Abstract
Muscle spindle density is extremely high in the deep muscles of the human neck. However, there is a paucity of information regarding the morphology and immunoreactivity of these muscle spindles. The objective of this study was to investigate the intrafusal fiber content and to assess the myosin heavy chain (MyHC) composition of muscle spindles from human deep neck muscles. In addition to the conventional spindles containing bag1, bag2, and chain fibers (b1b2c spindle), we observed a number of spindles lacking bag1 (b2c spindle) or bag2 (b1c spindle) fibers. Both bag1 and bag2 fibers contained slow tonic MyHCs along their entire fiber length and MyHCI, MyHCIIa, embryonic, and α-cardiac MyHC isoforms along a variable length of the fibers. Fetal MyHC was present in bag2 fibers but not in bag1 fibers. Nuclear chain fibers contained MyHCIIa, embryonic, and fetal isoforms with regional variations. We also compared the present data with our previous results obtained from muscle spindles in human biceps brachii and the first lumbrical muscles. The allotment of numbers of intrafusal fibers and the MyHC composition showed some muscle-related differences, suggesting functional specialization in the control of movement among different human muscles.
M
Muscle spindles contain specialized fibers, called intrafusal fibers, innervated by both sensory neurons and γ-motor neurons. There are three types of intrafusal fibers, nuclear bag1, nuclear bag2, and nuclear chain fibers, classified according to their distinct myosin ATPase (mATPase) reactions (Ovalle and Smith 1972). The morphological features of muscle spindles in different human muscles are more diverse than in other mammals. We have reported that the human muscle spindles in different muscles exhibit distinct features that most likely reflect the functional demands of these particular muscles (Eriksson and Thornell 1987,1990; Eriksson et al. 1994; Liu et al. 2002; Soukup et al. 2003). In human biceps brachii muscle, the muscle spindle density is much lower than in jaw and lumbrical muscles but each biceps spindle has a unique morphological identity (Liu et al. 2002). In contrast, spindles in the human masseter are exceptionally large and are more complex than those of limb muscles (Eriksson and Thornell 1987,1990). The deep muscles of the human neck, i.e., the rectus capitis posterior major, the rectus capitis posterior minor, the obliquus capitis inferior, and the obliquus capitis superior, are located deep in the suboccipital region. These muscles are quite small and short. They function in fine rotatory movements of the head and help maintain the stability of the cervical spine. The deep neck muscles have an unusually high muscle spindle density (Cooper and Daniel 1963; Peck et al. 1984; Kulkarni et al. 2001), almost five times higher than that of the splenius capitis and three times that of the semispinalis capitis muscle (Peck et al. 1984). However, the intrafusal fiber content and MyHC composition of muscle spindles from the deep muscles of the neck have not been studied in detail. The MyHC composition is considered the best marker for the functional properties of human skeletal muscle fibers because it correlates with contraction force and velocity (Larsson and Moss 1993; Bottinelli and Reggiani 2000; Bottinelli 2001).
In an attempt to correlate muscle spindle structure and function in human muscles, the fiber type content, general morphology, and MyHC composition of muscle spindles in the deep muscles of the human neck were examined and compared with the data available for other human muscles.
Materials and Methods
Deep muscles of the neck (rectus capitis posterior major, rectus capitis posterior minor, obliquus capitis inferior and obliquus capitis superior) were collected at autopsy from two females, ages 26 and 17, three males, ages 55, 21, and unknown, without known muscle related disorders. The samples were collected according to the ethical recommendations of the Swedish Transplantation Law, with the approval of the Medical Ethical Committee, Umeå University.
The muscle samples were mounted, rapidly frozen in propane chilled with liquid nitrogen, and stored at −81C until sectioned. Frozen specimens were serially sectioned at −23C using a Reichert Jung cryostat (Leica; Nussloch, Germany). Sections to be used for demonstration of mATPase activity after preincubation at pH 10.4, 4.6, and 4.3 (Dubowitz 1985) were 8 μm thick; those for immunocytochemistry were 5 μm thick.
Antibodies and Labeling
Monoclonal antibodies specific for different MyHC isoforms were used (Table 1). The specificities of these antibodies have been carefully assessed for human muscles (Liu et al. 2002). The short names of antibodies are listed in Table 1. All antibodies were diluted in 0.01 M PBS containing 0.1% bovine serum albumin (BSA) and were used at their optimal dilution.
Immunocytochemistry
Sections were processed for immunocytochemistry as previously described (Liu et al. 2002), using the indirect peroxidase-anti-peroxidase (PAP) technique (Sternberger 1979). The PAP products were purchased from Dakopatts (Glostrup, Denmark). Incubation with the appropriate primary antibody was performed for 1 hr at 37C. In control sections, the primary antibody was replaced by normal serum from the same species and at the same dilutions. No staining was observed in these sections.
All sections were examined and photographed with a Nikon microscope (Eclipse, E800; Tokyo, Japan) equipped with a Spot camera (RT color; Diagnostic Instruments, Sterling Heights, MI).
Survey
Twenty-two muscle specimens were sectioned and stained. A total of 199 muscle spindles were examined, and 36 of these were studied in consecutive serial transverse sections covering a distance of approximately 2 mm. Three regions were examined: the A region, including equator and juxtaequatorial parts, containing the periaxial fluid space; the B region, extending from the end of the periaxial fluid space to the end of the capsule; and the C region, corresponding to the extracapsular portion of the spindle (Barker and Banks 1994). Each region was further subdivided into an inner (proximal to the equator) and outer (distal to the equator) portion.
Statistical Analyses
StatView software (3rd ed; SAS Institute, Cary, NC) was used for statistical analysis. Statistic comparison of the intrafusal fiber numbers between different muscles was performed with an unpaired t-test. Differences were considered significant at p<0.05.
Results
General Morphological Features
The 199 muscle spindles studied contained a total of 1039 intrafusal fibers. Two hundred and two fibers were classified as bag1 fibers, 185 as bag2, and 649 as chain fibers. The remaining three fibers were considered as exceptions because of their abnormal histochemical or immunocytochemical staining patterns (see below). The number of intrafusal fibers per spindle varied considerably between four and 15 (Table 2). On average, there were 7.2 (range 4–15) fibers per spindle and in each spindle there were 1.3 (range 0–3) bag1, 1.0 (range 0–4) bag2, and 4.9 (range 2–11) chain fibers.
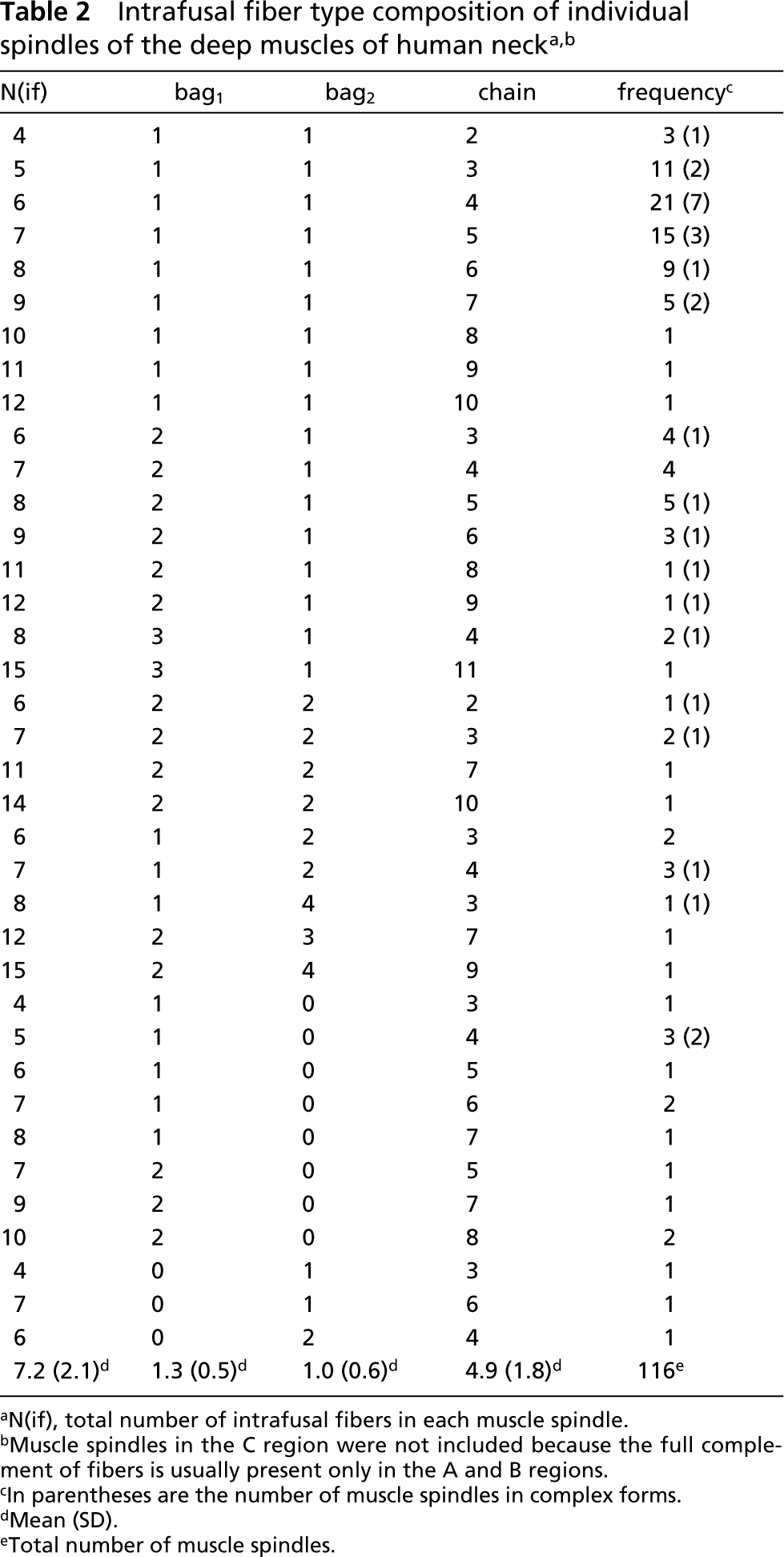
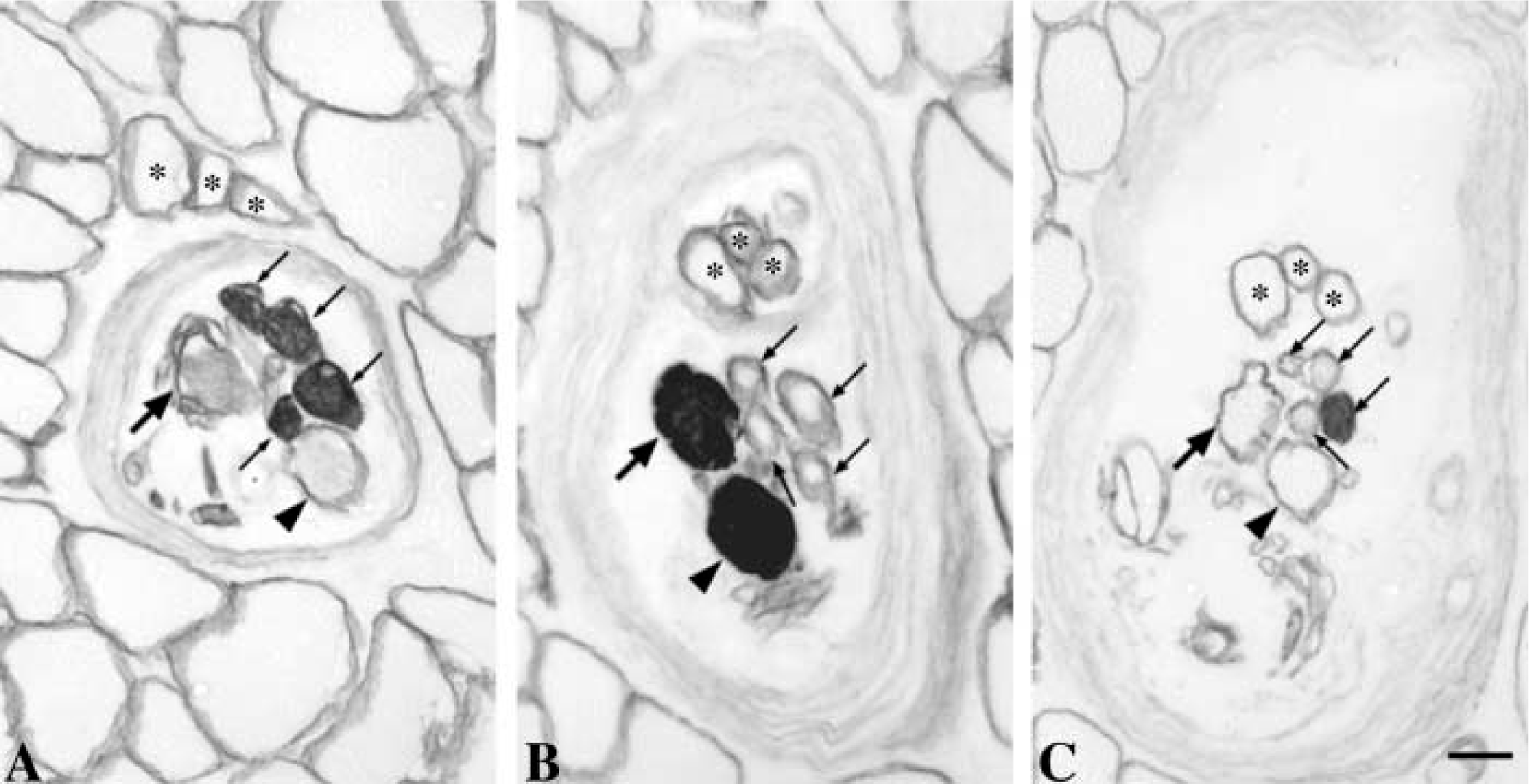
Although most spindles studied contained all three fiber types (b1b2c spindle), 13% of the spindles contained only one type of bag fiber (Figure 1); 10.4% lacked bag2 fibers (b1c spindle) whereas 2.6% lacked bag1 (b2c spindle). Two of 12 b1c spindles were part of two pairs of paired spindles, whereas the remaining were single receptors. These b1c spindles were found in three different subjects.
Only 20 of 116 muscle spindles examined in the A and/or B regions, where the full complement of intrafusal fibers is present, presented a unique allotment of numbers of bag1, bag2, and chain fibers. Twenty-one muscle spindles had exactly the same intrafusal fiber content: one bag1, one bag2, and four chain fibers.
Monoclonal antibodies used to detect different MyHC isoforms
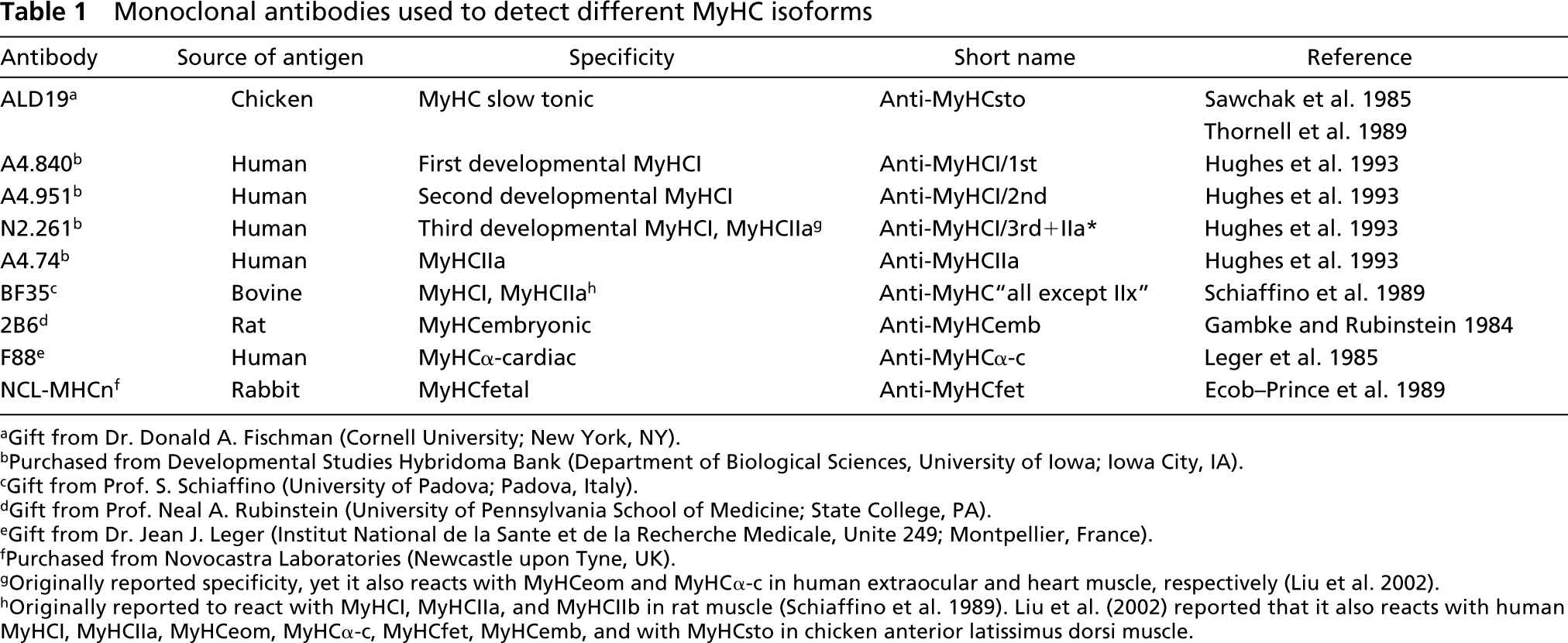
aGift from Dr. Donald A. Fischman (Cornell University; New York, NY).
bPurchased from Developmental Studies Hybridoma Bank (Department of Biological Sciences, University of Iowa; Iowa City, IA).
cGift from Prof. S. Schiaffino (University of Padova; Padova, Italy).
dGift from Prof. Neal A. Rubinstein (University of Pennsylvania School of Medicine; State College, PA).
eGift from Dr. Jean J. Leger (Institut National de la Sante et de la Recherche Medicale, Unite 249; Montpellier, France).
fPurchased from Novocastra Laboratories (Newcastle upon Tyne, UK).
gOriginally reported specificity, yet it also reacts with MyHCeom and MyHCα-c in human extraocular and heart muscle, respectively (Liu et al. 2002).
hOriginally reported to react with MyHCI, MyHCIIa, and MyHCIIb in rat muscle (Schiaffino et al. 1989). Liu et al. (2002) reported that it also reacts with human MyHCI, MyHCIIa, MyHCeom, MyHCα-c, MyHCfet, MyHCemb, and with MyHCsto in chicken anterior latissimus dorsi muscle.
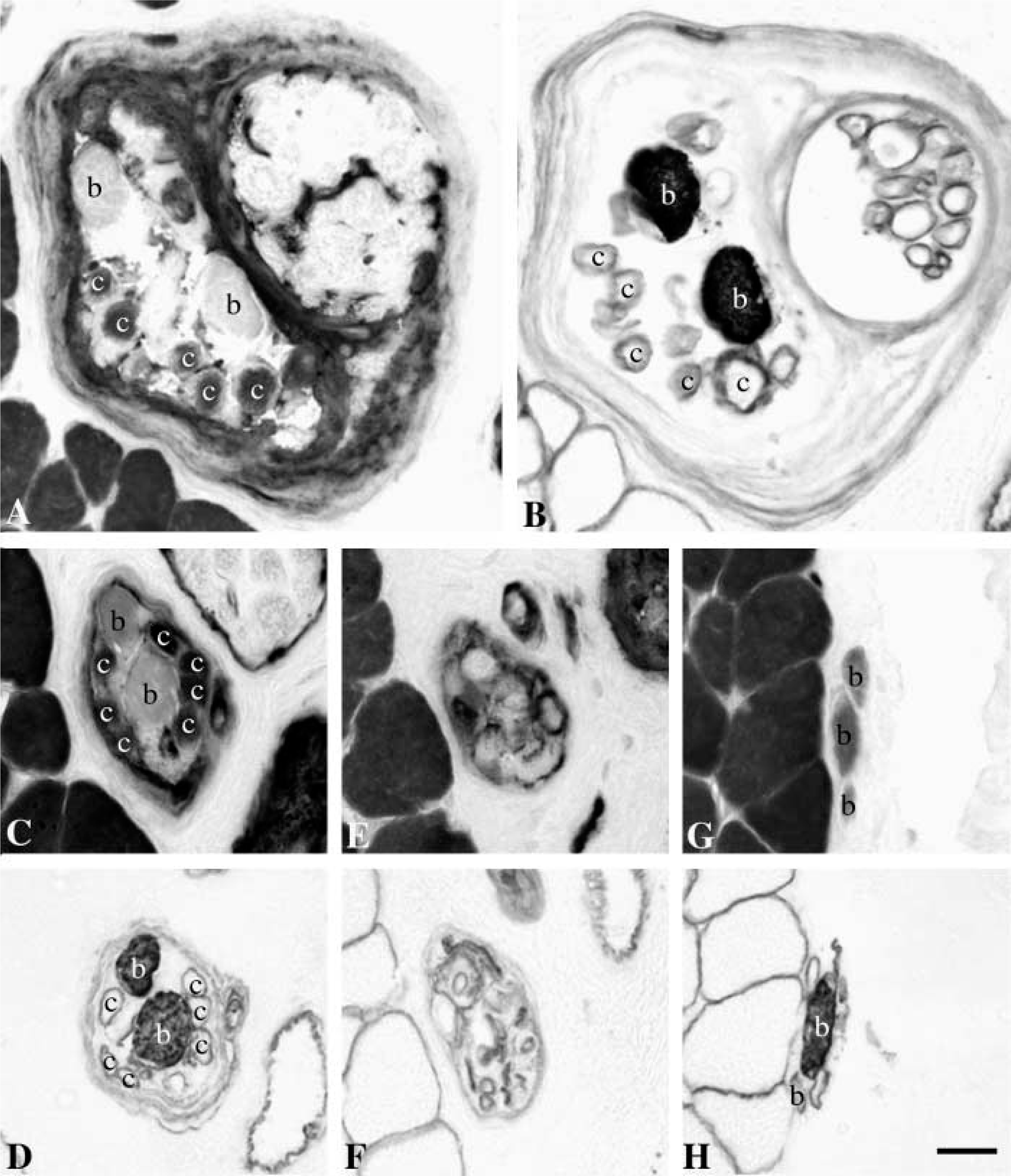
In transverse sections, nuclear bag fibers were in general much larger than nuclear chain fibers. The diameter of the two types of bag fibers was most often quite similar and rather constant along their length. However, some fibers exhibited dramatic changes in diameter along their length (Figure 2). The nuclear bag fibers usually extended beyond the capsule, whereas the chain fibers usually ended within the capsule, yet some exceptions were observed. In seven spindles, bag2 fibers terminated surprisingly in the A region at least in one pole, whereas the bag1 and chain fibers continued further along the spindle. In one spindle, we observed nine chain fibers together with one bag1 and one bag2 fiber in the C region.
Most spindles appeared as single isolated receptors, but 22% of the total number of muscle spindles and 36% among those followed along serial consecutive sections occurred in different forms of linkage. The linked forms observed in the present study generally fitted the previous descriptions of parallel and paired muscle spindles in cat neck muscles (Richmond and Abrahams 1975; Bakker and Richmond 1981,1982). The latter was the most commonly found in the present study. Two of 36 spindles followed in serial consecutive sections showed a third type of complex form. Each of these two spindles was first encountered in the C region, where it contained one bag1 and one bag2 fiber. The bag1 fiber tapered off and finally disappeared in the C region. The remaining bag2 fiber continued and a new bag1 fiber appeared, either in the C region or in the B region, of the new spindle. This new spindle, consisting of the new bag1 fiber together with the common bag2 fiber and five chain fibers, was then followed along the B and A regions. In addition to these three complex forms, one spindle had an extension of connective tissue from the spindle capsule which enclosed three extrafusal fibers, starting in the outer A region, for a certain distance (Figure 3).
In two spindles, at the end of the polar region, bag1 fibers adhered to the capsular wall, exhibiting no myofibrils for a short distance, and then parted from the wall to continue further outside the capsule (Figure 4).
mATPase Activity
The mATPase staining features of nuclear bag1, bag2, and chain fibers at different pH values were generally in accordance with our earlier report in human biceps brachii muscle (Liu et al. 2002). Some exceptions, however, were observed. In brief, nuclear bag1 fibers exhibited light staining along their whole fiber length at pH 10.4 (Figures 5A–5C). After preincubation at pH 4.3, these fibers showed low mATPase activity in the A region (Figure 5G) and occasionally even lost their activity in the equator. However, their mATPase activity gradually increased from low to moderate as bag1 fibers entered the B and C regions (Figures 5H and 5I). Nuclear bag2 fibers mostly displayed weak mATPase staining at pH 10.4 in the equatorial and juxtequatorial regions and moderate staining in the remaining parts of the fibers (Figures 5A–5C), although random variability was observed. The mATPase activity after preincubation at pH 4.3 was low in the equatorial region, low or moderate in the inner A region, and became intermediate to high along the rest of the length of the bag2 fibers (Figures 5G–5I). In 58% of the spindles, the staining intensities of both bag1 and bag2 fibers at pH 4.6 were the same as at pH 4.3. In the remaining bag fibers, however, the staining intensities observed at pH 4.6 were darker than those observed at pH 4.3. Nuclear chain fibers were always darkly stained over their entire length at pH 4.6 (Figures 5D–5F) and were darkly stained in the A and inner B regions but lightly stained in the remaining parts at pH 10.4 (Figures 5A–5C). In general, chain fibers displayed low or very low mATPase activity at pH 4.3, but 28% of the chain fibers showed intermediate or high activity in the A and B regions (Figures 5G–5I).
aN(if), total number of intrafusal fibers in each muscle spindle.
bMuscle spindles in the C region were not included because the full complement of fibers is usually present only in the A and B regions.
cIn parentheses are the number of muscle spindles in complex forms.
dMean (SD).
eTotal number of muscle spindles.
One bag fiber with unusual mATPase staining was encountered. This bag fiber displayed exactly the same staining features as bag1 fibers in the intracapsular region (Figures 5A, 5B, 5D, 5E, 5G, and 5H). In the extracapsular region, however, this fiber stained like a bag2 fiber (Figures 5C, 5F, and 5I) and it was therefore called “mixed bag1/bag2“ fiber.
Immunoreactivity
The staining patterns observed in the A, B, and C regions of nuclear bag1, bag2, and chain fibers with a battery of nine antibodies are summarized in Figure 6.
Nuclear Bag1 Fibers
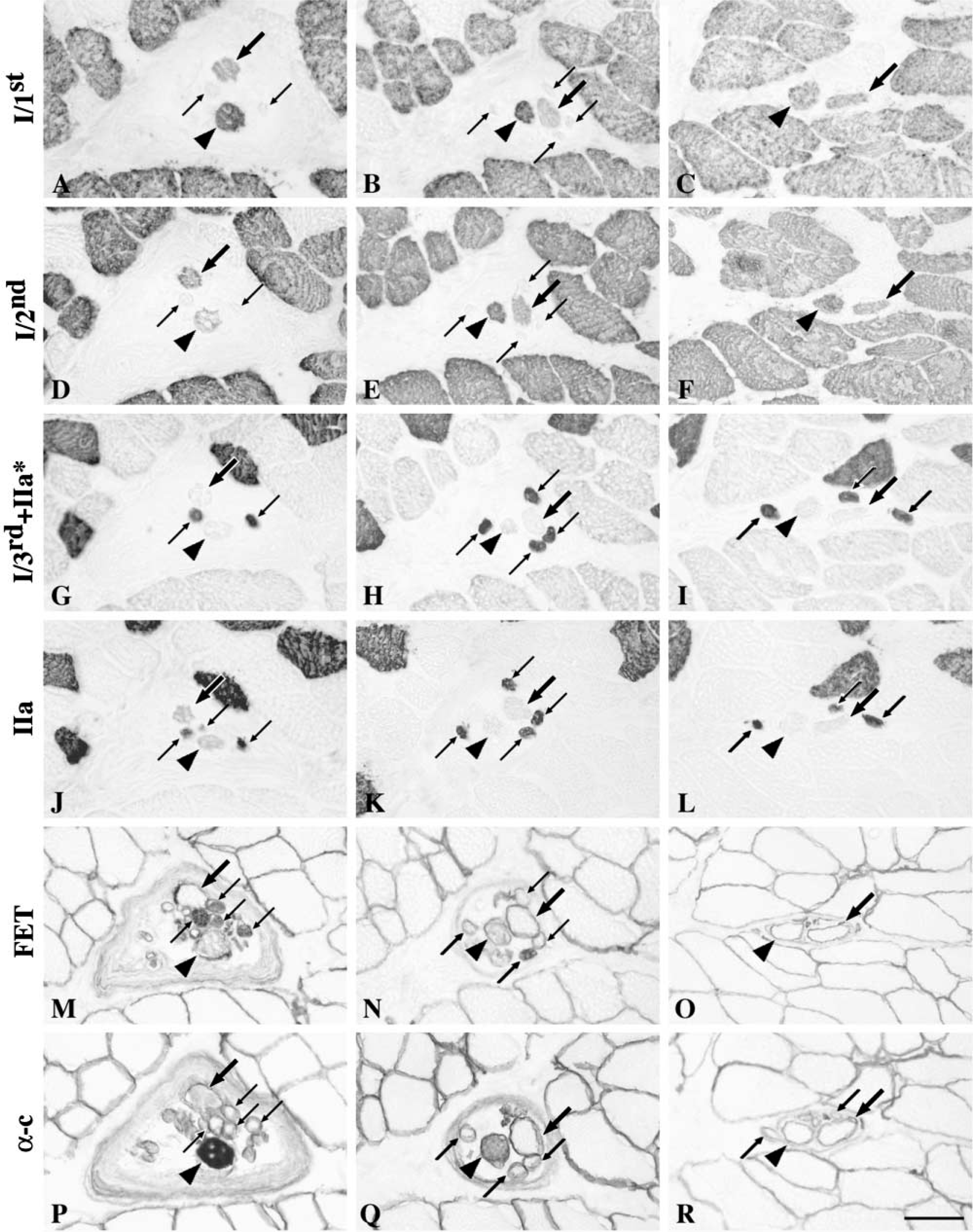
All nuclear bag1 fibers were strongly stained with anti-MyHCsto (Figures 5J–5L) and showed no staining with anti-MyHCfet (Figure 7M–7O) along their entire length. The vast majority of bag1 fibers showed similar staining patterns with the two anti-MyHCI antibodies. Their staining intensity with both anti-MyHCI/1st and anti-MyHCI/2nd MAbs gradually increased along the fiber length from the A to the C region (Figures 7A–7F). Bag1 fibers were weakly stained with anti-MyHCI/3rd+IIa∗ only in the outer B and C regions (Figures 7G–7I). Clearly, bag1 fibers reacted with anti-MyHCIIa antibody (Figure 7J–7L). The number of positive fibers was much higher in the A and B regions than that in the C region, in which only three of 31 bag1 fibers were stained. Bag1 fibers usually showed no staining with anti-MyHC“all except IIx” in the equatorial region, and they were either unstained or weakly stained in the juxtequatorial region. Their staining intensity gradually increased along the fiber length from the B into the C region (Figures 5M–5O). The majority of the bag1 fibers showed weak to moderate staining with anti-MyHCemb in the A region, fewer fibers were stained in the B region, and only 29% of the fibers were stained in the C region (Figures 5P–5R). Approximately one half of the bag1 fibers in the A region and 16% in the B region were labeled by anti-MyHCα-c, whereas they showed no staining in the C region (Figures 7P–7R).
Nuclear Bag2 Fibers
Nuclear bag2 fibers were strongly and evenly stained with anti-MyHCsto along their entire fiber length (Figures 5J–5L) except for three fibers that showed less reactivity at their tapering ending in the C region. Bag2 fibers were strongly and homogeneously stained with anti-MyHCI/1st and anti-MyHCI/2nd in both the C and B regions (Figure 7B, 7C, 7E, and 7F). In the A region, 97% and 94% of the bag2 fibers were stained with anti-MyHCI/1st (Figure 7A) and anti-MyHCI/2nd (Figure 7D), respectively. In general, bag2 fibers were unlabeled by anti-MyHCI/3rd+IIa∗ in the inner A region and weakly labeled along the remaining of their length (Figures 7G–7I). Although bag2 fibers mostly did not react with anti-MyHCIIa, a number of fibers were weakly or very weakly stained in the A and B regions (Figures 7J–7L). The staining intensity of bag2 fibers with anti-MyHC“all except IIx” gradually increased along the fiber length from the A to the C regions (Figures 5M–5O). Bag2 fibers did not react with anti-MyHCfet in the C region but 15% of these fibers were stained in both the A and B regions (Figures 7M–7O). The majority of bag2 fibers in the A and B regions were weakly or moderately stained with anti-MyHCemb (Figures 5P–5R). Bag2 fibers generally showed a higher level of reactivity with anti-MyHCα-c than with bag1 fibers in the same region (Figures 7P–7R).
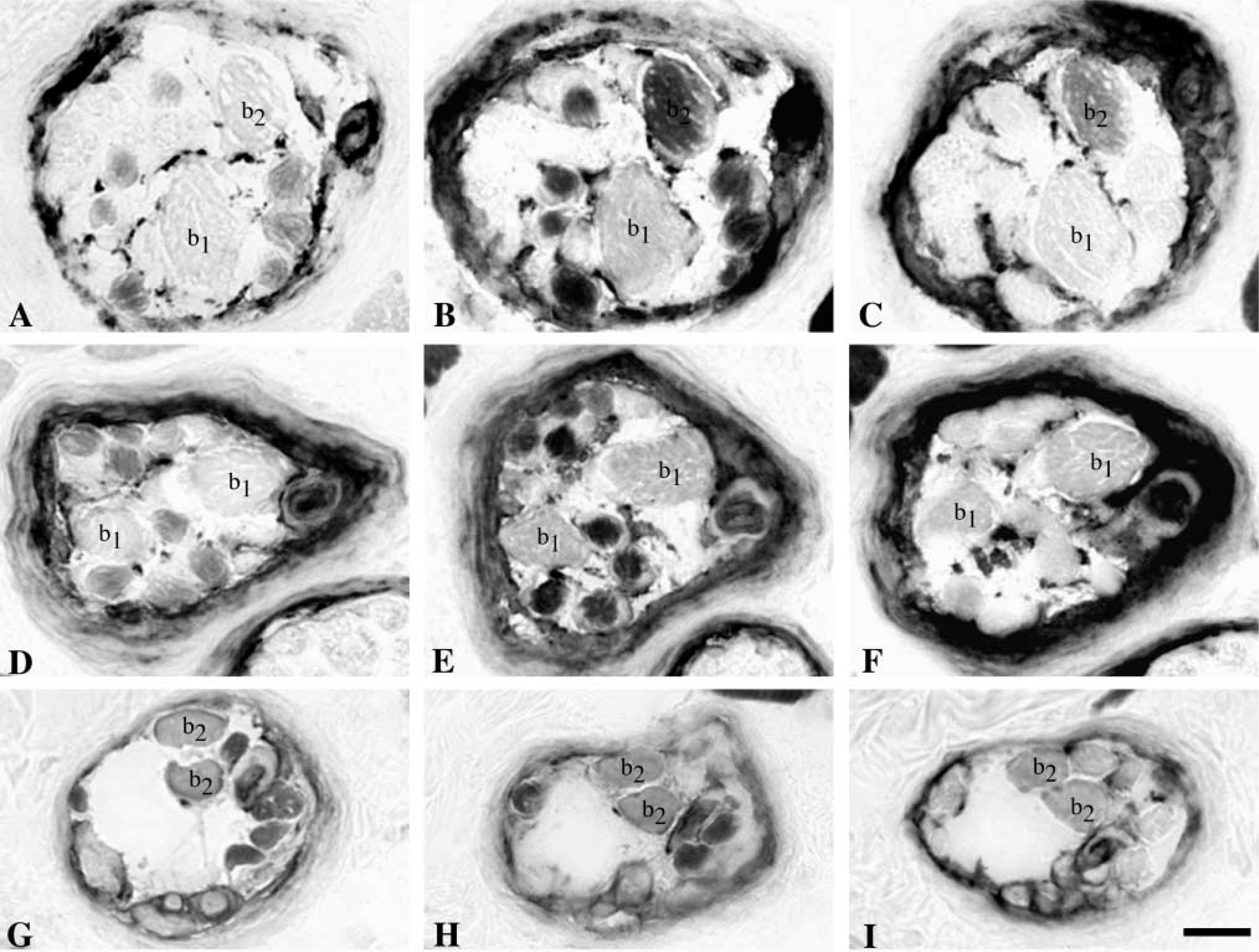
Identification of three types of muscle spindles. Serial transverse sections through intracapsular regions of a b1b2c spindle (
Nuclear Chain Fibers
The nuclear chain fibers showed no staining with anti-MyHCsto, anti-MyHCI/1st, anti-MyHCI/2nd, and anti-MyHCα-c but were strongly stained with anti-MyHC“all except IIx” (Figures 5 and 7). Almost all chain fibers showed strong staining with anti-MyHCI/3rd+IIa∗ (Figures 7G–7I) and anti-MyHCIIa (Figures 7J–7L). The chain fibers that were less stained with anti-MyHCI/3rd+IIa∗ showed also absent or weak staining with anti-MyHCIIa, except in one spindle where two chain fibers were weakly stained by anti-MyHCI/3rd+IIa∗ but strongly stained by anti-MyHCIIa. Nineteen percent of chain fibers in the A region, 47% in the B region and 64% in the C region showed no staining with anti-MyHCfet, whereas the remainder generally showed decreasing levels of staining intensity from the A to C regions (Figures 7M–7O). Chain fibers showed much higher staining intensity to anti-MyHCemb than bag fibers, although a few of chain fibers in all three regions were negative (Figures 5P–5R). As a general rule, fibers negative to anti-MyHCemb were also negative to anti-MyHCfet.
Exceptions
Although most intrafusal fibers clearly fitted into one of the three fiber types with the general staining patterns described above, two atypical intrafusal fibers were encountered in one spindle. These two fibers, seen in the A region, were similar to chain fibers in diameter and showed low mATPase activity at pH 10.4 and high activity at pH 4.3. They showed no staining with anti-MyHCfet, weak staining with anti-MyHCsto, and moderate to strong staining by all the other MAbs used in the present study (Figure 8).
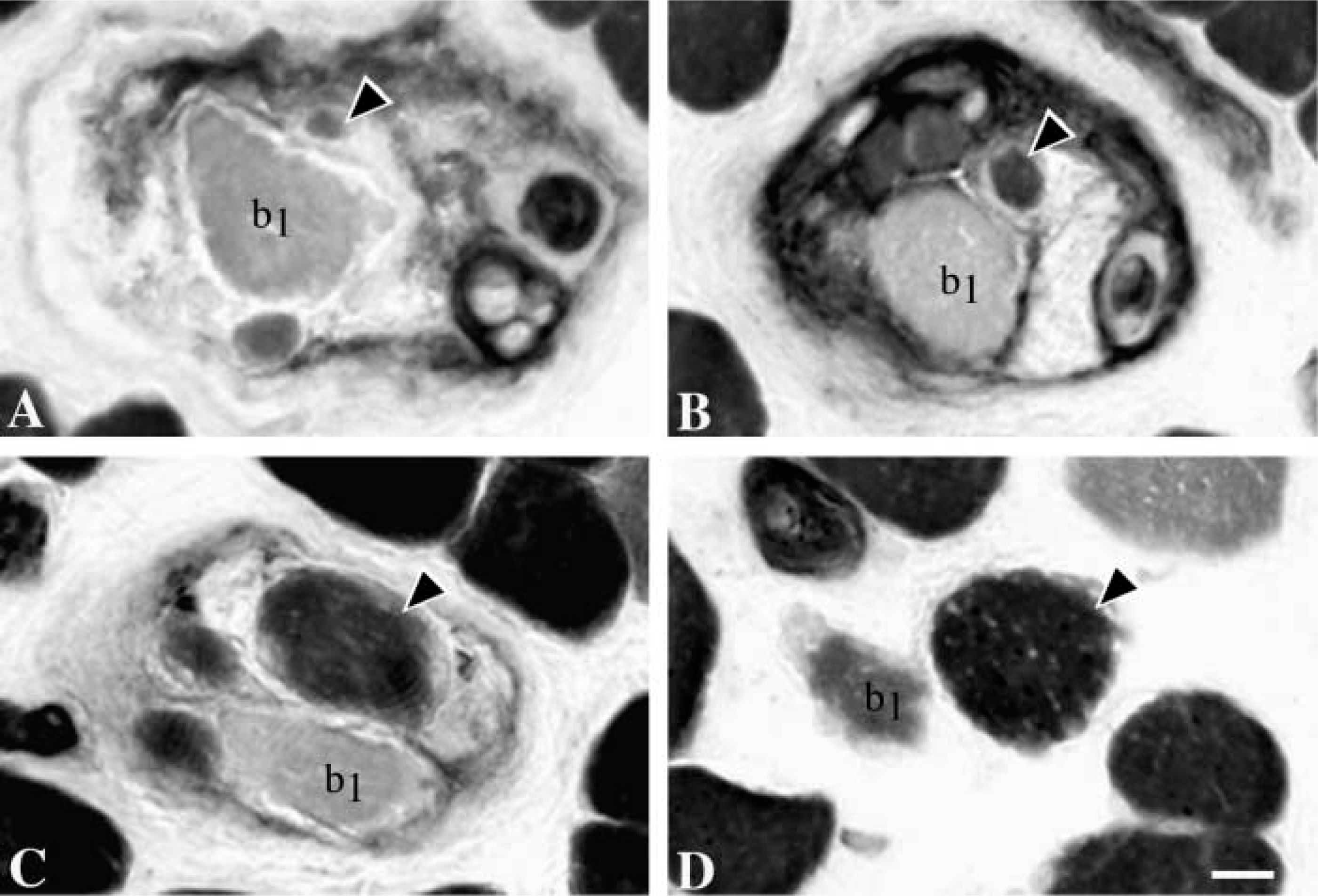
Dramatic changes in diameter along the length of a bag2 fiber (arrowhead). Serial cross-sections through intracapsular (
Discussion
The major highlights provided by the present study regarding the spindles in human deep neck muscles can be summarized as follows: (a) some spindles lacked bag2 fibers; (b) there was a low incidence of spindles with a unique allotment of intrafusal fiber types; (c) there was a low number of intrafusal fibers per spindle; (d) the content of MyHCemb and MyHCfet varied along the length of nuclear chain fibers; and (e) there were wide variations in MyHC composition among intrafusal fibers.
One-bag-fiber Spindles
Three categories of muscle spindles, b1b2c, b1c, and b2c, were identified. Our findings show that human neck muscle spindles can lack either bag1 or bag2 fibers. Moreover, 10 of 12 b1c spindles existed as single isolated receptors, indicating that the b1c spindles in human neck muscles are in fact rather common and are not likely to represent a developmental deviation. The ability to distinguish between b1c and b2c spindles relies on the precise classification of the two types of bag fibers. In the outer A and B regions, the bag2 fiber can be clearly distinguished from the bag1 fiber by its higher staining intensity after preincubation at pH 4.6 and pH 4.3 (Figure 1). Moreover, immunocytochemistry can also be used as an additional way of identifying intrafusal fiber types, and therefore the use of both these criteria in the present study allowed a reliable classification of nuclear bag1 and bag2 fibers.
Three extrafusal muscle fibers (asterisk,
Transient disappearance of intrafusal fibers. Serial sections taken through the juxtaequatorial (
The absence of bag2 fibers has previously been reported in a small number of muscle spindles in human biceps brachii muscle (Liu et al. 2002). Because of the lack of relevant physiological data on such b1c spindles in humans, their functional significance remains unknown. However, we can speculate that spindles lacking bag2 fibers might have a relatively higher dynamic sensitivity, and they might reflect adaptation of the fusimotor system to the particular task of controlling the head posture and movements.
One-bag-fiber spindles have been described in the cat (Banks et al. 1979; Kucera 1982; Kucera and Walro 1987), particularly in the neck muscles (Richmond and Abrahams 1975; Bakker and Richmond 1981,1982; Richmond et al. 1986; Abrahams and Richmond 1988). However, all the one-bag-fiber spindles in cat muscles selectively lack bag1 fibers (with one reported exception; Kucera 1982), usually appear as part of spindle complexes (Richmond and Abrahams 1979; Bakker and Richmond 1981,1982; Richmond et al. 1986; Abrahams and Richmond 1988), and are restricted to the regions near tendons or tendinous inscriptions in dorsal neck muscles (Bakker and Richmond 1981). We did not find any preferential location for the b1c spindles in our samples of the human deep neck muscles.
Intrafusal Fiber Type and Number
We have previously shown that each muscle spindle in the human biceps brachii has a unique identity, revealed in part by a unique allotment of numbers of bag1, bag2, and chain fibers (Liu et al. 2002). In the human first lumbrical muscle, which is a short muscle with high spindle density, the muscle spindles also have unique combinations of nuclear bag1, bag2, and chain fibers (Soukup et al. 2003). However, the present study revealed that only 17% of the muscle spindles in the deep muscles of the neck had unique intrafusal fiber complements, and as many as 18% of the muscle spindles studied in the A and B regions contained one bag1, one bag2, and four chain fibers. We compared our previous results with regard to the intrafusal fiber content from human biceps brachii (Liu et al. 2002) and the first lumbrical muscles (Soukup et al. 2003) with the deep neck muscles and found statistically significant differences. Muscle spindles in the lumbrical muscle have the highest number of intrafusal fibers, whereas those in the deep neck muscles contain the lowest fiber number (Figure 9). These differences were mainly due to a lower number of bag1 fibers in deep neck muscles compared to biceps brachii, and a lower number of bag1 and bag2 fibers in biceps compared to lumbrical muscle. The difference between the first lumbrical and the deep neck muscles was due to variation in the number of all three intrafusal fiber types.
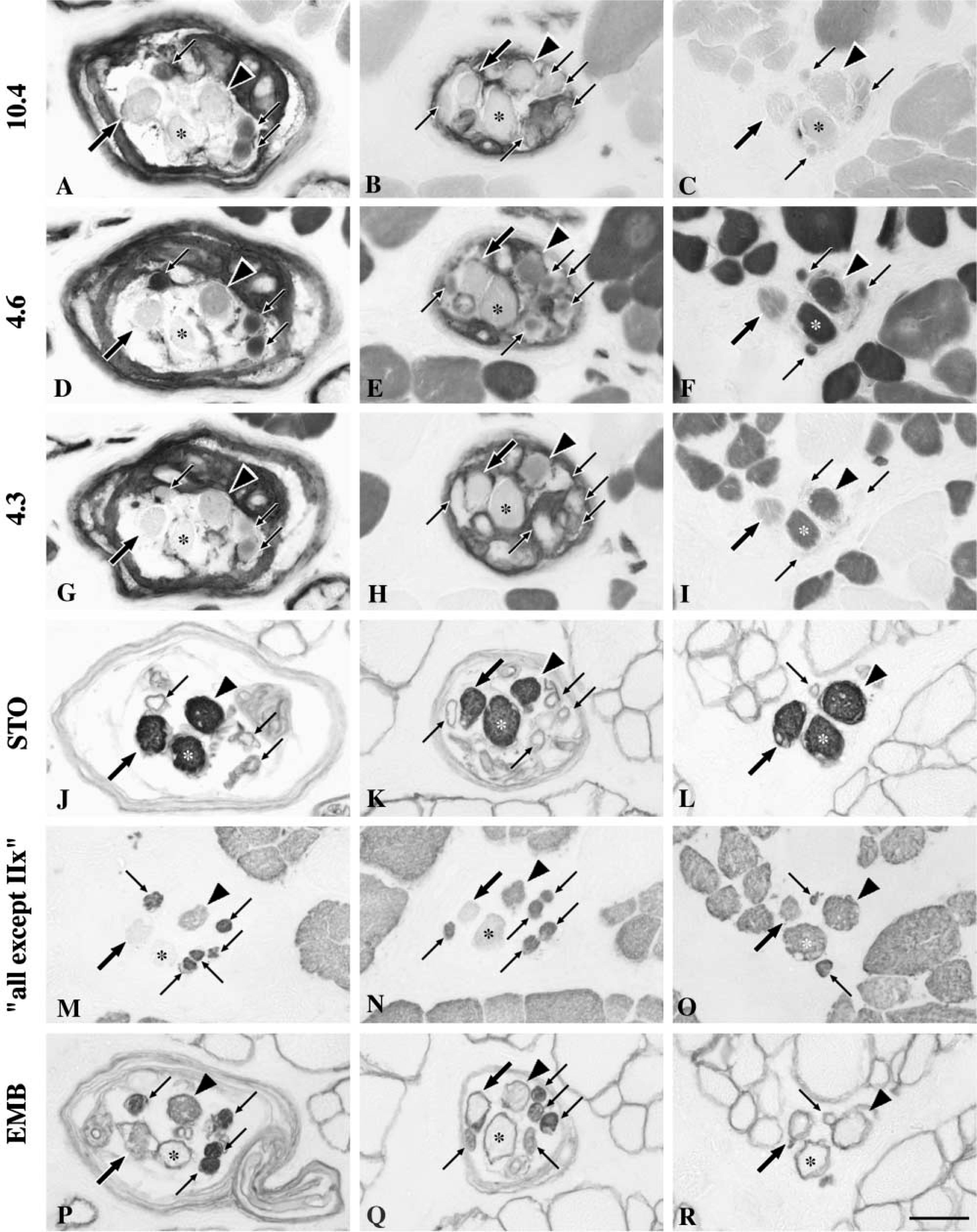
mATPase and immunocytochemical staining features of intrafusal fibers. Serial cross-sections of a spindle stained for the demonstration of mATPase at pH 10.4 (
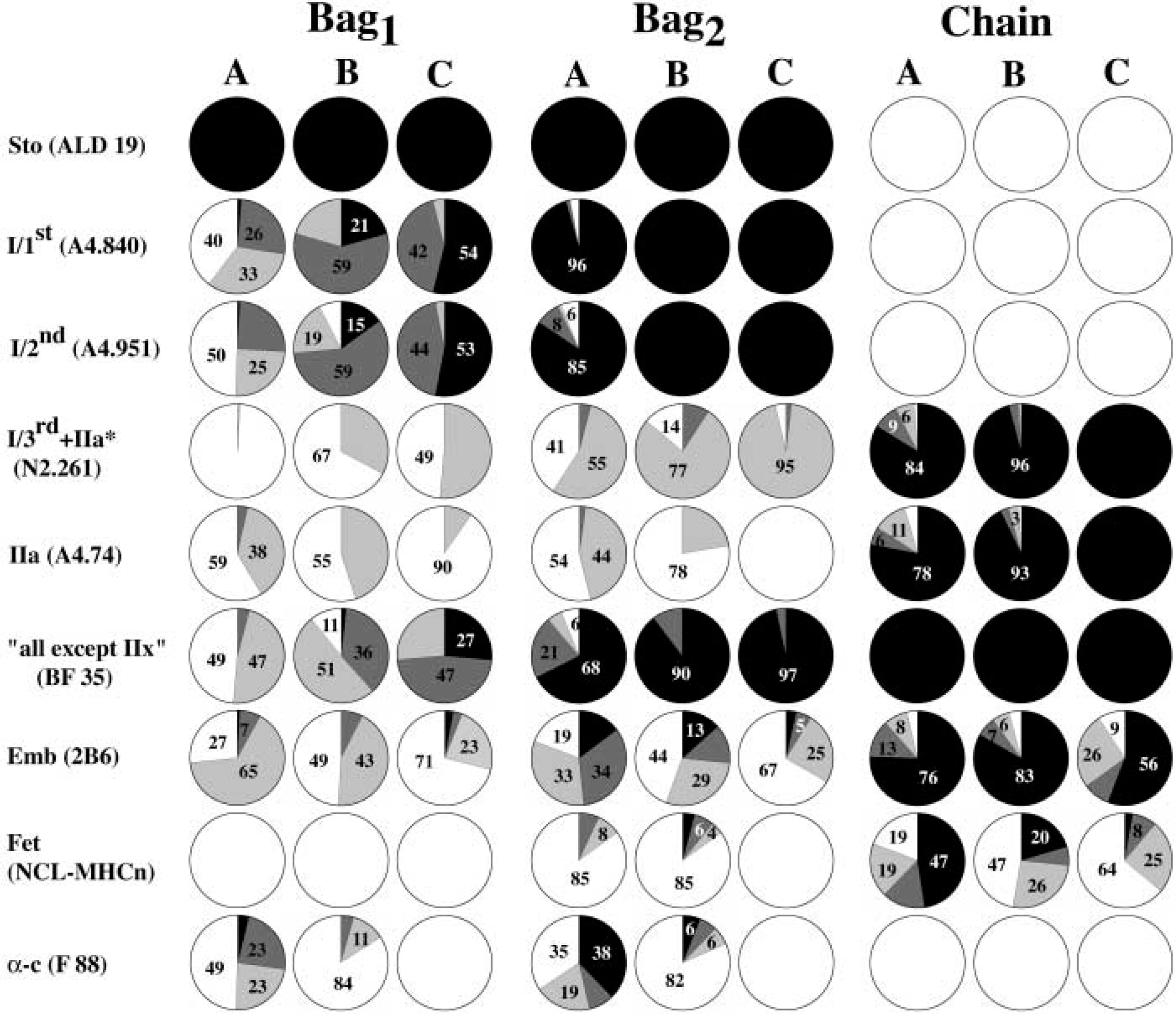
Schematic summary of the staining patterns of nuclear bag1, bag2, and chain fibers. The relative proportion (%) of each fiber type displaying the different staining levels in the A, B and C regions is shown. Black, strong staining; dark gray, moderate staining; light gray, weak staining; white, no staining.
MyHC Composition in Intrafusal Fibers
This is, to the best of our knowledge, the first investigation to examine the MyHC composition of intrafusal fibers in the deep muscles of the human neck. The immunocytochemical staining profiles of the intrafusal fibers reported here generally paralleled those of human biceps brachii (Liu et al. 2002), although with important exceptions.
The most striking difference was the variation in MyHCemb and MyHCfet content of nuclear chain fibers. The distribution of the embryonic and fetal myosin varied between the chain fibers from different spindles, and along the length of a given chain fiber. These variations seemed to correlate with the variability in mATPase activity noted along the length of individual nuclear chain fibers in the neck muscle spindles. Nuclear chain fibers usually have uniform mATPase activity and a homogeneous MyHC composition along their entire length in muscle spindles of human biceps brachii (Liu et al. 2002) and the first lumbrical muscles (Soukup et al. unpublished data).
There were wide variations in MyHC composition among the intrafusal fibers studied. In fact, we found more variation in the staining patterns along the intrafusal fibers in the deep neck muscles than in the biceps brachii, as shown in Figure 6. For example, staining with MAb A4.840 did generally increase along the length of nuclear bag1 fibers, but the transition in staining level was spread along the A and B regions. The widely variable staining features revealed in the present study are likely to reflect a wider range of contractile properties of the intrafusal fibers and the more complex architecture and functions of these small deep neck muscles. The deep neck muscles have limited lever action because of their small size (Kamibayashi and Richmond 1998), and in the rhesus monkey they are preferentially recruited during small turns, whereas larger, multiarticular muscles also became active during larger faster turns (Corneil et al. 2001).
MyHC expression in different regions. Serial cross-sections of one spindle stained with anti-MyHCI/1st (
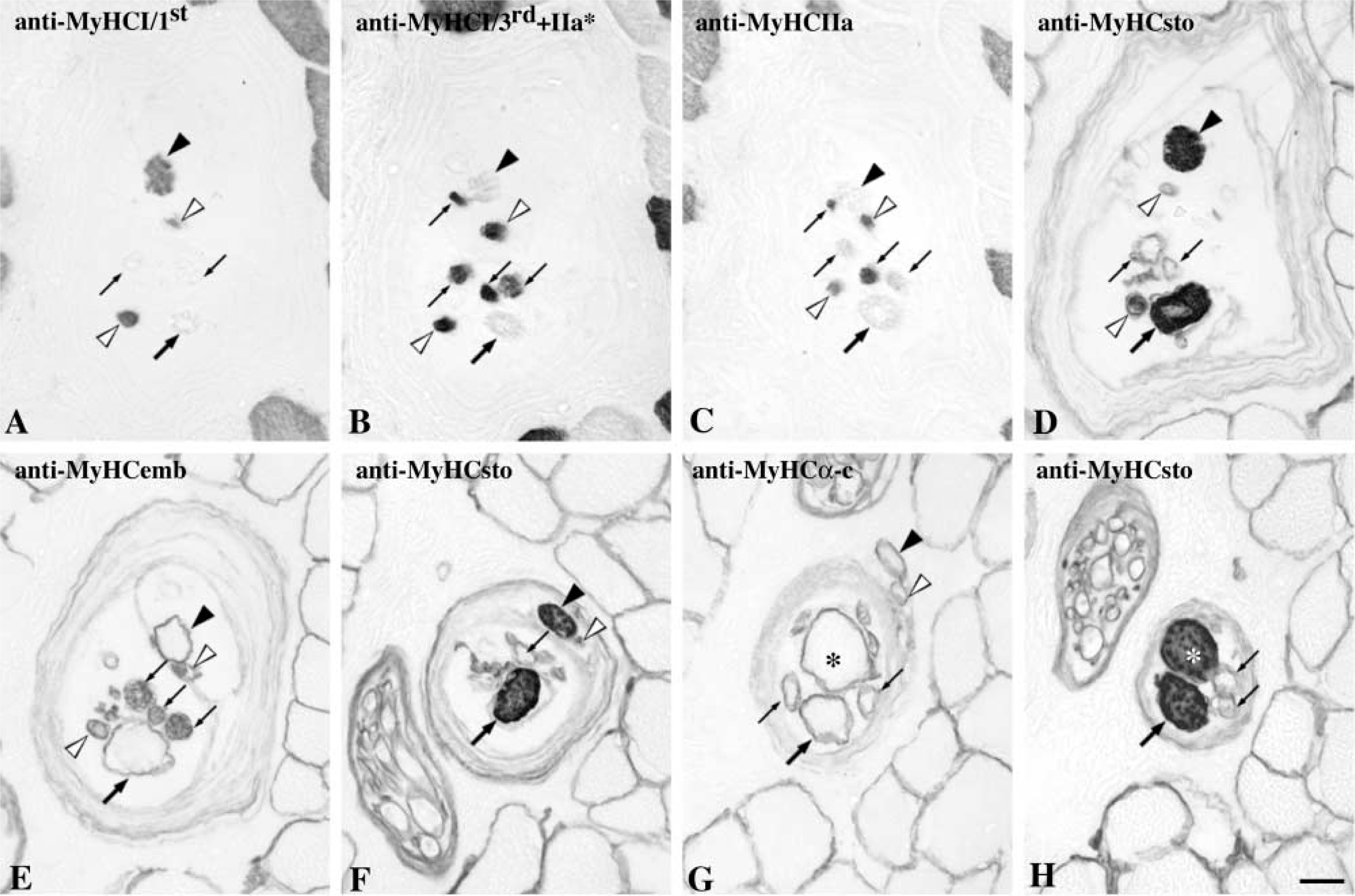
Intrafusal fibers with unusual staining features. Note that the two fibers marked with open arrowheads stained as nuclear bag2 fibers with anti-MyHCI/1st (
The high muscle spindle density and the special features of the muscle spindles in the deep neck muscles allow not only great precision of movement but also adequate proprioceptive information needed both for control of head position and movements and for eye/head movement coordination (Bakker et al. 1984; Edney and Porter 1986; Porter 1986). This study clearly revealed that the muscle spindles of the deep neck muscles have distinct morphological features and MyHC composition. Our data further support the concept that each muscle is unique in both fiber type composition and sensorimotor organization.
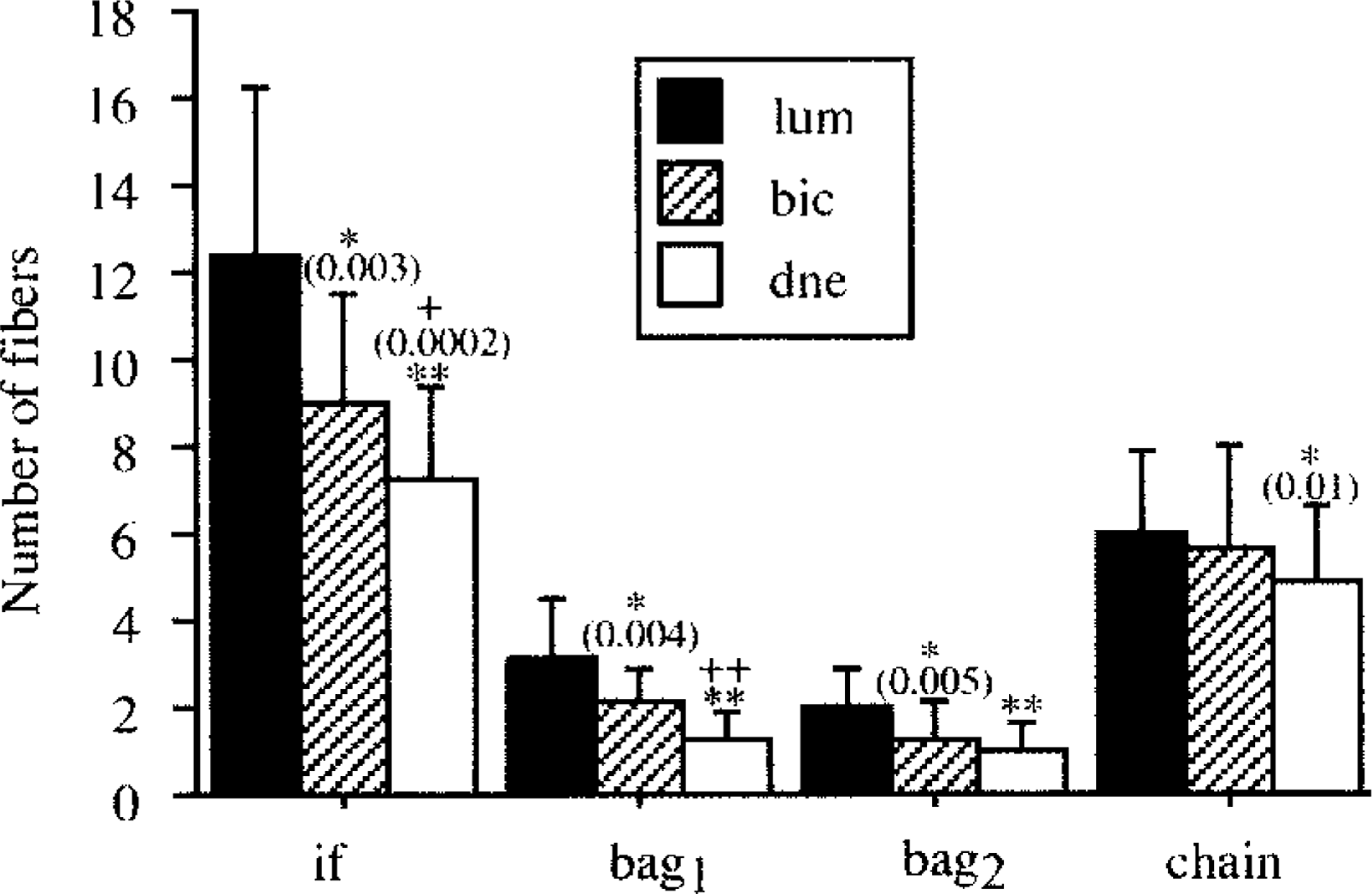
Comparison of intrafusal fiber complement (mean ± SD) between human first lumbrical (lum), biceps brachii (bic), and deep neck (dne) muscles. The asterisk indicates statistically significant difference from the first lumbrical muscle and “+” indicates statistically significant difference from biceps brachii. p values are given in parenthesis. ∗∗, ++ p<0.0001. if indicates all intrafusal fibers.
Footnotes
Acknowledgements
Supported by grants from the Medical Faculty of Umeå University and by The Swedish Research Council.
We thank Drs N. Rubinstein, D.A. Fischman, G.S. Butler-Browne, J.J. Leger, and S. Schiaffino for kindly providing antibodies. The excellent technical assistance of Margaretha Enerstedt is also acknowledged.