Abstract
Amplification of immunological signals with catalyzed reporter deposition (CARD) allows improved detection of scarce tissue antigens in light and electron microscopy. The technique takes advantage of the oxidation ability of horseradish peroxidase (HRP), in the presence of hydrogen peroxide, to yield the accumulation of one of its specific reporter-tagged substrates. This immunocytochemical approach continues to be improved by the introduction of new reporter molecules tagged to tyramine or to other HRP substrates. In this study we introduced a novel HRP substrate tagged to Nanogold particles. The amplification protocol is based on the application of a specific primary antibody, a biotinylated secondary antibody, streptavidin-HRP, and an HRP substrate coupled to Nanogold, followed by silver intensification. In addition to amplification of immunological signals of high resolution, direct accumulation of Nanogold particles at target sites by enzymatic activity of HRP improves the efficiency of the technique compared to other amplification protocols. Moreover, this approach combines the CARD amplification potentials with the ultrasmall gold probe and the silver intensification method. Immunolabeling obtained by light and electron microscopy, as well as immunodot assay using this new amplification strategy, appear to be highly sensitive, specific, and of enhanced intensity.
The catalyzed reporter deposition approach (CARD) is now widely used in cell biology for membrane immunoassays, Western blotting, in situ hybridization, and immunocytochemistry (Bobrow et al. 1989; Adams 1992; Wigle et al. 1993; Kerstens et al. 1995; Mayer and Bendayan 1997, 1999; Schöfer et al. 1997; Humbel et al. 1998). A majority of studies have reported greater sensitivity with reduced background and enhancement of antigen detection between 8- and 10,000-fold, depending on the level of resolution and the protocol used (Bobrow et al. 1991; Adams 1992; Merz et al. 1995; Hunyady et al. 1996; Werner et al. 1996; Erber et al. 1997). The procedure is based on the action of the immunologically introduced HRP to catalyze the deposition of substrate reporter molecules by a short half-life free radical mechanism (Bobrow et al. 1989). This amplification procedure was recently adapted to electron microscopy to reveal intracellular antigens with high resolution by modifying the original protocol. New reporter molecules coupled to tyramine were introduced and allowed the sensitive detection of antigenic sites with the particulate colloidal gold marker (Mayer and Bendayan 1999).
To expand the possibilities of this approach we have now developed a novel probe, the HRP substrate-Nanogold, which takes advantage of the small 1.4-nm Nanogold particles and of the silver enhancement protocol. Both the Nanogold and the silver enhancement contribute to higher labeling intensities. Small gold particles such as those of 1–5-nm colloidal gold and Nanogold are known to give higher labeling densities (Lackie et al. 1985; Ghitescu and Bendayan 1990; Hainfeld and Furuya 1992; Dulhunty et al. 1993), partly because of reduced steric hindrance and the overall ratio between immunoglobulin molecules and gold particles. In addition to its small size, Nanogold offers advantages such as its stable covalent linkage to proteins and its neutral isoelectric point which prevents interactions with negatively charged molecules on tissue sections. The silver intensification protocol also provides several advantages by improving detection limits (Holgate et al. 1983) and generating high contrast, non-diffusible, permanent black staining (Holgate et al. 1983; Taatjes et al. 1987). Hacker et al. (1997) and Zehbe et al. (1997) have already used the combination of the CARD technique with streptavidin-Nanogold and silver autometallography in in situ hybridization (ISH) experiments. They reported single gene copy sensitivity in detecting viral DNA and very high sensitivity for RNA ISH. They also suggested that CARD-Nanogold could have the potential to replace in situ polymerase chain reaction in many applications.
In this study we used three different immunolabeling approaches in light and electron microscopy to assess the amplification obtained with a novel HRP substrate-Nanogold probe. The simple indirect streptavidin-Nanogold protocol was compared to biotinyl-LC-LC-tyramide/streptavidin-Nanogold and to HRP substrate-Nanogold. The results obtained with the latter approach appear superior in allowing the immunolocalization of low amounts of antigen-antibody complexes. Moreover, the CARD-Nanogold amplification system was assessed on membrane immunoassay and was proved to be quite sensitive.
Materials and Methods
Preparation of a Novel HRP Substrate-Nanogold
A new HRP substrate consisting of ligands other than tyramine was covalently coupled to a 1.4-nm Nanogold-type gold cluster according to previously described techniques (Hainfeld 1987). The reagent product was purified by size exclusion chromatography using an Amicon GH-25 matrix in aqueous buffer. The Nanogold peak was collected and rotary evaporated to dryness and resuspended in 20% methanol. This prototype reagent was generated and provided by Nanoprobes (Stony Brook, NY).
Immunocytochemistry
For light microscopic immunocytochemistry, fragments of normal rat pancreas were fixed in Bouin's fixative and embedded in paraffin according to standard techniques. Five-μm-thick sections were mounted on glass slides, deparaffinized, and rehydrated through decreasing concentrations of ethanol. After washing in 0.01 mol/liter PBS, pH 7.2, the sections were incubated with 3% H2O2 in PBS for 10 min to quench endogenous peroxidase activity. The sections were then briefly washed in PBS and incubated with the specific guinea pig anti-porcine insulin antibody (ICN; Costa Mesa, CA) diluted 1:200 or 1:10,000 in PBS for 2 hr at room temperature (RT). The tissues were washed twice for 5 min in PBS and once for 5 min in 0.15 mol/l NaCl, 0.05% Tween-20 (Sigma-Aldrich Canada; Oakville, Ontario, Canada), 0.1 mol/liter Tris-HCl buffer, pH 7.5 (Tris-Tween buffer), before incubation in Tris-1% bovine serum albumin (BSA) solution for 30 min. Then the biotinylated goat anti-guinea pig IgG antibody (GAG-B; Jackson Immunoresearch Laboratories, West Grove, PA) diluted 1:500 in Tris, pH 7.5, containing 0.5% BSA (Tris-BSA) was applied to the sections for 1 hr at RT. After three washes of 5 min each in Tris-Tween buffer, the tissue sections intended for the indirect simple labeling protocol were incubated for 30 min with streptavidin-Nanogold (Nanoprobes) diluted 1:500 in Tris buffer. The sections intended for the amplification protocol were incubated for 30 min with streptavidin-HRP (NEN Life Science; Boston, MA) diluted 1:500 in Tris-BSA, washed in Tris-Tween buffer for 15 min, and incubated for 10 min with the HRP substrate-Nanogold complex diluted 1:50 in 0.05 mol/liter Tris-HCl, pH 7.5, containing 0.03% H2O2. The sections processed for simple and amplified protocols were then washed for 15 min in Tris-Tween buffer and rinsed several times in ultrapure water. Enhancement of the Nanogold was performed by incubating the slides in the LI Silver enhancement solution (Nanoprobes) for about 20–30 min. For each of the protocols, the gold-silver development was allowed to proceed until reaching an optimal signal: maximal specific reaction with low levels of background. Sections were then thoroughly rinsed with water for 5 min, mounted in PBS-50% glycerol without counterstaining, and observed under a Leitz Orthoplan microscope (Leica Microsystems; Montreal, Quebec, Canada).
For the electron microscopic immunocytochemical study, small fragments of normal rat pancreas were fixed by immersion with 0.1 mol/liter phosphate-buffered 1% glutaraldehyde for 2 hr at RT, washed in the same buffer, dehydrated in methanol, and embedded in Unicryl at −25C as described previously (Bendayan 1995). Thin sections were cut, mounted on Parlodion-carbon-coated nickel grids, incubated for 30 min on a drop of 1% ovalbumin in PBS, and transferred to a drop of the rabbit anti-human amylase antibody (Sigma-Aldrich) diluted 1:10 or 1:200 in PBS or the guinea pig anti-insulin antibody diluted 1:200 or 1:10,000 for 2 hr at RT or overnight at 4C. The grids were rinsed for 15 min with PBS and placed on a drop of the 1% ovalbumin solution for 15 min. The sections to be processed for simple indirect labeling were incubated with the Nanogold-conjugated goat anti-rabbit IgG or with the Nanogold-conjugated goat anti-guinea pig IgG (Nanoprobes) diluted 1:100 in PBS for 30 min at RT. The sections to be processed for the CARD amplification protocol were incubated with the biotinylated goat anti-rabbit IgG (GAR-B; Sigma-Aldrich) diluted 1:100 in PBS or with the biotinylated anti-guinea pig IgG antibody diluted 1:500 in PBS for 1 hr, washed in the same buffer, incubated with streptavidin-HRP 1:500 for 30 min, washed twice in PBS and once in Tris, and incubated with the HRP substrate-Nanogold complex diluted 1:25 in 0.05 mol/liter Tris-HCl, pH 7.5, containing 0.03% H2O2 for 10 min. After these incubations, the tissue sections were washed with PBS for the simple protocol and with Tris buffer for the amplified protocol. The grids were then thoroughly washed with ultrapure water, silver-enhanced in the HQ Silver solution (Nanoprobes) for 4 min., washed again with water, air-dried, and stained with uranyl acetate. Grids were examined with a 410 SL Philips electron microscope.
The biotinyl-long chain-long chain-tyramide (biotinyl-LC-LC-tyramide) protocol was also tested for its amplification properties. For a fair comparison, streptavidin-Nano-gold and silver enhancement were used as the detection step for the deposited biotin molecules. For the tyramide amplification protocol in electron microscopy, we followed the previously described technique (Mayer and Bendayan 1999). Briefly, the grids were incubated on drops of 2% BSA in PBS, transferred onto drops of the anti-amylase (1:200 in PBS) overnight at 4C, washed, and incubated on a drop of GARB (1:100 in PBS) for 60 min. The sections were then washed, transferred to the BSA solution, incubated on a drop of streptavidin-HRP (1:500 in PBS) for 30 min, washed again, and incubated for 10 min with the biotinyl-LC-LC-tyramide probe diluted 1:50 in PBS containing 0.03% H2O2. After several washes, the accumulated biotin molecules were detected by a 30-min incubation with the streptavidin-Nanogold (1:500 in PBS). The grids were then silver-enhanced with the HQ Silver solution for 4 min, stained with uranyl acetate, and examined in the electron microscope.
The specificity of the immunolabelings was assessed by several control experiments, including antigen adsorption, omission of the primary or secondary antibody, omission of the streptavidin-HRP reagent, or omission of the HRP substrate-Nanogold.
Immunodot Assay
Normal rabbit IgGs were obtained from Jackson Immunore-search (Bio/Can; Mississauga, Ontario, Canada) and serial dilutions in PBS were made from a starting concentration of 10 μg/ml down to 0.001 μg/ml. One μl of each solution was spotted on a strip of nitrocellulose membrane (0.45 μm) (Gibco BRL; Burlington, Ontario, Canada) and allowed to dry for 30 min. The membrane was then blocked for 60 min with 5% BSA in PBS containing 0.05% Tween (PBST), incubated with the biotinylated anti-rabbit IgG diluted 1:500 with 1% BSA in PBST for 2 hr at RT, and washed for 15 min with PBST buffer. The strip was incubated with streptavidin-HRP (1:500 in PBST containing 1% BSA) for 30 min at RT, washed in PBST buffer for 15 min, rinsed in Tris, and amplified using the HRP substrate-Nanogold complex diluted 1:200 in 0.05 mol/liter Tris-HCl, pH 7.5, containing 0.03% H2O2 for 10 min at RT. After washing with Tris-Tween buffer and deionized water, the nitrocellulose membrane was treated with the LI Silver enhancement solution for 30 min, washed in water, and air-dried. For control, the same protocol was followed with one exception, the omission of the incubation with the HRP substrate-Nanogold.
Results
In our comparative study of the CARD-Nanogold amplification system by light and electron microscopy, we first applied the simple protocol, making use of the Nanogold coupled to a species-specific secondary antibody. Amylase antigenic sites in pancreatic acinar cells were revealed. In electron microscopy, this simple indirect labeling technique along with a rather diluted primary antibody, followed by silver enhancement, resulted in a particulate black signal over the different cellular compartments involved in protein secretion. With the anti-amylase antibody diluted 1:200, specific labeling but of low intensity was present over the rough endoplasmic reticulum, the Golgi apparatus, the condensing vacuoles, and the secretory granules (Figure 1A). For the amplification protocols, using the same antibody dilution, the biotinyl-long chain-long chain-tyramide probe combined with the streptavidin-Nanogold reagent and silver enhancement led to higher intensities of labeling over structures containing amylase. However, some background staining was obtained over nuclei and mitochondria (not shown). To improve the results, a novel signal amplification protocol was developed using a new probe, the HRP substrate-Nanogold. This HRP substrate-Nanogold protocol consists of direct and specific deposition of Nanogold on its reaction with peroxidase. Immunolocalization of amylase in the acinar cells using this novel approach led to significant increases in signal compared to that obtained with the simple approach. Electron-dense particulate deposits in high numbers were present over cellular compartments containing amylase (Figure 1B). This labeling was associated with low background staining over nuclei and mitochondria. Moreover, application of this protocol with a more concentrated primary antibody led to very high labeling intensities detectable at low magnification and allowing a general overview of the labeled tissue (Figure 1C). Despite the lack of counterstaining, the silver-intensified reaction product permitted clear outlining of the labeled structures. The dark labeling present over the RER, the cisternae of the Golgi apparatus, the condensing vacuoles, and the secretory granules made possible a high definition of amylase-secreting cells and organelles at low magnification (Figure 1C). In addition, nuclei and mitochondria showed very little labeling, demonstrating the overall high specificity of the reaction.
By light microscopy, immunodetection of insulin antigenic sites in rat pancreatic islet cells revealed a strong positive signal when the primary antibody was used at 1:200 dilution with both the simple (streptavidin-Nanogold) and the amplified (HRP substrate-Nanogold) immunolabeling technique (Figures 2A and 2C). The staining appeared as a black precipitate localized over the major part of the islets of Langerhans, contrasting with the surrounding acinar cells devoid of labeling. Differences between the simple and the amplified technique were more apparent when the primary antibody against insulin was used at much higher dilutions. The use of the anti-insulin antibody at 1:10,000 dilution, in combination with the simple indirect labeling technique (streptavidin-Nanogold) and silver enhancement, resulted in lack of staining over the pancreatic islets (Figure 2B). In contrast, the same highly diluted antibody in combination with the HRP substrate-Nanogold was able to generate a positive specific result. Black staining was observed over the insulin-secreting cells of the pancreatic islets, with no background over the exocrine parenchyma (Figure 2D). Furthermore, the same protocol at the electron microscopic level supported those results (Figures 3A and 3B). With the primary antibody diluted at 1:200, very intense labeling, revealed by the silver-enhanced gold particulate marker, was obtained over the dense core of the insulin secretory granules, with almost no diffusion of the reaction product into the halo of the granules (Figure 3A). Moreover, a similar positive labeling was also obtained with the anti-insulin diluted 1:10,000 (Figure 3B).
The specificity of the labelings obtained with both the simple and the amplified technique in light and electron microscopy was confirmed by the control experiments. Addition of the corresponding antigens to the antibodies or omission of the primary antibody abolished the labeling. Other control experiments, such as omission of the biotinylated secondary antibodies or omission of the streptavidin-HRP reagent, also led to absence of labeling (Figure 4A). However, protocols in which the HRP substrate-Nanogold reagent was omitted but otherwise completed with the streptavidin-HRP step and the silver intensification procedure gave a positive signal of low intensity (Figure 4B).
Determination of the sensitivity of the CARD-Nanogold amplification method was evaluated by an immunodot assay with known concentrations of protein. Silver deposits that revealed the proteins bound on the nitrocellulose membrane were still detectable at protein concentrations of 0.005 and 0.001 μg/ml. Because only 1 μl of these dilutions was spotted on the membrane, the positive reactions show that an amount of protein between 5 and 1 pg can be detected with this amplification technique (Figure 5). However, as with the control experiment involving omission of the HRP substrate-Nanogold reagent in immunoelectron microscopy (Figure 4B), the silver intensification procedure was able to reveal the HRP molecules on immunodots (not shown). Nevertheless, the sensivity of the reaction was much lower than with the amplification procedure.
Discussion
Back in the early 1990s the CARD technique was essentially based on the deposition of reporter molecules, such as biotin and FITC, through the reaction of HRP on its substrate tyramide. It has since been extended to various other reporter molecules (Plenat et al. 1997; van Gijlswijk et al. 1997; Hopman et al. 1998; Mayer and Bendayan 1999). Tyramine is a phenolic compound bearing a free hydroxyl group that reacts with HRP and a free amino group that can act as a linking end with reporter molecules. The reaction proceeds through intermediate free radicals. In the presence of H2O2, HRP catalyzes the oxidation of the phenol moiety of the tyramide that forms a highly reactive radical. The reporter-tagged activated tyramine binds rapidly and covalently to electron-rich amino acids of proteins in the immediate vicinity of the immobilized HRP (Gross and Sizer 1959; Bobrow et al. 1992). This system was first applied in electron microscopy, where application to postembedding immunocytochemistry led to the deposition of a dense filamentous reaction product over specific labeled structures (Mayer and Bendayan 1997). Subsequent improvements of the technique yielded amplified labeling revealed by colloidal gold particles (Mayer and Bendayan 1999). Up to now, no other hydrogen-donating substrates of HRP were used with the CARD approach. As a matter of fact, the technique can also be applied with other enzyme/substrate systems to enhance immunological signals. Bobrow et al. (1989, 1992) reported successful amplifications with other enzymatic systems, such as β-galactosidase-biotinylcoumarin-galactose. Catalytic deposition of the novel HRP substrate used in the present study presumably follows the same free radical mechanism as tyramide. Moreover, it has been coupled to Nanogold particles, easily visualized after silver enhancement, which eliminates additional detection steps and provides the technique with several advantages.
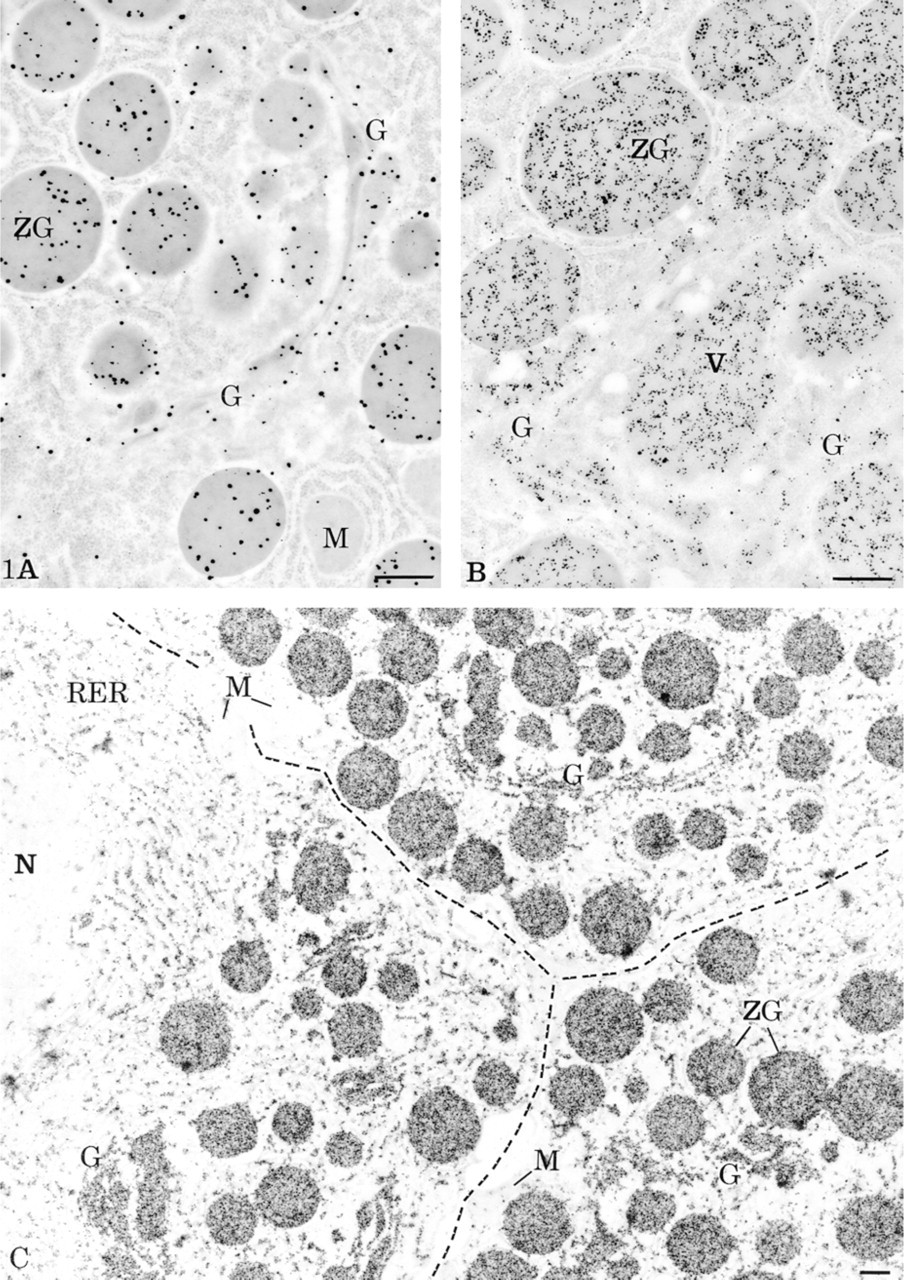
Localization of amylase in rat pancreatic acinar cells by electron microscopy. (
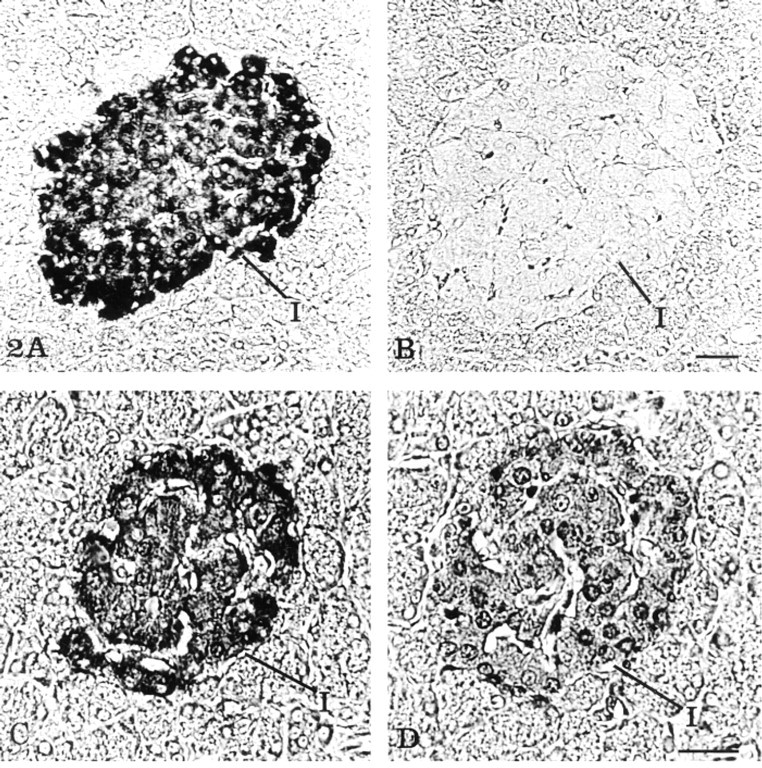
Localization of insulin on serial sections of rat pancreatic tissue at the light microscopic level. (
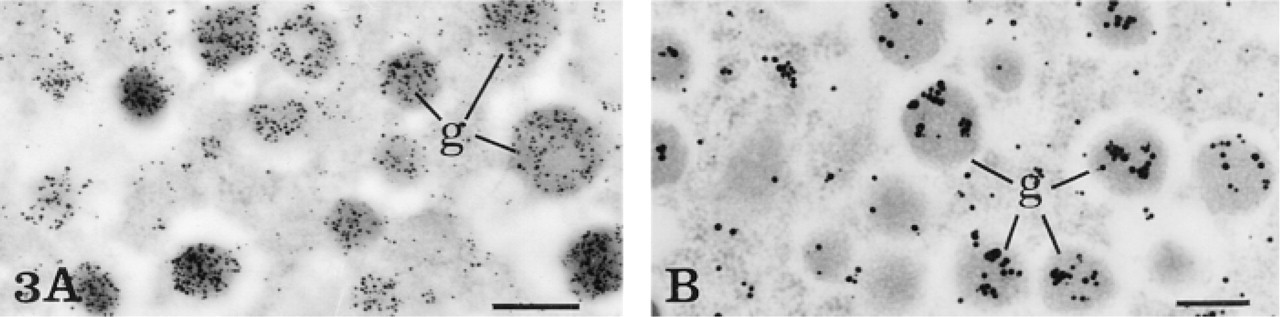
Localization of insulin in a rat pancreatic B-cell at the electron microscopic level. (
The present work demonstrates that the CARD-Nanogold system can be employed successfully as a highly sensitive, high-resolution immunoenzymatic technique for light and electron microscopy. The approach requires five successive incubation steps with (a) the specific primary antibody, (b) the corresponding biotinylated secondary antibody, (c) the streptavidin-HRP, (d) the HRP substrate-Nanogold complex, and finally (e) the silver enhancement. The results obtained with the amplification protocol at the electron microscopic level support the fact that accumulation of labeled substrate allows a single antigen-antibody complex to be revealed by multiple reporter molecules (Bobrow et al. 1989). The anti-amylase antibody was used at a dilution of 1:200 with both the simple and the amplified protocol. At this dilution, the antibody can bind only to a definite number of antigen molecules, according to its binding affinity. Compared to the simple labeling protocol, the CARD-Nanogold amplification system yields much higher labeling intensities, demonstrating greater efficiency in detection for an equal number of antigen-antibody complexes. By light microscopy, this increase in detection efficiency was reflected by shorter silver enhancement times providing signals as intense or even of higher intensity than those obtained with the simple approach. The enhanced staining intensity and the reduced time in silver development is most likely due to an increase in the number of Nanogold particles that facilitates conglomeration of silver deposits and therefore enhances staining by light microscopy (Lackie et al. 1985).
On the other hand, we have also demonstrated an increase in the detection limit of antigens with the use of the HRP substrate-Nanogold probe. For this purpose, a highly diluted antibody against insulin was used to simulate conditions in which antigen-antibody complexes are present in very small numbers. Incubation of pancreatic tissue with the anti-insulin antibody diluted 1:10,000, combined with the simple indirect protocol, resulted in an absence of signal. In contrast, the amplification protocol yield specific positive staining in both light and electron microscopy, making it possible to detect the signal generated by only few antigen-antibody complexes.
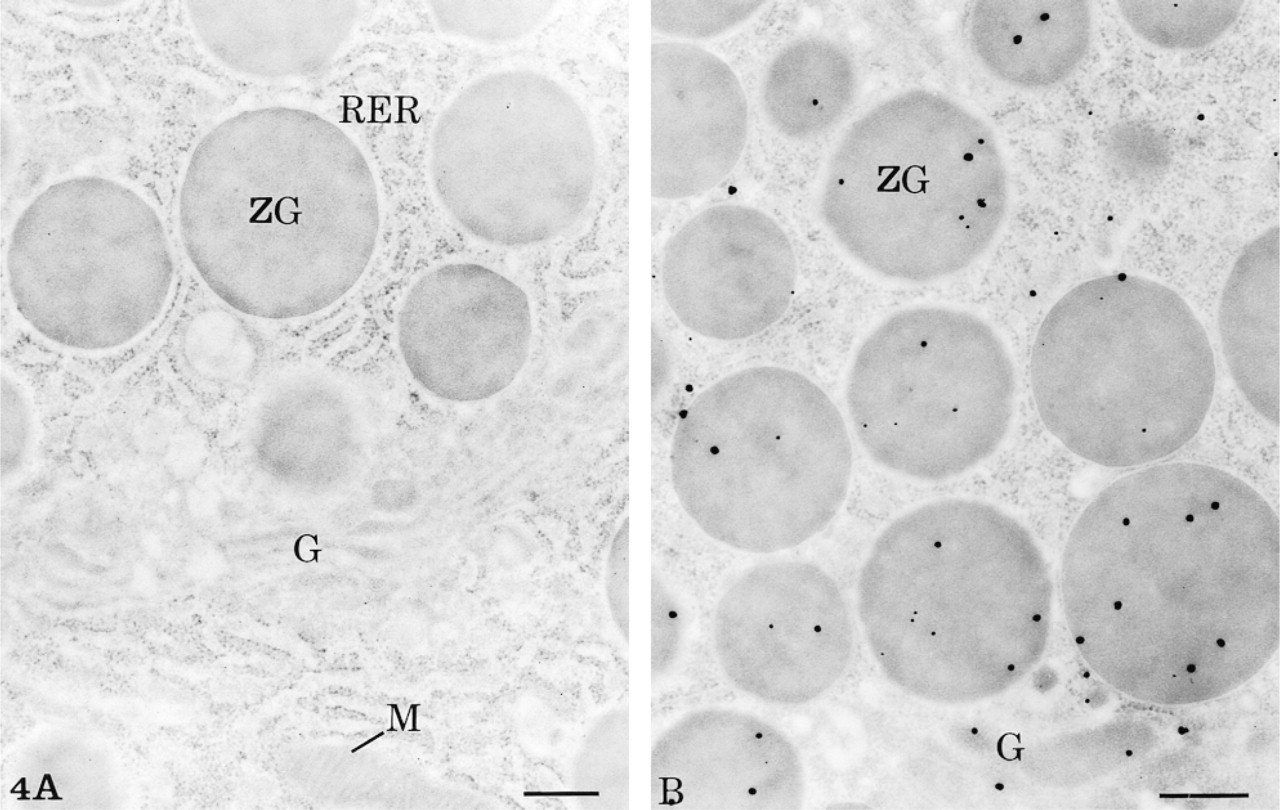
Control experiments. (
The amplification capabilities of the CARD approach combined with the small Nanogold probe, the silver enhancement, and a rather concentrated antibody (anti-amylase 1:10; Figure 1C) yield a very strong labeling over the pancreatic cells. Owing to this very intense signal, labeled cellular compartments could be detected at low magnification in electron microscopy without any counterstaining. This protocol, used with moderately concentrated antibodies, could be very useful for the screening of immunostained tissue sections or even for labeled subcellular structures. Observations were further facilitated by the very low background found over nuclei and mitochondria. In what concerns the specificity of the results, this was confirmed by the control experiments. However, staining of low intensity was observed with the control protocol involving the omission of the HRP substrate-Nanogold, either by immunoelectron microscopy or by immunodot. One explanation for this low signal, which indeed appeared specific, could be the silver intensification of the hemic iron atom present in the peroxidase molecule. In this case, the metallic atoms may have been transformed into iron sulfides or selenides because, with only few exceptions, endogenous or exogenous metals in tissues must be present under these forms to be able to react with the silver developer (Danscher 1981, 1982, 1983). An alternative would be the direct silver amplification of the streptavidin-HRP, without transformation, due to a favorable chemical or spatial condition of the iron in complex molecules. Either way, staining under this control condition was observed only after incubation with streptavidin-HRP, and the low signal appeared to be specifically localized at target antigenic sites. Moreover, addition of an unrelated Nanogold compound did not modify or increase the staining. As for immunoelectron microscopy, in immunodot the sensitivity of this labeling was found to be much lower than with the complete amplification protocol.
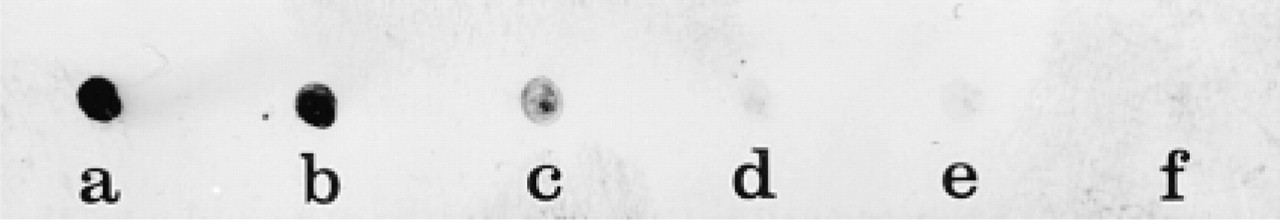
Immunodot assay. One μl of serial dilutions of rabbit IgGs starting at 10 μg/ml (10 ng of proteins) was spotted on to a nitrocellulose membrane. The HRP substrate-Nanogold amplification protocol generated a positive signal down to the lowest concentration of IgGs applied to the nitrocellulose strip (0.001 μg/ml or 1 pg of proteins). (
Sensitivity of the CARD-Nanogold amplification method was assessed with an immunodot assay with known concentrations of protein. Serial dilutions of normal rabbit IgG spotted on membrane down to 1 pg of protein were revealed. This result demonstrates a detection limit comparable to that of ELISA, enhanced luminol luminescence, and to the one-cycle amplification with biotinyl-tyramide and the two-cycle amplification with fluorescein-tyramide visualized with the streptavidin-HRP-DAB system used by Bobrow et al. (1991). Furthermore, our amplification method displays several advantages over other detection systems: the elimination of the carcinogenic DAB reagent, the rapid one-step silver intensification after deposition of Nanogold, the application to electron microscopy, and the detection by particulate markers offering good ultrastructural resolution.
The major drawback of the CARD-Nanogold technique relates to the silver enhancement step in its use for transmission electron microscopy. The homogenous 1.4-nm gold particles are difficult to visualize, and silver intensification is essential to increase their size. However, the silver enhancement process leads to a heterogeneity in the size of gold-silver particles, which hampers quantification of immunolabeling. The disparity in particle size may arise from the local gold label density that affects the percentage of enlarged particles and the final particle size. In addition, large particles might result from fusion of small ones, making quantification of immunolabeling density problematic and leading to underestimations (Stierhof et al. 1991; Takizawa and Robinson 1994; Humbel et al. 1995; Robinson et al. 1998). Therefore, comparison among different amplification protocols becomes difficult, and a silver enhancement technique that could yield more uniform enhanced particles is still needed for reliable quantitative evaluation and multiple labeling experiments.
The new HRP substrate-Nanogold probe, used as an amplification/detection step in our CARD protocol, leads to high-sensitivity immunolabeling. Moreover, it pushed back the detection limit afforded by the simple indirect Nanogold protocol. The combination of three amplification methods, i.e., the CARD protocol, the use of the small Nanogold probe, and the silver intensification technique, has provided improved, very sensitive, and intense immunolabeling by both light and electron microscopy.
Footnotes
Acknowledgements
Supported by grants from the Medical Research Council of Canada and the US National Institutes of Health. GM is recipient of a studentship award from the Medical Research Council of Canada.
We thank Dr Frederic R. Furuya of Nanoprobes for his aid in the preparation of the HRP substrate-Nanogold. We also thank Mr Jean Léveillé for photographic work.
