Abstract
Objective:
Bipolar disorders increase the risk of dementia and show biological and brain alterations, which resemble accelerated aging. Lithium may counter some of these processes and lower the risk of dementia. However, until now no study has specifically investigated the effects of Li on brain age.
Methods:
We acquired structural magnetic resonance imaging scans from 84 participants with bipolar disorders (41 with and 43 without Li treatment) and 45 controls. We used a machine learning model trained on an independent sample of 504 controls to estimate the individual brain ages of study participants, and calculated BrainAGE by subtracting chronological from the estimated brain age.
Results:
BrainAGE was significantly greater in non-Li relative to Li or control participants, F(2, 125) = 10.22, p < 0.001, with no differences between the Li treated and control groups. The estimated brain age was significantly higher than the chronological age in the non-Li (4.28 ± 6.33 years, matched t(42) = 4.43, p < 0.001), but not the Li-treated group (0.48 ± 7.60 years, not significant). Even Li-treated participants with partial prophylactic treatment response showed lower BrainAGE than the non-Li group, F(1, 64) = 4.80, p = 0.03.
Conclusions:
Bipolar disorders were associated with greater, whereas Li treatment with lower discrepancy between brain and chronological age. These findings support the neuroprotective effects of Li, which were sufficiently pronounced to affect a complex, multivariate measure of brain structure. The association between Li treatment and BrainAGE was independent of long-term thymoprophylactic response and thus may generalize beyond bipolar disorders, to neurodegenerative disorders.
Introduction
Bipolar disorders (BD) are severe, recurrent psychiatric conditions affecting 1–3% of the global population (Grande et al., 2016). Due to early onset and lifelong course, BD are the sixth leading cause of disability among all diseases (Whiteford et al., 2013). In addition, BD markedly increase the risk of dementia (Diniz et al., 2017), another major contributor to psychosocial burden. Despite these substantial societal costs and human suffering, we do not yet fully understand the links between BD and neurodegenerative disorders.
On the molecular level, episodes of depression or mania increase allostatic load (Kapczinski et al., 2008) and may lead to increased oxidative stress, low-grade systemic inflammation, reduction of neurotrophins (Berk et al., 2011), accelerated cellular aging (Do Prado et al., 2013) and telomere shortening (Powell et al., 2018)—mechanisms which also operate in dementias (Marchesi, 2011). On macroscopic level, BD have consistently been associated with structural brain changes (Hibar et al., 2016, 2018). These are most consistently found and most pronounced in mesiotemporal regions, including the hippocampus (Hajek et al., 2012; Hibar et al., 2016; Lyoo et al., 2010), which is also among the first regions showing atrophy in dementias (Harper et al., 2017). Furthermore, neurostructural alterations tend to be mild or even absent in the early stages of BD (Fusar-Poli et al., 2012; Hajek et al., 2019) and become more pronounced later in the course of illness (Hibar et al., 2016, 2018). The accumulation of biological and brain alterations in the course of BD is sometimes termed neuroprogression (Berk et al., 2011). On the whole system level, BD are associated with increased prevalence and earlier onset of comorbid age-related conditions (Kapczinski et al., 2008; Powell et al., 2018; Rizzo et al., 2014; Vancampfort et al., 2016) and with reduced life expectancy and premature death (Grande et al., 2016; Laursen, 2011; Walker et al., 2015). Taken together, these findings provide indirect evidence that BD are associated with accelerated aging, particularly of the brain.
Interestingly, certain medications may counter these processes. Replicated studies in tissue cultures and animal models have shown that pre-treatment with Li protects neurons from a range of toxic stimuli in a range of neurodegenerative models (Lauterbach and Mendez, 2011). Cross sectional, as well as prospective studies in human participants, including one randomized controlled trial (Lyoo et al., 2010), have repeatedly shown association between Li treatment and larger gray matter (GM) volumes (Hajek et al., 2012; Monkul et al., 2007; Moore et al., 2000). Pharmacoepidemiological studies showed a lowering of risk for Alzheimer dementia (AD) with repeated prescriptions of Li (Gerhard et al., 2015; Kessing et al., 2008). Last but not least, randomized controlled trials have demonstrated beneficial effects of Li on symptoms and biomarkers of amnestic mild cognitive impairment or AD (Forlenza et al., 2019; Matsunaga et al., 2015). All in all, there is sufficient evidence suggesting that Li affects the mechanisms involved in accelerated aging and neuroprogression, and that these effects may be detectable in human participants (Powell et al., 2018). However, no previous studies have investigated the association between Li treatment and brain aging.
Access to large samples of brain scans and analytical advances involving machine learning/pattern recognition, allow us to estimate the biological age of the brain from structural magnetic resonance imaging (MRI) (Cole et al., 2018; Kolenic et al., 2018; Koutsouleris et al., 2014). The discrepancy between the estimated brain age and chronological age, so called Brain Age Gap Estimate (BrainAGE), captures diffuse, multivariate neurostructural alterations as a single number. This mitigates the problem of multiple comparisons and yields relatively unbiased estimates of effect size. As a highly multivariate, composite index of brain structure, BrainAGE is ideally suited to investigate the complex effects of BDs and Li treatment on the brain. In addition, while many manuscripts have described an association between Li treatment and individual regions of interest (Hajek and Weiner, 2016), a summary measure, such as the BrainAGE, could provide new insights about the effects of Li on the whole brain level.
Here, for the first time, we applied this novel technology to study the effects of psychiatric medications on the brain. We hypothesized that the BrainAGE will be significantly greater in the non-Li than in the Li treated or control groups. In addition, we postulated that within subjects, only BD participants not treated with lithium will have a significantly greater estimated brain age than chronological age.
Materials and methods
We recruited BD participants from the Maritime Bipolar Registry (Hajek et al., 2005), Mood Disorders Program and associated Li treatment clinic at Dalhousie University (Halifax, NS, Canada) and controls through advertisement. BD participants had regular follow-up visits at the clinic, including monitoring of Li levels at least twice per year. Recruitment of Li treated patients from a specialized clinic ensured that Li levels fell within the therapeutic range. This prevented sub-therapeutic levels, which could be insufficient to elicit neuroprotective changes, or levels above the therapeutic range, which could lead to neurotoxicity. The study was approved by the Research Ethics Boards of IWK Health Center and Capital District Health Authority (Halifax, NS, Canada) and all included participants provided informed consent.
Inclusion/exclusion criteria
Participants with BD were required to (1) have the diagnosis of BD made by a psychiatrist and (2) be at least 20 years of age. Patients were excluded, if they had (1) the diagnosis of an organic mood disorder, (2) mood disorder not otherwise specified and (3) more than one lifetime course of electroconvulsive therapy (ECT) or ECT within the last 6 months. The neuropsychiatrically healthy individuals were excluded if they had a personal history of psychiatric disorders. Participants from any group were excluded if they (1) met any MRI exclusion criteria, or suffered from; (2) substance abuse in the last 12 months; (3) cerebrovascular disease; (4) neurodegenerative disorders; or (5) macroscopic pathology on MRI. BD participants were divided into those with versus without Li treatment at the time of scanning, referred to as Li and non-Li group respectively. The non-Li group was further divided into those who were Li-naïve and those who had had past history of Li treatment.
Diagnostic assessments
For the detailed description of the diagnostic assessment, please see Hajek et al. (2005). Briefly, the diagnostic interviews were performed by pairs of clinicians, according to the Schedule for Affective Disorders and Schizophrenia, Lifetime version (SADS-L) (Endicott and Spitzer, 1978). Diagnostic information was reviewed in a blinded fashion during research team consensus meetings, which included a minimum of two psychiatrists. We also used National Institutes of Mental Health (NIMH) life charts (NIMH-LCMTM) (Denicoff et al., 2000) to obtain a detailed description of clinical course and Alda Scale to assess prophylactic response to Li (Grof et al., 2002; Manchia et al., 2013). Each control participant also underwent the SADS-L interview and was included if found to have no personal or family history of Axis I psychiatric disorders.
MRI methods
All magnetic resonance acquisitions were performed with a 1.5 Tesla General Electric Signa scanner (General Electric Medical Systems, Fairfield, Connecticut) and a standard quadrature head coil. After a localizer scan, a T1-weighted spoiled gradient recalled scan was prescribed (flip angle = 40°, echo time = 5 ms, repetition time = 25 ms, field of view = 24 cm x 18 cm, matrix = 256 x 160 pixels, number of excitations = 1, no interslice gap, 124 slices, 1.5 mm thick).
BrainAGE estimation
We applied one of the original machine learning methods for assessing brain age, which was designed by the Structural Brain Mapping Group, Department of Psychiatry, University of Jena, Jena, Germany (Franke et al., 2010). We published extensive validation of this method and justification of each individual processing step in a previous methodological manuscript (Franke et al., 2010). This method accurately and reliably estimates the age of individual brains across wide age range (Franke et al., 2012, 2010), and we used it previously (Hajek et al., 2019; Kolenic et al., 2018).
The analyses included (1) preprocessing of the raw T1-weighted image data using standard voxel-based morphometry pipelines in SPM (www.fil.ion.ucl.ac.uk/spm/), running under MATLAB (www.mathworks.com) (Franke et al., 2010). Prior to processing, we performed detailed visual and statistical quality controls. T1-weighted images were corrected for bias-field inhomogeneity, then spatially normalized and segmented into GM, white matter (WM) and cerebrospinal fluid within the same generative model, while accounting for partial volume effects, applying adaptive maximum a posteriori estimations, and using a hidden Markov Random Field model. Subsequently, we applied (2) data reduction using smoothing and principal component analysis (MATLAB Toolbox for Dimensionality Reduction, http://ict.ewi.tudelft.nl/~lvandermaaten/Home.html), in or-der to reduce computational costs, avoid severe over-fitting and produce a robust and widely applicable age-estimation model. Lastly, we performed (3) estimation of brain age using relevance vector regression (RVR). We utilized RVR, which was introduced as a Bayesian alternative to support vector machines (SVM) for obtaining sparse solutions to pattern recognition tasks. In contrast to SVM, it does not require additional parameter optimization during the training phase. Also, SVM uses the ‘non-typical’ cases, whereas RVR uses the ‘most-typical’ cases for finding the optimal regression line. These analytical refinements likely contribute to the fact that the RVR was shown to be more stable and accurate then SVM in capturing the typical age-related changes across the whole brain, while avoiding overfitting. We utilized a linear kernel, since age estimation accuracy is not improved when using non-linear kernels, which may also increase the risk of overfitting.
We trained the RVR model using an independent sample of 504 healthy individuals (230 males) from the IXI database, aged 19–86 years (www.brain-development.org). We applied a cross validation approach, which produced an unbiased estimate of performance in new individuals. We used the resulting age prediction model to individually estimate brain age in our study participants, thus completely separating the training and testing phase. We used ‘The Spider’ (http://people.kyb.tuebingen.mpg.de/spider/) toolbox, running under MATLAB, to train the model and predict individual brain ages. The brain age model explained over 80% of the baseline age variance (R2 = 0.83) in healthy subjects in this study (testing sample), with mean absolute error (MAE) of 4.62 years and an excellent agreement between chronological age and brain age (intraclass correlation [ICC] = 0.94, 95% confidence interval [CI] = [0.90, 0.97], p < 0.001). This is comparable to our previous validation of the method (MAE = 5.08, R2 = 0.83) (Franke et al., 2010) and to other models, i.e., MAE = 4.6 years, R2 = 0.83 (Koutsouleris et al., 2014), and MAE = 4.31, R2 = 0.79 (Schnack et al., 2016). Our outcome measure was the whole brain BrainAGE, which is the difference between estimated brain age and chronological age (Franke et al., 2010). To evaluate the contribution of each tissue type to the whole brain changes, we also acquired separate BrainAGE estimates from only GM and only WM.
Statistical analyses
All statistical analyses were conducted in R Studio (R version 3.3.2). To compare clinical and demographic variables, we used t-test, one-way analysis of variance (ANOVA) or Chi-square test, as appropriate. We initially tested for an association between age or sex and BrainAGE to select which demographic variables to control for. We then performed an analysis of covariance (ANCOVA) with BrainAGE as the dependent variable, group (Li, non-Li, control) as the grouping variable, while covarying for demographic variables, which were significantly associated with BrainAGE. This approach followed the most recent recommendations for analyses using BrainAGE (Le et al., 2018a). This primary analysis was followed by post hoc pairwise comparisons, corrected using Tukey’s Honest Significant Difference test. To compare brain and chronological age within subjects, we used paired t-test.
In exploratory analyses, we repeated the primary analyses using BrainAGE calculated separately from GM and WM. To explore association between BrainAGE and clinical variables, we utilized Pearson correlations or two sample t-tests, where appropriate. For time-related variables, we used multiple regression to control for chronological age. Within the BD group, we separately explored associations between anticonvulsant or antipsychotic (AP) treatment and BrainAGE, using ANCOVA and controlling for age. Medications, which were significantly associated with BrainAGE were then included in the ANCOVA model, alongside Li treatment. We also tested, whether the association between Li treatment and BrainAGE would persist after controlling for treatments with both APs and anticonvulsants.
Results
We recruited 84 BD participants (43 non-Li and 41 Li treated) and 45 controls. The groups were comparable in sex and age (Table 1). BrainAGE was associated with age, r(127) = –0.49, p < 0.001, but not sex, t(127) = –0.85, p = 0.40. Thus, we adjusted for age in the following analyses.
Sample description.
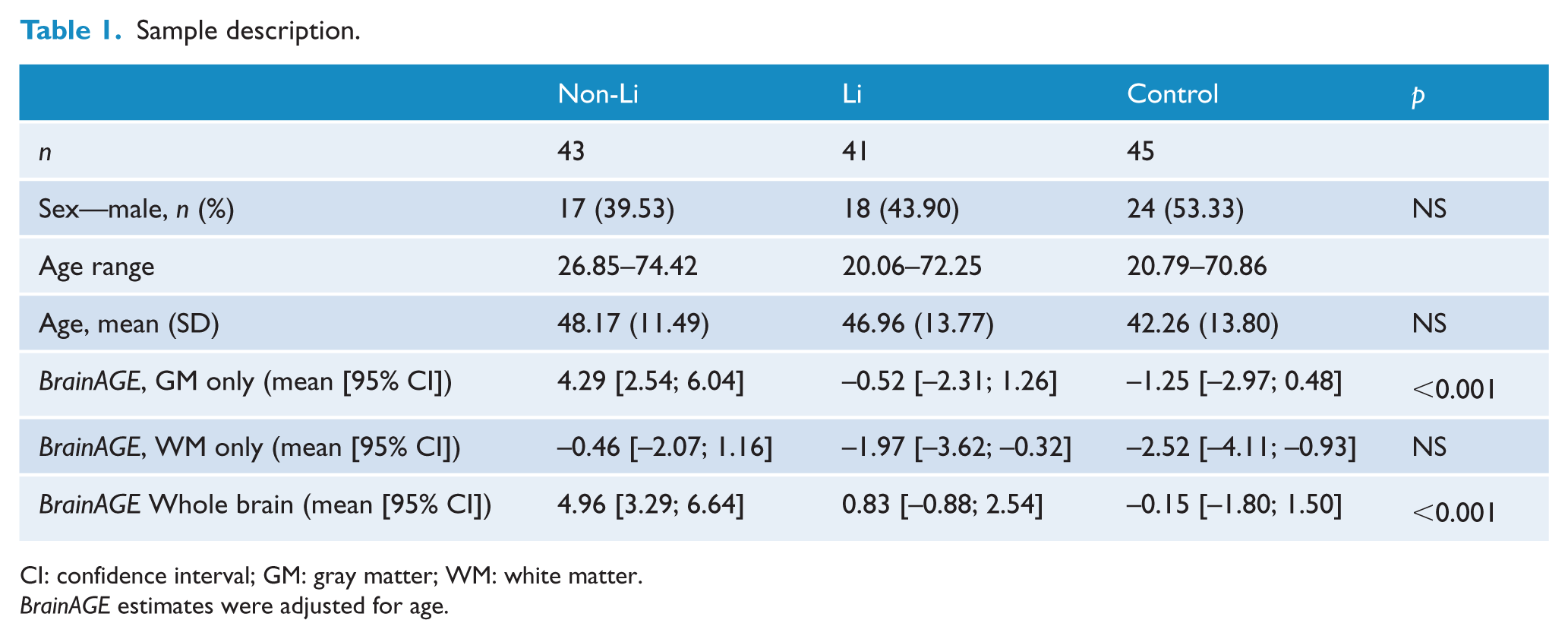
CI: confidence interval; GM: gray matter; WM: white matter.
BrainAGE estimates were adjusted for age.
Primary analyses
The Li-treated, non-Li and control participants differed in BrainAGE, F(2, 125) = 10.22, p < 0.001, Table 1, Figure 1. Post hoc comparisons showed significantly greater BrainAGE in non-Li relative to Li, (mean difference = 4.10, 95% CI = [1.24, 6.96], p corr = 0.002), or control (mean difference = 4.96, 95% CI = [2.16, 7.76], p corr < 0.001) participants, with no differences between the Li-treated participants and controls (mean difference = 0.86, 95% CI = [–1.97, 3.69], p corr = 0.75).
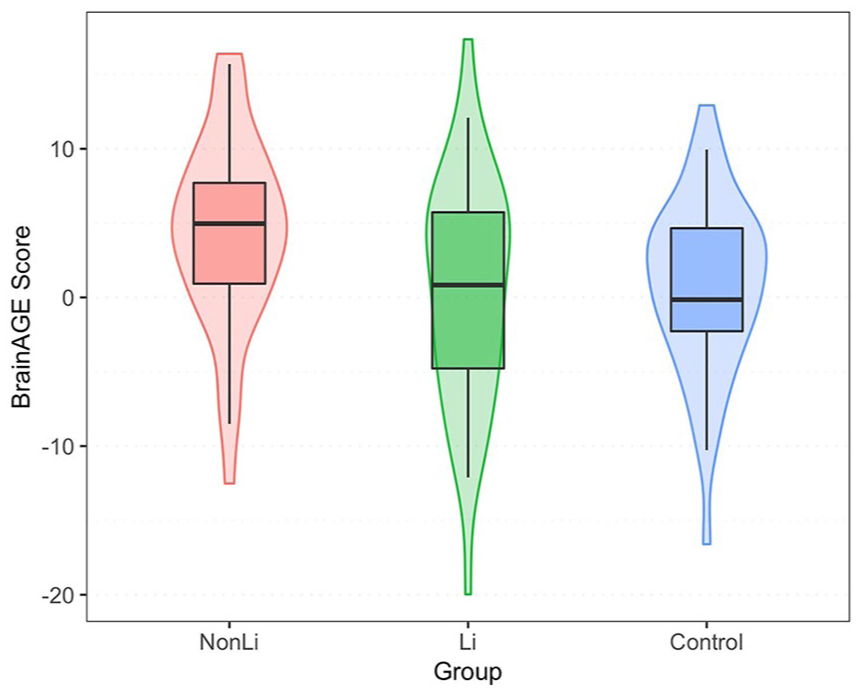
Boxplot and violin plot of BrainAGE distribution in the Li, non-Li and control participants. The box shows the interquartile range, the vertical line inside the box is the mean age adjusted BrainAGE. 95% of observed BrainAGE scores fell inside the whiskers.
The estimated brain age in non-Li BD participants was higher than their chronological age by an average of 4.28 ± 6.33 years, matched t(42) = 4.43, p < 0.001, whereas the Li-treated participants had a comparable estimated brain and chronological age, mean difference = 0.48 ± 7.60 years, matched t(40) = 0.41, p = 0.69, Figure 2.
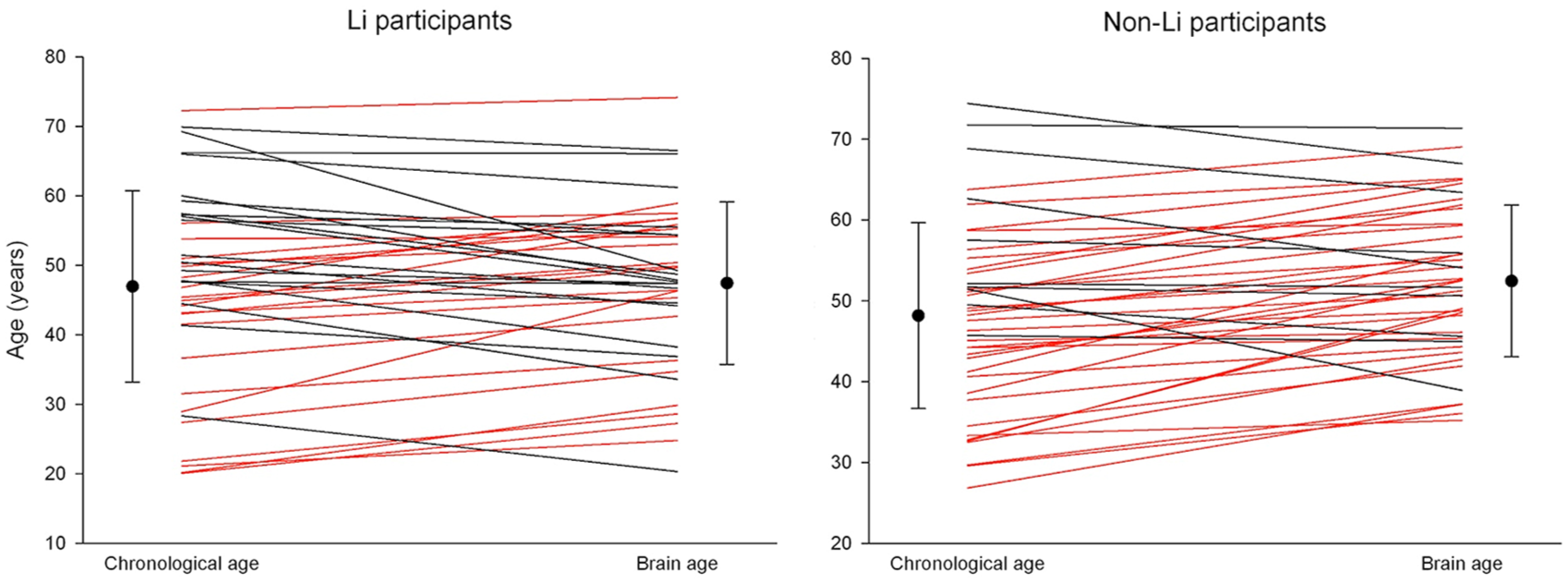
Within subject comparison of the chronological age and the estimated brain age, separately in the Li-treated and non-Li participants. Red color denotes participants with greater estimated brain than chronological age.
Exploratory analyses
We found the same pattern of between-group differences in BrainAGE calculated from GM, F(2, 125) = 11.48, p < 0.001, while BrainAGE calculated from WM did not significantly differ between the groups, F(2, 125) = 1.71, p = 0.19, Table 1. The proportion of participants who had a greater estimated brain than chronological age was higher among the non-Li (76.7%) than the Li-treated participants, 53.7%, χ2(1) = 4.94, p = 0.03, Figure 2.
Effects of medications and other clinical variables
The Li and non-Li groups were comparable in relevant clinical variables (Table 2).
Demographic and clinical information of participants with bipolar disorders.
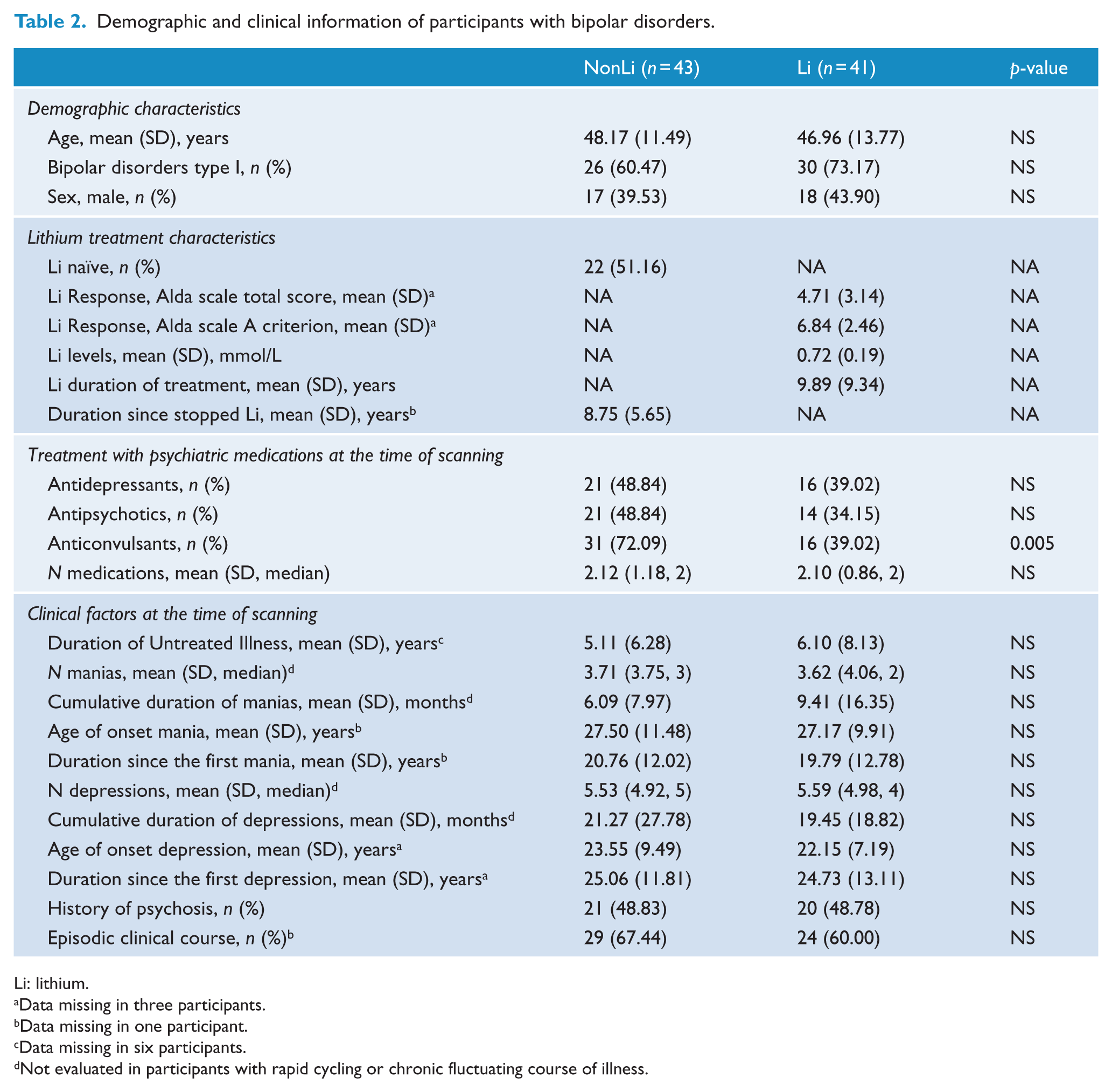
Li: lithium.
Data missing in three participants.
Data missing in one participant.
Data missing in six participants.
Not evaluated in participants with rapid cycling or chronic fluctuating course of illness.
We found a significant difference between Li-treated, previously Li-treated, and Li-naïve participants, F(2, 80) = 6.30, p = 0.003. Post hoc comparisons demonstrated that participants currently treated with Li showed significantly lower BrainAGE than previously Li-treated participants (mean difference = –5.35, 95% CI = [–9.09, –1.62], p corr = 0.003) who had comparable BrainAGE to the Li naïve group (mean difference = 2.34, 95% CI = [–1.90, 6.59], p corr = 0.39). The past duration of Li treatment in the previously Li treated participants was 8.06 ± 9.29 years, range 0.125–32 years and the latency since Li discontinuation was 8.75 ± 5.65 years. There was no association between latency since Li discontinuation, past or current duration of Li treatment and BrainAGE.
BrainAGE was greater in participants with, relative to those without, current AP treatment, F(1, 81) = 6.61, p = 0.01. Factorial ANOVA with current Li treatment (yes, no) and current AP treatment (yes, no) and age as covariate showed significant main effects of each treatment, but in opposite directions—i.e., positive association between AP treatment and BrainAGE, F(1, 80) = 4.84, p = 0.03, and negative association between Li treatment and BrainAGE, F(1, 80) = 8.69, p = 0.004. Consequently, we found the highest BrainAGE in non-Li, AP treated participants (5.90, 95% CI = [3.73, 8.07]) and the lowest BrainAGE in Li-treated participants without AP treatment (–0.67, 95% CI = [–2.65, 1.32], Figure 3). History of psychotic symptoms was not associated with BrainAGE, F(1, 81) = 0.36, p = 0.55.
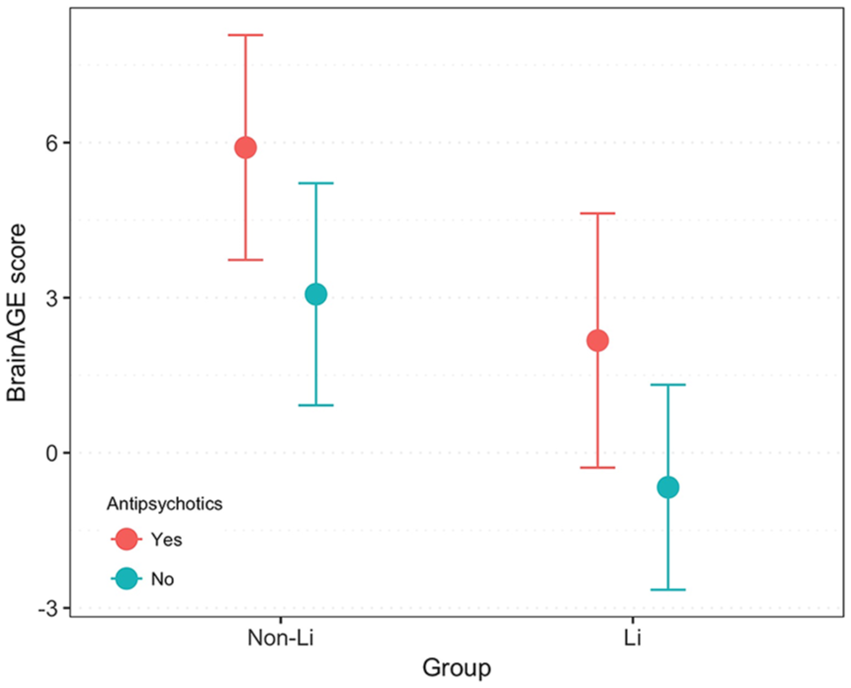
BrainAGE in Li and non-Li participants, grouped by treatment with antipsychotics—age adjusted means and 95% confidence intervals.
Even Li-treated participants with partial prophylactic treatment response (Alda scale total score < 7) showed lower BrainAGE than the non-Li group, F(1, 64) = 4.80, p = 0.03.
There were no differences in BrainAGE between participants with, relative to those without current anticonvulsant treatment, F(1, 81) = 2.00, p = 0.16. The association between Li treatment and BrainAGE remained significant, F(1, 79) = 6.32, p = 0.01, when we controlled for exposure to both APs and anticonvulsants.
BrainAGE was not associated with number of concomitant medications, number and duration of manias or depressions, duration since the first manic or depressive episode, exposure to antidepressants, Li levels, clinical course, duration of untreated illness, or history of psychotic symptoms.
Discussion
Among BD participants, only those not currently treated with Li had an advanced estimated brain age relative to their chronological age, as well as greater BrainAGE scores relative to their Li-treated counterparts or controls. In contrast, BD patients with similar illness burden and ongoing Li treatment showed comparable brain and chronological age and comparable BrainAGE scores to controls. Only current, not past exposure to Li was associated with lower BrainAGE. Exploratory analyses suggested that exposure to APs was associated with greater BrainAGE, even among Li-treated participants. Consequently, BrainAGE was lowest in Li-treated participants without AP exposure and highest in AP-treated participants without Li treatment. There were no differences in BrainAGE between Li responders and non-responders. The association between Li treatment and BrainAGE was greater in the GM than in the WM.
This is the first study to examine the effect of Li treatment on the brain age. Our findings are congruent with cross sectional and longitudinal studies, which found a positive association between Li treatment and neurostructural measures (Hibar et al., 2018; Lyoo et al., 2010; Monkul et al., 2007) and also with studies, which reported brain volumetric alterations in BD participants not treated with Li (Hibar et al., 2018). In keeping with our findings, a previous study has also suggested that the effects of Li were more pronounced in the GM than in the WM (Sassi et al., 2002). More broadly, our findings are also in line with epidemiological studies reporting that BD are a risk factor for dementia, a prototypical condition of advanced brain aging, and that Li treatment may lower this risk to the population level (Gerhard et al., 2015; Kessing et al., 2008). The association between Li treatment and BrainAGE is also congruent with studies showing positive effects of Li on biomarkers of Alzheimer’s disease and cognitive performance (Forlenza et al., 2019; Matsunaga et al., 2015).
Two previous studies found no association between BrainAGE and BD. However, neither of these studies adjusted for Li treatment or provided information about the illness burden (Nenadić et al., 2017; Shahab et al., 2018). Another investigation reported that participants in the early stages of BD had comparable BrainAGE to controls and comparable brain and chronological age (Hajek et al., 2019). In addition, participants with established BD showed BrainAGE alterations, which were intermediate between controls and participants with schizophrenia (Koutsouleris et al., 2014). The advanced BrainAGE later in the course of illness, as demonstrated in this study, but not in participants at risk or early in the course of BD (Hajek et al., 2019), is in line with other morphometric studies and may support the neuroprogressive nature of BD.
The association between Li exposure and BrainAGE may have several explanations. It is possible that it reflects differences between Li-treated and untreated participants in clinical factors, which may themselves be associated with brain structure. However, the Li and non-Li participants in our study were comparable in relevant clinical variables, including the duration of illness, number of episodes, cumulative time spent ill and clinical course, making this explanation unlikely.
Second, it is possible that the Li responsive participants are a specific subgroup and that only the Li non-responsive illness is more prone to ‘neuroprogression’. Importantly, treatment response to Li was not a selection criterion in this study. As is typically the case in a clinical setting, the Li-treated group contained both full and partial Li responders. This is related to the fact that full response may be unattainable, or Li may be used for antisuicidal effects or as an augmentation. Also, many of the non-Li participants were Li naïve and there could have been Li responders among them. All in all, potential differences between responders and non-responders did not provide a sufficient explanation for the findings of this study.
Third, instead of detecting positive effects of Li, perhaps we captured the negative effects of other medications—i.e., the non-Li group showed larger BrainAGE as a result of exposure to treatments other than Li. This is unlikely, as Li and non-Li groups differed only in exposure to anticonvulsants, which were not associated with BrainAGE. Interestingly, treatment with AP medication was associated with advanced BrainAGE, but Li treatment remained negatively associated with BrainAGE even when we controlled for exposure to other mood stabilizers (APs and anticonvulsants). Taken together the most parsimonious interpretation is that the observed differences in BrainAGE were related to differential exposure to Li.
Interestingly, only current, not past Li exposure was negatively associated with BrainAGE. Participants who stopped Li treatment on average 8.75 ± 5.65 years ago showed significantly greater BrainAGE than those currently treated with Li. It is possible that treatment duration for past Li users was not long enough to garner the associated structural changes. However, this appears unlikely as the average past exposure of Li in this group was 8.06 ± 9.29 years, and other studies found positive changes in global GM volume following just 4 weeks of Li treatment (Moore et al., 2000). Alternatively, perhaps participants with particularly pronounced brain alterations were less likely to respond to Li and stopped the treatment because of inadequate response. The lack of association between Li treatment response and BrainAGE and the fact that some of the previously Li treated participants had used Li for years, makes this explanation unlikely. It seems more probable that similar to other clinical outcomes, the neuroanatomical benefits associated with Li treatment are eventually lost following the cessation of treatment.
One key question is, whether the putative neuroprotective effects of Li are direct or indirect. Perhaps Li protects brain structure simply by preventing episodes of illness, which lead to brain alterations—an indirect effect secondary to treatment response. In this case, the effects of Li would be isolated to participants with mood disorders, who respond to the treatment. Alternatively, Li may directly influence the biochemical pathways associated with neurotrophic and antiapoptotic mechanisms. In this case, the effects of Li on brain structure would not be restricted to Li responders and would generalize to other conditions. In support of the direct effect, even Li-treated participants with partial prophylactic response showed lower BrainAGE than the non-Li group. This is also in keeping with a previous study, where the effects of Li on hippocampal volumes were found even in participants who continued to have episodes of illness while on Li (Hajek et al., 2014). Furthermore, Monkul et al. (2007) reported a positive association between Li and brain structure in control participants without BD. In addition, a large body of preclinical literature showed that Li prevented neuronal damage in a range of neurotoxic models, including those unrelated to BD (Lauterbach and Mendez, 2011). Finally, randomized controlled trials have shown positive effects of Li on symptoms and biomarkers of amnestic mild cognitive impairment or AD in participants without BD (Forlenza et al., 2019; Matsunaga et al., 2015). Taken together, these findings strongly suggest that the effects of Li on brain structure are direct and do not represent an epiphenomenon of treatment response. This is similar to the antisuicidal properties of Li, which are also independent of treatment response and occur even among non-responders to treatment (Ahrens and Müller-Oerlinghausen, 2001).
The diffuse actions of Li make it difficult to identify the specific targets that contribute to the putative neuroprotective effects. Preclinical research has suggested that Li functions through the inhibition of glycogen synthase kinase-3, and by upregulating neuroprotective proteins such as brain-derived neurotrophic factor and B-cell lymphoma 2 (Quiroz et al., 2010). However, some of these actions are shared with other medications, such as APs or anticonvulsants, which are negatively associated with brain structure (Andreasen et al., 2013; Fusar-Poli et al., 2013; Ho et al., 2011). Perhaps the positive effects of Li on brain structure are related to some unique pharmacodynamic property of Li or unique combination of lithium’s effects.
Our exploratory analyses revealed some interesting findings which require replication. We noted a nominal association between AP treatment and greater BrainAGE, even among Li-treated participants. This is in keeping with previous results from human and animal studies, which have reported reduced brain volume in those chronically exposed to AP treatment (Andreasen et al., 2013; Fusar-Poli et al., 2013; Ho et al., 2011). More importantly, this is in keeping with a previous study, in which brain age acceleration rate was positively associated with cumulative AP intake (Schnack et al., 2016). However, our study was not specifically designed to test the effects of AP, thus making the interpretation of this finding difficult. Psychotic disorders have repeatedly been associated with lower GM volumes and advanced BrainAGE, regardless of treatment (Hajek et al., 2019; Nenadić et al., 2017; Pantelis et al., 2003). In this study, history of psychotic symptoms, was however not associated with advanced brain age. It is also possible that the effects of AP on the brain are mediated by AP-induced metabolic changes, including obesity and type II diabetes, which are themselves associated with neurostructural alterations and advanced BrainAGE (Kolenic et al., 2018). Thus, the association between AP treatment and advanced BrainAGE could be indirect and needs to be more thoroughly tested in future studies.
This study has several limitations. We used a cross-sectional, naturalistic design. Only a prospective, randomized study could establish a causal link between Li treatment and preserved BrainAGE. However, it would be virtually impossible to achieve an average of 9.89 years of Li exposure in such a study. Also, it would be difficult to justify and obtain funding for a randomized controlled trial without evidence from naturalistic investigations for the association between BrainAGE and Li treatment, which we showed for the very first time. While we did not detect significant differences between Li responders and non-responders, there may still be neural differences between these groups, which might emerge with larger samples or different methods. Although called BrainAGE, this metric is sensitive to disease effects, which act in the same direction as aging (Kolenic et al., 2018; Koutsouleris et al., 2014; Nenadić et al., 2017). We used it as a cumulative measure of diffuse brain structural alterations. Due to limitations of MRI, we cannot describe the histological features that contributed to the global alterations as captured by the BrainAGE. We did not collect genetic, neurocognitive data, nor did we use other brain imaging modalities.
The advantages of this study include detailed clinical assessments in a large sample of well-characterized patients who continue to be prospectively followed; an interesting measure of brain structure, and a conservative application of machine learning where we completely separated training from testing. The results of this study were robust, persisted in subgroup analyses, and were consistent with much of the previous research. Testing effects of medications on multiple regions or voxels decreases statistical power and if uncontrolled may increase the risk of false positives. As BrainAGE takes into account alterations throughout the whole brain, while obviating the need for multiple comparisons/region of interest selection (Cole et al., 2018), it could serve as a sensitive outcome measure in pharmaconeuroimaging studies (Le et al., 2018b).
To conclude, BD were associated with greater, whereas Li treatment with lower discrepancy between brain and chronological age. These findings support the neuroprotective effects of Li, which appear to be sufficiently pronounced to affect a complex, multivariate measure of brain structure, such as the BrainAGE. The negative association between Li treatment and brain age was detectable only in the currently treated participants, but not in those with past Li exposure. Similar to other clinical outcomes of Li treatment, the beneficial effects of Li on brain structure may eventually dissipate, following the cessation of treatment. Importantly, the association between Li treatment and BrainAGE was independent of long-term prophylactic response and occurred even in participants with partial Li response. This raises the possibility that the effects of Li on brain structure may generalize beyond BD and might be of benefit to those with neurodegenerative disorders. Future studies should prospectively investigate the association between Li treatment and BrainAGE to test whether Li can prevent accumulation of brain alterations associated with BD and to assess the prognostic significance of advanced brain age on clinical outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by funding from the Canadian Institutes of Health Research (103703, 106469 and 142255), Nova Scotia Health Research Foundation, Dalhousie Clinical Research Scholarship to T. Hajek, Brain & Behavior Research Foundation (formerly NARSAD); 2007 Young Investigator and 2015 Independent Investigator Awards to T. Hajek, the Ministry of Health, Czech Republic (grants number 16-32791A, 16-32696A). The sponsors of the study had no role in the design or conduct of this study; in the collection, management, analysis and interpretation of the data; or in the preparation, review, or approval of the manuscript. The authors report no biomedical financial interests or potential conflicts of interest.
