Abstract
Despite more than 25 years of research establishing the antidepressant efficacy of repetitive transcranial magnetic stimulation, there remains uncertainty about the depth and breadth of this evidence base, resulting in confusion as to where repetitive transcranial magnetic stimulation fits in the therapeutic armamentarium in the management of patients with mood disorders. The purpose of this article is to provide a concise description of the evidence base supporting the use of repetitive transcranial magnetic stimulation in the context of the stages of research that typically accompanies the development of evidence for a new therapy. The antidepressant efficacy for the use of repetitive transcranial magnetic stimulation in the treatment of depression has been established through a relatively traditional pathway beginning with small case series, progressing to single-site clinical trials and then to larger multisite randomised double-blind controlled trials. Antidepressant effects have been confirmed in numerous meta-analyses followed more recently by large network meta-analysis and umbrella reviews, with evidence that repetitive transcranial magnetic stimulation may have greater efficacy than alternatives for patients with treatment-resistant depression. Finally, repetitive transcranial magnetic stimulation has been shown to produce meaningful response and remission rates in real-world samples of greater than 5000 patients. The evidence for the antidepressant efficacy of repetitive transcranial magnetic stimulation therapy is overwhelming, and it should be considered a routine part of clinical care wherever available.
Keywords
In 2021, psychiatric treatment of mood disorders such as major depressive disorder (MDD) consists mostly of the use of antidepressant medications, forms of psychotherapy and electroconvulsive therapy, all of which have their basis in developments from over 50 years ago. Repetitive transcranial magnetic stimulation (rTMS) has been under development as a novel antidepressant treatment for over 25 years, although in Australia it is only relatively recently becoming a more routine part of clinical practice. The development and evaluation of rTMS over such an extended time span of clinical and regulatory development have contributed to a lack of clarity in the broader psychiatric community as to the role of rTMS and to the evidence base that supports its use, as views have formed about its value at various points in time and do not necessarily reflect the quantity and quality of data currently available informing its use. The purpose of this article is to provide a concise and accessible overview of the evidence supporting the use of rTMS treatment in the management of patients with MDD so that readers can make informed conclusions as to the potential benefit of this therapy. This is timely and important given the context of the recent Australian Federal government decision to fund public provision of rTMS treatment of MDD through the Medicare Benefit Scheme.
Developing and evaluating evidence
To do this, it is important to use a valid framework for evaluating the available evidence when considering the efficacy of rTMS or any intervention. A variety of approaches have been proposed for rating the levels of research evidence following an original report by the Canadian Task Force on Periodic Health Examinations (1979) which described four levels of evidence from expert opinion through to a randomised controlled trial with proper randomisation. Similar approaches have been developed by the Oxford Centre for Evidence-Based Medicine (CEBM) (UK), the National Health and Medical Research Council (NHMRC; Australia) and others.
Evidence accumulates over several phases. A new potential treatment will be first evaluated in case reports or case series or potentially in an open-label clinical trial, and then one or more placebo-controlled randomised trials are typically conducted. The results of clinical trials are then typically synthesised in systematic reviews and meta-analyses. The process of evidence synthesis is also increasingly recommended to include evaluation of the validity and reliability of the accumulated evidence such as the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach recommended by many organisations, including the National Institute for Health and Care Excellence (NICE) in the United Kingdom, the Canadian Task Force on Preventive Health Care and the World Health Organization (WHO).
Network meta-analysis are a relatively recent and important addition to the tools available when assessing the evidence available for a particular condition. For most disorders, there are likely to be two or more potential interventions of interest, and repeatedly comparing treatments to one another in multiple meta-analyses is not likely to be a useful approach. Network meta-analysis techniques were developed to allow comparisons of more than two interventions in a single analysis to produce consistent estimates of the relative effects of all interventions compared to each other (Dias and Caldwell, 2019).
The final method used in the synthesis of research evidence is that of the umbrella review (Aromataris et al., 2015). This involves the synthesis of information from the highest forms of evidence available for a treatment, namely, other systematic reviews and meta-analysis. Finally, but beyond the synthesis of existing data, evaluation of a new intervention can also be informed by the collection of post-approval outcomes.
The evidence for the effectiveness of rTMS in depression
So what evidence has been collected to date evaluating the potential use of rTMS therapy for patients with MDD? (see Figure 1). The first outcome data were collected in a series of case reports (e.g., George et al., 1995) and non-controlled (e.g., Conca et al., 1996) and controlled (George et al., 1996; Pascual-Leone et al., 1996) pilot clinical trials which suggested that rTMS did produce antidepressant benefits. These studies were followed by a very large number of small investigator-initiated single-site sham-controlled clinical trials. Not unsurprisingly, given the number of trials and the limited sample size/power of most, not all of these single-site small studies showed beneficial effects, but the vast majority did show a benefit of active rTMS therapy over sham stimulation. These studies also provided important information in regard to the benefits of different forms of rTMS and the safety of this intervention. Of note, the methods of sham control in these trials have progressively improved over time (see Box 1), and many have reported the success of blinding (e.g., George et al., 2010), a rare feature in trials reporting the success of other types of treatments in mental health conditions.
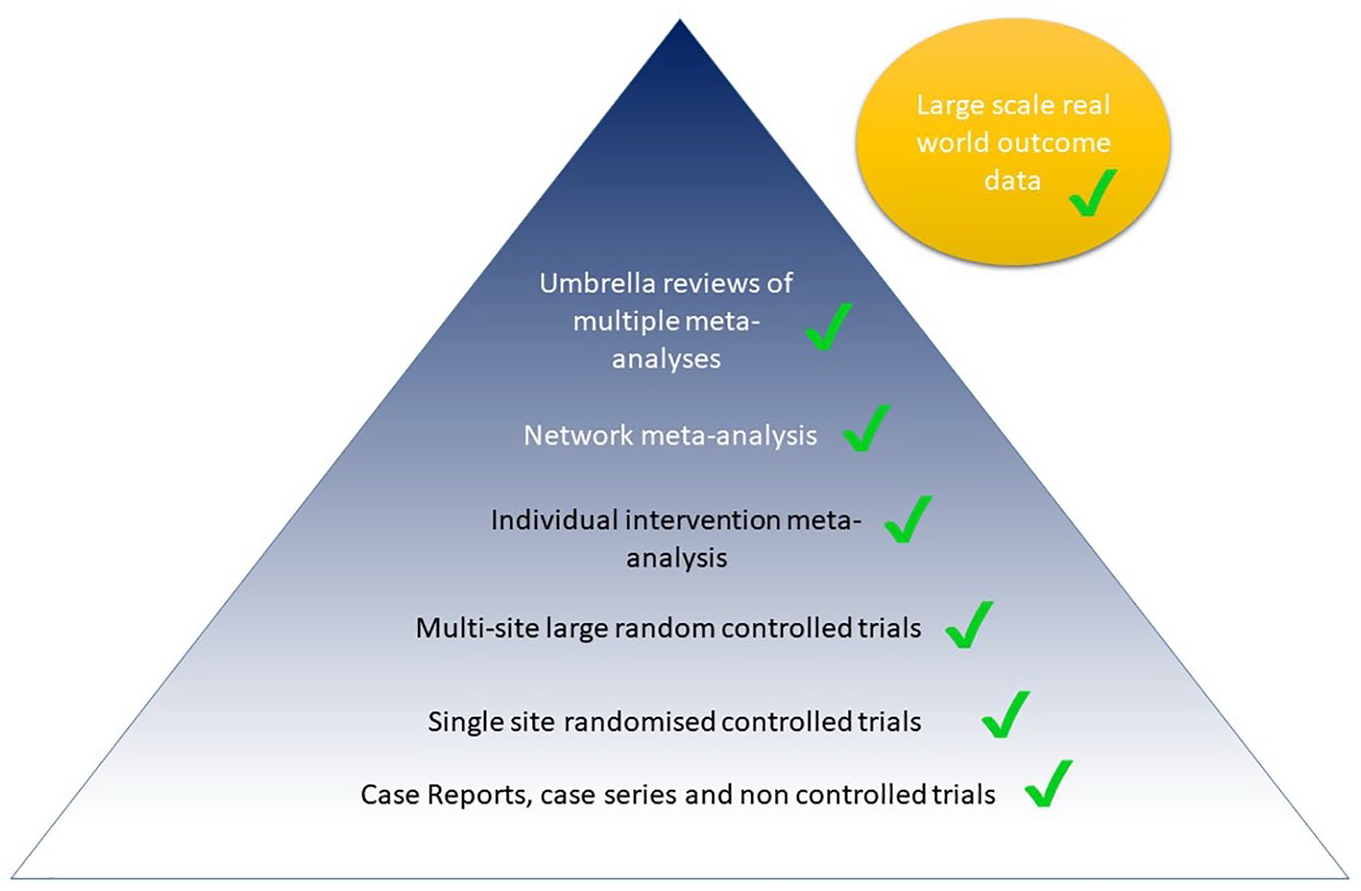
Evidence for the use of rTMS in depression.

TMS: transcranial magnetic stimulation; rTMS: repetitive transcranial magnetic stimulation.
In the early to mid-2000s, two of the next evidence ‘steps’ started to occur simultaneously: the conduct of larger multisite clinical trials and meta-analyses. The first and most significant multisite clinical trial was conducted in 300 patients in the United States, Canada and Australia and showed positive antidepressant effects of rTMS that were sufficient to lead to regulatory approval by the Food and Drug Administration (FDA) in the United States in 2008 and created a de facto standard for rTMS administration methods in regard to dose and other stimulation parameters (O’Reardon et al., 2007). The use of these stimulation parameters was adopted in a second, this time independently (National Institute of Mental Health [NIMH]) funded, clinical trial conducted in the United States and published in 2010, which also showed positive antidepressant effects (George et al., 2010).
The first meta-analyses of antidepressant rTMS clinical trials were published back in 2001 and 2002, although these included a very limited number of trials. The meta-analysis by McNamara et al. included five trials (McNamara et al., 2001), the study by Holtzheimer et al. (2001) included 12 trials and the largest, that of Martin et al. (2002), included 14 trials (
Since the early 2000s, the meta-analyses of rTMS studies in depression have grown, both in substance and in the level of confidence with which they can make conclusions about rTMS treatment. By 2010, Slotema et al. were able to publish a meta-analysis of 34 trials including 1383 patients randomised to active or sham stimulation. This included studies in which patients were free of antidepressant medication (number of studies = 7), stable on medication (
Over the last 10 years, a considerable number of new meta-analyses have been published exploring the use of rTMS therapy: these have increasingly focused on addressing specific questions about the application of the technique, rather than just exploring the overall efficacy. For example, Teng et al. used meta-analytic techniques to demonstrate that greater antidepressant effects were achieved with longer periods of treatment but modest doses of stimulation per day (Teng et al., 2017), and Wei et al. showed that there was no difference between active and sham groups in regard to dropout rates in rTMS studies, indicating a high degree of acceptability and tolerability of treatment (Wei et al., 2017). In addition, a series of meta-analyses have been conducted exploring the relative efficacy of several different forms of rTMS therapy. Although the vast majority of rTMS studies have been conducted using high-frequency stimulation applied to the left dorsolateral prefrontal cortex (DLPFC), a significant number of studies have also investigated stimulation of the right DLPFC and the sequential bilateral application of both of these approaches, as well as a number of novel paradigms including priming rTMS (pTMS) and theta burst stimulation (TBS). Independent meta-analyses have demonstrated the antidepressant benefits of low-frequency right-sided rTMS (Berlim et al., 2013b; Schutter, 2010) and sequential bilateral rTMS (Berlim et al., 2013a; Sehatzadeh et al., 2019), as well as the standard left high-frequency approach.
In recent years, a number of network meta-analysis have been conducted in which rTMS has been a treatment of focus. Different forms of rTMS were compared to investigate whether there was evidence of differential efficacy by Brunoni et al. (2017) in a study including 81 rTMS trials with a total of 4233 patients. High-frequency left-sided, low-frequency right-sided, sequential bilateral rTMS and pTMS were all found to be more effective than sham stimulation for both response and remission rates.
The second form of network meta-analysis published including rTMS studies has been the exploration of a range of interventions for patients with treatment-resistant depression (TRD). In a study notably published by authors from the pharmaceutical industry, Papadimitropoulou et al. (2017) looked at the antidepressant effects of a variety of drugs and non-drug treatments at 2, 4, 6 and 8 weeks after commencement of the intervention. rTMS was clearly the superior intervention when considering the 4- and 6-week outcomes overall (although this was not mentioned in the abstract). For example, in regard to response rates after 6 weeks of therapy, rTMS was the only treatment where there were significant effects, and rTMS also ‘ranked first’ in regard to remission rates at the same time point.
A recent umbrella review has also evaluated the evidence for the efficacy of rTMS using the rigorous GRADE approach to assess the quality of evidence across existing meta-analysis (Razza et al., 2020). ‘High-quality evidence’ was found to support the efficacy of high-frequency left-sided rTMS and sequential bilateral rTMS.
The final form of evidence worthy of consideration, as indicated above, is information on the outcomes of a treatment once it has been used in real-world clinical practice. A considerable number of these studies have been published, but these have been universally dwarfed in scale by a recent publication. This study described the outcomes of 5010 patients treated at 103 practices (Sackeim et al., 2020) on an intention-to-treat basis (ITI) and for treatment completers who had received at least 20 rTMS sessions. Response and remission rates were 58% and 28% in the ITI group and 83% and 62% in patients who completed at least 20 treatments – clearly clinically meaningful effects.
In understanding this literature, it is important to note that the vast majority of trials demonstrating the effectiveness of treatment have been conducted in patients with significant levels of TRD. Although there is evidence that rTMS can be effective in patients earlier in their illness course (Voigt et al., 2019), the vast majority of trials are being conducted in TRD. For example, in an analysis that we conducted by pooling results from over 1100 patients in a series of clinical trials, we found a response rate of over 45% in a group of patients who averaged more than 5.5 failed medication trials (Fitzgerald et al., 2016).
There is one additional format in which evidence is synthesised but which is not typically read by most clinicians. These are the reports developed as part of the process of the evaluation of new treatments for regulatory approval or funding. These evaluations are typically systematic, comprehensive and utilise a number of methods of systematic review and meta-analysis described above. For example, rTMS has been recommended for public funding through the Medical Services Advisory Committee in Australia based on a formal application sponsored by the Royal Australian and New Zealand College of psychiatrists. Funding has been approved by the Federal government and should be implemented in late 2021.
A comprehensive assessment of the effectiveness, safety and cost-effectiveness of TMS therapy was recently published by the Ontario health technology assessment group in Canada (Ontario, 2021). This assessment included the evaluation of 58 primary studies and nine systematic reviews and network meta-analysis and was subject to evaluation using the GRADE framework. The authors of this independent and comprehensive evaluation concluded that ‘
Tolerability and safety
Fortunately, rTMS therapy has an excellent safety record and tolerability. The only side effects shown to be reported in more than 5% of patients undergoing rTMS in a recent meta-analysis were discomfort at stimulation site or headache (~20% of patients) (Zis et al., 2020). Most tellingly, the discontinuation rates in patients having real rTMS in clinical trials are actually numerically lower (not statistically different) from the rates seen in sham-treated patients (2.7%) (Zis et al., 2020). These rates are dramatically lower than typical antidepressant medication discontinuation rates. There are no long-term adverse consequences of rTMS therapy as seen with other options for patients with treatment-resistant depression, such as the metabolic consequences of atypical antipsychotics or the risk of renal or thyroid impairment with lithium therapy. There is a small risk of seizure induction during rTMS treatment, although this is very low – less than 0.02 seizures per 1000 treatment sessions (Lerner et al., 2019).
Ongoing challenges
As can be inferred by the results of the studies described, rTMS has robust and clinically meaningful antidepressant efficacy, and this has recently been of sufficient significance to warrant approval of public funding in Australia through the Medicare Benefit Scheme. Multiple rTMS devices are approved for clinical use by the Therapeutics Goods Administration (TGA) in Australia and listed on the Australian Register of Therapeutic Goods (ARTG). However, and unfortunately, like all other existing treatments for depression, rTMS is not ‘curative’ and there are significant ongoing issues with its use and implementation that require further research and clinical development in parallel to its expansion into more widespread clinical applications. For example, it is clear that engagement in rTMS is a time-consuming commitment for patients, and this is associated with meaningful financial costs. There is considerable research, including that being led in Australia (Fitzgerald et al., 2018, 2020), underway to try and develop more rapidly active and efficient methods of rTMS administration, but these are not yet ready for mainstream clinical use. In addition, although rTMS has been shown to be as effective as other options for patients with TRD (although it is worthy of note that meaningful comparisons of the efficacy of rTMS to ECT have not been conducted [Fitzgerald, 2020] and ECT remains the treatment of choice for the most severe forms of depression, including depression with psychotic features), we believe that efficacy can be improved. There is significant ongoing research aimed at improving efficacy through developments like the enhanced individualisation of treatment parameters such as targeting (Cash et al., 2020) and potentially the use of neurophysiological tools such as electroencephalography (EEG) to aid in the prediction of the likelihood of successful clinical response (Bailey et al., 2018, 2019).
Clearly in parallel with these activities, there is a pressing need to educate the psychiatric and medical community in regard to the role of rTMS. Clearly, the vast majority of currently practicing psychiatric clinicians will have received limited training in regard to the use of rTMS therapy, and as such, there is a considerable need to expand knowledge and understanding through a variety of educational and training pathways.
Conclusions
Clearly, rTMS therapy for depression has been evaluated across a wide range of research studies commencing with a series of small clinical trials and progressing to larger multisite trials. The research evidence supporting its efficacy has been summarised in numerous meta-analyses and subsequently in umbrella reviews and network meta-analyses. Individual meta-analyses and the recent umbrella review (Razza et al., 2020) have clearly confirmed that rTMS is an effective treatment for patients with MDD and is safe and well tolerated. Network meta-analysis have confirmed that there are several forms of rTMS that are effective and importantly that it has comparable, if not superior, efficacy when compared to other interventions for patients with TRD.
Although rTMS is by no means a perfect treatment and ongoing developmental work is required to enhance its efficacy and efficiency of clinical delivery, we think that the evidence presented above, collated progressively over the last 25 years of research, clearly shows that it has a meaningful and substantial place now in the management of patients with TRD. It should be made available to and be presented to patients as an alternative treatment option when there has been insufficient clinical benefit from initial trials of antidepressant medication (see Figure 2 and Table 1). The evidence for, and place of, rTMS in treatment should be reflected in clinical practice guidelines, clinical service provision, and the education and training of psychiatric clinicians. The comprehensive research conducted in the development of rTMS by groups all around the world has convincingly established that it is an effective and valuable treatment. The focus now should be on how to make rTMS most appropriately available and accessible to as wide a range of suitable patients as possible.
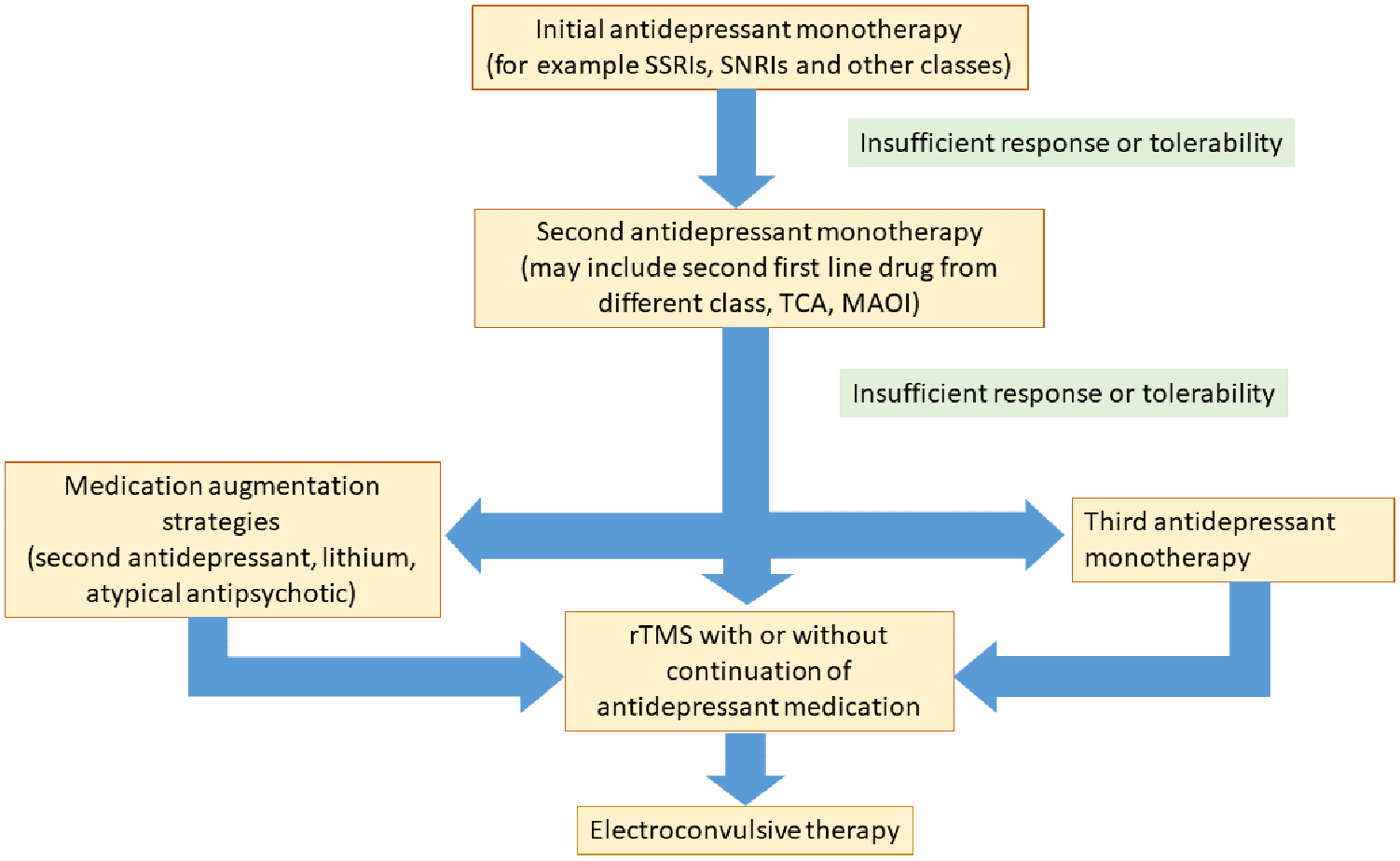
The place of rTMS in the biological treatment of patients with treatment-resistant depression.
Factors contributing a decision to provide rTMS treatment following two failed medication trials.

rTMS: repetitive transcranial magnetic stimulation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: In the last 3 years, P.B.F. has received equipment for research from Nexstim and Brainsway Ltd. He is a founder of rTMS Clinics Australia. M.S.G. has received equipment for research from Brainsway and Magstim. P.B.F. is supported by an NHMRC Investigator Grant (1193596).
