Abstract
Objective:
Attention deficit hyperactivity disorder is a frequent comorbid condition in adults with bipolar disorder. We performed a meta-analysis aimed at assessing sociodemographic and clinical correlates of attention deficit hyperactivity disorder in bipolar disorder.
Method:
We searched main electronic databases up to June 2021. Random-effects meta-analyses, with relevant meta-regression and quality-based sensitivity analyses, were carried out to estimate the association between attention deficit hyperactivity disorder and putative correlates, grading the quality of evidence.
Results:
We included 43 studies, based on 38 independent samples. Attention deficit hyperactivity disorder participants were more likely to be males (odds ratio = 1.46; p < 0.001) and unemployed (odds ratio = 1.45; p = 0.045), and less likely to be married (odds ratio = 0.62; p = 0.014). They had an earlier onset of bipolar disorder (standardized mean difference = −0.36; p < 0.001); more mood episodes (standardized mean difference = 0.35; p = 0.007), particularly depressive (standardized mean difference = 0.30; p = 0.011) and mixed (standardized mean difference = 0.30; p = 0.031) ones; higher odds of using antidepressants (odds ratio = 1.80; p = 0.024) and attempted suicides (odds ratio = 1.83; p < 0.001) and lower odds of psychotic features (odds ratio = 0.63; p = 0.010). Moreover, they were more likely to have generalized anxiety disorder (odds ratio = 1.50; p = 0.019), panic disorder (odds ratio = 1.89; p < 0.001), social phobia (odds ratio = 1.61; p = 0.017), eating disorders (odds ratio = 1.91; p = 0.007), antisocial personality disorder (odds ratio = 3.59; p = 0.004) and substance (odds ratio = 2.29; p < 0.001) or alcohol (odds ratio = 2.28; p < 0.001) use disorders. Quality of the evidence was generally low or very low for the majority of correlates, except for bipolar disorder onset and alcohol/substance use disorders (high), and suicide attempts (moderate).
Conclusion:
Comorbid bipolar disorder/attention deficit hyperactivity disorder may have some distinctive clinical features including an earlier onset of bipolar disorder and higher comorbid alcohol/substance use disorder rates. Further research is needed to identify additional clinical characteristics of this comorbidity.
Introduction
Bipolar disorder (BD) is a severe mental illness affecting about 1–2% of the population worldwide, requiring both pharmacological and non-pharmacological interventions (Bartoli et al., 2018; Carvalho et al., 2020; Fiorillo et al., 2015; McIntyre et al., 2020). It is characterized by a chronic and recurring course, a marked impact on the quality of life and high rates of psychiatric and medical comorbidities (Bartoli et al., 2013; Carvalho et al., 2020; McIntyre et al., 2020). Among these, attention deficit hyperactivity disorder (ADHD) is a frequently concurrent condition, involving about one in six adults suffering from BD (Schiweck et al., 2021). Several hypotheses have been proposed to explain the reasons behind the high frequency of this comorbidity. These disorders might stem from a common genetic or biopsychosocial diathesis, with shared mechanisms and vulnerabilities (O’Connell et al., 2021; Van Hulzen et al., 2017). ADHD may be thus a form of juvenile BD occurring before the onset of a first full affective episode (Brancati et al., 2021). An alternative hypothesis might regard the association between ADHD and BD merely as an epiphenomenal link of two different disorders (Mucci et al., 2019; Youngstrom et al., 2010; Zepf, 2009). Indeed, from a clinical standpoint, ADHD and BD have plenty of overlapping symptoms, such as distractibility, heightened motor activity, irritability, emotional dysregulation and mood lability (Faraone et al., 2019). Thus, the similarities in terms of clinical features may make the boundaries between these disorders blurred and unclear. Nonetheless, BD symptoms represent a marked deviation from the individual’s euthymic state, while ADHD features are chronic, trait-like and probably refer to deviations from developmental trajectories (Skirrow et al., 2012). Therefore, the comorbidity of BD and ADHD adds a layer of complexity to the whole diagnostic process. As a consequence, ADHD is often untreated in individuals with BD, with no more than 10% of subjects being properly diagnosed and receiving specific treatment for ADHD (Bond et al., 2012). Moreover, although recent systematic data attempted to clarify the epidemiological impact of ADHD in BD (Bartoli et al., 2022; Sandstrom et al., 2021; Schiweck et al., 2021), findings on possible sociodemographic and clinical correlates of this comorbidity are sparse and somehow inconsistent. The identification of factors associated with comorbid ADHD could be useful to clarify if these subjects might represent a clinical subpopulation with specific unmet needs, thus requiring tailored treatments. In particular, we hypothesized that individuals with co-occurring BD and ADHD might be characterized by specific clinical features, for example, in terms of number of mood episodes, suicidal behaviors and additional comorbid conditions, such as anxiety and substance or alcohol use disorders (Bond et al., 2012; Hegerl et al., 2010; Klassen et al., 2010; Mucci et al., 2019; Sandstrom et al., 2021; Schiweck et al., 2021). To shed light on this topic, we performed a systematic review and meta-analysis of observational studies aimed at identifying sociodemographic and clinical correlates of ADHD among adults with BD and assessing the quality of existing evidence.
Methods
This meta-analysis was carried out according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 Statement (Page et al., 2021a). The Study protocol registration was completed in Open Science Framework registries on 29 July 2021 (doi: 10.17605/OSF.IO/JV7MY).
Eligibility criteria
We included observational studies testing the cross-sectional association between one or more sociodemographic or clinical characteristics and comorbid current or past ADHD in BD. We excluded studies (1) not providing information on ADHD, (2) involving individuals with mood disorders but without specific data on BD, (3) including samples with a mean age <18 years, (4) providing insufficient data on correlates to be included in the meta-analysis, as well as (5) multiple works, based on data from the same sample, to avoid duplicate results. We also excluded studies with incomplete data or not undergoing peer-review process, such as conference abstracts, dissertations and gray literature.
Search strategy and selection of studies
We searched Embase, Ovid MEDLINE and APA PsycInfo databases (via Ovid) for articles indexed up to 14 June 2021, without language restrictions, using the following search phrase: ((bipolar or mania) and ADHD).mp., with ‘.mp’ code meaning that it included title, abstract, heading words and keywords. A supplemental, post hoc, non-systematic search on Google Scholar was carried out to check whether additional studies were retrievable. We also performed a manual search of the reference lists of two relevant reviews (Sandstrom et al., 2021; Schiweck et al., 2021). Four authors independently completed the preliminary screening based on titles and abstracts. Full texts were then retrieved to assess studies according to inclusion criteria for final eligibility. Disagreements concerning suitability for inclusion were resolved by discussion and consensus, involving all authors.
Data extraction
We used an Excel data extraction template including key items for all eligible studies: year of publication, country, setting, inclusion criteria, sample size, mean age, gender proportion, mood phase (i.e. euthymia, depression and/or mania/hypomania), methods to diagnose ADHD, sociodemographic and clinical correlates of ADHD, and main findings. Four authors independently extracted data for blind check of accuracy. Authors of studies with unclear or partial data were contacted by email for additional information to reduce the risk of selective reporting bias by including possible unpublished findings.
Quality assessment
We evaluated the risk of selection bias by checking whether compared groups (BD with vs BD without ADHD) were similar in terms of main characteristics, that is, age and gender, considering acceptable a difference of maximum 3 years in mean age and of 5% in men proportion. Second, we assessed the representativeness of studies included in our meta-analysis, verifying whether participants were not selected from special populations in terms, for instance, of age, gender or clinical characteristics. Finally, we evaluated studies for potential sources of information and misclassification bias. First, we checked whether studies used adequate instruments to assess ADHD, such as the Diagnostic Interview for ADHD in Adults (DIVA) or the Wender Utah Rating Scale (WURS), as well as other appropriate diagnostic interviews (Vallejo-Valdivielso et al., 2019), instead of assessments merely based on non-structured, clinical evaluations or on clinical charts/databases review. Second, since a diagnosis of ADHD during manic or depressive phases is not recommended (Skirrow et al., 2012), we checked whether participants were tested in euthymic/remission phase.
Data analysis
Different meta-analyses were carried out on each correlate for which data were available from at least five different studies. When two or more studies had partial or entire sample overlap for a specific correlate, the study with the largest amount of information was included in the analysis. Meta-analyses of the association between ADHD and relevant correlates were based on odds ratio (OR) with 95% confidence interval (CI) and standardized mean difference (SMD) with 95% CI, for categorical and continuous variables, respectively. Pooled estimates were obtained by weighting each study according to a random-effects model for meta-analysis. Heterogeneity across studies was evaluated using standard cut-offs for I2 statistic (Higgins et al., 2003).
Publication bias was assessed using Egger’s test for correlates based on at least 10 studies (Page et al., 2021b), with the related funnel plot based on the trim-and-fill method (Duval and Tweedie, 2000) for analyses showing a p-value <0.10. Heterogeneity-based sequential sensitivity analyses were performed for analyses with significant findings based on I2 > 50% and at least 10 studies, leaving out the minimum number of studies up to an I2 value below the desired preset threshold of 50% (Patsopoulos et al., 2008). Random-effects meta-regression analyses were carried out to test whether the different timeframes considered for the diagnosis of ADHD (current or past) influenced the effect estimates. Moreover, sensitivity analyses were carried out, sequentially excluding studies with low quality in each of the considered items (age and gender comparability, representativeness, appropriate methods for ADHD diagnosis). Analyses were performed in Stata Statistical Software, Release 17 (StataCorp LLC: College Station, TX).
Grading of the evidence
For each correlate found to be significant, we critically appraised the quality of evidence, classifying it as very high, high, moderate, low or very low, according to relevant upgrades or downgrades in considered items: magnitude of the effect, risk of bias, precision of effect size, consistency of findings and publication bias (Schünemann et al., 2021). The magnitude of the effect was evaluated according to the standard cut-offs for SMD magnitude (0.2 small, 0.5 medium, 0.8 large and 1.3 very large) (Rosenthal, 1996); to evaluate the effect magnitude for ORs, these were converted to effect sizes dividing the ln(OR) by 1.81 (Chinn, 2000). We upgraded the quality of evidence by one level if the effect magnitude was large or very large, whereas we downgraded it by one level if the magnitude was small. The effect of risk of bias on findings was evaluated by verifying whether sensitivity analyses excluding studies with lower quality in each evaluated item (age comparability, gender comparability, representativeness and ADHD diagnosis) were consistent with the findings of the overall analysis, downgrading by one level if at least one quality-based sensitivity analysis was not consistent with the overall analysis. Precision was evaluated by checking whether the 95% CI was <0.4 of effect size, downgrading the quality of evidence by one level if 95% CI was ⩾0.4. Consistency was evaluated by verifying whether the value of I2 was lower than 50%. For variables including less than 10 studies, we downgraded by one level the quality of evidence if the I2 was ⩾50%; for variables including at least 10 studies, we downgraded by one level if the I2 was ⩾50% and the result of the heterogeneity-based sensitivity analysis was not consistent with that of the overall analysis. Finally, publication bias was evaluated according to Egger’s test (Egger et al., 1997) for variables with at least 10 studies (Page et al., 2021b), downgrading by one level if (1) less than 10 studies were included or (2) Egger’s test p-value was <0.10 and the trim-and-fill method showed inconsistency with the findings of the overall analysis (Duval and Tweedie, 2000).
Results
Screening and study characteristics
The study selection process is described in Figure 1. Our systematic search generated 2975 unique articles. The screening of titles and abstracts identified 94 potentially eligible studies. After a full-text revision, 43 studies, based on 38 independent samples, met the eligibility criteria and were included in the meta-analysis (Aksoy et al., 2010; Angst et al., 2013; Ateşci et al., 2010; Bener et al., 2016; Bennett et al., 2019; Berkol et al., 2014; Bernardi et al., 2010; Biederman et al., 2008; Di Nicola et al., 2014; Ghaffary et al., 2013; Gomes et al., 2019; Grigoroiu-Serbanescu et al., 2020; Harmanci et al., 2016; Hegarty et al., 2012; Henin et al., 2007; Jhanda et al., 2018; Karaahmet et al., 2013; Karanti et al., 2020; Koc and Kesebir, 2014; Lan et al., 2015; Makris et al., 2012; Martz et al., 2021; McIntyre et al., 2010; Merikangas et al., 2011; Nierenberg et al., 2005; Oğuz et al., 2014; Pattanshetti et al., 2016; Perroud et al., 2014; Perugi et al., 2013; Pinna et al., 2019; Prunas et al., 2019; Richard-Lepouriel et al., 2019; Rydén et al., 2009a, 2009b; Sachs et al., 2000; Salarvan et al., 2019; Sentissi et al., 2008; Tamam et al., 2008; Torres et al., 2018; Torres et al., 2015; Townsend et al., 2013; Tüysüzoğlu et al., 2011; Walsh et al., 2020). Full search strategy, search result and screening results are described in Supplement 1. The list of the articles excluded after full-text review is reported in Supplement 2. The quality assessment of the studies included in the meta-analysis is reported in Supplement 3. The characteristics of the included studies are reported in Table 1. Additional data are available upon request by contacting the corresponding author.
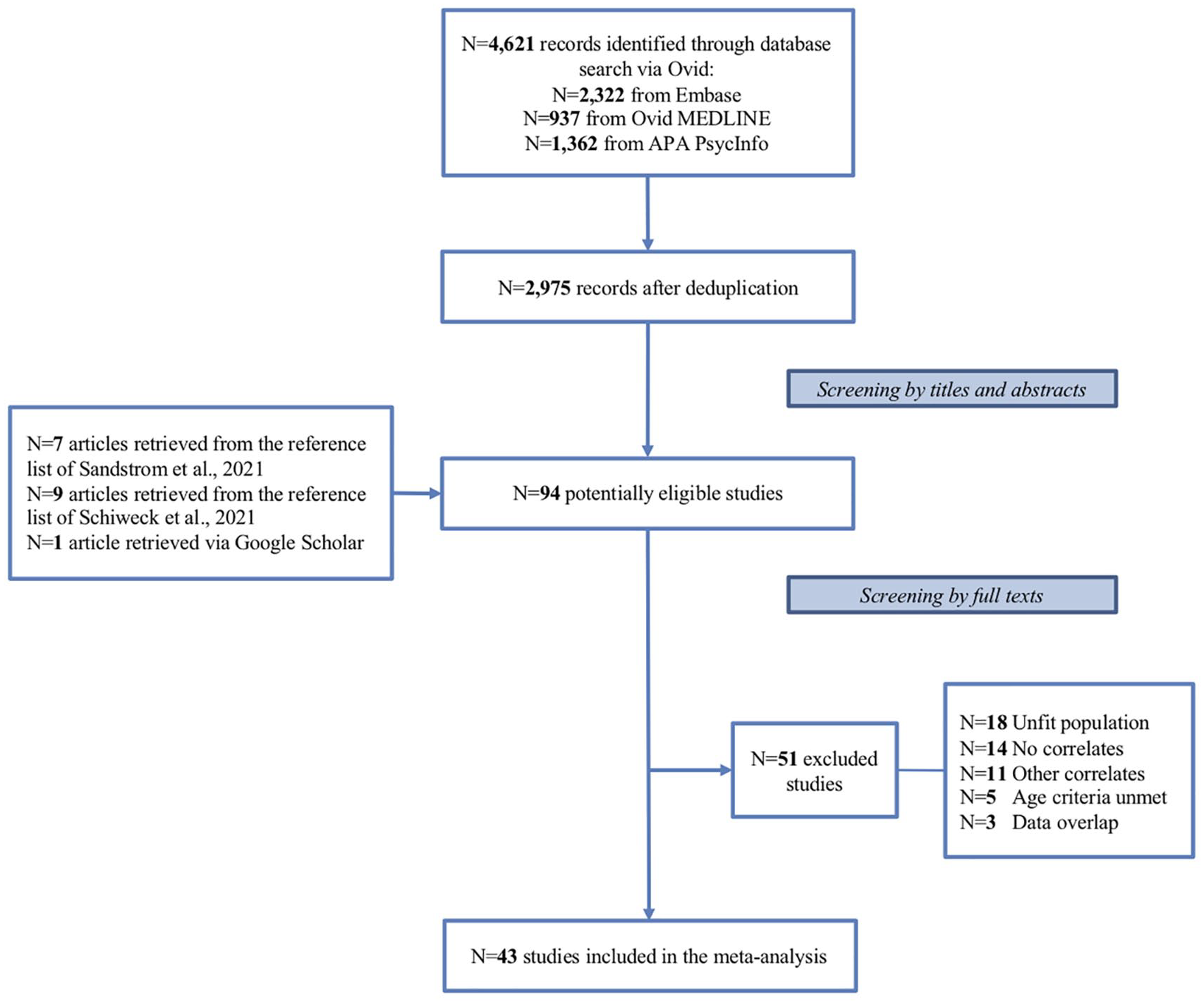
Flowchart of the inclusion process.
Characteristics of studies included in the meta-analysis.
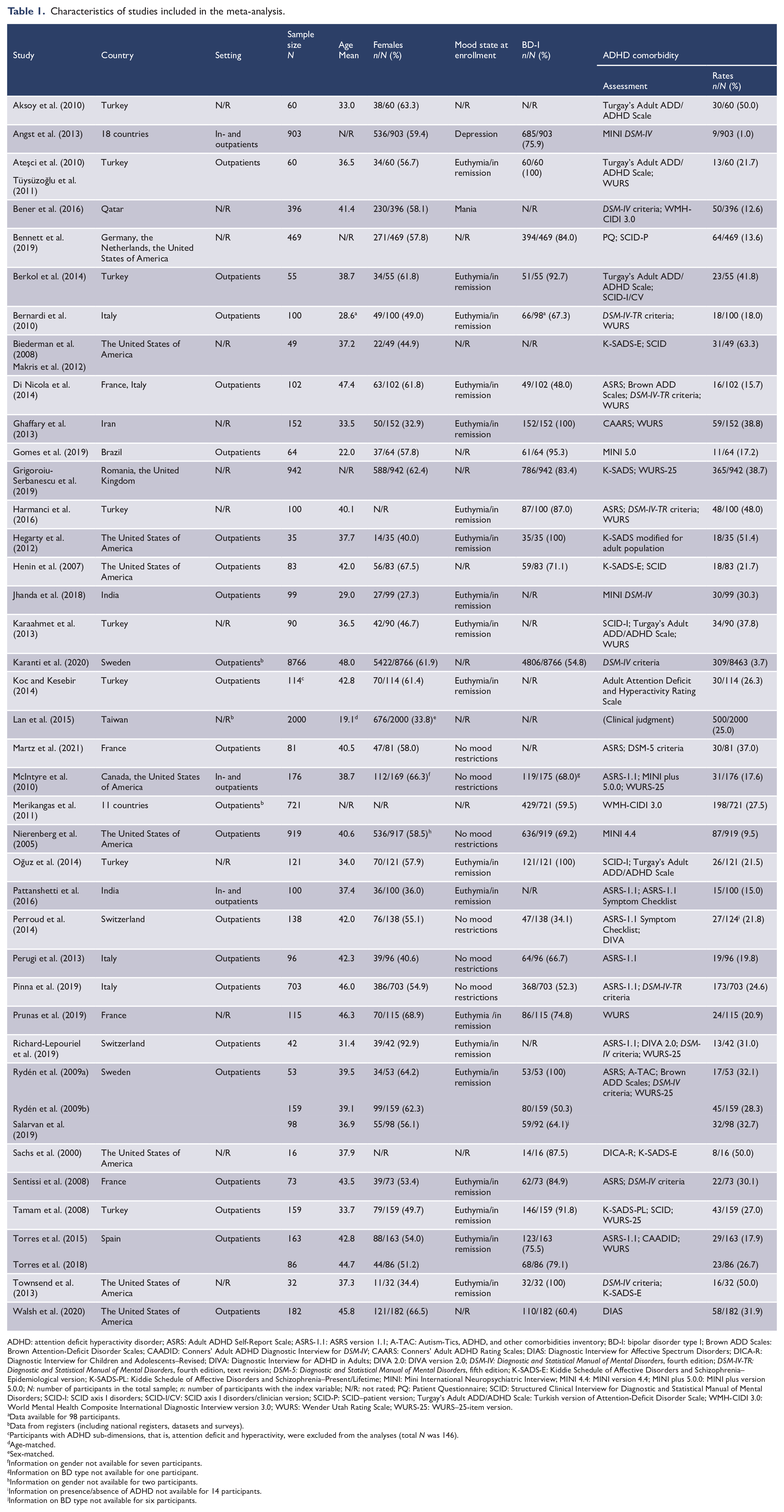
ADHD: attention deficit hyperactivity disorder; ASRS: Adult ADHD Self-Report Scale; ASRS-1.1: ASRS version 1.1; A-TAC: Autism-Tics, ADHD, and other comorbidities inventory; BD-I: bipolar disorder type I; Brown ADD Scales: Brown Attention-Deficit Disorder Scales; CAADID: Conners’ Adult ADHD Diagnostic Interview for DSM-IV; CAARS: Conners’ Adult ADHD Rating Scales; DIAS: Diagnostic Interview for Affective Spectrum Disorders; DICA-R: Diagnostic Interview for Children and Adolescents–Revised; DIVA: Diagnostic Interview for ADHD in Adults; DIVA 2.0: DIVA version 2.0; DSM-IV: Diagnostic and Statistical Manual of Mental Disorders, fourth edition; DSM-IV-TR: Diagnostic and Statistical Manual of Mental Disorders, fourth edition, text revision; DSM-5: Diagnostic and Statistical Manual of Mental Disorders, fifth edition; K-SADS-E: Kiddie Schedule of Affective Disorders and Schizophrenia–Epidemiological version; K-SADS-PL: Kiddie Schedule of Affective Disorders and Schizophrenia–Present/Lifetime; MINI: Mini International Neuropsychiatric Interview; MINI 4.4: MINI version 4.4; MINI plus 5.0.0: MINI plus version 5.0.0; N: number of participants in the total sample; n: number of participants with the index variable; N/R: not rated; PQ: Patient Questionnaire; SCID: Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders; SCID-I: SCID axis I disorders; SCID-I/CV: SCID axis I disorders/clinician version; SCID-P: SCID–patient version; Turgay’s Adult ADD/ADHD Scale: Turkish version of Attention-Deficit Disorder Scale; WMH-CIDI 3.0: World Mental Health Composite International Diagnostic Interview version 3.0; WURS: Wender Utah Rating Scale; WURS-25: WURS–25-item version.
Data available for 98 participants.
Data from registers (including national registers, datasets and surveys).
Participants with ADHD sub-dimensions, that is, attention deficit and hyperactivity, were excluded from the analyses (total N was 146).
Age-matched.
Sex-matched.
Information on gender not available for seven participants.
Information on BD type not available for one participant.
Information on gender not available for two participants.
Information on presence/absence of ADHD not available for 14 participants.
Information on BD type not available for six participants.
Features associated with ADHD comorbidity in BD
Twenty-nine correlates—including sociodemographic characteristics, history of mood episodes, clinical features, current pharmacological treatment, psychiatric comorbidities and substance use disorders—were meta-analyzed. A summary of findings is reported in Table 2.
Summary of findings.
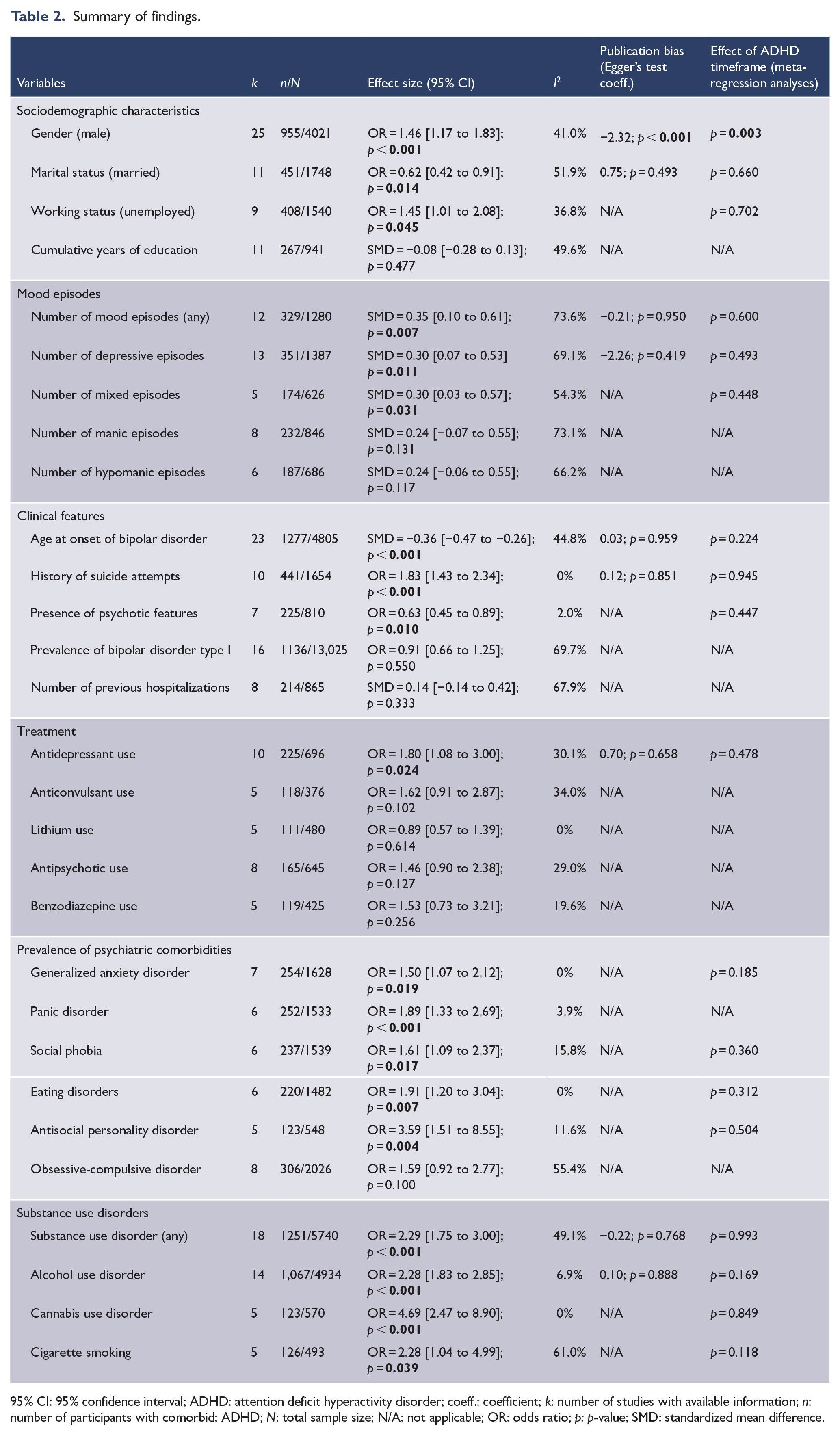
95% CI: 95% confidence interval; ADHD: attention deficit hyperactivity disorder; coeff.: coefficient; k: number of studies with available information; n: number of participants with comorbid; ADHD; N: total sample size; N/A: not applicable; OR: odds ratio; p: p-value; SMD: standardized mean difference.
Sociodemographic characteristics
Data showed that participants with ADHD were more likely males (k = 25; OR = 1.46; 95% CI = [1.17, 1.83]; I2 = 41.0%; Egger’s p < 0.001), even though the association was confirmed only among those with past ADHD according to meta-regression analysis. Moreover, comorbid participants were more likely to be unemployed (k = 9; OR = 1.45; 95% CI = [1.01, 2.08]; I2 = 36.8%) and less likely to be married (k = 11; OR = 0.62; 95% CI = [0.42, 0.91]; I2 = 51.9%; Egger’s p = 0.493), although this finding was not confirmed by the heterogeneity-based sequential sensitivity analysis (k = 10; OR = 0.68; 95% CI = [0.46, 1.02]; p = 0.061; I2 = 40.1%). We did not find any difference in terms of education (k = 11; SMD = −0.08; 95% CI = [–0.28, 0.13]; I2 = 49.6%). Forest plots are shown in Supplementary Figures 1–4 in Supplement 4.
Egger’s test showed probable publication bias for gender (coeff.: −2.32; p < 0.001). However, when applying the trim-and-fill method, the result was consistent with that of the overall analysis (OR = 1.96; 95% CI = [1.56, 2.46]) (Supplementary Figure 5 in Supplement 4). The relevant meta-regression analyses for ADHD timeframe were reported in Table 2. The quality-based sensitivity analyses were reported in Supplement 5.
Mood episodes
ADHD comorbidity was associated with more mood episodes of any polarity (k = 12; SMD = 0.35; 95% CI = [0.10, 0.61]; I2 = 73.6%; Egger’s p = 0.950), depressive episodes (k = 13; SMD = 0.30; 95% CI = [0.07, 0.53]; I2 = 69.1%; Egger’s p = 0.419) and mixed episodes (k = 5; SMD = 0.30; 95% CI = [0.03, 0.57]; p = 0.031; I2 = 54.3%). Sequential sensitivity analyses confirmed the overall findings for both any polarity (k = 9; SMD = 0.56; 95% CI = [0.37, 0.75]; I2 = 34.4%) and depressive episodes (k = 11; SMD = 0.42; 95% CI = [0.24, 0.62]; I2 = 47.5%), respectively. Forest plots are reported in Supplementary Figures 6–8 in Supplement 4. The relevant meta-regression analyses for ADHD timeframe were reported in Table 2. The quality-based sensitivity analyses were reported in Supplement 5.
No significant differences between the two groups were identified by the meta-analyses on the number of previous manic (k = 8; SMD = 0.24; 95% CI = [–0.07, 0.55]; I2 = 73.1%) or hypomanic (k = 6; SMD = 0.24; 95% CI = [–0.06, 0.55]; I2 = 66.2%) episodes (Supplementary Figures 9–10 in Supplement 4).
Clinical features
Participants with ADHD had a lower mean age at BD onset (k = 23; SMD = −0.36; 95% CI = [–0.47, –0.26]; I2 = 44.8%; Egger’s p = 0.959). Moreover, they were more likely to have attempted suicide (k = 10; OR = 1.83; 95% CI = [1.43, 2.34]; I2 = 0%; Egger’s p = 0.851) and had lower odds of having psychotic features (k = 7; OR = 0.63; 95% CI = [0.45, 0.89]; I2 = 2.0%). Finally, no differences were estimated in terms of BD-I diagnosis (k = 16; OR = 0.91; 95% CI = [–0.14, 0.42]; I2 = 69.7%) and number of previous hospitalizations (k = 8; SMD = 0.14; 95% CI = [−0.14 to 0.42]; I2 = 67.9%). Forest plots are shown in Supplementary Figures 11–15 in Supplement 4. The relevant meta-regression analyses for ADHD timeframe were reported in Table 2. The quality-based sensitivity analyses were reported in Supplement 5.
Treatment
Participants with ADHD comorbidity were more frequently treated with antidepressants (k = 10; OR = 1.80; 95% CI = [1.08, 3.00]; I2 = 30.1%; Egger’s p = 0.658) (Supplementary Figure 16 in Supplement 4). Meta-regression analysis based on ADHD diagnosis timeframe and the quality-based sensitivity analyses are reported in Table 2 and Supplement 5, respectively. Conversely, meta-analyses showed that the prescription of anticonvulsants, lithium, antipsychotics and benzodiazepines did not significantly differ between the two groups (Supplementary Figures 17–20 in Supplement 4).
Psychiatric comorbidities
Several additional psychiatric conditions were more likely in participants diagnosed with ADHD: generalized anxiety disorder (k = 7; OR = 1.50; 95% CI = [1.07, 2.12]; I2 = 0%); panic disorder (k = 6; OR = 1.89; 95% CI = [1.33, 2.69]; I2 = 3.9%); social phobia (k = 6; OR = 1.61; 95% CI = [1.09, 2.37]; I2 = 15.8%); eating disorders (k = 6; OR = 1.91; 95% CI = [1.20, 3.04]; I2 = 0%) and antisocial personality disorder (k= 5; OR = 3.59; 95% CI = [1.51, 8.55]; I2 = 11.6%) (Supplementary Figures 21–25 in Supplement 4). No differences in terms of concurrent obsessive-compulsive disorders were estimated (k = 8; OR = 1.59; 95% CI = [0.92, 2.77]; I2 = 55.4%) (Supplementary Figure 26 in Supplement 4). Meta-regression analyses were reported in Table 2 and related quality-based sensitivity analyses in Supplement 5.
Substance use disorders
Participants diagnosed with ADHD were more likely to report any substance use disorders (k = 18; OR = 2.29; 95% CI = [1.75, 3.00]; I2 = 49.1%; Egger’s p = 0.768), alcohol use disorder (k = 14; OR = 2.28; 95% CI = [1.83, 2.85]; I2 = 6.9%; Egger’s p = 0.888), cannabis use disorder (k = 5; OR = 4.69; 95% CI = [2.47, 8.90]; I2 = 0%) and cigarette smoking (k = 5; OR = 2.28; 95% CI = [1.04, 4.99]; I2 = 61.0%). Forest plots are displayed in Supplementary Figures 27–30 in Supplement 4, meta-regression analyses in Table 2 and quality-based sensitivity analyses in Supplement 5.
Grading of the evidence
The overall assessment of the quality of evidence is displayed in Table 3. The body of evidence on the association between putative variables and comorbid ADHD could be deemed of high quality for age at onset of BD and concurrent alcohol or substance use disorders. Evidence of moderate quality was found for a history of suicide attempts. Evidence was of low quality regarding gender (male), number of previous mood episodes of any polarity and concurrent panic, antisocial personality and cannabis use disorders. The remaining body of evidence was deemed of very low quality.
Grading of the evidence.
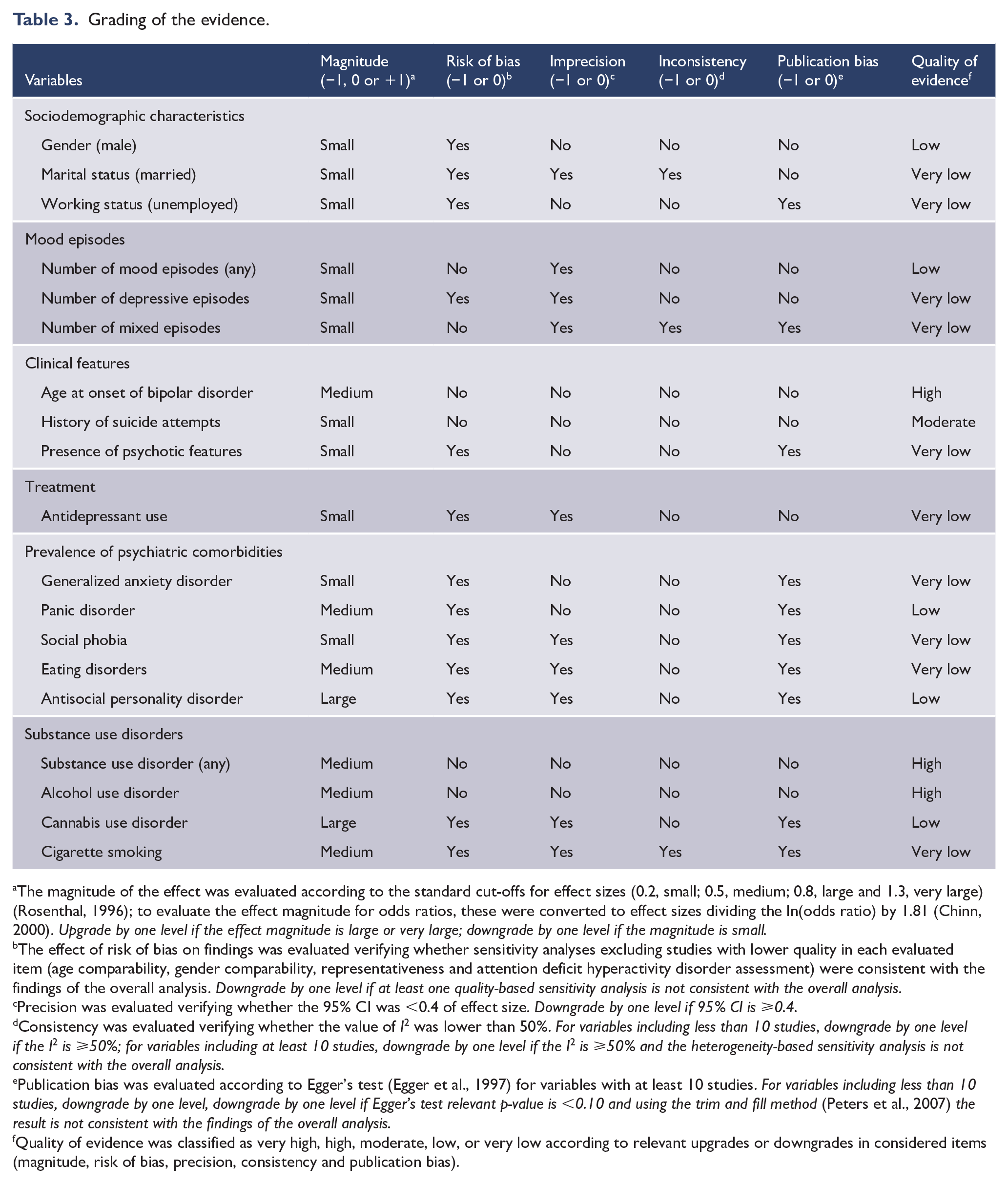
The magnitude of the effect was evaluated according to the standard cut-offs for effect sizes (0.2, small; 0.5, medium; 0.8, large and 1.3, very large) (Rosenthal, 1996); to evaluate the effect magnitude for odds ratios, these were converted to effect sizes dividing the ln(odds ratio) by 1.81 (Chinn, 2000). Upgrade by one level if the effect magnitude is large or very large; downgrade by one level if the magnitude is small.
The effect of risk of bias on findings was evaluated verifying whether sensitivity analyses excluding studies with lower quality in each evaluated item (age comparability, gender comparability, representativeness and attention deficit hyperactivity disorder assessment) were consistent with the findings of the overall analysis. Downgrade by one level if at least one quality-based sensitivity analysis is not consistent with the overall analysis.
Precision was evaluated verifying whether the 95% CI was <0.4 of effect size. Downgrade by one level if 95% CI is ⩾0.4.
Consistency was evaluated verifying whether the value of I2 was lower than 50%. For variables including less than 10 studies, downgrade by one level if the I2 is ⩾50%; for variables including at least 10 studies, downgrade by one level if the I2 is ⩾50% and the heterogeneity-based sensitivity analysis is not consistent with the overall analysis.
Publication bias was evaluated according to Egger’s test (Egger et al., 1997) for variables with at least 10 studies. For variables including less than 10 studies, downgrade by one level, downgrade by one level if Egger’s test relevant p-value is <0.10 and using the trim and fill method (Peters et al., 2007) the result is not consistent with the findings of the overall analysis.
Quality of evidence was classified as very high, high, moderate, low, or very low according to relevant upgrades or downgrades in considered items (magnitude, risk of bias, precision, consistency and publication bias).
Discussion
To our knowledge, this is the first systematic review and meta-analysis aimed at identifying characteristics associated with comorbid ADHD in BD. Based on data from 43 observational studies, including 38 independent samples, we assessed a total of 29 correlates. Our findings showed that study participants with BD and ADHD were more likely to be males, unemployed and not married. Moreover, they showed distinctive clinical features, that is, an earlier onset of BD and a higher number of lifetime mood episodes. In particular, depressive and mixed episodes, attempted suicides and antidepressant treatments were more frequent in comorbid participants, while we estimated lower odds of psychotic features. Furthermore, in terms of additional comorbidities, participants with ADHD diagnosis were also more prone to be affected by antisocial personality disorder, substance or alcohol use disorders and other mental disorders, such as anxiety and eating disorders. All these findings were not influenced by the timeframe considered for ADHD diagnosis (current or past), except for gender. Indeed, the association between ADHD and male gender seemed to hold true only among participants with a past diagnosis of ADHD, which is consistent with epidemiological findings reporting that the male-to-female ratio of ADHD decreases from childhood to adulthood (Moffitt et al., 2015; Williamson and Johnston, 2015).
Our study complements findings of previous meta-analyses estimating the prevalence rates of ADHD in BD (Sandstrom et al., 2021; Schiweck et al., 2021). Nonetheless, it only partially supports the hypothesis that subjects with comorbid ADHD might represent a subpopulation of people with BD with specific clinical features. An appropriate interpretation of our findings needs to consider that the quality of evidence was generally low or very low for the majority of correlates in comorbid ADHD participants, except for earlier BD onset and rates of comorbid alcohol/substance use disorders (high quality) and suicide attempts (moderate quality). The remaining associations were influenced by several issues, such as small magnitude of the effect, risk of bias of included studies, imprecision and inconsistency of overall estimates, as well as an uncertain probability of publication bias. Additional research is certainly needed to substantiate our meta-analytic evidence. Nonetheless, some preliminary explanations could be proposed on the clinical characteristics possibly associated with comorbid ADHD in BD. First, the pattern of inattention and/or hyperactivity/impulsivity superimposed on BD could contribute to the poorer relational, social and occupational functioning of comorbid subjects (Asherson et al., 2014; Michielsen et al., 2015; Strakowski et al., 2010). Similarly, the estimated higher likelihood of concurrent substance use disorder may be due to emotional dysregulation, impulsivity, dysfunctional reward mechanisms and sensation-seeking, all mutual symptoms of BD and ADHD (Asherson et al., 2014; Carrà et al., 2006; Faraone et al., 2019; Lee et al., 2019). Indeed, a common neurobiological substrate shared by BD, ADHD and substance use disorders, characterized by dysfunctions of dopamine in the brain reward circuitry, has been extensively studied (Regnart et al., 2017). Moreover, emotional dysregulation could be accountable also for the higher frequency of mood episodes (especially depressive and mixed ones), the increased likelihood of comorbid anxiety and the higher odds of having attempted suicide among individuals with both BD and ADHD. Consistently, we found antidepressant use to be more frequent in comorbid BD-ADHD individuals, although we estimated no differences in the prescription of other drug classes, namely, lithium, anticonvulsants, antipsychotics and benzodiazepines. Surprisingly, despite its well-proven efficacy for suicide prevention in BD (Bartoli, 2022; Smith and Cipriani, 2017) lithium use was lower than expected, and this is likely to be explained by the general, limited evidence base on treatments for comorbid ADHD in BD (Asherson et al., 2014; Bond et al., 2012). Although the mutual influence between BD and ADHD may suggest the need of specific treatment plans, there is a lack of studies on effective treatments for comorbid BD and ADHD. It has been suggested that an appropriate approach might include a staged treatment, in which first the mood episodes are addressed with mood stabilizers, and only afterwards ADHD symptoms are managed with additional specific medications (Bond et al., 2012; Scheffer, 2007). However, pharmacological strategies should consider the suboptimal treatment responses among individuals with comorbid BD-ADHD (Klassen et al., 2010), possibly linked to the synergistic interaction of these disorders (Hegarty et al., 2012; Townsend et al., 2013). Future research should thus clarify whether comorbid ADHD might affect the efficacy of standard treatments for BD as well as how medications for ADHD might influence the course of BD (Asherson et al., 2014; Girard and Joober, 2017). Moreover, a more comprehensive management considering the biopsychosocial relevance of this comorbidity should include non-pharmacological interventions, like cognitive behavioral therapy, which has been suggested as an effective second-line option in adults with ADHD and BD (Bond et al., 2012). In addition, psychoeducation-based interventions aimed at self-empowerment and coping with symptoms (Schiweck et al., 2021) should be considered.
The findings of this meta-analysis should be interpreted with caution considering some limitations and methodological issues. First, since it is based on cross-sectional data, it is not possible to hypothesize any causal inference. Second, the heterogeneity across studies in terms of study design, setting, included population, mood phase and methods to diagnose ADHD must be taken into account when interpreting our findings. In particular, the identification of ADHD among people with BD from many of the studies included in our meta-analysis might have been influenced by the high variability in terms of diagnostic tools used (structured clinical interviews and self-report rating scales) and by the lack of multiple assessments involving also family members and caregivers (Seixas et al., 2012). Although the general debate about the diagnostic validity of the BD-ADHD comorbidity (Youngstrom et al., 2010), including the limited available recommendations for the diagnosis of ADHD in people with comorbid mental disorders (Pettersson et al., 2018), we were able to sufficiently distinguish quality issues in terms of methods used to detect ADHD. Third, data on several correlates were available only from a limited number of studies, hampering the precision of estimates for several variables. Similarly, some characteristics could not be explored due to the lack of sufficient data from eligible studies, thus preventing a more comprehensive assessment. Fourth, even if we attempted to reduce reporting bias by contacting corresponding authors for additional information, we obtained useful information just for a limited number of studies. While relevant tests showed low probability of publication bias for several variables, we cannot rule out that unpublished findings may have influenced the meta-analytic estimates for some correlates. Finally, in this meta-analysis, we considered BD participants reporting either adult or childhood ADHD. We did not estimate any effect on clinical correlates of the life stage at which ADHD was diagnosed. Nonetheless, we cannot rule out that adult and childhood/adolescent ADHD might simply be two separate clinical entities (Castellanos, 2015; Moffitt et al., 2015) with different clinical presentations in adults with comorbid BD.
As a whole, the findings of this meta-analysis only partially support the hypothesis that a diagnosis of ADHD in adults suffering from BD might identify a specific subgroup of individuals. Participants suffering from both ADHD and BD most likely have earlier BD onset and increased rates of comorbid alcohol/substance use disorders and suicidal behaviors. Nonetheless, further research is needed to identify additional clinical characteristics of this comorbidity, which may also represent peculiar targets of more adequate care programs.
Supplemental Material
sj-docx-1-anp-10.1177_00048674221106669 – Supplemental material for Clinical correlates of comorbid attention deficit hyperactivity disorder in adults suffering from bipolar disorder: A meta-analysis
Supplemental material, sj-docx-1-anp-10.1177_00048674221106669 for Clinical correlates of comorbid attention deficit hyperactivity disorder in adults suffering from bipolar disorder: A meta-analysis by Francesco Bartoli, Tommaso Callovini, Daniele Cavaleri, Riccardo Matteo Cioni, Bianca Bachi, Angela Calabrese, Federico Moretti, Aurelia Canestro, Marco Morreale, Christian Nasti, Dario Palpella, Susanna Piacenti, Renata Nacinovich, Ilaria Riboldi, Cristina Crocamo and Giuseppe Carrà in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors thank Reza Bidaki (Ghaffary et al., 2013), Sandeep Grover (Jhanda et al., 2018), Alina Karanti et al. (2020), Ismail Koc (Koc and Kesebir, 2014), Nader Perroud et al. (2014) and ![]() for having provided additional information regarding their articles.
for having provided additional information regarding their articles.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
