Abstract
In the unwounded cornea, tenascin-C localizes to a short stretch of the basement membrane zone at the corneoscleral junction or limbus. To determine whether the function of the limbus is affected by the absence of tenascin-C, mice possessing a deletion of tenascin-C and strain-matched wild-type mice are used in corneal debridement wounding experiments. The expression of integrins (α3, α9, and β4) in the tenascin-C knockout corneas is evaluated by producing polyclonal cytoplasmic domain antipeptide sera and performing immunofluorescence microscopy. In addition, we evaluate the localization of several other proteins involved in wound healing, including fibronectin, laminin β1, nidogen/ entactin, and VCAM-1, in both the tenascin knockout and wild-type mice. There are no differences in healing rate, scarring, or neovascularization after corneal debridement wounds. α9 integrin is expressed at the limbal border of unwounded tenascin-C knockout animals and is upregulated during migration only after the larger wounds. At 8 weeks after larger wounds, the localization of α9 again becomes restricted to the limbal border. Results show that tenascin-C is not required for development or maintenance of the corneal limbus or for normal re-epithelialization of corneal epithelial cells after debridement wounding.
T
Tenascin-C is a large modular glycoprotein consisting of a single fibrinogen-like domain and multiple EGF and FN-type III domains. The tenascin-C mRNA is subject to tissue-specific alternative splicing due to the inclusion or exclusion of a variable number of the exons encoding the FN-type III domains. As a result, multiple tenascin-C isoforms are generated, with distinct sizes ranging from 180 to 330 kD. In the corneas of humans and mice, the sizes of the tenascin isoforms produced range from 180 to 230 kD (Ljubimov et al. 1998; Matsuda et al. 1999). Tenascin-C molecules self-assemble. A single cysteine residue near the amino-terminus is required for the formation of oligomers consisting of six tenascin-C molecules within an assembly called a hexabrachion (Luczak et al. 1998). At the tenascin-C carboxy-terminus, the fibrinogen-like domains form a network by interacting with other matrix molecules, such as fibronectins and laminins (Erickson 1993; Schenk and Chiquet-Ehrismann 1994).
Cells can adhere directly to tenascin-C via binding to members of the integrin family of receptors including α2β1, α8β1, α9β1, αvβ3, and αvβ6 (Prieto et al. 1993; Sriramarao et al. 1993; Schnapp et al. 1995; Yokosaki et al. 1995; Denda et al. 1998). Integrins are a large superfamily of cell adhesion molecules that function as αβ heterodimers to interact extracellularly with a variety of molecules within the ECM and/or with other receptors on adjacent cell surfaces. Intracellularly, integrins bind to the internal cytoskeleton of the cell and regulate a variety of signal transduction events (Hynes 1996).
In addition to mediating attachment of cells to matrix, tenascin functions in anti-adhesion (Chiquet-Ehrismann 1995b). The exact mechanism of the anti-adhesive affect of tenascin-C is likely to vary with cell type because in one study, tenascin-C anti-adhesion is lost when the EGF domains are deleted (Dorries et al. 1996), whereas in another report, the fibrinogen domain was determined to be anti-adhesive (Fischer et al. 1997). It may also be that tenascin-C hexabrachions interfere with the ability of cells to bind directly to other more adhesive ligands. Because of its ability both to promote and to interfere with cell adhesion, the net effect of secretion of tenascin into the extracellular matrix on the properties of a cell within a tissue is a function of the amount and type of tenascin present in the matrix, the numbers and types of integrins present on the surrounding cells, and the identities of the other proteins making up the matrix.
Despite many studies documenting tenascin-C as a molecule whose expression correlates with organogenesis, wound healing, and cancer, tenascin-C knockout mice develop normally and have the same fecundity and physical appearance as wild-type mice (Saga et al. 1992; Forsberg et al. 1996). However, closer analyses of the brains of these mice have revealed altered behaviors, including hyperlocomotion, poor swimming, and reduced ability to adapt to new situations (Fukamauchi et al. 1996), altered neurotransmitter activity and release (Fukamauchi et al. 1997, 1998), and increased gliosis in response to cerebral injury (Steindler et al. 1995). In addition to these central nervous system defects, tenascin-C knockout mice also show symptoms of suppressed hematopoiesis in the bone marrow (Ohta et al. 1998) and an enhanced inflammatory response to chemically induced dermatitis (Koyama et al. 1998).
Although tenascin-C is not required for the development of a viable mouse, inactivation of tenascin-C clearly alters cellular behavior and tissue architecture in cells and tissues that normally express it. During studies of the developing mouse eye, we observed that α9 integrin expression and localization became increasingly restricted to a subset of epithelial cells in the basal cell layer of a region called the corneoscleral junction or limbus. Tenascin-C also localized to the basement membrane zone beneath the α9-positive cells and in the underlying stroma (Stepp et al. 1995). These data suggest that α9-tenascin-C interactions may be involved in mediating adhesion and/or proliferation of the α9-positive limbal epithelial basal cells. The limbus is important to the health of the cornea because it is the location at which the corneal epithelial stem cells reside (Schermer et al. 1986; Cotsarelis et al. 1989; Lavker et al. 1991). Loss of these stem cells due to trauma or disease results in the ingrowth of conjunctival cells and blood vessels and destroys vision (Huang and Tseng 1991). During epithelial cell differentiation in the cornea and especially after corneal injury, the corneal epithelial stem cells in the limbus proliferate and give rise to transit-amplifying (TA) cells. The TA cells go on to proliferate and migrate away from the limbus towards the center of the cornea (Lavker et al. 1991; Lehrer et al. 1998). Protein surface markers exclusive for the limbal stem cell surface have yet to be identified. Our experiments have shown that the overall amount of α9 expressed and the numbers of cells that are α9-positive increases as the limbal basal cell population is forced to proliferate and TA cells migrate towards the center of the cornea after an epithelial injury (Stepp and Zhu 1997). α9 integrin therefore is expressed not only on the stem cells themselves but also on the TA cells. Tenascin-C localization is retained at the basement membrane zone beneath the epithelial cells at the limbal border during re-epithelialization. In addition, an increase in the distribution of tenascin-C occurs within the corneal stroma at later times after epithelial cell migration is complete (Stepp and Zhu 1997).
Although it has been shown that the corneal epithelial stem cells are restricted to the basal cell compartment at the limbus, it is not at all clear what keeps them there. It has been postulated that basement membrane specializations exist at the border between the limbus and peripheral cornea that function as a barrier which the stem cells do not normally cross (Gipson 1989; Kolega et al. 1989). Injuries that destroy the limbus, such as chemical wounds or burns, result in localized loss of stem cells. The only way to save vision in such patients is to transplant stem cells from a limbus obtained either from a healthy contralateral eye or from a donor eye (Kenyon and Tseng 1989; Tseng and Tsai 1991; Tsubota et al. 1999). A detailed study of the basement membrane zone beneath the limbus showed that there were differences in the molecular structures of the Type IV collagen and laminins at the conjunctiva-limbal and limbal-peripheral corneal borders (Ljubimov et al. 1995). Moreover, tenascin-C expression at the corneal basement membrane zone in human (Tervo et al. 1990; Maseruka et al. 1997) and mouse (Stepp and Zhu 1997) has been shown to be tightly restricted to a short stretch at the border between the limbus and the peripheral cornea.
To determine whether α9-tenascin-C interactions function to maintain the corneal-scleral junction, we used the tenascin knockout mouse, generated by Saga et al. (1992), in corneal epithelial debridement wound healing studies. After injury, we assessed the quality of wound closure (time to closure, scarring) as well as the distribution of epithelial integrins, entactin, and laminins in control and healing corneal epithelium in the tenascin-C knockout mouse. Finally, we evaluated the localization of additional α9 integrin ligands besides tenascin-C to determine whether there is evidence of compensation for the lack of tenascin by changes in expression of osteopontin or VCAM-1.
Materials and Methods
Animals
Tenascin-C-deficient mice were generated by Saga et al. (1992) and donated to us by M. Kusakabe (Riken, Japan) and H. Erickson (Duke University, Raleigh, NC). Tenascin-C KO mice were maintained on a GRS/A genetic background and wild-type GRS/A and Balb/C mice were used as controls. The original characterization of these mice has been described by Saga et al. (1992).
Wound Model
All experiments on mice were conducted in compliance with the recommendations of the Association for Research in Vision and Ophthalmology and with the GWU IACUC. Manual debridement wounds were created on the corneas of 8-week-old tenascin-C knockout mice and their wild-type counterparts as described previously (Stepp and Zhu 1997). Mice were placed under general anesthesia and eyes numbed with a topical anesthetic. Corneas were then scraped with a dull scalpel to remove the epithelium within a 1.5-mm central corneal area that had been demarcated with a dull trephine (small wound) or from a 3.0-mm area comprising the entire corneal surface, excluding a 0.5 mm area adjacent to the limbus (large wound). Animals were sacrificed by lethal overdose at times ranging from 12 to 48 hr for the small wounds and 1 day to 2 months for the large wounds. To determine the time of wound closure, corneas were stained with a vital dye and evaluated under the dissecting microscope. Scarring and neovascularization were assessed under the dissecting microscope. Enucleated eyes were frozen in Tissue Tek II OCT compound (Lab Tek; Naperville, IL) for immunohistochemistry.
Immunohistochemical Analysis
The frozen corneas were sectioned (10 μm) and stained with the primary antibodies to the basement markers laminin β1, entactin, and perlecan (A. Ljubimov; Cedars-Sinai Medical Center, Los Angeles, CA) as well as J-18 (laminin 5; J. Jones, Northwestern University Medical School, Chicago, IL). Corneas were also stained using antibodies to tenascin (Mtn-12; T-3413, Sigma, St Louis, MO), VCAM-1 (429:MVCAM.A, #01810D, Pharmingen, San Diego, CA), osteopontin (C. Giachelli; U. of Washington, Seattle, WA), and fibronectin (R.O. Hynes; Mass. Inst. of Tech./HHMI, Cambridge, MA) and with antibodies to the integrins α3, α9, and β4. The polyclonal integrin sera were generated using standard procedures (Covance Laboratories; Vienna, VA) from the human amino acid sequences encoded by the C-terminal cytoplasmic domains of the three integrins. The α3 peptide was EMK SQP SET ERL TDD Y, the α9 peptide was RYK EII EAE KNR KEN EDS WDW VQK, and the β4 peptide was KKK TTS GSL STH MDQ QFF QT. For α3 and β4, the generation of these sera involved injection of two rabbits each with a KLH-conjugated peptide. The longer peptide comprising the complete cytoplasmic domain of α9 was used for generation of α9 sera after four rabbits injected with a shorter peptide failed to generate sera useful for immunofluorescence. Of four rabbits injected with the longer peptide, only one generated a serum that recognizes α9 on human or mouse tissues. Biochemical and immunohistochemical characterization of these sera are described in Results. For double labeling, a rat anti-mouse β4 monoclonal antibody (346-11A; #09491D; Pharmingen) was used along with the rabbit α9 polyclonal sera. The images were viewed and captured using an Olympus BX60 or with confocal microscopy using the BioRad MRC 1000 program. All images were imported into Adobe Photoshop 4.0.
Immunoblot Analyses
Immunoblotting was carried out as described previously (Stepp et al. 1996). To characterize the α3 and α9 sera, mouse liver was used as a source of protein extracts. For β4, mouse corneal epithelium was used. Tissue extracts were run unreduced on 7.5% SDS-PAGE gels and proteins transferred to the nitrocellulose membranes. Membranes were cut into several strips after blocking with blocking buffer consisting of 5% milk in Tris-buffered saline containing 0.1% Tween-20. Strips were incubated separately with dilutions of each of the three integrin sera. Peptide inhibition was performed by preincubating diluted sera with 50μg/ml peptide for 30 min to 1 hr before adding to the nitrocellose strips. Specificity of peptide inhibition was determined by preincubating sera with two peptides: (a) the peptide used to generate the serum, and (b) one of the integrin peptides from a different integrin α- or β-chain. HRP-conjugated donkey anti-rabbit IgG was used as secondary antibody and proteins were detected by ECL Western Blot Detection reagent (Amersham; Arlington Heights, IL). BioMax film (Kodak; Fisher, Rochester, NY) was used to obtain images, which were then scanned and processed using Adobe Photoshop.
Tenascin RT-PCR
To confirm the absence of tenascin-C mRNA in tissues obtained from tenascin-C KO mice, total RNA was isolated from liver obtained from either tenascin-C KO or wild-type GRS/A mice. Mice were sacrificed by lethal injection, liver tissues rapidly frozen and powdered in liquid nitrogen, and total RNA extracted using RNAzol according to the manufacturer's instructions (Tel-Test; Friendswood, TX). Total RNA per well of 25 μg was used for RT-PCR studies with tenascin primers. Primers for mouse G3PDH (Life Technologies/Gibco; Gaithersburg, MD) and β-actin (Clontech; Palo Alto, CA) served as controls. The primers for tenascin were made against the region flanking the translation start site of the mouse tenascin gene to direct the amplification of a tenascin-C rtDNA product of 834 bp (Forsberg et al. 1996). Their sequences are as follows: 3′ CAG GTA CTT CTT CAC GGA GC and 5′ GCA GTC TTC CCC AGT GAA AC. RT-PCR was performed using a Perkin Elmer Thermal Cycler and the SuperScript One-Step RT-PCR System (Life Technologies/Gibco) for 40 cycles at 60C. Products were separated on an ethidium bromide-impregnated 2% agarose gel (E-gel; Invitrogen, Carlsbad, CA), sized by comparison to a 100-bp DNA ladder (Life Technologies/Gibco) and photographed under UV light.
Results
Localization of Tenascin-C in the Unwounded Cornea Is Restricted to the Periphery
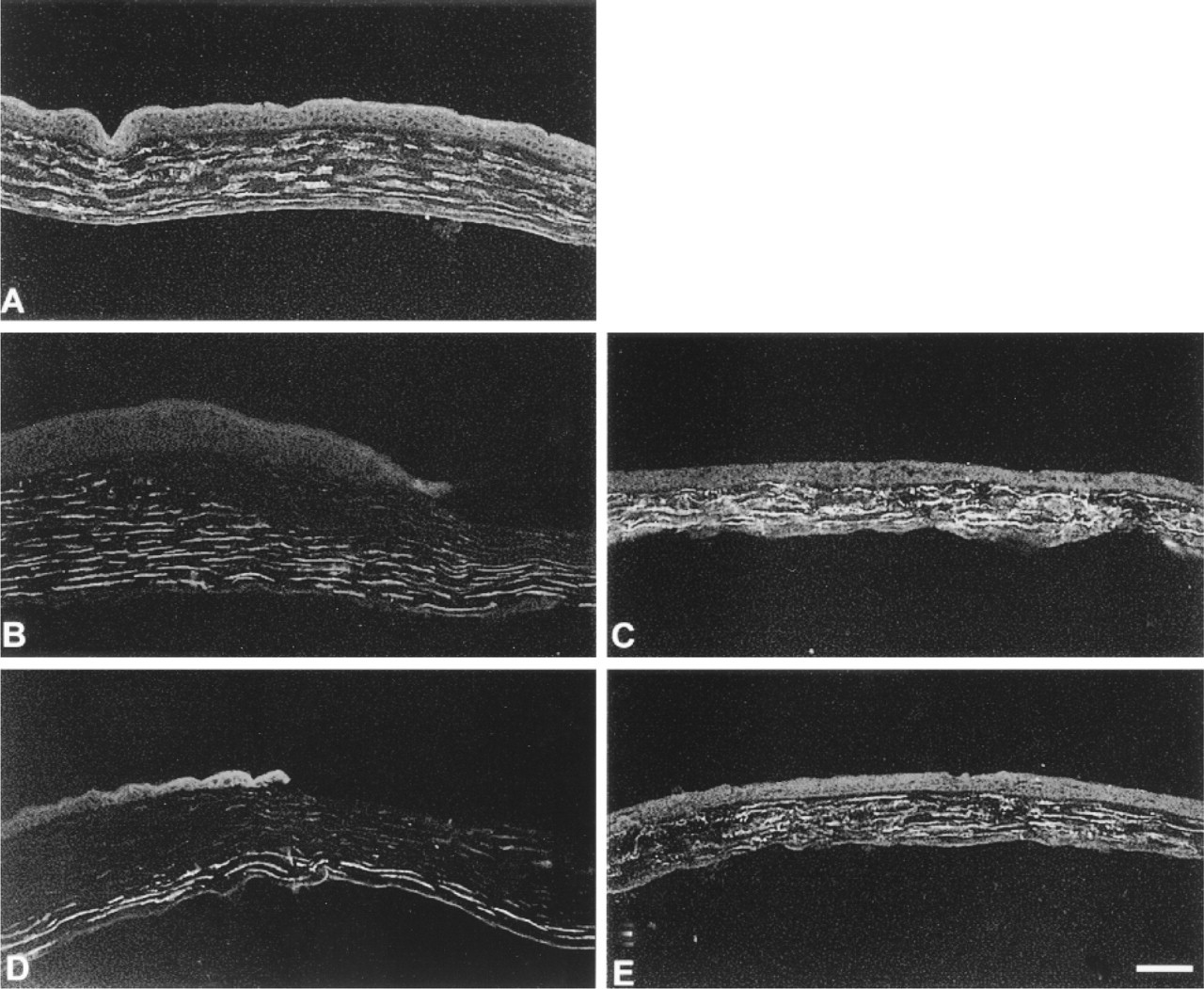
A rat monoclonal antibody recognizing mouse tenascin-C was used on the pigmented inbred GRS/A wild-type mice and the GRS/A tenascin-C KO mice. As expected, tenascin-C localization was high at the limbal region (Figure 1A) and was absent in the central cornea (Figure 1B) in mice containing the intact tenascin-C gene. No tenascin-C was observed in the KO mouse cornea in the limbal area (Figure 1C) or central cornea (Figure 1D). To further confirm that tenascin-C was not expressed in the tissues of the knockout mice, RT-PCR was performed using tenascin-C primers derived from the sequence of the mouse tenascin-C gene flanking the translational start site (Figure 1E). RNAs isolated from the tenascin-C KO and wild-type mouse liver are capable of amplifying control primers for G3PDH and β-actin, but only the wild-type liver RNA amplifies the tenascin-C primers. These data show that neither the tenascin-C protein nor its mRNA is detected in these tenascin-C KO mice.
Polyclonal Sera Against α3, α9, and β4 Were Generated and Characterized for Use in Immunofluorescence Studies
Peptide antibody reagents were generated and characterized first by immunofluorescence and their specificity confirmed by immunoblotting. Data on the characterization of these sera by blotting are shown in Figure 2. For α3, α9, and β4, a single band of the appropriate kD was observed for each serum (Figure 2, Lanes 2, 5, 8, respectively) which was not present when sera were preincubated with the peptide used for their production (Figure 2, Lanes 4, 7, and 10, respectively). Peptide blocking was specific because addition of the α9 peptide to either the α3 or β4 serum did not interfere with its ability to interact with α3 or β4 (Figure 2, Lanes 3 and 9), nor did the addition of the α3 peptide interfere with the α9 binding (Figure 2, Lane 6). Using immunofluorecence microscopy, all three of these newly prepared sera were tested against other sera known to be specific against each of these integrins. Identical resuits were obtained in the corneas of mice, rats, and humans. Although data from rats and humans are not presented, mouse data are described below.
Localization of Several Epithelial Integrins and Basement Membrane Markers Is Not Altered in Corneas of Tenascin-C KO Animals
To determine whether the differentiation of the epithelial cells of the cornea is altered by the disruption of tenascin-C gene expression, we localized α3, β4, and α9 integrins in the unwounded tenascin-C KO cornea using immunofluorescence (Figure 3). Compare to the wild-type GRS/A animals, the localization of α3 and β4 in the tenascin-C KO animals was identical, and therefore data from only the tenascin-C KO tissues are shown. The localization of α3 was restricted primarily to the basal and basolateral membranes of the basal cells (Figure 3A). β4 was more abundant at the basal cell basal membrane (Figure 3B), where it is known to be present in hemidesmosomes. However, β4 was also present at the lateral membranes between the basal cells. Like α3 and β4, the localization of α9 was similar in the tenascin-C KO and wild-type animals. α9 was localized exclusively to the epithelial cells at the corneoscleral junction between the limbus and the peripheral cornea (Figure 3C) and was not detected in the central cornea (Figure 3D). Therefore, tenascin-C expression at the corneoscleral junction is not a prerequisite for the localization of α9 integrin to this site. To further evaluate the tenascin-C KO cornea, the localization of several basement membrane proteins was evaluated, including perlecan, laminin β1, laminin γ1, entactin/nidogen, and laminin-5. Shown in Figures 3E and 3F are the results for entactin/nidogen and laminin β1, respectively. The basement membrane zone is linear and continuous and there is no evidence of any abnormalities in the tenascin-C KO animals. Taken together, the localization of integrins and basement membrane markers indicates that the tenascin-C KO cornea is similar to other wild-type control, unwounded corneas.
Tenascin-C is a component of the anatomic border formed at the corneoscleral junction in the normal cornea and has been postulated to play a structural role (Stepp and Zhu 1997). The restriction of α9-positive cells to the limbal border in the unwounded tenascin-C KO cornea suggests that the anatomic border normally formed by tenascin-C is not a requirement for the formation of a normal limbal junction. Given the ability of tenascin-C to mediate either adhesion or anti-adhesion, depending on the cell type and nature of the substrate (Chiquet-Ehrismann 1995b), subtle differences in the molecular structure of this border might well affect either the retention of stem cells at this site or the ability of the α9-positive, early transit-amplified cells to migrate past this border after being forced to proliferate in response to wounding.
Despite the Lack of Tenascin-C, α9-positive Cells Are retained at the Limbal Border After Epithelial Cell Migration Has Been Induced by Debridement Wounding
Both smaller wounds, which close within 24 hr without a significant increase in cell proliferation, and larger wounds, which take 2-3 days to re-epithelialize and require cell proliferation during migration, were made on the corneal surface of the tenascin-C KO mice and tissues were evaluated for the rate of wound closure, for signs of neovascularization and scarring, and for the localization of integrins. These types of wounds remove only the epithelial cells and leave the basement membrane intact and native (Stepp et al. 1996; Stepp and Zhu 1997).
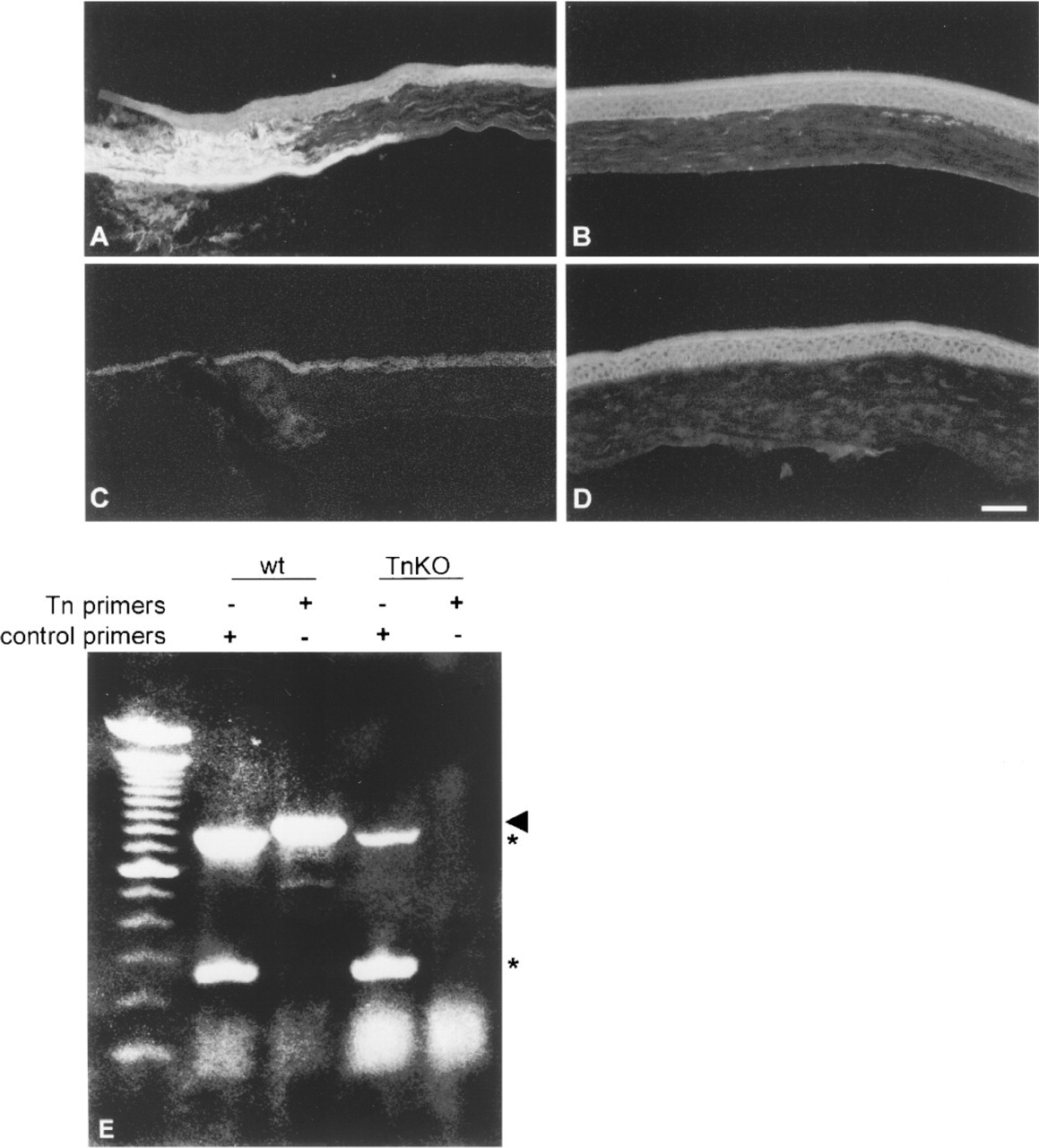
Tenascin-C expression and localization in the unwounded corneas of wild-type and tenascin-C KO mice. (
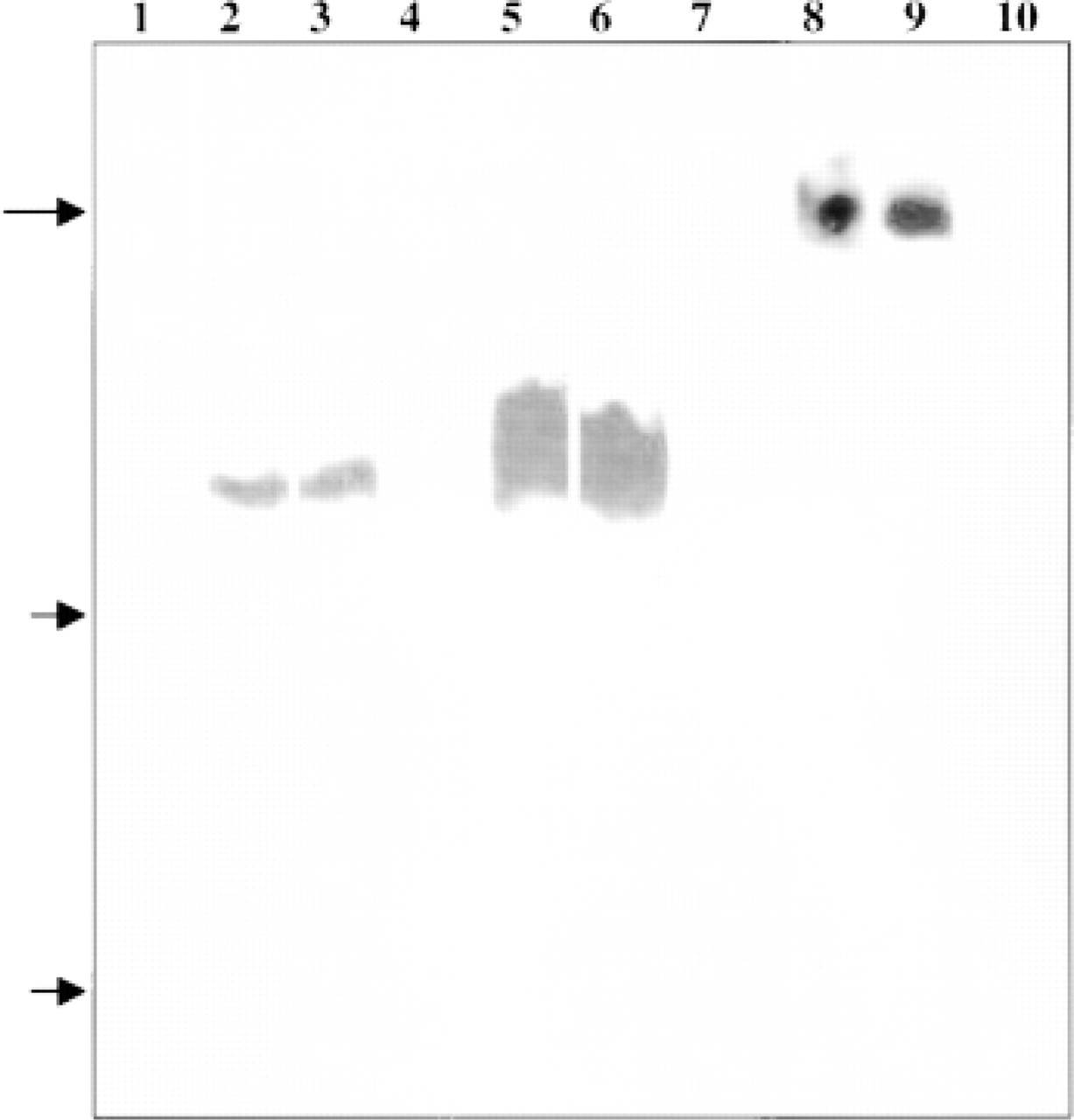
Integrin antibody characterization by immunoblotting. In Lanes 1-7, mouse liver tissue was extracted, size-separated on 7.5% SDS-PAGE, and the gel blotted onto nitrocellulose. In Lanes 8-10, corneal epithelial extracts were used rather than liver tissues. Lane 1 shows a strip incubated with a preimmune sera. Lanes 2-4 show the results obtained with the affinty-purified α3 serum (Lane 2), the α3 serum after preincubation with the α9 peptide (Lane 3), and α3 serum after preincubation with the α3 peptide (Lane 4). Lanes 5-7 show the results obtained with the affinty-purified α9 serum (Lane 5), α9 serum after preincubation with the α3 peptide (Lane 6), and α9 serum after preincubation with the α9 peptide (Lane 7). Lanes 8-10 show the results obtained on corneal tissue extracts incubated with the β4 serum (Lane 8), β4 serum after preincubation with the α9 peptide (Lane 9), and β4 serum after preincubation with the (4 peptide (Lane 10). Arrows indicate the mobilities of molecular weight markers of kD of 207, 120, and 78.
The rate of wound closure was determined after the removal of a 1.5-mm area of epithelial cells from the corneal surface of a total of 42 tenascin-C KO and 30 GRS/A wt mice at 12, 18, 24, and 48 hr. For large wounds, a total of 43 tenascin-C KO and 30 GRS/A wt were evaluated at 1, 2, 3, 6, and 12 days after initial injury. We observed no difference in the length of time to wound closure between KO and wild-type strains. In addition, there was no increase in scarring or neovascularization within the first 6 weeks post wounding. Although there were no differences between the wild-type and KO GRS/A mice, large wounds were generally observed to take 3 days to close in the GRS/A strain of mice rather than 2 days, which is standard for 8-week-old Balb/c mice. This difference indicates that variation in healing rates exists among different mouse strains.
Immunofluorescence studies performed on tissues at various times after wounding indicated that both the tenascin-C KO and GRS/A wt mouse corneas showed similar changes in integrin localization during healing. The data presented in Figures 4 and 5 are from tenascin-C KO animals. After smaller wounds, the localization of α9 to the basal cells at the limbus was retained at 12 hr (Figure 4A) and 18 hr (Figure 4C) and was absent at the leading edge at both time points (Figures 4B and 4D). α3 localized to the basolateral membranes and at the basal surface of the basal cells at the leading edge at both 12 and 18 hr after injury (Figure 4E and 4F, respectively). β4 appeared to increase in abundance in the cytoplasm and on the membranes of cells at the leading edge and back towards the periphery, and the relative abundance of β4 appeared higher at 18 hr (Figure 4H) compared to 12 hr (Figure 4G). After small debridement wounds in the rat, we have shown, using a biochemical approach, that β4 expression increases during migration and decreases as soon as migration is complete (Stepp et al. 1996). Thus, after the small wound, the expected changes in integrin expression and localization occur in the tenascin-C KO mouse.
Thus far, the tenascin-C KO mouse cornea has responded similarly to wild-type corneas to debridement wounding. Unlike the small wound, in which a wedge of cells makes up the leading edge, in the large wound a single-cell layer sheet of cells makes up the leading edge, with all of its cells derived from proliferation of transit-amplifying cells from the limbus. Past studies have shown that α9 is expressed later during migration after these larger wounds and during restratification (Stepp and Zhu 1997). However, it has not been shown how soon α9 is upregulated after the large wounds or whether there are also changes in β4 expression and localization after the larger wounds. In Figure 5, we used confocal microscopy to evaluate simultaneously the localization of α9 and β4 integrins after large wounds in dual-labeling experiments conducted on tissues from tenascin-C KO mice; similar results were obtained from wild-type mice. In Figure 5A, at 1 day after wounding, a gradient of β4 is observed, with β4 becoming more abundant away from the leading edge. In Figure 5A, note also that at the tip of the leading edge (arrowhead), a portion of the epithelial sheet made up of three or four cells has loosened and has come off the stroma. This is a common occurrence after larger wounds, indicating that the sheet is loosely adherent and more so at the leading edge. β4 appears to be localized primarily to the basal surface of the migrating sheet of cells. α9 localization at 1 day after large wounds also increases towards the periphery, with the three or four cells at the very tip of the leading edge expressing little if any α9. The normal restriction of α9 within the limbus is lost at 24 hr after larger wounds; earlier times were not evaluated.
By 2 days, both α9 and β4 were expressed abundantly, as shown in Figure 5B in a typical section taken from a region behind the leading edge during migration. Although Figures 5A and 5B show a single layer of migrating epithelial cells expressing both α9 and β4, the staining for β4 remains most abundant at the basal surface, where the cells are in contact with the underlying matrix. α9 is most abundant at the basolateral cell surfaces and within the cytoplasm. Comparing the localization of α9 and β4 during migration after the large wound (Figure 5) with their localization after the smaller wounds (Figure 4A-4D) reveals important differences. β4 appears to increase in amount at longer times after both types of injury and to be abundant at both basal and nonbasal cell membranes in response to both small and large wounds. α9 localization shows more dramatic differences; only after the larger wounds is α9 present on actively migrating corneal epithelial cells in the central cornea. By 8 weeks after injury in the TN KO mouse, migration and restratification are complete and the expression of α9 in the central cornea is again restricted to the cells within the limbus; there is little if any α9 in the central cornea (Figure 5C). The pattern shown is identical to that observed in control mice. To the left of the arrow in Figure 5C, the α9-positive conjunctival and limbal basal cells are seen to express a low level of β4 at their basal surfaces. To the right of the arrow towards the center of the cornea, cells possess less α9 and more β4. The inset shows the transition zone between limbus and peripheral cornea at a higher magnification. Note that cells at this site display dramatic differences in α9 and β4 localization and expression. Taken together, these data show that the ability of the epithelial cells of the cornea to heal in response to manual debridement wounds is not affected by the lack of tenascin-C.
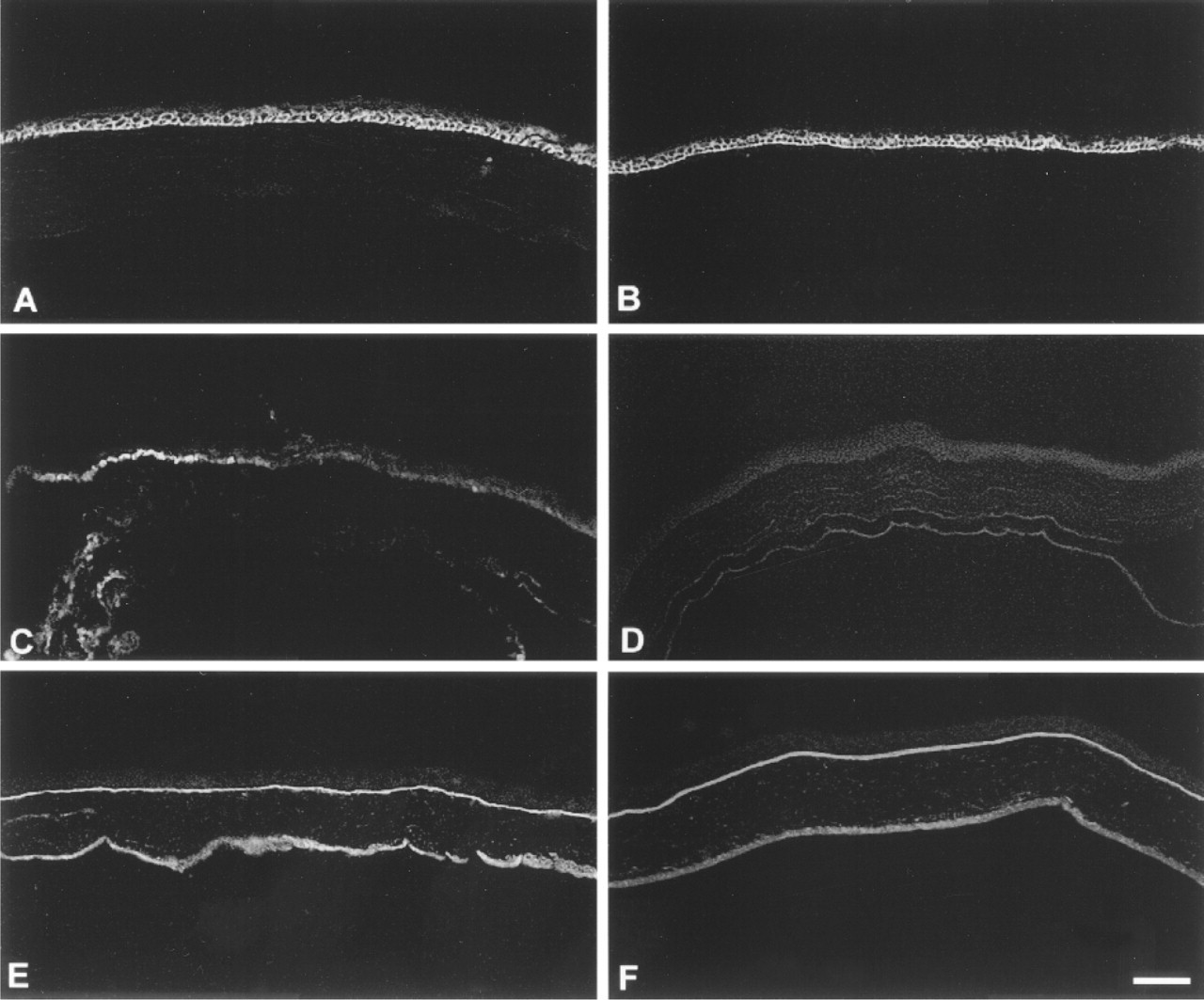
Integrin and ECM protein expression in unwounded tenascin-C KO mouse.
VCAM-1 Expression in Healing Corneas Is also Comparable Between the Wild-type and the Tenascin-C KO Mice
Recent data show that, in the vasculature, α9β1 integrin on leukocytes can function as a receptor for VCAM-1 on activated endothelia (Taooka et al. 1999). To evaluate whether there are any locations at which α9 and VCAM-1 might be co-distributed in either normal or wounded wild-type and tenascin-C KO corneas, we evaluated these tissues for expression of VCAM-1 and present the data from tenascin-C KO animals in Figure 6. Similar data are obtained from wt animals. VCAM-1 was not expressed by the epithelial cells in the cornea (Figure 6A). However, VCAM-1 did appear to be expressed on the normal, unwounded fibroblasts in the stroma. At 18 hr after small wounds, the staining for VCAM-1 disappeared at the anterior surface of the central cornea (Figure 6B). By 48 hr, VCAM-1 was again present in this region (Figure 6C). At 2 days after the larger wounds, a marked loss of VCAM-1 staining in the stroma was observed over the entire corneal surface, with VCAM-1-positive fibroblasts visible only along the posterior aspect of the stroma (Figure 6D). By 6 days, a normal-appearing VCAM-1 staining profile was observed. In addition to tenascin-C and VCAM-1, another ligand for α9 has been demonstrated. A proteolytically cleaved fragment of osteopontin has been shown to mediate α9β1 adhesion and migration (Smith and Giachelli 1998). We performed immunofluorescence microscopy using sera against osteopontin on tenascin-C KO corneas as well as on the wild-type GRS/A mice and found no evidence for expression of osteopontin in the cornea before or after debridement wounding in KO or wild-type mice (not shown). Therefore, in the tenascin-C KO mouse cornea, there is no compensation for the lack of one α9 ligand, tenascin-C, by the upregulation of the other known ligands, VCAM-1 and osteopontin.
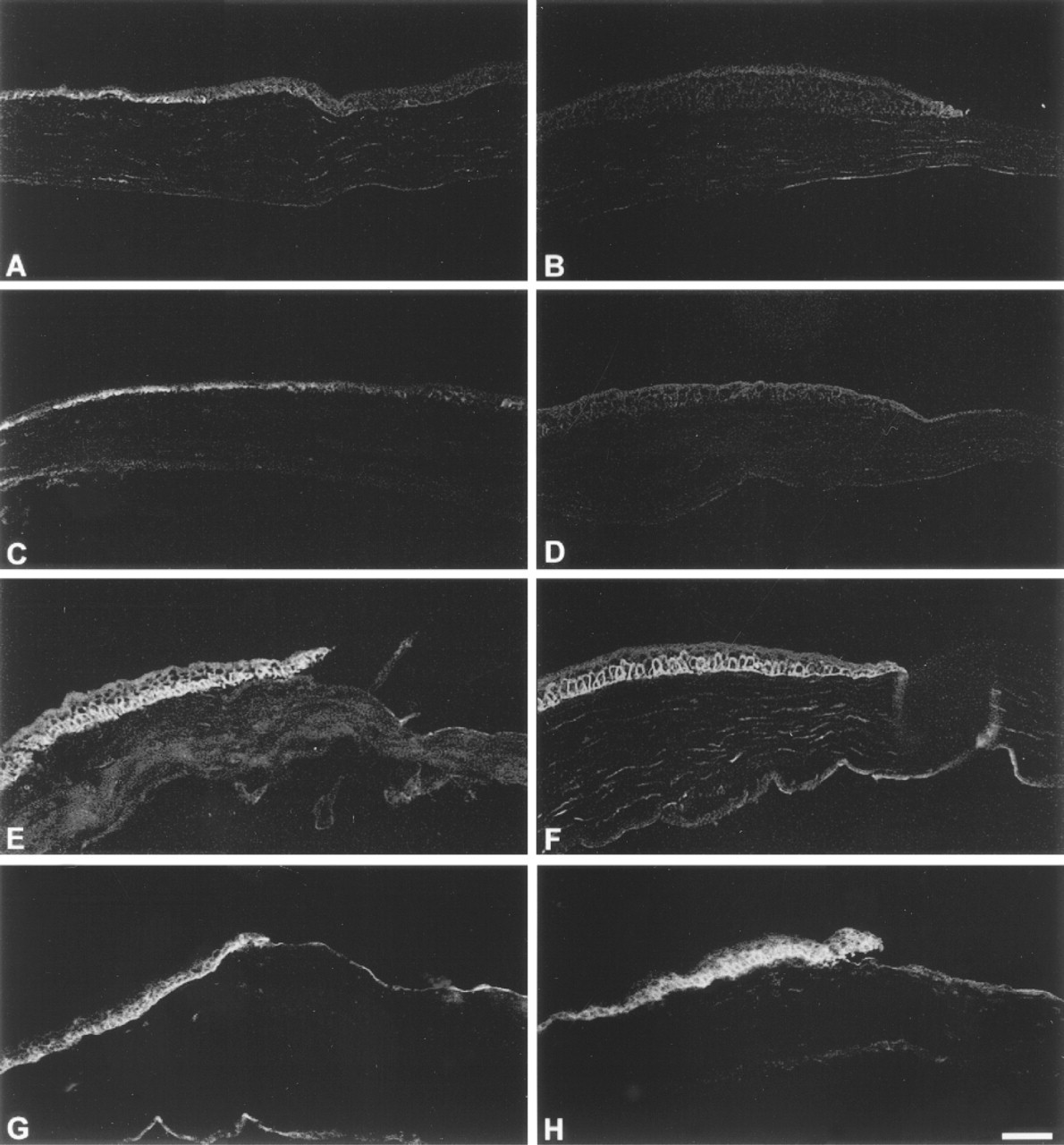
Integrin expression in tenascin-C KO mouse after small wound. (
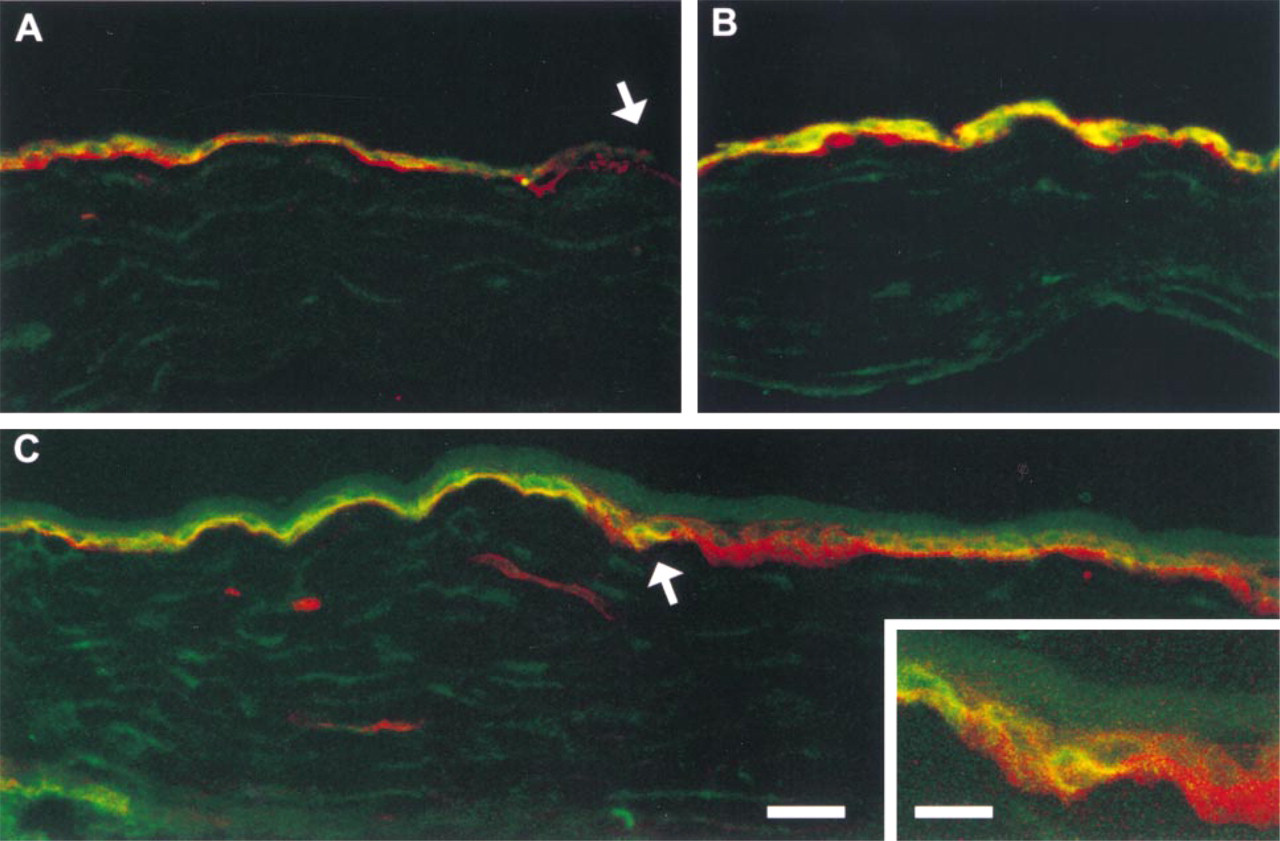
Integrin expression in tenascin-C KO mouse after large wound. (
Discussion
Re-epithelialization Is Not Delayed in the Absence of Tenascin Several years of studies had shown that tenascin-C is upregulated in response to epidermal wounding (Betz et al. 1993; Schenk et al. 1995; Aukhil et al. 1996) and corneal (Tervo et al. 1991; van Setten et al. 1992; Maseruka et al. 1997; Stepp and Zhu 1997; Tuori et al. 1997). Forsberg et al. (1996) performed full-thickness skin punch injuries on tenascin-C-deficient animals and found no differences in healing rate, epithelial cell proliferation, or wound contraction. These authors did observe a decrease in the accumulation of fibronectin within the wound bed in the KO animals compared to the controls at both early and late times after injury. Matsuda et al. (1999) recently demonstrated, using a linear incisional wound model of corneal injury, that there were no differences in healing rate or scarring unless the KO mouse corneas were sutured closed at the time of injury. In normal animals, these authors observed that using a suture induces a more robust increase in tenascin-C expression. Interestingly, Matsuda et al. (1999) also demonstrated a reduction in the level of fibronectin in the corneal stroma of the tenascin-C KO sutured corneas compared to control corneas. The reduced accumulation of fibronectin in the wounds of the tenascin-C KO animals observed by Forsberg et al. (1996) and Matsuda et al. (1999) suggests that tenascin-C-fibronectin interactions may function in part to regulate the accumulation of fibronectin after wounding. It is also an intriguing possibility that tenascin-C KO dermal and stromal fibroblasts downregulate fibronectin expression to compensate for the absence of the anti-adhesive properties of tenascin-C in the wound bed.
VCAM-1 expression in unwounded and healing corneas of tenascin-C KO mice. (
The larger-sized debridement wounds we perform induce tenascin-C expression both in the stroma and by the epithelial cells. This increase in tenascin-C occurs immediately after closure of the wound at 3 to 6 days (Stepp and Zhu 1997). However, we do not observe any increase in scarring of the corneas of the tenascin-C KO mice in response to the larger wounds. Fibronectin was also evaluated and no evidence for its accumulation in either the wild-type or knockout tissues was obtained (not shown). Although our studies do not directly assess cell proliferation in the limbal region, the fact that the timing of closure is not delayed after the large wounds that require cell proliferation during migration, coupled with the healthy corneal appearance at times up to 6 weeks post-wounding, suggests that the limbal region in the tenascin KO mice is fully functional. Taken together, our data and those of Forsberg et al. (1996) and Matsuda et al. (1999) show that tenascin-C KO mice can heal skin and corneal wounds normally. However, Matsuda et al. (1999) showed that specific types of corneal injuries induced increases in scarring. The use of tenascin-C KO mouse corneas in appropriate wounding models may allow the development of treatments to minimize or eliminate corneal scaring after suturing. Given that epidermal healing and corneal wound healing have many similarities, new treatments developed to minimize scarring in the cornea are likely to function similarly in skin.
α9 and β4 Integrin Expression Is Increased During Wound Healing in Normal and Tenascin-C KO Mice
We are unable to document any differences in integrin expression in the corneas of the tenascin-C KO mouse. Analysis of α9 localization in control and wounded corneas revealed no differences between control and tenascin-C KO animals. The localization of α9 varies in the KO animals after wounding in precisely the same way as in Balb/c and GRS/A mice. The same is true for α3 and β4 integrins. There are no changes in α3 and the increased expression of β4 occurs during migration after the small wound as it does in normal animals. For the larger wounds, we show that α9 is upregulated at 1 day after injury and continues to increase at Day 2 in both the tenascin-C KO and wild-type animals.
The absence of one member of a receptor-ligand pair has been shown to disrupt the localization of the other member of that pair. In the α8 integrin KO mouse, various ligands of α8β1 integrin, including fibronectin, vitronectin, and tenascin, are reduced in expression and localization during development (Muller et al. 1997; Lipschutz 1998). α3 KO mice have disrupted localization of laminin basement membrane components (DiPersio et al. 1997). However, there is no evidence for any alteration in the localization of α9 in the absence of tenascin-C. Other α9 ligands (VCAM-1 and osteopontin) do not compensate for the lack of tenascin-C in the tenascin-C KO mouse.
We see no evidence of localization or expression of either VCAM-1 or osteopontin in the cells of the corneal epithelium of the tenascin-C KO or the wild-type animals, despite the abundant amount of α9 localized to these tissues in response to the large wound. Dynamic upregulation of expression and localization for α9 integrin after injury in both normal and tenascin-C KO animals is unlikely to occur in the absence of function. Our data suggest that the exact ligand or counter-receptor for epithelial α9 integrin remains to be determined. Given that after the large wound, the tenascin-C KO mouse corneal epithelial cells express none of the known ligands for α9, they would provide an excellent system to use to discover the nature of any additional α9 integrin ligands-counter-receptors.
Tenascin-C Is Not Required for Maintenance of Corneoscleral Junctions After Wounding
The importance of the corneoscleral junction in the maintenance of corneal health cannot be overestimated (Tsubota et al. 1999). Identification of the molecules critical for maintaining its structure and function will aid in developing treatments for patients with conditions that result in corneal stem cell deficiency. Gipson (1989) performed an ultrastructural analysis of the limbal area in humans and rabbits to determine what, if any, differences could be observed in the ultrastructure of the basement membrane at the limbus and that of the central cornea. That study documented a reduced number of hemidesmosomes at the limbal zone compared to the central cornea. Also noted was that the basement membrane appeared thicker and partially duplicated, with laminin and Type VII collagen evident both at and beneath the limbal basement membrane zone. Others have shown collagen and laminin isoform differences at the limbal-corneal border (Ljubimov et al. 1995; Qin et al. 1997). The exact nature of the border remains to be determined, as do the molecular components required for its function.
The extracellular matrix environment at the limbus is likely to play a role similar to that of the stromal microenvironment in the bone marrow. In the bone marrow, specific stromal-stem cell interactions mediate the proliferation rate and retention of stem cells (Papayannopoulou et al. 1995; Vermeulen et al. 1998). Inhibiting the function of α4 integrin in the marrow causes the hematopoietic stem cells located there to migrate into the bloodstream. This observation has provided insight into hematopoiesis and has provided a method for obtaining large numbers of hematopoietic stem cells from patients. Tenascin-C is also important in the bone marrow; several hematapoietic precursor cells interact with tenascin-C in the marrow and this interaction stimulates stem cells to proliferate (Seiffert et al. 1998). In addition, the proliferation of cells derived from the marrow of tenascin-C KO animals exhibits reduced colony-forming capacity, and the longevity of the tenascin-C KO cell cultures is reduced compared to that of wild-type animals (Ohta et al. 1998). Both defects can be corrected by exogenous addition of soluble tenascin-C.
Although tremendous progress has been made in understanding how to obtain and manipulate the stem cells involved in hematopoiesis, we know little about regulation and maintainance of the stem cells needed for the corneal epithelium. Keeping their niche at the corneoscleral junction free from trauma is important, but we must also identify the proteins, both cell-associated and those present within the microenvironment at the limbus, that are required to maintain the health of these stem cells. Only then can we hope to develop culture methods and treatments to help those suffering from the loss of stem cells caused by damage to the limbus. Our studies demonstrate that one of the known components of this junction, tenascin-C, is not necessary for the limbus to retain its normal function in preserving the stem cell population after injury. Additional studies and the identification of the cell-cell and cell-substrate interactions necessary for limbal stem cell maintenance and function are necessary to address the problem of corneal stem cell deficiency.
Footnotes
Acknowledgments
Supported by NIH RO1 EYO 8512-09 (to MAS).
We wish to thank Drs H.P. Erickson and M. Kusakabe for providing the GRS/A tenascin-C and the GRS/A wild-type mice, Dr Robyn Ruffner and the GWU Center for Microscopy and Image Analysis for assistance with the photography and imaging, and Dr Bernard Zook and the staff at the GWUMC Animal Research Facility for caring for the mice.
