Abstract
Background:
Previous studies have implicated childhood trauma and abnormal brain-derived neurotrophic factor in the pathogenesis of schizophrenia. Here, we explored whether brain-derived neurotrophic factor levels mediated the relationship between childhood trauma and psychopathological symptoms in patients with first-episode schizophrenia.
Methods:
Patients with first-episode schizophrenia (n = 192) and healthy controls (n = 136) were enrolled. Childhood traumatic experiences and psychopathology were assessed by Childhood Trauma Questionnaire and Positive and Negative Syndrome Scale, respectively. Enzyme-linked immunosorbent assay was used to quantify brain-derived neurotrophic factor levels.
Results:
The patients with first-episode schizophrenia experienced more severe childhood trauma and had lower serum brain-derived neurotrophic factor levels than healthy controls. Emotional abuse and Childhood Trauma Questionnaire total score showed positive correlation with Positive and Negative Syndrome Scale positive, general psychopathological subscore and total score. Emotional neglect showed positive correlation with Positive and Negative Syndrome Scale positive subscore. Physical neglect was positively associated with Positive and Negative Syndrome Scale negative subscore. Emotional neglect and Childhood Trauma Questionnaire total score were negatively correlated with serum brain-derived neurotrophic factor levels. The serum brain-derived neurotrophic factor levels mediated the relationship between both Childhood Trauma Questionnaire total score and Positive and Negative Syndrome Scale total score and negative symptoms in the patients. The brain-derived neurotrophic factor levels also mediated the relationship between emotional neglect and Positive and Negative Syndrome Scale total score in the patients.
Conclusion:
Childhood trauma might contribute to the clinical symptoms of schizophrenia by affecting brain-derived neurotrophic factor levels. Perhaps we can prevent schizophrenia by reducing childhood traumatic experiences.
Keywords
Introduction
Schizophrenia is a group of chronic, multifactorial and severe mental disorders that are characterized by abnormalities in cognitive, emotional, behavioral activities and significant occupational and social function impairment (Koshiyama et al., 2021). At present, the etiology of schizophrenia is not entirely clear, despite numerous hypotheses of the etiology of schizophrenia (Valton et al., 2017). According to the neurodevelopment hypothesis, neuroplasticity and abnormalities in brain development play a central role in the pathophysiology of schizophrenia (Murray et al., 2017). Brain-derived neurotrophic factor (BDNF) promotes the development, differentiation, growth and regeneration of neurons (Sato, 2017). However, whereas some studies showed lower BDNF levels in patients with first-episode schizophrenia (PFES) in comparison with healthy controls (HC) (Çakici et al., 2020; Song et al., 2015), others showed the opposite (Skibinska et al., 2019) or no differences (Kawashima et al., 2009). Likewise, how BDNF levels relate to the severity of psychiatric symptoms as measured by Positive and Negative Syndrome Scales (PANSS) for schizophrenia remains unclear. Some studies showed higher BDNF levels in positive correlation with symptom severity (Binford et al., 2018; Kudlek Mikulic et al., 2017) and other studies showed the opposite (Chiou and Huang, 2016) or no correlations (Bakirhan et al., 2017).
Studies have accumulated to show a significant effect of childhood trauma on the occurrence of schizophrenia (Sallis et al., 2020). Childhood trauma not only increases but may be causally related to the risk of schizophrenia (Cancel et al., 2019); cumulative childhood trauma has a dose–response relationship with the occurrence of schizophrenia (Mall et al., 2019). Patients with schizophrenia exposed to childhood trauma have earlier illness onset (Neill et al., 2020), more severe auditory hallucinations and delusions (Bailey et al., 2018), more suicide attempts and suicidal behavior (Mohammadzadeh et al., 2019), poorer cognitive function (Wells et al., 2020) and poorer response to treatment (Misiak and Frydecka, 2016), which are risk factors for treatment resistance (Hassan and De Luca, 2015). Mall et al. (2019) showed that physical abuse was positively correlated with PANSS total score, general psychopathology and PANSS positive symptoms, whereas physical neglect was negatively correlated with PANSS negative symptoms. Other studies showed that only total childhood trauma score is positively correlated with PANSS positive symptoms in PFES but not chronic schizophrenia (Kilicaslan et al., 2017). Thus, the relationship between childhood trauma, particularly the nature of the traumatic experiences, and symptom severity remains to be clarified.
How childhood trauma increases the risk of schizophrenia is not clear. Childhood trauma may affect brain structure and function by undermining the neuroprotective effects of BDNF (Van Velzen et al., 2016). Investigating whether or how BDNF levels mediate the inter-relationship between childhood trauma and symptom severity in the PFES may facilitate research in this direction.
Material and methods
Participants and study procedures
We recruited 192 patients who were hospitalized in Beijing Huilongguan Hospital. Inclusion criteria were as follows: (1) meeting the diagnostic criteria of schizophrenia according to the Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV); (2) total disease duration less than 3 years; (3) medicated with antipsychotics for less than 2 weeks; and (4) age from 18 to 45 years. The exclusion criteria were as follows: (1) comorbidity of DSM-IV axis I or other severe mental disorders; (2) organic brain or other serious medical disease; (3) drug or alcohol dependence, or substance abuse; (4) mental retardation; (5) pregnancy or lactation; and (6) treatment with immunomodulators and antioxidants. We recruited from the community 136 HC that matched gender and age of the patients. This study was approved by the Ethics Committee of Beijing Huilongguan Hospital. All participants signed a written informed consent prior to the study.
Clinical assessment
We used the Childhood Trauma Questionnaire (CTQ) to assess the participants’ childhood traumatic experiences, a validated and widely used questionnaire to evaluate childhood trauma (Bernstein et al., 2003). The CTQ consists of 28 items, each of which is rated from 1 (never) to 5 (always). The questionnaire assesses five factors: emotional abuse (EA), emotional neglect (EN), physical abuse (PA), physical neglect (PN) and sexual abuse (SA). Each factor is assessed via five items, and the score for each factor ranges from 5 to 25, with higher score indicating more severe trauma. The reliability of the CTQ has been confirmed earlier in PFES (Simpson et al., 2019).
The PANSS was used to assess clinical symptom severity. The scale consists of 30 items: 7 items for the positive subscale, 7 items for the negative subscale and 16 items for the general psychopathology subscale. Each item is operationally defined with specific scoring criteria. All participants were assessed by a trained psychiatrist along with clinical examination. The score for each item ranges from 1 (none) to 7 (extreme). All attending psychiatrists were trained for PANSS assessment before the study began, and the assessments showed an intra-class correlation coefficient (ICC) ⩾ 0.80.
Measurement of plasma mature BDNF levels
Plasma samples were obtained between 7:00 a.m. and 9:00 a.m. from HC and PFES. All participants provided 5 mL of fasting venous blood from the cubital vein. Blood samples were stored in coagulant tubes in an ice box at 4°C for 4 hours after standing for 1 hour at room temperature, after which the plasma was separated by centrifugation (5000 r/min, 10 minutes). The separated plasma was stored at −80°C. We used enzyme-linked immunosorbent assay (ELISA) to detect BDNF levels. Two measurements were obtained for each plasma sample and the average calculated. Measurement of BDNF levels was carried out in strict accordance with the ELISA kit instructions (Biosensis, USA). The clinical data were blinded while the technicians analyzed all of the samples.
Statistical analyses
For demographic data, continuous and categorical variables were compared using t-tests and chi-square tests between groups. The CTQ total and subtype scores and BDNF levels were normally distributed. We included gender and educational level as covariates in multivariate analysis of covariance (MANCOVA) to compare the differences in CTQ total and subtype score between PFES and HC. We also used one-way analysis of covariance (ANCOVA) with gender and educational level as covariates to compare BDNF levels between PFES and HC. Multiple linear stepwise regression was used to analyze the influence of subtypes of childhood trauma on BDNF levels between PFES and HC again with gender and educational level as covariates. The same method was used to compare the influence of childhood trauma subtypes on PANSS scores for PFES. Simple linear regression analysis was used to analyze the relationship between BDNF levels and PANSS score for PFES with gender and educational level as covariates. The same method was used to compare the influence of CTQ total score on BDNF levels and PANSS score. We present all data values in mean ± standard deviation (SD) and employed a p < 0.05 (two-sided) as a criterion of statistical significance. G*Power 3.1.9.2 has been used to calculate power. The specific calculation process is as follows: we included predictors according to the mediating effect equation, including 2 covariates (gender, education), 2 tested predictors (total CTQ score, BDNF level), the total number of predictors is 4; total sample size is 192; effect size f2 is 0.15; and α error probability is 0.05. We used power analysis for linear multiple regression: mixed model, R2 increases, finally the calculated power is 0.99.
According to the results of linear regression, we used SPSS’s PROCESS macro for mediation effect analysis. In the mediation model, we used emotional neglect score and CTQ total score as independent variables; PANSS total score and PANSS negative symptom score as dependent variables, and BDNF levels as a mediator. Educational level and gender were included as covariates. We selected model 4 in PROCESS (see Results), and a bootstrap method was used to test the significance of the mediating effect. The 95% confidence interval (CI) was shown for total, indirect and direct effects with 5000 bootstrap samples. If A 95% CI for indirect effect did not include a zero, value indicated the mediating effect is considered significant at the level of 0.05.
Results
Socio-demographics, clinical characteristics, CTQ score and BDNF levels in PFES and HC
Education level but not age or sex was significantly different between the PFES and HC (Table 1). Thus, educational level was used as a covariate in the analysis. t-test shows that men have higher BDNF levels than women (730.04 ± 339.19 pg/mL vs 649.91 ± 306.60 pg/mL). Thus, gender was included as a covariate in the analysis. There was no correlation between age and each of CTQ score, PANSS score and BDNF levels. The mean age at illness onset was 26.1 ± 7.3 years and the mean illness duration was 12.0 ± 15.9 months for the patients. Chlorpromazine equivalent antipsychotic dosage (CPZ) in patients was 348.28 ± 220.39 mg/d (Andreasen et al., 2010; Tachibana et al., 2016). Linear regression results showed that CPZ had no effect on psychopathological symptoms (βPositive score = 0.011, p = 0.884; βNegative score = 0.015, p = 0.796; βGeneral score = −0.076, p = 0.300; βTotal score = −0.030, p = 0.669) and BDNF levels (β = −0.108, p = 0.136) in PFES. MANCOVA showed that the subscore and total score of childhood trauma were higher in patients as compared to HC. One-way ANCOVA revealed that the BDNF levels were lower in patients as compared to HC (Table 1).
Participant demographics, CTQ and PANSS score, and BDNF levels.
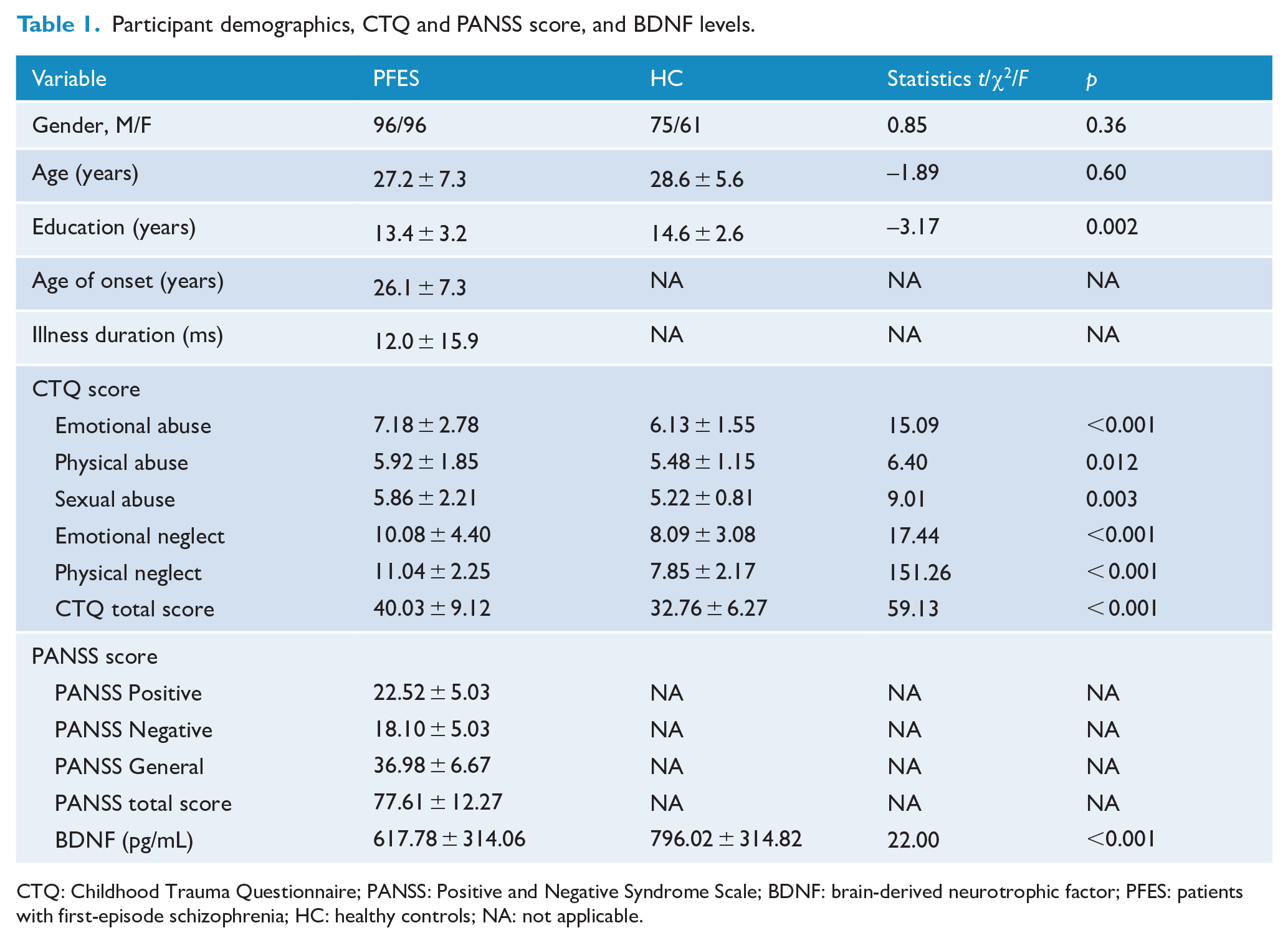
CTQ: Childhood Trauma Questionnaire; PANSS: Positive and Negative Syndrome Scale; BDNF: brain-derived neurotrophic factor; PFES: patients with first-episode schizophrenia; HC: healthy controls; NA: not applicable.
Linear regression analysis of childhood trauma and BDNF levels in PFES and HC
Linear regression analysis showed that emotional neglect and total CTQ score had a negative predictive effect on BDNF levels, with gender and educational level as covariates (βEN = −0.181, p = 0.013; βtotal = −0.177, p = 0.015), while other subtypes of CTQ had no predictive effect. The subtypes of CTQ and the total score did not significantly predict the BDNF levels for HC (Supplementary Material Tables 1 and 2).
Linear regression analysis of childhood trauma and PANSS score in patients
Multiple linear stepwise regression analysis with gender and educational level as covariates showed that emotional abuse and emotional neglect were positively correlated with PANSS positive score (βEN = 0.203, p = 0.009; βEA = 0.187, p = 0.015). Positive correlations were also found between emotional neglect, emotional abuse and PANSS total scores (βEN = 0.196, p = 0.008; βEA = 0.308, p < 0.001). A positive correlation was also shown between physical neglect and PANSS negative scores (β = 0.212, p = 0.003). There was a positive correlation between emotional neglect and PANSS general psychopathology score (β = 0.353, p < 0.001). The total childhood trauma score was correlated with PANSS positive score (β = 0.303, p < 0.001), negative score (β = 0.222, p = 0.002), general psychopathology score (β = 0.367, p < 0.001) and total score (β = 0.433, p < 0.001; Supplementary Material Tables 3 and 4).
Linear regression analysis of BDNF levels and PANSS score in patients
Simple linear regression analysis with gender, educational level and chlorpromazine equivalent antipsychotic dosage as covariates showed that BDNF levels were significantly negatively correlated with PANSS negative score (β = −0.598, p < 0.001) and total score (β = −0.303, p < 0.001; Supplementary Material Table 5).
Mediation analysis
In mediating effect models, we used CTQ measures as predictors and PANSS negative and total score as outcome measures. BDNF levels were used as the mediator in all models. We focused on the relationships among CTQ total score, BDNF levels, PANSS negative score and PANSS total score in models 1 and 2 (Figures 1 and 2). We also focused on the relationships among emotional neglect, BDNF levels and PANSS total score in model 3 (Figure 3). For models 1–3, the indirect and indirect effects were significant, suggesting partial mediation.
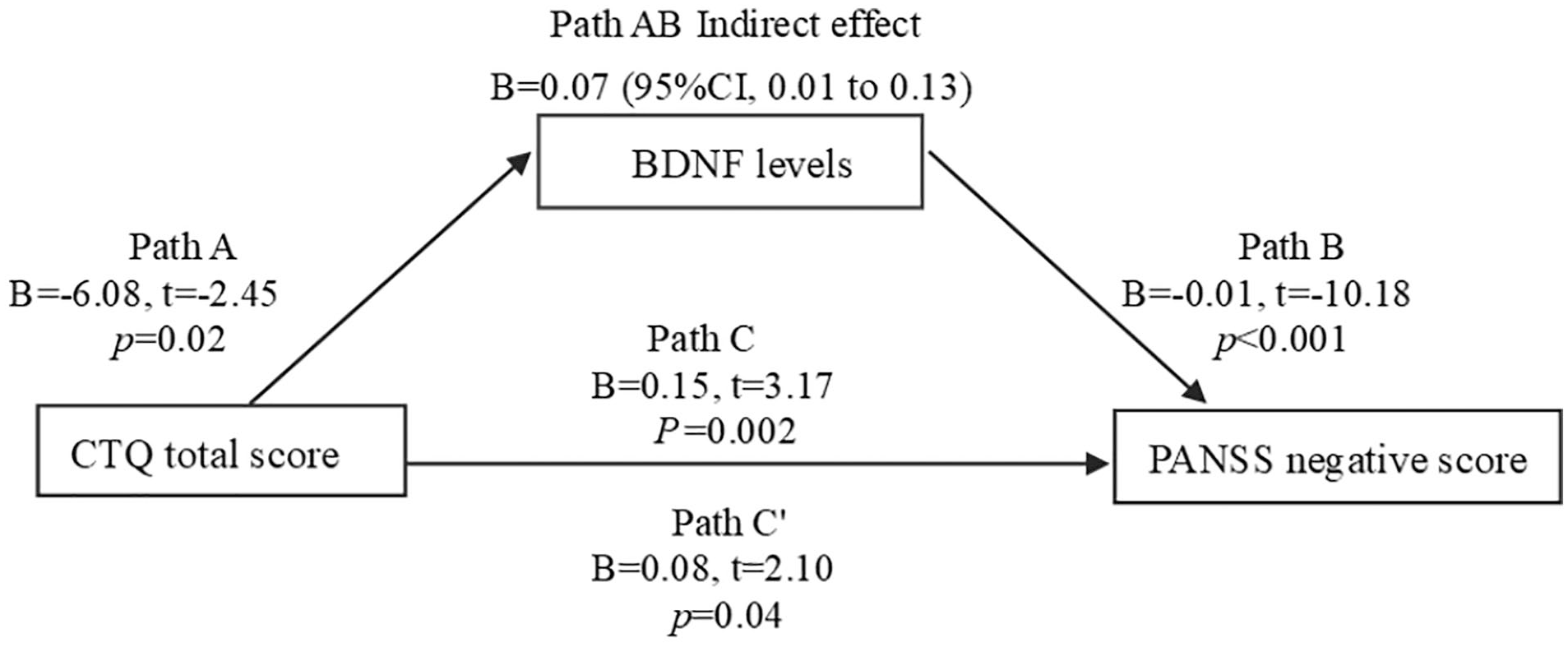
Model 1 path diagram of the mediation model (X = CTQ total score; Y = PANSS negative score; BDNF levels as mediators). Path C′ and Path C represent the direct and total effect between CTQ total score and PANSS negative score. Path AB represents the mediation effect and is significant at p < 0.05 in the model.
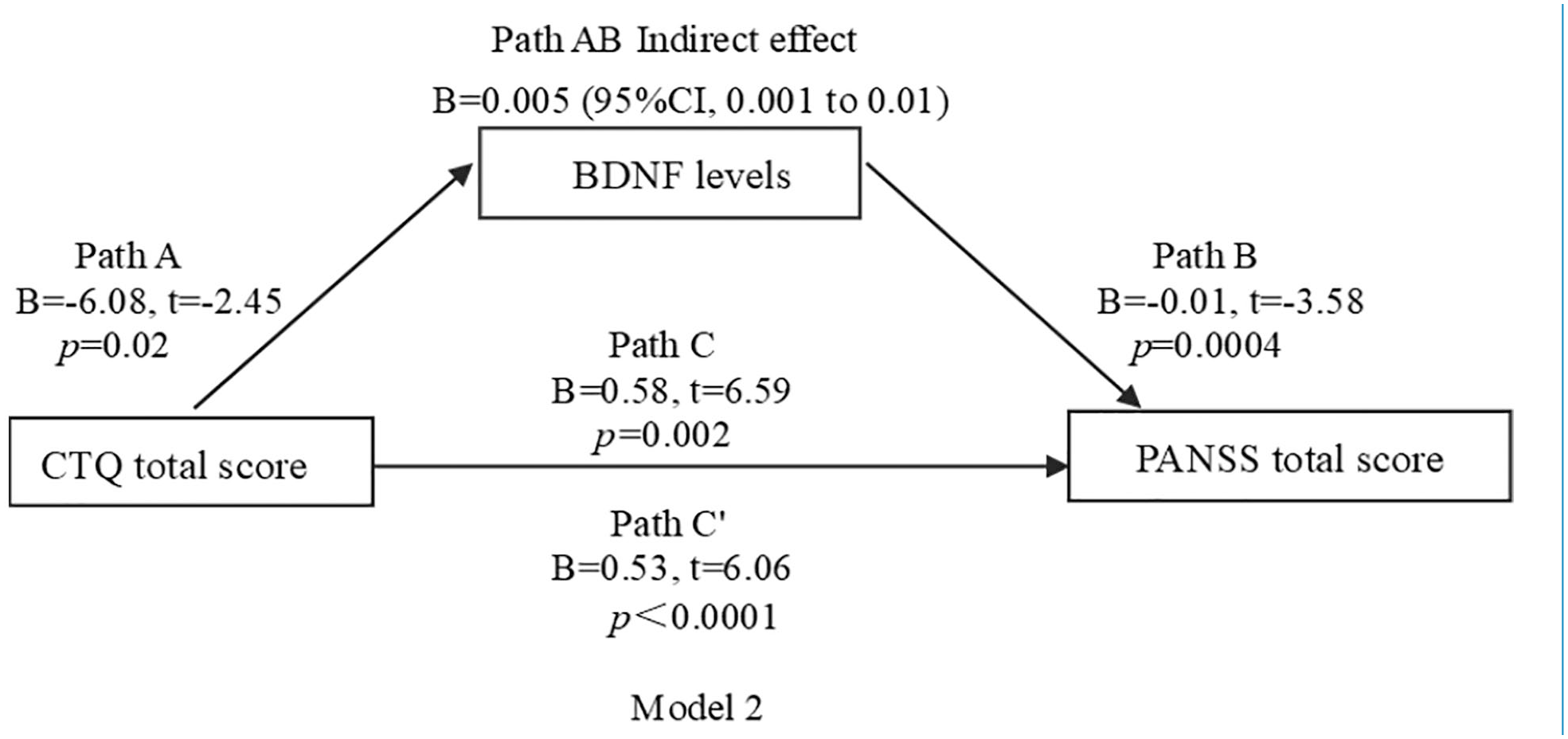
Model 2 path diagram of the mediation model (X = CTQ total score; Y = PANSS total score; BDNF levels as mediators). Path C′ and Path C represent the direct and total effect between CTQ total score and PANSS total score, respectively. Path AB represents the mediation effect and is significant at p < 0.05 in the model.
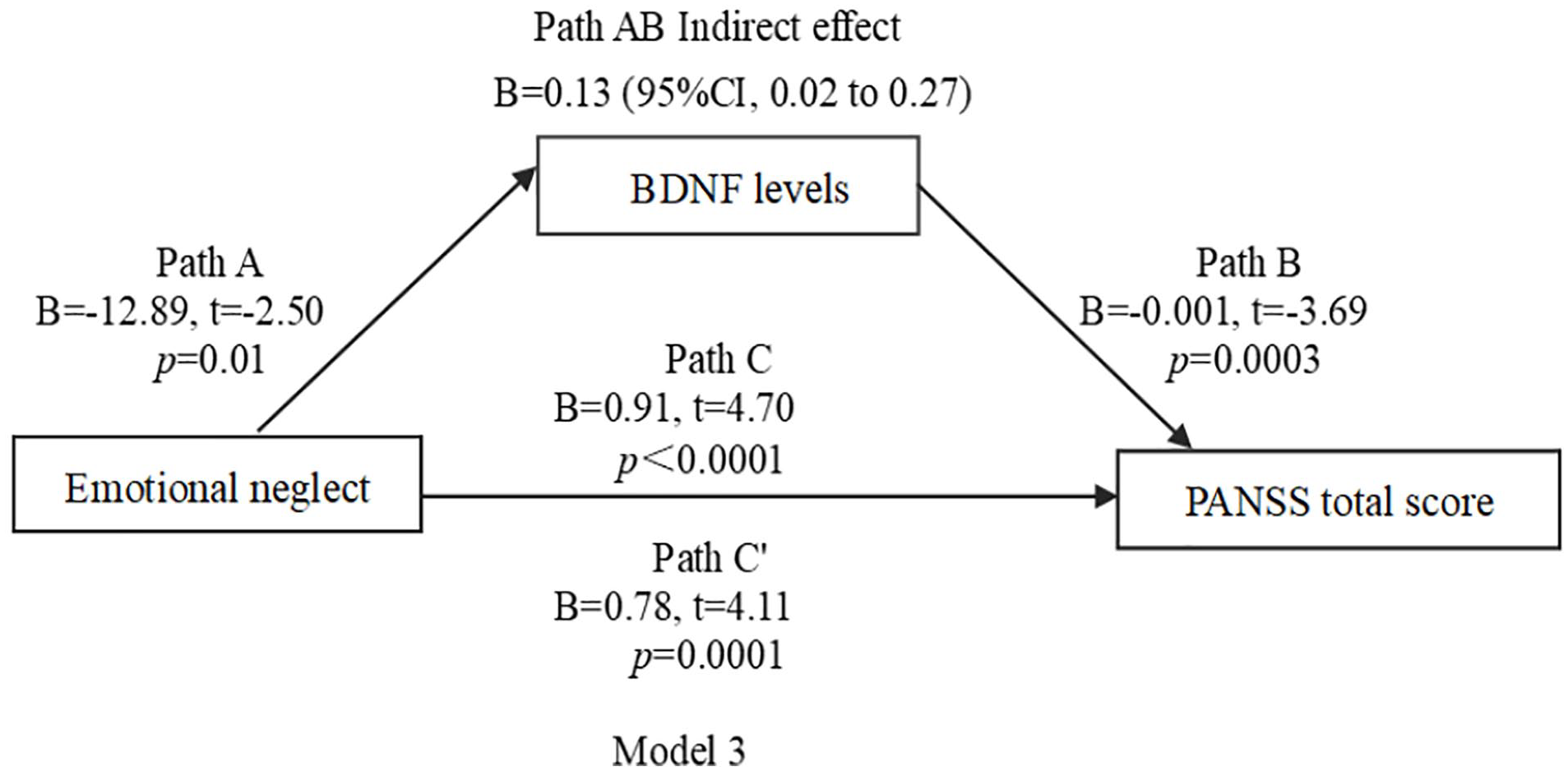
Model 3 path diagram of the mediation model (X = emotional neglect; Y = PANSS total score; BDNF levels as mediators). Path C′ and Path C represent the direct and total effect between emotional neglect and PANSS total score, respectively. Path AB represents the mediation effect and is significant at p < 0.05 in the model.
Discussion
Our study showed that each CTQ subscore and CTQ total score were higher in patients than in HC, indicating that PFES experienced more childhood trauma than did HC, in accordance with previous studies (Morgan et al., 2020). We found that emotional abuse and emotional neglect were positively associated with PANSS positive symptoms in patients, in contrast to previous reports that positive symptoms such as hallucination and delusion are primarily related to sexual abuse (Bailey et al., 2018; Hailes et al., 2019) and that PANSS positive symptoms are related to physical abuse (Kilicaslan et al., 2017). However, longitudinal studies have also shown that emotional neglect and emotional abuse in childhood are associated with later delusions and hallucination symptoms (Abajobir et al., 2017). Studies have shown that emotional neglect is related to the white matter abnormalities in cortico-limbic circuitry for PFES (Asmal et al., 2019), and white matter abnormalities are associated with PANSS positive symptoms (Kalin, 2019). These earlier findings can be fruitfully considered along with the current results that emotional abuse and emotional neglect were correlated with PANSS total scores in PFES. Emotional neglect was also positively correlated with PANSS general psychopathology in PFES. Thus, emotional abuse and emotional neglect may play a role in the development of psychiatric symptoms of schizophrenia. Emotional neglect and abuse are the most common experiences of childhood trauma in male patients with schizophrenia, which are associated with an ultra-high-risk (UHR) for psychosis (Fusar-Poli et al., 2017). In addition, we found that physical neglect was positively correlated with PANSS negative scores. Ramsay et al. showed that childhood emotional neglect is significantly associated with negative symptoms in PFES (Ramsay et al., 2011). Other studies showed that both physical and emotional neglect are correlated with negative symptoms (Struck et al., 2020). Our study also showed that the total childhood trauma score was positively associated with PANSS positive symptom, negative symptom, general psychopathology and total scores, in contrast to previous findings that was not positively correlated with PANSS negative symptom (Chae et al., 2015).
Our study showed that the BDNF levels were reduced in patients compared to HC, as with the majority of previous studies (Çakici et al., 2020). Our study also found that BDNF levels were negatively correlated with PANSS negative symptoms and total scores, which is consistent with some previous studies (Aydın et al., 2020; Rizos et al., 2008). However, other studies have shown that BDNF levels are only negatively associated with PANSS positive symptoms (Gonzalez-Pinto et al., 2010). Studies have also shown no correlation between PANSS score and BDNF levels in patients with schizophrenia (Bakirhan et al., 2017). These discrepancies remain to be investigated.
Our study was the first to report that higher emotional neglect and CTQ total scores were associated with lower BDNF levels in patients with schizophrenia. This may be related to the lower expression of BDNF mRNA caused by childhood trauma and life stress, which leads to a decrease in BDNF levels in PFES (Mondelli et al., 2011). A recent review showed that childhood trauma is associated with reduced BDNF gene expression and global DNA methylation in PFES (Tomassi and Tosato, 2017). In addition, earlier studies also showed that the increase in inflammatory factor interleukin (IL)-6 is a mediator of psychosocial stress and decreased BDNF mRNA expression (Jawahar et al., 2015; Mondelli et al., 2011). Moreover, childhood trauma, especially emotional neglect and emotional abuse, can lead to dysfunction of the hypothalamus–pituitary–adrenal (HPA) axis (Nishi, 2020). Dysfunction of the HPA axis leads to an increase in glucocorticoids and a decrease in glucocorticoid receptors in the hippocampus and other brain regions and decreased BDNF levels (Buck et al., 2019). In turn, the decrease in BDNF levels may lead to abnormal neuronal development and maturation, including apoptosis, in the hippocampus and other cortical and subcortical structures, enhancing the risk of schizophrenia (Harrisberger et al., 2015; Zugman et al., 2015).
We also demonstrated BDNF levels as a mediator between both CTQ total score and PANSS total score and negative symptoms in PFES. These findings suggest that abnormal BDNF levels, as caused by environmental factors, especially childhood trauma, may be an important risk factor of schizophrenia. That is, childhood trauma leads to a decline in BDNF levels, which in turn leads to the development of schizophrenia. In particular, decrease in BDNF levels served as an intermediary between emotional neglect and PANSS total score for PFES. Emotional neglect is a common type of childhood trauma in both schizophrenics and neurotypical Chinese populations (Fang et al., 2015; Xie et al., 2018). Many Chinese children are now raised by grandparents, and Chinese parents place more emphasis on their children’s achievement than emotional needs (Fu et al., 2018), posing a risk to emotional development.
A number of limitations need to be considered for the current work. First, this was a cross-sectional study with childhood trauma assessed retrospectively and subject to recall bias. On the contrary, data from retrospective assessment of childhood trauma in patients with psychiatric patients appeared to be reliable, with a very small recall bias as compared with prospective assessments (Scott et al., 2012). Second, BDNF levels were quantified for plasma rather than the brain, although previous animal studies have shown similar maturation and aging processes for plasma and brain BDNF (Karege et al., 2002) and a positive correlation between plasma and cerebrospinal fluid (CSF) BDNF levels in patients with schizophrenia (Pillai et al., 2010). Third, the mechanism by which BDNF reduction is caused by childhood trauma remains unclear and needs to be further explored.
Conclusion
Our study showed that PFES experienced more severe childhood trauma and reduced BDNF levels as compared to HC. Childhood trauma was positively correlated with psychiatric symptoms and BDNF levels were negatively correlated with PANSS total score and negative symptoms in PFES. BDNF levels mediated the relationship between childhood trauma and psychiatric symptoms in the patients. Childhood trauma may lead to schizophrenia through neurodevelopmental mechanisms, a critical issue to address in future work.
Supplemental Material
sj-docx-1-anp-10.1177_00048674211031478 – Supplemental material for The mediating effect of brain-derived neurotrophic factor levels on childhood trauma and psychiatric symptoms in patients with first-episode schizophrenia
Supplemental material, sj-docx-1-anp-10.1177_00048674211031478 for The mediating effect of brain-derived neurotrophic factor levels on childhood trauma and psychiatric symptoms in patients with first-episode schizophrenia by Leilei Wang, Yi Yin, Yanfang Zhou, Junchao Huang, Ping Zhang, Song Chen, Hongzhen Fan, Yimin Cui, Xingguang Luo, Shuping Tan, Zhiren Wang, Chiang-Shan R Li, Baopeng Tian, Li Tian, L Elliot Hong and Yunlong Tan in Australian & New Zealand Journal of Psychiatry
Footnotes
Author Contributions
Y.T. had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Y.T. contributed to the concept and design of the study. All authors contributed to acquisition, analysis or interpretation of data. L.W. contributed to drafting of the manuscript. Y.T., C.-S.R.L., L.E.H. and B.T. contributed to critical revision of the manuscript for important intellectual content. Y.Y., H.F., J.H. and Y.T. contributed to statistical analysis. Y.T. helped in obtaining funding. Y.T. contributed to administrative, technical or material support.
Availability of Data and Materials
The data that support the findings of this study are available from the corresponding author upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Supports were received from the National Key R & D Program of China (2016YFC1307000), National Natural Science Foundation of China (81761128021, 81771452) and the National Institute of Health (R01MH112180 and R01MH116948).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
