Abstract
Objective:
To provide a qualitative view and quantitative measure of sleep disturbances across and between early stages – clinical ultra high-risk and first episode – of psychotic and bipolar disorders.
Methods:
Electronic databases (PubMed, Cochrane, Embase, PsychINFO) were searched up to March 2021 for studies comparing sleep measures between individuals with an early stage and controls. Standard mean deviations (Cohen’s d effect sizes) were calculated for all comparisons and pooled with random-effects models. Chi-square tests were used for direct between-subgroups (ultra high-risk vs first episode) comparisons of standard mean deviations. The effects of age, sex ratio, symptoms and treatment were examined in meta-regression analyses.
Results:
A database search identified 13 studies that contrasted sleep measures between individuals with an early stage (N = 537) and controls (N = 360). We observed poorer subjective sleep quality (standard mean deviation = 1.32; 95% confidence interval, [1.01, 1.62]), shorter total sleep time (standard mean deviation =−0.44; 95% confidence interval, [−0.67, −0.21]), lower sleep efficiency (standard mean deviation = −0.72; 95% confidence interval, [−1.08, −0.36]), longer sleep onset latency (standard mean deviation = 0.75; 95% confidence interval, [0.45, 1.06]) and longer duration of wake after sleep onset (standard mean deviation = 0.49; 95% confidence interval, [0.21, 0.77]) were observed in early stages compared to controls. No significant differences were observed for any of the reported electroencephalographic parameters of sleep architecture. No significant between-subgroups differences were observed. Meta-regressions revealed a significant effect of the age and the antipsychotic status on subjective measures of sleep.
Conclusion:
The early stage population presents with significant impairments of subjective sleep quality continuity, duration and initiation. Systematic assessments of sleep in early intervention settings may allow early identification and treatment of sleep disturbances in this population.
Introduction
Early diagnosis and intervention in psychiatry receive growing recognition and support (McGorry et al., 2018). In clinical staging models, early phases of psychotic and bipolar disorders have been coined as ‘High-Risk’ status and ‘First-Episode’ (FE). The High-Risk status (also referred as ‘ultra high-risk’ – UHR) is an inclusive construct that captures potentially prodromal psychiatric symptoms in young help-seekers (Fusar-Poli et al., 2013; Geoffroy and Scott, 2017). An FE is considered to be the first point of an established diagnosis of psychotic or bipolar disorder. In addition to symptoms, both UHR and FE statuses are associated with severe cognitive deficits that alter performance on everyday life activities (Donde et al., 2020; Lavallé et al., 2020; Fusar-Poli et al., 2012, 2013). Half of the individuals with an UHR status will convert to a psychotic or bipolar disorder, while the remaining will either convert to other disorders or return to a full remission (Fusar-Poli et al., 2017; Piras et al., 2014). Descriptions of early stages provide significant advances for accurate early detection, prediction of the risk to conversion and development of tailored early interventions services (McGorry, 2015). However, high variability of outcomes combined with a substantial risk of disability are still observed at early stages, which stresses the need for more fine-grained clinical profiling of these specific populations (Bozzatello et al., 2019).
We believe that focusing on sleep disturbances, a very common and shared at-risk clinical dimension of psychiatric disorders, is a promising and unique opportunity to enrich the clinical evaluation of early stages, as well as to propose early personalized interventions including chronotherapeutics. Sleep is a multifactorial phenomenon that results from the interaction of complex hormonal, homeostatic and neurobiological processes. Disturbances of sleep affect one out of five adults worldwide (Ohayon, 2011). Sleep disturbances are associated with an increased risk of cardiometabolic disorders, including hypertension, myocardial infarction, stroke and morbid obesity (Bacaro et al., 2020; Larsson and Markus, 2019; Nuyujukian et al., 2019; Viot-Blanc and Levy, 2006). Significant sleep complaints are experienced by up to 80% of remitted patients with psychotic and bipolar disorders (Geoffroy and Gottlieb, 2020; Geoffroy et al., 2015; Kaskie et al., 2017; Laskemoen et al., 2019; Meyer et al., 2020). In parallel, these disorders are associated with several objective disturbances of sleep-wake cycle as shown by polysomnography and actigraphy investigations (Baglioni et al., 2016; Castelnovo et al., 2018; Chan et al., 2017; Geoffroy et al., 2015; Zangani et al., 2020). Severity of sleep disturbances correlates with worse cognitive performances and poorer functional outcomes (Kaskie et al., 2019; Morton and Murray, 2020) and has been demonstrated as a possible risk factor for onset and relapse of psychotic and mood disorders (Geoffroy et al., 2020; Melo et al., 2016; Pancheri et al., 2019; Soehner et al., 2019). In addition, sleep problems might strongly contribute to the 10–15 years reduction of life expectancy demonstrated in patients (Hayes et al., 2017).
Sleep disturbances can be measured as sleep complaints according to sleep questionnaires, sleep continuity, duration and initiation parameters modifications according to actigraphy or polysomnography, and sleep architecture modification according to polysomnography. High-quality reviews and meta-analyses have already demonstrated significant disturbances of these measures in UHR and FE populations (Clarke et al., 2020; Davies et al., 2017). However, no quantitative analyses to date have synthesized sleep measures differences including early stages all at once, as well as comparisons between those stages. In parallel, no biomarkers are yet available to distinguish between clinical stages and help create profiles to enhance prediction and therapeutic selection in early intervention services (McGorry et al., 2014). These are important issues as more and more early intervention services provide care for both UHR and FE populations (Kollias et al., 2020; Leuci et al., 2020; Pelizza et al., 2019). In addition, the effect of potential confounders such as treatment effect on sleep has not been investigated in early stages. Finally, new studies have been published since the last reviews and allow new insights and possible analyses.
To address these gaps, we conducted a systematic review and meta-analysis of the literature with three aims:
To provide a qualitative view and quantitative measure of sleep disturbances across early stages of psychotic and bipolar disorders;
To compare sleep markers between different early stages;
To examine the effect of potential moderators on the observed effect sizes.
We believe that a better understanding of sleep disturbances in patients at the early stages of psychotic and bipolar disorders could contribute to the development of better-targeted treatments in this specific population.
Methods
Recommendations of the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) guidelines for systematic review and meta-analysis were followed (Moher et al., 2015). The protocol was previously registered at the PROSPERO register (#192164, Protocol S1) (Chien et al., 2012).
Literature search strategy
Eligibility
Inclusion criteria were as follows:
English or French language studies published in peer-review journals, with article types suitable for inclusion in a systematic review and meta-analysis (all except conference posters, reviews, meta-analyses, unpublished studies, opinions and comments);
Studies including participants with a clinical UHR for psychosis (UHR-P status diagnosed with the Structured Interview for Prodromal Syndromes Criteria [SIPS]; Miller et al., 2003) or the Comprehensive Assessment of At-Risk Mental States (CAARMS; Yung et al., 2005), a clinical UHR for bipolar disorder (UHR-BD status, diagnosed using specific criteria developed by the ORYGEN Youth Health group in Australia; Bechdolf et al., 2010), or an established clinical diagnosis of FE of psychosis (FE-P), FE of schizophrenia (FE-SZ) or FE of bipolar disorder (FE-BD) (criteria of the Diagnostic and Statistical Manual of Mental Disorders [DSM] III to 5 or the International Classification of Diseases [ICD]-9 to 10, in which FE-P corresponds to a duration of psychosis between 1 and 6 months, FE-SZ between 6 and 24 months, and FE-BD to a duration of hypomania or mania more that 4 and 7 days, respectively);
Studies including control participants without any established clinical diagnosis of psychiatric and neurological condition according to the DSM or the ICD;
Subjective sleep outcomes as assessed using validated rating scale; and/or sleep continuity, duration or initiation outcomes as measured with actigraphic or polysomnographic records; and/or neurophysiological outcomes of sleep architecture as measured with polysomnography;
Studies with a between-group design (i.e. comparison of sleep measures between patients and/or control groups) and providing sufficient statistical indices for sleep outcomes.
Search strategy
We searched the PubMed and additional databases (Cochrane, Embase and PsychINFO) using combinations of keywords referring to early stages and sleep with no limitation of date until March 2021 (Figure 1):
Early stage: ‘first episode affective’ OR ‘first episode depression’ OR ‘first episode mania’ OR ‘first episode psychosis’ OR ‘ultra high risk’ OR ‘UHR’ OR ‘at risk mental state’ OR ‘ARMS’ OR ‘clinical high risk’ OR ‘CHR’ OR ‘early psychosis’ OR ‘bipolar trait’ OR ‘bipolar vulnerability’ OR ‘bipolar trait’ OR ‘bipolar vulnerability’ OR ‘help-seeking’ OR ‘help-seeker’ OR ‘schizotypy’ OR ‘early intervention’.
Sleep: sleep OR insomnia OR circadian OR sleepiness OR wakefulness OR hypersomnolence OR drowsiness OR somnolence OR sleepy OR drowsy OR actigraph* OR polysomnograph*.
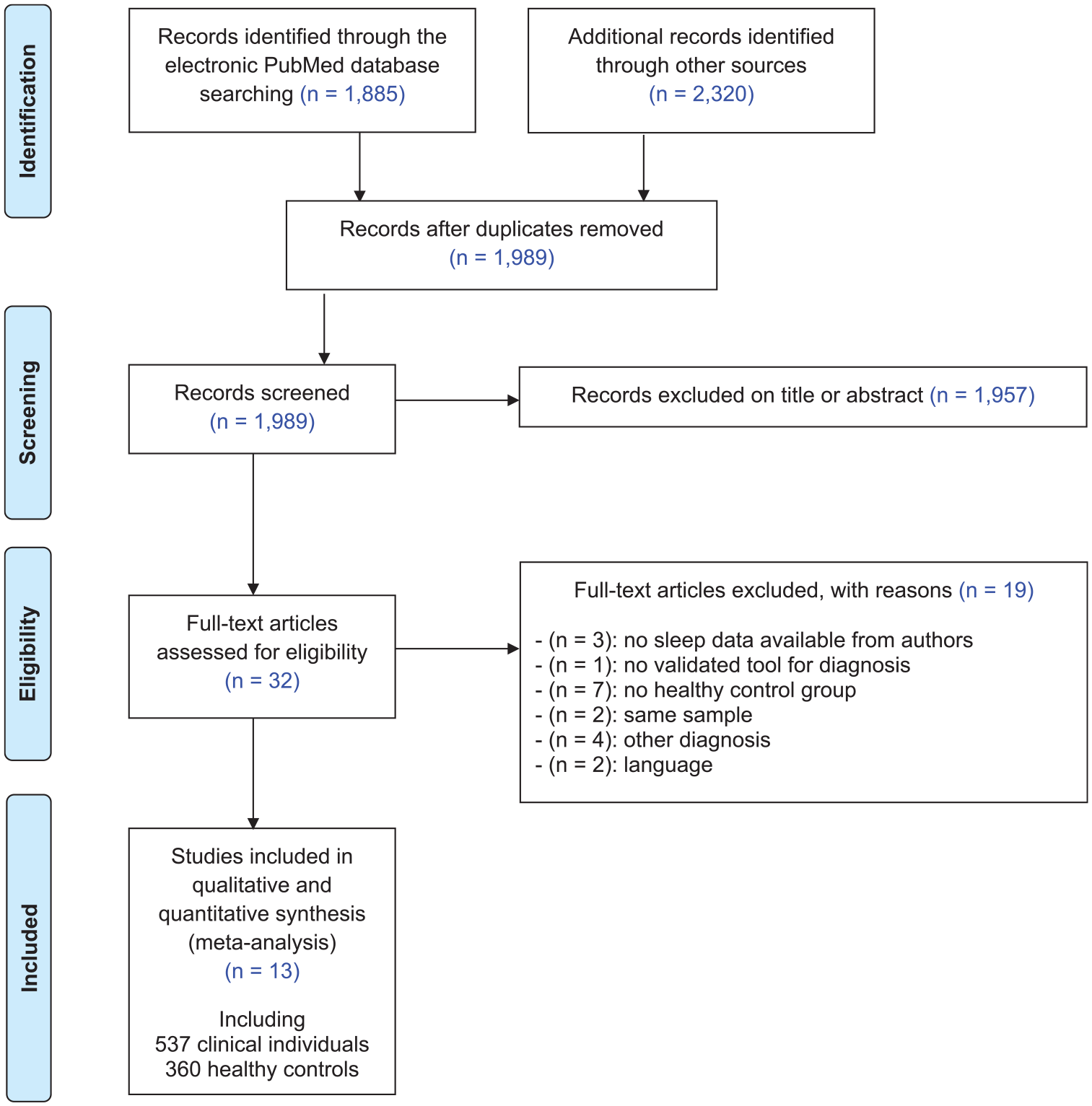
PRISMA flow diagram for the systematic review and meta-analysis.
After excluding duplicate references, two reviewers (C.D., A.J.) independently screened the title and abstract of each study identified by the search and applied the inclusion and exclusion criteria. Following this first screen, the same procedure was followed for full texts of eligible studies. The ‘similar articles’ findings in PubMed and the reference lists of identified studies were reviewed for additional references, although none were identified in this manner.
Data extraction
The following data were extracted when present: (1) population and clinical data (sample size, mean age, gender ratio, diagnosis, clinical symptoms), (2) statistical indices regarding subjective sleep outcomes as assessed using validated rating scale, and/or sleep continuity, duration and initiation outcomes as measured with actigraphy or polysomnography recordings and/or neurophysiological outcomes of sleep architecture as measured with polysomnography. We ensured that different participants were included in the different reports from the same research groups.
Quality assessment
To measure the overall quality of the included references, a global rating score was calculated for each study using the Standard Quality Assessment (QualSyst tool; Kmet et al., 2004).
Meta-analysis
Outcomes
All statistical analyses were carried out using jamovi software version 1.2. When provided, we extracted means and standard deviations (SD) for sleep outcomes in both patient and control groups. We calculated Cohen’s d effect sizes (also referred as standardized mean differences, SMDs) with 95% confidence intervals (95% CIs) for each sleep outcome when present in ⩾3 studies. We interpreted the magnitude of the effect sizes using Cohen’s interpretative guidelines (0.2 = small; 0.5 = medium; 0.8 = large effect size; Cohen, 1988). As methods and sample characteristics differed across included studies, a random-effect model through the restricted maximum likelihood (REML) method was used to synthesize the results.
Heterogeneity
Global heterogeneity of summary SMD was quantified using Higgins I2 statistics with the values of 25%, 50% and 75% reflecting a small, medium and large degree of heterogeneity, respectively (Higgins et al., 2003). Higgins I2 p-value was calculated from a Cochran’s Q-test.
Publication bias
Publication bias was assessed by visual inspection of the funnel plots. In addition, we calculated a Rosenthal’s fail-safe N (Orwin, 1983; Rosenthal, 1979) to explore if some statistically non-significant studies missing from an analysis would reduce the observed effect (SMD) to a level not statistically significantly different from zero if included. To this end, Rosenthal’s method calculates the ‘fail-safe’ number of additional studies (fail-safe N) with mean null result necessary to reduce the combined significance to a p = 0.05 (McDaniel et al., 2006). In other words, the fail-safe N is the number of missing studies averaging a z-value of zero that should be added to make the effect statistically non-significant. The fail-safe N is considered robust against publication bias when N > 5n + 10, where N = the number of published studies (Rosenberg, 2005).
Meta-regressions and subgroup analyses
Subgroup analyses according to patient status were performed for each outcome. Chi-square tests were used for direct between-subgroups comparisons (UHR vs FE) of SMDs. To explore the influence of other factors than patient status on effect sizes, we then investigated the effect of four continuous variables when present in ⩾3 studies using meta-regressions: (1) age, (2) sex (%male), (3) symptoms severity (validated rating scale score) and (4) antipsychotic medication (%treated).
Results
Literature search
As shown in Figure 1, the initial search returned 1989 references after duplicate removed. Following preliminary screening of the titles and/or abstracts, 1762 were excluded accordingly. Among the 32 references that were reviewed in detail, 13 independent studies were selected for systematic review and meta-analysis including 537 individuals with an early stage and 360 healthy controls (Fekih-Romdhane et al., 2020; Guenole et al., 2014; Kaskie et al., 2019; Lederman et al., 2017; Lunsford-Avery et al., 2015; Manoach et al., 2014; Poe et al., 2017; Poulin et al., 2003; Riemann et al., 1995; Stowkowy et al., 2020; Yazihan and Yetkin, 2020; Zanini et al., 2015).
Characteristics of selected studies
Among the 13 studies retained for the systematic review and meta-analysis, 4 studies assessed UHR samples (Lunsford-Avery et al., 2015; Poe et al., 2017; Stowkowy et al., 2020; Zanini et al., 2015), 9 examined FE samples (Fekih-Romdhane et al., 2020; Forest et al., 2007; Guenole et al., 2014; Kaskie et al., 2019; Manoach et al., 2014; Poulin et al., 2003; Riemann et al., 1995; Yazihan and Yetkin, 2020) and 1 involved both (Lederman et al., 2017). UHR samples were UHR-P and diagnosed by the CAARMS for 1 study (Lederman et al., 2017) or the SIPS for 3 studies (Lunsford-Avery et al., 2015; Poe et al., 2017; Stowkowy et al., 2020), or both UHR-P and UHR-BAP as diagnosed using OYH criteria for 2 studies (Stowkowy et al., 2020; Zanini et al., 2015). FE samples were FE-P with a duration of psychosis from 1 to 6 months in 5 studies (Forest et al., 2007; Guenole et al., 2014; Kaskie et al., 2019; Poulin et al., 2003; Yazihan and Yetkin, 2020), or FE-SZ with a duration of psychosis from 6 to 24 months in 4 studies (Fekih-Romdhane et al., 2020; Lederman et al., 2017; Manoach et al., 2014; Riemann et al., 1995). FE was diagnosed using DSM criteria in all 13 retained studies. No studies involving FE-BAP were identified by the literature search.
Subjective sleep was assessed using the Pittsburgh Sleep Quality Index (PSQI) total score in six studies that provided subjective sleep data (Fekih-Romdhane et al., 2020; Lederman et al., 2017; Lunsford-Avery et al., 2015; Poe et al., 2017; Stowkowy et al., 2020; Zanini et al., 2015). Two studies used the Epworth Sleepiness Scale, which was not sufficient for meta-analysis (Fekih-Romdhane et al., 2020; Zanini et al., 2015). Sleep continuity, duration, initiation and sleep architecture were measured in nine studies among which one used actigraphy (Lunsford-Avery et al., 2015) and eight used polysomnography recordings (Forest et al., 2007; Guenole et al., 2014; Kaskie et al., 2019; Manoach et al., 2014; Poulin et al., 2003; Riemann et al., 1995; Yazihan and Yetkin, 2020; Zanini et al., 2015). Details of the included studies are provided in Table 1.
Characteristics of the studies included in the meta-analysis.
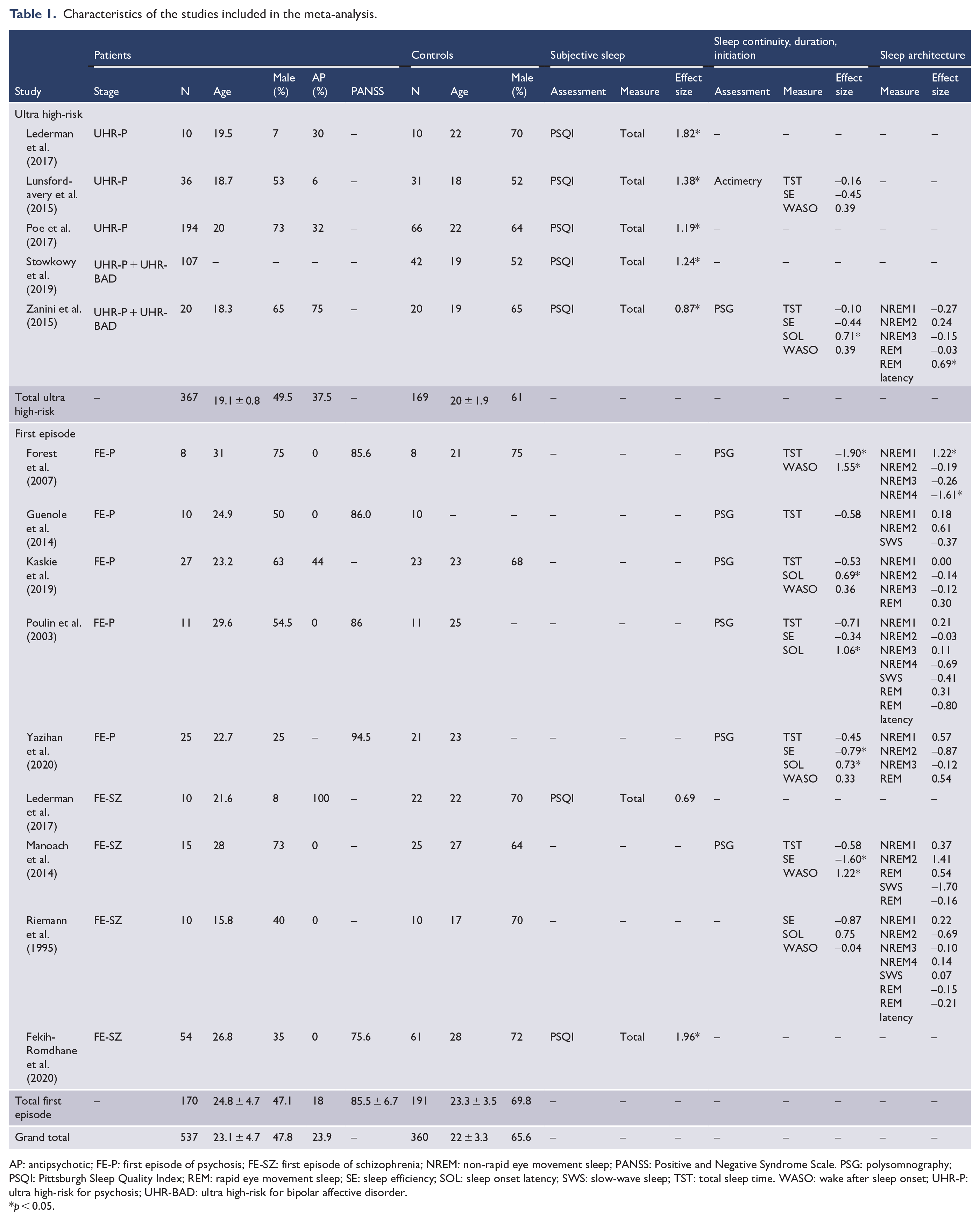
AP: antipsychotic; FE-P: first episode of psychosis; FE-SZ: first episode of schizophrenia; NREM: non-rapid eye movement sleep; PANSS: Positive and Negative Syndrome Scale. PSG: polysomnography; PSQI: Pittsburgh Sleep Quality Index; REM: rapid eye movement sleep; SE: sleep efficiency; SOL: sleep onset latency; SWS: slow-wave sleep; TST: total sleep time. WASO: wake after sleep onset; UHR-P: ultra high-risk for psychosis; UHR-BAD: ultra high-risk for bipolar affective disorder.
p < 0.05.
Overall, the quality assessment was satisfactory (mean: 20.7 ± 1.18). Total scores and details of this assessment are given in Supplemental Table S1.
Meta-analyses
Subjective sleep
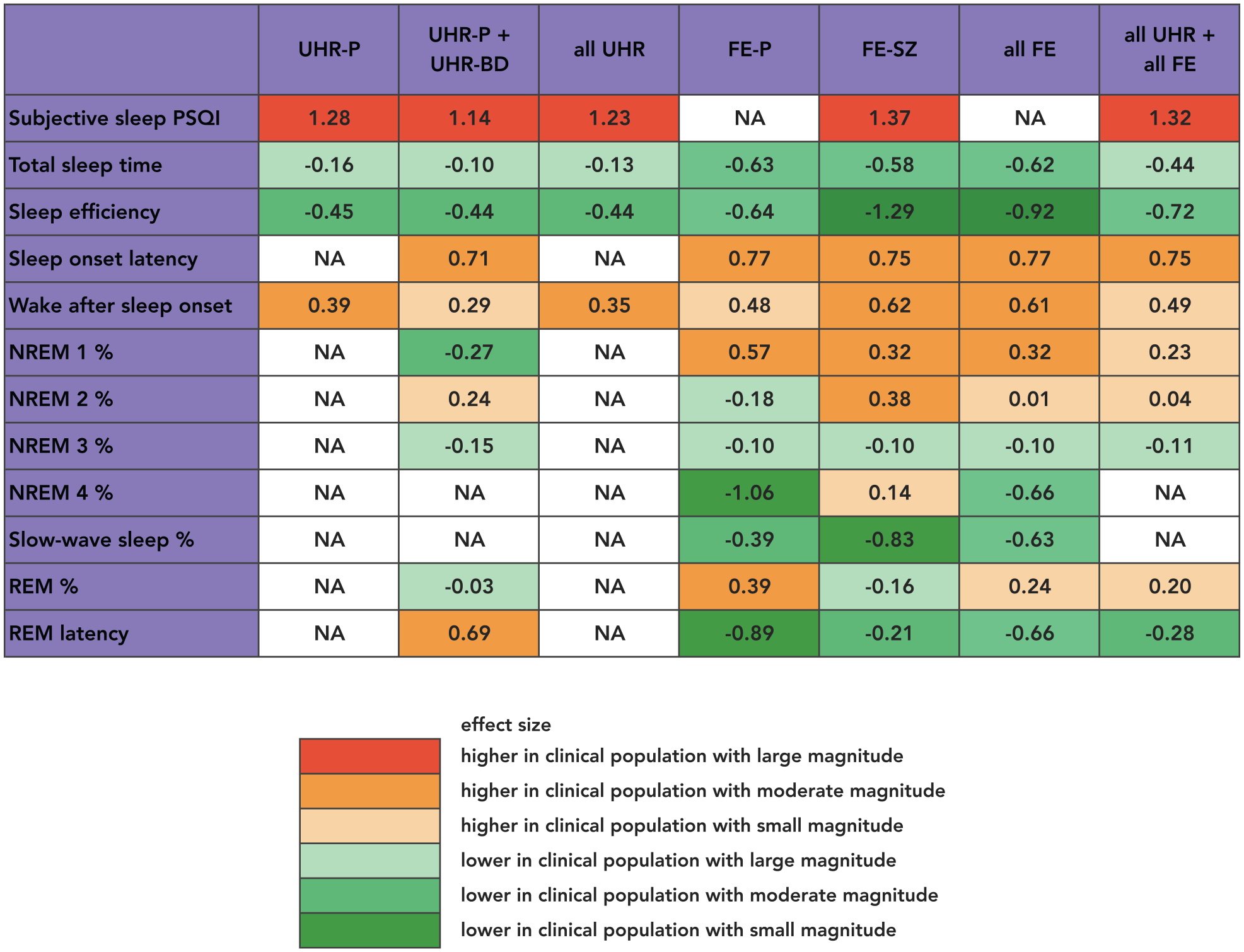
Individuals with early stages of psychotic or bipolar disorders had significant higher PSQI scores (i.e. worse sleep quality) with a large effect size [SMD(k(number of studies)=7) = 1.32 (1.01; 1.62), p < 0.001; I2 = 58%; Q-test p = 0.03]. Heterogeneity was recalculated post hoc with UHR-P + UHR-BD studies excluded (I2 = 64%; Q-test p = 0.02). Funnel plot was asymmetrical (Supplemental Figure S4), suggesting a publication bias which was not confirmed by robust fail-safe N (N = 414). Significant higher PSQI scores were confirmed for UHR individuals (k = 5), as well as for UHR-P individuals (k = 2), mixed groups of UHR-P + UHR-BD individuals (k = 2) and FE-SZ individuals (k = 2). PSQI could not be measured in FE-P individuals (Figure 2, Supplemental Figure S1).
Meta-analyses effect sizes (Cohen’s d) of comparisons of sleep measures between individuals at early stages of psychotic and bipolar disorders and healthy controls.
Sleep continuity, duration and initiation
Significant differences were observed between individuals with early stages of psychotic or bipolar disorders (UHR, 2 studies; FE, 7 studies) compared to healthy controls for each of the measured parameters of sleep continuity, duration and initiation (Figure 2, Supplemental Figure S2). More specifically, we observed in early stages:
Sleep continuity: lower sleep efficiency (i.e. total time in bed divided by total sleep time in %) with a medium effect size [SMD(k=5) = −0.72 (−1.08; −0.36), p < 0.001; I2 = 40.0%, Q-test p = 0.14; post hoc I2 = 62%, Q-test p = 0.02], which corresponds to 6% decrease of total sleep time/total time in bed ratio in comparison to healthy controls. Significant lower sleep efficiency was calculated for FE (k = 4) and UHR (k = 2), as well as for FE-P (k = 2) and FE-SZ (k = 2) individuals. Longer duration of wake after sleep onset (i.e. time spent awake after sleep onset per night in minutes) with medium effect size [SMD(k=7) = 0.49 (0.21; 0.77), p < 0.01; I2 = 39.0%, Q-test p = 0.13; post hoc I2 = 44.2%, Q-test p = 0.09], which corresponds to 9 minutes of longer waking periods after sleep onset in comparison to healthy controls. Significant longer wake after sleep onset was calculated for FE (k = 5), as well as for FE-P individuals (k = 3). No significant effects were observed across UHR (k = 2) and FE-SZ individuals (k = 2).
Sleep duration: shorter total sleep time (i.e. total time spent asleep in minutes) with a small effect size [SMD(k=8) = −0.44 (−0.67; −0.21), p < 0.001; I2 = 19.0%, Q-test p = 0.28; post hoc I2 = 21%, Q-test p = 0.29], which corresponds to 36 minutes decreased of sleep duration in comparison to healthy controls. Significant shorter total sleep time was calculated across FE studies (k = 6), as well as across FE-SZ (k = 5) individually. No significant differences in total sleep time were observed in UHR individuals (k = 2).
Sleep initiation: longer sleep onset latency (i.e. time taken for sleep onset in minutes) with medium effect size [SMD(k=5) = 0.75 (0.45; 1.06), p < 0.001; I2 = 0.0%, Q-test p = 0.92; post hoc I2 = 0.0%, Q-test p = 0.91] which corresponds to 15 minutes longer sleep onset in comparison to healthy controls. Significant longer total sleep time was calculated for FE individuals (k = 4).
For all measures of sleep continuity, duration and initiation, asymmetrical funnel plots (Supplemental Figure S4) and small fail-safe Ns (N < 30) suggested publication bias.
Sleep architecture
No significant differences were observed between individuals with an early stage (UHR, 1 study; FE, 7 studies) and healthy controls for any of the reported parameters of sleep architecture (Figure 2, Supplemental Figure S3). %NREM1: SMD(k = 8) = 0.23 (−0.03; 0.50), p > 0.05; %NREM2: SMD(k = 8) = 0.04 (−0.47; 0.56), p > 0.05; %NREM3: SMD(k = 6) = −0.11 (−0.39; 0.17), p > 0.05; %NREM4: SMD(k = 3) = −0.66 (−1.61; 0.29), p > 0.05; %Slow-wave sleep: SMD(k = 4) = −0.63 (−1.40; 0.15), p > 0.05; %REM: SMD(k = 8) = 0.20 (−0.05; 0.45), p > 0.05; REM latency: SMD(k = 4) = −0.28 (−1.07; 0.50), p > 0.05.
Between-group differences
No significant between-group differences (UHR vs FE) were observed across all calculated SMDs (all Ps > 0.05).
Meta-regressions
Meta-regressions revealed a significant effect of age and antipsychotic status on subjective measures of sleep across UHR and FE subgroups, such as sleep quality decreased with age (k = 6) and increased with the percentage of patients treated with antipsychotics (k = 6). In parallel, total sleep time decreased (k = 8) and wake after sleep onset increased (k = 7) with age. No additional effects of sex ratio and symptoms severity of clinical samples (as measured by the Positive And Negative Syndrome Scale) were observed across sleep measures (Supplemental Table S2).
Discussion
Here, we presented a review and meta-analysis of a large range of sleep measures in early stages of psychotic (UHR-P, FE-P, FE-SZ) and bipolar disorders (UHR-BD). The methodological novelty of the present updated meta-analysis was to combine stringent inclusion criteria similar to previous reviews (Clarke et al., 2020; Davies et al., 2017) with the additional consideration of the UHR-BD stage. Moreover, for the first time in the literature, between-stage comparisons and impact of potential confounders were calculated. The main result of our study is that early stages of psychosis (UHR-P, FE-P, FE-SZ) and ultra high risk for bipolar disorder (UHR-BD) are associated with significant alterations in subjective sleep quality assessed by the PSQI and in objective sleep quality parameters measured with actigraphy/polysomnography, including shorter total sleep time, lower sleep efficiency, longer sleep onset latency and longer duration of wake after sleep onset across various UHR and FE subgroups. These findings corroborate those of previous reviews involving either UHR or FE (Clarke et al., 2020; Davies et al., 2017). This supports the hypothesis that sleep disturbances appear during early stages and persist in established psychotic disorders (Geoffroy et al., 2015; Geoffroy and Gottlieb, 2020; Kaskie et al., 2017; Laskemoen et al., 2019; Meyer et al., 2020), while no conclusions about sleep status in FE-BD can be made. In contrast, we noted that specific sleep continuity, duration and initiation parameters did not differ between controls and individual early stage subgroups (e.g. no alteration of total sleep time in UHR). However, this result may be due to a lack of power, since early stages subgroups’ effect sizes mostly involved small numbers of studies (k = 2).
No significant differences emerged when examining direct between-subgroup comparisons (UHR vs FE) across all calculated effect sizes, suggesting similar quantitative patterns of sleep disturbances across early stages of psychotic and bipolar disorders. This indicates sleep disturbances as a trait marker of early stages in general, and thus irrelevant for distinguishing between stages. Nevertheless, recent actigraphy results have demonstrated that the extent of delayed sleep phase is associated with more severe or persistent phases of psychiatric disorders (Scott et al., 2016), which warrants further investigation by large-scale prospective studies including early stages to more chronic groups.
Sleep disturbances in early stages are likely to be the result of aberrant functioning of sleep/wake brain structures. In that sense, Lunsford-Avery et al. (2015) demonstrated that a reduction in the thalamus volume correlates with the severity of sleep quality and continuity alterations in UHR individuals. Moreover, a recent study showed reduced sleep spindles in FE-P and antipsychotic-naïve FE-SZ individuals, pointing to early disruption of thalamo-cortical networks that involve the thalamic reticular nucleus where spindles are generated (Kaskie et al., 2019; Manoach et al., 2014). In our study, no sufficient spindle data was available to be meta-analyzed but comparisons of neurophysiological markers of sleep architecture were conducted between early stages individuals and controls, yet yielding to no significant differences. This contrasts with the existing literature showing abnormal latency and relative duration of sleep stages in psychotic and bipolar disorders and suggests that such alterations might emerge in more altered brain functions that only disrupt in later stages of the disorders (Benson et al., 1991; Markovic et al., 2020; Poulin et al., 2003; Rao et al., 2002). In other words, alterations of sleep architecture seem to be associated with more severe or persistent stages of illness and are not yet detectable in UHR and FE stages.
The meta-regressions showed a general decrease in the sleep quality, continuity and duration with age, including worse subjective quality of sleep, decreased total sleep time and increased wake after sleep onset. This is reflecting both association of normative aging with reduced ability to initiate and maintain sleep (Mander et al., 2017) and likely deterioration of brain structures involved in sleep processes along the course of the disorders (Kay and Dzierzewski, 2015). However, investigations of shared biological substrates between sleep disturbances and psychiatric conditions throughout the course of age are still lacking. In parallel, meta-regressions indicated significant improvement in sleep subjective quality with the prescription of antipsychotic in early stages, which is consistent with the demonstrated improvement of insomnia symptoms with second-generation antipsychotics in schizophrenia (Monti et al., 2017). Finally, the absence of effects regarding symptoms severity on sleep parameters reinforces the concept of a trait-level of sleep disturbances across all early stages of psychotic and bipolar disorders.
Several limitations of this study should be acknowledged. First, the results of meta-analyses of sleep continuity, duration and initiation parameters should be taken with caution, as the presence of heterogeneities and publication bias were significant. In particular, the publication bias where studies that are statistically significant are more likely to be published than those with less encouraging results suggest a risk of a substantial overestimation of the true effect size, i.e. the significance and magnitude of specific sleep differences between early stages and controls (Lin and Chu, 2018). In parallel, polysomnography and actimetry findings in UHR groups relative to control groups are hard to interpret and compare with FE groups, given the scarcity of information available in the literature. Second, the results corresponding to the UHR-P + BD subgroup must be interpreted with caution, given that only two UHR-P + BD studies were included in the meta-analyses. Moreover, no data regarding individual UHR-BD subgroup was identified, which forces us to temper generalizations of our results to this specific subgroup. However, larger heterogeneity measures when excluding UHR-P + BD studies suggests that using transdiagnostic at-risk groups does not increase heterogeneity of sleep comparisons with controls. In parallel, no study including FE-BD was identified by the literature search, which prevents any conclusions about sleep status in this early stage of bipolar disorder. Moreover, our study only included early stages of bipolar disorder that were diagnosed with specific clinical criteria. We thus may have missed individuals with an early stage that were diluted in broader samples such as adolescent depression (Rao et al., 2002). The difficulty of precisely defining the early stages of bipolar disorder has been recently pointed out by The International Society for Bipolar Disorders Taskforce (Kupka et al., 2021). Although sleep disturbances have been suggested as a core dimension present in almost all patients with bipolar disorder from the very onset of this disorder (Geoffroy and Gottlieb, 2020; Malhi et al., 2018), future sleep studies including broader samples conceived to experience emerging bipolar disorder (e.g. FE-BD, adolescent depression with risk factors for bipolar disorder offspring of parents with bipolar disorder; Pouchon et al., 2021) are warranted. Third, it is worth noting that the vast majority of included studies measured objective sleep parameters using polysomnography, which by design alter the normal sleep schedule and habit. Finally, our samples included unavoidable differences between groups regarding cognitive profiles, education levels and symptoms assessed by UHR instruments (SIPS and CAARMS). Although critical, these variables were not consistently reported across studies and thus we could not analyze their effect in meta-regressions. Furthermore, heterogeneity inherent to different instruments to measure clinical symptoms and cognitive ability across studies must be considered.
Altered markers of subjective sleep and continuity, duration and initiation of sleep characterize early stages of psychotic disorders and at-risk stage of bipolar disorders, while disturbance of sleep architecture seems to be associated with more severe or persistent stages. Our results suggest that assessments of sleep subjective quality and sleep continuity, duration and initiation parameters that are readily implemented in clinical settings should be part of the early intervention armamentarium. This would be helpful for early identification of sleep disturbances and efficient chronotherapeutic interventions that may reduce the clinical impact on patients (Bradley et al., 2018; Gottlieb et al., 2019). As a perspective, our results support further investigations of sleep disturbances’ predictive value in the transition from UHR to full-established psychotic or bipolar disorder, as well as their relevance as potential endophenotypes of these disorders.
Supplemental Material
sj-docx-7-anp-10.1177_00048674211068395 – Supplemental material for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis
Supplemental material, sj-docx-7-anp-10.1177_00048674211068395 for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis by Clément Dondé, Antoine Jaffiol, Charles Khouri, Arnaud Pouchon, Renaud Tamisier, Michel Lejoyeux, Marie-Pia d’Ortho, Mircea Polosan and Pierre A Geoffroy in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-pdf-1-anp-10.1177_00048674211068395 – Supplemental material for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis
Supplemental material, sj-pdf-1-anp-10.1177_00048674211068395 for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis by Clément Dondé, Antoine Jaffiol, Charles Khouri, Arnaud Pouchon, Renaud Tamisier, Michel Lejoyeux, Marie-Pia d’Ortho, Mircea Polosan and Pierre A Geoffroy in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-pdf-2-anp-10.1177_00048674211068395 – Supplemental material for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis
Supplemental material, sj-pdf-2-anp-10.1177_00048674211068395 for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis by Clément Dondé, Antoine Jaffiol, Charles Khouri, Arnaud Pouchon, Renaud Tamisier, Michel Lejoyeux, Marie-Pia d’Ortho, Mircea Polosan and Pierre A Geoffroy in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-pdf-3-anp-10.1177_00048674211068395 – Supplemental material for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis
Supplemental material, sj-pdf-3-anp-10.1177_00048674211068395 for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis by Clément Dondé, Antoine Jaffiol, Charles Khouri, Arnaud Pouchon, Renaud Tamisier, Michel Lejoyeux, Marie-Pia d’Ortho, Mircea Polosan and Pierre A Geoffroy in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-pdf-4-anp-10.1177_00048674211068395 – Supplemental material for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis
Supplemental material, sj-pdf-4-anp-10.1177_00048674211068395 for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis by Clément Dondé, Antoine Jaffiol, Charles Khouri, Arnaud Pouchon, Renaud Tamisier, Michel Lejoyeux, Marie-Pia d’Ortho, Mircea Polosan and Pierre A Geoffroy in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-pdf-5-anp-10.1177_00048674211068395 – Supplemental material for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis
Supplemental material, sj-pdf-5-anp-10.1177_00048674211068395 for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis by Clément Dondé, Antoine Jaffiol, Charles Khouri, Arnaud Pouchon, Renaud Tamisier, Michel Lejoyeux, Marie-Pia d’Ortho, Mircea Polosan and Pierre A Geoffroy in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-xlsx-6-anp-10.1177_00048674211068395 – Supplemental material for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis
Supplemental material, sj-xlsx-6-anp-10.1177_00048674211068395 for Sleep disturbances in early clinical stages of psychotic and bipolar disorders: A meta-analysis by Clément Dondé, Antoine Jaffiol, Charles Khouri, Arnaud Pouchon, Renaud Tamisier, Michel Lejoyeux, Marie-Pia d’Ortho, Mircea Polosan and Pierre A Geoffroy in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
