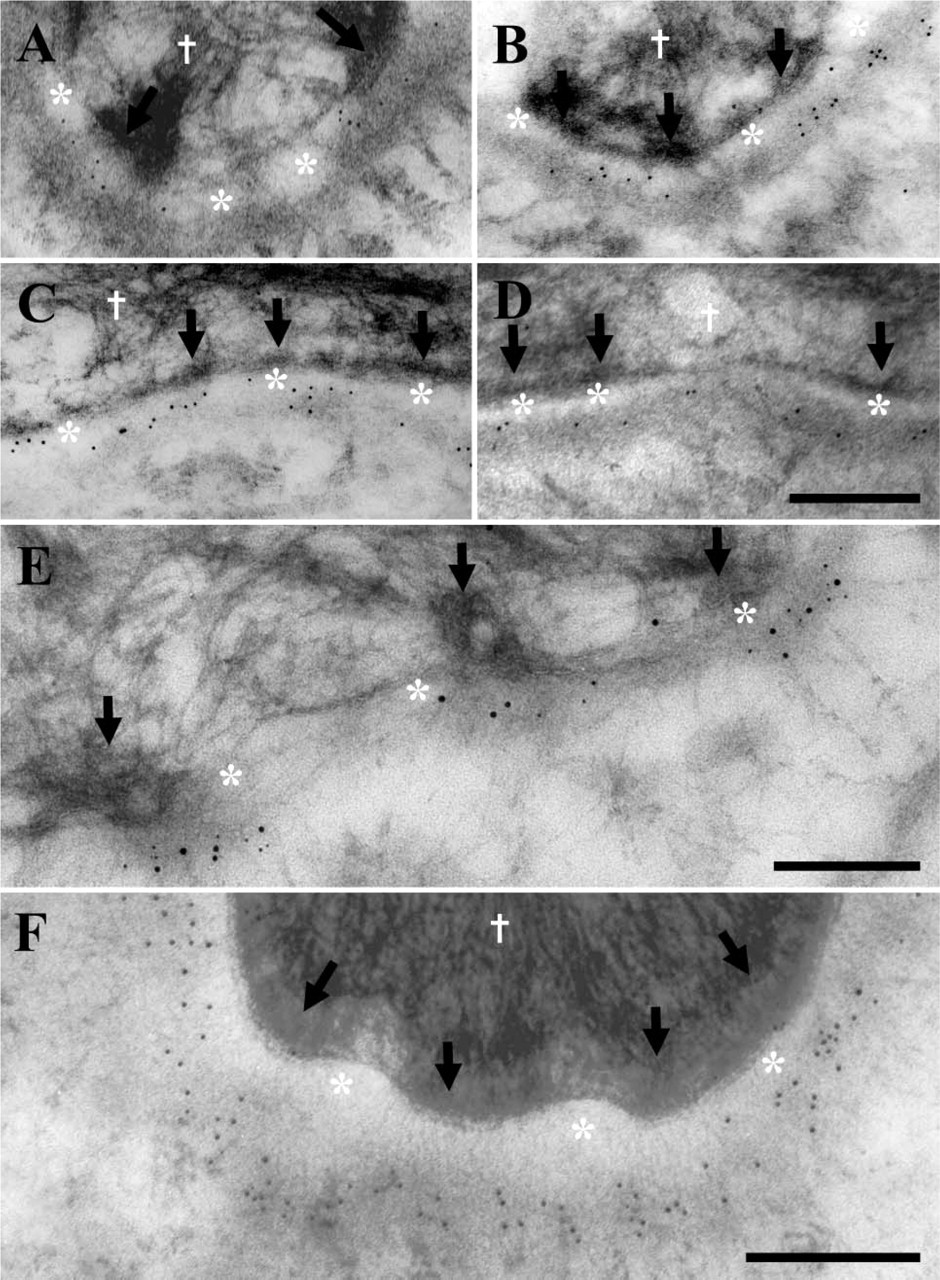
The majority of laminin 5 and laminin 10/11 chains are restricted to the lamina densa beneath HDs, whereas collagen IV is expressed continuously along the basal lamina. Postembedding immunoelectron microscopy with anti-α5 chain (laminin 10) (
Immunogold Electron Microscopy and Quantitative Analysis
Labeling of control interfollicular epidermal sections showed that the majority of laminin-10 α5 chains (Figures 5A-5D; Table 2), β1, γ1, and 11 chain (β2 chain, data not shown) were restricted to under the cytoplasmic HD outer plaques. This was in contrast to collagen IV, which was not as restricted to beneath epidermal HDs plaques (see Figure 5F, 61% collagen IV vs 82% laminin 5). The difference between all laminin and collagen values was statistically significant using the one-way ANOVA (p<0.001) and Student's t-test (p<0.000). All four α5, β1, β2, and γ1 chain antibodies and antiserum showed a remarkable similarity in the percentage of labeling associated with the HD attachment plaque and anchoring filament complex beneath HDs, ranging between 84% and 92% (see Table 2). Furthermore, these values reflect an almost identical (HD restricted) expression pattern to the previously reported values for laminin-5 subunits (Masunaga et al. 1996; McMillan et al. 2003b) (part of these findings are also included in Table 2). Our data are very similar to those of laminin 5 (α3 chain) that on average demonstrated 82% of labeling restricted to beneath HDs (see Table 2) at a distance ranging from 35 to 45 nm below the plasma membrane at the LL-LD junction (Masunaga et al. 1996; McMillan et al. 2003b). The precise distance of the 4C7 epitope on the C-terminal portion of the α5 chain was 53.07 nm (±6.69 SD) from the plasma membrane (arrows, Figures 5A-5D). This is 18 nm lower than the G1 domain of the laminin-5 α3 chain described previously (see Figure 1) (McMillan et al. 2003b). The difference between these two α chain mean values was statistically significant using the one-way ANOVA (p>0.01) and Student's t-test (p=0.009). However, visual examination of the distribution of these two antigens revealed overlapping values ∼30–40 nm beneath the plasma membrane, the only difference being that the α5 chain showed a wider range of labeling that extended deeper in the LD compared with the α3 chain. The remaining three β1-, β2-, and γ1-chain antibodies recognized, as yet unidentified, epitopes on specific laminin chains and were therefore not included in the plasma membrane distance measurements. However, all three antibodies showed upper LD labeling (not shown), the majority of which were restricted to beneath HDs similar to the α5 chain. The three β1-, β2-, and γ1-chain antibodies were excluded from the distance measurements but were scored for their localization either beneath visible HD attachment plaques (as defined by McMillan and Eady 1996) or within inter-HD areas.
Double labeling for the α5 chain of laminins 10/11 (highlighted by 5-nm small gold particles) and whole anti-laminin 5 antiserum (shown by the larger 15-nm gold particles) shows a similar labeling pattern in the LD beneath electron densities presumed to be HDs (Figure 5E). HD plaques are visible within the keratinocyte cytoplasm (white cross) and the dermal-epidermal junction is separated by the LL (Figure 5E, asterisks). Together our data suggest that the α5 chains (including β1, β2, and γ1 chains, see Table 2) show a restricted expression pattern beneath HDs, similar to laminin 5 but unlike collagen IV.
Discussion
We have demonstrated that the α5, β1, and γ1 chains show a similar localization to laminin 5 in the human interfollicular epidermal basement membrane. These data support the presence of multiple laminin isoforms beneath HDs in the basement membrane at several different epidermal sites. A very different localization of collagen IV within the LD but not restricted to beneath HDs was observed. These data suggest a complex network of interactions between different basement membrane components beneath the epidermis (Ghohestani et al. 2001; McMillan et al. 2003a; Miner and Yurchenco 2004).
In addition we have demonstrated that the expression of the α5 and β2 chains is independent of laminin 5, as demonstrated by residual staining in HJEB patients’ skin. The reduction in α5 and β2 chain expression in HJEB was only observed over separated, blistered areas of skin, suggesting that this effect is due to separation-induced antigen degradation in vivo. This was supported by reduced collagen IV staining in split areas of EB skin. This was confirmed after reduced collagen IV staining was observed within separated areas of HJEB skin samples (data not shown). Our results also suggest that the presence of other laminins cannot fully compensate for defects in laminin-5-deficient HJEB skin (McMillan et al. 1997, 1998). The α5 and β2 chains were also normally expressed in all other EB samples harboring defects in plectin, collagen XVII (bullous pemphigoid antigen 2), the α6β4 integrin, and collagen VII.
In previous reports (Aumailley and Rousselle 1999), the laminin β2 chain was not expressed in the epidermal basement membrane of neonatal foreskin. However, we showed weak, variable β2 chain expression (in thigh and arm skin) and absences in other body sites (scalp skin). We conclude that the lack of β2 staining may be due to several factors: antigen masking, a low-level expression, or site- or age-specific variations in human laminin β2 chain expression that may include posttranslational protein processing seen in other laminin isoforms (Miner et al. 1997).
Laminin 5, together with several HD-associated antigens, is expressed in a specific pattern around late anagen hair follicles that excludes staining around the dermal papilla area (Akiyama et al. 1995; Nutbrown and Randall 1995). Laminin-10 chains also show this similar expression pattern in late anagen hair follicles. Unlike laminin-5 and laminin-10 chains, we failed to observe any β2 chain expression (laminins 7/11) in any part of the adult hair follicle; however, this may be due to a low level of antigen expression or masking of the β2 chain epitope. The significance of these findings may be related to the specific growth phases of the lower non-permanent portion of the hair follicle.
In the laminin α5 chain knockout mouse, an unusual disruption in hair follicle morphogenesis was demonstrated (Li, et al., 2003). Li et al. (2003) reported that, in control mice, laminin 10 was present in murine elongating hair germs when other laminins were down-regulated, suggesting a specific role for this laminin in hair follicle development and follicular keratinocyte migration. Mouse skin lacking laminin 10 also contained fewer hair germs and follicles compared with control mice, and after transplantation experiments this skin showed a failure of hair germ elongation and defective basement membrane assembly. Intriguingly, treatment of these mice with purified exogenous laminin 10 corrected these defects and restored hair follicle development. Given that human hair follicles are slow cycling and the majority remains in the late anagen phase and shows different growth characteristics to murine follicles, our failure to demonstrate such growth phase-specific differences in the expression of laminin 10 during the hair cycle stages is not surprising.
The presence of multiple laminin isoforms beneath HDs suggests the hypothesis that there are laminin subunits possibly with overlapping functions that form focal clusters of laminin molecules. This was in contrast to collagen IV, which was not restricted to HDs and localized to the LD region. Ultrastructural data show that the α3 chain (laminin 5) is closer to the plasma membrane than the α5 chain (with only an 18-nm difference, see Table 2). This might suggest that the α5 chain is more closely associated with a LD component such as collagen IV. However, given the size of both laminin chains of ∼80–100 nm (as determined by rotary shadowing experiments), our data suggest a significant overlap occurs between α3 and α5 chains (Marinkovich et al. 1992; Vailly et al. 1994). Further studies using a larger battery of antibodies are required to determine the orientation of these laminin components.
Together these data show for the first time that laminin 10/11 chains are restricted to beneath HDs similar to laminin 5 but distinct from collagen IV. Our data suggest a specific localization of multiple laminin isoforms in the epidermal basement membrane beneath HDs and support the hypothesis that several laminins in close association may promote stable cell attachment among different basement membrane molecules.
Footnotes
Acknowledgements
This work was supported by a grant-in-aid of Scientific Research A (13357008, HS) and Health and Labor Sciences Research Grants (Research into Specific Diseases) H13-Saisei-02 and H17-Saisei 12, by a grant from the Japanese Society for the Promotion of Science (JSPS) grant #00345 to J.R.M., and by a grant-in-aid for JSPS fellows’ research expenses (#00345). This work was also supported by a grant from the Japanese Health Science Foundation for Research Residents, for class “A” researchers (JRM).
We gratefully acknowledge the technical support of Ms. M. Sato and Ms. K. Sakai in this study. We also thank Dr. T. Masunaga for kindly providing the data from the γ2 chain study (![]() ) and Dr. M.P. Marinkovich for his kind gift of his polyclonal laminin 5 antibody. Hybridoma supernatants D18 and 2E8 (produced by Drs. J. Sanes and E. Engvall) and M3F7 (from Dr. H. Furthmeyer) were obtained from the Developmental Studies Hybridoma Bank, developed under the auspices of the National Institute of Child Health and Human Development (NICHD) and maintained by the University of Iowa, Department of Biological Sciences, Iowa City, Iowa.
) and Dr. M.P. Marinkovich for his kind gift of his polyclonal laminin 5 antibody. Hybridoma supernatants D18 and 2E8 (produced by Drs. J. Sanes and E. Engvall) and M3F7 (from Dr. H. Furthmeyer) were obtained from the Developmental Studies Hybridoma Bank, developed under the auspices of the National Institute of Child Health and Human Development (NICHD) and maintained by the University of Iowa, Department of Biological Sciences, Iowa City, Iowa.
