Abstract
Objectives:
We investigated the association of the aldehyde dehydrogenase 2 (ALDH2) polymorphism (rs671), which is involved with the dopaminergic function, and with changes in cytokine levels and cognitive function, in a 12-week follow-up study in patients with bipolar disorder.
Methods:
Patients with a first diagnosis of bipolar disorder were recruited. Symptom severity and levels of plasma cytokines (tumor necrosis factor α, C-reactive protein, interleukin 6 and transforming growth factor β1) were examined during weeks 0, 1, 2, 4, 8 and 12. Neurocognitive function was evaluated at baseline and endpoint. The ALDH2 polymorphism genotype was determined.
Results:
A total of 541 patients with bipolar disorder were recruited, and 355 (65.6%) completed the 12-week follow-up. A multiple linear regression analysis showed a significant (p = 0.000226) association between the ALDH2 polymorphism and changes in C-reactive protein levels. Different aspects of cognitive function improved in patients with different ALDH2 genotypes. Only patients with the ALDH2*1*1 genotype showed significant correlations between improvement of cognitive function and increased transforming growth factor -β1.
Conclusion:
The ALDH2 gene might influence changes in cytokine levels and cognitive performance in patients with bipolar disorder. Additionally, changes in cytokine levels and cognitive function were correlated only in patients with specific ALDH2 genotypes.
Introduction
Bipolar disorder (BD), characterized by mood dysregulation, is recurrent and often chronic. BD-I and BD-II are two of its major subtypes. The lifetime prevalence rate of BD-I is about 2–3% and of BD-II is about 1–3% (Merikangas et al., 2007). The clinical characteristics of BD include social and occupational functional impairment, comorbidity, suicide attempts and a high familial risk, which are commonly seen in both subtypes (Mitchell et al., 2004). Genetic factors can be important in the etiology of BD, which is supported by evidence from family, twin and adoption studies (McGuffin et al., 2003). In addition to genetics, inflammation has been involved in the pathophysiology of BD (Goldstein et al., 2009). Meta-analyses (Fernandes et al., 2016; Goldsmith et al., 2016) have reported that proinflammatory cytokines—including C-reactive protein (CRP), interleukin-6 (IL-6) and tumor necrosis factor (TNF-α)—increased during acute manic and depressive episodes and decreased after treatment.
The dopaminergic system may play a central role in BD (Cousins et al., 2009) because dysregulated dopamine signaling is implicated in its pathogenesis (Greengard, 2001). As one of the predominant catecholamine neurotransmitters, dopamine has been linked to mood and cognition (Missale et al., 1998). Dopamine is also important for bridging the nervous and immune systems (Sarkar et al., 2010). Dopamine receptors are present in immune cells (Sarkar et al., 2010) and in glial cells (Kuric et al., 2013). In addition, evidence suggests that dopaminergic agonists might inhibit cytokine secretion and T cell proliferation and thus regulate immune functions and inflammatory reactions (Kuric and Ruscher, 2014). Therefore, it has been proposed (Aalberse and Schuurman, 2002) that changes in cytokine levels may be modulated by dopaminergic genes, while dopaminergic genes may also contribute to susceptibility for BD.
ALDH2 is the major ALDH isozyme for the oxidation of dopamine (Lamensdorf et al., 2000) by catalyzing the oxidation of DOPAL (3,4-dihydroxyphenylacetaldehyde) into DOPAC (3,4-dihydroxyphenylacetic acid) (Elsworth and Roth, 1997). One polymorphism of the ALDH2 gene (rs671) yields two alleles: ALDH2*1 and ALDH2*2. The ALDH2*1/*1 genotype encodes the active enzyme that metabolizes acetaldehyde, whereas the ALDH2*1/*2 and *2/*2 genotypes (ALDH2*2+ allele) encode partially and completely inactive enzymes (Crabb et al., 1989). The ALDH2*2 allele can be found in about half of the Asian population (Agarwal and Goedde, 1992), including in Han Chinese in Taiwan, but it is rarely seen in Caucasians. The ALDH2 gene is well known for its association with drinking behavior and for the risk of developing alcohol dependence. Although we did not find any individual association between the ALDH2 gene and BD (Lee et al., 2012; Wang et al., 2014), we previously reported (Lee et al., 2010) that the ALDH2 and DRD2/ANKK1 genes significantly interacted in BD-II. This significant interaction suggests that the ALDH2 gene may be associated with BD-II, possibly because of its influence on dopamine metabolism. This result also raised the question of whether the ALDH2 gene is associated with cytokine levels in BD.
Patients with BD continue to manifest cognitive dysfunction even after well-defined clinical remission of BD (Martinez-Aran et al., 2004; Rubinsztein et al., 2000) reported that even in euthymic states, patients with BD still showed poorer ability on executive function and verbal memory than did healthy controls, and that there was a high correlation between neuropsychological performance and global assessment performance. Cognitive impairment might also negatively affect a patient’s drug adherence, treatment effect and prognosis (Simonsen et al., 2008). Some (Zhao et al., 2015) have suggested that the pathophysiology of BD, such as dopamine dysregulation, may be responsible for cognitive impairment in euthymic patients with BD. Changes in the dopaminergic system are known to contribute to cognitive impairment (Nieoullon, 2002). A possible association was reported between dopamine receptor genes (Zhao et al., 2015) and dopaminergic genes (Simpson et al., 2014) with cognitive impairment in BD. However, most studies have focused on cross-sectional associations but not on longitudinal changes of cognitive function during treatment. In the current study, we explored the genetic associations between ALDH2, a candidate dopaminergic gene, and longitudinal changes in cytokine levels and cognitive function. Because immune dysfunction has been associated with cognitive impairment (Rosenblat et al., 2015), we investigated the correlation between cognitive function and cytokine levels, and the influence of the dopaminergic gene on this correlation.
Aim of the study
The aim of the study was to explore the association of the ALDH2 polymorphism with changes in cytokine levels and cognitive function in a 12-week follow-up study in patients with BD. We also examined the correlation between cytokine levels and cognitive function stratified by the ALDH2 polymorphism.
Methods
Patient selection
The research protocol was examined and approved by the Institutional Review Boards for the Protection of Human Subjects at National Cheng Kung University Hospital and at Tri-Service General Hospital. All patients signed informed consent forms after the study protocol had been completely explained.
This study is a secondary analysis of the combination of 2 clinical trials. The original studies were (1) a randomized, double-blind, controlled 12-week trial that investigated the add-on effect of memantine on BP-II treated using valproate (VPA) (trial registration: NCT01188148) (Lee et al., 2013) and (2) a randomized, double-blind, controlled 12-week trial investigating the add-on effect of dextromethorphan (DM) in BD treated using VPA (trial registration: NCT01188265) (Chen et al., 2014). We analyzed all the BD patients, but not the healthy controls, from the two studies.
Patients with BD-I and BD-II were enrolled from outpatient and inpatient settings. All participants were initially interviewed by an attending psychiatrist. The patients then underwent a structured interview with a clinical psychologist to confirm Diagnostic and Statistical Manual of Mental Disorders-Fourth edition (DSM-IV) diagnoses, using the Chinese Version of the Modified Schedule of Affective Disorder and Schizophrenia-Life Time (SADS-L) (Endicott and Spitzer, 1978), which has good inter-rater reliability (Huang et al., 2004). The exclusion criteria were having any major mental illnesses, borderline personality disorder, drug abuse or dependence or cognitive disorders other than BD-I and BD-II.
The DSM-IV-TR (Text Revision) criteria require a minimum duration of 4 days for the diagnosis of hypomania. However, the current study used a 2-day duration for hypomania suggested by epidemiologic data (Judd et al., 2003) as more prevalent in community samples. Therefore, the diagnosis of BD-II in the current study is based on the 2-day minimum for hypomania.
Study design
The patients were given add-on DM (30 mg/day or 60 mg/day), or memantine (5 mg/day), or a placebo for 12 weeks after a baseline assessment augmenting their open-label VPA treatment (500 mg or 1000 mg daily). Symptom severity was assessed at baseline, and then treatment responses were measured on weeks 1, 2, 4, 8 and 12. Concomitant medications were limited to lorazepam (<8 mg/daily) for insomnia, risperidone (1–6 mg/daily) for manic episodes, fluoxetine (20 mg/daily) for depressive episodes and an anticholinergic drug was permitted for extrapyramidal syndrome.
The clinical severity of depressive symptoms was assessed using the 17-item Hamilton Depression Rating Scale (HDRS) (Hamilton, 1967), and manic symptoms using the 11-item Young Mania Rating Scale (YMRS) (Young et al., 1978). The minimum recruitment criterion was an HDRS ⩾ 18.
We used the Wisconsin Card Sorting Test (WCST) and the Conners’ Continuous Performance Test (CPT) to assess cognitive function. The WCST evaluates several types of executive functions, including categorization, abstract reasoning, maintaining sets, set switching, strategic planning and modulating impulsive responses; it is a validated performance test for frontal lobe dysfunction (Axelrod et al., 1996). WCST scores were based on the total number of errors (TNE), perseverative errors (PE), perseverative responses (PR), number of categories completed (NCC) and number of trials required to complete the first category (TCC).
The CPT measures performance: number of errors of omission and errors of commission, hit reaction time (HRT), HRT standard error (HRT SE), variability and detectability (d′). Errors of omission are the number of failures to respond to the target stimulus, errors of commission are responding to a non-target (X) stimulus, HRT is the mean response time (milliseconds [ms]) for all target responses over the full six trial blocks, HRT SE is the consistency of response times and expresses the SE response to targets and detectability (d′) is the patient’s ability to discriminate between targets and non-targets.
Blood samples and genotyping
At baseline, 10 mL of blood was collected from the antecubital vein of every participant. DNA was extracted from the lymphocytes of the blood samples. The ALDH2 gene was genotyped using a modified protocol (Dandre et al., 1995). All samples were retyped and double-checked. The genotype error rate was less than 5%.
Whole blood was centrifuged at 3000 g for 15 min at 4°C to isolate plasma, which was immediately stored at −80°C. An antibody pair assay system (Flexia; BioSource Intl., Camarillo, CA) was used to quantify cytokine levels. The blood samples were processed and analyzed according to the manufacturer’s instructions. The cytokine levels of TNF-α, CRP, IL-6 and TNF-1β were measured at baseline and on weeks 1, 2, 4, 8 and 12.
Statistical analysis
The demographic and clinical characteristics of the patients were based on the ALDH2 genotypes at baseline and endpoint, including the YMRS and HDRS scores and cytokine levels. Categorical variables were compared using χ2 tests, and continuous variables were compared using independent t tests. We combined the ALDH2*1/*2 + *2/*2 genotypes into a single group for data analysis because only 36 (6.7%) patients had the ALDH2*2/*2 genotype. This combination is based on the decreased acetaldehyde metabolic effect of these two genotypes: the ALDH2*2/*2-encoded enzyme is inactive, and the ALDH2*1/*2-encoded enzyme has only one-tenth of the metabolite rate of the ALDH2*1/*1-encoded enzyme (Keung and Vallee, 1998). Because the distribution of all cytokine levels was positively skewed (Table 1), we used arithmetic transformations (log [x + 1]) for all cytokine levels to produce approximately normal distributions for further analysis. We used the intent to-treat (ITT) analysis by including all randomized patients who had taken at least one dose of the study drug, had one baseline assessment, and had at least one assessment after baseline. All outcome variables—HDRS, YMRS, and cytokine levels—were analyzed in the ITT set, and missing data were filled in using the last observation carried forward (LOCF) method.
Characteristics and cytokine levels of bipolar patients at baseline and endpoint based on different ALDH2 genotypes.
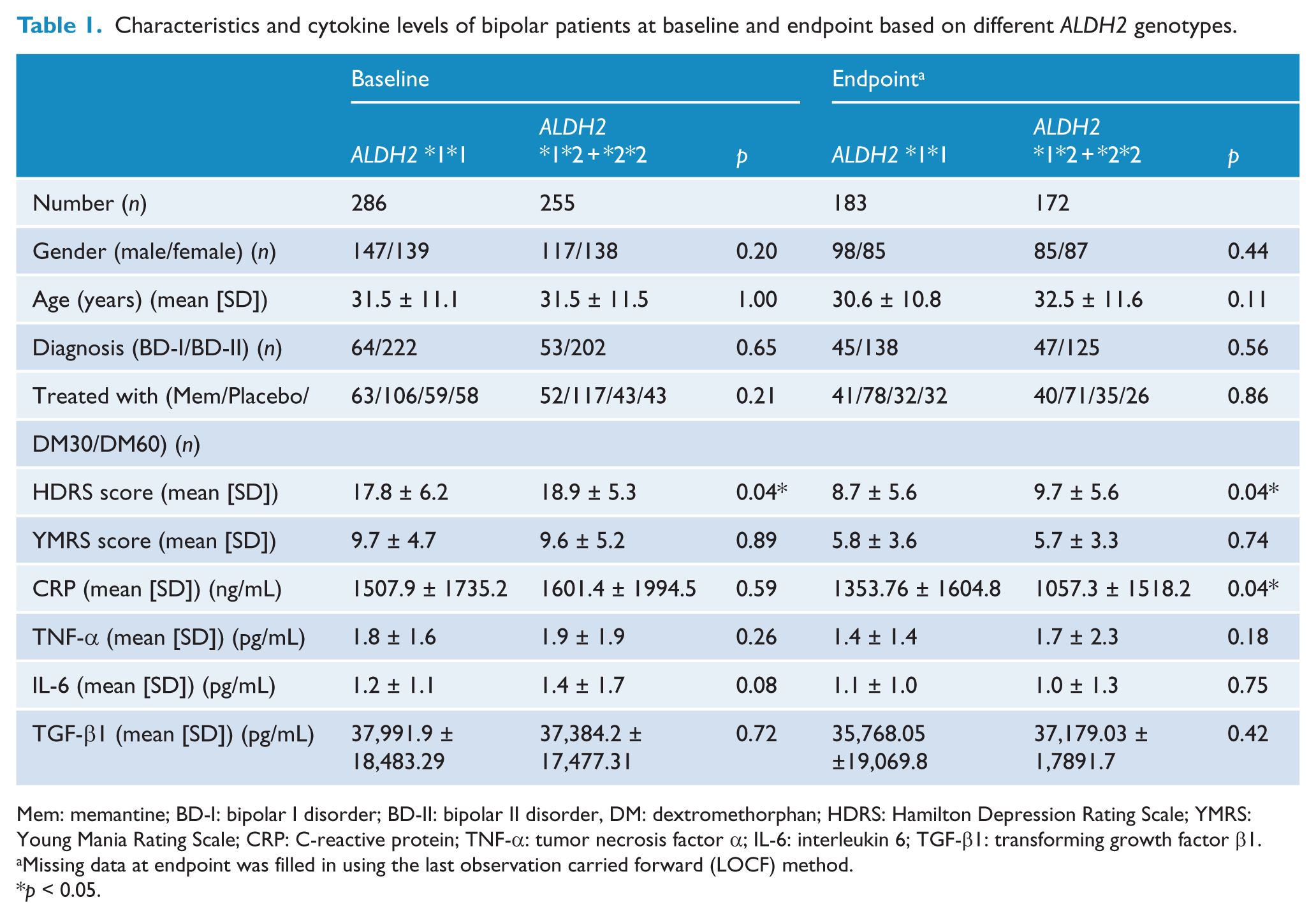
Mem: memantine; BD-I: bipolar I disorder; BD-II: bipolar II disorder, DM: dextromethorphan; HDRS: Hamilton Depression Rating Scale; YMRS: Young Mania Rating Scale; CRP: C-reactive protein; TNF-α: tumor necrosis factor α; IL-6: interleukin 6; TGF-β1: transforming growth factor β1.
Missing data at endpoint was filled in using the last observation carried forward (LOCF) method.
p < 0.05.
The generalized estimating equation (GEE) method (Zeger et al., 1988) was used to evaluate the association between the ALDH2 polymorphism and changes in symptoms and cytokine levels after 12 weeks of follow-up. Six GEE models were run using clinical severity (HDRS, YMRS) and cytokine levels as dependent variables. Treatment duration, ALDH2 genotypes, ALDH2 genotype × treatment duration, treatment given (add-on DM, memantine or placebo), gender, age and clinical severity (for cytokine levels only) were included as independent variables. The interaction term of the ALDH2 genotype × treatment duration was considered the association of the ALDH2 genotype with changes in clinical symptoms or cytokine levels.
After we had divided the patients into the ALDH2*1/*1 and the ALDH2*1/*2 + *2/*2 groups, we used a multivariate analysis of covariance (MANCOVA), controlled for age, gender, baseline HDRS, YMRS scores and augmented treatment received to assess the performance differences in WCST and CPT between baseline and endpoint. We used a principal component analysis (PCA) to reduce the WCST and CPT measures to several composite scores. The association between each composite score with each cytokine level stratified by the ALDH2 genotypes was then analyzed using Pearson’s correlation.
SPSS 18 for Windows (SPSS Inc., Chicago, IL) was used for statistical computations. Significance was set at p < 0.05. The power analysis was done using G-Power 3 software, and the effect size conventions were determined accordingly (Buchner et al., 1996).
Results
In all, 541 (309 + 232) patients with BD were recruited. All patients were confirmed to have no history of taking antipsychotics or mood stabilizers. Of 541, 355 (65.6%) completed the 12-week follow-up and 186 (34.4%) dropped out. The reasons for discontinuing are published elsewhere (Chen et al., 2014; Lee et al., 2013).
Most of the demographic and clinical characteristics of the patients were not significantly different in the ALDH2 *1*1 and *1*2 + *2*2 genotype groups at baseline or endpoint (Table 1), except that patients with the ALDH2 *1*2 + *2*2 genotype had higher HDRS scores at baseline (p = 0.04) and endpoint (p= 0.04) and a lower level of CRP at endpoint.
A multiple linear regression analysis that controlled for gender, age, HDRS and YMRS scores, and the type of treatment given showed a significant (p = 0.000226) association between the ALDH2 polymorphism and the changes in CRP levels after 12 weeks of treatment (Table 2). The CRP level was significantly lower in patients with the ALDH2*1*2 + *2*2 genotype than in those with the ALDH2*1*1 genotype (Figure 1). However, the ALDH2 polymorphism was not associated with changes in clinical symptoms or other cytokine levels (Table 2).
The association of the ALDH2 polymorphism with changes in symptoms and cytokine levels after 12 weeks of follow-up.
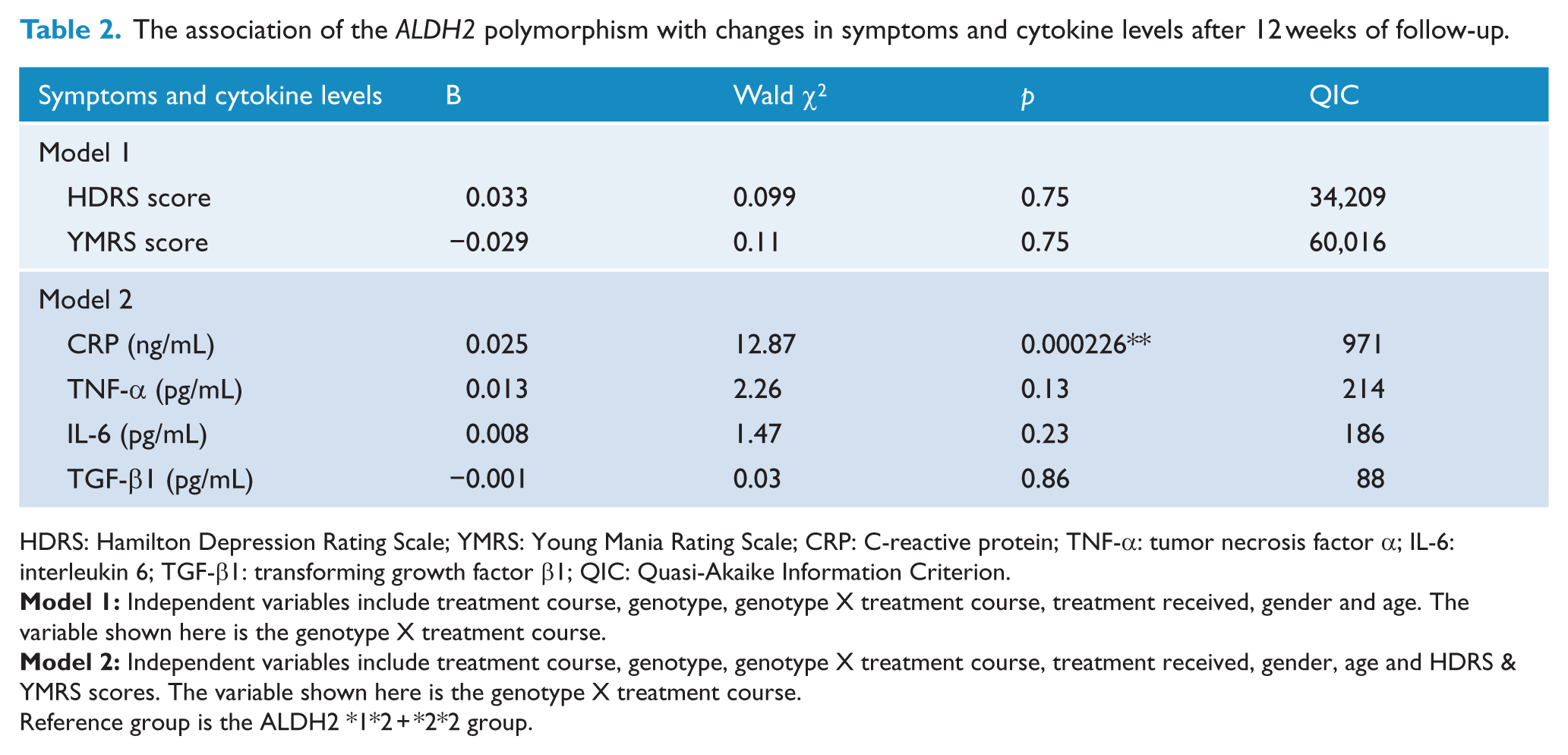
HDRS: Hamilton Depression Rating Scale; YMRS: Young Mania Rating Scale; CRP: C-reactive protein; TNF-α: tumor necrosis factor α; IL-6: interleukin 6; TGF-β1: transforming growth factor β1; QIC: Quasi-Akaike Information Criterion.
Reference group is the ALDH2 *1*2 + *2*2 group.
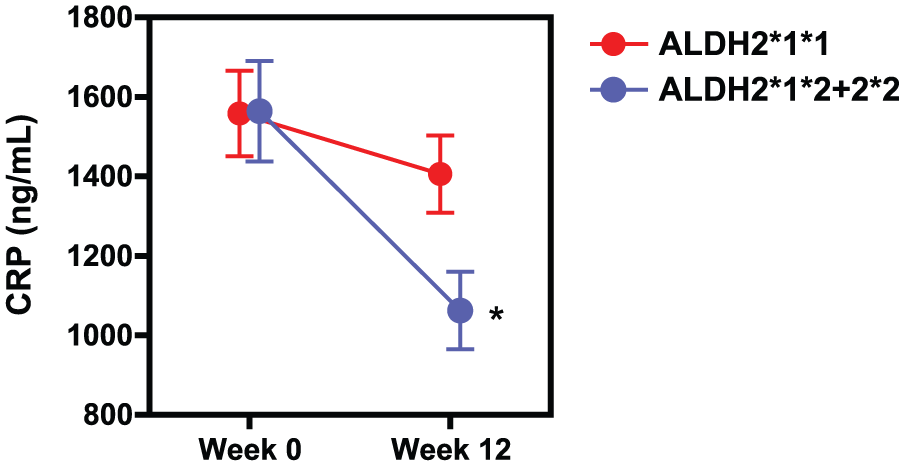
Association between the ALDH2 (rs671) polymorphism and changes in CRP levels in patients with BD over 12 weeks of treatment.
After 12 weeks of treatment, WCST and CPT cognitive performances significantly improved in patients with the ALDH2*1*1 genotype, but not in those with the ALDH2*1*2 + *2*2 genotype. The differences found in WCST subsets were TNE (p = 0.000002), PE (p = 0.000089), PR (p = 0.00040), NCC (p = 0.000145) and TCC (p = 0.009), and in the CPT subsets was Hit RT Std. Error T-Score (p = 0.16) (Table 3), which is normally related to distractibility. Patients with the ALDH2*1*2 + *2*2 genotype showed significant improvement in Commission T (p = 0.024) and Detectability (d′) T-scores (p = 0.031) (Table 3), both of which are related to impulsivity in CPT.
Performance of each cognitive task at the baseline and endpoint in the ALDH2 *1*1 and ALDH2 *1*2 + *2*2 genotype groups.
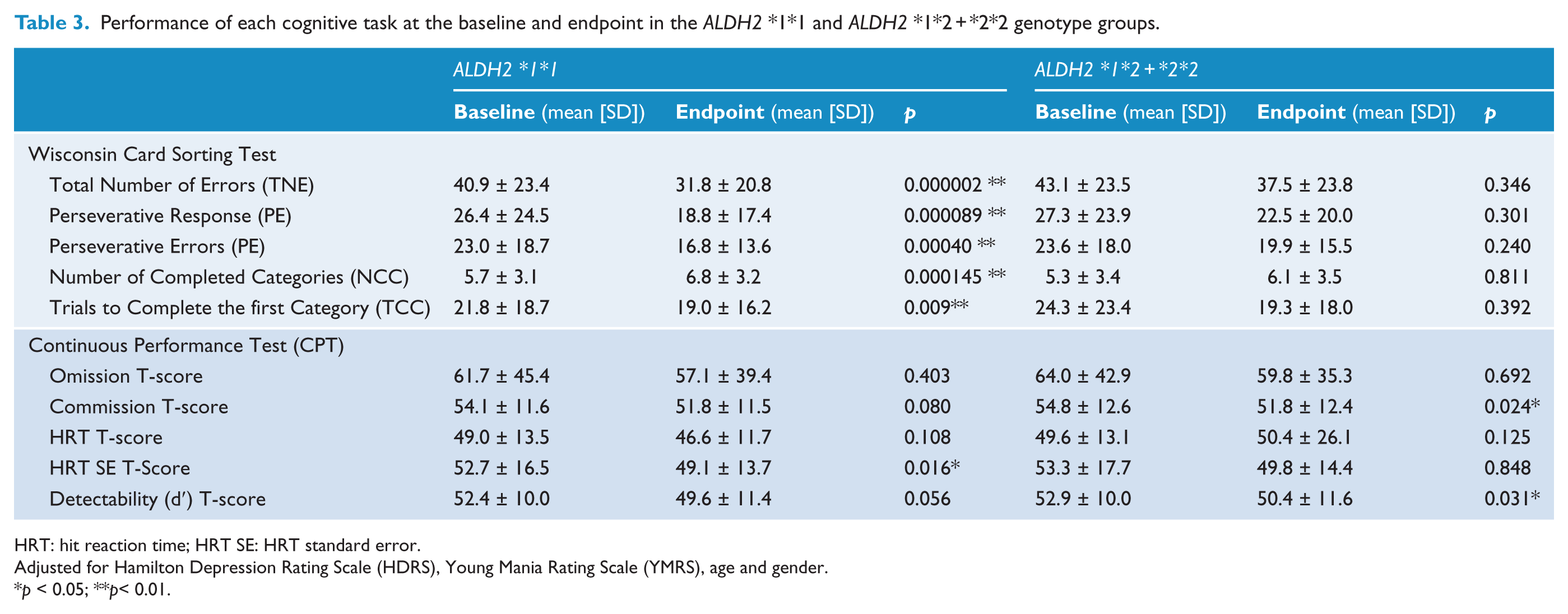
HRT: hit reaction time; HRT SE: HRT standard error.
Adjusted for Hamilton Depression Rating Scale (HDRS), Young Mania Rating Scale (YMRS), age and gender.
p < 0.05; **p< 0.01.
Factor analysis was used to condense the WCST and CPT measures to reduce the type I error. Three factors with eigenvalues > 1.00 from the varimax rotation were retained: Factor 1 was a composite score for WCST, factor 2 for CPT impulsivity and factor 3 for CPT distractibility (Table S1). Negative correlation between plasma levels of transforming growth factor (TGF)-β1 with factor 1 and factor 2 was significant only in patients with BD and the ALDH2*1*1 genotype (Table 4). There was no correlation between cognitive function and cytokine levels in patients with BD and the ALDH2*1*2 + *2*2 genotype.
Correlation between cytokine levels and factor scores of cognitive functions.
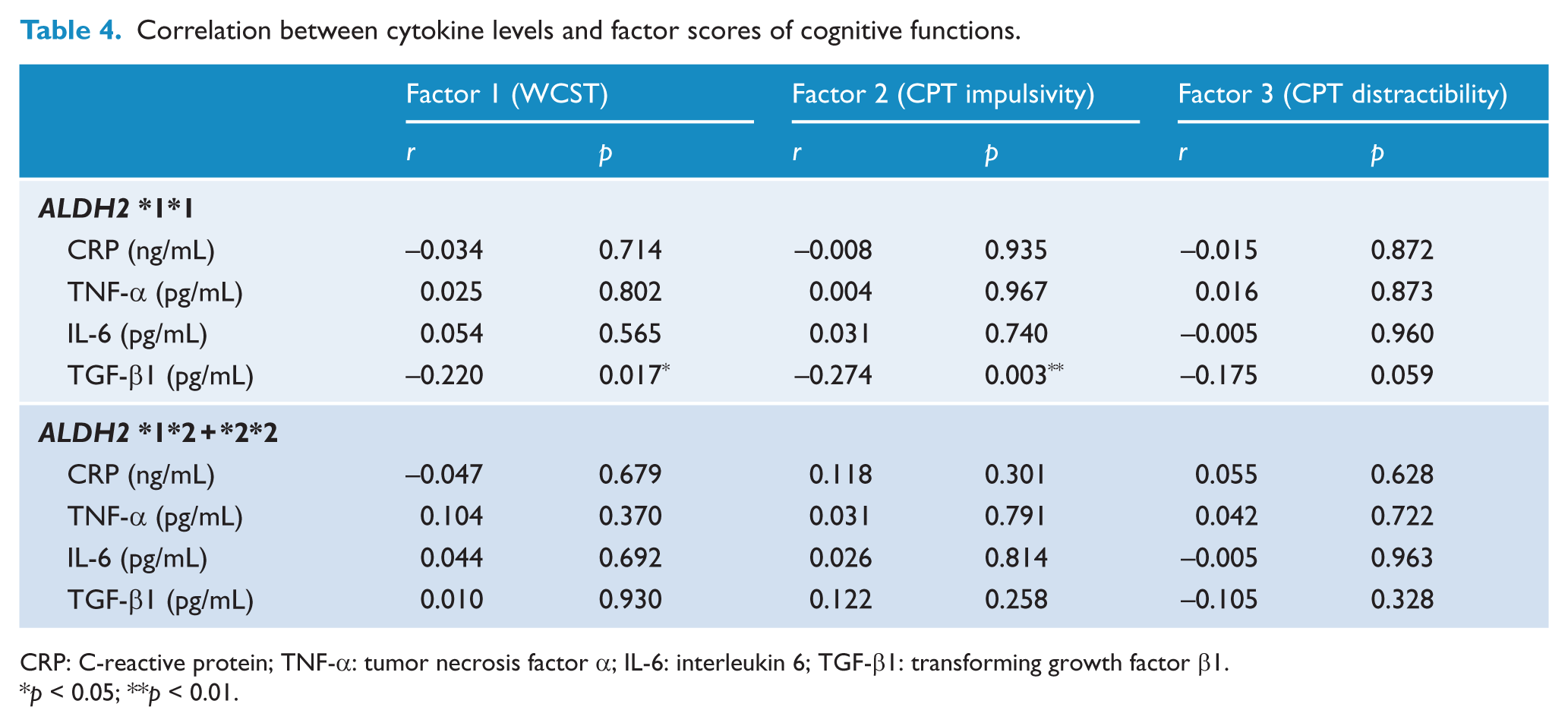
CRP: C-reactive protein; TNF-α: tumor necrosis factor α; IL-6: interleukin 6; TGF-β1: transforming growth factor β1.
p < 0.05; **p < 0.01.
The study had a power of approximately 0.68 to detect a small effect, and 1.00 to detect medium and large effects for multiple regression analysis (n = 541). In this power analysis, the effect-size conventions were determined as follows (Buchner et al., 1996): small effect size = 0.02, medium effect size = 0.15 and large effect size = 0.35 for the multiple regression model (α = 0.05). The study had a power of approximately 1.0 for MANCOVA when the effect size was set at 0.25.
Discussion
Our most important findings after 12 weeks of treatment were these. First, the change in CRP level was significantly associated with the ALDH2 gene. Second, WCST and CPT distractibility scores improved only in patients with the ALDH2 *1*1 genotype, and CPT impulsivity scores improved only in patients with the ALDH2 *1*2 + *2*2 genotype. Third, changes in cognitive function and plasma TGF-β1 levels were significantly negatively correlated only in patients with the ALDH2 *1*1. We found preliminary evidence that the ALDH2 gene affects changes in CRP levels and cognitive performance in patients with BD. In addition, a correlation between cognitive function and cytokine levels occurred only in patients with specific genotypes of the ALDH2 gene.
The ALDH2 (rs671) polymorphism and changes in CRP levels were positively associated in patients with BD; however, the ALDH2 (rs671) polymorphism and changes in clinical severity or of cytokine levels were not associated in patients with BD. CRP levels significantly decreased in patients with the ALDH2 *1*2 + *2*2 genotype, but not in those with the ALDH2 *1*1 genotype. Bian et al. (2010) and Xu et al. (2011) reported in their cross-sectional studies, that the ALDH 2*2 allele was associated with elevated plasma CRP levels after the onset of acute myocardial infarction (AMI) with greater risk for AMI in Han Chinese populations. We found, however, that BD patients with the ALDH 2*2 allele (the same as the ALDH 2*1*2 + *2*2 genotype) had baseline CRP levels that were not significantly different from those of patients with the ALDH2 *1*1 genotype. This discrepancy might be related to different illnesses studied, which suggests different roles in inflammation for the ALDH2 gene in different illnesses. Because no one has ever used a longitudinal study to investigate the effect of the ALDH2 (rs671) polymorphism on cytokine levels, ours is the first to show that, after treatment, patients with the ALDH2 *1*2 + *2*2 genotype had significantly lower CRP levels than did those with the ALDH2 *1*1 genotype. In the metabolism of dopamine, the ALDH2 *1*2 and *2*2 genotypes are associated with reduced ALDH2 enzymatic activity, which might slow down the oxidation of DOPAL into DOPAC and cause DOPAL to accumulate (Elsworth and Roth, 1997). DOPAL is considered toxic in in vivo dopamine metabolite, which might lead to the death of dopamine neurons (Fitzmaurice et al., 2013) and supposedly higher levels of serum hs-CRP (Xu et al., 2011). We hypothesize that treatment for BD, normally involving anti-inflammatory effect, attenuates its underlying pathophysiology, which is inflammation. Thus, patients with the ALDH2 *1*2 and *2*2 genotypes and accumulated neurotoxic metabolite will benefit more from add-on low-dose MEM and DM treatment than will patients with the ALDH2 *1*1 genotypes, who theoretically accumulate less neurotoxic metabolite. We were, however, unable to distinguish the effects of the ALDH2 (rs671) polymorphism on changes in clinical severity and other cytokine levels. Although (Guo et al., 2015) reported that the ALDH2 enzyme ameliorated IL-6- and TNF-α-induced inflammation by inhibiting autophagy in VA-13 cells. However, no previous research on this topic has fully clarified the effects of the ALDH2 (rs671) polymorphism.
This is also the first report of an association between the ALDH2 gene and cognitive function in BD. The ALDH2 gene has been associated with cognitive function (Fitzmaurice et al., 2013; Hao et al., 2011). The ALDH2 *2 allele has been associated with a risk of Alzheimer’s disease in Asian patients (Hao et al., 2011). DOPAL accumulation, which occurs people with the ALDH2 *1*2 and *2*2 genotypes, has been is hypothesized to be the pathogenesis of Parkinson’s disease (Fitzmaurice et al., 2013), a neurodegenerative disease frequently associated with cognitive function decline (Michel et al., 2014). We found that different subtypes of cognitive functions improved in patients with different ALDH2 genotypes. The WCST and CPT distractibility scores improved only in patients with the ALDH2 *1*1 genotype, and the CPT impulsivity score improved only in patients with the ALDH2 *1*2 + *2*2 genotype. Because DOPAL accumulates in patients with the ALDH2 *1*2 + *2*2 genotype, we hypothesize that DOPAL accumulation prevents improvement in the executive function and in distractibility scores in patients with BD. In contrast, the impulsivity scores of patients with BD and the ALDH2 *1*2 + *2*2 genotype showed significant improvement. One might suspect that the accumulation of DOPAL attenuates impulsivity. ALDH2 *1*2 + *2*2 genotypes are known to protect against drinking behavior and alcohol dependence. However, additional mechanistic study is needed to support our hypothesis.
In the current study, after stratification of the ALDH2 genotypes, significant correlations between increases in TGF-β1 levels and improvements in cognitive function (WCST and CPT impulsivity) were found only in patients with the ALDH2 *1*1 genotype. TGF-β1 was known for protecting neurons against different kinds of insults. (Li et al., 2013), in an animal study, that TGFβ1 treatment improved cognitive function. We hypothesize that, because patients with BD and the ALDH2*1*1 genotype are less affected by the accumulation of neurotoxic DOPAL, their cognitive performance benefit more from higher levels of TGF-β1. The present study evaluated the correlation between changes in cognitive function and in cytokine levels instead of their correlation in a cross-section design. In addition, our study population was larger than that of other studies. We believe that our findings provide new evidence that help clarify the association between inflammatory markers and cognitive function in BD. However, we found no association between CRP, TNF-α or IL-6 and cognitive function in patients with either genotype. A cross sectional inverse relationship between CRP and cognitive function was reported in patients with BD (Dickerson et al., 2013); however, no previous studies have investigated the longitudinal correlation between changes in CRP and cognitive function. The association between cognitive function decline and an increase in inflammatory markers, especially IL-6, has been reported only in healthy elderly African Americans (Goldstein et al., 2015), which highlights the influence of ethnicity. Future studies with longer follow-ups and more candidate cytokine biomarkers are needed to confirm our finding.
Our study has some limitations. First, there are 2486 known variants of the ALDH2 gene, but we investigated only one SNP. Although we did not genotype for the other 2000+ variants mentioned above, if corrected for multiple testing according to the number of all variants, current positive findings might not hold up. In addition, cognition might be a multigenically driven process. Using only a single variation in a single gene may have limited power to uncover the genetics of cognition in BD. Future studies a multiple-gene approach, or even epigenetics, is warranted. Second, we measured cytokine levels in plasma because Besedovsky and Del Rey (2011) reported that peripheral cytokine levels might reflect those in the brain; however, that correlation is still inconclusive (Pan and Kastin, 1999). Third, medication permitted in the study might have affected cognitive performance and obscured the changes in cytokine levels. We tried our best to limit concomitant treatment medication to only three drugs. In addition, when analyzing the association between the ALDH2 polymorphism and changes in symptoms and cytokine levels, add-on MEM and DM were controlled for; our findings should still be taken with caution. In addition, although risperidone and fluoxetine can also influence dopamine activity, when we analyzed the association between the ALDH2 gene and cognitive function, we did not control for the use and doses of risperidone and fluoxetine. The current association between the ALDH2 gene and cognitive function might be obscured because we did not control these two medications. Third, other factors that might affect cytokine levels, such as BMI, alcohol drinking and tobacco smoking, were not controlled for in the current study. Fourth, instead of using the DSM-IV-TR criterion of a 4-day duration for hypomania, we used the 2-day duration, which is not widely accepted; therefore, our positive finding in the current study might not apply to patients with BD-II diagnosed according to the DSM-IV-TR criterion. Fifth, none of the patients in the current study had ever taken antipsychotics, antidepressants or mood stabilizers before; thus, our findings might not apply to patients with a history of their use. However, we were unable to confirm whether the patients had ever used benzodiazepines, which are legally accessible in Taiwan without a prescription. Finally, we focused on the changes in cytokine levels and cognitive function after a 12-week pharmacological intervention. A longer follow-up is required to evaluate longer-term changes in cytokine levels and cognitive function.
Conclusion
Our study provides initial evidence of the association between the ALDH2 (rs671) polymorphism and change in CRP levels after 12 weeks of treatment. We also found that different subtypes of cognitive functions improved in patients with different ALDH2 genotypes. After the ALDH2 genotypes had been stratified, significant correlations between increases in TGF-β1 levels and improved cognitive function were found in patients with the ALDH2 *1*1 allele. Our findings suggest that this polymorphism, which modulates dopamine metabolism, might also affect changes in CRP levels and cognitive function, and that changes in cytokine levels were correlated with cognitive function only in patients with unchanged dopamine metabolism. Our findings support an association between dopamine metabolism, cognitive function and cytokine levels. However, the causal relation and the underlying mechanisms of this association require additional study. Knowing the mechanisms might increase our understanding of cognitive decline and inflammatory disturbances in BD.
Footnotes
Acknowledgements
We thank Ms. I-Chin Wang for her assistance in preparing this manuscript. R.B. Lu and C.S. Chen are joint corresponding authors and contributed equally to this manuscript. NCT01188265 (![]() ) trial date was from June 2007 to June 2011 in National Cheng Kung University Hospital and Tri-Service General Hospital. NCT01188148 (https://register.clinicaltrials.gov/) trial date was from 1 August 2008 to 31 July 2012 in National Cheng Kung University Hospital and Tri-Service General Hospital.
) trial date was from June 2007 to June 2011 in National Cheng Kung University Hospital and Tri-Service General Hospital. NCT01188148 (https://register.clinicaltrials.gov/) trial date was from 1 August 2008 to 31 July 2012 in National Cheng Kung University Hospital and Tri-Service General Hospital.
Author contributions
S.Y.L. wrote the first draft. S.Y.L., T.Y.W., S.L.C., Y.H.C. and L.J.W. managed the laboratory work and statistics. Y.H.Y. supervised the statistics. S.Y.L., T.Y.W., P.S.C., S.Y.H., N.S.T., I.H.L., K.C.C., Y.K.Y. and R.B.L. managed participant recruitment. R.B.L. and C.S.C. supervised this work and edited the manuscript. All authors reviewed the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported in part by grant NSC98-2314-B-006-022-MY3 (to RBL) and grant NSC100-2314-B-006-048-MY3 (to SYL) from the Taiwan National Science Council, grant DOH 95-TD-M-113-055 (to RBL) from the Taiwan Department of Health, grant NHRI-EX-97-9738NI (to RBL) from the Taiwan National Health Research Institute and a grant from the National Cheng Kung University Project for Promoting Academic Excellence and Developing World Class Research Centers.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
