Abstract
Objective:
Conventional antidepressant treatments result in symptom remission in 30% of those treated for major depressive disorder, raising the need for effective adjunctive therapies. Inflammation has an established role in the pathophysiology of major depressive disorder, and minocycline has been shown to modify the immune-inflammatory processes and also reduce oxidative stress and promote neuronal growth. This double-blind, randomised, placebo-controlled trial examined adjunctive minocycline (200 mg/day, in addition to treatment as usual) for major depressive disorder. This double-blind, randomised, placebo-controlled trial investigated 200 mg/day adjunctive minocycline (in addition to treatment as usual) for major depressive disorder.
Methods:
A total of 71 adults with major depressive disorder (Diagnostic and Statistical Manual of Mental Disorders–Fourth Edition) were randomised to this 12-week trial. Outcome measures included the Montgomery–Asberg Depression Rating Scale (primary outcome), Clinical Global Impression–Improvement and Clinical Global Impression–Severity, Hamilton Anxiety Rating Scale, Quality of Life Enjoyment and Satisfaction Questionnaire, Social and Occupational Functioning Scale and the Range of Impaired Functioning Tool. The study was registered on the Australian and New Zealand Clinical Trials Register: www.anzctr.org.au, #ACTRN12612000283875.
Results:
Based on mixed-methods repeated measures analysis of variance at week 12, there was no significant difference in Montgomery–Asberg Depression Rating Scale scores between groups. However, there were significant differences, favouring the minocycline group at week 12 for Clinical Global Impression–Improvement score – effect size (95% confidence interval) = −0.62 [−1.8, −0.3], p = 0.02; Quality of Life Enjoyment and Satisfaction Questionnaire score – effect size (confidence interval) = −0.12 [0.0, 0.2], p < 0.001; and Social and Occupational Functioning Scale and the Range of Impaired Functioning Tool score – 0.79 [−4.5, −1.4], p < 0.001. These effects remained at follow-up (week 16), and Patient Global Impression also became significant, effect size (confidence interval) = 0.57 [−1.7, −0.4], p = 0.017.
Conclusion:
While the primary outcome was not significant, the improvements in other comprehensive clinical measures suggest that minocycline may be a useful adjunct to improve global experience, functioning and quality of life in people with major depressive disorder. Further studies are warranted to confirm the potential of this accessible agent to optimise treatment outcomes.
Introduction
Major depressive disorder (MDD) is a leading cause of disability worldwide with severe impact on productivity and quality of life. Conventional treatments that target monoamines, in particular serotonin and noradrenaline, provide inadequate remission rates of around 30% on average (Rush et al., 2006). The Royal Australian and New Zealand College of Psychiatrist guidelines for mood disorders support the use of adjunctive therapies (Malhi et al., 2015). Adjuncts are needed to enhance standard treatments by focusing on pathways not usually targeted by standard therapy. The inflammatory hypothesis of depression was first proposed almost 20 years ago (Maes, 1995). It incorporates the monoamine hypothesis with other biological factors and suggests that depression arises from increased immune activation (Maes et al., 2013), oxidative and nitrosative stress (Bilici et al., 2001; Maes et al., 2000) and alteration of the kynurenine pathway, leading to changes in monoamine levels, neuronal growth and neuroprotective factors characteristic of depression. Antidepressants enhance neuronal growth and critically blocking neurogenesis eliminates the antidepressant effects of known antidepressants (O’Leary and Cryan, 2014).
The development of novel therapies that target the above-mentioned biological pathways is supported by observations that depression is accompanied by increased levels of pro-inflammatory cytokines, e.g., cyclo-oxygenase-2 (COX-2) expression (Maes et al., 2013). Consequently, COX-2 inhibitors are also of interest as adjunctive treatments in depression (Faridhosseini et al., 2014). Similarly, agents with immune effects relevant to the pathophysiology of depression such as infliximab, celecoxib, curcumin, aspirin, statins and N-acetylcysteine (Berk et al., 2013) also have potential antidepressant properties – supported by recent meta-analyses (Fernandes et al., 2016; Kohler et al., 2014).
Minocycline is a tetracycline antibiotic that has been used clinically for long-term acne treatment (usually dosed at 100 mg/day) but has gained significant interest in psychiatry because of its mechanism independent of its microbial action (Goulden et al., 1996). Preclinical studies have shown that minocycline can inhibit microglial activation, leading to reductions in inflammation and oxidative stress (Yrjanheikki et al., 1998). In various models, minocycline reduces interleukin-1β (IL-1β) (Homsi et al., 2009), prostaglandin-E2 (PGE2) and inhibits COX-2 expression in a dose-dependent manner (Kim et al., 2004). Increases in tumour necrosis factor-alpha (TNFα), catalase and malondialdehyde levels are also attenuated by minocycline treatment (Dean et al., 2012). Minocycline has also been shown to significantly increase B-cell lymphoma-2 (Bcl-2) RNA expression both alone and in the presence of oxidative stress (Kernt et al., 2010) and to reverse decreased neurogenesis in preclinical models (Kim et al., 2009). Furthermore, in an interferon-α (IFN-α) model, minocycline blocked the depressogenic effects of immune-inflammatory activation and reversed inhibited proliferation of neural stem cells (Zheng et al., 2015). Minocycline reduced swim latency in the Porsolt swim test, both alone and in combination with antidepressants in rats (Molina-Hernandez et al., 2008). Low doses of both current antidepressants and glutamate antagonists in combination with sub-threshold doses of minocycline have antidepressant effects, suggesting that long-term adjunctive minocycline may reduce the required dose of standard antidepressants (Molina-Hernandez et al., 2008).
There is mounting clinical evidence of minocycline’s potential benefits in the treatment of psychiatric disorders (Dean et al., 2012). Minocycline has been trialled in open-label studies (150 mg/day) with efficacy reported in schizophrenia and psychotic depression (Dean et al., 2012) and more recently in autism (1.4 mg/kg/day) (Ghaleiha et al., 2016). A meta-analysis found that adjunctive minocycline, at doses similar to the current trial (ranging from 50–200 mg/day), benefited negative symptoms in schizophrenia (Oya et al., 2014).
There are case reports of significant amelioration of depressive symptoms with minocycline, and recurrence on discontinuation (Dean et al., 2012). Furthermore, an open-label study of psychotic depression showed that after 6 weeks of minocycline treatment (150 mg/day), significant improvement in depressive symptoms were reported on the Hamilton Depression Rating Scale (Miyaoka et al., 2012). Good tolerability and low adverse effects profiles, even at the relatively high dose of minocycline treatment (300 mg/day), were also reported. We report the findings of a 12-week multi-site randomised controlled clinical trial of 200 mg/day of minocycline as an adjunctive therapy for moderate–severe depression (Dean et al., 2014).
The primary hypothesis was that 12 weeks of adjunctive minocycline treatment would be superior to placebo in alleviating the symptoms of depression, measured using the Montgomery–Asberg Depression Rating Scale (MADRS). Secondary hypotheses were that 12 weeks of adjunctive minocycline treatment would reduce other symptom measures and improve overall quality of life and functioning with sustained improvement 4 weeks following treatment discontinuation.
Methods
Study design
This study was a randomised controlled trial of 200 mg/day of minocycline compared with placebo (parallel groups) over 12 weeks of treatment. A follow-up visit was conducted 4 weeks following the end of the treatment phase. The study was conducted at (1) Barwon Health and The Geelong Clinic and (2) The Melbourne Clinic in Victoria, Australia, in collaboration with Deakin University and The University of Melbourne. A third trial site was also located in Bangkok, Thailand, in collaboration with Chulalongkorn University. The trial recruitment period was from August 2013 through until August 2015. All investigators completed Good Clinical Practice (GCP) training. Inter-rater reliability was conducted using standardised role-plays. The study was approved by the relevant Human Research Ethics Committees (also known as the Institutional Research Board [IRB]) and was conducted in accordance with GCP and International Council for Harmonisation (ICH) Guidelines. The study was registered on the Australian and New Zealand Clinical Trials Registry: #ACTRN12612000283875. The complete study protocol has been previously published (Dean et al., 2014). All participants provided written informed consent prior to commencing the study.
Participants
Participants were eligible to take part in the study if they met criteria for unipolar depression, based on Diagnostic and Statistical Manual of Mental Disorders–Fourth Edition (DSM-IV) criteria (Mini International Neuropsychiatric Interview Plus – MINI-PLUS 5); scored ⩾25 on the MADRS; and had been on stable antidepressant therapy for at least 2 weeks before randomisation (if currently receiving any therapy, participants were not required to be undergoing any therapy to take part). Furthermore, if female, of child-bearing age and sexually active, they needed to be using effective contraception (other than the contraceptive pill, which is contraindicated due to drug interactions). Participants were excluded if they had a concurrent diagnosis of bipolar disorder I, II or not otherwise specified; had three or more failed adequate trials of an antidepressant or electroconvulsive therapy for the current episode; had a known or suspected systemic medical disorder; were using the contraceptive pill without additional contraceptive measures; were pregnant or breast feeding; had tetracycline use 2 months prior to the study; were concurrently taking >5 mg beta carotene or >300 µg retinol equivalent, isotretinoin or etretinate, anticoagulants other than aspirin, methoxyflurane or penicillin; or were enrolled in another clinical trial.
Participants were withdrawn from the study if they discontinued the trial medication for 7 consecutive days, ceased effective contraception, became pregnant or withdrew consent. Participants who require tetracycline antibiotic treatment during the course of the study were withdrawn. A participant may also have been withdrawn due to adverse events at the discretion of the participant or researcher.
Investigational product
Blindness was maintained by ensuring that the packaging, appearance and colour of minocycline and placebo capsules were identical. Commercial minocycline tablets were purchased by the manufacturer (Pharmaceutical Packaging Professionals, Melbourne, Australia) and over-capsulated. Matching placebo capsules were then produced. Participants were directed to take one capsule (100 mg), twice a day, with food.
Randomisation and concealment
Participants were assigned randomly and consecutively to treatment with minocycline or placebo in a double-blind fashion. Randomisation was conducted according to Consolidated Standards of Reporting Trials (CONSORT) guidelines using block permutations. Randomisation codes were generated by a computer-based random number generator by an independent researcher and held by the central pharmacy (Barwon Health) in case unblinding was required. The investigators, participants and statistician were blind to the treatment allocation. The trial medications were dispensed by the trial pharmacist, and returned trial medication was audited.
Outcome measures
Demographic data were collected at baseline and included age, gender, weight, self-reported psychiatric history, family psychiatric history, smoking status, comorbid physical illness, current medication and comorbid psychiatric diagnosis (taken from Mini International Neuropsychiatric Interview [MINI]). Adverse events were recorded at each visit and were posed as open-ended questions about any issues with the trial or the study medication.
Validated measures of depressive symptomatology, global clinical impression, quality of life and functional status were completed at baseline and at weeks 4, 8, 12 and 16 (after discontinuation of treatment). These scales were all validated in Thai, allowing congruence between sites. Efficacy measures included the MADRS (primary outcome), the Clinical Global Impression–Improvement (CGI-I) and Clinical Global Impression–Severity (CGI-S) scales, the Patient Global Impression (PGI) scale and the Hamilton Anxiety Rating Scale (HAM-A). Quality of life was assessed using the Quality of Life Enjoyment and Satisfaction Questionnaire–Short Form (Q-LES-Q-SF). The Social and Occupational Functioning Scale was used to measure functioning over the duration of the study. Participant safety was monitored throughout the study. Adverse events were recorded at each visit and reviewed by the Principal Investigator. Supplementary Table 1 outlines the scales, their focus and the associated references.
Biological investigations
Participants were asked to provide a blood sample as an optional addition to the study. Serum, plasma and whole blood samples were collected at baseline and at week 12 to explore changes in immune-inflammatory, oxidative stress and neurotropic factors. These investigations are ongoing and will be published elsewhere.
Statistical analysis
T-tests and chi-square tests were used to compare baseline characteristics between the intervention and control groups. Number of previous depressive episodes was collected categorically (1 = 1–5, 2 = 5–10, 3 = 10–15, 4 = 15–20 and 5 = ⩾20). Main (MADRS) and secondary outcome analyses were based on all randomised participants with at least one post-baseline observation (slightly modified intention-to-treat analysis). Mixed models, repeated measures (MMRM) analyses of variance (ANOVAs) split-plot in time were then conducted. Model parameters were estimated using generalised estimating equations (GEEs) accounting for correlation within participants with an exchangeable working correlation matrix. To assess the impact of the intervention on primary and secondary outcomes, time by treatment interactions were examined in a model that included the fixed categorical effects of treatment, visit and treatment-by-visit interaction. This statistical approach allowed control for covariates when testing for treatment group effects (created at the patient level), and also avoided biased imputation of results for those participants who did not complete their follow-up assessment. The overall p-values for intervention by follow-up interaction were reported. Follow-up by intervention interaction impacts and their 95% confidence intervals (CIs) were reported. Effect sizes (ESs) (follow-up by intervention effect) were calculated using Cohen’s d. Responder (⩽50% reduction on the MADRS) and remitters (<7 on the MADRS) were compared dichotomously using Pearson’s chi-square.
A sample size of 35 in each arm achieves 80% power to detect a difference of 2.6 ± 4.5 change in MADRS score (primary outcome) at week 12 in a repeated measurements design that assumes a compound symmetry covariance structure with a standard deviation is 4.5 for both arms and baseline–week 12 correlation of 0.5 between observations on the same subject. This is equivalent to a Cohen’s D ES of 0.55. A Type I error of 0.05 is considered for the sample size calculation. An ES of 0.4 or greater for difference in MADRS scores in randomised controlled trials of antidepressants vs placebo has been suggested as necessary to suggest clinical utility for an antidepressant used as monotherapy. A sample of 71 is therefore powered to detect moderate to large ESs and trends towards significance in areas of smaller effect.
Results
Participants were recruited and followed up between August 2013 and December 2015. A total of 144 potential participants were screened, of which 71 were randomised to receive minocycline or placebo. All participants completed at least one post-baseline visit. A total of 57 participants completed the 12-week treatment phase and 55 completed the 4-week follow-up assessment. The CONSORT flow chart (Figure 1) illustrates participant flow. The capsule audit revealed an adherence rate of 87.9%; however, 32.4% of participants did not return empty capsule bottles (and therefore were not audited).
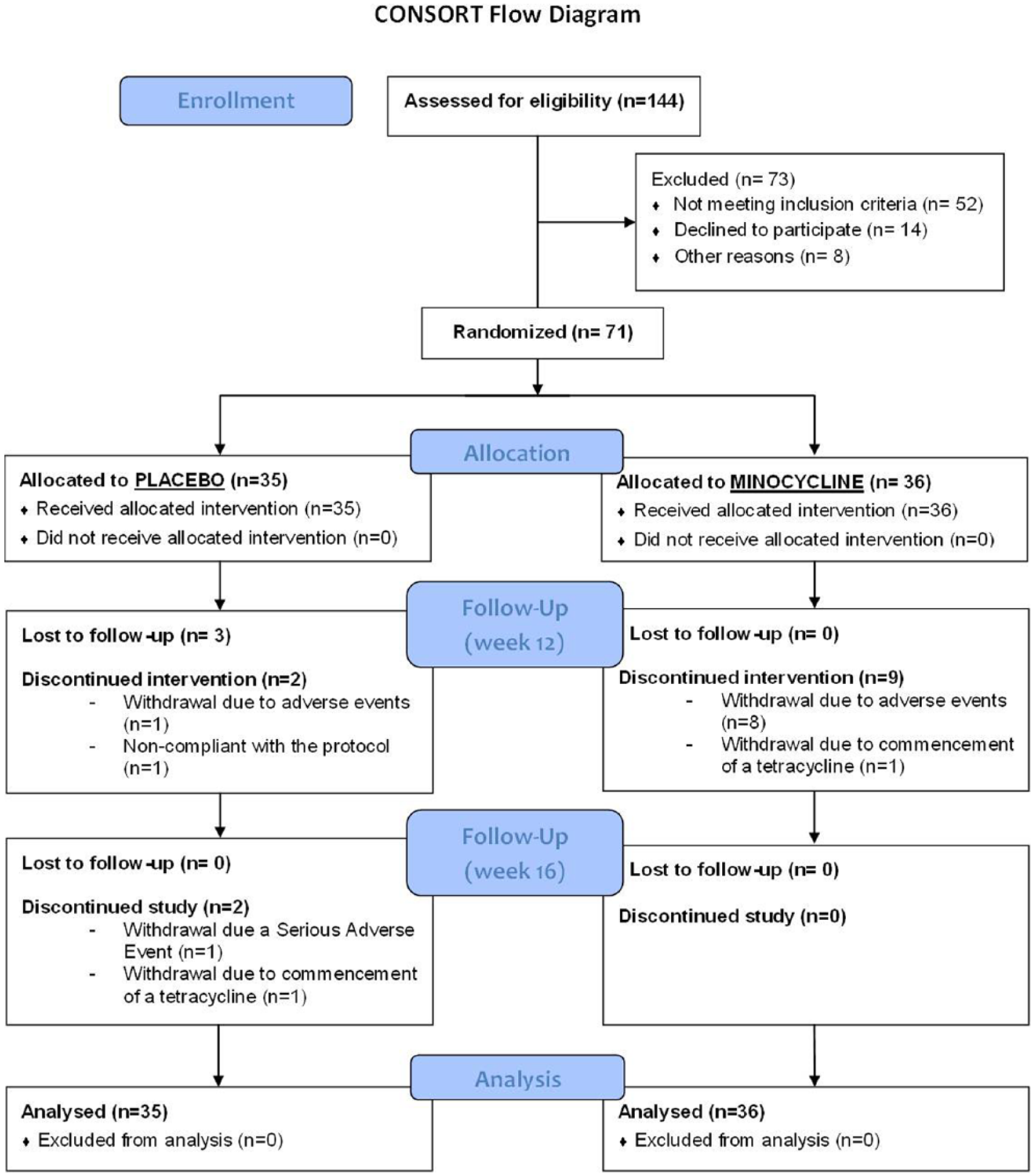
CONSORT diagram.
Participants did not differ significantly on any baseline demographics except gender, with both groups having significantly more women than men (reflecting the incidence of the disorder in the population). There were no differences between minocycline or placebo groups on baseline medication. The cohort had moderate to severe depressive symptoms with baseline MADRS scores of 31.7 (±4.0) and 31.0 (±4.6) in the placebo and minocycline groups, respectively, and a mean duration of illness (since diagnosis) of approximately 14 years suggesting a chronic course of illness. Similarly, approximately 73% of the sample met criteria for melancholic depression. The rates of comorbid anxiety disorders were high (approximately 42%), and are the most common comorbid disorder seen in this cohort. Rates of substance use, including tobacco and alcohol, were less than 5%. Table 1 demonstrates the characteristics of the sample.
Baseline characteristic of the sample (N = 71).
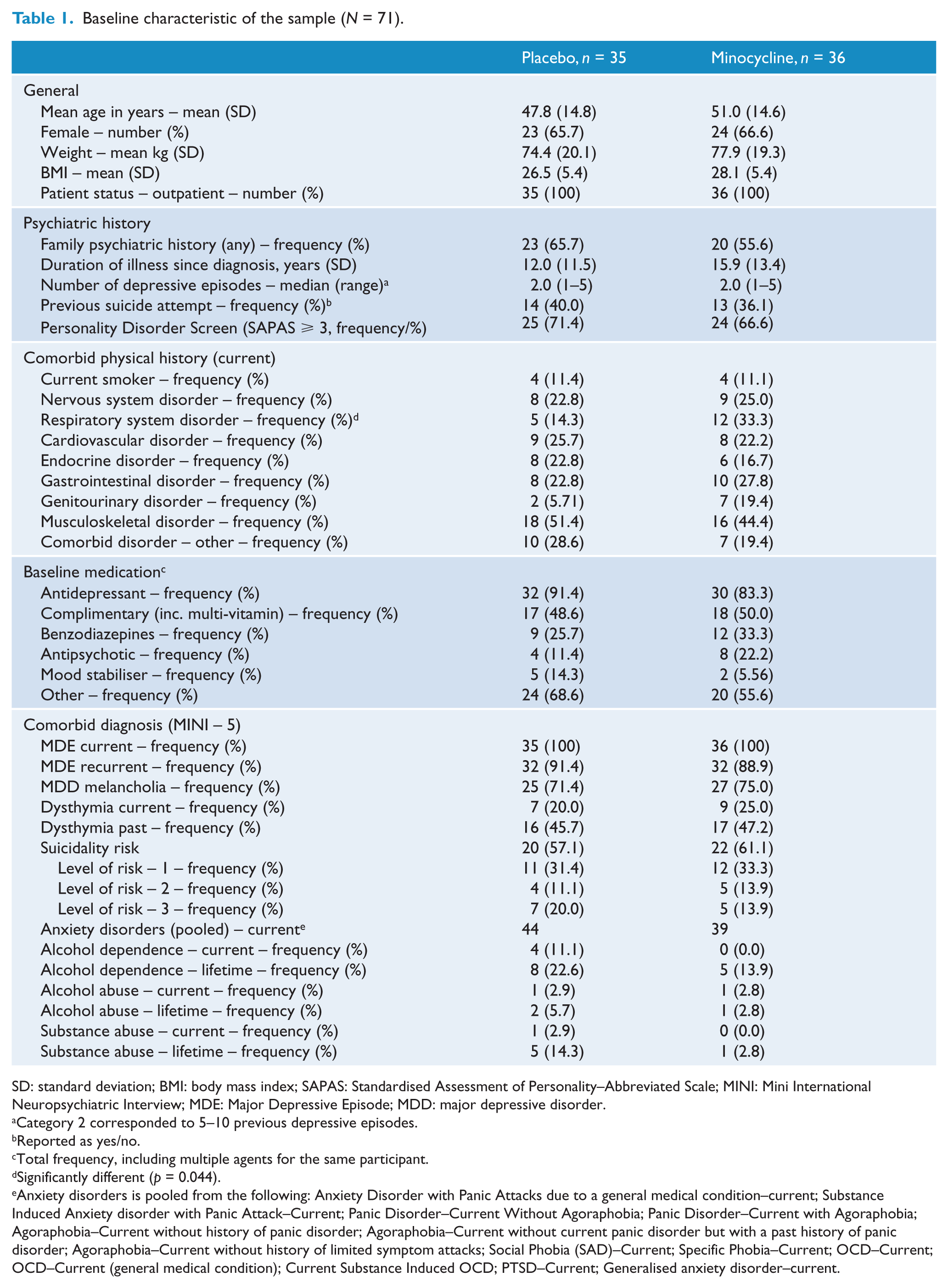
SD: standard deviation; BMI: body mass index; SAPAS: Standardised Assessment of Personality–Abbreviated Scale; MINI: Mini International Neuropsychiatric Interview; MDE: Major Depressive Episode; MDD: major depressive disorder.
Category 2 corresponded to 5–10 previous depressive episodes.
Reported as yes/no.
Total frequency, including multiple agents for the same participant.
Significantly different (p = 0.044).
Anxiety disorders is pooled from the following: Anxiety Disorder with Panic Attacks due to a general medical condition–current; Substance Induced Anxiety disorder with Panic Attack–Current; Panic Disorder–Current Without Agoraphobia; Panic Disorder–Current with Agoraphobia; Agoraphobia–Current without history of panic disorder; Agoraphobia–Current without current panic disorder but with a past history of panic disorder; Agoraphobia–Current without history of limited symptom attacks; Social Phobia (SAD)–Current; Specific Phobia–Current; OCD–Current; OCD–Current (general medical condition); Current Substance Induced OCD; PTSD–Current; Generalised anxiety disorder–current.
Based on the MMRM analyses (see Tables 2 and 3), there were no significant differences in the change in the primary outcome, MADRS scores, Figure 2(a), across the 12 weeks of treatment between the minocycline and placebo groups, ES (95% CI) = 0.46 [−7.1, 3.2], p = 0.02. However, there was a four-point difference between groups at treatment endpoint. There were also no significant differences at week 16 on the MADRS, ES (CI) = 0.41 [−6.9, 3.5].
Primary and secondary outcome measures based on MMRM analysis – all (N = 71) – mean (SD).
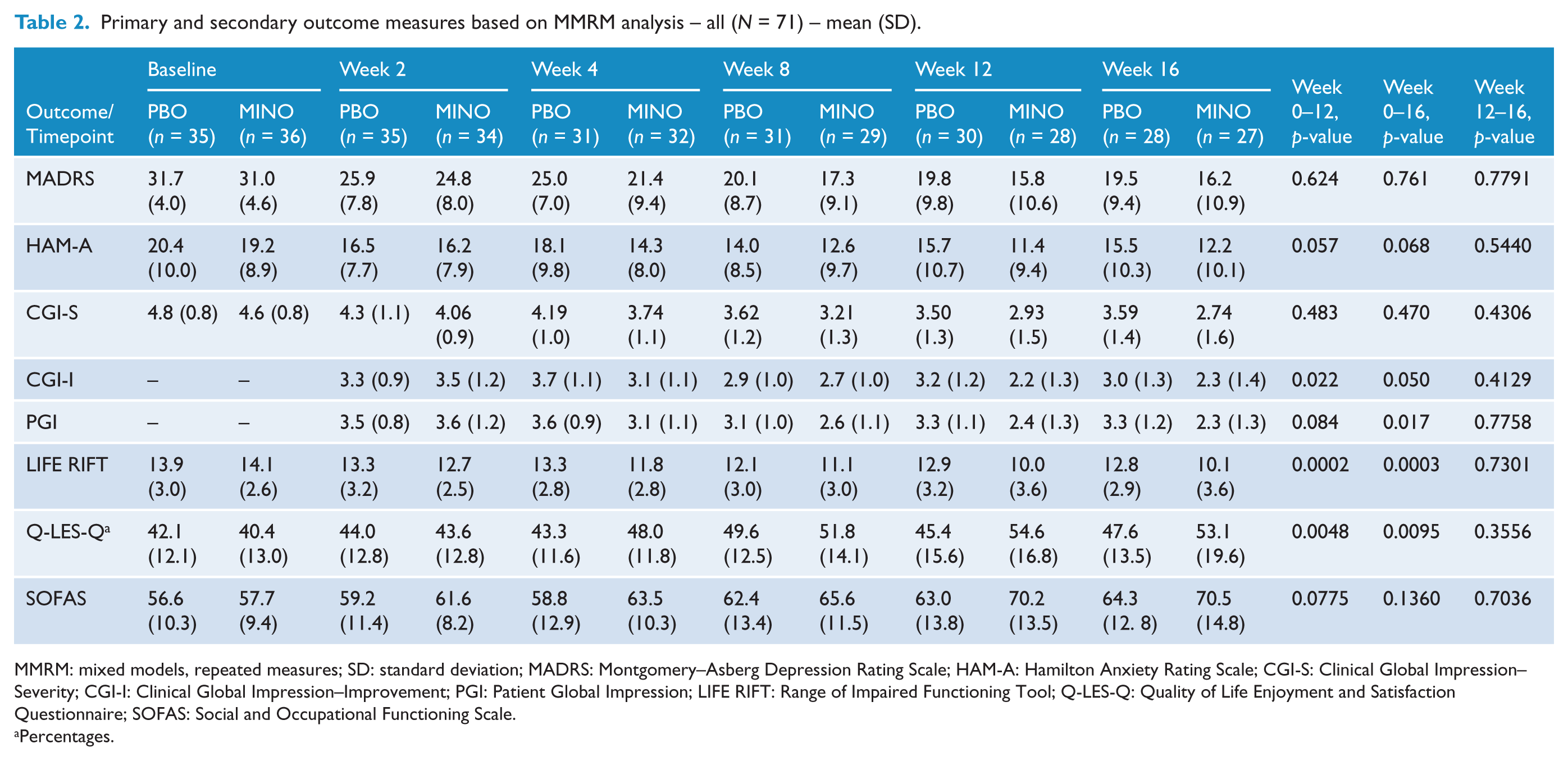
MMRM: mixed models, repeated measures; SD: standard deviation; MADRS: Montgomery–Asberg Depression Rating Scale; HAM-A: Hamilton Anxiety Rating Scale; CGI-S: Clinical Global Impression–Severity; CGI-I: Clinical Global Impression–Improvement; PGI: Patient Global Impression; LIFE RIFT: Range of Impaired Functioning Tool; Q-LES-Q: Quality of Life Enjoyment and Satisfaction Questionnaire; SOFAS: Social and Occupational Functioning Scale.
Percentages.
Confidence intervals and effect sizes on primary and secondary outcomes based on MMRM analysis.
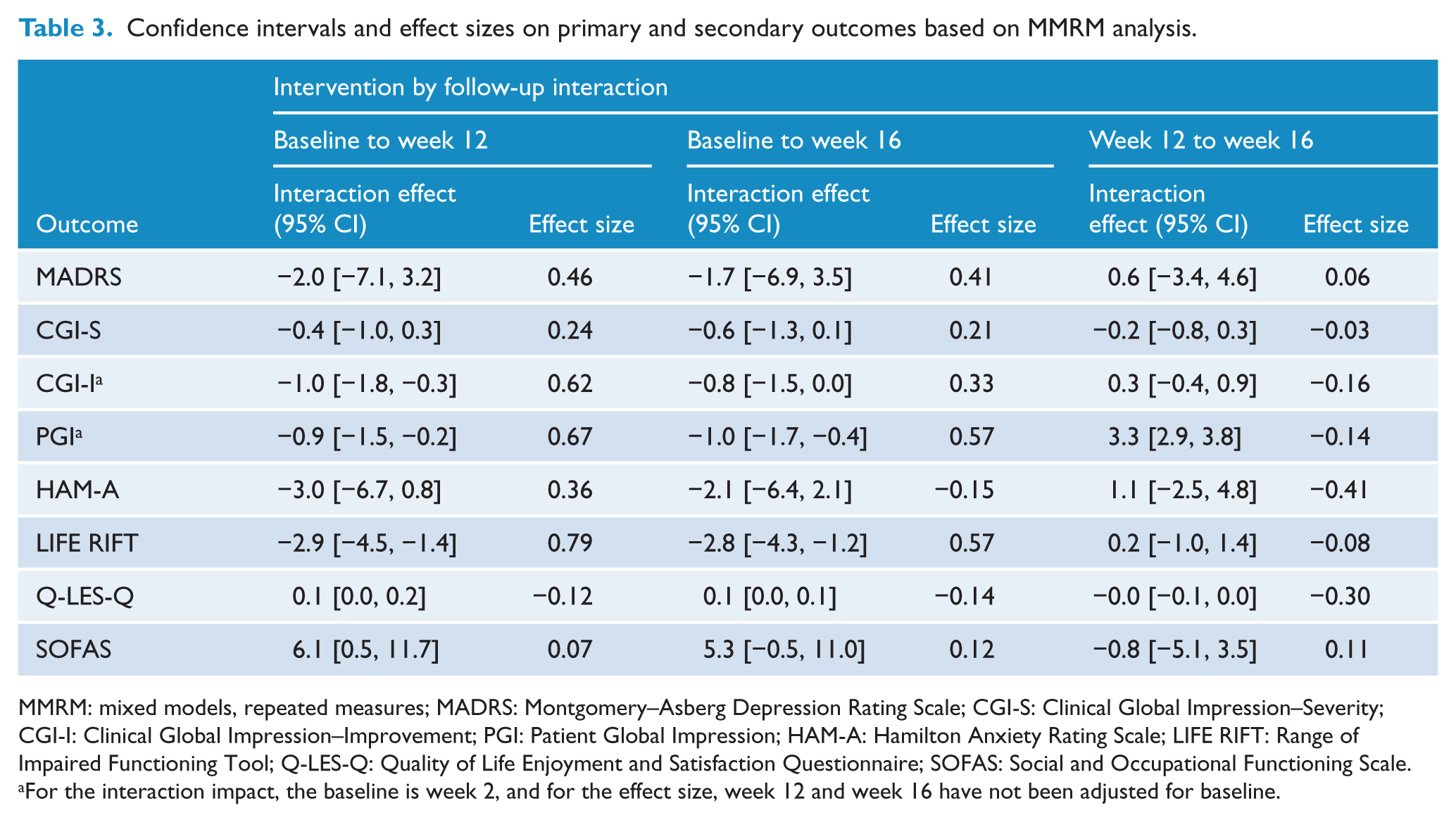
MMRM: mixed models, repeated measures; MADRS: Montgomery–Asberg Depression Rating Scale; CGI-S: Clinical Global Impression–Severity; CGI-I: Clinical Global Impression–Improvement; PGI: Patient Global Impression; HAM-A: Hamilton Anxiety Rating Scale; LIFE RIFT: Range of Impaired Functioning Tool; Q-LES-Q: Quality of Life Enjoyment and Satisfaction Questionnaire; SOFAS: Social and Occupational Functioning Scale.
For the interaction impact, the baseline is week 2, and for the effect size, week 12 and week 16 have not been adjusted for baseline.
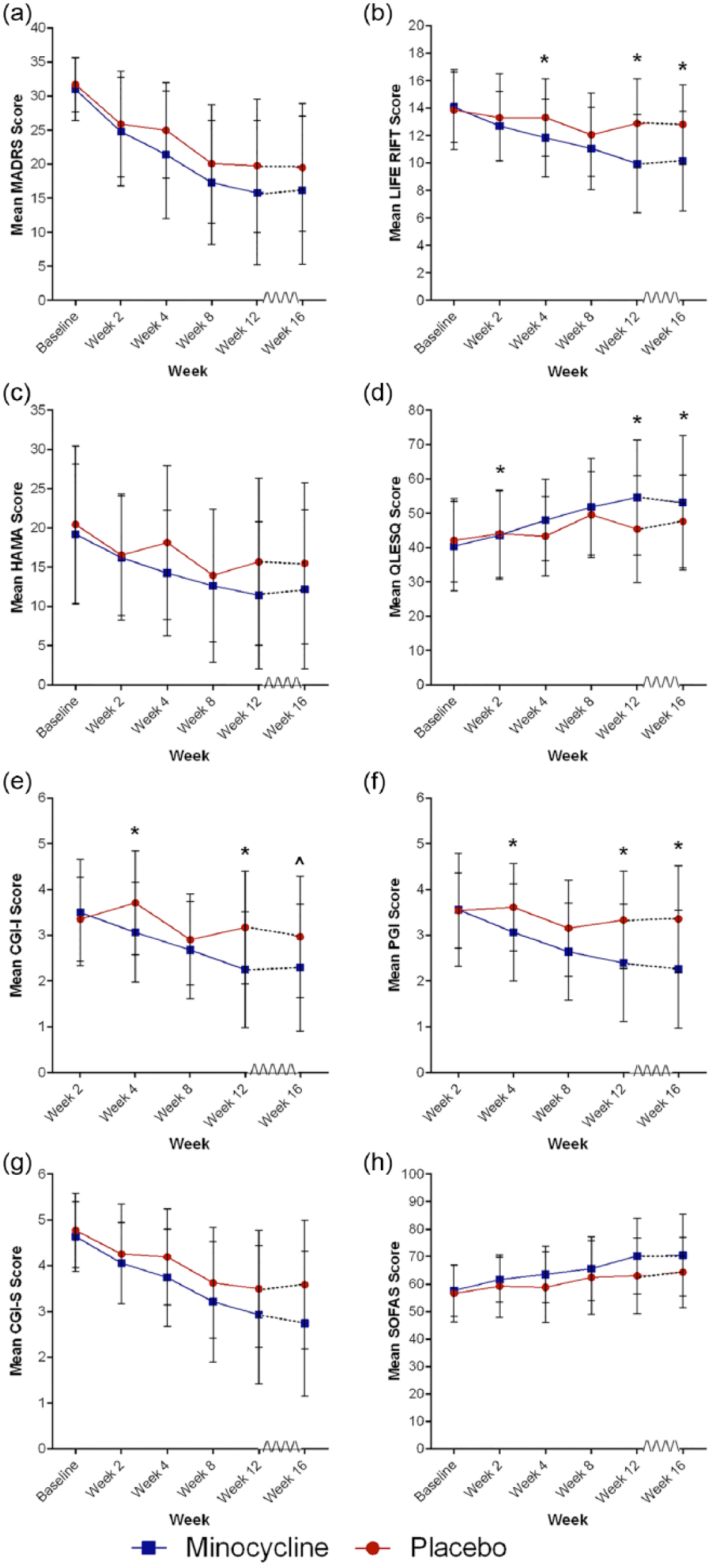
Change in outcome measures over 12 weeks of treatment and 4 weeks post-treatment discontinuation.
While not statistically significant, there was a trend towards improvements in anxiety symptoms – HAM-A, Figure 2(c) – following 12 weeks of minocycline treatment, ES (CI) = −0.36 [6.7, 0.8], p = 0.057, and 4-week follow-up phase, ES (CI) = −0.15 [−6.4, 2.1], p = 0.068. There was, however, significant improvement, ES (CI) = 0.62 [−1.8, −0.3], p = 0.022, over the 12-week treatment phase on the CGI-I, Figure 2(e), which was also maintained in the follow-up period, ES (CI) = 0.33 [−1.5, 0.0], p = 0.050. This improvement was reflected in the patient-rated PGI, Figure 2(f), improvement scale over the 16-week period, ES (CI) = −0.14 [2.9, 3.8], p = 0.017. The PGI did not reach significance at the end of the 12-week treatment phase, but did indicate a strong trend, ES (CI) = 0.57 [−1.7, −0.4], p = 0.084. Data can be viewed in Tables 2 and 3 and Figure 2.
There were highly significant improvements in functioning and quality of life. Specifically, the minocycline group showed improvements over time to week 12 and maintained at week 16 on the Range of Impaired Functioning Tool (LIFE RIFT) scale, Figure 2(b), ES (CI) = 0.79 [−4.5, −1.4], p = 0.0002 and ES (CI) = 0.57 [−4.3, −1.2], p = 0.0003, respectively. This was also seen on the Quality of Life Enjoyment and Satisfaction Questionnaire (Q-LES-Q), Figure 2(d), which was also significant over time to week 12 and week 16, ES (CI) = −0.12 [0.0, 0.2], p = 0.0048 and ES (CI) = −0.14 [0.0, 0.1], p = 0.0095. The SOFAS Figure 2(h), showed a non-significant trend towards improvements in the minocycline group over the 12-week treatment phase, ES (CI) = 0.07 [0.5, 11.7], p = 0.0775. Data are shown in Tables 2 and 3 and Figure 2.
We conducted further exploratory analyses. Due to the high number of those rated as melancholic, we also explored that subset (Tables 4 and 5) which did not show any further differences. The effect of personality disorder traits on outcomes were explored using the Standardised Assessment of Personality–Abbreviated Scale (SAPAS) (Table 1) as a mediator. There were no differences between treatment groups on the rate of personality traits, based on total SAPAS score, t(69) = −0.907, p = 0.36. A GEE model including the SAPAS as a covariate showed no significant effect of personality traits on the MADRS. There were also no significant differences between responders (⩽50% reduction) or remitters (<7) on the MADRS.
Primary and secondary outcome measures based on MMRM analysis – Melancholic Depression Subgroup only (N = 52) – mean (SD).
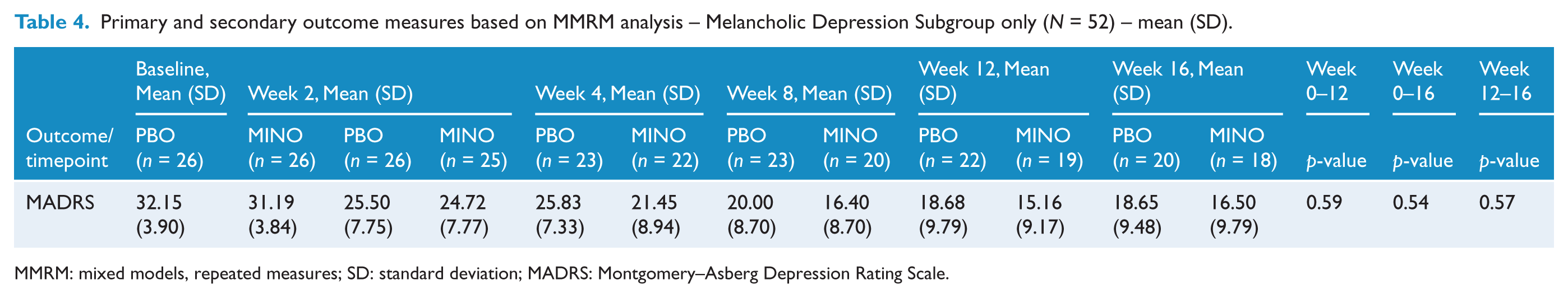
MMRM: mixed models, repeated measures; SD: standard deviation; MADRS: Montgomery–Asberg Depression Rating Scale.
Confidence intervals and effect sizes on primary and secondary outcomes based on MMRM analysis – Melancholic Depression Subgroup.

MMRM: mixed models, repeated measures; MADRS: Montgomery–Asberg Depression Rating Scale.
There were no significant difference in the total number of adverse events (p = 0.999) or on any individual events (see Supplementary Table 2) based on Mann–Whitney analysis. The most commonly reported adverse events were pain (placebo; n = 4, minocycline; n = 4), rash (placebo; n = 4, minocycline; n = 4), heartburn (placebo; n = 3, minocycline; n = 4), nausea (placebo; n = 1, minocycline; n = 5), dizziness (placebo; n = 3, minocycline; n = 3) and the common cold (placebo; n = 4, minocycline; n = 2).
Discussion
This is the first randomised controlled trial of adjunctive minocycline in MDD. While the four-point difference between minocycline and placebo in the MADRS scores (primary outcome measure) was not significant, this is on par with the magnitude of change seen with conventional antidepressants (Fournier et al., 2010). There were significant improvements in several important outcomes including global impression, functioning and quality of life, which are increasingly being recognised as more comprehensive measures of clinical outcomes. The current study was powered to achieve a moderate ES – the upper expectations of antidepressant trials. Indeed, the clinical effect indicates that with a smaller ES expectation (and therefore larger sample size), the efficacy of minocycline as an adjunctive antidepressant may be demonstrated, thus providing the necessary proof of concept for clinical utility.
The change in both clinician-rated and participant-rated global impression reflects an overall improvement, supported by the similar gains in quality of life and functioning scales. It may be postulated that the lack of effect on MADRS was due to power, or that the global improvements are being driven by changes that are not ‘core’ depressive symptoms. The high incidence of comorbid anxiety and depressive disorders (seen in 42% of the sample) further supports the need for studies of adjunctive minocycline targeting both comorbid anxiety in depression and specifically anxiety disorders. Similarly, minocycline could be driving changes in biology that were not explored in the current study (changes in energy for example) and these may account for the global improvements. It is not clear why there is a discrepancy between core depressive symptoms and overall improvements in quality of life and functioning. Further research is required to explore these effects.
Given the chronicity of the cohort (with an average duration of illness of approximately 14 years), the clinical improvement quality of life and functioning is potentially impactful. The fact that changes after only 12 weeks of minocycline treatment in a group who are receiving conventional treatment options points to the potential effectiveness of adjunctive minocycline in non-remitted individuals. It is of note that a subgroup analysis was performed finding similar results in the term of direction of the effects and intervention impact in those who are taking antidepressants (87% of the sample) when compared to the entire sample (data not shown as this was not an a priori analysis). The congruence between clinician ratings (CGI-I and LIFE RIFT) and participant ratings (PGI and Q-LES-Q) supports that improvements were felt both by the objective clinician and the subjective experience of the participant. The primacy of patient experience is congruent with the increased attention to the recovery model, and highlights the importance of patient-rated measures (Barber, 2012). Furthermore, this model of recovery highlights that improvement in functioning and quality of life are vital to people with disabling mood disorders and should not be neglected.
There are limitations to this study which may explain the negative primary outcome. While it may be a true negative finding, the sample size in the current pilot provides insufficient power to make definitive statements on the primary outcome. Future studies should consider increasing the sample size, similar to those of standard antidepressant trials. The role of personality comorbidity in depression trials is a particular concern in contemporary trials. It is always harder to demonstrate a main effect in a cohort that includes many refractory individuals, a characteristic of many contemporary trials. The fact that this study has shown improvement in this cohort is indicative of minocycline’s clinical potential. The relatively high levels of baseline depressive symptoms and rates of melancholia mean that this result is not necessarily generalisable to different cohorts of people with the heterogeneous construct of depression.
Minocycline is already approved for use in humans and is used long term for the treatment of acne. It is ‘off-patent’ and relatively inexpensive compared to current antidepressants. Therefore, this can be easily translated into treatment practice as a potentially affordable and safe adjunctive option in MDD treatment. This is further supported by our adverse events data that showed no difference over the 12 weeks of treatment. There were low rates of the most severe adverse reactions associated with typical minocycline use in adults. These include vertigo, photosensitivity and skin hyperpigmentation. Vertigo was reported by two participants in the minocycline group (compared with zero in the placebo group), and there were no reports of photosensitivity or skin hyperpigmentation. Three cases of sunburn were reported, that may be indicative of photosensitivity; however, two were reported in the placebo group (and only one in the minocycline group). Further data generated by this study, including the exploration of peripheral immune-inflammatory activation and also the mixed-methods review of case notes (both to be presented elsewhere), will augment this new knowledge, facilitating the development of next-generation antidepressant drugs, and will shed valuable new light on the pathophysiological mechanisms underpinning depression.
Footnotes
Acknowledgements
The authors would like to acknowledge the service support of Barwon Health, Deakin University, The Melbourne Clinic, The University of Melbourne and Chulalongkorn University. M.A., O.M.D. and M.B. would like to thank the Cooperative Research Centre (CRC) for Mental Health.
Declaration of Conflicting Interests
O.M.D. has received grant support from the Brain and Behavior Foundation, Simons Autism Foundation, Stanley Medical Research Institute, Deakin University, Lilly, National Health and Medical Research Council (NHMRC) and Australasian Society for Bipolar and Depressive Disorders (ASBDD)/Servier. M.A. has received grant support from ASBDD/Lundbeck and Deakin University. G.S.M. has received research support from AstraZeneca, Eli Lilly, Organon, Pfizer, Servier and Wyeth; has been a speaker for AstraZeneca, Eli Lilly, Janssen-Cilag, Lundbeck, Pfizer, Ranbaxy, Servier and Wyeth; and has been a consultant for AstraZeneca, Eli Lilly, Janssen-Cilag, Lundbeck and Servier. M.B. has received grant support from National Institutes of Health (NIH), Simons Autism Foundation, Cancer Council of Victoria, CRC for Mental Health, Stanley Medical Research Foundation, MBF, NHMRC, Beyond Blue, Geelong Medical Research Foundation, Bristol Myers Squibb, Eli Lilly, GlaxoSmithKline, Organon, Novartis, Mayne Pharma and Servier. M.B. has been a speaker for AstraZeneca, Bristol Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, Lundbeck, Merck, Pfizer, Sanofi Synthelabo, Servier, Solvay and Wyeth; and served as a consultant to AstraZeneca, Bristol Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, Lundbeck and Servier. M.B. is a co-inventor on two provisional patents regarding the use of N-acetylcysteine (NAC) and related compounds for psychiatric indications, assigned to the Mental Health Research Institute. B.K., M.M., C.H.N., M.M., L.B., A.S., S.T., A.B.S., H.M., D.J.S. and N.D. have no declarations of conflict of interest. The funders had no input into the data collection, analysis or interpretation; trial design; patient recruitment; or any aspect pertinent to the study.
Funding
The authors would like to acknowledge the funding support of the Brain and Behavior Foundation and the Australasian Society of Bipolar and Depressive Disorders/Servier. O.M.D. was supported by an Alfred Deakin Postdoctoral Research Fellowship. The study was sponsored by the Mental Health Research Institute. Funding Sources: Australasian Society for Bipolar and Depressive Disorders/Servier (Grant/Award Number: ‘ASBDD/Servier Grant’), Deakin University (Grant/Award Number: ‘Alfred Deakin Postdoctoral Research Fellowship’) and Brain and Behavior Foundation (Grant/Award Number: ‘Young Investigator Grant – 18740’).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
