Abstract
Deep brain stimulation is an emerging therapy for treatment-refractory obsessive-compulsive disorder patients. Yet, accessibility is limited, treatment protocols are heterogeneous and there is no guideline or consensus on the best practices. Here, we combine evidence from scientific investigations, expert opinions and our clinical expertise to propose several clinical recommendations from the pre-operative, surgical and post-operative phases of deep brain stimulation care for treatment-refractory obsessive-compulsive disorder patients. A person-centered and biopsychosocial approach is adopted. Briefly, we discuss clinical characteristics associated with response, the use of improved educational materials, an evaluative consent process, comprehensive programming by an expert clinician, a more global assessment of treatment efficacy, multi-disciplinary adjunct psychotherapy and the importance of peer support programs. Furthermore, where gaps are identified, future research suggestions are made, including connectome surgical targeting, scientific evaluation of hardware models and health economic data. In addition, we encourage collaborative groups of data and knowledge sharing by way of a clinical registry and a peer group of programming clinicians. We aim to commence a discussion on the determinants of deep brain stimulation efficacy for treatment-refractory obsessive-compulsive disorder patients, a rare and severe patient group, and contribute to more standardized and evidence-based practices.
Keywords
Deep brain stimulation (DBS) is a form of focal neurostimulation that modulates electrical activity in subcortical structures through the implantation of permanent stimulating electrodes. DBS for treatment-refractory obsessive-compulsive disorder (TR-OCD) can be accessed under a humanitarian exemption in the United States and Europe, yet remains unapproved in Australia, despite sufficient evidence for regulatory approval (Acevedo and Rossell, 2022; Mosley et al., 2022). The proposed changes to the Royal Australian and New Zealand College of Psychiatrists clinical memorandum on DBS for psychiatric conditions indicate that DBS for OCD may be acceptable outside of a clinical trial if performed by an appropriately experienced multi-disciplinary team, with rigorous assessments and monitoring. Yet, treatment accessibility issues remain, with no definitive referral processes and poor awareness of DBS throughout psychiatric services. Furthermore, the Mental Health Act prohibits DBS for OCD in New South Wales, and the Northern Territory. In our recent systematic review (Acevedo et al., 2021), we identified several disparities in treatment approaches, which can confound patient outcomes, highlighting the need for standardization. Currently, there are no clinical guidelines or expert consensus outlining best practice in managing OCD DBS patients. In this Viewpoint, we combine scientific evidence with our clinical experience, to present recommendations for each stage of the care process for people with OCD undergoing DBS, informed by biopsychosocial and person-centered approaches. In addition, at each stage, we make recommendations for further research to improve future evidence-based practices.
Pre-operative
The pre-operative phase should encompass a full psychiatric and medical workup, patient and carer education and informed consent. The precise definition of ‘treatment resistance’ for the screening of DBS varies slightly across settings, but usually includes failure of ⩾2 selective serotonin reuptake inhibitors, 1 trial of clomipramine, ⩾1 trial of augmentation with an atypical antipsychotic and one complete course of cognitive behavioral therapy (CBT) incorporating the principles of exposure and response prevention (ERP).
Screening
It is established that some psychiatric comorbidities may interfere with the therapeutic response of DBS or increase the risk of psychiatric adverse events; common exclusions include bipolar disorder, schizophrenia and severe autism spectrum disorder. Later age of onset and greater insight into illness have been associated with better clinical response. Particular symptom domains (hoarding and symmetry), comorbid personality disorder and perfectionism are associated with poor response (Alonso et al., 2015; Denys et al., 2010; Graat et al., 2021). Having said this, these clinical and demographic factors have low predictive validity at the individual level and should not of themselves serve as exclusion criteria.
Patients pursuing DBS for OCD arguably have insight that they have OCD and have a need for treatment but have varying levels of insight into their symptoms vis-à-vis ego-syntonic and ego-dystonic aspects of OCD. For example, perfectionism, symmetry and hoarding are commonly appraised as ego-syntonic by patients, which can blunt the effectiveness of post-operative ERP and may explain the association with limited efficacy. Symptom insight should be investigated further to better classify what components are related to response, and whether a marker of insight can be used as a baseline predictive tool.
Despite extensive investigation, baseline clinical predictors are not yet established, creating uncertainty as to who would benefit most. Experienced teams are broadening their samples to include severe psychiatric comorbidities (Denys et al., 2020). Therefore, comorbidities, OCD subtypes, age of onset and level of insight should be considered as part of a patient’s complete profile. We suggest that a collaborative registry is required to achieve sufficiently powered investigations to refine predictors of response. This does not necessarily mean that people with a poorer prognostic profile would be precluded from DBS, but such knowledge will allow more nuanced education about potential risks and benefits. A registry with longitudinal data will allow for the refinement of selection guidelines.
Education
Unrealistic expectations can negatively impact satisfaction post-DBS and are correlated with poor psychosocial adjustment and functional outcomes (in Parkinson’s disease and epilepsy, no identified data in OCD) (Hasegawa et al., 2014; Maier et al., 2016). Furthermore, despite expressing realistic expectations, patients may hope for more (Thomson et al., 2021). Both family members and patients should not expect a cure, but be well informed of, and committed to, the post-operative regimen (as described below).
Studies in major depressive disorder (MDD) have demonstrated that DBS patients generally report realistic expectations of outcomes, but they also (retrospectively) identified difficulties with attending to, processing and retaining the educational material presented as part of the consent process (Thomson et al., 2021). In our experience with OCD, compulsions can interrupt consenting discussions, and repeated questioning suggests poor retention. Thus, a method of education involving verbal and visual shared decision-making materials, case vignettes and evaluation of the delivered information across several sessions is recommended. A video of a DBS patients discussing their experiences paired with a booklet of visual statistics on the likelihood of various benefits and risks would theoretically enhance retention. Furthermore, patients should receive education on practical and maintenance aspects of the DBS hardware both pre- and post-operatively; for example, many patients do not appreciate the amount of time required for charging their battery.
Previously, simple but effective educational tools (booklet, interactive app) for DBS have shown value in pre-operative decision-making, follow-up satisfaction and addressing unrealistic hopes (Dinkelbach et al., 2017; Yen et al., 2021). Information delivered by other DBS patients may have a stronger impact in preparing individuals for personally difficult experiences during recovery (Dinkelbach et al., 2017; Thomson et al., 2021).
Consent
Neuropsychological difficulties and desperation associated with disease progression can render informed consent challenging. However, psychiatric patients should not be deemed incapable of decision-making and denied treatment, rather precautions should be implemented to ensure ethical principles are met, particularly autonomy and beneficence. Informed consent requires (1) disclosure of all medically relevant information about diagnosis, prognosis and therapy; (2) adequate mental capacity to understand their situation and the information provided; and (3) autonomous decision-making.
The only identified consent tool used in OCD DBS patients includes a revised version of the Informed Consent Evaluation Feedback Tool (ICEFT-R) by McLaughlin et al. (2021), which assesses understanding of the purpose, benefits, risks and voluntary nature of the research. An updated version of the ICEFT-R assessing expectations and autonomy could provide another safeguard in addition to the mental health tribunal (or equivalent) review, against neuroethical concerns.
Surgery
The surgical procedure involves implantation of electrodes within striatal or basal ganglia regions. Electrodes are inserted based on a pre-operative stereotactic magnetic resonance imaging trajectory; test stimulation and behavioral observations are conducted to assess preliminary efficacy and side effects. Once the electrode placement is confirmed, an implantable pulse generator is inserted into the chest, and patients undergo a period of recovery prior to activation of stimulation.
Stimulation location
The stimulation location is the most important determinant of clinical efficacy, yet there are no agreed criteria to select a target according to the characteristics of the patient. Often, the target is selected based on the neurosurgeons’ experience, as millimeter precision is critical.
Different targets can achieve similar efficacy through modulation of the same white matter network. White matter connectivity (but not anatomical location of electrodes) can predict individual response through modulation of a shared network (Baldermann et al., 2019; Li et al., 2020; Mosley et al., 2021; Smith et al., 2021) and stimulation of a particular tract within the striatum that likely modulates neighboring structures (Morishita et al., 2014). Each surgical target can be thought of as a ‘gateway’ into a network (rather than a single region) and may have different clinical effects depending on individual connectivity patterns. Connectome (whole brain connectivity) analysis is the only method to demonstrate individual predictive ability and has shown utility in surgical targeting in MDD patients (Riva-Posse et al., 2018). Therefore, connectome surgical targeting should be implemented in future OCD research in line with the expertise of the surgical team.
Hardware
Neurosurgical teams implant several DBS models from different device companies (e.g. Medtronic, Abbott, Boston Scientific), and we are aware of no formal comparison of this hardware in terms of efficacy and safety in OCD. The Medtronic 3389 model has been used in slightly more cohorts and recent investigations, yet there is no apparent preference or superiority of a particular model for a chosen target. Considering the connectome approach, in which several closely neighboring targets can modulate a shared network, a greater span may be considered preferable to provide access to different ‘gateways’. Formal scientific evaluation of hardware models could inform best practice.
Due to relatively high stimulation amplitude in OCD patients, battery depletion occurs approximately every 14 months (Denys et al., 2020; Ooms et al., 2017) and requires additional surgery and costs. Patients can experience severe distress around the possibility of stimulation turning off and may show dramatic symptom rebound when it does occur. Despite higher initial costs, rechargeable batteries are more cost-effective in the long term (Ooms et al., 2017) and help prevent interruptions to therapy. Rechargeable batteries are now standard practice and wireless rechargeable devices should be offered to all patients in order to avoid continuous surgery and laborious charging. However, there is limited accessibility to wireless rechargeable devices in Australia, and external recharging can become an intrusion on patient’s lives.
Post-operative
Programming
DBS parameters (amplitude, pulse width, frequency, location) need to be refined throughout the follow-up management and may have varying effects on the neural elements depending on the surgical target. Standard values have largely been adopted from movement disorder applications and may not be optimal for psychiatric conditions. Refinement of stimulation parameters, particularly the location, is a critical factor in achieving the best outcomes (Acevedo et al., 2021). Currently, the only established method is the Montgomery algorithm (Montgomery, 2010), which is not condition-specific, and deviations may be necessary when managing difficult cases, which comes with practical experience and cannot be described in a series of simple steps. An expert guideline (van Westen et al., 2021) and literature review (Morishita et al., 2014) on programming for OCD patients have proposed several practical suggestions for managing different behavioral responses, as well as non-response, in the context of neurocircuitry. However, there is a need for an expert consensus on DBS programming for OCD and advice on complex scenarios (Holland et al., 2020; Morishita et al., 2014), such as those available for movement disorders (e.g. Picillo et al., 2016). Table 1 outlines features that will need to be addressed in such guidelines.
Features of proposed guidelines for stimulation parameters for DBS in OCD.

DBS: deep brain stimulation; OCD: obsessive-compulsive disorder.
Scientific evaluations have rarely reported on programming methods for DBS in OCD (Kahn et al., 2021; Widge and Dougherty, 2015), thus direct evaluation of stimulation parameters will provide empirical evidence to support expert guidelines (e.g. Okun et al., 2007). We encourage a collaborative peer group between experienced programming clinicians and trainees across hospital sites and disciplines. Also, establishing a criterion for the minimum level of experience for programming clinicians would ensure best practices are followed.
DBS can modulate anxiety almost immediately, yet the effect on compulsions and avoidance may take weeks to months, and obsessive-compulsive symptoms are context-dependent. During the initial programming phase, we suggest that re-assessment occurs every 2 weeks. This is because some patients experience cognitive/affective side effects, particularly hypomania, and frequent monitoring for side effects is necessary. Also, frequent assessments allow a continuous titration of therapy, in order to reach the optimal stimulation parameters in an efficient time frame. Although the majority of patients reach response in 6 months, around 25% of cases may take up to 20 months to do so (van Westen et al., 2021); even then, fluctuations in the patient’s state may require ongoing fine-tuning. Thus, during this optimization phase of DBS, programming should be dependent on the clinician’s discretion and the patient’s mental state.
Some centers commence programming based on a pre-defined contact (e.g. most dorsal location). However, the precise location of the lead can deviate from the intended location, depending on the surgical procedure. Thus, consideration of post-operative neuroimaging to identify the contact with the closest proximity to the anatomical target (or circuit) may fast track the programming process and allow patients to receive benefit with less programming burden. Future applications may improve personalized care by the development of programming methods based on symptom subtypes. In addition, patients sometimes desire to have greater control over DBS programming or experience fluctuations in symptoms; the ability to switch between stimulation modes would provide greater flexibility in these instances.
Assessment of efficacy
While the Yale-Brown Obsessive-Compulsive Scale (YBOCS) is the ‘gold standard’ rating scale for obsessive and compulsive symptomatology, it is arguably not adequate for assessing the broader effects of DBS in OCD patients (De Haan et al., 2015; van Westen et al., 2021). It has been argued that the ability to achieve personally valuable goals related to psychosocial dimensions should be prioritized in addition to symptomatic improvement, identifying a need for patient-reported and objective functional outcomes to be assessed (Synofzik and Schlaepfer, 2008). Recent trial protocols are recognizing the complexities of DBS and incorporating several primary endpoints including social and occupational functioning (McLaughlin et al., 2021). Non-responders (according to the YBOCS) often have marked improvement in comorbid symptoms and quality of life and thus continue DBS (Graat et al., 2021). Furthermore, the effects of DBS are not limited to primary OCD or comorbid symptoms. Thus, the patient and clinician perspective may be incongruent; Parkinson’s DBS patient satisfaction is correlated with determinants of health rather than symptomatic improvement (Nam et al., 2020). Therefore, we suggest assessment of outcomes from DBS for OCD encompasses symptomatic and functional domains, using both objective and subjective measures.
Multi-modal clinical intervention
While DBS alone may result in a significant improvement in symptom burden for some patients, for many further improvements are possible with the addition of psychological and social therapies. Evidence-based psychological therapy consolidates and augments DBS effects, therein the journey begins. DBS weakens the association between obsessions and anxiety, and CBT, particularly ERP is necessary to break habitual rituals (Mantione et al., 2014). As soon as the patient can engage with the clinician for ERP, they should; engagement can be used as a marker that DBS is effective.
Commitment from family/carers is necessary to ensure they are not enabling the OCD symptoms. Families and carers of OCD DBS patients can experience profound transitions from a life consumed by a co-dependent relationship as a caregiver, into normality. Thus, patients and family members may experience ‘burden of normality’ through recovery from a life consumed by OCD (Bosanac et al., 2018), and appropriate psychotherapy, family therapy, psychoeducation and peer support should be offered.
Within our cohort, we found that consolidation of exposure therapy in the home setting was necessary to target rituals, which over years of living with OCD can become habitual. Although DBS may allow an individual to engage in previously intolerable behaviors, they will not do so unless deeply engrained ‘coping’ mechanisms are mitigated—a process that may take years. It is well established that ERP is necessary to optimize DBS, yet a more personalized model of care has not been described.
We propose adjunct therapy beyond pure CBT, involving an eclectic approach from evidence-based treatments including (but not limited to) exposure response programs, dialectic behavioral therapy, acceptance and commitment therapy and psychoeducation. Some individuals may also require a trauma informed care (TIC) approach to address hypervigilance and hyperarousal. Also, active support in naturalistic settings and consideration of biopsychosocial determinants is important. It is also important to shift the patient’s insight to understand pathological and habitual rituals, gain trust in the self and an awareness that recovery is achievable. To set the stage for this post-implantation work, we suggest it is beneficial to meet with a mental health clinician pre-operatively to commence building rapport and trust, establish baseline severity and identify and set realistic functional goals. Similarly, it is important to assess the psychosocial environment pre-operatively to inform follow-up management (Schläpfer et al., 2021). Collectively, this approach may improve psychosocial outcomes and difficulties faced through the burden of normality syndrome.
Peer support
Within mental health services, peer support promotes hope and the belief that recovery is possible; it can also help build self-esteem and enhance self-efficacy and self-management of social scenarios (Repper and Carter, 2011). Stories of hope are powerful, and often necessary to apprise and facilitate a reappraisal within OCD patients that recovery is possible. A peer support service from the pre-operative to long-term follow-up could assist in decision-making, goal setting, alleviation of fears and psychosocial adjustments. Peer support is an evidence-based model; core values and principles are common across models (Watson, 2019), and toolkits can assist in the implementation of these at the individual and organization level. The empirical testing of a peer support model for OCD patients undergoing DBS is needed.
Conclusion
In this Viewpoint, several recommendations for the management of OCD DBS patients are proposed; a summary is provided in Table 2. Importantly, we encourage pre-operative education through accessible and multimodal tools; comprehensive programming and psychotherapy by a multi-disciplinary and collaborative approach to targeting symptomology, and psychosocial functioning; peer support programs for patients and family members; and collaborative initiatives to integrate data and share knowledge, including a registry to increase robustness of data and translational outcomes. We also highlight the importance of a person-centered approach in the pursuit of precision medicine. DBS requires significant resources and investment from the clinician and patient yet provides individuals the ability to regain control over their lives. Clinical and research protocols should be optimized and standardized, to ensure resources are appropriately allocated and the best possible outcomes are achieved. Future applications should incorporate tractography-guided surgical targeting, with appropriate scientific evaluation. Additional research and health economic data are needed to understand the value of these recommendations.
Recommendations for OCD DBS patient care: best practices and areas where more research is needed.
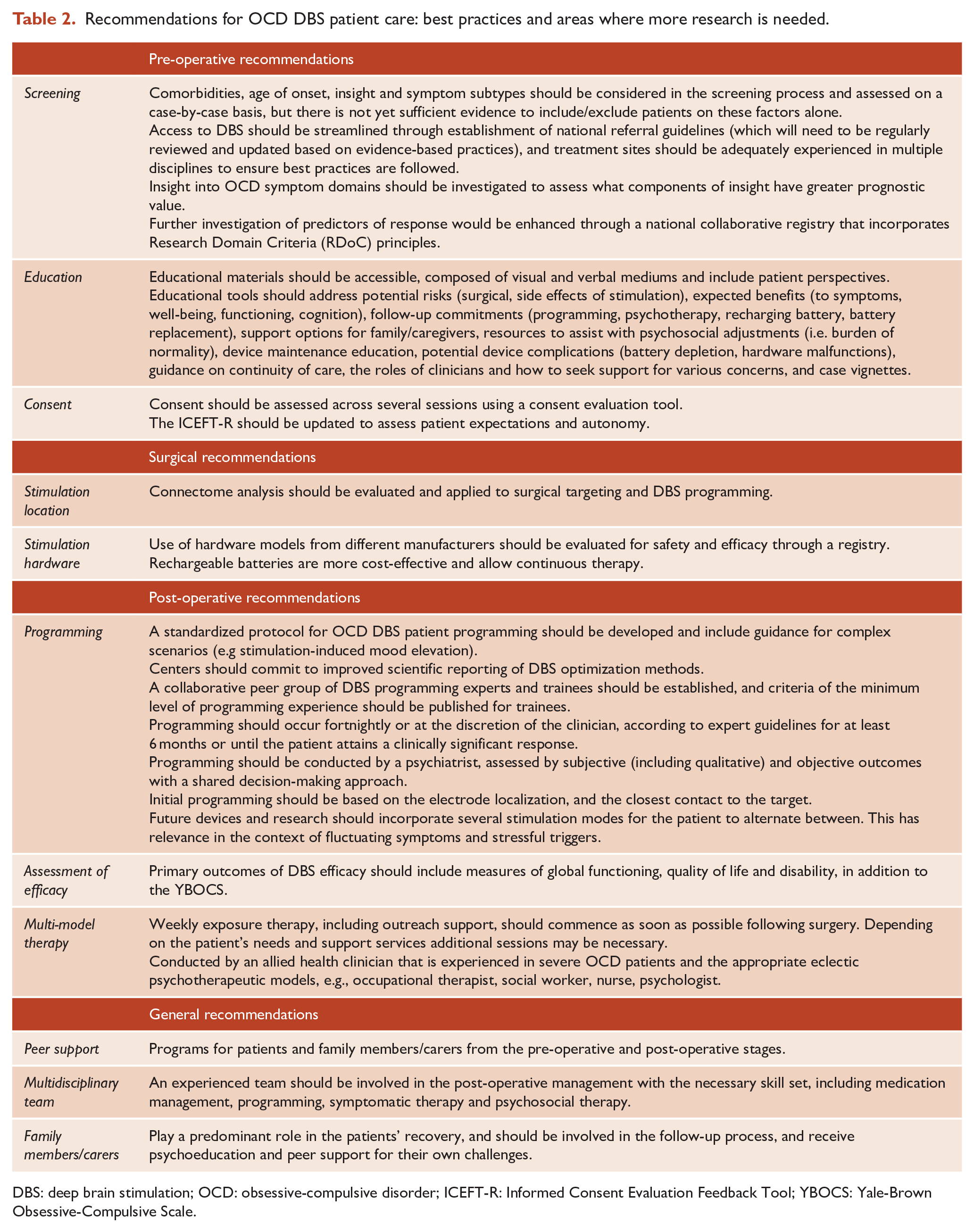
DBS: deep brain stimulation; OCD: obsessive-compulsive disorder; ICEFT-R: Informed Consent Evaluation Feedback Tool; YBOCS: Yale-Brown Obsessive-Compulsive Scale.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported the Australian Government Research Training Program Scholarship, Senior National Health and Medical Research Council (NHMRC) Fellowship (GNT1154651) and perpetual IMPACT philanthropy grant (IPAP2017/1439).
