Abstract
Objective:
Over a half century, lithium has been used as the first-line medication to treat bipolar disorder. Emerging clinical and laboratory studies suggest that lithium may exhibit cardioprotective effects in addition to neuroprotective actions. Fractalkine (CX3CL1) is a unique chemokine associated with the pathogenesis of mood disorders and cardiovascular diseases. Herein we aimed to ascertain whether lithium treatment is associated with favorable cardiac structure and function in relation to the reduced CX3CL1 among patients with bipolar disorder.
Methods:
We recruited 100 euthymic patients with bipolar I disorder aged over 20 years to undergo echocardiographic study and measurement of plasma CX3CL1. Associations between lithium treatment, cardiac structure and function and peripheral CX3CL1 were analyzed according to the cardiovascular risk. The high cardiovascular risk was defined as (1) age ⩾ 45 years in men or ⩾ 55 years in women or (2) presence of concurrent cardiometabolic diseases.
Results:
In the high cardiovascular risk group (n = 61), patients who received lithium as the maintenance treatment had significantly lower mean values of left ventricular internal diameters at end-diastole (Cohen’s d = 0.65, p = 0.001) and end-systole (Cohen’s d = 0.60, p = 0.004), higher mean values of mitral valve E/A ratio (Cohen’s d = 0.51, p = 0.019) and superior performance of global longitudinal strain (Cohen’s d = 0.51, p = 0.037) than those without lithium treatment. In addition, mean plasma levels of CX3CL1 in the high cardiovascular risk group were significantly lower among patients with lithium therapy compared with those without lithium treatment (p = 0.029). Multiple regression models showed that the association between lithium treatment and mitral value E/A ratio was contributed by CX3CL1.
Conclusion:
Data from this largest sample size study of the association between lithium treatment and echocardiographic measures suggest that lithium may protect cardiac structure and function in patients with bipolar disorder. Reduction of CX3CL1 may mediate the cardioprotective effects of lithium.
Introduction
Patients with bipolar disorder have at least 2 times higher mortality rate and a 10-year reduction in life expectancy as compared with the general population (Chesney et al., 2014; Crump et al., 2013; Hayes et al., 2015; Westman et al., 2013). Moreover, research from Eastern and Western countries has consistently reported that cardiovascular disease is the major cause leading to the excessive and premature deaths in bipolar population (Chen et al., 2020; Chesney et al., 2014; Correll et al., 2017; Crump et al., 2013; Hayes et al., 2015; Westman et al., 2013). In particular, emerging evidence has suggested that congestive heart failure is the major comorbidity that contributes to the high cardiovascular mortality in patients with bipolar disorder (Chen et al., 2020). To meaningfully reduce the risk of cardiovascular deaths in patients with bipolar disorder, an early detection and management of cardiac structural and functional abnormalities before the progression to heart failure are urgently called for in this at-risk population.
Over the past half-century, lithium has been used as the first-line medication to treat the mood symptoms of bipolar disorder (Grunze et al., 2018; Yatham et al., 2018). One of the major mechanisms accounting for the mood-stabilizing effects of lithium involves neuroprotective actions (Rybakowski et al., 2018; Soeiro-de-Souza et al., 2012). Of particular note, emerging clinical data have suggested that lithium therapy reduces the risk of cardiovascular disease in patients with bipolar disorder (Lan et al., 2015; Prosser and Fieve, 2016; Tsai et al., 2020).). Furthermore, laboratory research has demonstrated that lithium at therapeutic levels (0.39 ± 0.06 mEq/L) ameliorates pathological hypertrophy and cardiac fibrosis in the rat model of myocardial infarction (Lee et al., 2017). Collectively, the available evidence from clinical and laboratory studies suggest that lithium therapy may potentially exhibit cardioprotective effects in addition to its well-known neuroprotective actions (Chen et al., 2019). Despite the aforementioned evidence, few studies have examined the effect of lithium treatment on cardiac structure and function in association with biomarkers involved in the pathogenesis of bipolar disorder and cardiovascular disease.
Apart from traditional cardiovascular risk factors, several pathophysiological processes have been proposed as the putative pathogenesis bridging bipolar disorder and cardiovascular disease (Goldstein et al., 2015, 2020). In particular, chemokines, a family of low-molecular-weight chemotactic cytokines, have been shown to be associated with both neuropsychiatric disorders and cardiovascular diseases (Apostolakis and Spandidos, 2013; Stuart and Baune, 2014; Trettel et al., 2020). Among various kinds of chemokines, fractalkine (CX3CL1) is synthesized as a trans-membrane chemokine by neurons and presents in a soluble form upon cleavage by metalloproteases (Trettel et al., 2020). Both trans-membrane and soluble form CX3CL1 bind to receptor CX3CR1 in microglia and mediates the bidirectional cross-talk between neurons and microglia in the pathological brains (Biber et al., 2007; Cardona et al., 2006; Hughes et al., 2003; Poniatowski et al., 2017). These processes are involved in the pathogenesis of mood disorders by modulating the release of inflammatory cytokines, synaptic plasticity and neuron apoptosis (Stuart and Baune, 2014). Besides, CX3CL1 has been implicated in the different stages of atherogenesis, including the recruitment of leukocytes (Schulz et al., 2007; Stolla et al., 2012), proliferation and migration of vascular smooth muscle cells (Lucas et al., 2003; White et al., 2010) and formation of atherosclerotic plaque (Nie et al., 2014). In turn, adverse cardiac remodeling in the ischemic heart leads to cardiac dysfunction and progression of heart failure (Bhatt et al., 2017). Together with laboratory research which has demonstrated that lithium is related to the decreased CX3CL1 levels by disrupting Golgi-mediated CX3CL1 trafficking to the cell membrane (Fuster-Matanzo et al., 2017), the existing data suggest that CX3CL1 is a potential therapeutic target and biomarker in the study of whether lithium treatment has cardioprotective effects in patients with bipolar disorder.
The aim of this study was to ascertain whether lithium treatment was associated with favorable cardiac structure and function in patients with bipolar disorder using both standard and novel echocardiographic measurements. In addition, we explored whether plasma CX3CL1 were related to the cardioprotective actions of lithium. We hypothesized that lithium treatment was associated with better cardiac structure and function in relation to the reduced CX3CL1 among patients with bipolar disorder.
Material and methods
Participants
This cross-sectional study recruited participants from the psychiatric department at Taipei Medical University Hospital who met the following inclusion criteria: (1) age ⩾ 20 years; (2) a primary diagnosis of bipolar I disorder confirmed by two research psychiatrists according to the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders-Fourth Edition Text Revision Axis I disorders (SCID-I); and (3) remission of mood symptoms as defined by total scores of <6 on the Young Mania Rating Scale (YMRS) and <7 on the 21-item Hamilton Depression Rating Scale continuously (HDRS-21) for more than 8 weeks. Exclusion criteria were as follows: (1) any substance use disorder except for tobacco use; (2) any mental disorder associated with a general medical condition; (3) any type of dementia; and (4) inability to undergo an echocardiographic examination. After enrollment in this study, participants’ medications used to treat bipolar disorder or medical diseases remained the same as prescribed from the primary care physician. The study protocol was approved by the Joint Institutional Review Board of Taipei Medical University. All participants provided written informed consent before participating in this study.
Clinical variables
Each participant underwent physical examination, including blood pressure, body weight and body height, on the day of echocardiographic study. Blood pressure was measured twice in the sitting position using a mercury sphygmomanometer. The mean of the 2 values was used for data analyses. Data on demographic characteristics, disease course of bipolar disorder, history of medical comorbidities and details of psychopharmacological treatment were obtained by reviewing the medical records as well as interviewing patients and their reliable companions. The extracted data from medical records was rechecked by two psychiatrists involved in this study in order to decrease individual errors.
Since this was a naturalistic study and medications used to treat bipolar disorder were prescribed according to the primary care psychiatrist’s judgment, patients who received lithium as the maintenance treatment in this study may take different kinds of psychotropic medications during the disease course of bipolar disorder. To estimate lithium exposure in relation to the duration of bipolar disorder, we calculated the ratio of lithium exposure in relation to bipolar illness chronicity (RDIC) using our previously described formula: total lifetime years of lithium treatment / total years since the onset of bipolar disorder (Tsai et al., 2020). The greater RDIC value indicated a more consistent treatment with lithium during the disease course of bipolar disorder. We defined the onset of bipolar disorder as the occurrence of mood symptoms, either depression or mania, which caused psychosocial dysfunction or resulted in psychiatric hospitalization.
To investigate whether lithium was associated with favorable cardiac structure and function among patients with bipolar disorder at risk of cardiovascular diseases, we conducted stratified analyses according to the cardiovascular risk. Considering that patients with bipolar disorder begin to have increased risk of cardiovascular and metabolic morbidities in midlife (Goldstein et al., 2020), the high cardiovascular risk group was defined according to the following criteria: (1) age ⩾ 45 years in men or ⩾ 55 years in women or (2) presence of coronary heart disease, stroke, hypertension, diabetes mellitus, hyperlipidemia, or obesity (Davis et al., 2015; Towfighi et al., 2011). In addition, given prior findings that the association of bipolar disorder with cardiovascular risk is greater among female patients (Goldstein et al., 2020), we evaluated the association of lithium treatment with cardiac structure and function in the female subgroup.
Biochemistry and CX3CL1 assay
On the day of echocardiographic examination, heparinized blood samples were collected through venous puncture between 8:30 a.m. and 9:30 a.m., following an overnight fast beginning at midnight. Biochemical analyses, including serum fasting glucose, triglyceride, total cholesterol and low-density lipoprotein, were conducted in the Department of Laboratory Medicine at Taipei Medical University Hospital. Remaining plasma samples were collected in separate tubes and stored at −80 °C until analysis. Plasma CX3CL1 levels were measured in duplicate using commercially available enzyme-linked immunosorbent assay (ELISA) kits (Research and Diagnostic Systems, Minneapolis, MN, USA). The mean minimum detectable doses were 18 pg/mL. The intra- and interassay coefficients were <8% and <10%, respectively.
Standard echocardiography and speckle tracking imaging
All patients underwent standard (M mode, two dimensional and color Doppler imaging) and novel echocardiography (two dimensional speckle tracking imaging) performed by the same board-certified cardiologist blinded to the clinical data. Examinations were performed with a median frequency (3–8 MHz) transducer using the same cardiac ultrasonographer Vivid 7 with software version 103.0.1 (GE Healthcare Bio-Sciences Corporation, Piscataway, NY, USA). Images were stored digitally and analyzed offline using the EchoPAC PC software version 108.1.5 (GE Healthcare Bio-Sciences Corporation).
M-mode imaging was analyzed to determine cardiac structural parameters, including interventricular septal thickness at end-diastole (IVSd), left ventricular posterior wall thickness at end-diastole (LVPWd), left ventricular internal diameter at end-diastole (LVIDd) and left ventricular internal diameter at end-systole (LVIDs). The ratio of peak velocity blood flow from left ventricular relaxation in early diastole (mitral valve E wave) to peak velocity blood flow in late diastole caused by atrial contraction (mitral valve A wave) was computed using color Doppler imaging to evaluate left ventricular diastolic function. To assess left ventricular systolic function, left ventricular ejection fraction (LVEF) was estimated using both the 2-D and modified Simpson’s method. In addition, global longitudinal strain was calculated by the summation of longitudinal strain values derived from the speckle tracking images in 2-chamber, 4-chamber and apical long axis view. The Q lab offline analyses used for speckle tracking imaging were confirmed with the same analyzer to ensure the consistency of tracking. Given that the strain is a measure of tissue deformation, the value of cardiac strain during the systolic phase is negative. Furthermore, the more negative the cardiac strain value, the greater the systolic function performance. To quantify the degree of changes in echocardiographic variables, effect size was calculated using Cohen’s d. Values between 0.21 and 0.50 denoted a small effect size, those between 0.51 and 0.80 denoted a moderate effect size, and those >0.80 denoted a large effect size.
Statistical analysis
Two-group comparisons were analyzed using an independent sample t test or Mann–Whitney U test for continuous variables and Pearson’s χ2 test or Fisher’s exact test for categorical variables. For the four-group comparisons, a one-way analysis of variance (ANOVA) test with post hoc Tukey’s test was used. Pearson product-moment correlation was performed to examine the correlations between continuous variables. A multiple linear regression was conducted to examine the contribution of CX3CL1 to cardiac function. Variables were selected based on the effect size r obtained from the pairwise correlation as well as the cardiovascular professionals’ clinical experience and literature (Alpert et al., 2016; Nadruz et al., 2017). To evaluate whether lithium therapy was associated with echocardiographic values depending on the level of cardiovascular risk, general linear model was applied to examine the interaction effect of lithium treatment (yes/no) and cardiovascular risk (high/low) on the echocardiographic measures. P < 0.05 was considered statistically significant in this study.
Results
Demographic and clinical characteristics between patients with and without lithium therapy
In total, we recruited 100 participants with euthymic bipolar I disorder (37 men and 63 women; mean age, 43.7 ± 13.3 years) in this study (Table 1). Fifty-three of the patients received lithium as the maintenance treatment at the entry into study. Compared with patients who did not receive lithium as the maintenance treatment (n = 47), patients with lithium therapy had significantly higher ratios of lithium exposure in relation to bipolar illness chronicity (i.e. RDIC). Besides, patients with lithium therapy were less likely to receive valproic acid or carbamazepine as the maintenance treatment when compared with those patients without lithium therapy. The two groups did not significantly differ in other demographic and clinical characteristics of bipolar disorder.
Demographic and clinical variables of patients with bipolar disorder (N = 100).
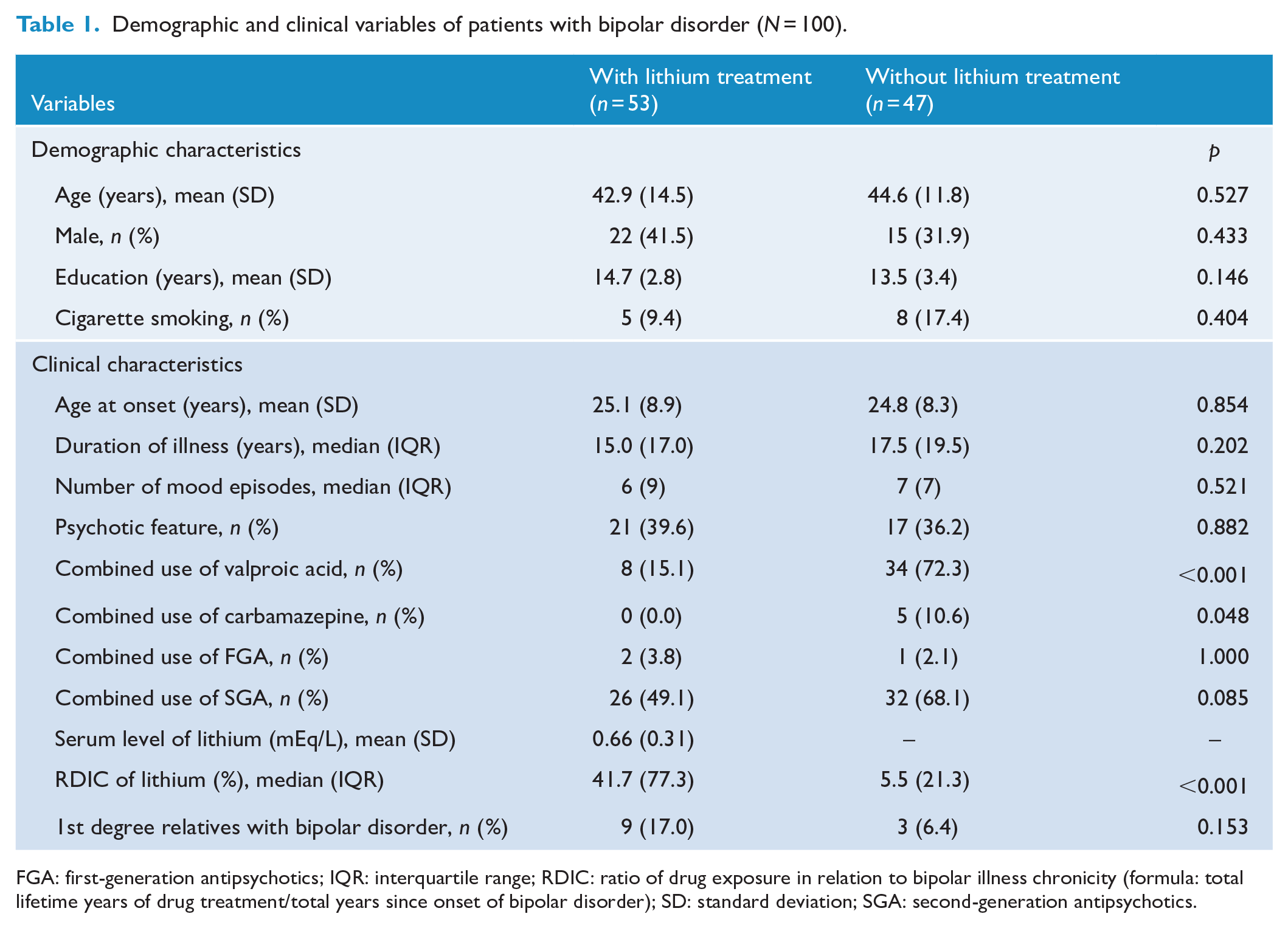
FGA: first-generation antipsychotics; IQR: interquartile range; RDIC: ratio of drug exposure in relation to bipolar illness chronicity (formula: total lifetime years of drug treatment/total years since onset of bipolar disorder); SD: standard deviation; SGA: second-generation antipsychotics.
Cardiometabolic profiles between patients with and without lithium therapy
Table 2 compares the cardiometabolic profiles between patients with and without lithium treatment. The results showed that the cardiometabolic profiles, including cardiometabolic comorbidities and measures, were comparable between the two groups.
Cardiometabolic variables of patients with bipolar disorder (N = 100).
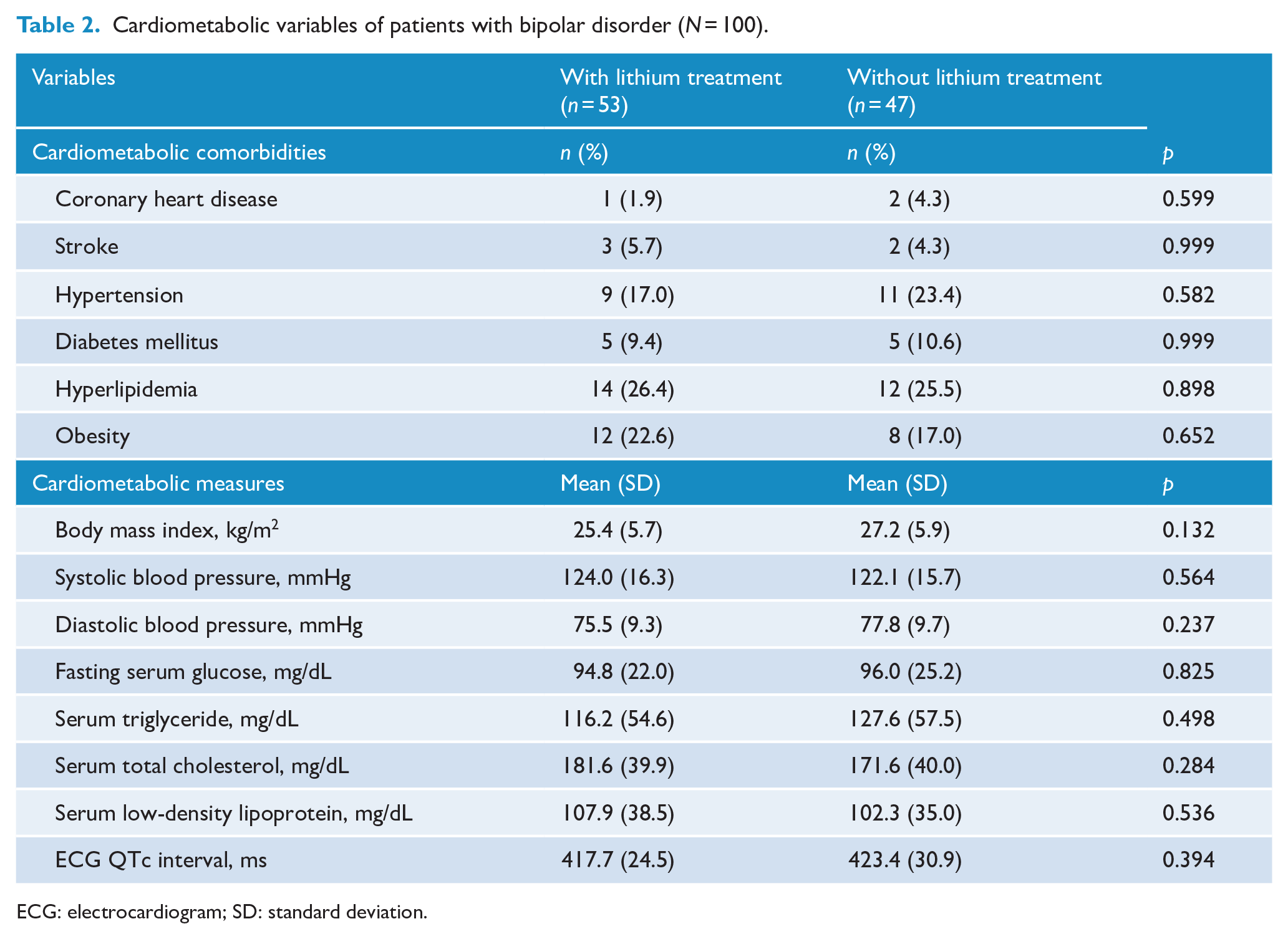
ECG: electrocardiogram; SD: standard deviation.
Echocardiographic measures between patients with and without lithium therapy
Table 3 presents the echocardiographic measures between patients with and without lithium treatment. Patients with lithium therapy had significantly lower mean values of LVIDd (Cohen’s d = 0.65, p = 0.001) and LVIDs (Cohen’s d = 0.60, p = 0.004) and higher mean mitral valve E/A ratio (Cohen’s d = 0.51, p = 0.019) than in those without lithium therapy. Although LVEF was not significantly different between the two groups, patients with lithium therapy had superior performance on the global longitudinal strain than did those without lithium treatment (Cohen’s d = 0.51, p = 0.037).
Echocardiographic variables of patients with bipolar disorder (N = 100).
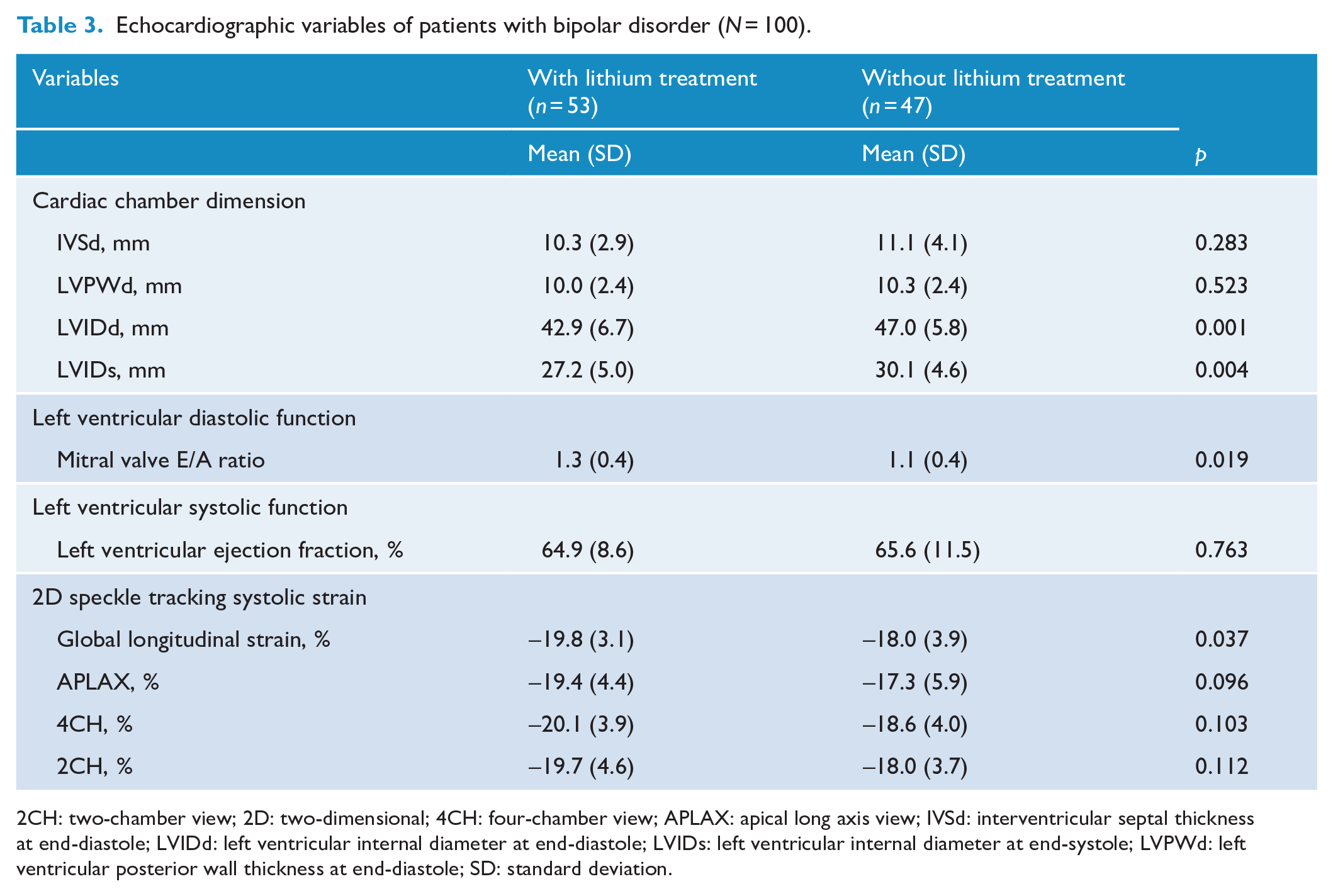
2CH: two-chamber view; 2D: two-dimensional; 4CH: four-chamber view; APLAX: apical long axis view; IVSd: interventricular septal thickness at end-diastole; LVIDd: left ventricular internal diameter at end-diastole; LVIDs: left ventricular internal diameter at end-systole; LVPWd: left ventricular posterior wall thickness at end-diastole; SD: standard deviation.
Echocardiographic measures between patients with and without lithium therapy stratified by cardiovascular risk
Table 4 illustrates the echocardiographic measures between patients with and without lithium treatment stratified by the cardiovascular risk. Among the high cardiovascular risk group, 5 (22.7%) of male patients and 25 (64.1%) of female patients were aged below 45 years and 55 years, respectively. They were stratified into the high cardiovascular risk group because of the cardiometabolic comorbidities (i.e. coronary heart disease, stroke, hypertension, diabetes mellitus, hyperlipidemia, or obesity). Significantly, patients with lithium therapy in the high cardiovascular risk group had lower mean values of LVIDd (Cohen’s d = 0.72, p = 0.007) and LVIDs (Cohen’s d = 0.62, p = 0.018) and higher mean mitral valve E/A ratio (Cohen’s d = 0.75, p = 0.005) than did those without lithium treatment. Moreover, the performance of the global longitudinal strain in the high cardiovascular risk group was greater among patients with lithium therapy than that among patients without lithium treatment (Cohen’s d = 0.68, p = 0.037). Although echocardiographic measures were not significantly different between patients with and without lithium treatment in the low cardiovascular risk group, the mean values of LVIDd and LVIDs were decreased in patients with lithium therapy than did those without lithium treatment. Considering that sample size of the low cardiovascular risk group was smaller than that of the high cardiovascular risk one, no significant difference in LVIDd and LVIDs between patients with and without lithium treatment in the low cardiovascular risk group may result from insufficient statistical power rather than differential associations of lithium. To evaluate whether lithium was associated with LVIDd and LVIDs depending on the level of cardiovascular risk, general linear model was applied to test the interaction effect of lithium treatment (yes/no) and cardiovascular risk (high/low) on the LVIDd and LVIDs. The results showed that lithium treatment interacted with cardiovascular risk on the LVIDd (F = 4.66, p = 0.033) but not LVIDs (F = 0.95, p = 0.332).
Echocardiographic variables of patients with bipolar disorder stratified by cardiovascular risk (N = 100).
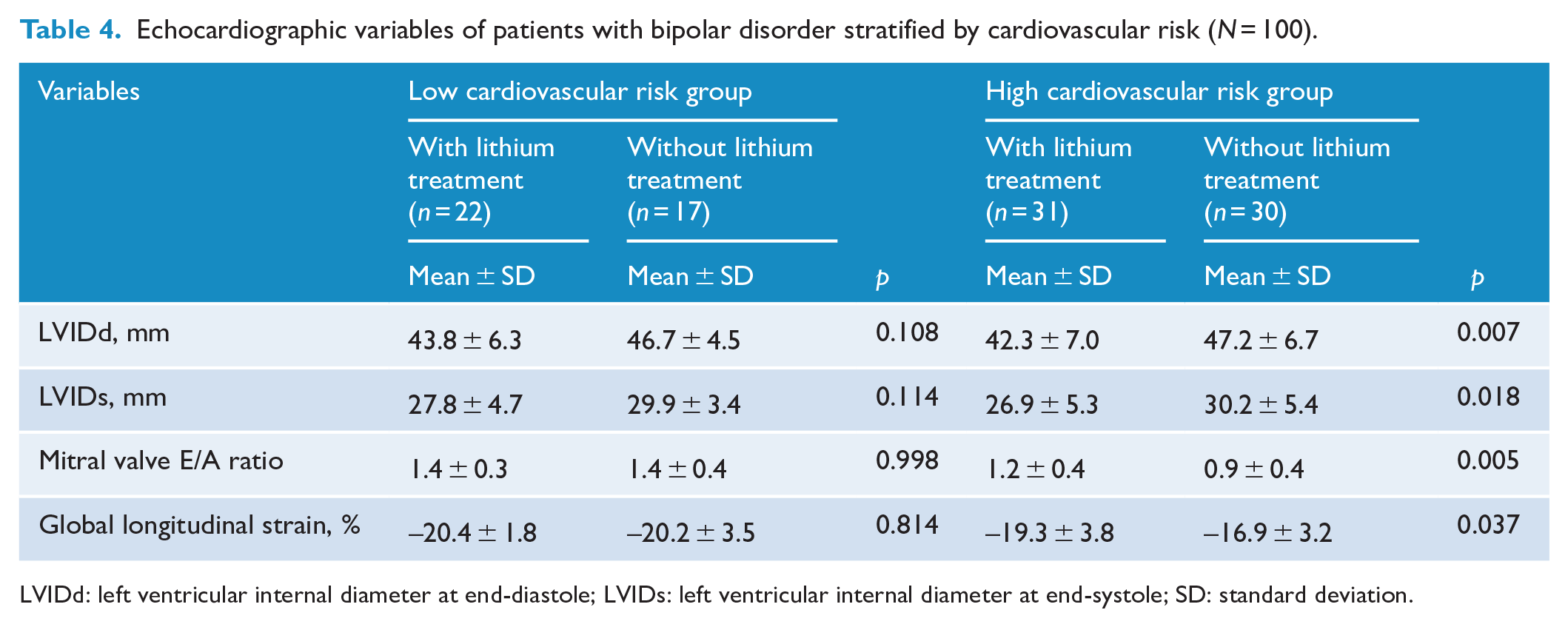
LVIDd: left ventricular internal diameter at end-diastole; LVIDs: left ventricular internal diameter at end-systole; SD: standard deviation.
Analysis of association between peripheral CX3CL1 and cardioprotective potential of lithium
To explore whether CX3CL1 is associated with the cardioprotective potentials of lithium, we first compared the plasma CX3CL1 levels between patients with and without lithium treatment in both low and high cardiovascular risk groups (e-Figure 1, in the Supplement). The mean plasma CX3CL1 levels were significantly elevated in the high cardiovascular risk group without lithium treatment (579.5 ± 131.4 pg/mL) than those with lithium treatment (464.9 ± 137.7 pg/mL, p = 0.029) and the low cardiovascular risk group regardless of lithium treatment (with lithium treatment: 395.1 ± 176.5 pg/mL, p < 0.001; without lithium treatment: 391.0 ± 151.4 pg/mL, p < 0.001). We also examined the relationship between plasma CX3CL1 levels, RDIC of lithium and echocardiographic parameters. Significantly, mitral valve E/A ratio was negatively correlated with plasma CX3CL1 level (r = –0.219, p = 0.036) (e-Figure 2, in the Supplement). In addition, the mitral valve E/A ratio was positively correlated to RDIC of lithium (p = 0.019) (Table 5, model 1). However, this correlation became non-significant when CX3CL1 and cardiometabolic variables were entered into the regression model (Table 5, model 2). The findings suggest that the association between the RDIC of lithium and mitral value E/A ratio was contributed by CX3CL1 in patients with bipolar disorder.
Regression models for mitral valve E/A ratio in patients with bipolar disorder (N = 100).
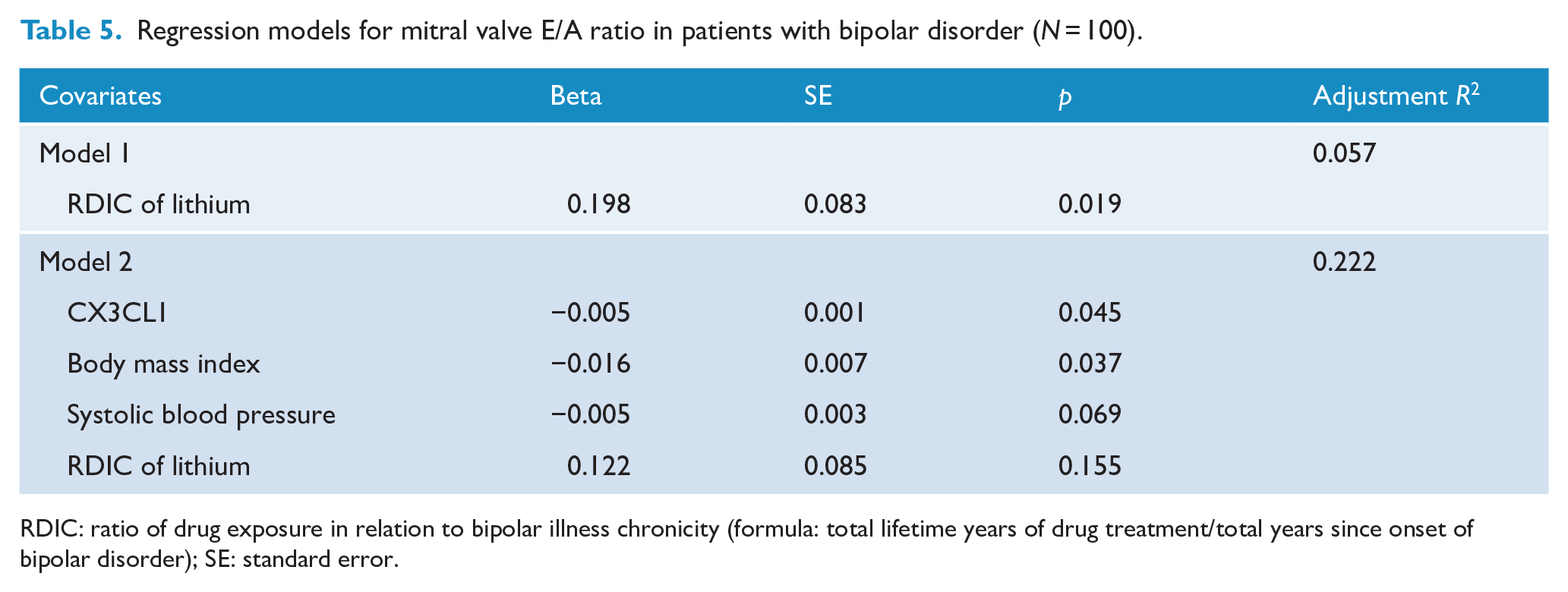
RDIC: ratio of drug exposure in relation to bipolar illness chronicity (formula: total lifetime years of drug treatment/total years since onset of bipolar disorder); SE: standard error.
Secondary analysis of female subgroup
In secondary analyses of the female subgroup, there were no significant differences between female patients with and without lithium treatment in terms of the clinical characteristics and cardiometabolic profiles except for the RDIC of lithium and frequency of valproic acid use (e-Tables 1 and 2 in the Supplement). In addition, female patients with lithium therapy tended to have lower mean values of LVIDd (Cohen’s d = 0.63, p = 0.017) and LVIDs (Cohen’s d = 0.38, p = 0.057) and higher mean values of mitral valve E/A (Cohen’s d = 0.44, p = 0.074) than did those without lithium treatment (e-Table 3 in the Supplement). However, the difference in the latter two echocardiographic measures did not reach statistical significance. As regards the association between plasma CX3CL1 levels, RDIC of lithium and echocardiographic parameters, the mitral valve E/A ratio was negatively correlated with plasma level of CX3CL1 (r = –0.344, p = 0.009). In addition, the mitral valve E/A ratio was positively correlated to the RDIC of lithium (p = 0.046). This correlation became non-significant when CX3CL1 and cardiometabolic variables were entered into the regression model (e-Table 4 in the Supplement).
Discussion
To minimize the effects of the severity of bipolar affective symptoms and state-dependent pathophysiology on the heart and inflammation, we recruited patients with bipolar disorder in full remission to investigate the association of lithium treatment with cardiac structure and function. For the first time, we discovered that lithium was associated with better performance on several echocardiographic parameters in patients with bipolar disorder, including the LVIDd, LVIDs, mitral valve E/A ratio and global longitudinal strain. In addition, the association between the lithium treatment and mitral value E/A ratio was contributed by plasma CX3CL1. The findings suggest that CX3CL1 may be a potential state-independent biomarker in the future studies of the lithium’s effects on cardiovascular risk in patients with bipolar disorder. Of particular note, previous prospective research has suggested that greater mood symptom burden is associated with higher cardiovascular mortality in patients with bipolar disorder (Fiedorowicz et al., 2009). In this present study, we did not find significant correlations between number of mood episodes and echocardiographic measures (data not shown). Given the limitation that recall bias may exist in the cross-sectional study, future prospective studies are necessary to evaluate mood symptoms longitudinally and examine their associations with the lithium’s effects on cardium.
Limited studies have included echocardiographic measures to investigate the effect of lithium therapy on cardiac structure and function in patients with bipolar disorder. One study used standard echocardiography to examine the effects of lithium on cardiac function in 30 patients with bipolar disorder and healthy controls (Zencir et al., 2015). The authors did not find any significant difference in cardiac function, such as LVEF and mitral valve E/A ratio, between the two groups. By contrast, we found that in the high cardiovascular risk group, lithium-treated patients had higher mean mitral valve E/A ratios than those without lithium treatment. Under physiological conditions, most of the blood fills left ventricle during the early diastole passive filling phase. Therefore, the mitral valve E-wave (i.e. peak velocity blood flow from left ventricular relaxation in early diastole) is typically higher than the mitral valve A-wave (i.e. peak velocity blood flow in late diastole caused by atrial contraction). However, when left ventricle diastolic dysfunction occurs, the early diastolic passive filling of ventricle is impaired and more blood enters into ventricle at the time of atrial phase filling. Thus, the mitral valve A-wave is reversely higher than the mitral valve E-wave. In particular, the mean mitral valve E/A ratio (0.9) in our patients without lithium treatment among the high cardiovascular risk group was close to the abnormal cutoff point of mitral valve E/A ratio (⩽0.8), whereas that (1.2) in patients with lithium treatment was within the normal range (0.8–2.0) (Prasad et al., 2019). Considering that abnormal mitral valve E/A ratio is referred to as cardiac diastolic dysfunction, our results suggest that lithium may preserve cardiac diastolic function in patients with bipolar disorder at high cardiovascular risk.
In this study, no significant difference was observed in the LVEF between patients with bipolar disorder with and without lithium treatment. However, the performance of the global longitudinal strain in the high cardiovascular risk group was greater in patients with lithium therapy than it was in those without lithium treatment. In the human heart, subepicardial fibers are oriented clockwise, and subendocardial fibers are oriented counter-clockwise (Trivedi et al., 2019). Therefore, the heart deforms along the radial, circumferential and longitudinal axes. Of the three deformation components, longitudinal strain is a measurement used to evaluate cardiac systolic function in the longitudinal axis, allowing a closer study of subendocardial fibers. Given that the subendocardial layer is most sensitive to ischemic insults (Algranati et al., 2011), longitudinal strain allows for investigating early changes in cardiac systolic function prior to the clinical manifestation of heart failure (e.g. LVEF < 40%) in our patients with bipolar disorder. Considering that global longitudinal strain predicts long-term risk of cardiovascular disease (Al Saikhan et al., 2019; Biering-Sørensen et al., 2017; Russo et al., 2014), our study informs future longitudinal research to evaluate whether lithium therapy reduces the risk of major cardiovascular events in a bipolar disorder cohort.
Pathophysiological mechanisms of bipolar disorder probably accelerate the occurrence of cardiovascular disease (Goldstein et al., 2015, 2020). CX3CL1 is a unique chemokine involved in the pathogenesis of mood disorders and atherogenesis (Lucas et al., 2003; Nie et al., 2014; Schulz et al., 2007; Stolla et al., 2012; Stuart and Baune, 2014; White et al., 2010). Following the ischemic insults, adverse cardiac remodeling leads to various kinds of cardiac structural and functional abnormalities, eventually resulting in the heart failure (Bhatt et al., 2017). In our present analyses, we found that mean plasma CX3CL1 level was significantly higher in patients with high cardiovascular risk than that in those with low cardiovascular risk. In addition, the elevated plasma CX3CL1 levels in the high cardiovascular risk group were reduced in patients receiving lithium treatment. Together with findings from multiple regression analyses which suggested that plasma CX3CL1 levels contributed to the association between lithium therapy and mitral valve E/A ratio, our data may provide a novel biomarker in the future study targeting the cardioprotective effects of lithium in patients with bipolar disorder.
Mechanisms underlying the association between lithium treatment and reduced plasma CX3CL1 levels are not fully elucidated. Recent laboratory research has demonstrated that inhibition of glycogen synthase kinase-3β (GSK-3β) leads to a 50% decrease in the levels of the mature membrane-bound form of CX3CL1 in primary cultured neurons treated with lithium (Fuster-Matanzo et al., 2017). In addition, mice treated with GSK-3β inhibitors, including lithium, exhibited decreased levels in both membrane-bound and soluble CX3CL1 in the brain probably due to the interruption of Golgi-mediated trafficking to the cell membrane (Fuster-Matanzo et al., 2017). However, limited studies thus far have investigated the mechanisms for the role of CX3CL1 in the cardioprotective actions of lithium. To afford mechanistic insights into the cardioprotective effects of lithium, future laboratory studies are required to explore the underlying mechanisms for the inhibitory effects of lithium on CXCL1 in cardiovascular system.
We acknowledge that our study has several methodological limitations. First, the cross-sectional design precluded the determination of the direction of causation in this study. However, results of this association study inform future prospective research to evaluate cardioprotective effects and mechanisms of lithium treatment among patients with bipolar disorder. Second, although this was so far the largest sample size study of the associations between lithium therapy and echocardiographic measures, this study was still limited by unequal numbers of participants in the stratified analyses. Furthermore, this study was underpowered to examine sex difference in the association of lithium treatment with cardiac structure and function. To elucidate whether lithium protects cardium among those patients with a high cardiovascular risk, future research should plan with statistical power and accommodate equal numbers of participants in each study subgroup. Third, we did not collect echocardiographic data from a control group. Previously, a meta-analysis and large-sample community studies have suggested that the expected values of left ventricular global longitudinal and mitral valve E/A ratio in healthy individuals were −18.9% to −20.4% and 1.0 to 1.6, respectively (Caballero et al., 2015; Yingchoncharoen et al., 2013). Accordingly, our patients with high cardiovascular risk who received lithium treatment actually had cardiac function comparable to that of healthy individuals, whereas that of patients without lithium treatment was worse than the general population. Fourth, some studies have reported that lithium may alter cardiac electrical conduction and cause adverse electrocardiographic effects (Mamiya et al., 2005; Mehta and Vannozzi, 2017; Vancampfort et al., 2016). The findings contradict our observation that lithium therapy was associated with better cardiac structure and function in patients with bipolar disorder. Nevertheless, we should note that many of these reports were related to the use of high or toxic doses of lithium and were thus different from our study on lithium within the therapeutic ranges. Further research must investigate the effects of lithium on cardiac health in a dose-dependent manner. Fifth, we did not measure oxidative stress and endothelial dysfunction, which are other biological mediators that may affect pathogenesis of cardiovascular diseases in patients with bipolar disorder (Goldstein et al., 2015, 2020). For a better understanding of the mechanisms underlying the cardioprotective actions of lithium, future studies need to include these potential markers. Sixth, physical activity may affect inflammatory response system and modulate risk of cardiovascular diseases (Goldstein et al., 2020). Considering that patients with bipolar disorder tend to have higher levels of sedentary behaviors compared with the general population (Vancampfort et al., 2016), the physical activity level should be included as an important variable in the future analysis. Last, we only recruited participants with bipolar I disorder in this study. This may constrain the generalizability of present findings to other subtypes of bipolar disorder.
In conclusion, from this largest sample size study of the association between lithium therapy and echocardiographic measures, we found that lithium treatment is associated with favorable cardiac structure and function among patients with bipolar disorder. In addition, reduction in circulating CX3CL1 levels may mediate the cardioprotective effects of lithium. To comprehend the fundamental value of lithium therapy in bipolar disorder, future prospective studies should strengthen our findings by additionally measuring other biomarkers reflecting the common pathogenesis of bipolar disorder and cardiovascular diseases across the various mood states.
Supplemental Material
sj-doc-1-anp-10.1177_00048674211062532 – Supplemental material for Cardioprotective potential of lithium and role of fractalkine in euthymic patients with bipolar disorder
Supplemental material, sj-doc-1-anp-10.1177_00048674211062532 for Cardioprotective potential of lithium and role of fractalkine in euthymic patients with bipolar disorder by Pao-Huan Chen, Cheng-Yi Hsiao, Shuo-Ju Chiang, Ruei-Siang Shen, Yen-Kuang Lin, Kuo-Hsuan Chung and Shang-Ying Tsai in Australian & New Zealand Journal of Psychiatry
Footnotes
Author Contributions
P.H. Chen and C.Y. Hsiao contributed equally to this study. P.H. Chen reviewed the literature, interpreted data and wrote the first draft of manuscript. C.Y. Hsiao undertook echocardiographic imaging acquisition and analysis, interpreted data and co-wrote the first draft of manuscript. S.J. Chiang, R.S. Shen and K.H. Chung coordinated in the collection and interpretation of data. Y.K. Lin undertook statistical analysis. S.Y. Tsai designed the study, wrote the proposal, supervised the data collection and edited the manuscript. All authors contributed to and approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by research grant from the Ministry of Science and Technology, Taiwan (MOST 106-2314-B-038-050-MY3). The Ministry of Science and Technology, Taiwan, has no involvement in study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision of article publication.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
