Abstract
Objective:
Deep brain stimulation can be of benefit in carefully selected patients with severe intractable obsessive-compulsive disorder. The aim of this paper is to describe the outcomes of the first seven deep brain stimulation procedures for obsessive-compulsive disorder undertaken at the Neuropsychiatry Unit, Royal Melbourne Hospital. The primary objective was to assess the response to deep brain stimulation treatment utilising the Yale-Brown Obsessive Compulsive Scale as a measure of symptom severity. Secondary objectives include assessment of depression and anxiety, as well as socio-occupational functioning.
Methods:
Patients with severe obsessive-compulsive disorder were referred by their treating psychiatrist for assessment of their suitability for deep brain stimulation. Following successful application to the Psychosurgery Review Board, patients proceeded to have deep brain stimulation electrodes implanted in either bilateral nucleus accumbens or bed nucleus of stria terminalis. Clinical assessment and symptom rating scales were undertaken pre- and post-operatively at 6- to 8-week intervals. Rating scales used included the Yale-Brown Obsessive Compulsive Scale, Obsessive Compulsive Inventory, Depression Anxiety Stress Scale and Social and Occupational Functioning Assessment Scale.
Results:
Seven patients referred from four states across Australia underwent deep brain stimulation surgery and were followed for a mean of 31 months (range, 8–54 months). The sample included four females and three males, with a mean age of 46 years (range, 37–59 years) and mean duration of obsessive-compulsive disorder of 25 years (range, 15–38 years) at the time of surgery. The time from first assessment to surgery was on average 18 months. All patients showed improvement on symptom severity rating scales. Three patients showed a full response, defined as greater than 35% improvement in Yale-Brown Obsessive Compulsive Scale score, with the remaining showing responses between 7% and 20%.
Conclusion:
Deep brain stimulation was an effective treatment for obsessive-compulsive disorder in these highly selected patients. The extent of the response to deep brain stimulation varied between patients, as well as during the course of treatment for each patient. The results of this series are comparable with the literature, as well as having similar efficacy to ablative psychosurgery techniques such as capsulotomy and cingulotomy. Deep brain stimulation provides advantages over lesional psychosurgery but is more expensive and requires significant multidisciplinary input at all stages, pre- and post-operatively, ideally within a specialised tertiary clinical and/or academic centre. Ongoing research is required to better understand the neurobiological basis for obsessive-compulsive disorder and how this can be manipulated with deep brain stimulation to further improve the efficacy of this emerging treatment.
Introduction
Obsessive-compulsive disorder (OCD) is a neuropsychiatric disorder with a worldwide prevalence of between 1% and 3% (Karno et al., 1988). Although the presentation may vary, it is most commonly characterised by recurrent obsessions and/or compulsions. Obsessions are recurrent and anxiety-provoking thoughts, images or impulses that dominate the thinking of an individual. Compulsions are recurrent mental acts or behaviours that often occur in relation to obsessional cues and can act to reduce the experience of anxiety or distress (American Psychiatric Association, 2013). While historically OCD was viewed as a chronic disorder which was intractable and non-responsive to treatment (Van Balkom et al., 1998), the prognosis has improved with the advent of psychological (exposure response prevention (ERP), cognitive behavioural therapy (CBT)) and pharmacological interventions (clomipramine, selective serotonin reuptake inhibitors (SSRIs) (Catapano et al., 2006; Denys, 2006; Meyer, 1966; Rosa-Alcázar et al., 2008). Despite the availability of these treatments, 20–30% of patients with OCD fail to improve and up to 10% develop a severe intractable form of the illness (Denys, 2006; Rasmussen and Eisen, 1992). Neurosurgical procedures for OCD are reserved for patients within this latter subgroup (Bear et al., 2010; Denys and Mantione, 2009).
The historical use and misuse of psychosurgery for a variety of psychiatric disorders, including schizophrenia, has overshadowed the benefits of stereotactic ablative treatments in patients with severe OCD (Pressman, 1998). ‘Modern’ surgical approaches have included anterior cingulotomy, subcaudate tractotomy, limbic leucotomy and anterior capsulotomy (Bear et al., 2010; Rosenfeld and Lloyd, 1999). Each of these procedures has been reported to be successful in between 30% and 70% of patients with intractable OCD (Feldman et al., 2001). In the largest modern prospective trial of anterior cingulotomy for severe OCD, 44 patients were followed for 32 months (Dougherty et al., 2002). In this study, one-third of patients who underwent the procedure experienced a significant improvement in OCD symptoms as measured by a 35% reduction in the Yale-Brown Obsessive Compulsive Scale (YBOCS).
Deep brain stimulation (DBS) involves the implantation of stimulation electrodes in specific highly localised brain regions with the aim of modifying brain activity. DBS has been extensively used in Parkinson’s disease, chronic pain, intractable tremor and dystonia (Benabid, 2003; Bittar et al., 2005; Kupsch et al., 2003; Rosenow et al., 2004; Visser-Vandewalle et al., 2004), and the first published cases in OCD were in 1999 by Nuttin and colleagues (Nuttin et al., 1999). DBS has a number of significant advantages over traditional lesional procedures, as it is adjustable, controlled, minimally destructive of tissue and reversible. Adverse effects of DBS may be related either to the procedure or stimulation. Procedure-related adverse effects include haemorrhage (1–2% of procedures), seizure induction (approximately 1%, usually in the first 24 hours following implantation) and infection (2–3%) (Benabid, 2003; Greenberg and Rezai, 2003; Hariz, 2002). Stimulation-related side effects, including effects on mood, cognition and sensory motor systems, are dependent on the stimulation site and are reversible with cessation of stimulation (Benabid, 2003; Greenberg and Rezai, 2003; Hariz, 2002).
The anatomical target for early studies of DBS in OCD was based on the known benefits of lesional surgery to the anterior limb of the internal capsule (Abelson et al., 2005; Anderson and Ahmed, 2003; Gabriëls et al., 2003). The promising outcomes of these early studies paved the way for increasingly larger studies of anterior capsule DBS (ACDBS). Gabriëls et al. (2003) found that ACDBS was effective in two-thirds of patients, whose YBOCS scores returned to normal over 33 months (Gabriëls et al., 2003). Using a similar, but slightly more ventral bilateral stimulator placement in the ventral capsule and striatum (VC/VS), Greenberg et al. (2006) followed 10 adult patients for a period of almost 36 months and reported a mean reduction in YBOCS scores from 36 to 22 with an associated improvement in global functioning (Greenberg et al., 2006). In a further follow-up study, Greenberg et al. reported that improved outcomes were found with more posterior lead placement, in the bed nucleus of stria terminalis (BNST; Greenberg et al., 2010). The most influential recent study used DBS of the bilateral nucleus accumbens in 16 patients and followed patients over a 72-week period (Denys et al., 2010). Significant improvements were observed in the majority of patients (mean YBOCS reduced from baseline of 33.7 to 18.0 over 8 months), though 25% of patients had a limited response to the DBS. Adverse effects of the procedure were generally mild with no significant adverse effects reported.
This study’s major aim was to ascertain outcomes of DBS in a selected group of OCD patients, monitoring OCD-related symptoms and level of functioning following DBS, and adverse effects. A secondary aim was to investigate the effect of DBS treatment on depressive and anxiety symptoms seen in OCD.
Methods
Legislative requirements
DBS for OCD is deemed to be a neurosurgical procedure for mental illness, under Section 100 of the Victorian Mental Health Act (State Government of Victoria, 2014). The Psychosurgery Review Board (PRB) has been established in order to ensure that the requirements of the Act are met. DBS cannot proceed without approval by the PRB and applications for DBS for OCD must satisfy the criteria set out by the PRB, including the patient giving informed consent and all other reasonable treatments having been tried without sufficient or long-lasting benefit.
Patient selection
Patients over the age of 18 years were referred to the Neuropsychiatry Unit at Royal Melbourne Hospital by their treating psychiatrist who had specialist OCD experience, for assessment of the suitability of DBS for severe treatment-resistant OCD. This study was not advertised, and patients were referred as part of their clinical treatment algorithm.
Inclusion and exclusion criteria
The assessment was undertaken via a staged process comprising an initial detailed referral from the primary treating psychiatrist and an inpatient assessment. The aim of the assessment process was to confirm the diagnosis and ensure that the criteria set out by the Psychosurgery Review Board of Victoria (PRB) were met. The PRB does not specifically define what constitutes ‘adequate pharmacological or psychological treatments’ for OCD nor does it provide a definition of intractable OCD. The minimum criteria for consideration of DBS for OCD in this cohort have been:
At least 5 years of severe OCD symptoms (YBOCS 28 or greater).
Adequate trials of at least three antidepressants including clomipramine, requiring a minimum of 12 weeks of treatment at maximally tolerated doses.
Augmentation with atypical antipsychotics.
If there was a significant co-morbid major depression with limited response to antidepressant therapy, then augmentation with mood stabilisers, or a trial of electroconvulsive therapy (ECT) and/or transcranial magnetic stimulation (TMS).
Psychological treatments including but not restricted to ERP and CBT. These treatments need to have been undertaken for at least 12 sessions on at least two different occasions.
Exclusion of pre-existing diagnoses of severe mental illness such as bipolar affective disorder or schizophrenia.
These criteria are consistent with other cohorts worldwide (Denys et al., 2010; Greenberg et al., 2006).
Pre-DBS assessments
The key components of an initial assessment are as follows:
Justification for the diagnosis of OCD including detailed review of all available medical, psychological and other opinions.
Exclusion of co-morbid disorders (e.g. substance dependence, personality disorders) that may contribute to the presentation.
Exclusion of a significant physical or neurological disorder that would be contraindicated in DBS.
Chronological history of biological treatments including pharmacological treatments, ECT or TMS.
Review of psychological therapies and neuropsychological testing.
Details of patient’s psychosocial background and general functioning.
The patient must be capable of fully informed consent and must have consented to the procedure.
During a pre-DBS assessment, the following clinical assessments were performed: neuropsychiatric review, neurological review, neuropsychological and psychological assessment, medication review, magnetic resonance imaging (MRI) and single-photon emission computed tomography (SPECT) imaging, electroencephalogram (EEG), as well as routine blood investigations. The patients completed the Neuropsychiatry Unit Cognitive Assessment (NUCOG) (Walterfang et al., 2006); YBOCS (Goodman et al., 1989); Social and Occupational Functioning Assessment Scale (SOFAS) (Morosini et al., 2000); Obsessive Compulsive Inventory (OCI) (Foa et al., 1998); and Depression, Anxiety and Stress Scales (DASS) (Brown et al., 1997).
As part of the assessment, all available clinical information was retrieved including from previous psychologists and psychiatrists. Structured clinical interview was not used; however, all subjects underwent a personality inventory.
Once a DBS neuropsychiatric assessment was completed, the patient, family and treating psychiatrist were informed of the decision to proceed with an application to the PRB. A PRB hearing was held with the patient and family in attendance. DBS can only be offered if approval is granted by the PRB according to the criteria of the local mental health act legislation.
All patients provided informed written consent to the surgical procedure and to the publication of their respective data in research papers. The project was approved by the local research and ethics committee.
Assessment tools
The YBOCS is a clinician-rated scale that provides a total score as a measure of overall OCD symptom severity, as well as an obsession (YBOCS-O) and compulsion (YBOCS-C) subscore. In this study, we used the widely accepted measure of greater than 35% improvement in the YBOCS to define a ‘full response’ to treatment (Denys et al., 2010; Goodman et al., 2010). We have defined a partial response as >15% and <35% improvement on the YBOCS, and non-response as <15% improvement. The OCI is a self-reported measure of 42 questions that measures the frequency and distress of specific symptom subsets (i.e. washing, hoarding). Each question is rated 0–4 for both frequency and distress giving an overall score for each (Foa et al., 1998). The DASS is a brief 21-item self-assessment tool of symptoms of depression, anxiety and stress (Henry and Crawford, 2005). It is not used as a diagnostic tool. The SOFAS is a tool used to measure social and occupational functioning and is based on the clinicians judgement of overall functioning (Morosini et al., 2000). A structured assessment of functioning and quality of life was not undertaken.
DBS surgery
DBS surgery was performed in two stages, with Medtronic 3387 electrodes used in five individuals, and St Judes electrodes in two individuals, at the discretion of the neurosurgeon. Stage one surgery was performed under local anaesthetic with insertion of electrodes using neuronavigational instruments, with fixation of electrodes to the skull. Microelectrode recording and evaluation of intra-operative response to macroelectrode stimulation was undertaken. Stage two surgery involved connection of the electrodes and extension leads to the implantable pulse generator (IPG), which was placed in a pouch fashioned under the pectoral fascia. Non-rechargeable IPGs were used in all cases. Post-operatively (within 48 hours), computed tomography (CT) of the brain was performed, and the images were fused with the pre-operative MRI to assess electrode placement. A summary of neurosurgical details for all seven patients is outlined in Table 1. Figure 1 demonstrates the electrode position for both the nucleus accumbens (NAc) and bed nucleus stria terminalis targets. Targets were chosen in discussion with the treating psychiatrist, neurologist and neurosurgeon, and in the case of Patient 4, targeting was changed intraoperatively due to poor response at the left NAc (See online Supplementary material for individual neurosurgical details).
Demographics at baseline assessment of patients who underwent DBS.
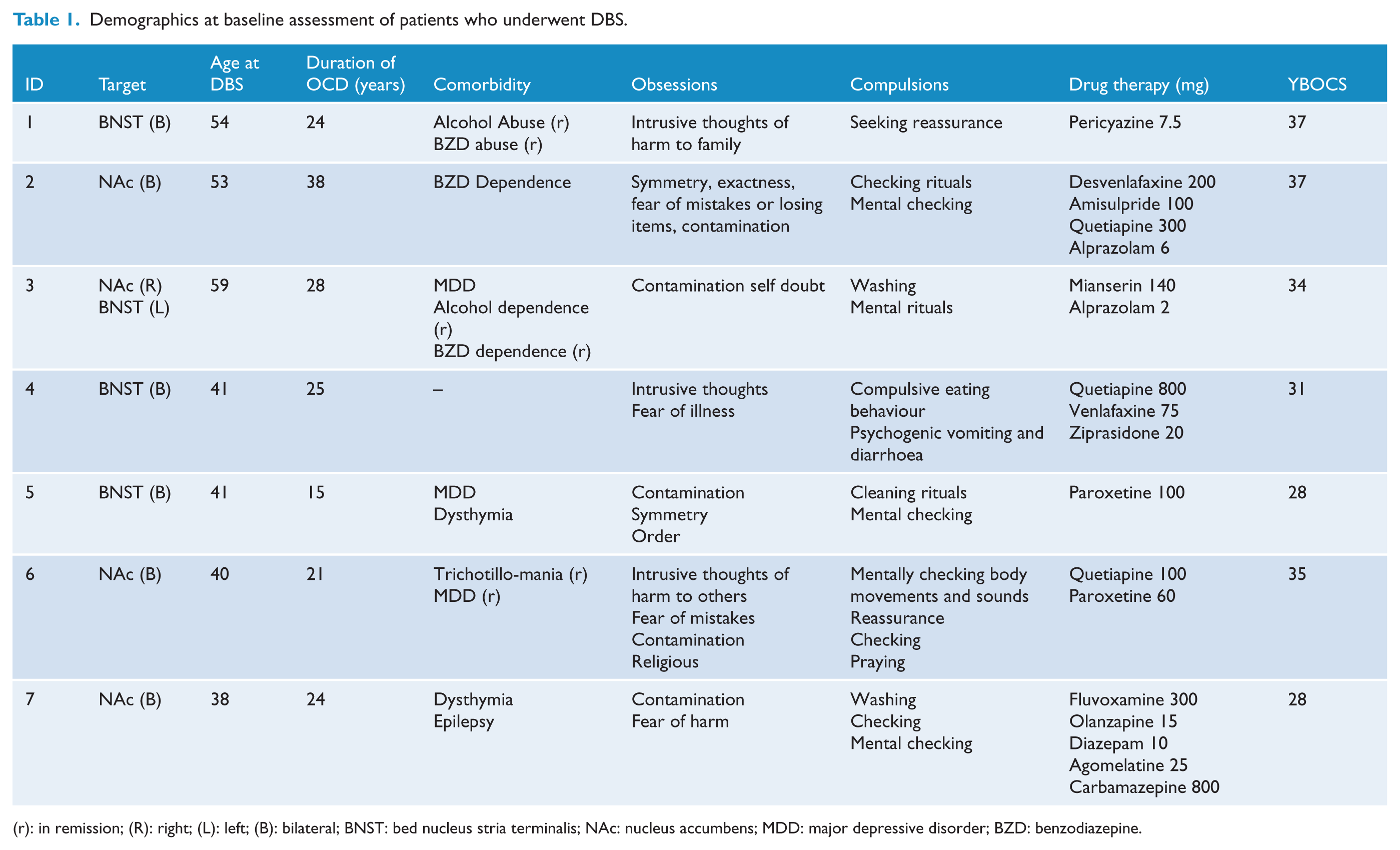
(r): in remission; (R): right; (L): left; (B): bilateral; BNST: bed nucleus stria terminalis; NAc: nucleus accumbens; MDD: major depressive disorder; BZD: benzodiazepine.
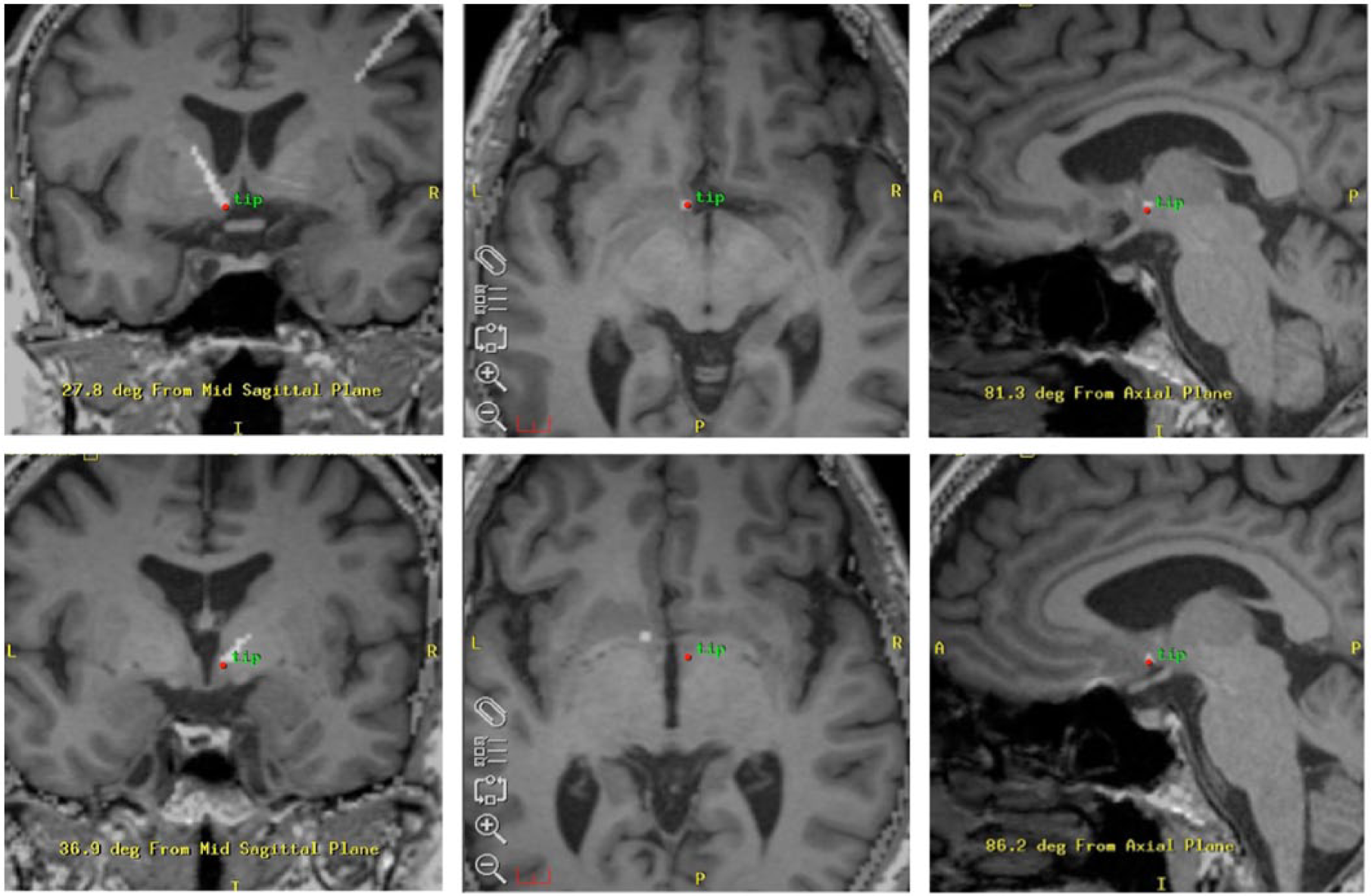
Electrode placement for nucleus accumbens and bed nucleus stria terminalis.
Stimulation protocol
Stimulation was typically commenced in the 2 weeks following stage two surgery and was titrated as per the treating neurologist. This was typically monopolar stimulation with uptitration of voltage to achieve clinical effect. If desired efficacy was not achieved with one active contact bilaterally, then a second contact was programmed, to give multiple-monopolar stimulation.
Outcome measures
Following DBS surgery, all patients were reviewed approximately every 6 weeks. Each review comprised psychiatric assessment including rating scales and neurological review of the stimulator settings. All ratings were undertaken unblinded to DBS settings. Medication adjustments were made at the discretion of the treating psychiatrist.
Statistical analysis
Due to the small size of the cohort, results described herein are predominantly descriptive; however, basic statistical analyses were undertaken using IBM SPSS Statistics for Mac, version 22. Data were tested for normality using the Shapiro–Wilk test, as well as inspection of histograms and Q-Q plots and found to be not normally distributed, thus non-parametric tests were used. The primary outcome measure (YBOCS) as well as the other secondary measures were analysed using the Wilcoxon signed-rank test.
Results
Demographic and clinical characteristics
The seven patients described satisfied the necessary criteria for psychosurgery and an application made to the PRB, which approved the procedures. The mean age of all patients who underwent DBS surgery was 46.4 years, with a mean duration of OCD symptoms of 25.9 years. All patients were on psychotropic medication at the time of surgery, as per Table 2. None were undergoing concurrent psychological treatment.
Changes in obsessive-compulsive disorder, anxiety, depression, stress and functional impairment from baseline to last follow-up.
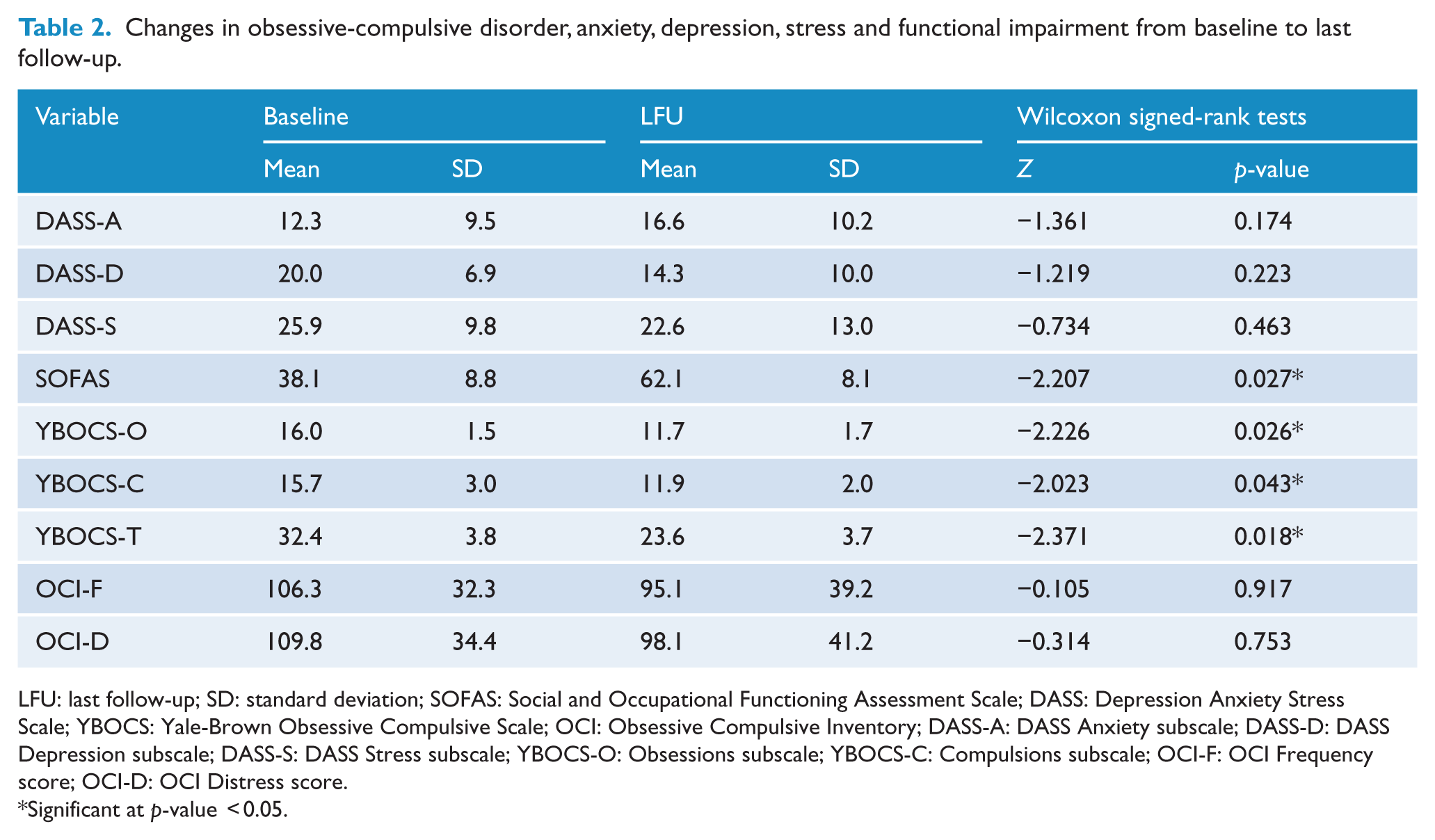
LFU: last follow-up; SD: standard deviation; SOFAS: Social and Occupational Functioning Assessment Scale; DASS: Depression Anxiety Stress Scale; YBOCS: Yale-Brown Obsessive Compulsive Scale; OCI: Obsessive Compulsive Inventory; DASS-A: DASS Anxiety subscale; DASS-D: DASS Depression subscale; DASS-S: DASS Stress subscale; YBOCS-O: Obsessions subscale; YBOCS-C: Compulsions subscale; OCI-F: OCI Frequency score; OCI-D: OCI Distress score.
Significant at p-value < 0.05.
Obsessive-compulsive symptoms
As demonstrated in Table 2, the mean reduction in OCD symptom severity as measured by the YBOCS was found to be statistically significant, for the total score as well as both subscales, indicating that DBS was effective in reducing symptom severity. The baseline scores for the YBOCS and OCI and the follow-up rating at the last visit for each subject are summarised in Table 3. Obsessive-compulsive symptoms improved for all patients on the YBOCS rating scale.
Summary of individual results from YBOCS and OCI scores from baseline to last follow-up.
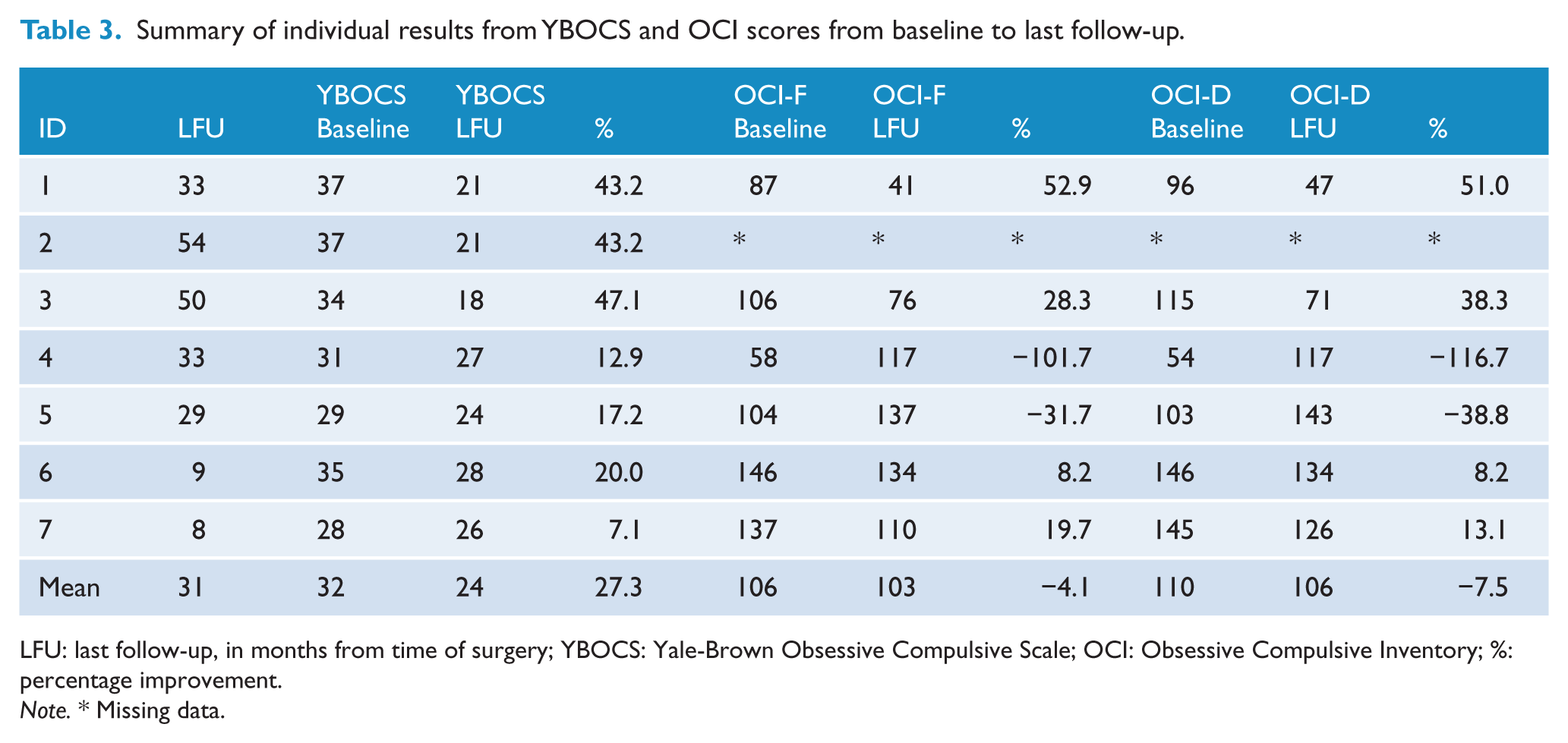
LFU: last follow-up, in months from time of surgery; YBOCS: Yale-Brown Obsessive Compulsive Scale; OCI: Obsessive Compulsive Inventory; %: percentage improvement.
Note. * Missing data.
Three patients from this cohort were defined as full responders, with two partial responders and two non-responders. Fluctuations in YBOCS scores were noted during the course of treatment. Significant deteriorations resulting in earlier review were associated with co-morbid depressive episodes, generalised anxiety symptoms or responsive to psychosocial stressors such as the death of a close relative.
Socio-occupational functioning
There was a statistically significant improvement in social and occupational functioning as measured by the SOFAS (Table 2). There was a mean improvement of 37% in the SOFAS score, with all patients scoring at 50 or above at last follow-up. This represents a change from ‘inability to function’ or ‘major impairment’ at baseline to ‘moderate’ or ‘some difficulty’ in socio-occupational functioning.
Adverse events
No life-threatening adverse events occurred following DBS. There were no postoperative concerns. Two patients developed hypomanic episodes that responded to reduction in stimulation alongside mood stabilising medication. Hypomania is a recognised and serious potential side effect in DBS (Naesström et al., 2016). These episodes did not require hospitalisation. One patient developed ageusia that spontaneously resolved. Another patient reported déjà-vu both during stage one surgery and at times of increased stimulation settings; however, this was self-limiting.
Depression and anxiety symptoms
Changes in DASS scores were not statistically significant. On average, Depression and Stress subscale scores on the DASS improved by 23.3% and 11.8%, respectively, at last follow-up compared to pre-surgery. Anxiety subscale scores on average deteriorated by 64.2% from baseline.
Qualitative outcomes
Meaningful positive life events were reported by patients that they attributed to DBS. These included undertaking overseas trips, attending family celebrations, returning to part-time work, enrolling in university studies and participation in leisure activities.
Discussion
This initial Australian experience of DBS for OCD has demonstrated that DBS is a safe and effective procedure in the treatment of severe OCD. We identified significant improvements in OCD symptom severity and functioning. The procedure was found to be safe, with the most serious adverse events being two episodes of hypomania. These were detected and managed without further significant risk or consequences such as hospitalisation.
DBS for severe intractable OCD was effective in our cohort, as demonstrated by a mean reduction in YBOCS of 27.3%, with 3/7 patients achieving a full response. These results are in keeping with other cohorts of DBS in OCD worldwide (of between 7 and 26 patients), who report a range of mean YBOCS reduction of between 17% and 72%, and response rates of 43–66% (Denys et al., 2010; Goodman et al., 2010; Greenberg et al., 2010; Islam et al., 2015; Mallet et al., 2008). The large variance in efficacy among different groups reflects the gaps in the evidence base, which is largely made up of case series with small patient numbers, as well as methodological inconsistencies (Fitzgerald and Segrave, 2015). To date, there have been no head-to-head randomised trials of implantation sites, nor consideration of symptom predominance (Brakoulias, 2013; Fitzgerald and Segrave, 2015). The definition of ‘full response’ constituting an improvement in YBOCS of 35% or more also sets the bar low for treatments in OCD. This is a well-established threshold, including for pharmacological trials, but continues to challenge the field. It is worth noting the probability of achieving full remission with serotonin reuptake inhibitors is 38% according to the same criteria (Catapano et al., 2006), and a review of augmentation with atypical antipsychotics found a mean YBOCS reduction of 10% (Veale et al., 2014). When considering that DBS is tried after other evidence-based treatments have failed, the improvements gained with DBS represent a meaningful outcome. This was supported by improvements in patient functioning. The contribution of regular psychiatric review and therapeutic rapport to improved symptoms and functioning must be considered, as has been shown in other psychiatric conditions such as major depressive disorder (Khan et al., 2012).
A statistically significant improvement in socio-occupational functioning was observed in this cohort, though significant impairments remained. This improvement was sufficient to enable meaningful activities in a majority of patients. Future work should assess the correlation of improvements in functioning with other measures such as quality of life and conduct a cost–benefit analysis.
Hypomania was the most severe adverse event in this cohort, though other cohorts have reported other severe adverse effects including infection, seizures, intracerebral haemorrhage, vertigo and visual changes (Naesström et al., 2016). Hypomania is a recognised side effect of DBS at various targets including the subthalamic nucleus (STN), NAc, VC/VS and ACDBS (Naesström et al., 2016). As such, assessment of mood forms an important part of ongoing regular mental state assessment following surgery, particularly with increasing stimulation parameters.
Depression and stress scores improved, though we identified individual variation, and DASS-D and -S scores in some patients worsened despite overall improvement in YBOCS. We identified fluctuations in anxiety scores during treatment with percentage improvements ranging from 47% to 100% and deterioration ranging from 0% to 300%. The worsening of Anxiety scores on the DASS in four of seven patients at last follow-up (with two recording no change and only one patient, an improvement) was an unexpected outcome and not in keeping with previous studies which have demonstrated anxiolytic effects of DBS (Denys et al., 2010) nor with studies which have concluded that the effect of DBS on OCD is likely related to the effects on mood and anxiety (Denys et al., 2010). There are a number of possible explanations for this finding. First, patients with higher levels of anxiety may be more likely to seek out and/or enter into treatment or have a more treatment refractory illness. Second, several patients noted that improvements in their OCD had enabled a greater level of ‘resisting’ symptoms and improved social functioning. This may increase intra- and interpersonal demands, which in turn could lead to greater levels of anxiety. Finally, we did not control for medication adjustments that were under the control of the patients’ treating psychiatrist.
This paper describes the results of a series of seven patients in a clinical research programme who underwent DBS treatment. The two major methodological limitations of this study were that we did not rate patients blind to DBS status or settings, nor did we standardise the DBS adjustments across all patients. Past randomised control trials have used standardised DBS protocols and periods of stimulation and ‘sham’ stimulation within the same patient (Denys et al., 2010). Given these studies and the small number of DBS procedures we anticipated, we chose to proceed with an open-label study. Despite these limitations, this study adds to field of research by adding further data to the relatively low case numbers, particularly regarding the BNST target, for which there have been relatively few published studies to date (Islam et al., 2015; Luyten et al., 2016).
The number of institutions offering DBS for OCD in Australia has grown in the previous few years as efficacy has been established. There are several ongoing challenges. These include but are not limited to:
Defining the most appropriate candidates for DBS surgery, that is, which factors may influence likely response to treatment.
Determining the best neuroanatomical target for DBS. To date, the results for varying neuroanatomical targets have been comparable, but are often applied ubiquitously in study groups.
Determining the most likely effective electrode contacts and stimulation settings. Currently, this is done via a stepwise ‘try and see’ approach as per the treating physician. In a condition such as Parkinson’s disease where the results are visible and occur much more quickly, this is more acceptable; however, the effects of stimulation can take weeks to months to improve OCD symptoms. This can lead to overstimulation and worsening of symptoms, or lengthy periods of ineffective or suboptimal treatment.
Accessibility and cost-effectiveness: At present, DBS remains an expensive treatment option that is only covered through self-funding, private health insurance or in rare instances work or transport insurance bodies. Average costing for an initial assessment, implantation process and device is approximately $80,000 AUD (not inclusive of public outpatient follow-up). The cost of replacement rechargeable batteries is approximately $25,000 AUD. DBS is only available in select states in Australia and is governed by the presiding Mental Health Tribunal. The main cost to the patient is in the upfront cost of the device and battery replacements, as the follow-up is usually conducted in a public outpatient clinic. Studying the cost-effectiveness of this treatment is extraordinarily difficult, as despite OCD being one of the most disabling psychiatric illnesses, patients are not often high utilisers of the health system, even when severely unwell. Without being able to demonstrate cost-effectiveness, it remains unclear whether DBS will attract government funding and thus continue to be a treatment that is only available to a select few.
Future directions
As further research of DBS in OCD is conducted, researchers should give consideration to reporting potential prognostic factors, in the hope that results may be pooled and analysed. Ideally, this could take place via an international registry akin to MSBase (Butzkueven et al., 2006). There is scope for further research to identify ‘biomarkers’ that may predict response to DBS, as there is emerging evidence of different neurobiological circuitry for different OCD symptom subtypes (Mataix-Cols et al., 2004). With regard to optimising electrode placement and programming, there have been preliminary studies, utilising techniques such as electroencephalography (EEG), functional MRI (fMRI) and magnetoencephalography (MEG) to better understand the mechanisms by which DBS works, as well as the underlying neuropathophysiology of OCD (Figee et al., 2013; Oswal et al., 2016). Other emerging imaging techniques include diffusion MRI tractography, which allows visualisation of white matter pathways, and modelling of brain connectivity and effects of electrical stimulation. Early studies have emerged in the last 2–3 years looking at these techniques and have reported its use in both surgical planning (Schlaepfer et al., 2013) and correlation with optimal stimulation parameters and connected cortical regions (Hartmann et al., 2015). This area requires further scientific inquiry with reproduction of findings to make firmer conclusions and thus extrapolate to factors that may influence suitability for DBS and optimum stimulation parameters.
In summary, the results of this study indicate that bilateral nucleus accumbens and bed nucleus stria terminalis DBS is effective and safe in the treatment of severe OCD. It is hoped that as newer imaging techniques become more available and biomarkers are identified, they will become incorporated into DBS treatment, offering patients more individualised and efficacious treatment.
Footnotes
Declaration of Conflicting Interests
S.F. received 0.5 FTE funding from Medtronic in the year 2016 as the DBS registrar at the Neuropsychiatry Unit. A.B. is employed by Medtronic and assisted in the creating of co-registered MRI and CT images for the paper.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
