Abstract
Objective:
Patients with bipolar disorder treated with lithium often require additional antipsychotics or anticonvulsants. However, the comparative effectiveness and safety of these agents as add-on to lithium has not been studied.
Methods:
This secondary analysis combined two similar 24-week trials on outpatients with bipolar disorder randomized to lithium (target serum level 0.4–0.6 mEq/L). Guideline-based adjunctive antipsychotics (Li+AP) and anticonvulsants (Li+AC) could be used if clinically indicated and was assessed at every study visit. Response was measured on the Clinical Global Impression scale and we performed adjusted mixed effects linear regression analyses. Analysis of variance tests compared metabolic measures including a binary diagnosis of metabolic syndrome before and after 24 weeks of treatment.
Results:
Among 379 outpatients (57% female, mean age 38 years, mean Clinical Global Impression 4.4), users of Li+AP (N = 50, primarily quetiapine and aripiprazole) improved to a similar degree (mean Clinical Global Impression improvement = 1.6, standard deviation = 1.5) as those using lithium-only (i.e. without adjunctive antipsychotics or anticonvulsants, N = 149, mean Clinical Global Impression improvement = 1.7, standard deviation = 1.4) (p = 0.59). Users of Li+AC (N = 107, primarily lamotrigine and valproate, mean Clinical Global Impression improvement = 1.2, standard deviation = 1.3) and users of Li+AP+AC (N = 73, mean Clinical Global Impression improvement = 1.1, standard deviation = 1.3) showed worse response compared to lithium-only users (all p < 0.01). When comparing Li+AP to Li+AC, users of Li+AP improved slightly better on general (p = 0.05) and manic symptoms (p = 0.01), but showed a worse development of glucose, triglycerides, and metabolic syndrome.
Conclusion:
Despite treatment-by-indication confounding, these findings are relevant for real-world treatment settings and emphasize the need for randomized trials on this clinically important topic.
Introduction
Bipolar disorder is a difficult-to-treat mental disorder and guidelines recommend different pharmacological approaches, primarily lithium, antipsychotics and anticonvulsants (Baldessarini et al., 2019; Malhi et al., 2018; Rhee et al., 2020; Yatham et al., 2018). In clinical practice, many patients require combination treatment with two or even more agents to achieve response (Glue and Herbison, 2015; Kessing et al., 2016; Köhler-Forsberg et al., 2020; Taylor et al., 2014). Guidelines recommend combining different drug classes, e.g., lithium with quetiapine or lamotrigine (Baldessarini et al., 2019; Malhi et al., 2018; Yatham et al., 2018). Although the use of lithium has declined during the recent years, it is still used by a third of patients with bipolar disorder (Kessing et al., 2016; Köhler-Forsberg et al., 2020; Rhee et al., 2020). Noteworthy, at least 20–30% of lithium users were treated with additional antipsychotics or anticonvulsants (Bjorklund et al., 2016; Kessing et al., 2016; Köhler-Forsberg et al., 2020; Rhee et al., 2020). Among patients treated with lithium, antipsychotics and anticonvulsants are considered equivalent augmentation treatment choices, i.e., clinicians can either chose an antipsychotic or an anticonvulsant as add-on for a patient with insufficient response to lithium (Baldessarini et al., 2019; Malhi et al., 2018; Yatham et al., 2018).
However, despite the clinical importance, few trials have studied the efficacy and safety of antipsychotics or anticonvulsants as add-on to lithium. The BALANCE trial (N = 330) found that lithium monotherapy and lithium plus valproate were more likely to prevent relapse than valproate monotherapy (Geddes et al., 2010). Suppes et al. (2013) (N = 523) found that maintenance quetiapine in combination with lithium, compared to lithium with placebo, reduced the risk of recurrence of a mood episode. Vieta et al. (2008b) (N = 384) found that adjunctive aripiprazole to lithium improved response of mania symptoms compared to lithium+placebo. Other small trials found that lithium+oxcarbazepine showed better treatment effects compared to lithium+carbamazepine (N = 52) (Juruena et al., 2009), that lithium+oxcarbazepine might have some prophylactic efficacy compared to lithium+placebo (N = 55) (Vieta et al., 2008a), and that the combinations of lithium+valproate and lithium+carbamazepine (N = 64) showed similar efficacy (Missio et al., 2019), respectively. However, to our knowledge, no trial has included patients treated with lithium and studied the comparative effectiveness of adjunctive antipsychotics to adjunctive anticonvulsants. Furthermore, previous studies did not include metabolic effects, representing a frequent side effect that may negatively affect compliance and somatic health (Firth et al., 2019; Pillinger et al., 2020), particularly when combining two or more mood-stabilizing drugs.
Due to the paucity of randomized trials on this important aspect, we aimed to study the real-world comparative effectiveness of guideline-based and clinically indicated (i.e. non-randomized) adjunctive treatment with antipsychotics or anticonvulsants among patients randomized to lithium. We performed a secondary analysis on data from two large similar clinical trials randomizing outpatients with bipolar disorder to lithium for 24 weeks. The treating clinicians could initiate additional treatment with other drugs, such as antipsychotics or anticonvulsants, based on clinical indication. Our a priori hypothesis was that lithium plus antipsychotics would show better clinical treatment effects than lithium plus anticonvulsants but worsen metabolic markers.
Among patients randomized to 24 weeks of lithium treatment, we aimed to study the following:
The comparative clinical mood-stabilizing effectiveness of add-on antipsychotics versus add-on anticonvulsants versus lithium treatment alone;
Effects on metabolic markers, measured via blood tests before and after the 24-week trial.
Methods
Setting
This study represents secondary analyses and is based on two similar 24-week multi-site, randomized comparative effectiveness trials, the Bipolar Clinical Health Outcomes Initiative in Comparative Effectiveness (Bipolar CHOICE) (Nierenberg et al., 2014) trial and the Lithium Treatment Moderate-Dose Use Study (LiTMUS) (Nierenberg et al., 2009). Bipolar CHOICE was conducted between 2010 and 2013 and included patients from 10 US clinics to compare lithium (target dose of 900 mg, corresponding to 0.6 mEq/L) to quetiapine (target dose of 300 mg) combined with other guideline-informed medications for bipolar disorder (but not with one another) consistent with typical clinical practice, referred to as adjunctive personalized medication therapy (APT). LiTMUS was conducted between 2007 and 2009 and included patients from six US clinics to compare optimized personalized treatment (OPT) with lithium (target dose of 600 mg, corresponding to 0.4 mEq/L) to OPT without lithium. The adjunctive treatment parts (i.e. APT and OPT) were based on the same guidelines and were hence comparable. Both trials found no overall differences in treatment efficacy between their respective randomized treatment arms (Nierenberg et al., 2013; Nierenberg et al., 2016). The mean serum lithium levels were 0.56 mEq/L in the Bipolar CHOICE trial and 0.45 mEq/L in the LiTMUS trial.
For this study, we included those two treatment arms randomized to lithium (Supplementary Figure 1) in order to have a large study population of outpatients with bipolar disorder treated with lithium and similar treatment protocols for additional drug use following the same guidelines.
All subjects provided verbal and written informed consent prior to participation. The Institutional Review Boards of the different sites approved the study protocol, and the rationale, design and specific methods of Bipolar CHOICE and LiTMUS are reported in detail elsewhere (Nierenberg et al., 2009, 2014). Data are only available by request to the senior author A.A.N.
Participants
For Bipolar CHOICE, 240 patients were randomized to lithium+APT, whereas LiTMUS randomized 141 patients to lithium+OPT. Two individuals who were randomized to lithium+OPT had to be excluded due to missing medication form data, i.e., missing data on additional drugs used during the trial. Supplementary Figure 1 is presenting a flowchart for identification of this study population. Supplementary Table 1 presents baseline data for the combined cohort and the lithium treatment arms from the two trials, indicating no baseline differences.
Both trials applied similar broad entry criteria to maximize sample heterogeneity and generalizability of the findings. All patients were aged between 18 and 68 years. At inclusion, participants were required to have a DSM-IV-TR bipolar I or bipolar II diagnosis and to be at least mildly symptomatic (Clinical Global Impression Scale for Bipolar Disorder [CGI-BP] (Spearing et al., 1997) ⩾ 3). Psychiatric diagnoses were determined using the extended Mini-International Neuropsychiatric Interview, an electronic version of a validated structured diagnostic interview (Sheehan et al., 1998). Clinical interviews obtained demographic information and medical history.
Adjunctive use of antipsychotics or anticonvulsants
Participants stayed on the randomized lithium treatment for the entire duration of the trial (i.e. lithium+APT in Bipolar CHOICE and lithium+OPT in LiTMUS). The additional medications were allowed to change during the 24-week study periods based on individual patient needs and accordance of medication changes with clinical guidelines. In both trials, the Medication Recommendation Tracking Form (Reilly-Harrington et al., 2013) was used at every study visit to assess clinical decision(s) regarding each drug used by each patient. This form was developed by the Bipolar Disorder Trials Network to comprehensively capture physician prescribing behavior and clinical decision-making. Hence, patients could be receiving antipsychotics and/or anticonvulsants at study initiation or they could initiate treatment with these drugs during the trial. Regarding anticonvulsants, we only included those agents with mood-stabilizing properties, i.e., lamotrigine, valproate and carbamazepine. We defined the following groups:
Lithium without additional treatment with an antipsychotic or anticonvulsant during the 24 weeks of the trial (hereafter referred to as the Li-only group),
Lithium with additional treatment with an antipsychotic but no use of an anticonvulsant (hereafter referred to as Li+AP),
Lithium with additional treatment with an anticonvulsant but no use of an antipsychotic (hereafter referred to as Li+AC),
Lithium with additional treatment with an antipsychotic and an anticonvulsant (hereafter referred to as Li+AP+AC).
In addition, we included information on the specific drugs (e.g. quetiapine or lamotrigine).
Clinical measures
At study baseline and each subsequent visit (same follow-up visits in both trials after 2, 4, 6, 8, 12, 16, 20 and 24 weeks), patients were assessed with the overall CGI-BP including the CGI subscales for depression and mania.
Before and after 24 weeks of treatment, all participants had the following anthropometric and metabolic markers assessed: Cholesterol, high-density lipoprotein (HDL), low-density lipoprotein (LDL), triglycerides, glucose, waist circumference, body mass index (BMI), thyrotropin-stimulating hormone (TSH), creatinine, blood pressure and pulse. Furthermore, for each participant, we calculated a binary diagnosis of metabolic syndrome based on National Cholesterol Education Program Adult Treatment Panel III (NCEP ATP III) criteria (Grundy et al., 2005).
Statistical analysis
Concerning potential baseline differences between the treatment groups, we performed chi-square tests and analysis of variance (ANOVA).
Regarding treatment effects during the 24 weeks of the trial, we performed mixed effects linear regression analyses using the CGI-BP overall score as the primary outcome measure. We performed secondary analyses using the CGI-BP subscales for depression and mania. All analyses were adjusted for age, sex, trial (i.e. Bipolar CHOICE or LiTMUS) and baseline CGI-BP severity.
We studied treatment effects for individuals using (1) Li+AP, (2) Li+AC and (3) Li+AP+AC. We always compared to the reference group, being the Li-only group.
To directly compare users of Li+AP to users of Li+AC, we excluded those individuals treated with Li-only and individuals treated with Li+AP+AC. This analysis was performed to directly compare guideline-based use of antipsychotics versus anticonvulsants as add-on to lithium.
Regarding anthropometric and metabolic markers, we performed ANOVA tests between the groups before and after the 24 weeks. We also compared the groups by performing ANOVA on the change in the markers from week 0 to week 24. We did not adjust for multiple testing as these markers are highly dependent of each other.
All analyses were performed using STATA 14.0. The accepted alpha level was 0.05.
Results
Characteristics
We included 379 outpatients with bipolar disorder randomized to lithium (mean serum levels 0.50–0.62 mEq/L during the trial), whereof 149 (39.3%) did not receive additional treatment with antipsychotics or anticonvulsants during the trial (i.e. the Li-only group). During the 24-week study period, a total of 50 (13.2%) were using antipsychotics as add-on to lithium (Li+AP), 107 (28.2%) were using anticonvulsants add-on (Li+AC), and 73 (19.3%) were using both antipsychotics and anticonvulsants add-on (Li+AP+AC). The Li+AP, Li+AC and Li+AP+AC groups differed from the Li-only group by having had more previous depressive and manic episodes and more previous psychiatric hospital contacts (Table 1). The specific antipsychotic and anticonvulsant drugs that were used as add-on to lithium are listed in Table 2. Quetiapine and aripiprazole were the most frequently used antipsychotics, whereas lamotrigine and valproate were the most frequently used anticonvulsants. Among those using Li+AP or Li+AC, the majority used only one type of additional drug during the study (Table 2).
Baseline characteristics for 379 outpatients with bipolar disorder randomized to lithium treatment, divided into users and non-users of clinically indicated (i.e. non-randomized) adjunctive antipsychotic and/or anticonvulsant drugs.
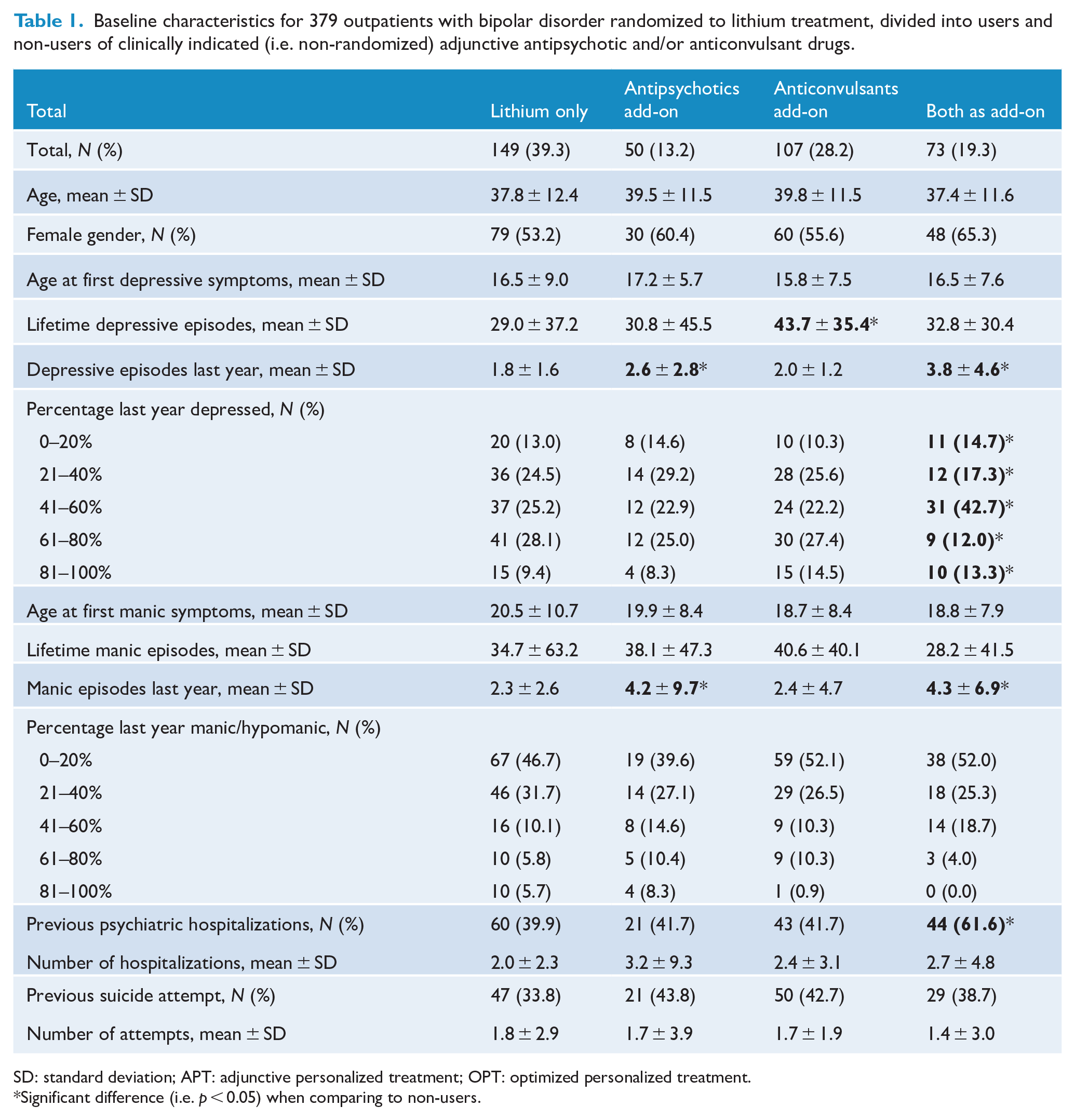
SD: standard deviation; APT: adjunctive personalized treatment; OPT: optimized personalized treatment.
Significant difference (i.e. p < 0.05) when comparing to non-users.
Drug utilization profiles among 379 patients with bipolar disorder treated for 24 weeks with lithium using guideline-based and clinically indicated adjunctive antipsychotics and/or anticonvulsants.
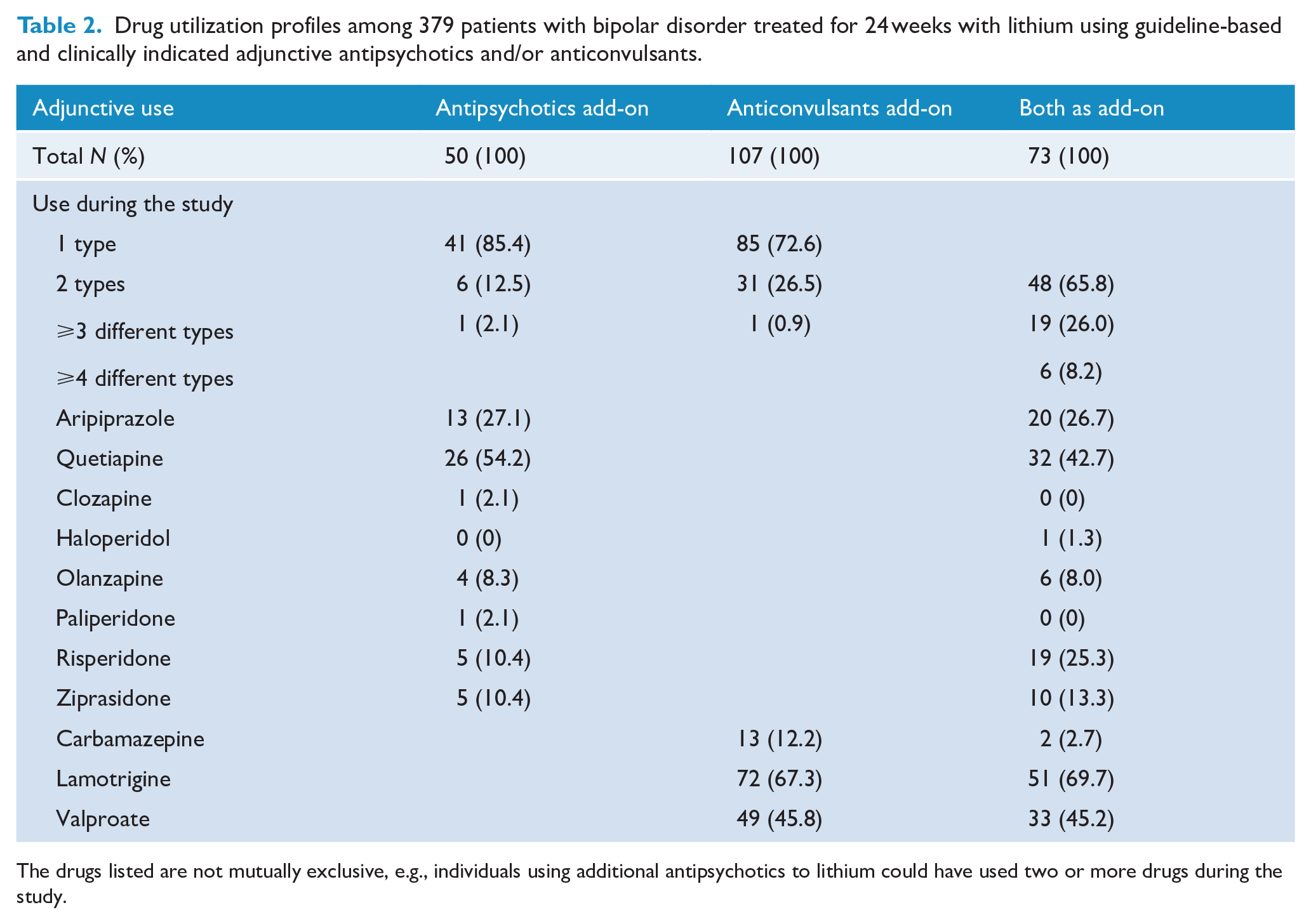
The drugs listed are not mutually exclusive, e.g., individuals using additional antipsychotics to lithium could have used two or more drugs during the study.
Clinical outcomes during the 24-week trial
First, we compared the Li+AP, Li+AC and Li+AP+AC groups with the Li-only group (Figure 1 and Supplementary Figure 2). The Li-only group showed the best response pattern during the first weeks of the study (mean CGI = 4.4 at baseline and mean CGI = 2.8 after 8 weeks), whereas the three groups with additional treatment (i.e. Li+AP, Li+AC and Li+AP+AC) showed a similar response pattern during the first 8 weeks (mean CGI = 4.4 at baseline and mean CGI at week 8 between 3.2 and 3.5). From week 16, the Li+AC group responded better and had a similar mean CGI-BP score after 24 weeks as the Li-only group (mean CGI = 2.7 after 24 weeks; overall p = 0.59 for difference during the 24 weeks in adjusted mixed effects analyses). On the other hand, those using Li+AC remained on the same CGI-BP score (mean of 3.2 or 3.3) between weeks 8 and 24. Individuals using Li+AP+AC worsened from week 8 (mean CGI = 3.5) to week 16 (mean CGI = 3.8) but improved during the subsequent weeks and had a similar mean CGI-BP score after 24 weeks as the Li+AC group (mean CGI = 3.3). Those using Li+AC (p = 0.001) and those using Li+AP+AC (p < 0.001) showed worse CGI overall treatment response compared to the Li-only group in adjusted mixed effects analyses. The differences in mean CGI-BP improvement were 0.7 CGI-BP points, corresponding to approximately 41% of the standard deviation of the improvement of Li-only users (Figure 1). Compared to the Li-only group, the Li+AC showed less improvement on the CGI depression and mania subscales and the Li+AP+AC group showed less improvement on the depression subscale (all p < 0.05; Supplementary Figure 2).
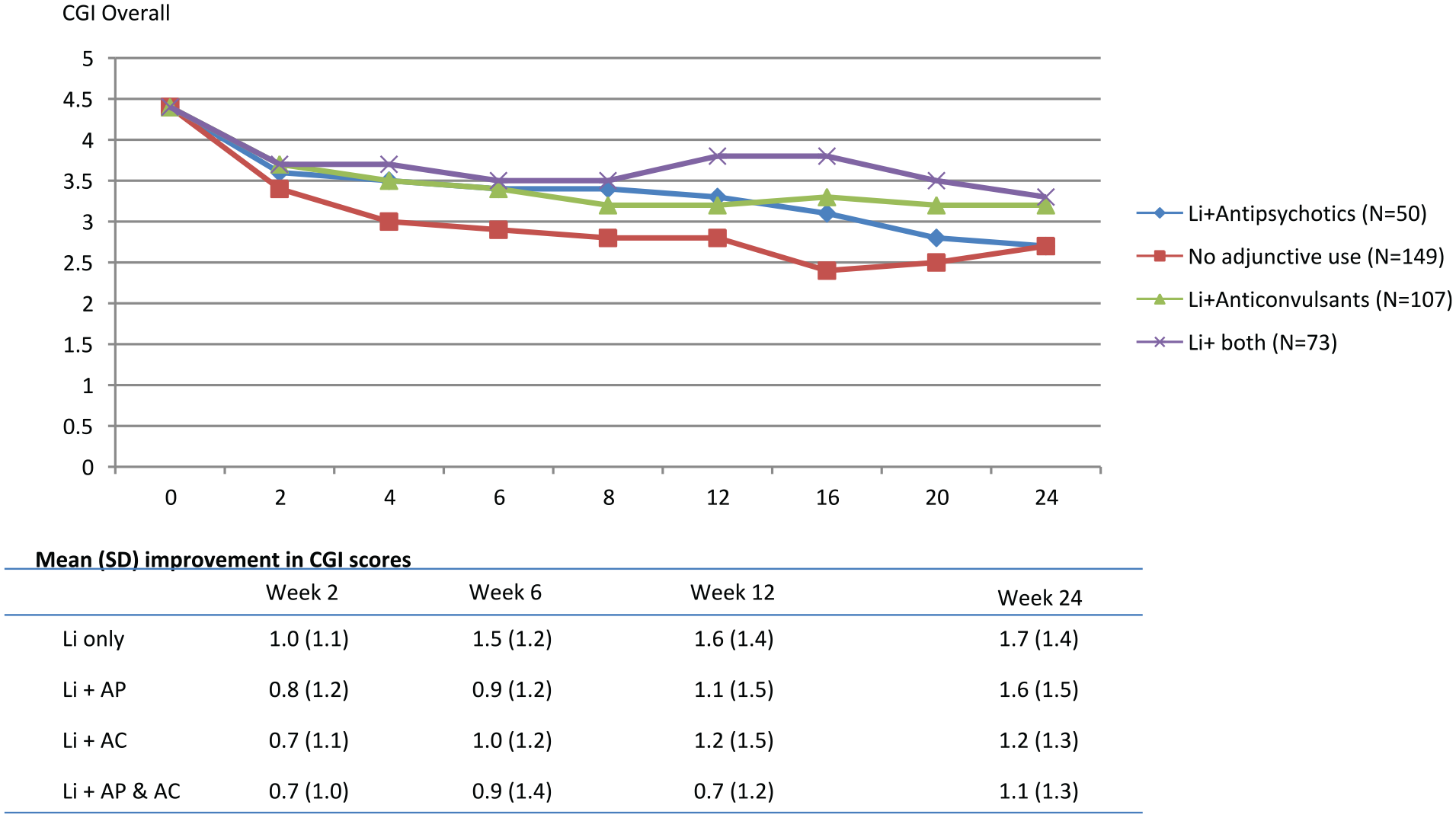
CGI overall treatment effects among 379 outpatients with bipolar disorder treated with 24 weeks of lithium treatment depending on adjunctive use of antipsychotics and/or anticonvulsants.
Lithium+antipsychotics versus lithium+anticonvulsants
Next, we compared the Li+AP and Li+AC groups in order to compare these two guideline-based add-on treatment options to lithium (Figure 2). We found no baseline differences between the two groups (Table 1).
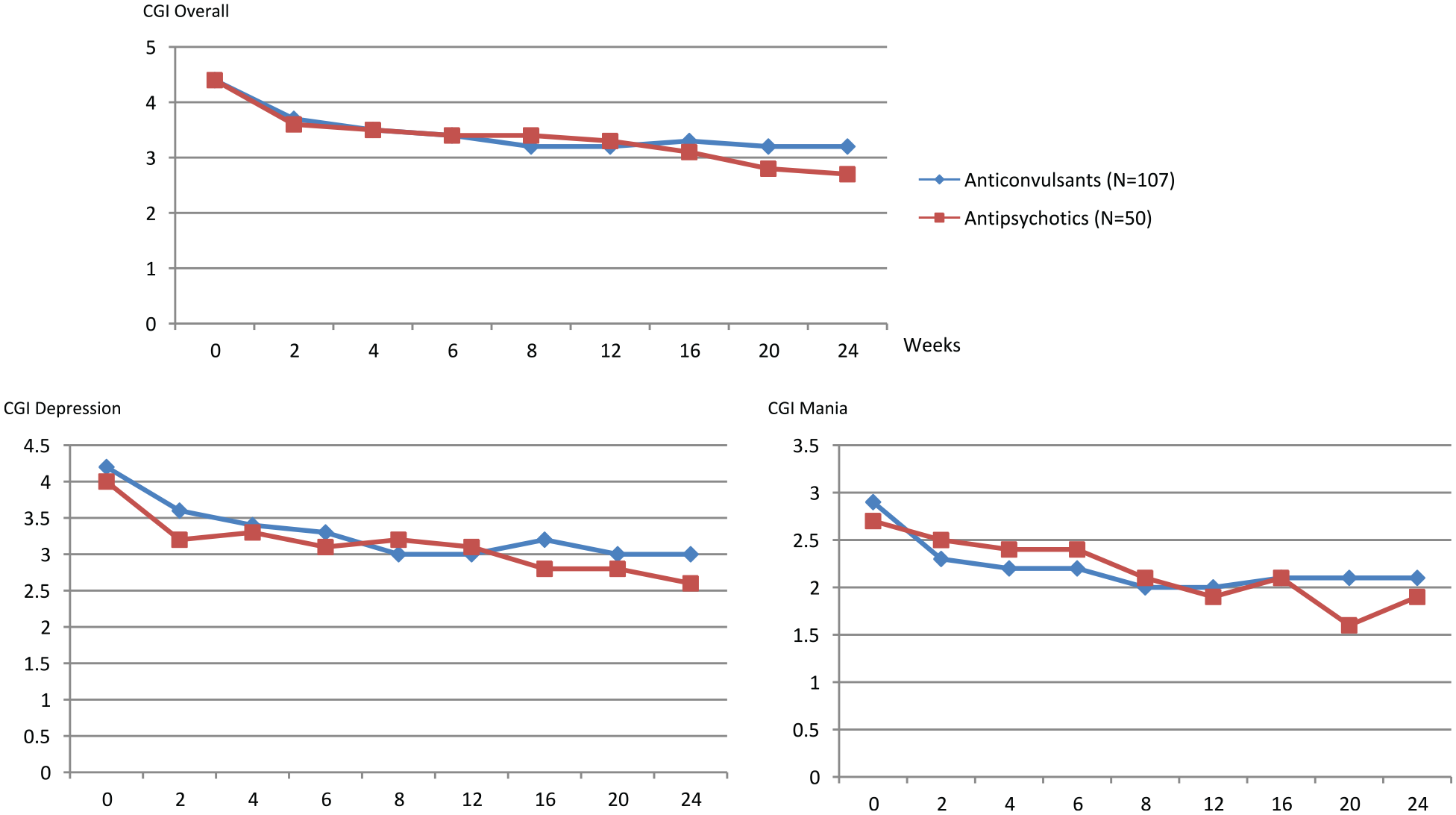
Among outpatients with bipolar disorder treated with lithium for 24 weeks, comparing clinically indicated adjunctive use of antipsychotics to adjunctive use of anticonvulsants.
Both groups showed similar mean response rates on the CGI-BP overall and the CGI-BP mania and depression subscales during the first 16 weeks of treatment. Those using Li+AP showed better response between week 16 and the 24-week visit, whereas those using Li+AC remained on the same level. Mixed effects analyses indicated that those using Li+AP, compared to the Li+AC group, were associated with significant better treatment response on the overall CGI-BP (p = 0.05) and the CGI-BP mania scale (p = 0.01).
Effects on metabolic measures
We found that individuals using Li+AP, compared to the Li-only group, had higher mean baseline values of cholesterol, while individuals using Li+AC, compared to Li-only, had lower glucose (Table 3).
Differences in metabolic measures (normal reference values shown below the table) before and after 24 weeks of lithium treatment with or without additional use of antipsychotics or anticonvulsants.
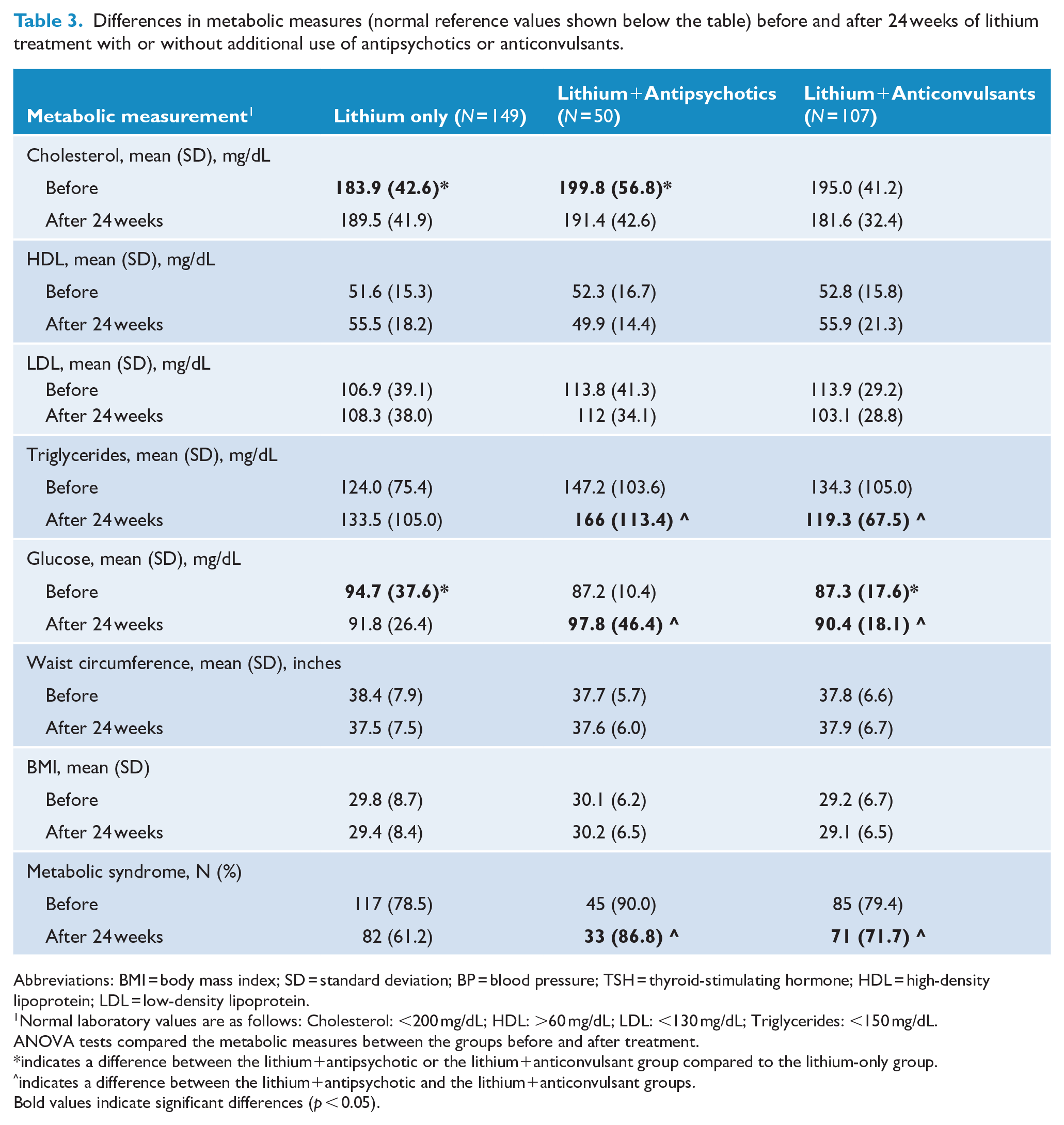
Abbreviations: BMI = body mass index; SD = standard deviation; BP = blood pressure; TSH = thyroid-stimulating hormone; HDL = high-density lipoprotein; LDL = low-density lipoprotein.
Normal laboratory values are as follows: Cholesterol: <200 mg/dL; HDL: >60 mg/dL; LDL: <130 mg/dL; Triglycerides: <150 mg/dL.
ANOVA tests compared the metabolic measures between the groups before and after treatment.
indicates a difference between the lithium+antipsychotic or the lithium+anticonvulsant group compared to the lithium-only group.
indicates a difference between the lithium+antipsychotic and the lithium+anticonvulsant groups.
Bold values indicate significant differences (p < 0.05).
Despite not reaching significance for all markers, those using Li+AP showed a trend toward a general worsening of several metabolic markers during 24 weeks of treatment. Particularly, we observed a decrease in HDL (from a mean of 52.3 to 49.9) and an increase in triglycerides (from a mean of 147.2 to 166) and glucose (from a mean of 87.2 to 98.8). Those using Li-only or Li+AC showed no change or a change in the opposite direction.
The majority of patients fulfilled the criteria for metabolic syndrome (80.7% at baseline). A decrease in the rate of metabolic syndrome was noted in the Li-only (78.5% at baseline and 61.2% at week 24) and the Li+AC groups (79.4% at baseline and 71.7% at week 24). The Li+AP group showed a higher rate (90.0% at baseline and 86.8% at week 24) and a significantly higher rate at week 24 compared to the Li-only group (p = 0.003) and the Li+AC group (p = 0.04).
We found no differences in TSH, creatinine, blood pressure and pulse (results not shown).
Discussion
This study pooled 379 outpatients with bipolar disorder of at least moderate severity treated with lithium for 24 weeks (mean serum levels 0.5–0.6 mEq/L). Guideline-based and clinically indicated use of additional medications to lithium showed that those using lithium+antipsychotics had similar treatment outcomes as those using lithium only, whereas those using lithium+anticonvulsants showed slightly worse outcomes. When comparing the two additional treatment groups directly (i.e. Li+AP versus Li+AC), we found that individuals using Li+AP showed slightly better treatment response compared to the Li+AC group, but also worse development of several metabolic markers, particularly triglycerides, glucose and metabolic syndrome. However, the Li+AP and Li+AC groups only differed during the last two visits (i.e. after 20 and 24 weeks of treatment) and the metabolic changes were modest. Furthermore, the add-on treatments were non-randomized, thus complicating causal interpretations. Nevertheless, our findings are based on a real-world representative bipolar disorder cohort following standard guidelines and were observed during a rather short treatment period of 24 weeks. Hence, our findings are of potential clinical importance and need to be tested in randomized clinical trials. In addition, this study emphasizes the large somatic burden among patients with bipolar disorder and thus a clear need for prevention and intervention, as 80% fulfilled the criteria for metabolic syndrome.
Cost–benefit evaluation of combination treatments for bipolar disorder
Patients with bipolar disorder often need a combination of two or more mood-stabilizing agents due to insufficient response to monotherapy (Kessing et al., 2016; Köhler-Forsberg et al., 2020; Rhee et al., 2020). Lithium is used by one out of three patients with bipolar disorder and many are in need of additional medications (Kessing et al., 2016; Köhler-Forsberg et al., 2020; Rhee et al., 2020). Despite this, few randomized trials have studied add-on treatments to lithium (Geddes et al., 2010, Juruena et al., 2009; Missio et al., 2019; Suppes et al., 2013; Vieta et al., 2008a; Vieta et al., 2008b) and no trial has directly compared antipsychotics to anticonvulsants as add-on to lithium. Despite not having randomized patients to add-on treatment to lithium, this study suggests small but interesting differences in treatment outcomes and side-effect profiles among patients treated with lithium and adjunctive use of antipsychotics or adjunctive use of anticonvulsants.
It is of interest that the difference on metabolic markers, such as blood triglyceride and glucose levels, was present already after 6 months of treatment, representing a rather short-term treatment for outpatients with bipolar disorder and with the present cohort representing a young group (mean age of 38 years).
Noteworthy, a recent meta-analysis on patients with schizophrenia suggested that an increased clinical efficacy of antipsychotics may be accompanied by worsened effects on metabolic markers (Pillinger et al., 2020). Our findings support this suggesting that among bipolar disorder patients with the need for add-on treatment to lithium, antipsychotics may increase the clinical efficacy compared to anticonvulsants, but that this may come at the cost of worsened metabolic effects. Hence, the present findings suggest that clinicians need to carefully balance the potential better efficacy of add-on antipsychotics to lithium versus the potential negative effects on metabolic markers and on somatic health in general and cardiovascular risk particularly. Furthermore, due to the limitations of this non-randomized study, our findings should be subjected to thorough examination in a randomized trial.
Strengths and limitations
Strengths include the large study population and the real-world setting. Bipolar CHOICE and LiTMUS had broad inclusion and limited exclusion criteria, thus representing patients seen in everyday clinical work. Add-on treatment to lithium was guideline-based and clinically indicated and assessed at every study visit, further strengthening the generalizability of our findings. Finally, our study cohort was larger than most previous trials and we also included measures of several important metabolic measures.
Our primary limitation is confounding by indication. Patients were not randomized to additional treatments, and additional use of antipsychotics and/or anticonvulsants could be initiated during the trial and was based on an evaluation by the treating clinician together with the patient. However, as guidelines consider antipsychotics and anticonvulsants as equal add-on treatment choices, this limitation supports the generalizability as it shows efficacy outcomes relevant for real-world clinical settings. Second, this study represents a secondary post hoc analysis, why cause-and-effect relationships cannot be drawn. Third, we only followed patients for 24 weeks, but metabolic changes often develop over longer periods. Fourth, the serum lithium levels were, by design of the original trials, rather low. Fifth, the trials were performed between 2007 and 2013, with guidelines having changed since then and additional antipsychotic drugs have been marketed. However, recent drug utilization studies have shown that those antipsychotics used in this study were still frequently used several years later (Köhler-Forsberg et al., 2020; Rhee et al., 2020). Sixth, other medications could be used, e.g., somatic medications, but this study focused on additional use of antipsychotics and anticonvulsants.
Conclusion
Among 379 patients randomized to rather low doses of lithium (mean serum lithium between 0.5 and 0.6 mEq/L), we found small but interesting differences in treatment and metabolic side effects between clinically indicated and guideline-based adjunctive antipsychotics versus adjunctive anticonvulsant. This study evaluated relatively low doses in a rather young cohort and a treatment duration of only 6 months, representing a rather short-term treatment for outpatients with bipolar disorder. Nevertheless, our findings emphasize the large somatic burden and a need for intervention on metabolic parameters among patients with bipolar disorder. Clinicians should carefully balance the need for additional antipsychotic use against their potential detrimental metabolic effects. Large randomized clinical trials are needed on patients treated with lithium performing head-to-head comparisons of adjunctive antipsychotics versus adjunctive anticonvulsants.
Supplemental Material
sj-docx-1-anp-10.1177_00048674221077619 – Supplemental material for Lithium plus antipsychotics or anticonvulsants for bipolar disorder: Comparing clinical response and metabolic changes
Supplemental material, sj-docx-1-anp-10.1177_00048674221077619 for Lithium plus antipsychotics or anticonvulsants for bipolar disorder: Comparing clinical response and metabolic changes by Ole Köhler-Forsberg, Louisa G Sylvia, Michael Thase, Joseph R Calabrese, Mauricio Tohen, Charles L Bowden, Melvin McInnis, Dan V Iosifescu, James H Kocsis, Edward S Friedman, Terence A Ketter, Susan L McElroy, Richard C Shelton, Vicki Fung, Michael J Ostacher and Andrew A Nierenberg in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-pptx-1-anp-10.1177_00048674221077619 – Supplemental material for Lithium plus antipsychotics or anticonvulsants for bipolar disorder: Comparing clinical response and metabolic changes
Supplemental material, sj-pptx-1-anp-10.1177_00048674221077619 for Lithium plus antipsychotics or anticonvulsants for bipolar disorder: Comparing clinical response and metabolic changes by Ole Köhler-Forsberg, Louisa G Sylvia, Michael Thase, Joseph R Calabrese, Mauricio Tohen, Charles L Bowden, Melvin McInnis, Dan V Iosifescu, James H Kocsis, Edward S Friedman, Terence A Ketter, Susan L McElroy, Richard C Shelton, Vicki Fung, Michael J Ostacher and Andrew A Nierenberg in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-pptx-2-anp-10.1177_00048674221077619 – Supplemental material for Lithium plus antipsychotics or anticonvulsants for bipolar disorder: Comparing clinical response and metabolic changes
Supplemental material, sj-pptx-2-anp-10.1177_00048674221077619 for Lithium plus antipsychotics or anticonvulsants for bipolar disorder: Comparing clinical response and metabolic changes by Ole Köhler-Forsberg, Louisa G Sylvia, Michael Thase, Joseph R Calabrese, Mauricio Tohen, Charles L Bowden, Melvin McInnis, Dan V Iosifescu, James H Kocsis, Edward S Friedman, Terence A Ketter, Susan L McElroy, Richard C Shelton, Vicki Fung, Michael J Ostacher and Andrew A Nierenberg in Australian & New Zealand Journal of Psychiatry
Footnotes
Clinicaltrials.gov Number
Bipolar CHOICE: NCT01331304.
LiTMUS: NCT00667745
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Dr Ole Köhler-Forsberg discloses honoraria for lectures for Lundbeck Pharma A/S.
Dr Sylvia was a shareholder in Concordant Rater Systems and has served in the past year as a consultant for United Biosource Corporation, Clintara, Bracket and Clinical Trials Network and Institute. Dr Sylvia receives royalties from New Harbinger. She has received grant/research support from NIMH, PCORI, AFSP and Takeda.
Dr Fung receives grant/research support from AHRQ and NIH. Her spouse is an employee of Vertex Pharmaceuticals.
Dr Thase has been an advisor/consultant: to Alkermes, AstraZeneca, Bristol-Myers Squibb, Eli Lilly, Forest Laboratories, GlaxoSmithKline, Janssen Pharmaceuticals, Lundbeck, MedAvante, Merck, Mylan, Neuronetics, Otsuka, Pamlab, PharmaNeuroboost, Pfizer, Rexahn, Roche, Shire, Sunovion, Supernus, Takeda and Teva, as well as the US Food and Drug Administration and the National Institute of Mental Health. During the same time frame, Dr Thase has received honoraria for talks from AstraZeneca, Bristol-Myers Squibb, Eli Lilly, Merck and Pfizer and he has received research grants from Alkermes, AstraZeneca, Eli Lilly, Forest, GlaxoSmithKline, Otsuka, PharmaNeuroboost and Roche, as well as the National Institute of Mental Health and the AHRQ.
Dr Calabrese receives federal funding from the Department of Defense, Health Resources Services Administration and NIMH; he receives research funding or grants from the following private industries or nonprofit funds: Cleveland Foundation, NARSAD and Stanley Medical Research Institute; he receives research grants from Abbott, AstraZeneca, Cephalon, GlaxoSmithKline, Janssen, Eli Lilly and Lundbeck; he serves on the advisory boards of Abbott, AstraZeneca, Bristol-Myers Squibb, Dainippon Sumitomo Pharma, Forest, France Foundation, GlaxoSmithKline, Janssen, NeuroSearch, OrthoMcNeil, Repligen, Schering-Plough, Servier, Solvay/Wyeth, Takeda and Supernus Pharmaceuticals; and he reports CME activities with AstraZeneca, Bristol-Myers Squibb, France Foundation, GlaxoSmithKline, Janssen, Johnson & Johnson, Schering-Plough and Solvay/Wyeth.
Dr Deckersbach’s research has been funded by NIH, NIMH, NARSAD, TSA, IOCDF, Tufts University, DBDAT, Cogito, Sunovion and Otsuka Pharmaceuticals. He has received honoraria, consultation fees and/or royalties from the MGH Psychiatry Academy, BrainCells Inc., Clintara, LLC., Systems Research and Applications Corporation, Boston University, the Catalan Agency for Health Technology Assessment and Research, the National Association of Social Workers Massachusetts, the Massachusetts Medical Society, Tufts University, NIDA, NIMH, Oxford University Press, Guilford Press, and Rutledge. He has also participated in research funded by DARPA, NIH, NIMH, NIA, AHRQ, PCORI, Janssen Pharmaceuticals, The Forest Research Institute, Shire Development Inc., Medtronic, Cyberonics, Northstar and Takeda.
Dr Tohen was a full-time employee at Lilly (1997–2008). He has received honoraria from, or consulted for, Abbott, AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, Lilly, Johnson & Johnson, Otsuka, Merck, Sunovion, Forest, Geodon Richter Plc, Roche, Elan, Alkermes, Allergan, Lundbeck, Teva, Pamlab, Wyeth and Wiley Publishing. His spouse was a full-time employee at Lilly (1998–2013).
Dr Bowden currently has no activities or consultant relationships to disclose.
Dr McInnis has received grants for research support from NIMH, the Heinz C Prechter Research Fund and the Michigan Institute for Clinical Health Research (MICHR). He has received consulting income from the Qatar National Research Foundation and Merck Pharmaceuticals.
Dr Kocsis has received research grants and contracts from AHRQ, NIMH, NIDA, Burroughs Wellcome Trust, Pritzker Consortium, Takeda, Forest, AstraZeneca and Roche. He is on the speaker’s bureau at Pfizer and Merck and on the advisory board at Corcept.
Dr Friedman receives royalties from Springer. He has served as an expert forensic consultant for Thomson Rhodes & Cowie P.C. and Berger and Zavesky Co. L.P.A. Dr Friedman received grant support from NIMH, AHRQ, Novartis, St Jude Medical, Medtronic, Repligen, AstraZeneca, Roche and Takeda and Neosync.
Between May 14, 2010 to present, Dr Ketter had the following financial interests/arrangements or affiliations that could be perceived as real or apparent conflicts of interest: grant/research support from Agency for Healthcare Research and Quality, AstraZeneca Pharmaceuticals LP, Cephalon Inc. (now Teva Pharmaceuticals), Eli Lilly and Company, Pfizer, Inc., Merck & Co., Inc. and Sunovion Pharmaceuticals; consultant/advisory board fees from Acadia Pharmaceuticals, Allergan, Inc., Avanir Pharmaceuticals, Depotmed, Forest Pharmaceuticals, Genentech, Janssen Pharmaceuticals, Merck & Co., Inc., ProPhase, Sunovion Pharmaceuticals, Teva Pharmaceuticals, Bristol-Myers Squibb Company and Cephalon, Inc; lecture honoraria from Abbott Laboratories, Inc., GlaxoSmithKline, Otsuka Pharmaceuticals, Pfizer, Inc. and AstraZeneca Pharmaceuticals LP; and royalties from American Psychiatric Publishing, Inc. In addition, Dr Ketter’s spouse was an employee of and held stock in Janssen Pharmaceuticals.
Dr McElroy is or has been a consultant to or member of the scientific advisory boards of Allergan, Avanir, Bracket, F. Hoffmann-La Roche Ltd., MedAvante, Mitsubishi Tanabe, Myriad, Naurex, Novo Nordisk, Opiant, Otsuka, Shire, Sipnose, Sunovion and Takeda. She is or has been a principal or co-investigator on studies sponsored by the Agency for Healthcare Research & Quality (AHRQ), Alkermes, Allergan, Avanir, Azevan, Cephalon, Forest, Marriott Foundation, Medibio, Myriad, National Institute of Mental Health, Naurex, Orexigen Therapeutics, Inc., Shire and Takeda. She is also an inventor on United States Patent No. 6,323,236 B2, Use of Sulfamate Derivatives for Treating Impulse Control Disorders, and along with the patent’s assignee, University of Cincinnati, Cincinnati, Ohio, has received payments from Johnson & Johnson, which has exclusive rights under the patent.
Dr Shelton has served as a consultant to Acadia Pharmaceuticals, Bristol-Myers Squibb, Cyberonics, Inc., Elan, Corp, Euthymics Bioscience, Cerecor Inc., Clintara LLC, Forest Pharmaceuticals, Janssen Pharmaceutica, Medtronic, Inc., MSI Methylation Sciences, Naurex, Inc., Nestle’ Health Science—Pamlab, Inc., Otsuka Pharmaceuticals, Pfizer, Inc., Ridge Diagnostics, Shire PLC, and Takeda Pharmaceuticals. Dr Shelton has received research grant support from Appian Labs, Elan, Corp, Euthymics Bioscience, Forest Pharmaceuticals, Janssen Pharmaceutica, Naurex, Inc, Novartis Pharmaceuticals, Otsuka Pharmaceuticals, Nestle’ Health Science—Pamlab, Inc., Repligen, Corp, Ridge Diagnostics and Takeda Pharmaceuticals.
Andrew A. Nierenberg is a consultant for the Abbott Laboratories, Alkermes, American Psychiatric Association, Appliance Computing Inc. (Mindsite), Basliea, Brain Cells, Inc., Brandeis University, Bristol-Myers Squibb, Clintara, Corcept, Dey Pharmaceuticals, Dainippon Sumitomo (now Sunovion), Eli Lilly and Company, EpiQ, L.P./Mylan Inc., Forest, Genaissance, Genentech, GlaxoSmithKline, Hoffman LaRoche, Infomedic, Intra-Cellular Therapies, Lundbeck, Janssen Pharmaceutica, Jazz Pharmaceuticals, MedAvante, Merck, Methylation Sciences, Naurex, NeuroRx, Novartis, Otsuka, PamLabs, Parexel, Pfizer, PGx Health, Ridge Diagnostics Shire, Schering-Plough, Somerset, Sunovion, Takeda Pharmaceuticals, Targacept and Teva; consulted through the MGH Clinical Trials Network and Institute (CTNI) for Astra Zeneca, Brain Cells, Inc, Dianippon Sumitomo/Sepracor, Johnson and Johnson, Labopharm, Merck, Methylation Science, Novartis, PGx Health, Shire, Schering-Plough, Targacept and Takeda/Lundbeck Pharmaceuticals. He receives grant/research support from American Foundation for Suicide Prevention, AHRQ, Brain and Behavior Research Foundation, Bristol-Myers Squibb, Cederroth, Cephalon, Cyberonics, Elan, Eli Lilly, Forest, GlaxoSmithKline, Janssen Pharmaceutica, Intra-Cellular Therapies, Lichtwer Pharma, Marriott Foundation, Mylan, NIMH, PamLabs, PCORI, Pfizer Pharmaceuticals, Shire, Stanley Foundation, Takeda, and Wyeth-Ayerst. Honoraria include Belvoir Publishing, University of Texas Southwestern Dallas, Brandeis University, Bristol-Myers Squibb, Hillside Hospital, American Drug Utilization Review, American Society for Clinical Psychopharmacology, Baystate Medical Center, Columbia University, CRICO, Dartmouth Medical School, Health New England, Harold Grinspoon Charitable Foundation, IMEDEX, Israel Society for Biological Psychiatry, Johns Hopkins University, MJ Consulting, New York State, Medscape, MBL Publishing, MGH Psychiatry Academy, National Association of Continuing Education, Physicians Postgraduate Press, SUNY Buffalo, University of Wisconsin, University of Pisa, University of Michigan, University of Miami, University of Wisconsin at Madison, World Congress of Brain Behavior and Emotion, APSARD, ISBD, SciMed, Slack Publishing and Wolters Klower Publishing ASCP, NCDEU, Rush Medical College, Yale University School of Medicine, NNDC, Nova Southeastern University, NAMI, Institute of Medicine, CME Institute, ISCTM. He was currently or formerly on the advisory boards of Appliance Computing, Inc., Brain Cells, Inc., Eli Lilly and Company, Genentech, Johnson and Johnson, Takeda/Lundbeck, Targacept and Infomedic. He owns stock options in Appliance Computing, Inc., Brain Cells, Inc and MedAvante; has copyrights to the Clinical Positive Affect Scale and the MGH Structured Clinical Interview for the Montgomery Asberg Depression Scale exclusively licensed to the MGH Clinical Trials Network and Institute (CTNI).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The Bipolar CHOICE trial was funded by the Agency for Healthcare Research and Quality (AHRQ): 1R01HS019371-01. The LiTMUS trial was funded by the NIMH: NO1MH80001.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
