Abstract
A multidisciplinary study involving lectin histochemistry, IHC, immuno-lectin blotting, and immunogold was carried out to determine the distribution of sugar residues in the glycoproteins of Rana esculenta oxynticopeptic cells. We considered animals in two experimental conditions, fasting and fed. It is known that, in mammals, the tubulovesicular membranes are rich in proteins with several functions. The proton pump H+,K+-ATPase, a heterodimeric complex with a catalytic α-subunit and a heavily glycosylated β-subunit, responsible for acid secretion, is the most abundant. No data have been published regarding the localization and the structures of H+,K+-ATPase in amphibians. In the water frog, the luminal membrane and tubulovesicular system of oxynticopeptic cells, which differ in morphology according to their functional stage, reacted with the primary gold-conjugated antibody against the H+,K+-ATPase α-subunit. By lectin histochemistry and immunoblotting, in the oxynticopeptic cells of R. esculenta we detected the presence of N-linked glycans having fucosylated (poly)lactosamine chains, which could correspond to the oligosaccharide chains of the β subunit. The latter are somewhat different from those described in mammals, and this is probably because of an adaptation to the different microenvironmental conditions in which the oxynticopeptic cells find themselves, in terms of their different habits and phylogeny.
T
In amphibians, a condition paralleling that of mammals can be found in the water frog Rana esculenta, in which pepsinogen is mainly produced by peptic cells clustered in esophageal glands, whereas hydrochloric acid is mainly produced by oxyntic cells arranged in gastric glands (Eksaeva 1958; Bani 1983; Bani et al. 1992; Gallego-Huidobro and Pastor 1996; Ferri et al. 2001). In contrast, in other frogs, such as Bombina variegata (Bani et al. 1992), Bufo melanostictus (Loo and Wong 1975), or Bufo viridis (Liquori et al. 2002), and in Caudata such as Triturus carnifex (Liquori et al. 2005), esophageal glands are lacking, and gastric glands present only oxynticopeptic cells. The gastric acid–secreting cells are characterized by complex membrane systems. Parietal (oxyntic) acid–secreting cells of mammalian gastric glands show intracellular canaliculi originating from invaginations of the plasma membrane (Ito et al. 1985). Acid-secreting cells in non-mammalian vertebrates lack intracellular canaliculi but exhibit an extensive smooth tubulovesicular system in the apical cytoplasm, which is functionally correlated to the plasma membrane (Forte and Forte 1970; Hirji 1982; Bani et al. 1992; Gabrowski et al. 1995; Liquori et al. 2000). The ultrastructural features of the acid-secreting cells differ according to their functional stage. The tubulovesicular compartment is reduced in extension during gastric acid secretion, when the number and length of the luminal membrane microvilli increase. This is probably related to the transfer and fusion of the smooth vesicles to the plasma membrane (Ogata 1997). In fasting conditions, the cytoplasm of these cells is filled with a conspicuous tubulovesicular membrane system. The apical surface is covered with short microvilli (Logsdon and Machen 1982).
Previously, glycosylated compounds associated with the tubular membrane system of the bullfrog oxynticopeptic cells have been studied by Forte and Forte (1970) and Beesley and Forte (1974) by chromatography and electrophoresis.
Beesley and Forte (1974) reported that acrylamide gel electrophoresis of bullfrog gastric microsomes showed at least 12 periodic acid-Schiff (PAS)-positive bands. The majority of these were thought to be glycoproteins.
The most abundant is H+,K+-ATPase, a gastric proton pump that in various vertebrates has been shown to be responsible for gastric acidification (Tyagarajan et al. 1996). The mammalian gastric H+,K+-ATPase is a heterodimeric complex with a catalytic α-subunit and a heavily glycosylated β-subunit that is essential for holoenzyme function and is involved in stabilizing the membrane structure as well as in regulating intracellular trafficking and in protecting against trypsinolysis (Chow et al. 1992; Tyagarajan et al. 1995; Crothers et al. 2004; Vagin et al. 2004). The α-subunit of H+,K+-ATPase is an integral glycoprotein with both amine and carboxyl groups facing the cytoplasm, whereas the H+,K+-ATPase β-subunit is an integral glycoprotein with one transmembrane domain, a short N-terminal cytoplasmic domain, and a large extracellular domain that is heavily glycosylated. Because of the presence of an extensive, glycoprotein-rich membrane system, the acid-secreting cells may be investigated by lectin histochemistry. In mammals, differences in the structure of the oligosaccharide chains may occur between species, even if they are phylogenetically closely related (Crothers et al. 1995; Appelmelk et al. 1996; Stewart et al. 1999). Variations have also been observed because of pathological alterations of the gastrointestinal tract (Thangarajah et al. 2002).
Abundant glycoprotein chains have been identified in stomach oxyntic cells in two frogs, Rana pipiens and Rana catesbeiana, by standard cytochemical methods (Forte and Forte 1970). Carbohydrate analysis showed that the glycans of these cells have an extremely low content in terms of two acid groups (i.e., uronic and sialic acids), whereas neutral sugars and hexosamines are relatively abundant. To our knowledge, no data have been published regarding the structure of the glycoprotein oligosaccharide chains in frog oxyntic cells or in other non-mammalian vertebrates. In this study, we localized and characterized the sugar residues in the gastric glands of R. esculenta (Amphibia, Anura) by lectin histochemistry, also in association with enzymatic digestion. The cellular and subcellular distribution of H+,K+-ATPase was also analyzed by immunofluorescence and immunogold using monoclonal antibodies against both the α- and β-subunits of this proton pump. By means of immuno- and lectin blotting, we also characterized the oligosaccharide chains of the H+,K+-ATPase β-subunit.
Materials and Methods
Adult female frogs (R. esculenta) were purchased from authorized dealers (Orefice; Arzano, Italy). They were euthanized with ether, and their stomachs were quickly removed. Two animals were in fasting condition, whereas the other two were regularly fed. For light microscopy, the samples were fixed in 10% formalin, dehydrated in a series of graded ethanol, and embedded in paraffin wax. Serial sections, 5 μm thick, were cut.
Carbohydrate Conventional Histochemistry
For histochemical studies, rehydrated sections were stained with the PAS procedure and Alcian blue (AB) at pH 2.5 to differentiate neutral and acidic glycoconjugates (Mowry and Winkler 1956).
Lectin Histochemistry
Binding with seven FITC-conjugated lectins and one phosphatase-conjugated lectin was performed to characterize and localize glycosidic residues in the gastric gland cells. The lectins used, their concentration, and their sugar specificities are listed in Table 1. For FITC-conjugated lectins (BSI-B4, ConA, PNA, LTA, SBA, UEA, WGA; Sigma, St. Louis, MO), rehydrated sections were incubated for 1 hr at room temperature with the conjugated lectin solution and rinsed in Tris-buffered saline (TBS) and mounted in a TBS-glycerine 70% mixture. For AAA phosphatase-labeled lectin (Vector Laboratories; Burlingame, CA), rehydrated sections were incubated for 1 hr at room temperature with the conjugated lectin in TBS (pH 7.4). The sections were rinsed in TBS and incubated in substrate working solution (BCIP/NBT Alkaline Phosphatase substrate Kit IV; Vector Laboratories) for 15 min at room temperature. The endogenous alkaline phosphatase activity was inhibited by the addition of levamisole to the working solution. The sections were washed in TBS (pH 9.5) for 5 min, counterstained with hemalum, dehydrated, cleared, and mounted with Entellan (Merck; Darmstadt, Germany). Lectin binding was also tested after β-elimination and α1,2-fucosidase digestion. β-elimination was performed with 0.2 M KOH in dimethylsulphoxide-H2O-ethanol (50:40:10) overnight at 45C and subsequent neutralization with 10 mM HCl. Only the O-linked glycans are removed from glycoproteins by this method (Ono et al. 1983). Fucosidase digestion was performed before LTA, UEA, and PNA lectins, by overlaying hydrated sections for 48 hr at 37C, in a humid Petri dish, with a solution containing 1,2-fucosidase (Sigma) diluted 1:200 in TBS 250 mM (pH 5.0).
Characteristics of the plant lectins used

Gal, galactose; GalNAc, N-acetylgalactosamine; GlcNAc, N-acetylglucosamine; L-Fuc, L-fucose; MαM, methyl-α-mannopyranoside; TACT, N,N′,N″-triacetylchitotriose.
Controls for the lectin labeling procedures included (a) substitution of the lectin with TBS and (b) incubation with the lectin with addition of the appropriate inhibitory sugar to confirm specificity of lectin labeling, as detailed in Table 1. Control of enzymatic activity was performed by incubating sections in enzyme-free TBS.
IHC
For IHC assay, the rehydrated sections were microwave pretreated in pH 6.0 citrate buffer for three 5-min cycles for antigen retrieval (Gown et al. 1993). The sections were treated with the blocking buffer (1% normal goat serum in TBS; Sigma) for 30 min at room temperature and incubated overnight with the primary rabbit antibody against the α -subunit of porcine H+,K+-ATPase (Calbiochem; San Diego, CA) diluted 1:500 in blocking buffer at 4C or with the primary mouse antibody against the β-subunit of pig H+,K+-ATPase (Novus Biologicals; Littleton, CT) diluted 1:500 in blocking buffer at 4C. After several rinses in TBS, sections were incubated with the secondary antibody as follows: in anti-rabbit Alexa Fluor 488 (Molecular Probes; Eugene, OR) diluted 1:500 for 5 hr at room temperature for α-subunit of H+,K+-ATPase or in anti-mouse Alexa Fluor 568 (Molecular Probes) diluted 1:500 for 5 hr at room temperature for β-subunit of H+,K+-ATPase. After several washes in TBS, sections were mounted in a TBS-glycerine 70% mixture. Negative controls were performed by omitting the primary antibodies or using antibodies preadsorbed with the immunizing peptide. Sections of rat alimentary tract were tested in parallel as positive controls. Expected positive results were obtained in oxyntic cells of rat gastric glands.
Semiquantitative Analysis
The images were captured using an epifluorescence E600 photomicroscope equipped with a DMX 1200 digital camera (Nikon; Kawasaki, Japan), with and without weak transillumination.
The IHC staining and the histochemical labeling of lectins used was assessed by two independent observers. The intensity of the labeling was classified as follows in Table 2: no labeling (0), weak labeling (1), moderate labeling (2), strong labeling (3).
Membrane Isolation
After being removed, stomachs were rinsed and transferred in ice-cold isolation medium (20 mM TRIS-HCl, 1 mM EDTA, 5 mM EGTA, 220 mM mannitol, 70 mM sucrose, 200 mM PMSF, 1 μg/μl leupeptin, and 1 μg/μl pepstatin; pH 7.4) at a weight-to-volume ratio of 1:10 before being submitted to apical scraping by cover slide. The mucosal layer was removed, resuspended in fresh isolation medium, and homogenized with 10 strokes in 1 min in a Potter-Elvehjem homogenizer at a speed of 100 rpm. The homogenate was filtered through a double layer of hydrophilic gauze and spun at 500 × g for 15 min at 4C to remove nuclei and unbroken cells. The pellet was discarded, and the supernatant was collected and centrifuged at 6000 × g (15 min, 4C) to eliminate the mitochondrial fraction. Finally, the supernatant was harvested and centrifuged at 150,000 × g (1 hr, 4C) to obtain a pellet containing the plasma and tubulovesicular membranes. The pellet was resuspended in isolation medium and used for the subsequent immuno- or lectin blotting analyses or frozen at −80C. The protein content of the sample membranes was essayed by the Lowry method (Lowry et al. 1951).
Semiquantitative analysis of lectins used
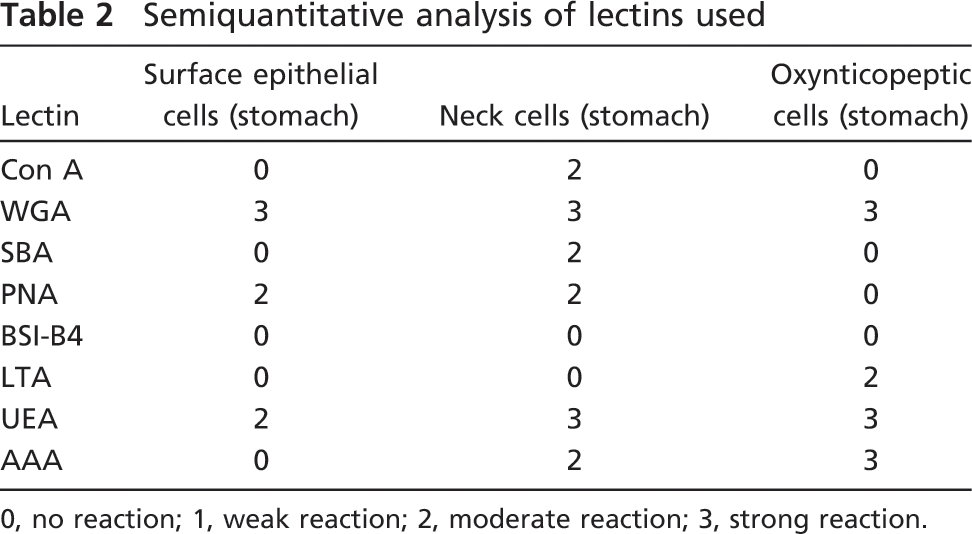
0, no reaction; 1, weak reaction; 2, moderate reaction; 3, strong reaction.
Electron Microscopy
For electron microscopy (EM), samples of stomach were fixed in a mixture of 3% paraformaldehyde and 1% glutaraldehyde in 0.1 M PBS, at pH 7.4, for 4 hr at 4C; some were postfixed in 1% OsO4 in PBS for 20 min at 4C. Fixed samples were washed in several changes of PBS, dehydrated in graded alcohols, and embedded in Epon-Araldite (TAAB; Reading, UK). Sections of 2 μm thick were heat stained with PAS–Toluidine blue borate and PAS–Emallume (Millonig 1976). Ultrathin sections for EM were mounted on formvar-coated nickel grids and stained routinely with uranyl acetate and lead citrate (Reynolds 1963).
Immunoelectron Microscopy
For immunoelectron microscopy, ultrathin non-osmicated sections were mounted on formvar-coated gold grids. The grids were incubated for 30 min at room temperature with blocking buffer (TBS-1% normal goat serum; Sigma). Sections were incubated overnight with the primary rabbit antibody against the α-subunit of porcine H+,K+-ATPase (Calbiochem) diluted 1:500 in blocking buffer at 4C and with the primary mouse antibody against the β-subunit of pig H+,K+-ATPase diluted 1:500 in blocking buffer at 4C. The grids were rinsed with PBS and incubated with anti-rabbit IgG 10-nm gold-conjugated antibody (Sigma) diluted 1:10 in TBS for 1 hr at room temperature. After several rinses in TBS and in distilled water, the grids were lightly stained with uranyl acetate-lead citrate. Immunolabeling controls were performed using antibodies preadsorbed with immunizing peptides or by omitting the primary antibodies. The grids were observed under a Morgagni 268 electron microscope (FEI; Hillsboro, OR).
Deglycosylation
For N-glycosidase F (PNGase F) digestion, 60 μg of the membranes prepared as above were incubated at 37C for 4 hr in the presence of 2 U of N-glycosidase F (Boehringer Mannheim; Mannheim, Germany). The enzymatic reaction was stopped by adding one volume of 2 × Laemmli buffer and boiling the resulting mixture at 90C for 4 min. Samples were analyzed by immunoor lectin blotting.
Immuno- and Lectin Blotting Analyses
Aliquots (50 μg protein) of membranes prepared as above were heated to 90C and electrophoresed in an SDS/13% acrylamide gel (Mighty Small II; Amersham Biosciences, San Francisco, CA) using a low molecular mass protein ladder (Amersham Biosciences; Buckinghamshire, UK). The resolved proteins were transferred electrophoretically onto nylon membranes that were blocked in 5% (w/v) low-fat milk in blocking solution (20 mM Tris-HCl, 0.15 M NaCl, 1% Triton-X100, pH 7.5) for 1 hr.
For the immunoblotting studies, the membranes were incubated with an affinity-purified mouse monoclonal antibody directed against the β-subunit of pig H+,K+-ATPase (Novus Biologicals) at a final dilution of 1:1000 in blocking solution. After washing (4 × 15 min) in blocking buffer, the membrane was incubated for 1 hr with peroxidase-conjugated goat anti-mouse IgG antibody (Sigma). After washing in blocking solution (3 × 10 min), blots were visualized for peroxidase activity by enhanced chemiluminescence (ECL plus kit; Amersham Biosciences). Negative controls were performed by omitting the H+,K+-ATPase antibody.
For the lectin blotting analysis, the nylon membranes were incubated with AAA-phosphatase at a final concentration of 40 μg/ml blocking solution, WAG-peroxidase at a final concentration of 50 μg/ml blocking solution, or UEA-peroxidase at a final concentration of 10 μg/ml blocking solution, conjugated lectins directed against the α-1,6-fucose, N-acetyl-glucosamine, and α-1,2-fucose residues of the glycosidic portion of the glycoproteins, respectively. After washing (5 × 10 min) in blocking solution, the membranes incubated with AAA-phosphatase were visualized by the nitroblue tetrazolium/5-bromo-4-chloro-3-indoyl phosphate (NBT/BCIP) (Vector Laboratories) and ECL-plus methods, respectively. For the NBT/BCIP method, after being washed with the blocking solution, membranes were incubated with 10 mM TRIS-HCl, pH 9.5, for 5 min under stirring, and the lectin-reactive bands were revealed with the NBT/BCIP solution as reported by the manufacturer.
Results
In this study, we investigated the gastric mucosa of R. esculenta by conventional histochemistry to identify the presence of glycoproteins and lectin histochemistry to characterize the oligosaccharide chains of these glycoproteins.
Histology
The stomach of R. esculenta is lined with a single layer of mucous surface cells. These were PAS positive but not alcianophilic. The gastric mucosa contains numerous glands. The fundic glands are mainly of the branched tubular type and consist of mucous neck cells, endocrine cells, and oxynticopeptic cells, secreting mainly hydrochloric acid (Figures 1A–1C). The glandular lumen was wide in the fasting animals (Figure 1A) and rather reduced in the fed ones (Figure 1B). In the sections stained with PAS–Toluidine blue, oxynticopeptic cells often showed several, small pepsinogen granules (Figure 1C). Lipid droplets were frequent in the basal area of the cytoplasm (Figure 1C). Oxyntico-peptic cells did not differ significantly between the oral and aboral fundus.
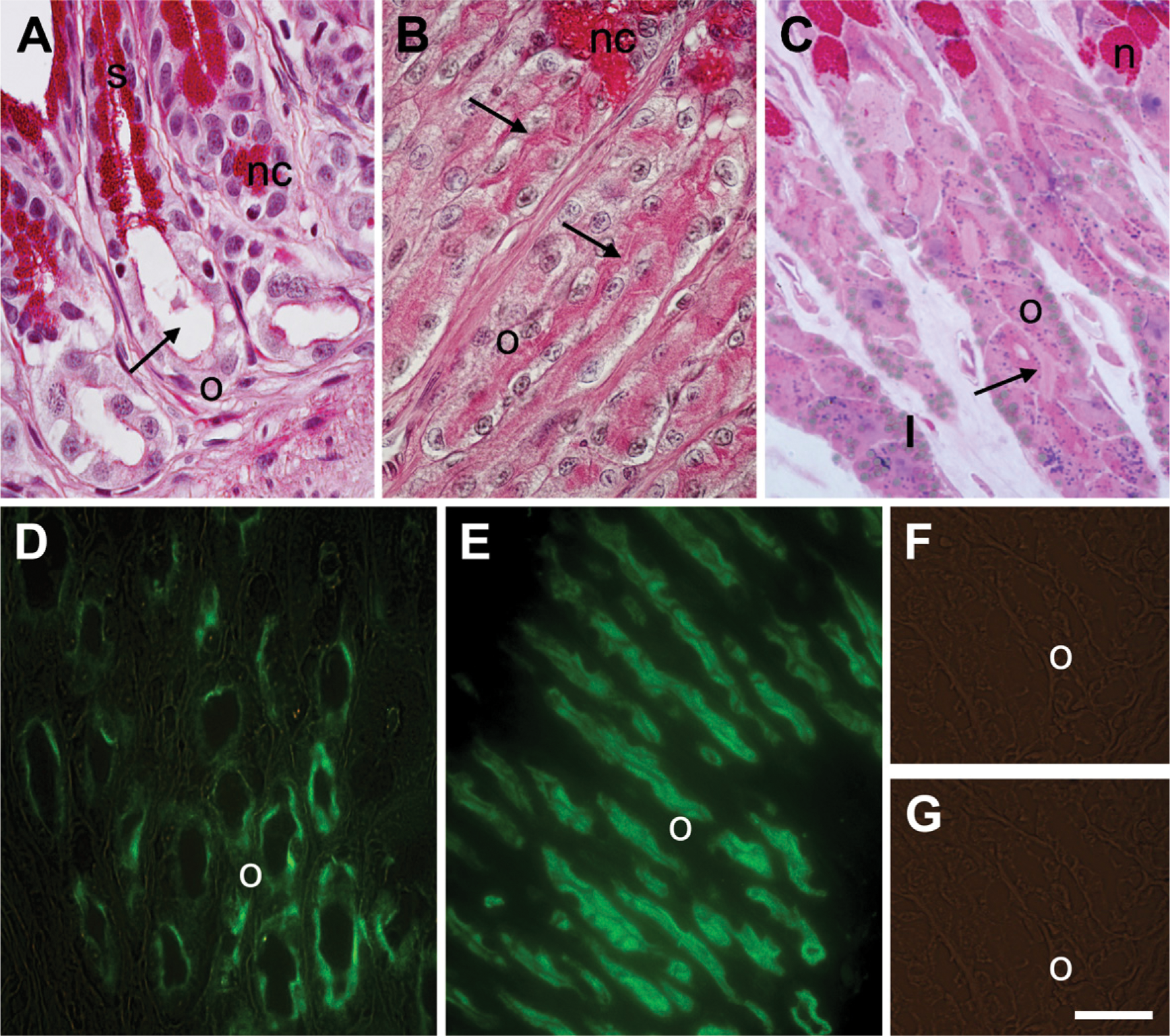
Gastric mucosa of Rana esculenta in fasting and fed conditions. Histochemical and IHC stainings. (
Carbohydrate Conventional Histochemistry
The luminal membrane and the apical cytoplasm of cid-secreting cells, as well as the mucous surface cells and the glandular neck cells, were PAS positive (Figures 1A–1C) but not alcianophilic. PAS staining decreased after β-elimination. In fasting animals, a thin layer of apical cytoplasm made up of oxynticopeptic cells stained intensely with PAS reaction (Figure 1A). In the fed animals, the PAS-positive layer was thicker compared with the fasting animals. This layer corresponds to the luminal membrane system, characterized by several, long microvilli that reduced the glandular lumen (Figures 1B and 1C).
IHC
In the fasting animals, the PAS-positive layer also reacted with the primary antibody against the H+,K+-ATPase α-subunit (Figures 1D). A thicker layer was immuno-reactive in the fed animals (Figure 1E). Surface mucous cells and glandular neck cells did not show affinity for this antibody (Figures 1D and 1E). In both fasting and fed animals, labeling was not observed in control sections when the primary antibody was substituted by PBS (Figure 1F) or when antigen-preadsorbed antibodies were used. No immunostaining was seen with the anti-H+,K+-ATPase β subunit antibody (Figure 1G).
Lectin Histochemistry and Enzymatic Digestion
We localized and characterized the glycosidic residues of oligosaccharide chains by lectin histochemistry. Lectin binding sites did not change qualitatively between the fed and fasted animals. In oxynticopeptic cells, the same areas that were PAS positive also strongly bound WGA (Figure 2A), AAA (Figure 2B), and UEA (Figure 2C), and less intensely, LTA (Figure 2D). No binding was seen with the other lectins tested. Labeling with LTA decreased after α1,2-fucosidase digestion, indicating Fuc residues linked α1,2 to the outermost Gal of the oligosaccharide chains, whereas binding with UEA was abolished. Pretreatment with α1,2-fucosidase imparted reactivity with PNA (Figures 2E and 2F), indicating a Fuc α1,2 Gal linkage. Surface epithelial cells reacted with UEA, PNA, and WGA, the latter showing the highest reactivity (Figures 2A, 2C, and 2E). Mucous neck cells showed affinity for all the lectins tested, except for LTA and BSI B4 (Figure 2D).
In control sections, no staining was observed after substituting the horseradish peroxidase-conjugated lectins with PBS. Interestingly, the lectin binding was significantly inhibited by addition of the corresponding hapten sugar. Results of lectin histochemistry are summarized in Table 2.
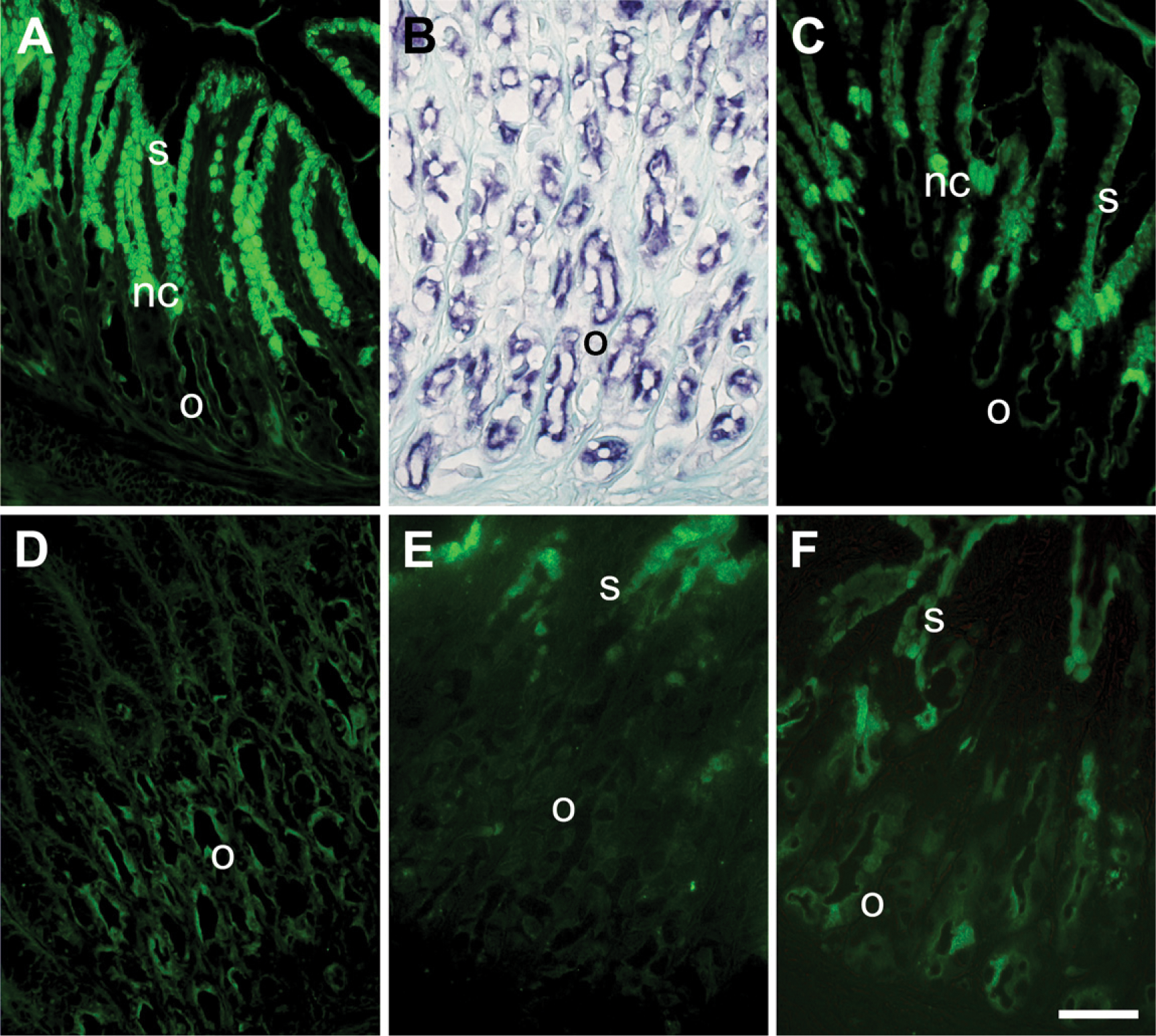
Gastric mucosa of R. esculenta. Lectin labeling on paraffin sections. (
Immuno- and Lectin Blotting
Immunoblotting experiments were carried out using isolated frog gastric mucosal membranes prepared as described in Materials and Methods. Incubation with a monoclonal antibody directed against the β-subunit of H+,K+-ATPase showed three immunoreactive bands with molecular masses of ∼32, 58, and 67 kDa, respectively (Figure 3A). Immunoblotting of mucosal membranes deglycosylated by the PNGase enzyme showed a considerable reduction in intensity of the 67- and 58-kDa bands, with a parallel strong increase in the immunoreactivity of the 32-kDa band. Experiments of lectin blotting carried out with conjugated WGA, UEA, and AAA showed multiple reactive bands, among which the 58-kDa band corresponded to the glycosylated form of the H+K+-ATPase β-subunit (Figure 3B).
EM
The fine structure of the surface mucous cells did not present significant differences between the fasting and fed animals, whereas the most significant changes occurred in the oxynticopeptic cells.
Surface Mucous Cells
The surface mucous cells were cylindrical in shape (Figure 4A). Their luminal membranes had short microvilli, whereas their lateral membranes showed several interdigitations. The supranuclear cytoplasm was filled with numerous small, elongated mucous granules with a variable electron density. Near the basal membrane (Figure 4A), several tonofilaments were seen.
Mucous Neck Cells
The mucous neck cells were filled with rounded or polygonal mucous granules that were large and closely packed (Figure 4B). They showed uniform moderate electron density.
Oxynticopeptic Cells
The oxynticopeptic, mainly acid-secreting cells, of R. esculenta differ from mammalian parietal cells in that they lack intracellular canaliculi. In fasting animals, the oxynticopeptic cells showed a relatively developed rough endoplasmic reticulum (rER), localized mainly in the basal part of the cell (Figures 4C and 4D). The smooth endoplasmic reticulum (sER) was well developed, similar to the mammalian parietal cell counterpart, and consisted of short tubules and vesicles extending into the supranuclear cytoplasm. The luminal membranes of the oxynticopeptic cells were lifted in short and stocky microvilli. Some cells contained granules quite similar to the zymogen granules of the chief cells (Figures 4C and 4D).
In fed animals, the luminal membrane of the oxyntico-peptic cells was characterized by several long microvilli that projected into the glandular lumen that appeared significantly reduced. The tubulovesicular system was reduced and consisted mostly of expanded vesicles (Figures 4E and 4F).
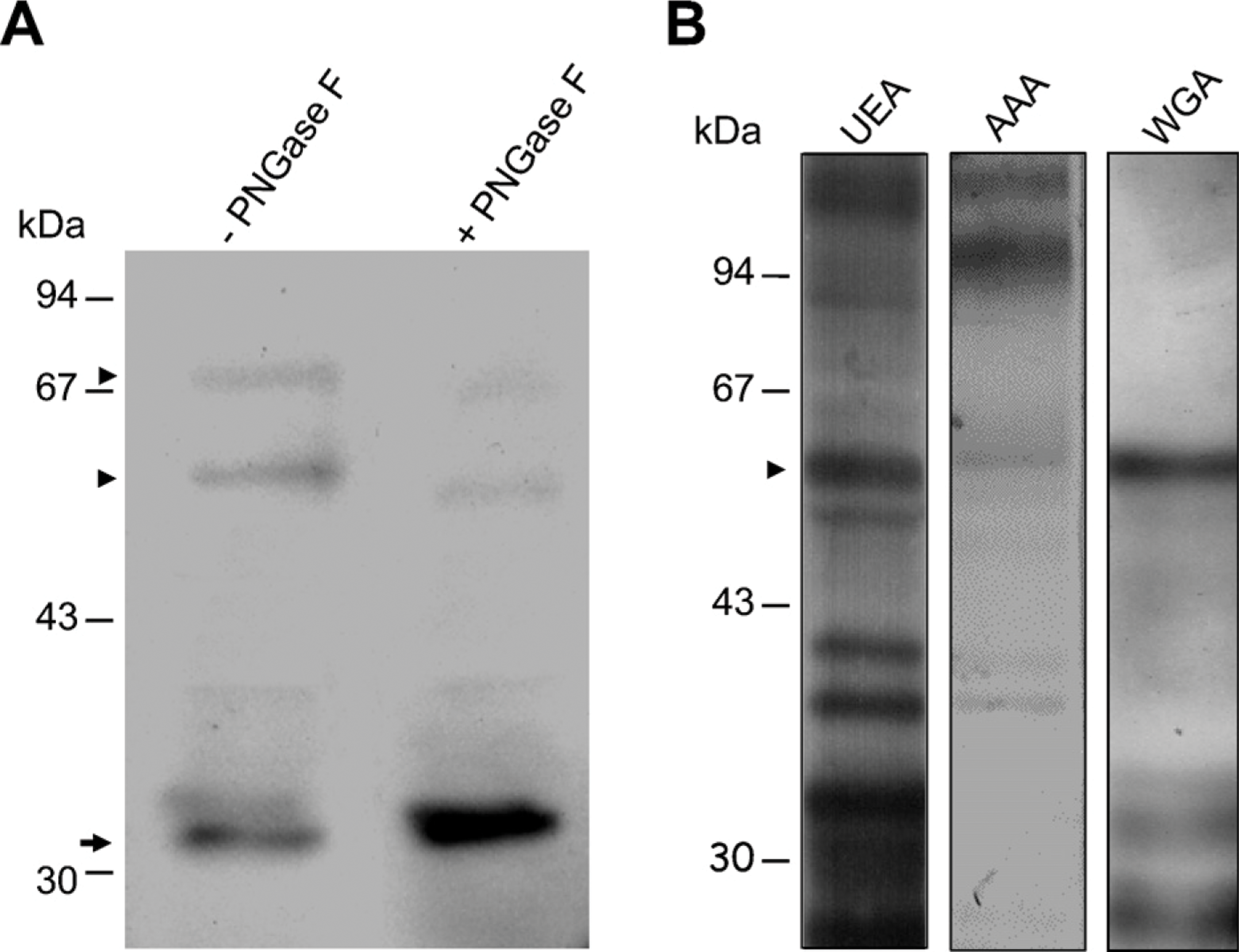
Lectin and immunoblotting analyses. (
Endocrine Cells
Various types of cells with ultrastructural features similar to mammalian endocrine cells were also found in fundic glands.
Immunoelectron Microscopy
Immunogold staining with the anti-H+,K+-ATPase α-subunit resulted in numerous gold particles on the microvilli of the luminal membranes and on the membranes of the tubulovesicular system. In the fed animals, the number of gold particles was higher than in the fasted animals (Figures 4D and 4F). Gold particles were also seen on the Golgi complex, rER, and some lysosomes. No immunostaining was seen with the anti-H+,K+-ATPase β-subunit antibody.
Discussions
The histological and ultrastructural features of R. esculenta gastric glands are known (Wright et al. 1957; Geuze 1971a; Bani et al. 1992; Gallego-Huidobro and Pastor 1996). No data have been published regarding the lectin-binding pattern of frog gastric glands. The results of our study showed that the epithelial surface cells of the stomach of R. esculenta mainly produce neutral glycoproteins with glycosidic residues of (GlcNAc β1,4)n and Gal β1,3 GalNAc and Fuc α1,2 linked to the outermost Gal of the oligosaccharide chains. The gastric glands consist of mucous neck, oxynticopeptic, and endocrine cells. Mucous neck cells produce neutral mucins with glycosidic residues of mannose and/or glucose: (GlcNAc β1,4)n, GalNAc, and Gal β1,3 GalNAc. These cells show binding sites for AAA and UEA, but not for LTA, and thus probably contain Fuc-linked α1,6 to the innermost GlcNAc of the core in N-linked glycans or terminal Fuc linked to the outermost Gal of the oligosaccharide chains. The oxynticopeptic cells of gastric glands of R. esculenta secrete mainly hydrochloric acid, pepsinogen being secreted mostly by the peptic cells located in the esophageal glands (Liquori et al. 2002). Similar to mammalian parietal cells, the oxynticopeptic cells present an extended membrane system correlated to HCl secretion, although the plasma membrane does not fold into intracellular canaliculi. In any case, the ultrastructural features of the amphibian oxynticopeptic cells change in relation to the functional status (Geuze 1971b). In this study, the features of the oxynticopeptic cells in fed and fasting frogs corresponded to those previously described (Logsdon and Machen 1982). In fact, in fasting frogs, the oxintopeptic cells presented a very extensive tubulovesicular system, whereas the luminal membrane had few, short microvilli. In contrast, the oxynticopeptic cells in fed animals showed features typical of active secreting cells, with a reduced tubulovesicular system with enlarged cisternae and an extensive system of microvilli.
In R. esculenta, the luminal membrane and tubulo-vesicular system of the oxynticopeptic cells reacted with the primary gold-conjugated antibody against the H+,K+-ATPase α-subunit. In the fed animals, the immunoreactivity against the H+,K+-ATPase α-subunit was higher than in the fasting ones because of a higher content of H+,K+-ATPase or, alternatively, more binding sites of the antibody were available to the H+,K+-ATPase. In light microscopy, the same areas also appeared to be PAS positive and thus were rich in glycoproteins. Histochemical reactions indicates that these glycoproteins had prevalently N-linked neutral oligosaccharide chains, which were variously fucosylated, as shown by the binding with LTA, UEA, and AAA lectins. The binding pattern of these lectins varied because their specificity toward fucose depends on the oligosaccharide chain structure. It is known that the binding of different lectins with affinity to a given sugar can vary in relation to the links between the sugar and its neighbors (Spicer and Schulte 1992). AAA has a major binding affinity to Fuc α1,6 linked to the proximal GlcNAc of the N-linked glycan core and to terminal Fuc with an α1,3 or α1,4 linkage. LTA and UEA-I have higher affinity for Fuc with an α1,2 linkage (Alonso et al. 2003).
In the oxynticopeptic cells of R. esculenta, we hypothesized the presence of glycans having (poly)lactosamyl chains with fucose α1,2 linked to galactose, as indicated by the increased affinity for PNA after α1,2-fucosidase treatment. Fucose α1,3 linked to GlcNAc was also present, as indicated by unchanged LTA affinity after α1,2 fucosidase treatment. Furthermore, AAA labeling indicated the presence of an α1,6-fucosylated core in the N-glycans. It is known that, in mammals, the tubulo-vesicular membranes are rich in proteins with several functions. The proton pump H+,K+-ATPase is the most abundant. It is responsible for secreting acid into the stomach lumen. H+,K+-ATPase is a heterodimer, with a catalytic α-subunit and an associated β-subunit. The latter is a heavily glycosylated protein essential for holoenzyme function (Tyagarajan et al. 1997).
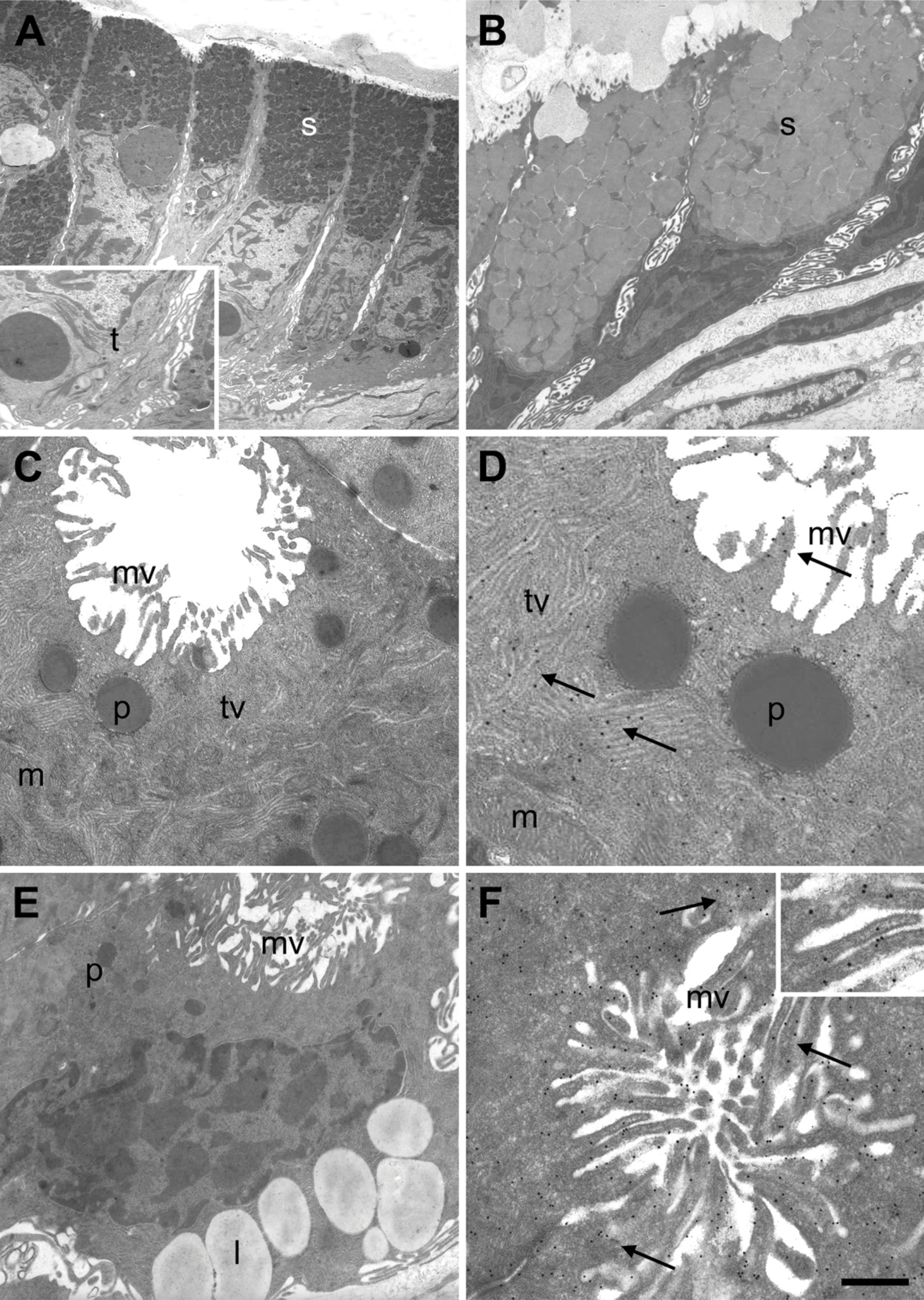
Electron micrographs of secretory cells in gastric mucosa of R. esculenta in fed and fasting conditions. (
In the rabbit (Tyagarajan et al. 1996, 1997), β-H+,K+-ATPase is an integral glycoprotein with a short N-terminal cytoplasmic domain, one transmembrane domain, and an extensive extracellular domain that is heavily glycosylated. The extracellular domain consists of polypeptide chains with seven potential sites of N-glycosylation. Two oligosaccharide chains are oligomannosidic; the others are of the complex type, tri- or tetra-antennary, with branches having lactosamine or polylactosamine structure and terminating in α-linked Gal residues, with a fucosylated core (Tyagarajan et al. 1997). α-Galactosyl residues are found in the rat, but not in the mouse, pig, or dog (Stewart et al. 1999).
The glycosylation sites are conserved among different species of mammals, although differences in the nature of the oligosaccharide chains may occur between species (Crothers et al. 1995; Appelmelk et al. 1996; Stewart et al. 1999). In all the species studied, the oligosaccharide chains are asialylated. The presence of branched fucosylation on mouse and human gastric β-subunit has been suggested (Appelmelk et al. 1996).
It has been hypothesized that the H+,K+-ATPase β-subunit oligosaccharide chains may play a role in the trafficking (Chow et al. 1992) and protection of the enzyme in the harsh acidic environments of the stomach (Chow and Forte 1993). In all species examined, glycans of H+,K+-ATPase β-subunits are asialylated (Tyagarajan et al. 1997). The extracellular side of the H+,K+-ATPase β-subunit has been shown to be more resistant to trypsinolysis. Resistance to trypsinolysis is not affected by terminal-galactose removal, but it is reduced by complete deglycosylation (Tyagarajan et al. 1995; Crothers et al. 2004). It can be supposed that the intrinsic folding of β-H+,K+-ATPase, its oligosaccharide chains, and the links between the α- and β-subunits allows the luminal domain of the protein to resist against gastric juice lysis. The β-H+,K+-ATPase with a highly mannosylated core has the same sensitivity to trypsinolysis as the fully mature form. Thus, it could be inferred that the glycoside composition of the oligosaccharides is less important than the sole presence of N-linked oligosaccharides. The complete absence of N-linked oligosaccharides inhibits H+,K+-ATPase activity. Several pathologies arise as a consequence of malfunctioning glycosylation mechanisms, so a better knowledge of glycan functions could help to prevent them (Thangarajah et al. 2002).
We can hypothesize that, in R. esculenta, the β-subunit of H+,K+-ATPase will also be the most abundant glycoprotein in the tubulovesicular system of oxynticopeptic cells and thus be the major cause of histochemical staining of these cells.
The comparison between the results of immunolectin blotting carried out with antibody against the β-subunit of H+,K+-ATPase with the lectins and the results of conventional histochemistry reactions raises speculation that the β-subunit of H+,K+-ATPase in the frog is a neutral glycoprotein with N-linked oligosaccharide chains. The non-reactivity of IHC can be attributed to steric hindrance that did not allow the antibody β-H+,K+-ATPase to bind itself to the specific epitopes of the polypeptide chain. The immunoblotting experiments showed three immunoreactive bands with molecular masses of ∼32, 58, and 67 kDa, respectively. A band at 58 kDa could correspond to β-H+,K+-ATPase, and a less intense band at 67 kDa could correspond to an isoform of the same protein. This glycoprotein shows a band of ∼32 kDa, which corresponded to a protein core to which are bound oligosaccharide chains with α1,6-fucosylated cores and polylactosamine antennas, asialylated, rich in N-acetylglucosamine with Fuc α1,2 linked to subterminal galactose and/or α1–3 to the N-acetylgalactosamine.
In conclusion, our study allowed us to show the presence of H+,K+-ATPase in the luminal membrane and the tubulovesicular system of the oxynticopeptic cells of a frog and to hypothesize the structural pattern of the glycosidic component of the β-subunit of H+,K+-ATPase. The oligosaccharide chains of the β-subunit are somewhat different from those described in mammals, and this is probably because of an adaptation to the different microenvironmental conditions in which the oxynticopeptic cells find themselves in relationship to different habits and phylogeny.
Comparative morphofunctional studies between amphibians and mammals could help in clarifying the pathological consequences of altered oligosaccharide chain sequences.
