Abstract
Objective:
Cognitive impairment is prevalent in schizophrenia. Macrophage migration inhibitory factor which is released into the circulation under stress or inflammation, is associated with cognition and also plays an important role in immunity. However, no study has investigated the relationship between macrophage migration inhibitory factor and cognitive function in first-episode schizophrenia patients at baseline or after treatment. This study investigated the pre- and post-risperidone treatment correlations between serum macrophage migration inhibitory factor levels and cognitive function in first-episode schizophrenia patients.
Methods:
A total of 83 first-episode schizophrenia patients who received risperidone monotherapy and 57 healthy controls – matched for sex, age, smoking status, education (years), marital status and waist-to-hip ratio – were included. Macrophage migration inhibitory factor levels were measured before and 10 weeks after treatment in the patient group and at baseline in the controls. Pre- and post-treatment cognitive functions in patients were assessed using the MATRICS Consensus Cognitive Battery.
Results:
At baseline, macrophage migration inhibitory factor levels were significantly higher in first-episode schizophrenia patients than those in healthy controls (p < 0.01) and decreased in patients after 10 weeks of risperidone treatment compared with baseline (p < 0.05). The MATRICS Consensus Cognitive Battery total score and the sub-scores for the Trail Making Test, Symbol Coding, Letter Number Sequence, Maze and Brief Visuospatial Memory Test–Revised improved significantly after risperidone treatment. After controlling for age, sex, education, waist-to-hip ratio and smoking status, partial correlation analysis showed a positive correlation between baseline macrophage migration inhibitory factor levels and patients’ baseline MATRICS Consensus Cognitive Battery verbal memory scores (r = 0.29, p = 0.01). Macrophage migration inhibitory factor changes correlated negatively with verbal memory changes (r = −0.26, p = 0.04). Multiple linear regression analysis identified a definite correlation between the changes in word memory test score and macrophage migration inhibitory factor level (β = −0.09, p = 0.04).
Conclusion:
Macrophage migration inhibitory factor may be involved in the process of cognitive impairment in first-episode schizophrenia and repair mechanisms following risperidone treatment.
Introduction
Schizophrenia is a chronic and highly disabling psychiatric disorder with a lifetime prevalence of approximately 1% worldwide (Chang et al., 2017). It is clinically characterized by neurocognitive impairment and affects many aspects of memory, attention, executive function and information processing (Condray and Yao, 2011). Current studies confirm that immune function imbalances can lead to mental illnesses (Ganguli et al., 1993). Studies examining the perinatal period have reported that prenatal infections and immunological changes in pregnant women are associated with schizophrenia and foetal brain changes that can persist until adulthood (Blomström et al., 2016). In one study concerning the effectiveness of the immunosuppressant methotrexate in the treatment of schizophrenia, the results suggested that patients with schizophrenia were affected by activation of the immune system and inflammatory syndromes (Chaudhry et al., 2015). This may be explained by the following mechanism: when the body is subjected to traumatic injury or infection, the immune response causes inflammation (Anderson and Maes, 2013), resulting in a state of stress. Stress hormones reach the hypothalamus via the brainstem reticular formation, stimulate the hypothalamic–pituitary–adrenal (HPA) axis and promote the release and secretion of adrenocorticotropic hormone. This process increases the level of cortisol but also activates the body’s autoimmune responses, promoting the release of cytokines (Dantzer et al., 2008). These pro-inflammatory cytokines released by peripheral blood cells perturb the junction formation of vascular endothelial cells, resulting in increased blood–brain barrier permeability (Savitz and Harrison, 2018). The consecutive entry of inflammatory cells into the central nervous system, through the blood–brain barrier, activates inflammatory cascades and affects the brain, eventually resulting in cognitive function changes in affected patients.
The pathophysiological mechanisms in schizophrenia are strongly linked to neural, immunological and endocrine systems (Ganguli et al., 1993). Moreover, there is evidence that macrophage migration inhibitory factor (MIF) plays an important role in immunity and inflammation. MIF was first discovered in 1966 (Bloom and Bennett, 1966) and is a 12-kDa peptide monomer consisting of 114 amino acids. It is mainly present in the anterior pituitary gland, liver, kidney and brain, among other tissues (Nishibori et al., 1996). MIF is considered to be an important part of the endocrine system and HPA axis (Bernhagen et al., 1993) and is constitutively expressed by the anterior pituitary gland and released into the circulation under stress or inflammation. It is known to be a chemoattractant and has a wide range of physiological functions, including cytokine activity and receptor binding. It also prevents macrophages from pro-inflammatory migration out of capillary lymphokines. Its gene expression is exceptionally high in neurons within the cortex, hypothalamus, hippocampus, cerebellum and pons (Bacher et al., 1998). MIF has multiple effects as an inflammatory mediator (Leng et al., 2003). It can rapidly activate the extracellular signal-regulated kinase 1/extracellular signal-regulated kinase 2/mitogen-activated protein kinase (ERK1-ERK2-MAPK) pathway through sustained phosphorylation (Su et al., 2017). The chemokine receptors CXCR2 and CXCR4 effectively recognise MIF (Sinitski et al., 2020), and both receptors form complexes with CD74 to regulate biological behaviours, such as inflammatory cell recruitment (Schwartz et al., 2009). This process modifies the body’s response to infection and stress by activating innate and adaptive immune pathways (Fallica et al., 2014). MIF plays a central role in the pathogenesis of infectious diseases such as septic shock, tumours and also in metabolic diseases such as diabetes, atherosclerosis and rheumatic diseases; therefore, MIF is regarded as a therapeutic target in various inflammatory and autoimmune diseases (Sinitski et al., 2020).
Some pathological and physiological experiments have confirmed that MIF may be involved in the development of schizophrenia. For example, the MIF gene is located on chromosome 22q11.2 (Shishido et al., 2014) and the risk of mental illness and behavioural problems is high in patients with 22q11.2 deletion syndrome (Burnside, 2015). A 2017 meta-analysis of 41 studies showed that, compared with control participants, patients with schizophrenia had significantly increased microglial density (van Kesteren et al., 2017). In rodent microglia, MIF/CD74 interaction can induce cyclooxygenase-2 (COX-2) expression and upregulate prostaglandin E2 secretion (Wang et al., 2011). MIF also has a role in pathological cognitive impairment in Alzheimer’s disease, which is mediated by increased concentrations of inflammatory mediators, such as tumour necrosis factor-α (TNF-α), interleukin -6(IL-6) and interferon -γ(IFN-γ), in the cerebrospinal fluid (Popp et al., 2009). A previous study showed that MIF is involved in defence mechanisms in cognitive decline (Zhang et al., 2019). It has been suggested that MIF inhibition may delay neurodegeneration and improve cognitive function (Nasiri et al., 2020). Therefore, while MIF has been implicated in cognitive impairment, the association between MIF and cognitive impairment, as well as in activated repair mechanisms, specifically in schizophrenia, remains to be investigated. Moreover, to date, no study has identified whether MIF is involved in altered neurocognitive function following changes in schizophrenia treatment.
We hypothesized that MIF might be involved in the process of cognitive impairment and repair in patients with first-episode schizophrenia (FES-Sz). Our hypotheses were that these patients had increased MIF levels and impaired cognitive function. Furthermore, changes in cognitive function after schizophrenia treatment may be related to altered MIF levels. This study may help evaluate cognitive changes at different stages of schizophrenia and provide a better understanding of how MIF influences cognitive function, possibly leading to interventions at an early stage.
In our study, we included 83 patients with FES-Sz who had received 10-week risperidone monotherapy. We followed these patients to investigate the relationship between MIF levels and neurocognition. Peripheral inflammatory factors enter the central nervous system through the blood–brain barrier; therefore, changes in immune functions in the central nervous system can be assessed by measuring the serum or plasma cytokine levels (Hsuchou et al., 2012). We, therefore, investigated (1) whether there was a difference in MIF levels between patients with FES-Sz and healthy controls, (2) whether there was a correlation between MIF levels in patients with FES-Sz and their cognitive function and (3) whether there was a correlation between MIF levels and changes in cognitive function after patients with FES-Sz had been treated for 10 weeks with risperidone.
Methods
Study population
We enrolled patients with FES-Sz who had been admitted to our hospital between 2018 and 2019. The inclusion criteria comprised the following: (1) patients of Han Chinese ethnicity; (2) patients aged 18–45 years; (3) patients diagnosed with schizophrenia by two independent and experienced clinical psychiatrists, based on diagnostic criteria in the Diagnostic and Statistical Manual of Mental Disorders(4th Edition; DSM-IV); and (4) patients with an unmedicated first episode of schizophrenia defined as a disease duration ⩽60 months and with either no previous use of antipsychotics or usage of <14 days (Lieberman et al., 2003). The exclusion criteria comprised the following: (1) patients with comorbidities, including mental retardation, or those with past or present alcohol or other psychoactive substance abuse or dependence; (2) patients with allergic or autoimmune diseases; (3) patients who were pregnant or lactating; (4) patients with a history of diabetes mellitus or abnormal glycolipids, using lipid-lowering drug use, or following a low-sugar or low-fat diet; (5) patients with prior use of immunosuppressive agents; and (6) patients with other major physical or infectious diseases. We recruited a healthy control group through advertisements and information, and this group comprised residents of Changping District, Beijing. Previous studies have shown that people with schizophrenia generally have limited years of education; therefore, we recruited individuals with low education levels and matched them with our patient group. For all participants in this study, we collected their complete physical history, including routine physical examination results, laboratory test results (blood and urine analyses) and imaging findings to exclude those with a history of persistent infection, allergy or autoimmune diseases. None of the controls enrolled in our study received immunosuppressive drug therapy or took anti-inflammatory medication.
Informed consent was obtained from all participants, and the study was approved by the Institutional Ethical Review Board.
Cognitive assessments
Cognitive function was assessed using the MATRICS Consensus Cognitive Battery (MCCB), which comprises a total of 10 subscales, namely, the Trail Making Test (TMT), Symbol Coding (SC), Hopkins Verbal Learning Test–Revised (HVLT-R), WMS-III-Spatial Span (SS), Letter Number Sequence (LNS), Neuropsychological Assessment Battery-Mazes (NAB-MAZES), Brief Visuospatial Memory Test–Revised (BVMT-R), Category Fluency (CF), Mayer-Salovey-Caruso Emotional Intelligence Test-Managing Emotion (MSCEIT-ME) and the Continuous Performance Test (CPT). Before study initiation, two psychiatrists simultaneously attended a consistency training session and maintained an intraclass correlation coefficient of >0.85 after training. Using the MCCB, the cognitive function of all study participants was also assessed on the days of blood sampling.
Determination of MIF concentrations
Prior to blood sampling, the participants fasted overnight. Blood samples were obtained from patients with FES-Sz at baseline and after 10 weeks, whereas blood samples of healthy controls only were obtained only at baseline. The blood samples were collected with a coagulation tube at approximately 07:00 hours and coagulated at room temperature. Within 4 hours, the samples were centrifuged (3000 r/min, 10 minutes) to separate the serum, which was stored in a refrigerator at −80°C for later use. MIF was detected using an enzyme-linked immunosorbent assay (Andygene ELISA kit). All samples were assessed by the same technician, who was blinded to the clinical status of the participants. The sensitivity of the enzyme-linked immunosorbent assay was 8 pg/mL. The coefficients of variation within and between batches were 5% and 7%, respectively. The determination process was undertaken in strict accordance with the relevant instructions.
Statistical analysis
SPSS (IBM SPSS Statistics for Windows, Version 22.0) was used for all statistical analyses. Measurement data, conforming to the normal distribution, are expressed as mean ± standard deviation, and demographic data were compared using independent sample t or χ2 tests. Data not conforming to normal distribution are expressed as median (P25, P75). MIF levels between the two groups were compared using an independent sample t test. Pre- and post-treatment MIF levels in the patient group were compared using a paired t test. A partial correlation analysis was used for the correlation analysis between MIF level and cognitive function. In multiple regression analysis, the change in verbal memory scores was used as the dependent variable, and the change in serum MIF levels, sex, age, waist-to-hip ratio, smoking status and years of education was considered as independent variables to explore possible associations with changes in verbal memory score. The test was performed at an α level of 0.05.
Results
Table 1 summarizes the participants’ demographic characteristics. In this study, we enrolled 83 patients with FES-Sz (who received risperidone alone for 10 weeks) and 57 healthy controls. Among the 83 patients, 14 were excluded due to treatment changes or early patient discharge. There was no significant difference in age, sex, years of education and the waist-to-hip ratio between patients who dropped out and those who were followed up or between the patient and healthy control groups (p > 0.05) (Table 1).
Demographic and clinical characteristics of patients with schizophrenia and healthy controls.
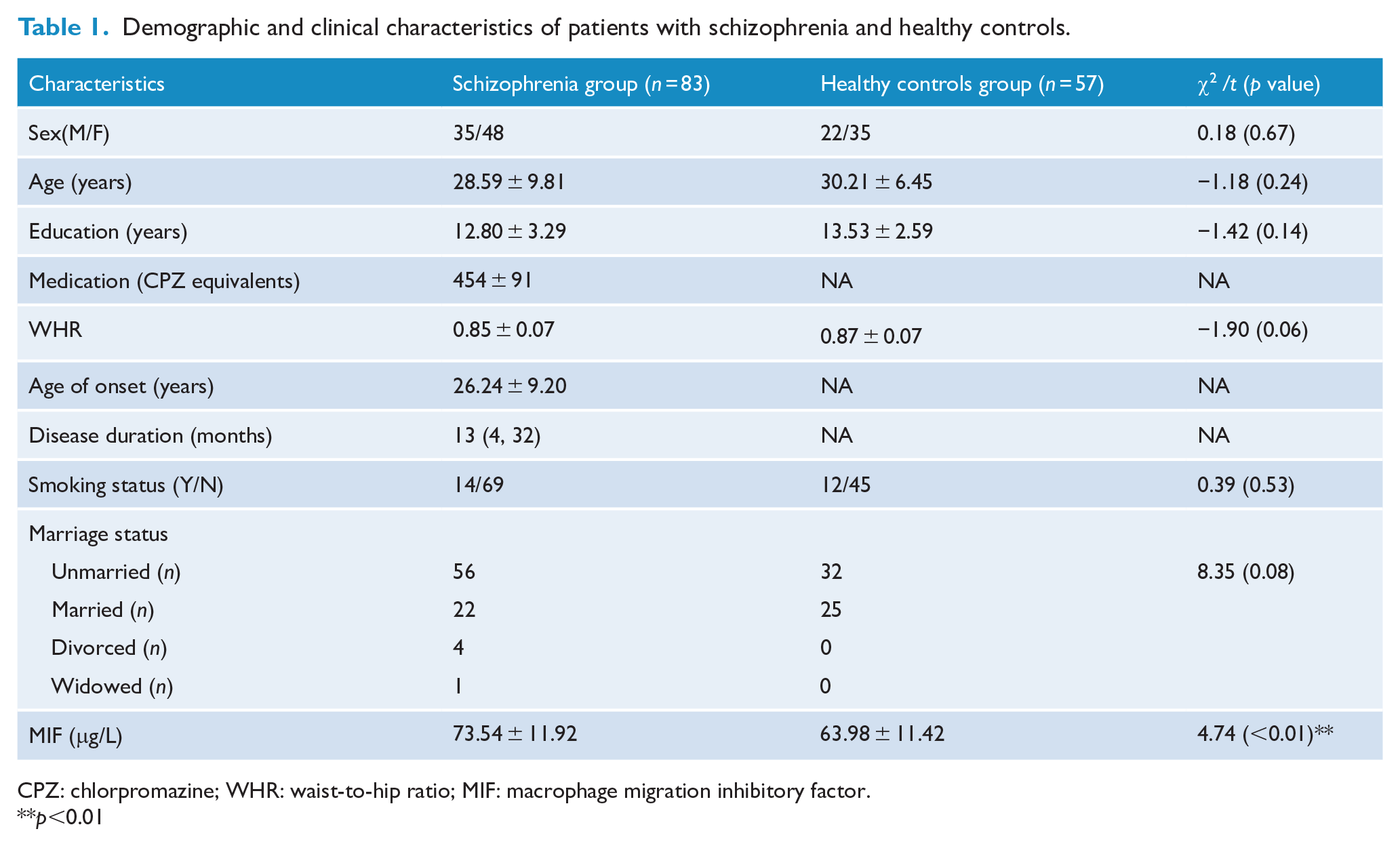
CPZ: chlorpromazine; WHR: waist-to-hip ratio; MIF: macrophage migration inhibitory factor.**p<0.01
Comparison of serum MIF concentration between patients with FES-Sz and healthy controls
The serum MIF concentration at baseline was significantly higher in patients with FES-Sz than in the healthy controls (t = 4.74, p < 0.01) (Figure 1).
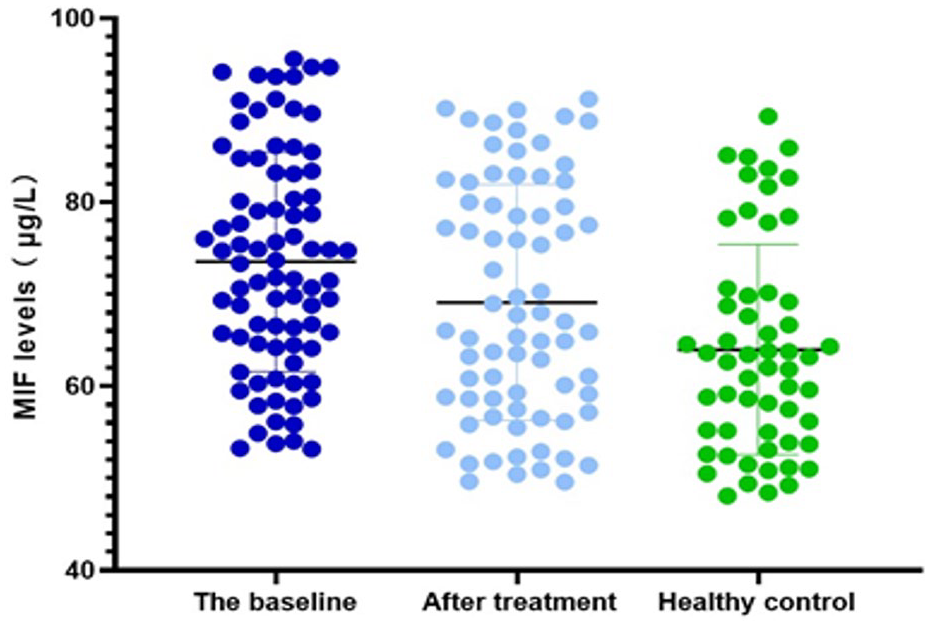
MIF levels in the schizophrenia group at baseline and after treatment and that in the control group.
Changes in pre- and post-treatment MIF levels and cognitive function in patients with FES-Sz
A paired t test was used to compare pre- and post-treatment serum MIF and cognitive function changes. MIF levels were significantly reduced after FES-Sz treatment relative to the baseline value (t = 2.24, p = 0.02). The MCCB total score (t = −2.61, p = 0.01) and the sub-test scores for TMT (t = −2.77, p < 0.01), SC (t = −2.12, p = 0.04), LNS (t = −2.87, p = 0.01), NAB-MAZES (t = −2.49, p = 0.02) and BVMT-R (t = −3.29, p < 0.01) significantly increased after risperidone treatment compared with the baseline scores in patients with FES-Sz (Table 2).
Changes in MIF levels and neurocognitive function before and after risperidone treatment.
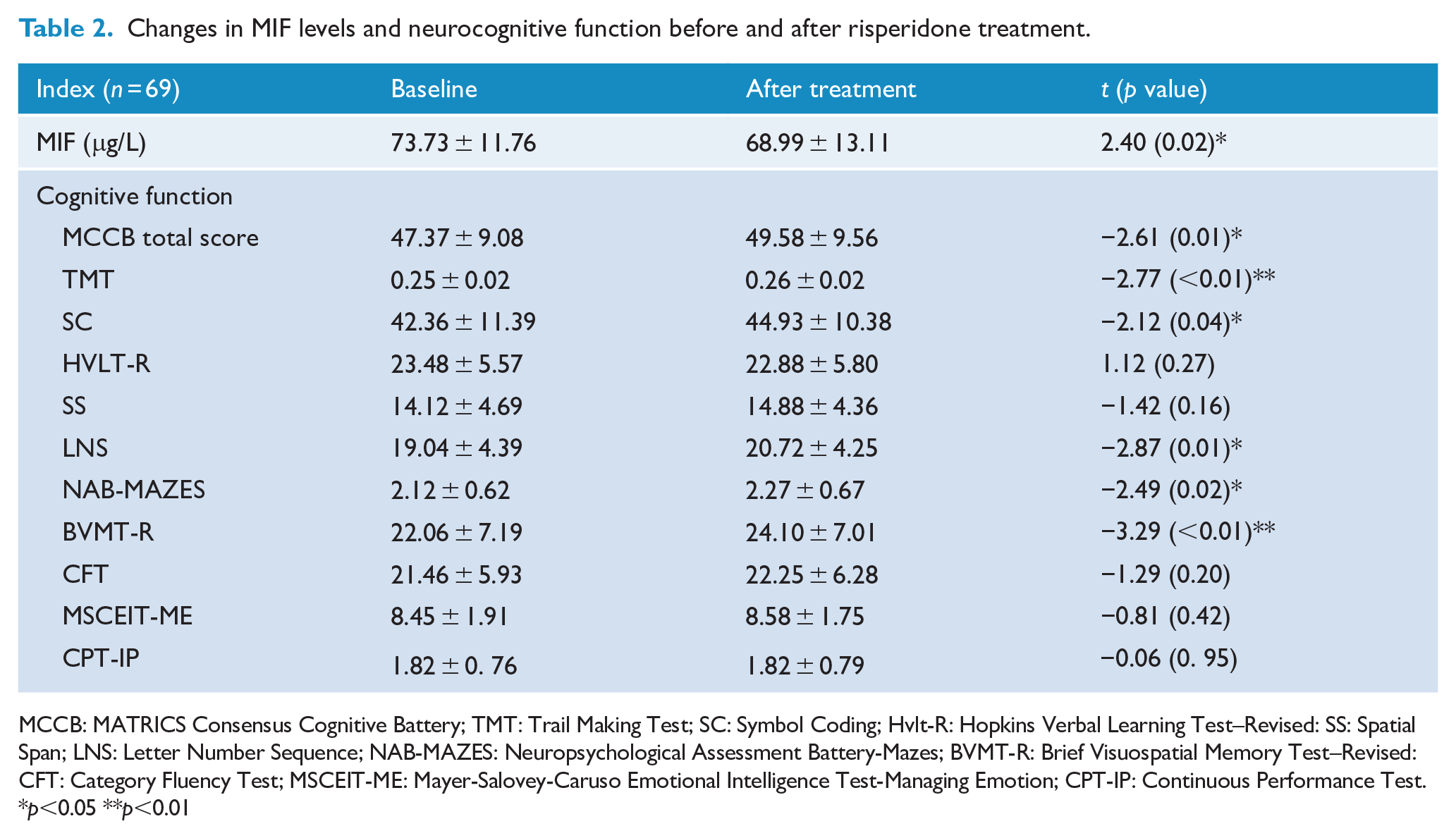
MCCB: MATRICS Consensus Cognitive Battery; TMT: Trail Making Test; SC: Symbol Coding; Hvlt-R: Hopkins Verbal Learning Test–Revised: SS: Spatial Span; LNS: Letter Number Sequence; NAB-MAZES: Neuropsychological Assessment Battery-Mazes; BVMT-R: Brief Visuospatial Memory Test–Revised: CFT: Category Fluency Test; MSCEIT-ME: Mayer-Salovey-Caruso Emotional Intelligence Test-Managing Emotion; CPT-IP: Continuous Performance Test.
p<0.05 **p<0.01
Correlation between MIF levels and cognitive function in patients with FES-Sz
At baseline, serum MIF level correlated with the MCCB scores. The change in serum MIF values correlated with the change in the MCCB scores. When controlling for age, sex, waist-to-hip ratio, smoking status and years of education as covariates, partial correlation analysis indicated a positive correlation between MIF levels at baseline and MCCB HVLT-R scores in the patient group (r = 0.29, p = 0.01). In contrast, MIF change values after risperidone treatment correlated negatively with the change in MCCB HVLT-R scores (r = −0.26, p = 0.04) (Figures 2 and 3). We did not find a correlation between serum MIF level and MCCB total score and other subscale scores (Tables 3 and 4). We conducted multiple linear regression analyses and identified a definite correlation between the change in word memory test score and the change in serum MIF level (β = −0.09, p = 0.04).
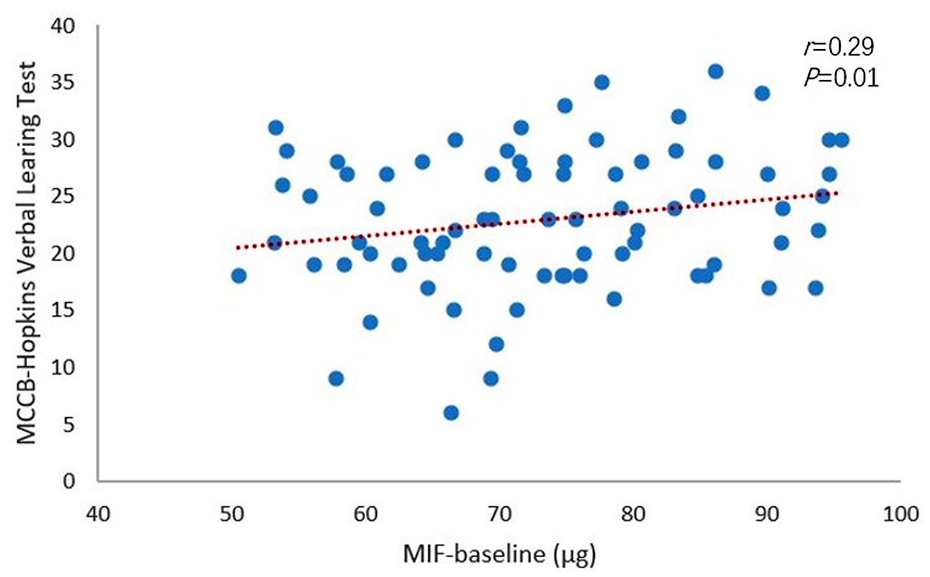
Serum MIF levels in the FES group positively correlate with verbal memory function.
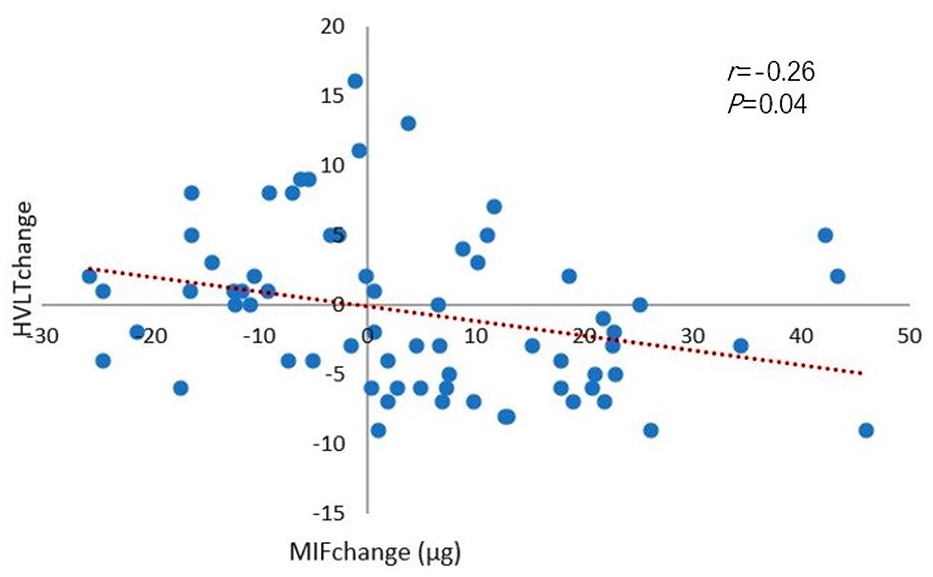
The change in serum MIF levels in the FES group negatively correlates with the change in verbal memory function.
Correlation between baseline serum MIF levels and baseline MCCB in patients with schizophrenia.
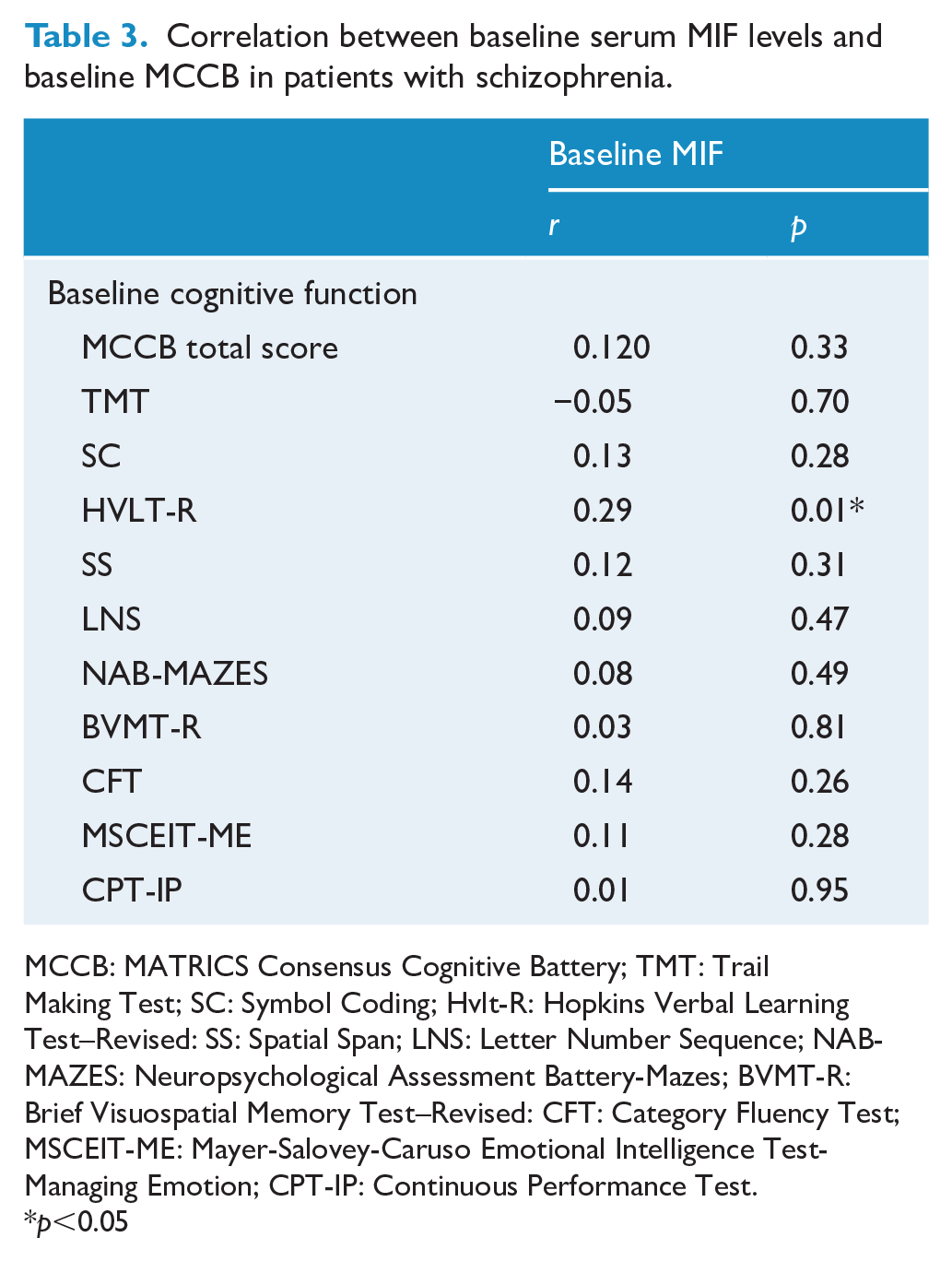
MCCB: MATRICS Consensus Cognitive Battery; TMT: Trail Making Test; SC: Symbol Coding; Hvlt-R: Hopkins Verbal Learning Test–Revised: SS: Spatial Span; LNS: Letter Number Sequence; NAB-MAZES: Neuropsychological Assessment Battery-Mazes; BVMT-R: Brief Visuospatial Memory Test–Revised: CFT: Category Fluency Test; MSCEIT-ME: Mayer-Salovey-Caruso Emotional Intelligence Test-Managing Emotion; CPT-IP: Continuous Performance Test.
p<0.05
Correlation between changes in serum MIF levels and change in MCCB in patients with schizophrenia.
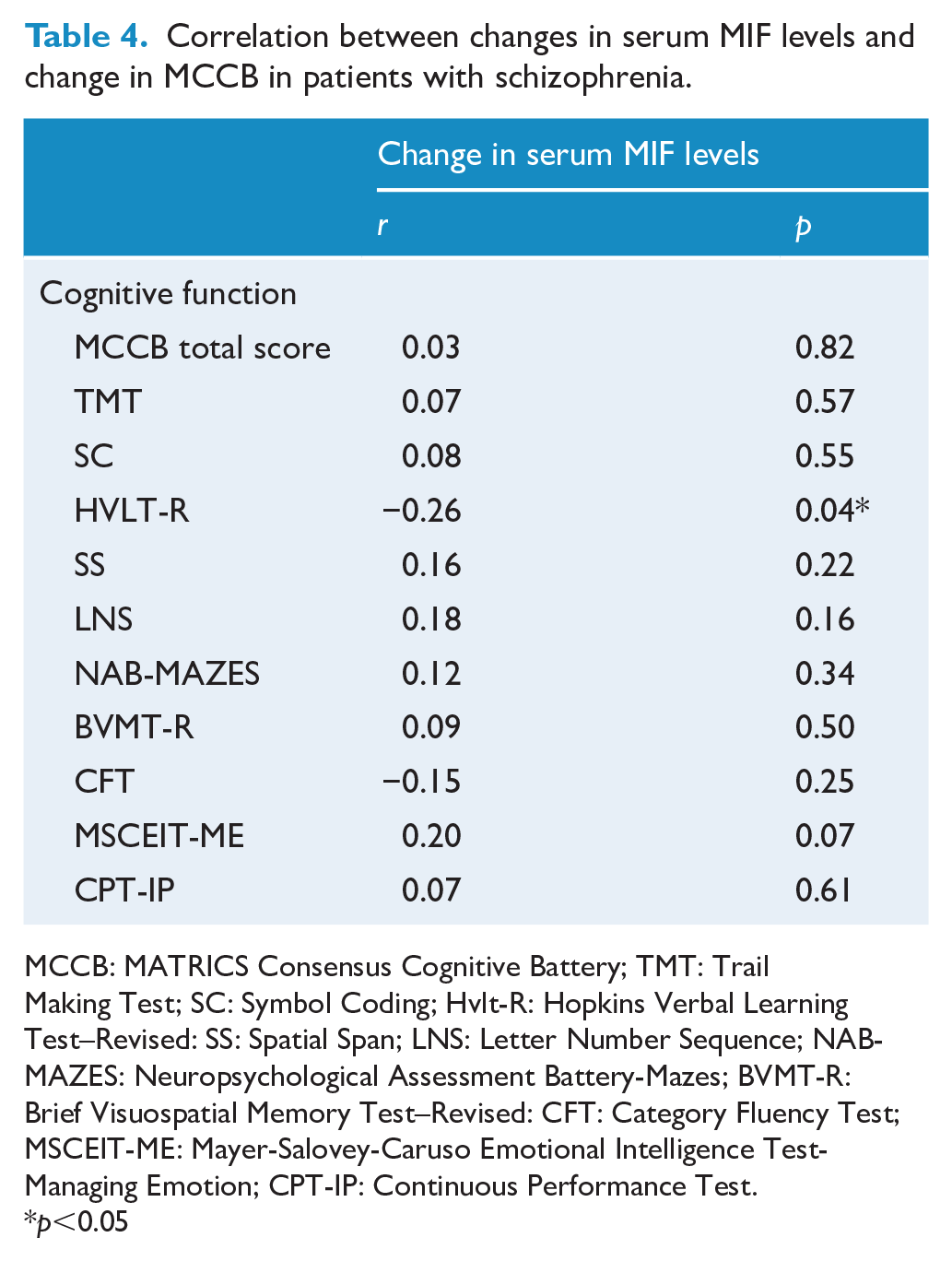
MCCB: MATRICS Consensus Cognitive Battery; TMT: Trail Making Test; SC: Symbol Coding; Hvlt-R: Hopkins Verbal Learning Test–Revised: SS: Spatial Span; LNS: Letter Number Sequence; NAB-MAZES: Neuropsychological Assessment Battery-Mazes; BVMT-R: Brief Visuospatial Memory Test–Revised: CFT: Category Fluency Test; MSCEIT-ME: Mayer-Salovey-Caruso Emotional Intelligence Test-Managing Emotion; CPT-IP: Continuous Performance Test.
p<0.05
Discussion
This is the first study to investigate the relationship between serum MIF concentrations and neurocognition in patients with FES-Sz. Our findings are as follows: (1) serum MIF levels were significantly higher in patients with FES-Sz than in healthy controls; (2) after 10 weeks of treatment with risperidone, serum MIF levels were significantly reduced and cognitive function improved in patients FES-Sz; and (3) in patients with FES-Sz, baseline MIF levels correlated positively with verbal memory function in neurocognition. Moreover, there was a negative correlation between the change in serum MIF levels and the change in the verbal memory function, with some predictive effect.
In our study, serum MIF levels in patients with FES-Sz were higher than those in healthy controls. Neuroimmunological studies have confirmed that infection or stress can induce the onset of schizophrenia, leading to increased levels of pro-inflammatory cytokines, resulting in neurotransmitter disorders and psychiatric symptoms (van Kesteren et al., 2017). Serum MIF has a documented role in the induction and regulation of various inflammatory diseases. It is involved in diverse physiological processes of the body in terms of both chemokine and cytokine activities. Our results suggest that MIF is expressed in high levels in patients with FES-Sz, which is consistent with the results reported in a previous study (Okazaki et al., 2018), suggesting that inflammation is involved in the development of schizophrenia. Owing to the unique physiological characteristics of this cytokine, elevated levels of MIF appear to play a dual role in the onset of schizophrenia. Moreover, this cytokine protects the body. It was found that, in mice, MIF is present in both the central and peripheral tissues and is an important part of the endocrine system and the HPA axis (Bernhagen et al., 1993). When the body is under stress, MIF is released into the bloodstream and can initiate a systemic stress response. MIF also directly modulates glucocorticoid-dependent immunosuppression (Flaster et al., 2007), and its levels simultaneously increase along with adrenocortical hormones in response to stress or trauma.
However, MIF overexpression in immune cells may have a detrimental role, leading to chronic inflammatory diseases in the central nervous system and peripheral blood. A previous study found that MIF stimulated the production of pro-inflammatory cytokines, including TNF-α, IL-1β and IL-6 (Bilsborrow et al., 2019). These cytokines can facilitate catecholaminergic neurotransmitter functions, indirectly increase the activity of hippocampal dopaminergic neurons and participate in the occurrence of mental disorders. Moreover, schizophrenia is characterised by microglial activation (Monji et al., 2013). MIF is an important pro-inflammatory factor that can promote the activation of microglia. Zhang et al. reported that, following activation of microglia and infiltrating macrophages, MIF directly acted on amyloid β (Aβ) protein deposits, inducing chronic neuroinflammation (Zhang et al., 2019). In turn, excessive activation of microglia cells has been reported to promote the release of substantial amounts of MIF, thus aggravating the neuroimmune response (Huleihel et al., 2017).
To date, no relevant studies have reported on the effects of MIF on patients with FES-Sz before and after risperidone treatment. Findings of this study showed improved cognitive function and decreased MIF levels following risperidone treatment, further verifying the involvement of neuroinflammatory processes in schizophrenia and implicating MIF in the pathogenesis of schizophrenia. These findings are consistent with the results of multiple clinical trials (Akhondzadeh et al., 2007; Laan et al., 2010), providing evidence that anti-inflammatory drugs can improve psychiatric symptoms. Also, that reduced inflammation improves cognitive function, and that the levels of most inflammatory cytokines in patients receiving antipsychotic treatment approach those of healthy controls gradually (Miller et al., 2011). There are opposing opinions concerning the effects of risperidone on cognitive function, with some suggesting that reducing risperidone dosage may improve cognitive function (Zhou et al., 2018). In contrast, others have reported that risperidone can modulate non-competitive N-methyl-
Our data suggest that in patients with FES-Sz, a high serum MIF level at baseline was associated with better verbal memory function. While there was no significant difference in verbal memory function before and after risperidone treatment, a previous study reported that verbal memory impairment is the most severe cognitive impairment in patients with FES-Sz (McCleery et al., 2014); therefore, any effects of treatment are likely to require considerable time to manifest. Risperidone treatment resulted in a decrease in serum MIF levels. The greater decrease in MIF after drug treatment, the less improvement of verbal memory. This suggests that MIF has a protective effect in schizophrenia. Schizophrenia involves a global impairment of cognitive function, while verbal memory relies on a network of regions, including the prefrontal cortex and medial temporal lobe structures (Antoniades et al., 2018), through which the hippocampus then acquires new information. In a recent study, MIF function was blocked using MIF antagonists to measure the expression patterns of MIF protein and MIF messenger RNA (mRNA) (Chai et al., 2020). They observed impaired hippocampal cell proliferation and synaptic growth and speculated that MIF might help to maintain the function of neural stem cells and promote the growth of neuronal processes during hippocampal development. MIF further activates the glucocorticoid and monoaminergic neurotransmitter systems through stress-induced activation of the HPA axis to regulate emotion-related memory functions and affect the neuroplasticity of memory function (Yirmiya and Goshen, 2011). MIF also exists in the mossy fibres of the dentate gyrus and hippocampal dendrite structure (Cox et al., 2013), which has a certain protective effect on nerve fibres. In a mouse model of Alzheimer’s disease (Zhang et al., 2019), inhibition of MIF can stimulate glial cells, resulting in an insufficient Aβ protein clearance rate, thereby affecting memory function. MIF can promote the activation of neuromicroglial cells and indirectly affect T cells, leading to the activation of meningeal medulloid cells, and regulate cognitive levels, such as verbal memory (Bacher et al., 2002). In a 6-week randomised, double-blind study of combined treatment with risperidone and celecoxib, COX-2 inhibitor (Müller et al., 2002), a group of schizophrenia patients treated with the combined therapy had significantly improved cognitive function than the group that received the antipsychotic drug alone. In a rat model of adjuvant arthritis, MIF inhibitors could induce the expression of inducible nitric oxide synthase and COX-2 (Zhang et al., 2018). MIF knockout in a stroke model revealed increases in caspase-3 activity, neuronal death and infarct size (Zhang et al., 2014). However, some study findings differ from those in our study.A previous study found that the upregulation of MIF is closely related to the metabolic syndrome of schizophrenia (Cui et al., 2018). The damage of the central insulin axis and abnormal metabolism can lead to neuronal damage, as well as an increase in astrocytes and microglia cells, so as to affect cognitive function (Bryll et al., 2020). Moreover, MIF antagonists have been shown to have protective effects on blood–brain barrier permeability, the infarct area and neurological dysfunction after ischemic injury (Liu et al., 2018). Our results in patients with FES-Sz suggested that MIF has a protective role in verbal memory; however, more inflammatory factors need to be included in the analysis as part of more comprehensive exploration.
This study had several limitations. First, our findings did not pass the Bonferroni correction (the p value should be <0.005, which is equal to 0.05/10, given that we had 10 subscales). However, some correlation can be seen at baseline and after treatment. We used partial correlation to exclude relevant confounding factors. There was almost no effect on constant correlation due to other factors. In future studies, the sample size should be increased, and the follow-up time needs to be prolonged to obtain conclusive results. Second, the pre- and post-treatment measurement of cognitive function in the same patients may have induced a learning effect that could have influenced the results. Third, only serum MIF levels were measured in this study; evaluation of MIF gene expression and detection of MIF variants could be the next step, which would be more helpful to study the pathogenesis of schizophrenia and identify its association with cognitive function. Fourth, the immune system is complex and involves various interactions between cytokines. This study did not examine other pro- and anti-inflammatory cytokines. Measuring the serum level of only one cytokine, such as MIF, can only reflect changes in this single cytokine. Regarding the relationship between the immune system and cognitive function in patients with schizophrenia, this approach is not sufficiently comprehensive to provide a reliable result. Therefore, future studies should not only focus on single cytokines but also cytokine networks and their activation.
In conclusion, schizophrenia has adverse effects on cognition, and our results suggest that the immune system is activated in FES-Sz. MIF is involved in the pathogenesis of schizophrenia; MIF levels increase in early disease stages and decrease following treatment with risperidone, indicating that MIF is a biomarker of this disease. In this study, we identified a potential correlation between high serum levels of MIF and verbal memory function. Future research should shift the study focus from individual cytokines to cytokine networks.
Footnotes
Author Contributions
J-J.Y., Q.Z., D-C.C., H-N.L. and J-Q.S. were responsible for clinical data collection. J-Q.S. was responsible for the laboratory experiments. H-N.L. and J-Q.S. were involved in evolving the ideas and editing the manuscript. J-J.Y., Q.Z., D-C.C. were responsible for study design, statistical analysis and manuscript preparation. All authors have contributed to and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Informed consent was obtained from all participants, and the study was approved by the Institutional Ethical Review Board of Beijing Huilongguan Hospital. Approval Number (2018-10).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Funds of Capital Health Research and Development of Special (2018-2-2131). The funding sources in this study had no role in the study design, data collection and analysis, decision to publish or preparation of the article.
Data Availability Statement
Data supporting the findings of this study are available from the corresponding author upon reasonable request.
