Abstract
Following on from the publication of the Royal Australian and New Zealand Journal of Psychiatry Mood Disorder Clinical Practice Guidelines (2020) and criticisms of how these aberrantly addressed repetitive transcranial magnetic stimulation treatment of depression, questions have continued to be raised in the journal about this treatment by a small group of authors, whose views we contend do not reflect the broad acceptance of this treatment nationally and internationally. In fact, the evidence supporting the use of repetitive transcranial magnetic stimulation treatment in depression is unambiguous and substantial, consisting of an extensive series of clinical trials supported by multiple meta-analyses, network meta-analysis and umbrella reviews. Importantly, the use of repetitive transcranial magnetic stimulation treatment in depression has also been subject to a series of health economic analyses. These indicate that repetitive transcranial magnetic stimulation is a cost-effective therapy and have been used in some jurisdictions, including Australia, in support of public funding. An argument has been made that offering repetitive transcranial magnetic stimulation treatment may delay potentially effective pharmacotherapy. In fact, there is considerably greater danger of the opposite happening. Repetitive transcranial magnetic stimulation is as, if not more effective, than antidepressant medication after two unsuccessful medication trials and should be a consideration for all patients under these circumstances where available. There is no meaningful ongoing debate about the use of repetitive transcranial magnetic stimulation treatment in depression – it is a safe, effective and cost-effective treatment.
Keywords
In a recent issue of the journal, Malhi et al. (2021a) restate claims made by the first author about repetitive transcranial magnetic stimulation (rTMS) treatment for depression that the ‘evidence for the efficacy of this intervention is still very much in its infancy’ and that ‘the jury is very much out as to whether there is a meaningful signal with rTMS delivery in the treatment of depression’. The authors support these claims only by references to the same lead author’s previous publications and with no systematic review of the evidence. As has been presented recently in this journal (Fitzgerald et al., 2021a) and described extensively elsewhere (e.g. Brunoni et al., 2017; Papadimitropoulou et al., 2017; Razza et al., 2020), these statements are inaccurate (see Table 1). In fact, there already exists a substantive evidence base supporting the use of rTMS in the treatment of depression that extends from multisite clinical trials to multiple meta-analyses, network meta-analyses, umbrella reviews and large-scale real-world treatment outcome data (Fitzgerald et al., 2021a). Scientific support for the efficacy and cost-effectiveness of rTMS for depression is by no means in ‘its infancy’: in fact it is consistent with that required to support the clinical use of a new treatment and is underwritten by a wealth of data exploring its neurobiological mechanisms (Luan et al., 2020) and real-world outcomes (Sackeim et al., 2020).
rTMS-related myths and corresponding evidence-supported statements.
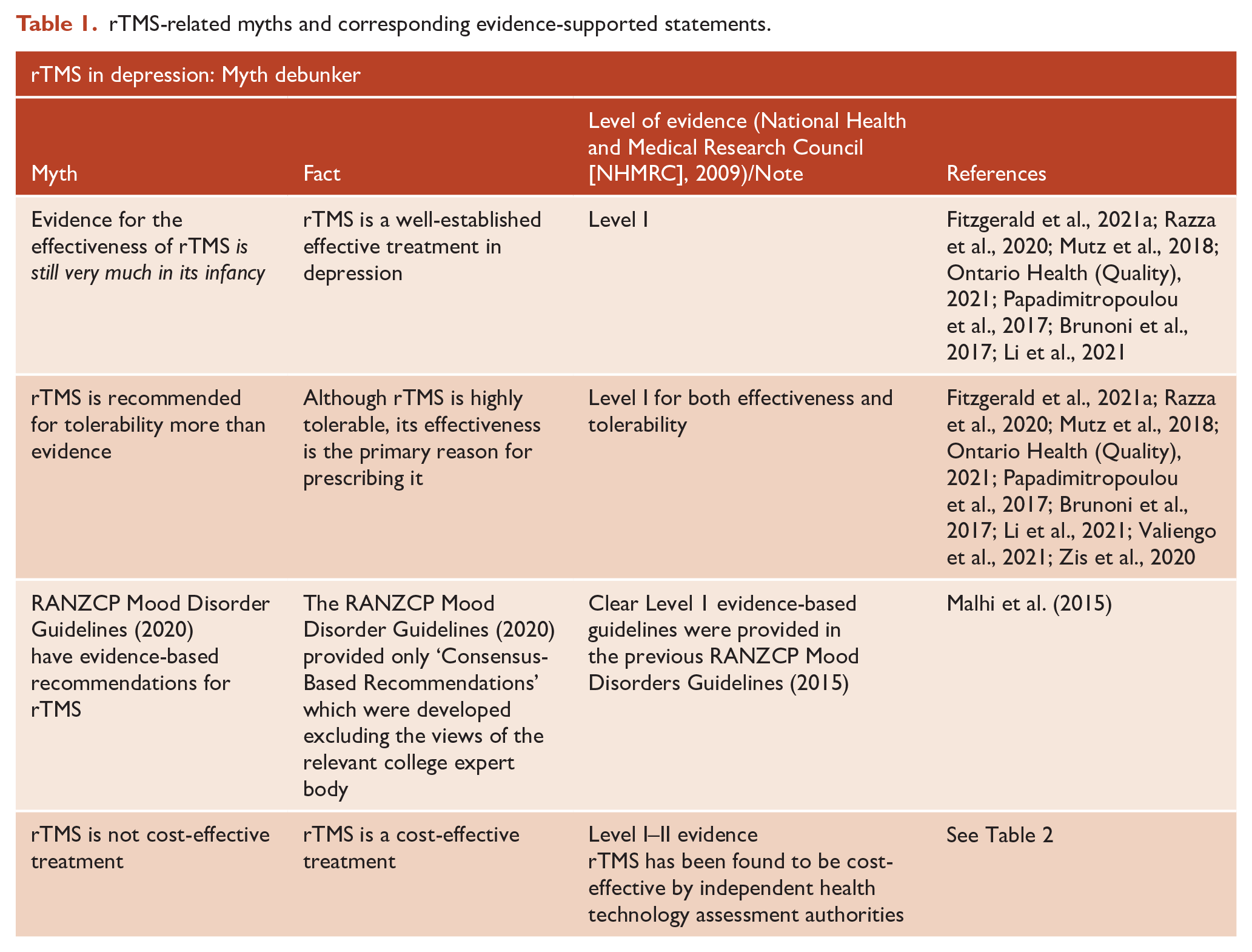
Malhi et al. (2021b) also cite the recently published RANZCP clinical practice guidelines for mood disorders. However, the way in which these guidelines addressed rTMS therapy has been criticised in regard to both the content and process of their development (Fitzgerald et al., 2021b). Most critically, the guidelines omitted the substantive level 1 evidence that supports the efficacy of rTMS (and which was included in a previous version of the college guidelines; Malhi et al., 2015). Instead, the 2020 guideline included ‘Consensus-Based Recommendations’ which were based on erroneous arguments and developed with a critical lack of due process (Fitzgerald et al., 2021b). Specifically, rTMS was addressed with an out-of-date and selective, not systematic, review. Second, the ‘consensus’ and consultation process were wholly inadequate. There was no formal consensus process involving individuals, especially experts in the field, outside of the authorship group. The guideline authors provided the draft guideline to the committee of the RANZCP’s Section for ECT and Neurostimulation but then ignored the opinion of the committee, who represent the relevant specialists in the field, which was that the guideline’s appraisal of the evidence for rTMS was substantially inaccurate and inadequate (Fitzgerald et al., 2021b).
The strong evidence supporting the efficacy of rTMS render arguments such as ‘When the efficacy of rTMS is questioned a common response is that because rTMS is highly tolerable there is no harm trying it’ meaningless. In fact, the true ‘hidden costs’ derive from denying patients timely access to rTMS, not when rTMS is offered instead of medication. Given that the evidence points to rTMS being more effective than continued pharmacological treatments once patients have failed several antidepressant trials (Fitzgerald et al., 2021a; Papadimitropoulou et al., 2017), then continuing further trials of various medication strategies (which in real-world practice can be protracted over years) instead of offering rTMS therapy constitutes withholding effective treatment and introducing the risk of harms associated with chronic or worsening depression. Repeated unsuccessful trials of medication are much more likely to lead to adverse consequences than a trial of rTMS. An initial trial of rTMS to establish efficacy is time-limited, does not require medication titration/withholding/washout and has about a 50% chance of achieving depression treatment response (Sackeim et al., 2020). Denying or delaying rTMS treatment can have real-world consequences. Clinically, we have seen patients denied access to rTMS who reported adverse outcomes of treatment denial or delay.
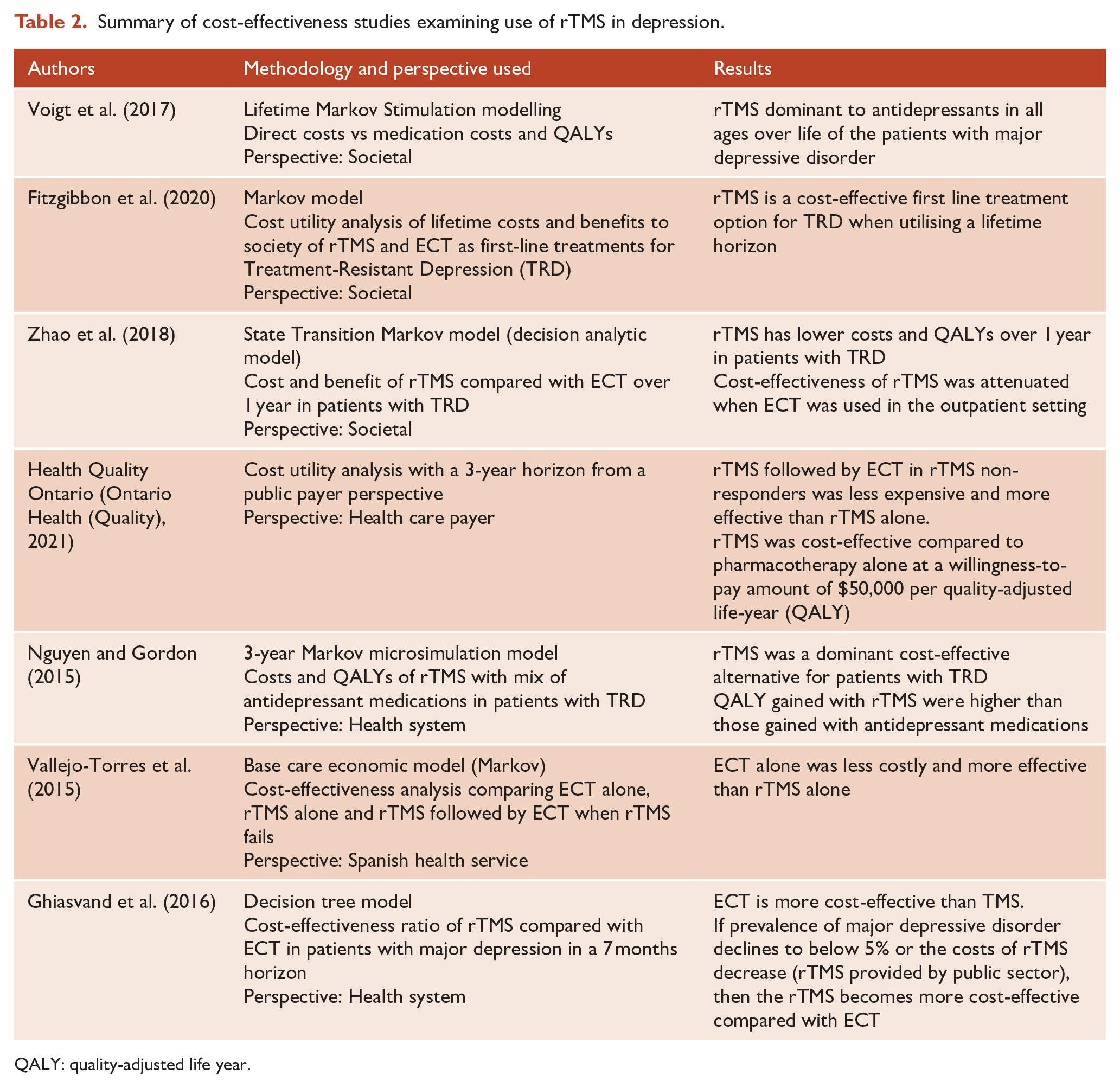
Given that Malhi et al. refer to ‘hidden costs’, we should consider the cost-effectiveness of rTMS in depression. Consistent with the data supporting its efficacy, the cost-effectiveness of rTMS is endorsed in the literature, both in academic studies and those conducted by funding bodies and groups that inform government policy (Nguyen and Gordon, 2015; Teng et al., 2020; Ontario Health (Quality), 2021; Voigt et al., 2017, and see Table 2). For example, an analysis by researchers from Griffith University concluded that rTMS was a ‘dominant/cost-effective alternative for patients with treatment resistant depressive disorder’ and produced higher quality-adjusted life years (QALY) than antidepressant medications at less cost (Nguyen and Gordon, 2015). The most recent analysis, by the independent Ontario Health Technology Assessment group, found that a course of rTMS followed by electroconvulsive therapy (ECT) was significantly more cost-effective than ECT alone, and rTMS was cost-effective compared to antidepressant medication at the frequently used standard of US$50,000 per QALY (Ontario Health (Quality), 2021). Another analysis that specifically evaluated the cost-effectiveness of rTMS if used earlier in the course of treatment and over a patient’s lifetime supported the use of rTMS after a single failed antidepressant trial (Voigt et al., 2017). This analysis also found that rTMS was significantly more cost-effective compared to repeat antidepressant medication trials, resulting in lower costs and better outcomes across a range of age groups. Across these analyses there is a clear indication that rTMS is cost-effective compared to antidepressant medication although there are less consistent findings when comparing rTMS to ECT.
Summary of cost-effectiveness studies examining use of rTMS in depression.
QALY: quality-adjusted life year.
Seeing that these responses and their supporting evidence have been detailed and discussed in recent publications, we also question the scholarly quality of Malhi et al. (2021a) and the value of its publication. It provides no primary data or new analysis. Rather, it revisits assertions that have been rebutted and negated. Clearly inaccurate statements such as the efficacy of rTMS being ‘very much in its infancy’ and that ‘the jury is very much out’ just create doubt and confusion, do not advance meaningful debate and risk damaging the scholarly reputation of the College journal. The journal’s readership and contributors expect its contents to be evidence-based and meet the highest standards of scholarly rigour.
The efficacy of rTMS has been consistently established by independent bodies most recently in Australia by the Medical Services Advisory Committee of the Department of Health. As with all therapies in clinical psychiatry, continued research is required to enhance the efficacy, efficiency and clinical utility of rTMS. We continue to engage in this research and welcome the publication of new, original research and primary evidence that nuances the clinical role and biological mechanisms of rTMS.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: In the last 3 years P.B.F. has received equipment for research from Medtronic Ltd, Neurosoft, Nexstim and Brainsway Ltd. He has served on scientific advisory boards for Magstim and LivaNova and acted as a founder for TMS Clinics Australia and Resonance Therapeutics. C.G. is a researcher with the Ramsay Neurostimulation Service at The Adelaide Clinic. She has no financial conflicts of interest to declare. C.L. is the Director of the TMS clinics at the Black Dog Institute and Ramsay Northside Clinic, Sydney. S.C. is director of TMS Specialists Clinics. F.N. is a private psychiatrist practising in ECT and TMS. K.E.H. is a founder of Resonance Therapeutics. L.C. is founder and co-director of a clinical neuromodulation centre (Qld Neurostimulation Centre). This centre was not involved in the present work. M.B. is the Clinical Investigator on an NHMRC-funded RCT using rTMS to treat OCD. P.C. is Director of TMS, Ramsay Health, S.A. S.G. is a clinician and researcher with the Ramsay Neurostimulation Service at The Adelaide Clinic. S.H. is Co-Director and researcher in the Neuromodulation unit at Sir Charles Gairdner Hospital Mental Health Service. S.P. is Director for ECT at Royal Brisbane and Women’s Hospital. S.S. is the Director of ECT and Neurostimulation (including TMS) at Gold Coast Health. There are no other financial conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: PBF is supported by an NHMRC Investigator Grant (1193596).
