Abstract
In addition to being the specialized site for transmission of force from the muscle to the tendon, the myotendinous junction (MTJ) also plays an important role in muscle splitting during morphogenesis. An early event in the formation of the MTJ is a regional deposition of basement membranes. We used immunocytochemistry to investigate the distribution of laminin chains during the development of MTJs in human limb muscle at 8-22 weeks of gestation (wg) and in adult MTJs. We used polyclonal antibodies and a new monoclonal antibody (MAb) against the human laminin α1 G4/G5 domains. At 8-10 wg, laminin α1 and laminin α5 chains were specifically localized to the MTJ. Laminin α1 chain remained restricted to the MTJ at 22 wg as the laminin β2 chain had appeared, whereas the laminin α5 chain became deposited along the entire length of the myotubes from 12 wg. In the adult MTJ, only vestigial amounts of laminin α1 and laminin α5 chains could be detected. On the basis of co-distribution data, we speculate that laminin α1 chain in the forming MTJ undergoes an isoform switch from laminin 1 to laminin 3. Our data indicate a potentially important role for laminin α1 chain in skeletal muscle formation.
M
In this study we investigated the distribution of different laminin chains in the forming and mature human MTJ. Our data indicate that initially both laminin α1 and laminin α5 chains appear to be selectively deposited at the MTJ in the absence of the laminin β2 chain. The laminin α1 chain remains restricted to this subdomain of the muscle basement membrane as the laminin β2 chain appears at the MTJ, whereas the laminin α5 chain becomes deposited along the entire basement membrane with time. The localized expression of laminin α1 chain at the MTJ is developmentally regulated, and further maturation occurs later in development or postnatally because this laminin chain was present only in vestigial amounts in the adult MTJ. Our data point to important roles for laminin α1 in MTJ formation and also indicate that during a developmental window the rare laminin-3 isoform might be present at the MTJ.
Materials and Methods
Muscle Samples
A total of 14 limb muscle samples were obtained from 10 human fetuses legally aborted at approximately 8, 9, 10, 14, 18, 20, and 22 weeks of gestation (wg). Four normal muscle biopsies and two muscle samples obtained post mortem were used to study the adult MTJ. In addition, sections from two muscle biopsies from patients who were diagnosed with Duchenne muscular dystrophy, were also used to further ascertain the specificity of the monoclonal antibody (MAb) against the laminin α1 chain. The muscle samples were collected with the approval of the Ethical Committee of the Medical Faculty, Umeå University and in accordance with the Declaration of Helsinki of 1975. The muscle samples were rapidly frozen in propane chilled in liquid nitrogen and stored at −80C until use. Serial cross-sections (5 μm thick) were cut in a Reichert-Jung cryostat and stored at −30C until processing for immunohistochemistry.
Western Blotting
In a pilot study, the polyclonal antibodies hLN-α1G4/G5 to human laminin α1 revealed discrete but very restricted staining at MTJ in human fetal muscle. To confirm the validity of this staining, we tested the reactivity of these antibodies by Western blotting. Human fetal leg muscle of11 wg was snapfrozen. The tissue was thawed and laminins were extracted for 2 hr on ice after sonication of the tissue in 10 mM Tris-HCl, pH 7.4, 0.15 M NaCl, and 10 mM EDTA containing protease inhibitors. After centrifugation for 20 min at 11,000 times; g, extracted proteins were subjected to electrophoresis under reducing conditions on 5% SDS-PAGE. As a control, human laminin-1 purified from conditioned JAR cell media (Champliaud and Gullberg, unpublished data) was run in a parallell lane. Electrophoretically separated proteins were transferred to nitrocellulose, incubated with antibodies to hLN-α1G4/G5 specific for the 400-kD laminin α1 chain and the blot was developed using the ECL system (Amersham; Poole, UK) as described (Tiger et al. 1997). On the basis of the immunohistochemical staining, the amount of laminin α1 chain was expected to be very low. This was also observed when the immunoblot was developed. Whereas a strong laminin α1 chain signal appeared in the control lane, a band at the laminin α1 chain position in the muscle extract was seen only after prolonged exposures (not shown).
Immunohistochemistry
The sections were processed for immunohistochemistry with the following previously characterized antibodies: MAb 4C12 against laminin β1/γ1 chain (Immunotech, Marseille, France; diluted 1:1000); MAb 1928 against laminin β1 chain (Chemicon, Temecula, CA; diluted 1:500); MAb C4 to laminin β2 chain (Hunter et al. 1989; diluted 1:1000); polyclonal antibodies hLN-α1G4/G5 against the 400-kD laminin α1 chain (Tiger et al. 1997; diluted 1:2000); MAb 5H2 against laminin α2 chain (provided by E. Engvall, La Jolla, CA; diluted 1:250); MAb BM-2 against laminin α3 chain (Rousselle et al. 1991; diluted 1:50 to 1:5000); MAb 4C7 (sold as MAb 1924; Chemicon) against laminin α5 chain (diluted 1:3000); MAb B8B11 (provided by M.-F. Champliaud, Charlestown, MA; diluted 1:100) against laminin γ2 chain; polyclonal antibodies R16 (Koch et al. 1999; diluted 1:1000) against laminin γ3 chain; MAb 143 DB7 against all known isoforms of tenascin-C (Tiitta et al. 1992; diluted 1:200); MAb 4A10 against tenascin-C (supernatant diluted 1:10; Gullberg et al. 1997); MAb A4.951 (purchased from the Developmental Studies Hybridoma Bank maintained by the Dept. of Biological Sciences, University of Iowa, Ames, IA; diluted 1:50) against slow myosin heavy chain (Hughes et al. 1993).
When this study was almost completed, the MAb 163 DE4 was made available. This MAb was generated by immunizing Balb/C mice with a recombinant laminin α1 chain polypeptide corresponding to the G4/G5 region of human laminin α1. We used this MAb (diluted 1:100) to confirm the data obtained with the polyclonal antibody to the laminin α1 chain.
The tissue sections were air-dried for 15-30 min, rehydrated in PBS for 5 min, and then incubated with 5% normal rabbit serum (Dako; Copenhagen, Denmark) for 15 min to inhibit unspecific staining. The sections were then incubated overnight with the appropriate primary antibody at 4C. The primary antibodies were diluted in PBS with 0.1% bovine serum albumin. Thereafter, the sections were washed in PBS and again incubated with normal rabbit serum for 15 min, followed by incubation with rabbit anti-mouse IgG (Dako) for 30 min at room temperature. After washing in PBS for 15 min, the sections were incubated with peroxidase-mouse anti-peroxidase complex (Dako) for 30 min and then washed in PBS for 15 min. Normal swine serum, swine anti-rabbit IgG, and peroxidase-rabbit anti-peroxidase complex were used on the sections treated with the polyclonal antibodies. Development of peroxidase was obtained by applying a solution containing 1 mg/ml of diaminobenzidine and H2O2 for 5-10 min, followed by rinsing in running water for 5 min. Finally, the sections were dehydrated in graded concentrations of ethanol followed by xylene and mounted with DPX.
Additional sections were processed using the Vectastain Elite ABC peroxidase system (Vector Laboratories; Burlingame, CA).
Double labelings were obtained with indirect immunofluorescence technique by sequential incubations with two primary antibodies raised in different species and using secondary antibodies conjugated to either red Cy3 (for rat MAb 1928; Jackson Immuno Research Laboratories; Avondale, PA) or to red or green Alexa fluorochromes (Molecular Probes; Eugene, OR).
Control sections were treated in the same way as described above, except that the incubation with the primary antibody was omitted. Additional control sections were treated with preimmune serum from the rabbit producing the polyclonal antibodies hLN-α1G4/G5 to evaluate the level of unspecific staining. The sections were examined and photographed in a Zeiss microscope equipped with epifluorescence, using a CCD camera (Dage-MTI; Michigan City, MI).
Results
Fetal Muscle
The antibodies against tenascin-C (Figures 1A and 2B) strongly labeled the tendons at all ages, and moderate staining was also observed between individual myotubes and clusters of myotubes at 8 and 10 wg. The myotubes were labeled by the MAb against slow myosin heavy chain (Figure 1B) as previously reported (Hughes et al. 1993). The presence of a basement membrane was assessed with MAb 4C12, which recognizes the laminin β1 and γ1 chains. This MAb strongly delineated the contours of the developing myotubes and clusters from 8 wg (Figure 1C), and there was no difference in staining intensity towards the MTJ.
Polyclonal antibodies against the laminin α1 chain specifically labeled the extracellular matrix at the MTJ in fetal limb muscle from 8 to 22 wg (Figures 1D and 2A-2F). These antibodies did not specifically label the myotubes for the remainder of their length. In sections treated with rabbit preimmune serum (Figure 1E), no corresponding staining was observed at the MTJ.
The MAb 163D4 against the laminin α1 chain (Figure 1F) likewise strongly stained the extracellular matrix at the MTJ at all fetal ages and did not stain the contours of the myotubes outside of the MTJ.
Staining with MAb 5H2 against the laminin α2 chain at 8-10 wg was weak and rather uneven (not shown). At later stages, the staining intensity increased and there was variation among different regions and muscles. The contours of the developing myotubes and the MTJ stained moderately to strongly from 12 wg (Figure 1G).
The MAb BM-2 against the laminin α3 chain did not label developing muscle at any fetal age (not shown).
The MAb 4C7 against the laminin α5 chain labeled the extracellular matrix at the MTJ weakly to moderately at 8 and 10 wg (not shown). This MAb did not stain the contours of the developing myotubes outside the MTJ at 8 wg. From 10 wg, the MAb 4C7 weakly stained the contours of the developing myotubes in some of the muscles. From 12 wg, 4C7 labeled the contours of the myotube clusters and individual myotubes in all limb muscles investigated (Figure 1H). Variation in staining intensity was observed among different muscles and among muscle fascicles after 12 wg, but there was no difference in staining intensity towards the MTJ. Larger blood vessels were labeled by MAb 4C7 at all ages.
The MAb 1928, which is specific for laminin β1 chain, stained the contours of the developing myotubes weakly to moderately and the MTJ strongly between 8 and 12 wg (Figure 2C). From 14 wg, this MAb strongly stained both the contours of the myotubes and the MTJs (Figure 2E). Double staining with this MAb and the polyclonal antibodies against the laminin α1 chain showed that these chains were colocalized at the MTJ (Figures 2C and 2E).
The MAb C4 against laminin β2 chain labeled the contours of the individual myotubes weakly to moderately and strongly labeled MTJs (Figure 2F) and motor endplates (not shown) from 20 wg, as previously reported (Wewer et al. 1997). Double-stained sections revealed co-localization of β2 and α1 chains of laminin at the MTJ (Figure 2F).
The antibodies B8B11 against γ2 chain and R16 against γ3 chain of laminin did not stain the extra-cellular matrix around myotubes or at the MTJ (not shown).
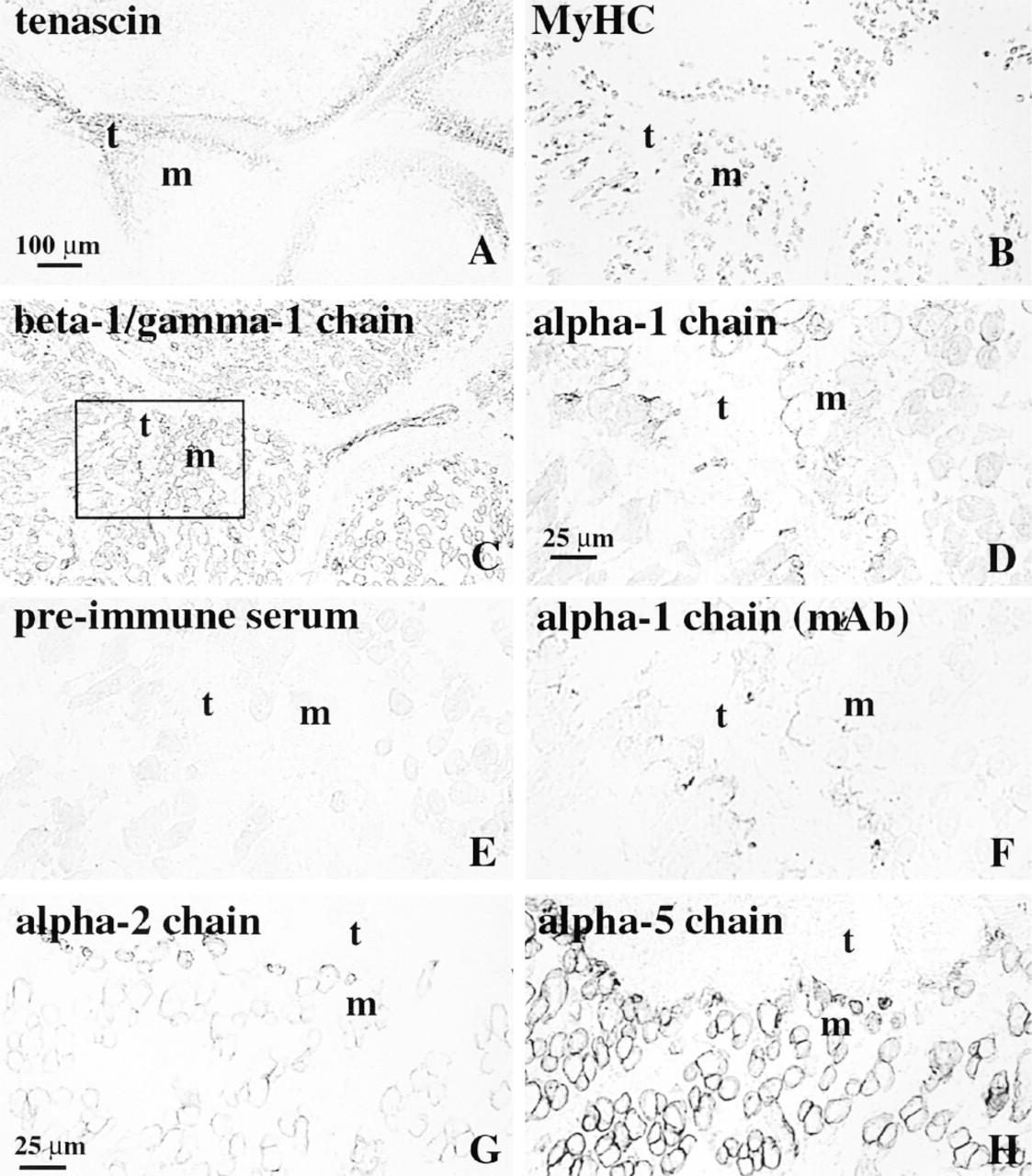
Serial cross-sections of human limb showing muscles (m) and tendons (t) at 10 (
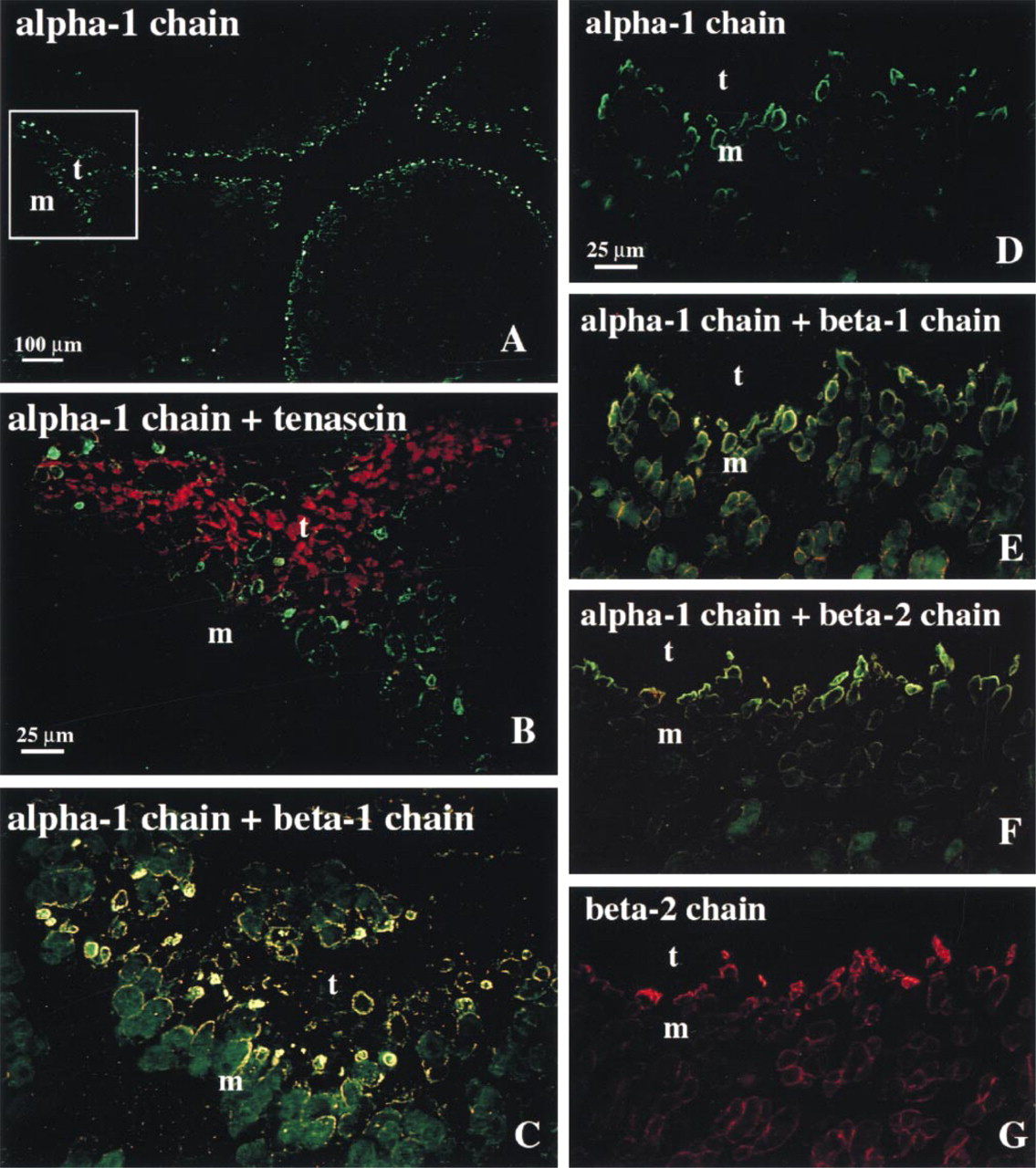
Immunofluorescence micrographs of cross-sections of human fetal muscle (m) and tendon (t) at 10 (
Adult Muscle
The staining pattern obtained with the polyclonal antibodies against laminin α1 chain (Figure 3A) was difficult to interpret and was not clearly different from the pattern seen in the sections treated with the corresponding preimmune serum (Figure 3B). The extracellular matrix of some of the adult MTJs was weakly stained by the MAbs against the laminin α1 chain (Figure 3C). The laminin α1 MAb did not label the extracellular matrix outside the MTJ. In sections from two patients suffering from Duchenne muscular dystrophy, the MAbs to laminin α1 chain failed to stain sarcolemmal basement membranes (not shown), confirming the results previously obtained with the polyclonal antibodies hLN-α1G4/G5 (Tiger et al. 1997).
The adult MTJs were weakly stained by the MAb against the α5 chain (Figure 3D) whereas the MAbs against the laminin α2 (Figure 3E) and β2 (Figure 3F) chains stained the MTJ strongly, in accordance with published results (Engvall et al. 1990; Wewer et al. 1997).
Discussion
The finding that defects in the laminin α2 chain can cause muscular dystrophy (Hillaire et al. 1994; Xu et al. 1994a,b; Helbling-Leclerc et al. 1995) has increased the interest in understanding the role of different laminins in muscle development. A detailed study of the transitions in laminin isoforms that occur during human development and disease is important to gain further understanding of the role of laminin isoforms in muscle function. In this study we have focused on the expression of laminin isoforms in the human MTJ.
During development and growth, the MTJ is subjected to dynamic changes. Electron microscopic studies of developing embryonic MTJ in avians indicate that basement membrane deposition is one of the first steps in MTJ formation (Tidball and Lin 1989). At later time points, junctional folds and myofibril insertions occur. Owing to the lack of a full panel of reagents to detect human laminin α-chains, knowledge about laminin isoforms in human muscle has been only fragmentary. We have recently shown that the MAb 4C7, suggested to recognize the laminin α1 chain, recognizes the a5 chain, and that laminin α1 is not prominently present in developing adult or dystrophic human skeletal muscle basement membranes (Tiger et al. 1997). Another consequence of the clarification of the specificity of 4C7 is that the laminin isoform previously recognized by 4C7 in certain tissues and thought to represent laminin 3 (α1β2γ1) really represents laminin 11(α5β2γ1). This updates the question of whether laminin 3 really exists in muscle.
In this study we have analyzed the laminin α1, α2, α3, α5, β1, β2, γ1, γ2, and γ3 chains in the developing MTJ. Laminin α1 chain was detected both with polyclonal antibodies and with a recently developed MAb. The specificity of the polyclonal antibody, which is raised to a recombinant protein encompassing the E3 domain, has been previously shown (Tiger et al. 1997). The MAb, raised to the same recombinant protein used to generate the polyclonal antibody (unpublished observations), does not react in immunoblotting, but it specifically immunoprecipitates the recombinant hLNα1 G4/G5 protein and human laminin 1.
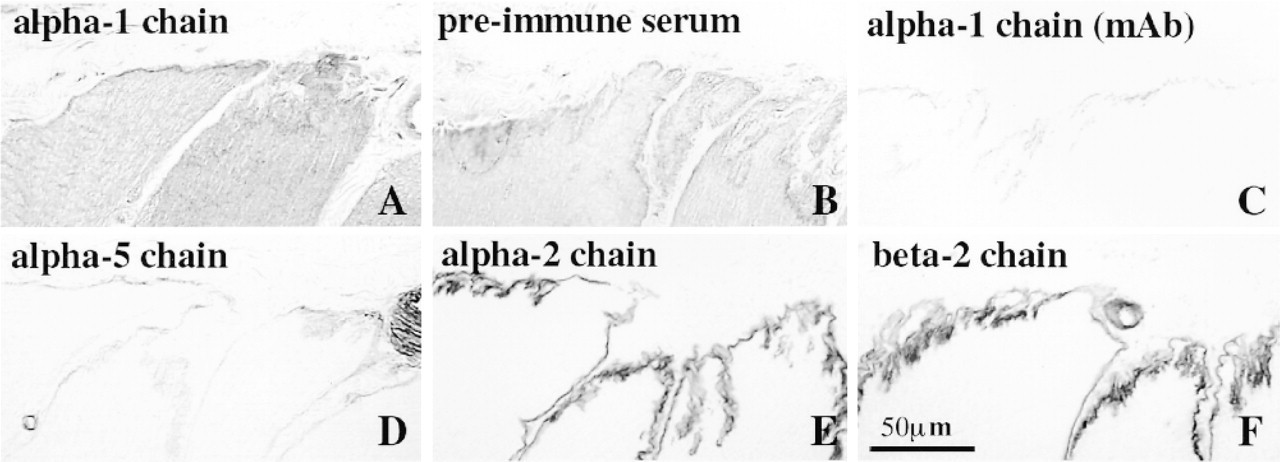
Serial cross-sections of adult human MTJs stained with polyclonal antibodies against α-1 chain (
The presence of a basement membrane at early time points was indicated by the staining observed with antibodies to laminin β1 and laminin β1/γ1 chains around the myotubes. The nature of the α-chain trimerizing with the β1/γ1 chains present in the early basement membranes detected around all myotubes is not known because none of the α-chains investigated were initially present in this pattern. Whether laminin α1 chain coexists with or replaces the early isoform of laminin α-chain at the MTJ remains to be determined.
Although reagents to laminin α1 chain in mouse have been available for some time, several groups have reported the lack of laminin α1 in developing skeletal muscle (Ekblom et al. 1990; Schuler and Sorokin 1995; Tiger et al. 1997). The restricted expression of laminin α1 chain to the MTJ is most likely the reason for the missed detection of laminin αl chain in previous reports. However, the presence of laminin αl in the forming MTJ has been reported in the developing mouse (Patton et al. 1997).
It is not known what laminin β-chains the laminin α1 chain associates with at the developing MTJ. It is expected that the amount of laminin α1 chain in muscle is very low, making biochemical and RNA characterizations very difficult to perform. The weak immunoblotting signal obtained from a muscle extract in the present study indicates that we are close to the limit of detection. Biochemical analysis of the chain composition of laminin α1 containing heterotrimers might be even more difficult. Likewise, in situ hybridization analysis will have to be very sensitive to detect the highly restricted laminin α1 expression. Analysis of transgenic mice having lacZ expressed under the control of the laminin α1 promoter might be the most feasible way to resolve the question of the origin of laminin α1 at the MTJ. On the basis of co-distribution patterns, we speculate that the laminin α1 chain is initially present as laminin 1 (α1β1γ1) but that as the laminin β2 chains start to accumulate, the laminin α1 chain may also exist at the MTJ in the form of laminin 3 (α1β2γ3). This is an interesting possibility that remains to be demonstrated biochemically. We cannot exclude the possibility that laminin α1 chain might form a heterotrimer with a novel β-chain. In our studies of laminin α1 chain synthesized in vitro or extracted from placenta, we have indirect data suggesting that laminin α1 chain does indeed associate with a novel β-chain in addition to its associations with laminin β1 and β2 in laminin 1 and laminin 3 (M.-F. Champliaud, personal communication).
The presence of laminin α1 chain in developing human muscle was restricted spatially to the MTJ and to some extent temporally, indicated by the weak or absent staining observed in the adult MTJ. We could not determine the exact time point for the downregulation of the laminin α1 chain from the MTJ owing to the fact that material older then 22 wg was not available. However, the presence of this laminin α-chain exclusively at the MTJ during the period of muscle morphogenesis suggests that it might play an important role in tendon attachment of the developing myotubes and that it might participate in such events as regulation of myotube growth as well as muscle splitting and shaping. Recent data revealed the existence of a sub-compartment at the ends of primary myotubes, where selective accumulation of the mRNA for a nuclear protein named MARP (muscle ankyrin repeat protein) occurs as a result of signaling from tendon mesenchyme (Baumeister et al. 1997). Both MARP and thrombospondin-4 are proposed to be involved in coupling of muscle formation and tendon morphogenesis (Baumeister et al. 1997). Data from Drosophila studies (Becker et al. 1997) also show the existence of reciprocal signaling events between muscle cells and the corresponding epidermal muscle attachment cells.
An important question pertains to what cells synthesize the laminin α1 chain accumulating at muscle endpoints. Laminin α1 chain can be synthesized either by muscle cells themselves or by other possibly fibroblast-like cells in the tendon. Evidence for the synthesis of laminins by fibroblasts exists in the literature (Kühl et al. 1984; Monical and Kefalides 1994), and the possible involvement of fibroblasts in regulating basement membrane assembly has been suggested by studies showing that the presence of fibroblasts contributes to basement membrane assembly on myotubes in vitro (Kühl et al. 1984; Sanderson et al. 1986). In either case, the initial expression of laminin α-chains at the muscle endpoints suggests the influence of signals from the adjacent tendon mesenchyme.
Laminin α3 chain was not present in basement membranes of muscle fibers. The distribution of laminin α4 chain has not yet been determined in developing human muscle, but in situ hybridization and immunolocalization studies in mouse have revealed laminin α4 chain in the developing skeletal muscle (Iivanainen et al. 1997). In mouse, the laminin α4 chain starts to be deposited on myotubes towards the end of the maturation of the primary myotubes (Patton et al. 1997). Postnatally, the laminin α4 chain disappears from the extrasynaptic sarcolemmal basement membrane. It is possible that laminin α4 chains are structurally “fitted” to accommodate basement membranes of growing cells. In this regard, it is interesting to note that laminin α4 lacks a short arm, which might influence its ability to homopolymerize and also its ability to interact with other ECM components and cells. Laminin α4 mRNA has been detected previously in human muscle (Richards et al. 1996) and it might therefore also be a major laminin α-chain in developing human muscle.
The early accumulation of laminin α2 and α5 chains at the MTJ was followed by expression of these isoforms along the entire length of the developing myotubes, suggesting that basement membrane remodeling starts at the endpoints of the developing muscle. The patterns of distribution of laminin α1, α5, and β2 chains in the basement membrane of developing human myotubes were in agreement with previous reports (Patton et al. 1997; Tiger et al. 1997; Wewer et al. 1997).
In summary, our data show that laminin α1 and laminin α5 are early basement membrane markers for the forming MTJ. At later points, laminin α5 is expressed around the entire myofiber, whereas laminin α1 remains restricted to the endpoints at times when important morphogenic events occur. Experiments are under way to determine the basis for the laminin α1 localization to the muscle endpoints.
Footnotes
Acknowledgments
Supported by grants from the Swedish Medical Research Council (12x-3934), Gustaf V:s Fond (DG), Bergvalls Stiftelse (DG), and the Medical Faculty of the University of Umeå.
We thank Mona Lindström for excellent technical assistance, Eva Engvall and Marie-France Champliaud for providing antibodies, and Peter Ekblom for support and stimulating discussions.
