Abstract
Keywords
Brain-derived neurotrophic factor (BDNF) is a 27 kDa polypeptide that plays an important role in the survival, differentiation and outgrowth of peripheral and central neurons during development and adulthood [1]. BDNF is highly expressed in the hippocampus, a region that is critically involved with learning and memory. There is a good correlation between BDNF mRNA expression and behavioural performance in various learning and spatial memory tests [2]. Recent reports have implicated BDNF in the pathophysiology of a number of disparate neuropsychiatric disorders including mood disorders [3] and schizophrenia [4].
The effects of stress on neurotrophic factors were first demonstrated by Smith et al., who showed that single or repeated episode(s) of immobilization stress markedly reduced BDNF mRNA levels in the hippocampus [5]. This observation has been repeated with other models of stress, including unpredictable stress [6], foot shock [7], and early maternal deprivation [8]. The underlying mechanism may be that of hypothalamic–pituitary–adrenal (HPA) axis activation and elevated adrenal corticosteroid secretion, which is supported by the observation that exogenous corticosterone administration decreases BDNF expression in the hippocampus in an animal model [5]. However, adrenalectomy does not completely abolish BDNF downregulation, suggesting that mechanisms in addition to HPA axis activation are also influential in regulating the BDNF response to stress.
Abnormalities of the HPA axis, manifested by hypercortisolaemia, are seen in bipolar disorder [9]. Reports of HPA axis dysfunction in schizophrenia are less consistent but a recent study described HPA axis overactivity in patients with first-episode schizophrenia [10]. We have recently investigated the effects of treatment with mifepristone (RU-486), a glucocorticoid-receptor (GR) antagonist, on mood and cognitive function in depressed patients with bipolar disorder [11] and schizophrenia [12]. Seven days of adjunctive mifepristone improves mood and spatial working memory in patients with bipolar disorder, but not schizophrenia. There are no reports of studies examining the effects of treatment with GR antagonists on BDNF expression. The aims of the present study were to investigate serum BDNF levels in symptomatic bipolar disorder and schizophrenia compared with healthy comparison subjects. Second, we sought to examine the change in BDNF levels following GR antagonism in bipolar disorder and schizophrenia.
Method
Details of the mifepristone treatment protocol have been described previously [11, 12]. Briefly, depressed patients with bipolar disorder (n = 20) and schizophrenia (n = 20), diagnosed according to DSM-IV criteria, were entered into a double-blind, placebo-controlled, crossover design study. Only men and post-menopausal women were eligible to take part. Patients were excluded if they had a previous head injury, neurological condition or other serious medical condition, or current alcohol or drug abuse. Patients had been stable on prescribed medication for at least 4 weeks prior to entering the study (Appendix I), and were prescribed adjunctive mifepristone, 600 mg day−1, or placebo for 7 days. Fourteen days following treatment, subjects entered the other arm of the treatment protocol (i.e. those initially prescribed mifepristone received placebo and vice versa) for 7 days, following which there was a further 14 day follow-up period. Mood symptoms and neuropsychological performance were assessed at baseline and at 14 days following mifepristone and placebo treatment. Serum cortisol and BDNF were measured at baseline, immediately following both treatments, and at 14 days after the end of both treatments. BDNF was measured in 14 healthy age- and sex-matched control subjects with no personal or family history of psychiatric disorder. All patients gave their fully informed consent to participate in this study, which received ethical approval from the Local Research Ethics Committee.
BDNF was estimated from serum stored at −70°C using enzyme-linked immunoassay (ELISA) according to the manufacturer's instructions (Promega, Southampton, UK). Absorbencies were measured at 450 nm using an automatic ELISA microplate reader. Intra-assay coefficient of variation (CV) was 12.5% at 25.6 pg mL−1; 12.3% at 53.3 pg mL−1; and 10.9% at 286.1 pg mL−1. Cortisol was estimated using Corti-cote radioimmunoassay (ICN Pharmaceuticals, Costa Mesa, CA, USA). Intra-assay CV was <9% across the assay range.
All data are reported as means and SD unless otherwise specified. Following confirmation that data met assumptions for parametric analysis, BDNF levels were analyzed using analysis of variance (ANOVA) with diagnosis (bipolar, schizophrenic or control) as a between-subjects factor. Significant main effects were examined post-hoc using the least significant difference method. The effect of treatment on cortisol and BDNF within each patient group was examined in the same way with the inclusion of sample as a within-subject factor. Pairwise comparisons were made by t-test. Associations between cortisol and BDNF were examined by Pearson's correlations. All cited p values were two-tailed, with a significance level set at 0.05. Analyses were performed using SPSS version 11 (SPSS, Chicago, IL, USA).
Results
Demographic data are presented in Table 1. At baseline there was no significant difference between groups with regard to BDNF levels (F = 0.014, df = 2,51, p = 0.986; Table 2).
Demographic details and illness characteristics
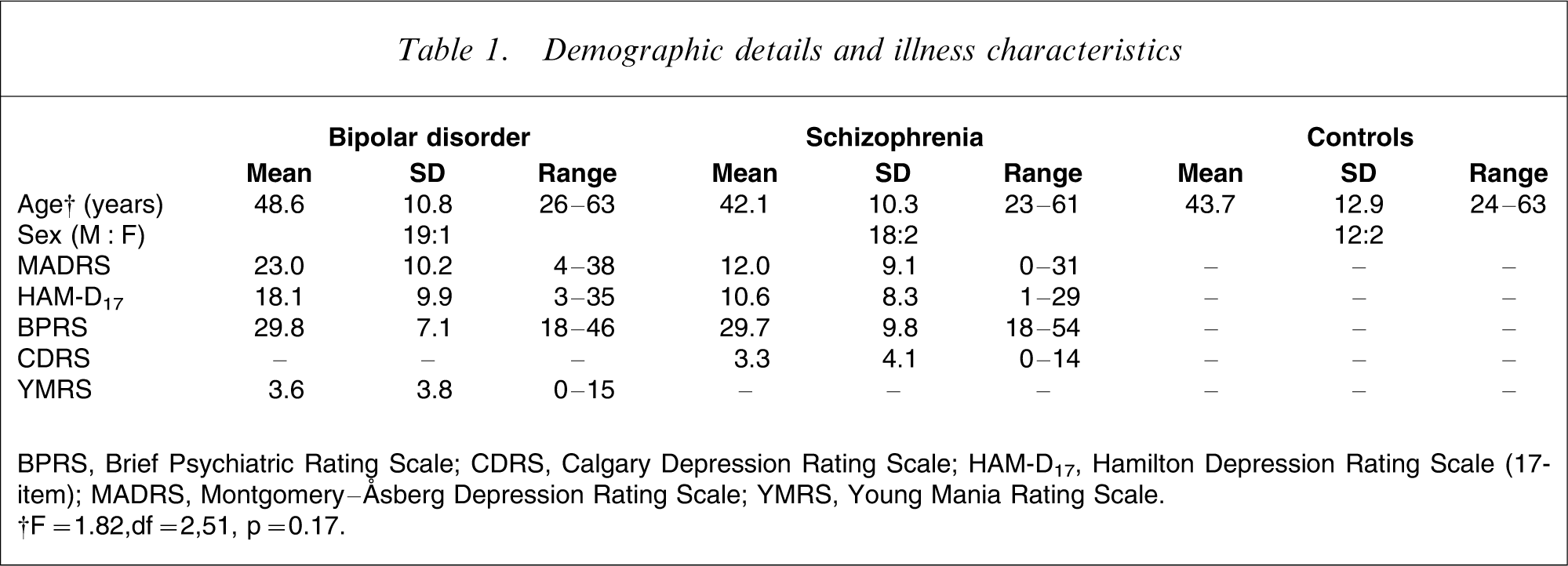
BPRS, Brief Psychiatric Rating Scale; CDRS, Calgary Depression Rating Scale; HAM-D17, Hamilton Depression Rating Scale (17-item); MADRS, Montgomery–Åsberg Depression Rating Scale; YMRS, Young Mania Rating Scale.
†F = 1.82,df = 2,51, p = 0.17.
BDNF and cortisol levels
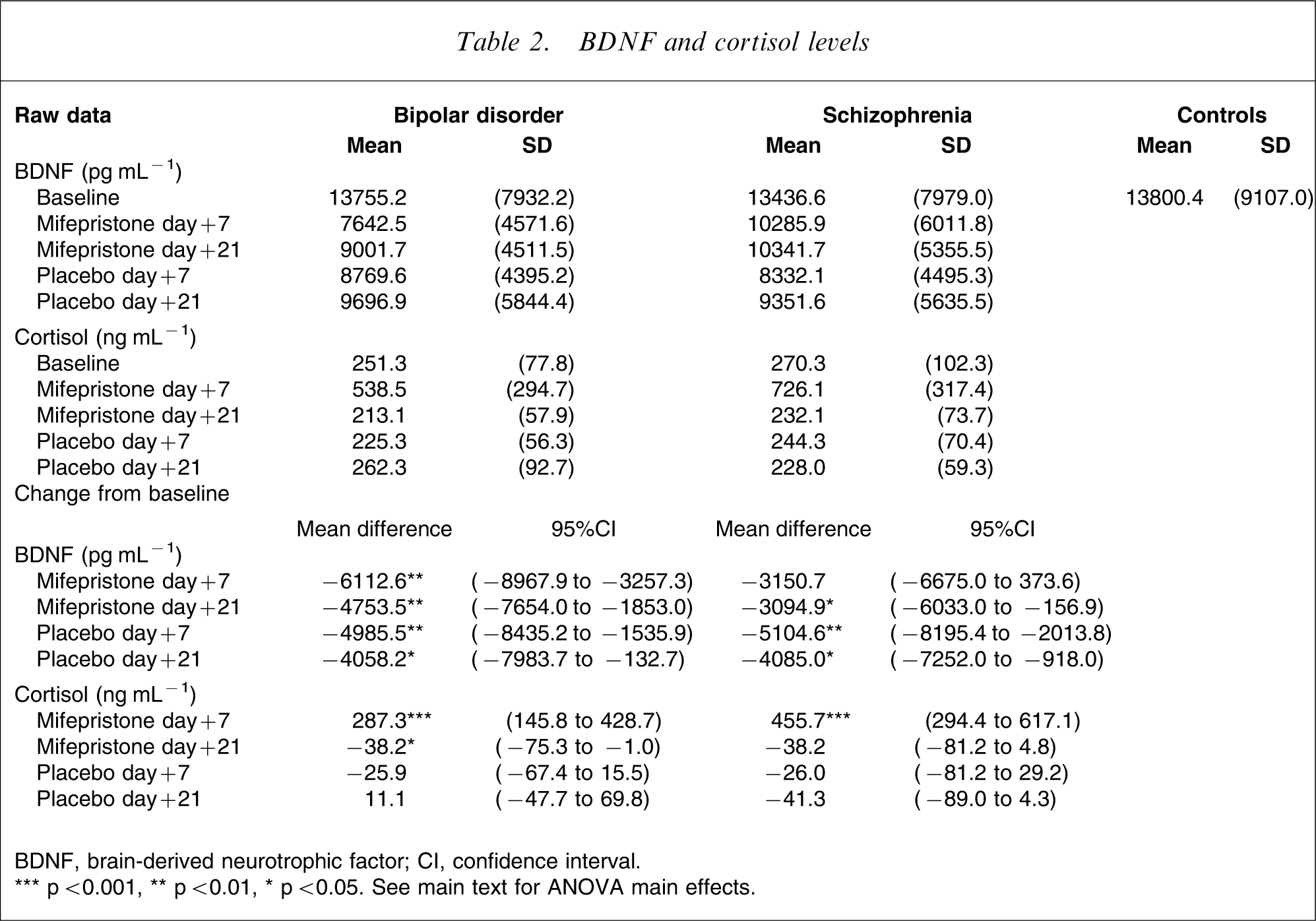
BDNF, brain-derived neurotrophic factor; CI, confidence interval.
∗∗∗ p < 0.001, ∗∗ p < 0.01, ∗ p < 0.05. See main text for ANOVA main effects.
From the analysis of treatment effects, there was no significant main effect of diagnosis on BDNF (F = 0.152, df = 1,35, p = 0.699), but a main effect of sample was observed (F = 8.964, df = 4,140, p < 0.001). Analysis of simple main effects revealed that BDNF levels at baseline were significantly higher than at all other time points, irrespective of treatment with mifepristone or placebo. There was no significant diagnosis by sample interaction (F = 1.028, df = 4,140, p = 0.386).
In the analysis of cortisol levels, there was no significant main effect of diagnosis (F = 2.318, df = 1,35, p = 0.137), but a main effect of sample was observed (F = 56.279, df = 4,140, p < 0.0001). Analysis of simple main effects showed that cortisol levels after 7 days of mifepristone treatment were significantly higher that at all other time points. Cortisol levels 14 days after cessation of mifepristone treatment were significantly lower than at baseline (p < 0.05). There was no significant diagnosis–sample interaction (F = 3.260, df = 4,140, p = 0.062).
The relationship between the cortisol and BDNF response to treatment was examined at each time point (post-treatment levels minus baseline). After 7 days of mifepristone treatment, there was a significant negative correlation between the cortisol and BDNF responses to mifepristone in schizophrenia patients (r = − 0.602, R2=0.362, p = 0.006) but no significant relationship at the same point in bipolar patients (r = 0.104, R2=0.011, p = 0.670; Figure 1). No other correlations at any time point were significant, nor were any following placebo.
Scatter plots of the cortisol (CORT) and brain-derived neurotrophic factor (BDNF) response to mifepristone at the point of cessation of treatment. (a) Schizophrenia; (b) bipolar disorder.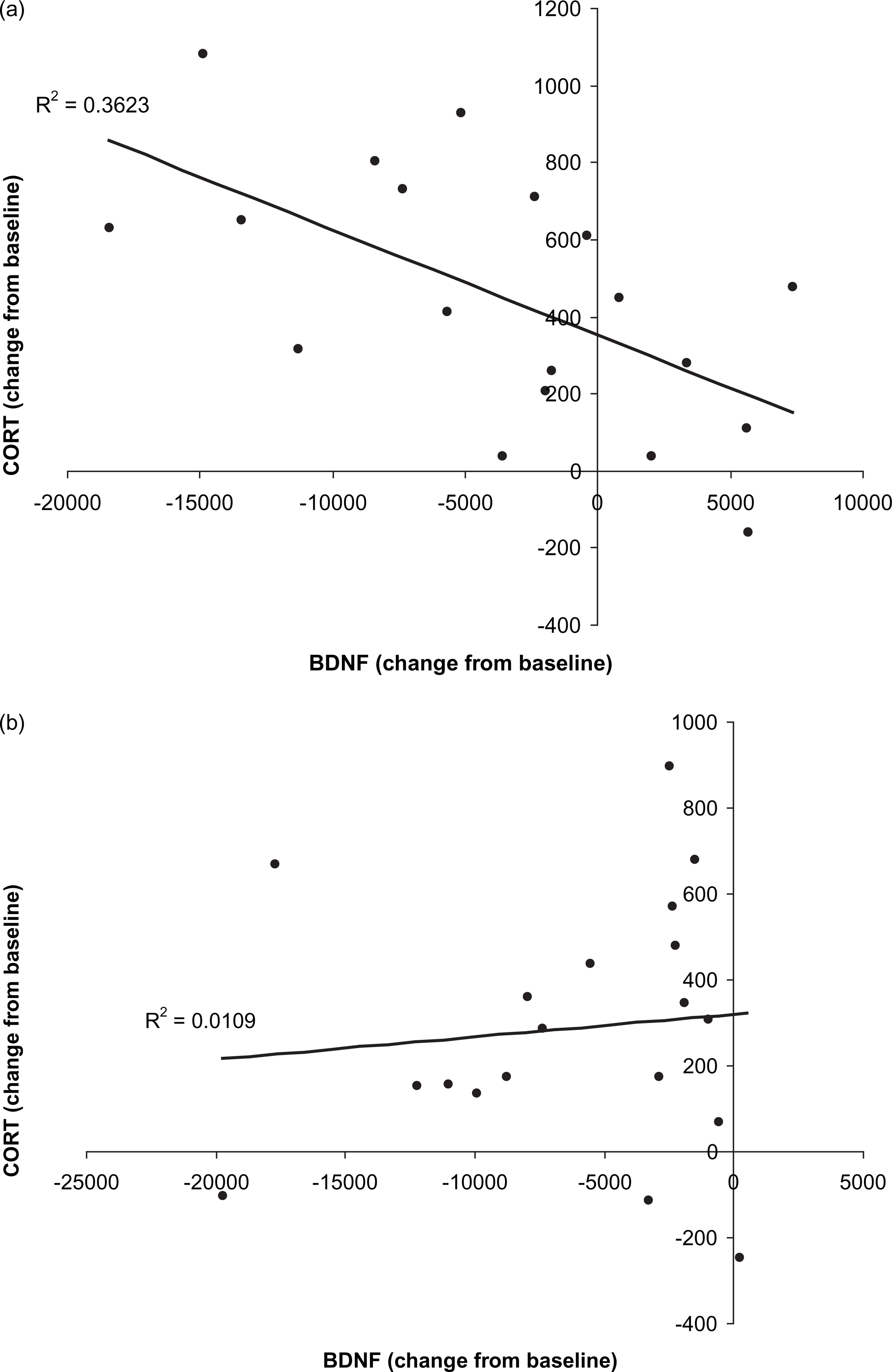
Discussion
This is the first study to examine the effects of antiglucocorticoid treatment on BDNF levels in humans. We found no differences in baseline levels of serum BDNF between depressed patients with bipolar disorder, schizophrenia and healthy controls. Several studies have reported that BDNF levels are decreased in drug-free and drug-naïve patients with major depressive disorder compared with healthy controls, and that they return to normal following successful treatment with antidepressant medication [13, 14]. In bipolar disorder, it has been demonstrated that BDNF levels are significantly decreased in manic and depressed patients compared to euthymic patients and controls [15]. This finding is supported by another recent report that found that BDNF levels in patients with bipolar disorder or schizophrenia were significantly decreased following the onset of symptoms, but in all instances then progressively increased towards control values during 1 year follow up subsequent to the first episode [16]. Other studies of serum BDNF levels in patients with schizophrenia are conflicting: one study has reported that BDNF levels are indistinguishable from healthy controls [17], but another study has reported decreased levels in medicated patients with chronic schizophrenia [18]. The effects of psychotropic drugs on BDNF expression are not well understood, and medication cannot be excluded as a confounding variable.
In the present study, at the cessation of treatment with mifepristone, cortisol levels were significantly increased, compared with baseline, in both patient groups. This effect results from blockade of the GR, and loss of the normal GR-mediated negative feedback on cortisol secretion. As predicted from pre-clinical studies that have investigated the effects of stress and exogenous corticosteroid administration on BDNF expression, hypercortisolaemia was associated with a reduction of BDNF levels. However, a reduction in peripheral BDNF was also observed following treatment with placebo in the absence of elevated cortisol levels. This phenomenon is not easily explained and requires further investigation. A large multicentre, parallel-group, placebo-controlled trial of mifepristone in bipolar disorder in currently underway, and the results of that study may provide further insights.
In patients with schizophrenia we observed a significant negative correlation between change in cortisol level and change in BDNF expression. This association was not present in patients with bipolar disorder. Bipolar disorder, unlike schizophrenia, is characterized by chronic HPA axis dysfunction [9], and the absence of a correlation between increased cortisol levels and decreased BDNF levels may reflect underlying pathophysiological differences between these two disorders, and longstanding GR dysregulation in bipolar disorder.
The mechanism(s) by which serum BDNF levels change following treatment with mifepristone are not known. The different response to hypercortisolaemia in bipolar disorder and schizophrenia suggests that the GR may have an important regulatory function, although effects of mifepristone that are independent of changes in cortisol or GR function cannot be excluded.
Although several studies have reported peripheral BDNF levels in a variety of neuropsychiatric disorders, the relevance of such measures to central BDNF expression is unclear. BDNF is more highly concentrated in brain tissue than blood [19], and it readily crosses the blood–brain barrier. BDNF levels in the brain and serum of rats undergo similar changes during maturation and ageing [20], suggesting that serum BDNF levels may reflect BDNF levels in the brain. Further investigation of the relationship between central and peripheral BDNF expression is required in order to clarify the relevance of serum BDNF measurement in human health and disease.
These preliminary results require replication in a larger sample size. In addition, pre-clinical studies and studies of serum BDNF in healthy subjects following antiglucocorticoid treatment are needed to investigate further the relationship between HPA axis function and BDNF expression, and the potential clinical significance of this association.
Footnotes
Acknowledgements
PM is the recipient of a National Alliance for Research in Schizophrenia and Depression (NARSAD) Young Investigator Award which provided financial support for this study. We thank the Stanley Medical Research Institute for their generous support of this study.
Appendix
Appendix 1. Medication details of bipolar and schizophrenia patients
| Bipolar patients (n) | Schizophrenia patients (n) | |
| Mood stabilizers† | ||
| Carbamazepine | 3 | 2 |
| Gabapentin | 3 | – |
| Lamotrigine | 6 | – |
| Lithium | 11 | – |
| Valproate | 5 | – |
| Antipsychotics‡ | ||
| Amisulpride | – | 1 |
| Clozapine | – | 3 |
| Fluphenazine | – | 1 |
| Haloperidol | – | 2 |
| Olanzapine | 3 | 7 |
| Quetiapine | 4 | 1 |
| Risperidone | 3 | 4 |
| Sulpiride | 1 | 2 |
| Zuclopenthixol | – | 1 |
| Antidepressants§ | ||
| Amitriptyline | 1 | – |
| Citalopram | 1 | – |
| Fluoxetine | – | 4 |
| Lofepramine | – | 1 |
| Mirtazapine | 5 | – |
| Paroxetine | 2 | – |
| Reboxetine | – | 1 |
| Sertraline | 1 | – |
| Trazodone | 1 | – |
| Venlafaxine | 6 | 1 |

†Nine bipolar patients on combination therapies: lithium + lamotrigine, n = 5; valproate + lamotrigine, n = 1; lithium + carbamazepine + gabapentin, n = 1; carbamazepine + gabapentin, n = 1; and lithium + valproate + carbamazepine, n = 1.
‡Two schizophrenia patients on combination therapies: olanzapine + haloperidol, n = 1; and risperidone + fluphenazine, n = 1.
§Four bipolar patients on combination therapies: venlafaxine + mirtazapine, n = 3; and citalopram + mirtazapine, n = 1.
