Abstract
Deep brain stimulation has shown promise for the treatment of severe, treatment-refractory obsessive-compulsive disorder. With the recent publication of the first Australian, randomised, sham-controlled trial of deep brain stimulation for obsessive-compulsive disorder, there are now four placebo-controlled trials demonstrating the efficacy of this therapy. Together with recent data identifying a biological substrate of effective stimulation that can predict response and that has been successfully reproduced, studies comparing and finding equivalent efficacy among different targets, as well as recent, large, open trials supporting the long-term effectiveness of deep brain stimulation, we argue that this should now be considered an accepted therapy for a select group of patients in the Australasian setting. We call on the Royal Australian and New Zealand College of Psychiatrists to revise their memorandum describing deep brain stimulation for obsessive-compulsive disorder as an ‘experimental’ treatment and recognise that it has proven efficacy. We stress that this should remain a therapy offered only to those with high treatment-refractory illnesses and only at specialised centres where there is an experienced multidisciplinary team involved in work-up, implantation and follow-up and also where frameworks are in place to provide careful clinical governance and ensure appropriate fully informed consent.
Keywords
Treatment-refractoriness and deep brain stimulation
In psychiatry, there are persons who, despite accurate diagnosis and the absence of confounding comorbidities, do not respond to the best-available, evidence-based therapies and remain highly unwell. Obsessive-compulsive disorder (OCD) is one condition that may present with ‘treatment-refractoriness’. With an estimated lifetime prevalence of between 1% and 2 % (Kessler et al., 2005), OCD is characterised by the intrusion of ego-dystonic, anxiety-provoking thoughts, urges or images (obsessions), generally accompanied by repetitive mental acts or behaviours (compulsions), which are carried out to neutralise the obsessions or to mitigate anxiety associated with them . The mainstay of treatment involves a combination of pharmacological (antidepressant) and psychological (cognitive-behavioural therapy; CBT) approaches (Fineberg et al., 2020). However, many sufferers still have clinically significant symptoms despite drug treatment (Erzegovesi et al., 2001) even after augmented therapy (Bloch et al., 2006). The most effective psychological treatment for OCD involves deliberate exposure to anxiety-provoking situations with the expectation that this anxiety (and the need to maintain compulsive rituals) will habituate over time. However, individuals with severe OCD commonly find this process intolerable and cannot engage fully in treatment or else do not habituate despite persistent exposure. The net result is that there exists a subgroup of people with OCD who are treatment-refractory, remain highly disabled and have limited therapeutic options available to them to improve their quality of life.
Deep brain stimulation (DBS) is a therapy that enables focal modulation of brain activity via implanted stimulating electrodes. Electrodes are precisely sited neurosurgically in a densely innervated subcortical region and connected to an implantable pulse generator (a brain pacemaker) that delivers continuous electrical stimulation. Stimulation parameters can be adjusted postoperatively to vary the position (the active contact), size and shape of the stimulation field within the brain. In addition to local stimulation effects, DBS can modulate activity in distant brain regions that are anatomically connected to the stimulation field. Thus, although device implantation requires surgery, chronic neurostimulation is provided (and can be adjusted) on an ambulatory basis. Once implanted, the device can be turned on and off, permitting a crossover, sham-controlled study of its effects within an individual participant. The electrodes can also be removed with minimal tissue damage, further distinguishing this therapy from surgical or gamma-knife lesioning procedures.
There is a significant volume of prior work that associates OCD with abnormal brain activity in frontolimbic circuits and demonstrates that these abnormalities can be normalised with electrical stimulation (Figee et al., 2013; Treu et al., 2021). DBS was first employed for the treatment of intractable OCD in the late 1990s (Nuttin et al., 1999), with initial targeting in the anterior limb of the internal capsule (ALIC) informed by prior work using ablative neurosurgery. Further studies reproduced encouraging preliminary outcomes (Abelson et al., 2005; Goodman et al., 2010; Greenberg et al., 2006), finding improved response with posterior migration of the target within the ventral capsule and ventral striatum (Greenberg et al., 2010). The anteromedial segment of the subthalamic nucleus (STN) has also been a promising target for neuromodulation (Mallet et al., 2008). An influential meta-analysis published 7 years ago examined available randomised, blinded, placebo-controlled data and concluded that DBS ‘may show promise for severe treatment-resistant OCD’ (Kisely et al., 2014). This sentiment is repeated in the Royal Australian and New Zealand College of Psychiatrists (RANZCP) memorandum on DBS (published in 2018) but with the rejoinder that ‘the limited number of research studies suggests that it should be considered experimental at this time’. Despite being published in 2018, this memorandum appears to overlook an important placebo-controlled trial published in 2016 (Luyten et al., 2016).
The authors of this Viewpoint article are all psychiatrists with direct experience of treating persons with DBS for OCD. We argue that there is now sufficient evidence to consider this therapy as accepted for carefully selected individuals in specialised treatment centres.
New data
A group of us (Mosley, Marsh, Mohan and Sachdev) recently published the first Australian, randomised, double-blind, sham-controlled trial of DBS for treatment-refractory OCD (Mosley et al., 2021). Electrodes were implanted in the bed nucleus of the stria terminalis (BNST), a region of the subcortex posterior to the nucleus accumbens (NAcc) and structurally connected to the amygdala and prefrontal cortex. In nine participants with long histories (decades) of treatment-refractory symptoms, we demonstrated a statistically significant difference between active and sham stimulation in a 3-month delayed-onset design (participants were randomised to have their device turned on or remained off for this period). After 12 months of open-label treatment, we reported a mean reduction in score on the Yale–Brown Obsessive-Compulsive Scale (YBOCS) of 49.6%. A reduction of 35% on this scale is globally accepted as being clinically significant, with seven of our nine participants meeting the threshold for a clinically significant response. Our participants had all previously undertaken exhaustive trials of pharmacological treatment (including clomipramine and several trials of antipsychotic augmentation) and exposure-oriented CBT (most had several courses and intensive residential therapy), as well as other psychotherapeutic strategies. We found that participants who responded were subsequently able to tolerate CBT and accrued additional benefit from a 10-session course. Interestingly, we were able to identify a neuronal tract connecting the amygdala and the ventrolateral prefrontal cortex that, when stimulated, was associated with a reduction in OCD symptoms, reinforcing the role of the BNST as a component of the ‘extended amygdala’ and supporting prior work suggesting that DBS acts via facilitating fear extinction (Fridgeirsson et al., 2020).
We consider this to be the fourth adequately reported placebo-controlled randomised controlled trial (RCT) (Table 1), adding to crossover trials of DBS at the STN (Mallet et al., 2008), NAcc/ALIC interface (Denys et al., 2010) and the BNST (Luyten et al., 2016) and comprising a total of 56 participants. Our blinded effect size (4.9 point difference in YBOCS between active and sham) was smaller than other sham-controlled trials of DBS in OCD (12 points in Luyten et al., 9 in Mallet et al. and 8 in Denys et al.), but importantly all of these studies used a crossover design after an open-label phase of stimulation optimisation. An advantage of a blinded phase at the start of follow-up is that participants were not exposed to stimulation prior to the blinded phase, thereby reducing the likelihood that they could recognise sensations associated with active stimulation and vice versa. A disadvantage was that due to a slow titration protocol, participants randomised to active stimulation in our trial spent less than the full phase at adequate stimulation intensities, thereby making this a less powerful approach to detecting treatment effects. This likely explains the failure of an earlier trial incorporating only a 1-month delayed-onset double-blind phase to reach statistical significance in six participants, despite a trend towards benefit for active stimulation over sham (Goodman et al., 2010).
Randomised, double-blind, sham-controlled studies assessing the efficacy of DBS for OCD.

DBS: deep brain stimulation; OCD: obsessive-compulsive disorder; $: not statistically significant; YBOCS: Yale–Brown Obsessive-Compulsive Scale.
Other supporting evidence
Four additional open-label studies in a further 100 participants have supported the aforementioned findings, with an average YBOCS reduction of 42%, 40%, 44% and 38 % after DBS of the ALIC/BNST (Denys et al., 2020; Kahn et al., 2021; Menchon et al., 2021; Naesström et al., 2021). An Australian case series of seven participants has also been published (Farrand et al., 2018). Other recent open-label cohorts have not only reported encouraging results but have also contributed to our mechanistic understanding of DBS, by identifying a mediating role of a common brain network that – when recruited by stimulation – reduces OCD symptoms. The existence of this common network explains why different subcortical targets could all be helpful in treating OCD, consistent with the present understanding of this condition as a circuit-based disorder rather than one of a circumscribed brain region. In essence, there may be several subcortical ‘access points’ to this network, which can be reached with DBS.
The basis of these findings is as follows: it is possible, through modelling the distribution of electrical stimulation in the brain, to identify cortical/subcortical brain regions connected by white matter fibres to the field of stimulation. It is also possible, through the use of diffusion neuroimaging, to model the course of white matter tracts passing through and adjacent to the stimulation field. Convergent findings in cohorts of 22 and 50 participants (comprising surgical targeting of the ALIC, NAcc and STN DBS at different centres) identified key fibre tracts connecting the anteromedial STN and medial dorsal thalamus with the lateral and medial prefrontal cortex via the ALIC (Baldermann et al., 2019; Li et al., 2020). Importantly, using cross-validation, the response of a given ‘out of sample’ participant could be predicted based on the structural connectivity of their stimulation field. This predictive ability was also found in our cohort, and the distribution of these independently generated key fibre pathways is remarkably alike (Figure 1). These ‘connectomic’ findings have since been replicated by a further two research groups in a total of 18 participants (Smith et al., 2021; van der Vlis et al., 2021). Finally, the relative benefits of two different targets (STN and ventral striatum) were compared in an elegant double-blind crossover trial in which six participants were implanted with electrodes at both locations and received periods of stimulation at each target in isolation (Tyagi et al., 2019), with both targets found to be equally effective in their anti-OCD effect. However, there were dissociable effects on mood and cognitive flexibility by stimulation site in this study that are worthy of further exploration so as to determine whether certain stimulation sites could be preferred based on individualised cognitive and affective comorbidities.
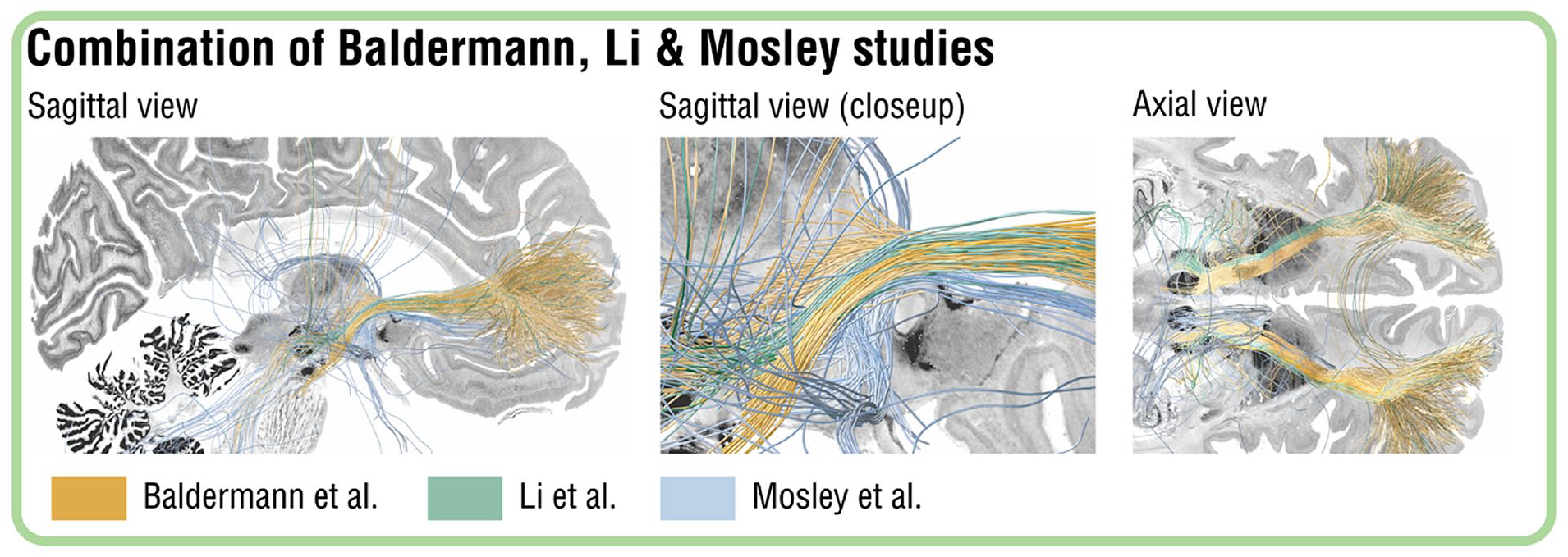
Overlapping connectivity fingerprint of effective DBS for OCD.
An accepted therapy?
In Europe and the United States, DBS for OCD is available to eligible participants via a Conformité Européene mark and a Humanitarian Device Exemption, respectively. While these labels do not endorse efficacy, they indicate that the procedure does not involve ‘unreasonable risk’ and allows for reimbursement to occur from insurance companies. At present, in Australia there is no mechanism for an individual with treatment-refractory OCD to receive DBS outside of a clinical trial (DBS is restricted to Parkinson’s disease, essential tremor and dystonia on the Medicare Benefits Schedule). Of greater concern, in New South Wales DBS for psychiatry remains prohibited under the Mental Health Act. Clinical trials have been an essential part of demonstrating the efficacy and safety profile of this therapy while safeguarding the rights of consenting participants. However, given the existing evidence of benefit outlined above, it seems increasingly unlikely that further sponsored trials will be undertaken by device manufacturers, while the funding landscape for Australian clinical trials as part of the National Health and Medical Research Council (NHMRC) and Medical Research Future Fund (MRFF) looks increasingly bleak, especially for costly therapies that benefit only a small number of participants, however unwell they may be.
We argue that there is now sufficient evidence of efficacy of DBS for OCD to establish it as an accepted therapy based on data from the aforementioned placebo-controlled clinical trials. Furthermore, there is strong supporting neuroanatomic evidence lending biological plausibility to these findings. In this, we echo the sentiments of the Task Force on Neurosurgery for Psychiatric Disorders, a committee of the World Society for Stereotactic and Functional Neurosurgery (WSSFN): ‘it would be unreasonable and imprudent to view this approach as purely investigational once sufficient evidence of safety and efficacy are demonstrated to validate the intervention as a treatment’ (Wu et al., 2020).
We share the opinion of the WSSFN that DBS should remain a treatment reserved for those individuals with demonstrated treatment-refractoriness and should only be carried out at dedicated, experienced units with strong affiliations with multidisciplinary research teams. Entry into our trial involved assessment by two independent psychiatrists with two separate YBOCS ratings in the severe range, as well as a corroborated history of treatment-refractoriness defined by insufficient response to at least (1) two trials of selective serotonin reuptake inhibitors at maximum tolerated dose for at least 12 weeks, (2) one trial of clomipramine at maximum tolerated dosage for at least 12 weeks, plus (3) one augmentation trial with an antipsychotic for at least 8 weeks in combination with one of the aforementioned drugs, plus (4) one complete trial of exposure-based CBT confirmed by a psychotherapist. In practice, our participants greatly exceeded these requirements, and their informed participation was endorsed by an independent mental health review tribunal.
An accepted therapy in practice
How would an acceptance of this therapy operate in practice? In addition to the requirement to demonstrate treatment-refractoriness (we propose the criteria above as a reasonable working definition), we suggest that units undertaking DBS for OCD should be accredited based on their experience with functional neurosurgery (i.e. minimum number of cases per year, surgical complication rates consistent with global rates) and the close involvement of at least one psychiatrist with experience in DBS programming. The informed consent of each person wishing to undertake DBS should be evaluated by a mental health review tribunal incorporating input from the usual treating psychiatrist and family caregivers. Follow-up should include exposure-oriented CBT and the involvement of other allied health clinicians to assist the individual with adapting to life with less significant symptoms. Each individual undertaking DBS should have a mandated follow-up period of at least 12 months with measures of treatment response recorded in a national registry. These results should be published to support advancement of the evidence base for this treatment.
Counter-arguments
It must be noted that there are several potential criticisms of our position. The total number of participants who have received DBS for OCD, while now approaching 300 worldwide, remains relatively low compared to most other evidence-based interventions. However, we argue it is reasonable to base assumptions of efficacy and safety on small numbers of participants in a therapeutic intervention that (1) involves neurosurgery, (2) should only be offered to the sickest, treatment-refractory individuals and (3), given the historical ethical issues relating to psychosurgery, should only be offered at a handful of specialised centres with an established track record in neuromodulation and where independent ethical oversight can take place. In this context, the demand that ‘more research is needed’ seems unreasonable, given, as we have outlined, the existence of four independent, randomised, sham-controlled trials of adequate duration at different centres, as well as data supporting the effectiveness of different subcortical targets in the brain connected by a common neuronal pathway.
There are risks associated with DBS that must be carefully considered, commensurate with it involving neurosurgery and implantation of a medical device. Intracerebral haemorrhage is rare but has been reported as a serious adverse event. The most common surgical complication is infection of the DBS system, which although not typically life-threatening necessitates removal of the device and a course of intravenous antibiotics before re-implantation can be considered. Unfortunately, this occurred in one of the participants in our trial. The most significant psychiatric risk is associated with sudden cessation of stimulation, which can occur due to battery depletion or lead fracture. Almost all published studies report a significant deterioration in OCD symptoms under such circumstances, which may also include depressive symptoms and suicidal ideation (Nuttin et al., 2003). In some cases, psychiatric symptoms ‘rebound’ to a greater level than at baseline (Ooms et al., 2014), postulated to reflect an ‘obsessive storm’. Many studies report the induction of stimulation-dependent impulsivity and hypomania in some participants during the initial titration of stimulation. However, these neuropsychiatric side effects tend to be relatively mild and transient; there are no reports of such patients requiring involuntary hospitalisation or treatment with mood stabilisers. There were no such adverse events in our study.
Finally, were DBS for OCD to attract federal health system funding in Australia, it might be argued that the costly nature of this surgery takes resources away from other deserving groups. Initial health economic analyses of DBS for OCD are promising (Moon et al., 2017; Ooms et al., 2017) and it is likely that the cost of this surgery is mitigated by the reduction in ongoing intensive psychiatric and psychological care, improved occupational functioning and reduced reliance on welfare. For example, in our study one person who had been unable to work now runs their own business and another who required full-time nursing home care is moving out into supported accommodation. In addition, as noted above, the uptake of this therapy is likely to remain low due to the limited pool of eligible candidates (in our study it took 4 years to recruit nine candidates from Queensland and New South Wales). This, along with the limited number of appropriately experienced surgical units, will constrain total expenditure.
Conclusion
In summary, there are now four randomised, placebo-controlled, double-blind trials demonstrating the efficacy of DBS for OCD, including in an Australian cohort, in addition to numerous open-label studies. We suggest that the RANZCP should recognise this as an accepted, evidence-based therapy for individuals with treatment-refractory OCD, with treatment-refractoriness defined as a failure of adequate pharmacotherapy and psychotherapy. Informed consent must be overseen by an independent mental health review tribunal and surgery offered at accredited centres incorporating mandatory follow-up and outcome reporting.
The barriers of reclassification of DBS as an accepted therapy in OCD include the fear of unregulated psychosurgery and uncertainty about health economics. However, these concerns are not borne out by the cautious worldwide uptake of this treatment in the last 20 years and preliminary economic analyses. These barriers reflect a historical bias that is no longer relevant in the current landscape of closely regulated healthcare provision. Indeed, given the multidisciplinary team required to conduct DBS surgery, it is nigh on impossible for such a procedure to be offered indiscriminately and outside of the tertiary settings we suggest above. It is our opinion that this therapy can relieve suffering in the most unwell individuals who have exhausted other treatment options and should be offered earlier in the course of illness once treatment-refractoriness has been demonstrated. To delay acceptance of this therapy is likely to prolong suffering, especially when there are no other new effective treatments on the horizon. Ultimately, DBS for OCD will be consigned to history if its continued misclassification as being experimental in nature is not reviewed in light of contemporary evidence and in the absence of ideological biases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: P.E.M. has received an honorarium from Boston Scientific for speaking at an educational meeting in 2018 and a Parkinson’s disease research grant from Medtronic in 2015. He has served on a scientific advisory board for TEVA pharmaceuticals (Huntington’s disease) and Noema pharmaceuticals (Tourette’s syndrome). D.V. has received consulting fees from TEVA pharmaceuticals and royalties from ACER for the NUCOG cognitive screening tool. S.F. undertook a DBS fellowship in 2016 that was partially funded by Medtronic and has received an honorarium from Abbvie for speaking at an educational meeting in 2020. A.M. and R.M. declare no conflict of interest. D.C. has received grant monies for research from Eli Lilly, Janssen Cilag, Roche, Allergen, Bristol-Myers Squibb, Pfizer, Lundbeck, Astra Zeneca and Hospira; travel support and honoraria for talks and consultancy from Eli Lilly, Bristol-Myers Squibb, Astra Zeneca, Lundbeck, Janssen Cilag, Pfizer, Organon, Sanofi-Aventis, Wyeth, Hospira, Servier and Seqirus; and is a current or past Advisory Board Member for Lu AA21004: Lundbeck; Varenicline: Pfizer; Asenapine: Lundbeck; Aripiprazole LAI: Lundbeck; Lisdexamfetamine: Shire; Lurasidone: Servier; Brexpiprazole: Lundbeck. He is founder of the Optimal Health Programme, currently operating as Optimal Health Australia, and is part owner of Clarity Healthcare. He is on the scientific advisory of The Mental Health Foundation of Australia. He does not knowingly have stocks or shares in any pharmaceutical company. P.S. was on the scientific advisory committee of Biogen Australia for the anti-dementia drug aducanumab.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
