Abstract
Studies of human prostate carcinoma (PCA) in animal model systems have been hampered by the paucity of human PCA cells available in tissue culture (Pretlow et al. 1994; Lalani et al. 1997). Most studies of progression and metastasis have relied on already metastatic human PCA isolates from various target organs that have been cultured for long periods of time, e.g., DU145, PC3, and LNCaP (Pretlow et al. 1994; Zhau et al. 1996; Lalani et al. 1997; Rubio et al. 1998; Yang et al. 1999). Human PCA primary tumors, isolated from patients and carried in athymic nude or SCID mice as xenografts (Pretlow et al. 1994; van Weerden et al. 1996), offer the opportunity to isolate PCA primary tumor cell lines that would be valuable for analyzing progression of this disease. More effective molecular and cellular analyses of progression mechanisms require isolates of such primary tumors of human PCA (Pretlow et al. 1994).
The xenograft approach for isolating human PCA tissue and its passage in nude mice offers an intermediate stage in the development of new cell systems (van Steenbrugge et al. 1994; van Weerden et al. 1996). The Pretlows and their colleagues have made significant advances in isolation and characterization of many independent isolates of PCA primary tumors from patients. These are maintained as xenografts in athymic nude mice (Pretlow et al. 1993; Wainstein et al. 1994; Cheng et al. 1996). This includes the CWR22 xenograft that was not metastatic in nude mice (Wainstein et al. 1994). They reported subsequent isolation of a “relapsed” variant of this xenograft (CWR22R), which was androgen-independent and metastatic to lungs of nude mice (Nagabhushan et al. 1996). A subline of the CWR22R xenograft (Pretlow xenograft #2152; Nagabhushan et al. 1996) was used by Jacobberger and colleagues to isolate and characterize a tissue-cultured cell line, CWR22Rv1 (Sramkoski et al. 1999). These cells display a karyotype similar to that of the original CWR22 xenograft (approximately 50 chromosomes), secrete prostate-specific antigen (PSA), display a specific point mutation in one exon of the androgen receptor gene (Tan et al. 1997), and generate primary tumors in the subcutis of nude mice pathologically characteristic of prostate carcinomas (Sramkoski et al. 1999).
CWR22Rv1 cells therefore offer an excellent opportunity to test possible metastatic spread to multiple organs from a cell population originally isolated from a human primary tumor and not from already metastatic subpopulations. If metastasis is observed with these cells to organs that are normally involved in the human disease, then we have a more relevant model system for evaluating mechanisms for PCA progression and metastasis.
Our laboratory has maximized sensitivity of progression and metastasis studies in animal models of tumor systems by transfecting a histochemical marker gene (e.g., bacterial lacZ or human placental alkaline phosphatase gene) into tumor cells, isolating relatively stable-expressing lacZ or PAP transfectants and testing for micrometastases in many target organs of athymic nude mice using ultrasensitive histochemical staining (Lin et al. 1990a,b; Kleinman et al. 1994). Use of histochemical markers permits ready visualization of both tagged tumor cells and neighboring host organ cells in the same field of vision. This is not possible with the use of fluorescent tags for tumor cells, such as luciferase or green-fluorescent protein (Rubio et al. 1998; Yang et al. 1999). These approaches with histochemical marker genes have been previously reviewed by us (Lin and Culp 1992; Culp et al. 1998a,b) and others (Kruger et al. 1999). They demonstrate our ability to detect single tumor cells in the vasculature and in target organs leading to micrometastases (Lin and Culp 1992; Lin et al. 1992; Culp et al. 1998a,b). They also enable us to evaluate relationships between neighboring tumor cells and host connective tissue cells at the highest resolution (Chang and Chung 1989; Culp et al. 1998a,b).
In the analyses reported here, the lacZ gene was transfected into cultured CWR22Rv1 cells, three clones were isolated, and the tumorigenic progression and metastasis of these clones were evaluated. These analyses identify one transfectant clone, referred to as LZ-CWR22R-H, that closely mimics metastatic progression normally observed in the human disease to lung, liver, and bone with a reasonable frequency. They also demonstrate remarkable stability of micrometastases in the lung and bone, as well as overt metastasis in the livers of this experimental animal system.
Materials and Methods
Isolation of lacZ-transfected CWR22R Cells
Early-passage CWR22Rv1 cells (Sramkoski et al. 1999), free of Mycoplasma, were obtained from Dr. James Jacobberger of this institution. This cell line (Sramkoski et al. 1999) was generated on an irradiated bed of STO mouse fibroblasts from the #2152 xenograft subline of CWR22R carried in nude mice by Dr. Thomas Pretlow and colleagues (Nagabhushan et al. 1996). Cells were grown in RPMI 1640 medium supplemented with 10% FCS and with penicillin and streptomycin. The bacterial lacZ gene, under regulation of the Rous sarcoma virus LTR on a bacterial plasmid carrying the neo R gene used for G418 selection (400 μg/ml), was transfected into CWR22R cells. Three clones (B, D, and H) were isolated as described previously (Lin et al. 1990a,b; Kleinman et al. 1994). These will be referred to as LZ-CWR22R-B, -D, or -H cells, respectively. The stability of lacZ expression during long-term culturing of these three clones, with or without selection drug, has been described previously (Culp et al. 1998a). Briefly, Clone D is highly unstable in the absence of selection drug, with most cells losing X-gal stainability within 7-10 passages; Clone B displays intermediate stability; and Clone H is highly stable for lacZ expression for >25 passages, even in the absence of selection drug.
Tumorigenicity Studies
Athymic nude mice (HSD nu/nu) were bred and housed in the Athymic Animal Facility of the CWRU/Ireland Cancer Center under AAALAC-I approval. All procedures were executed under supervision of the Animal Care and Use Committee of Case Western Reserve University. Nude mice (4-6-week-old males) received two sets of India ink tattoos on each side of their hind flanks, between which cells were injected into the subcutis (Kleinman et al. 1994). The indicated number of lacZ-transfected CWR22R cells, dispersed from cultures by trypsinization, were then injected sc and animals monitored daily for evolution of primary tumors. In some cases, cells (0.1 ml) were injected in a PBS vehicle, and in other cases they were injected in a Matrigel suspension (Pretlow et al. 1991). In all cases, the cultured population of Clone H cells stained with X-gal to >90% of the cells, indicating the excellent stability of expression of the lacZ gene in this particular clone.
Tissue Isolation and Histochemical Staining
Primary tumors were isolated from sacrificed animals when they were generally >4 mm in their shortest dimension and <10 mm in their longest. In most cases, the entire primary tumor and the surrounding normal mouse tissues were isolated as a unit for X-gal staining. In select cases, the primary tumor was isolated, bisected with a scalpel, and then the halves fixed and X-gal-stained. Lungs and other organs were isolated intact from sacrificed animals as described previously (Lin et al. 1990a,b; Kleinman et al. 1994). Fixation was performed at 4C for 1 hr with 2% (v/v) formaldehyde in PBS.
X-gal staining was executed on fixed tissues as described previously at pH 7.4 (overnight at room temperature) to minimize background tissue staining (Lin et al. 1990a,b; Lin and Culp 1992; Kleinman et al. 1994; Culp et al. 1998b). X-gal staining was also performed on tissues from animals not injected with tumor cells to evaluate possible background staining of tissues. This was particularly important in bone, in which some background staining is observed in the growth regions at the ends of long bones of the animal. X-gal-stained primary tumors, overt metastases, and micrometastases were photographed on a Nikon SMZU Dissecting Microscope with Kodak 160T film.
Materials
Matrigel was obtained from Collaborative Research (Bedford, MA), X-gal from Research Organics (Cleveland, OH), G418 and RPMI 1640 medium from Gibco (Grand Island, NY), potassium ferricyanide, potassium ferrocyanide, and formaldehyde from Sigma Biochemicals (St Louis, MO), tissue culture plastic ware from Becton-Dickinson Labware (Oxnard, CA), and fetal calf serum and RPMI 1640 medium from Irvine Scientific (Santa Ana, CA).
Results
Primary Tumor Development
When LZ-CWR22R Clone B, D, or H was injected into the subcutis of nude mice in a PBS vehicle, there were differences in the tumorigenicity of the three clones. Clone B yielded tumors in all animals (1). However, the vast majority of these tumors failed to give uniform X-gal stainability. For example, Figure 1A illustrates a tumor with only a minor fraction retaining expression of lacZ. In the case of Clone D, many animals yielded tumors when injected with >1 × 106 cells, but none of these tumors displayed any staining (not shown), consistent with the considerable instability of lacZ expression in cultured populations of Clone D. Because of this rapid loss of X-gal stainability by Clones B and D, they could not be studied any further to test for development of micrometastasis in various organs.
Results for Clone H were very different. Only 15-40% of the animals injected with either 1 × 106 or 5 × 106 cells in a PBS vehicle developed tumors, even as long as 6 months post injection (1). However, all these primary tumors in the subcutis were uniformly stainable with X-gal (Figure 1B), demonstrating the persistent expression of lacZ in this particular clone. Figure 1B shows a sizeable and highly stained primary tumor surrounded by fascia that is not X-gal-stainable. When any of these Clone H primary tumors were first sliced into halves with a scalpel and then fixed for X-gal staining, the uniformity of the stainability was again evident (Figure 1C). This shows that non-expressing variants of the tumor cells do not arise at later time points of primary tumor development (e.g., at the most interior regions of the primary tumor). The extensive vasculature of the primary tumor is also evident at these later times [red-staining blood vessels against the intense blue background (Figure 1C)].
In contrast to the relatively low tumorigenicity of Clone H when injected in PBS, the same cells injected in Matrigel were 100% tumorigenic when 1-5 × 106 cells were used (1). These Matrigel-borne primary tumors developed identically to PBS-borne tumors when pathological parameters were considered (not shown).
Micrometastasis to Multiple Organs of PBS-injected Cells
With PBS-injected Clone H cells, there was little evidence of overt metastases developing in most organs (except for liver; see below). However, lacZ stainability enables us to detect the very smallest of micrometastases. Micrometastases were observed in the lung for most mice with large tumors when PBS was used as the injection vehicle (Table 2). An example of one lung micrometastasis is shown in Figure 2A. With lower frequency, micrometastases were observed in other organs as well (2). Figure 2B shows a micrometastasis that has established itself reasonably close to a blood vessel in the liver. Figure 2C shows micrometastases close to a blood vessel in the brain. The relative distribution of micrometastases in animals bearing primary tumors and using PBS-injected cells was lung (>90%) >liver=bone (50%) >kidney=brain.
Tumorigenic competence of two independent lacZ transfectants of CWR22R cells a

aThe indicated number of cells were injected, either in PBS or Matrigel as indicated, into the subcutis of the hind flanks of nude mice. Injection sites were identified with tattoos. The indicated number of mice developed primary tumors at the injection site within 8 weeks and animals with tumors were harvested 17-153 days post injection, although animals were examined for at least 6 months to test for any tumorigenic potential.
bThese were separate experiments.
cVery early-passage Clone H cells compared in the same experiment.
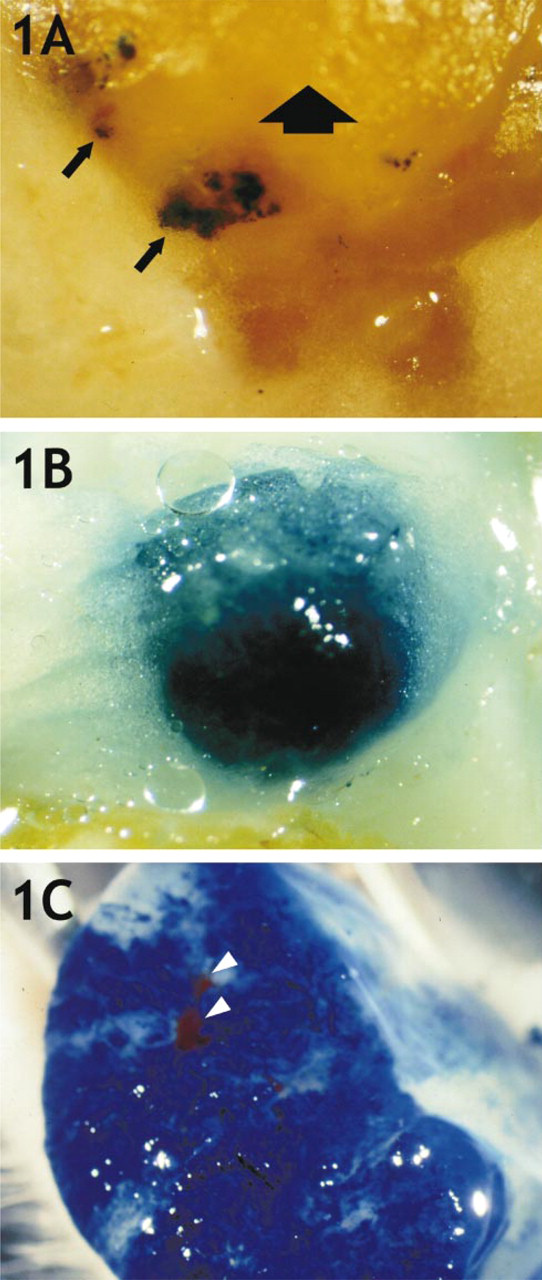
Primary tumors of lacZ transfectants. LZ-CWR22R Clone B or H cells (1 × 106) were injected into the subcutis of athymic nude mice. At the indicated times when the primary tumor had become large (>5 mm in the smallest dimension), animals were sacrificed and the region of the primary tumor excised from the surface of the animal. This region was then fixed and X-gal-stained as described in Materials and Methods. (
Tumor size and metastatic potential of Clone H cells a
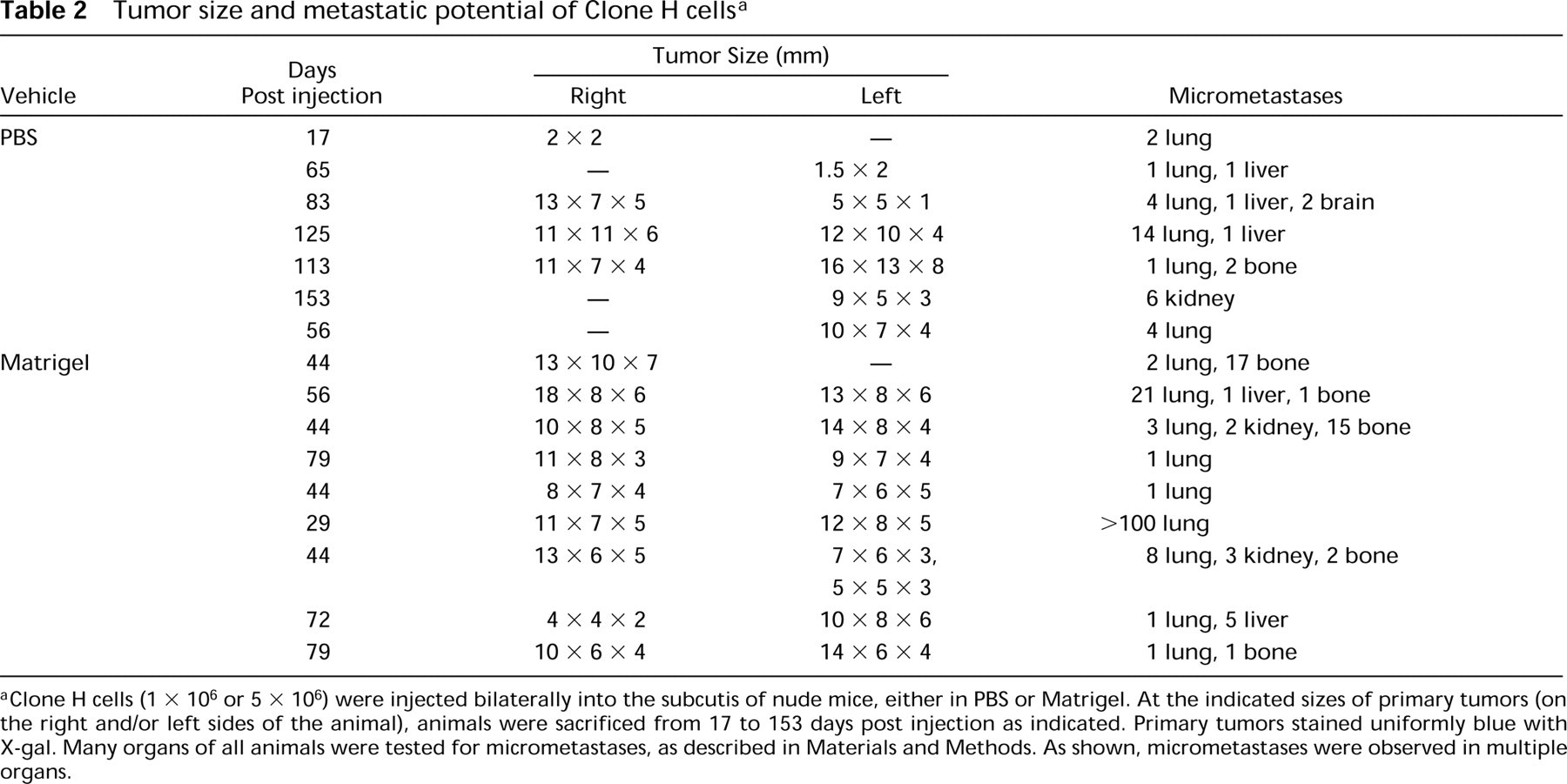
aClone H cells (1 × 106 or 5 × 106) were injected bilaterally into the subcutis of nude mice, either in PBS or Matrigel. At the indicated sizes of primary tumors (on the right and/or left sides of the animal), animals were sacrificed from 17 to 153 days post injection as indicated. Primary tumors stained uniformly blue with X-gal. Many organs of all animals were tested for micrometastases, as described in Materials and Methods. As shown, micrometastases were observed in multiple organs.
Micrometastasis to Multiple Organs of Matrigel-injected Cells
When Matrigel was the vehicle, Clone H primary tumors yielded micrometastases in the lungs of all animals examined (2). A lung micrometastasis is shown in Figure 3A, a liver micrometastasis in Figure 3B, and kidney micrometastases in Figure 3C. Micrometastasis frequency for all tumor-bearing animals was as follows: lung (100%) >bone (60%) >liver (20%) >>kidney or brain.
Several precautions were taken in these studies to guarantee that blue-staining foci truly contain tumor cells. First, a clonal population of CWR22Rv1 cells, shown to be tumorigenic in nude mice, was used for transfection of the lacZ gene. This obviates any possibility for another cell type from the original xenograft to be transfected with this gene. Second, a chromosome-integrating plasmid was used in these studies to obviate any episomal plasmid being expressed and possibly being transmitted to neighboring host tissue cells (Culp et al. 1998a,b). Third, as shown by our laboratory for tumor cells (Lin and Culp 1992) and many other labs using transgenic animals (Kruger et al. 1999), the polymerized blue product of X-gal digestion cannot be transmitted to neighboring host cells. Finally, the conditions used for fixation and X-gal staining of mouse tissues were developed to avoid any artifactual staining of tissue cell types (Lin and Culp 1992; Culp et al. 1998a,b). By comparing X-gal staining of whole organs with serial sections of X-gal-stained foci, we previously determined that single tumor cells could be detected by our Nikon SMZU dissecting microscope. Therefore, these foci are truly micrometastases of prostate carcinoma cells.
Micrometastasis to Bone
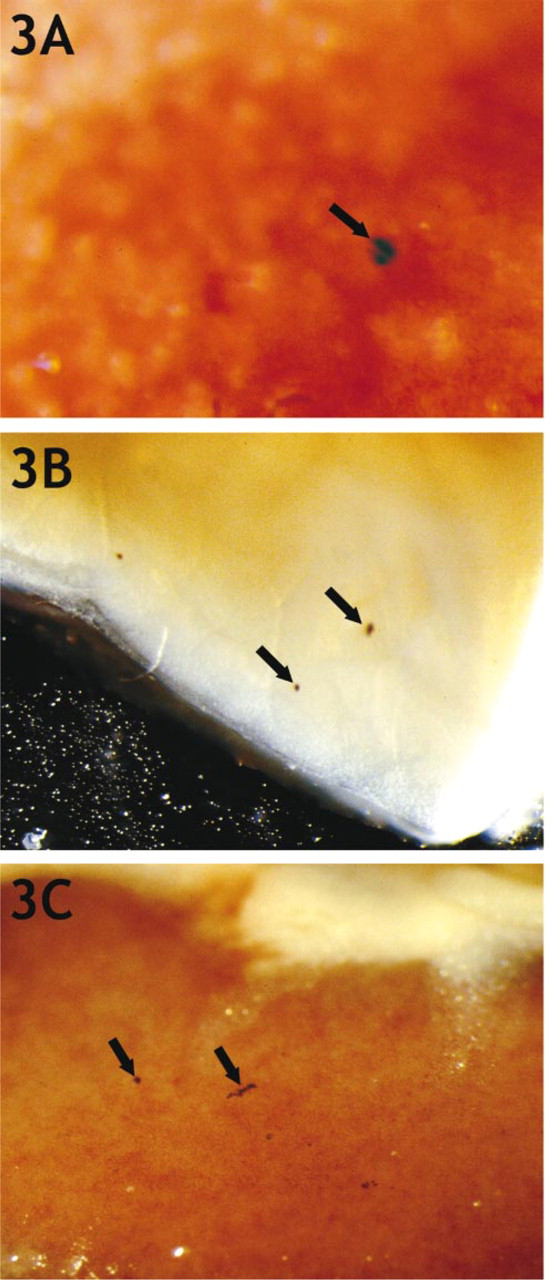
Bone has been the most difficult and elusive target for PCA metastasis studies in animal model systems (Pretlow et al. 1994; Lalani et al. 1997; Wu et al. 1998; Yang et al. 1999). However, this is a very important aspect of the progression of the human disease. Therefore, lacZ stainability affords us maximal sensitivity for detecting micrometastases in the various bone systems of the nude mouse. Bone micrometastases were very evident with Matrigel-injected cells (2). An example of micrometastases along the long bone of the animal is shown in Figure 4A for PBS-injected cells. In PBS-injected cells, micrometastases to spinal column were also occasionally observed (Figure 4B). Figure 4C shows micrometastases along the spine after Matrigel injection.
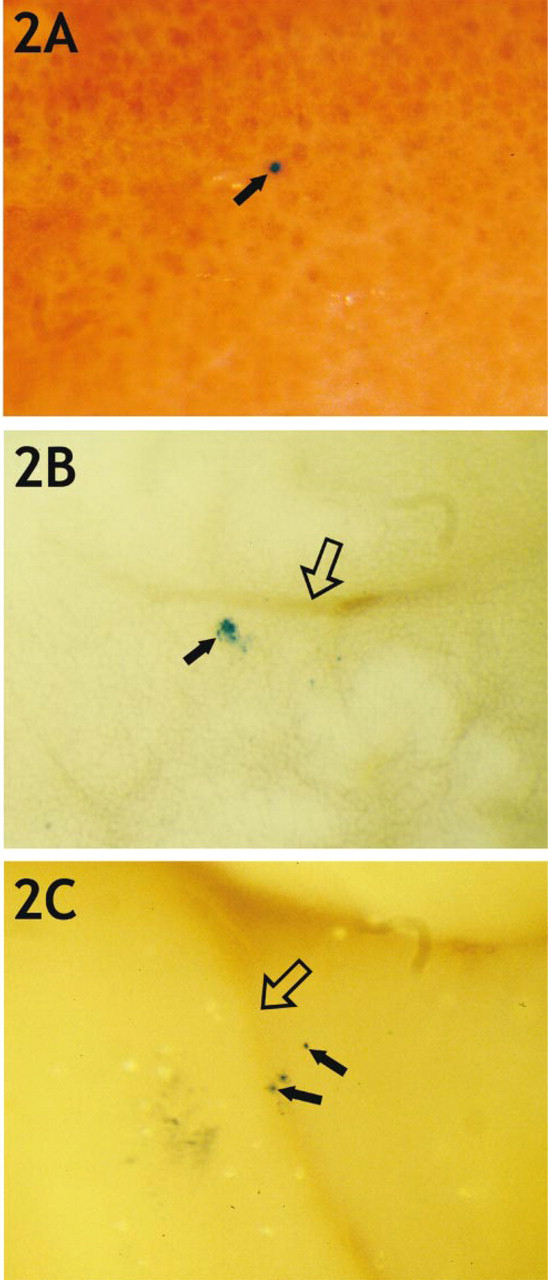
Micrometastasis to multiple target organs when Clone H cells were injected in a PBS vehicle. PBS-suspended Clone H cells were injected sc into nude mice. When the primary tumors had become large, the animals were sacrificed. Many internal organs were excised, fixed, and X-gal-stained to evaluate possible development of micrometastases. (
Micrometastasis when Clone H cells were injected in a Matrigel vehicle. Matrigel-suspended Clone H cells were injected sc into nude mice. When the primary tumors had become large, the animals were sacrificed. Many internal organs were excised, fixed, and X-galstained to evaluate development of micrometastases. (
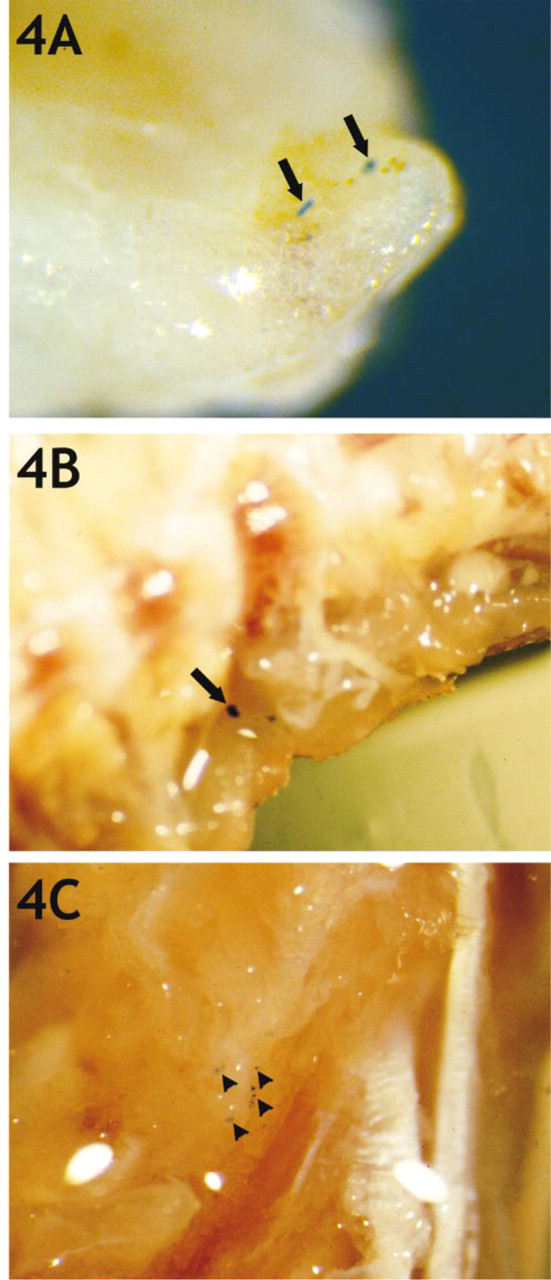
Micrometastasis to bone. Clone H cells were injected sc in either PBS or Matrigel vehicles. When primary tumors had become large, the animals were sacrificed for excision of many bones, followed by bone fixation and X-gal staining. (
Micrometastasis Outgrowth into Overt Metastases
The question arises of whether micrometastases in this LZ-CWR22R-H cell system can give rise to overt metastases. In none of the animals examined in this study were overt metastases ever observed in the lungs (not shown). This is a remarkable finding in light of the several hundred micrometastases that were scored in animals with very large primary tumors and in light of all animals that yielded lung micrometastases when primary tumors had developed. The same applied to bone micrometastases.
In contrast, overt metastases were observed in the livers of some animals. An example from a PBS-injected population is shown in Figure 5. Because micrometastases were much less common in the liver than in the lung and because comparable long time periods were involved, it is unlikely that insufficient time is the basis for poor outgrowth of overt metastases in the lung or bones. Apparently, there are environmental cues in the liver that permit effective outgrowth in the liver of Clone H cells, and these cues are missing in lung and in bone.
Discussion
These studies have identified a highly stable lacZ transfectant of a new human PCA cell line isolated from a primary tumor population, CWR22Rv1 (Sramkoski et al. 1999). This LZ-CWR22R-H cell line yields progression and metastatic competence that parallels the targets of the human disease, i.e., to lung, bone, and liver (Pretlow et al. 1994; Lalani et al. 1997). This is an important finding because animal model systems of PCA have not been very successful in documenting metastasis to liver and bone unless already metastatic human PCA isolates were used as the starting material (Wu et al. 1998; Yang et al. 1999). This progression pattern was evident without the need to introduce androgen into young nude mice, indicating the possible androgen-independence of these progression patterns.
Tumorigenesis of LZ-CWR22R-H cells was limited to 15-40% of animals injected when cells were injected sc in a PBS vehicle. However, all tumor-bearing animals yielded micrometastases in their lungs and in some cases in liver and bone, with brain and kidney micrometastases the most infrequent. The relative distribution of micrometastases with PBS-injected cells was lung (>90%) >liver=bone (50%) >>kidney= brain. The reasonably high level of progression of these cells into liver and bone offers optimism that this is a more accurate model of the human disease from an unselected primary tumor population of cells (Pretlow et al. 1994; Lalani et al. 1997).
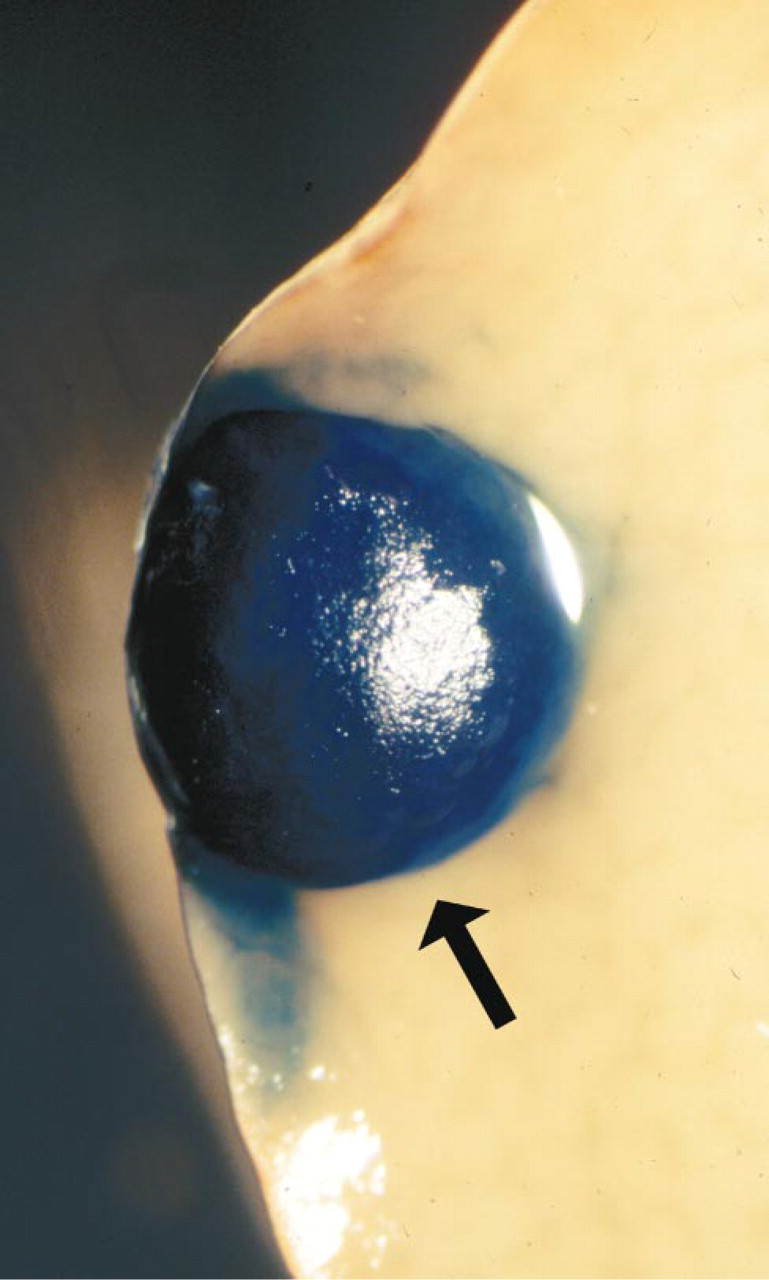
Overt metastasis in the liver. Clone H cells were injected sc in PBS. When primary tumors had become large (65 days post injection), animals were sacrificed for excision of many internal organs, followed by their fixation and X-gal staining. A large liver metastasis (large arow) was developing in the liver; it stained for lacZ expression throughout. Original magnification × 13.
When Matrigel, a now commonly used vehicle for PCA cells (Pretlow et al. 1991), was used as the injection vehicle for LZ-CWR22R-H cells, the pattern of tumor progression changed significantly. First, all animals that received bilateral injections developed primary tumors, many with tumors on both sides of the animal. Second, the pattern of metastasis changed quantitatively but not qualitatively. Micrometastasis frequency was as follows: lung (100%) >bone (60%) >liver (20%) >>kidney or brain. That progression to bone should be increased and that to liver decreased when Matrigel is used (compared to PBS injection) raises some interesting hypotheses that could be tested in future experiments. Matrigel may permit more facile selection and expansion of variants that are effective colonizers of bones. In contrast, there may be less effective expansion of variants that target the liver. Alternatively, Matrigel may permit many more cells to colonize the subcutis and thereby permit more rapid evolution of variants competent for migrating to bone structures. In any case, this system will be an effective model system for analyzing the molecular events that lead to evolution of bone and liver metastatic variants from a population of human PCA cells from a primary tumor of a human patient (Wainstein et al. 1994; Cheng et al. 1996).
A notable surprise in our analyses was the divergence in competence for forming overt metastases. With PBS- or Matrigel-injected cells, overt metastases were never observed in the lung or bone but were occasionally observed in the liver. That the liver received fewer tumor cells than either the lung or bones suggests that time or cell dosage was not a critical element in determining overt metastasis. Perhaps outgrowth in the lung and the bones is retarded by organ-specific environmental cues that are not present in the liver (Nicolson 1993; Radinsky 1995; Singh et al. 1997). It will be important to use orthotopic injection of LZ-CWR22R-H cells into the prostate gland to determine if these progression and overt metastasis patterns differ from those of subcutis-derived tumors (Fu et al. 1992; Stephenson et al. 1992; Sato et al. 1997).
This study is not the first to use a marker gene to track PCA cells in an animal model system. Rubio et al. (1998) evaluated PC-3 cells tagged with luciferase to develop lymph node micrometastases after IM injection and by highly sensitive detection of fluorescence. They provided evidence that the lymphatic system was the preferred route of migration for these cells. Similarly, Yang et al. (1999) tagged PC-3 cells with green-fluorescent protein (GFP) and tracked micrometastasis to many bones of the animal by taking advantage of the ultrasensitive fluorescence of the GFP tag after sc or orthotopic injection. Both these studies used PC-3 cells derived from a bone metastasis from a human patient. However, use of a fluorescent tag (e.g., luciferase or GFP) obviates any evaluation of the tagged tumor cells with neighboring host tissue cells (Chang and Chung 1989). Tumor cells tagged with histochemical marker genes can be readily evaluated at the single-cell level in any tissue in terms of their relationships with other cell types of the target organ, blood vessels, and other organ substructures (Lin et al. 1990a,b; 1992; Kleinman et al. 1994; Culp et al. 1998a,b).
The lacZ-tagged CWR22R tumor cells described here provide a valuable system for evaluating gene regulation at the level of single tumor cells. The ability to identify X-gal-stained single tumor cells in any tissue, combined with laser-capture microdissection (Emmert-Buck et al. 1996; Schutze and Lahr 1998; Simone et al. 1998) to target small groups of tumor cells, will enable us to test the levels of genes and their expression levels for possible relevance for metastatic progression of PCA. In addition, tagging of two genetically or phenotypically different CWR22R tumor derivatives with different histochemical marker genes (Lin et al. 1992) will provide a powerful paradigm, when combined with laser-capture microdissection, to evaluate the interrelationships of two different tumor subpopulations in different sites of the animal.
Footnotes
Acknowledgements
Acknowledgments
Some of these studies were supported in part by the Comprehensive Cancer Center of the Ireland Cancer Center at Case Western Reserve University (NCI-supported via P30-CA43703) for pilot studies on PCA metastasis. More extensive support was provided by research grant DAMD17-98-1-8587 from the US Army on PCA metastasis.
Athymic nude mouse experiments were conducted in the Athymic Animal Facility (AAALAC-I-approved) of the Case Western Reserve University/Ireland Cancer Center and were approved by the Animal Care and Use Committee of this University. The assistance of Pamela Steele and Kathy Pustai of this facility is greatly appreciated. Special gratitude is extended to Drs Thomas and Theresa Pretlow, as well as Joseph Giaconia of their laboratory, for conveying their extensive knowledge of PCA xenograft biology to the authors during the execution of these studies. Special gratitude is extended to Dr James Jacobberger of the Ireland Cancer Center for the donation of tissue culture-adapted CWR22Rv1 PCA cells.
