Abstract
The medical use of psychedelic substances (e.g. psilocybin, ayahuasca, lysergic acid diethylamide and 3,4-methylenedioxymethamphetamine) is attracting renewed interest, driven by a pressing need for research and development of novel therapies for psychiatric disorders, as well as promising results of contemporary studies. In this Viewpoint, we reflect upon the ‘Clinical Memorandum on Psychedelics’ recently released by the Royal Australian and New Zealand College of Psychiatrists and note subsequent developments including the application for down-scheduling of psilocybin and 3,4-methylenedioxymethamphetamine presently being considered by the Therapeutic Goods Administration and approvals for access via the Special Access Scheme. We suggest that this field is worthy of rigorous research to assess potential benefits, address safety parameters and clarify therapeutic mechanisms. To this end, we outline recent research findings, provide an overview of current knowledge relating to mechanisms of action and discuss salient aspects of the psychedelic-assisted psychotherapy treatment model. The sum of this research points towards medicinal psychedelics as a potential new class of psychiatric treatments when used within a medically supervised framework with integrated psychotherapeutic support. However, before widespread translation into clinical use can occur, appropriately designed and sufficiently powered trials are required to detect both potential positive and negative outcomes. Unique safety and regulatory challenges also need to be addressed. As for any new medical therapy, psychedelic research needs to be conducted in a rigorous manner, through the dispassionate lens of scientific enquiry. Carte blanche availability to practitioners, without specific protocols and appropriate training, would be potentially harmful to individuals and detrimental to the field.
Keywords
Introduction
Following an initial phase of research and subsequent decline between the 1950s and 1970s, the medical use of psychedelic substances is seeing renewed academic and media attention in Australia. This has been driven, inter alia, by changing attitudes following the legalisation of medicinal cannabis; an increasing number of international research studies reporting promising results; and the continuing unmet medical need relating to various psychiatric conditions including major depressive disorder (MDD), generalized anxiety disorder (GAD), alcohol and drug use disorders, and post-traumatic stress disorder (PTSD) (Nutt et al., 2020; Reiff et al., 2020).
There are around 100 clinical trials using psychedelics currently being conducted globally, focused primarily on the ‘classic’ psychedelics, namely psilocybin, ayahuasca and lysergic acid diethylamide (LSD), as well as the entactogen 3,4-methylenedioxymethamphetamine (MDMA). Initial results from these studies indicate that psychedelics may provide a new class of treatments for mood, anxiety and substance use disorders (Luoma et al., 2020).
In this context, the Royal Australian and New Zealand College of Psychiatrists (RANZCP) published a ‘Clinical Memorandum on Psychedelics’ in May 2020. The memorandum notes limited but emerging evidence of therapeutic benefit for a range of mental illnesses, with further research required to assess efficacy, effectiveness and safety. Psychological support delivered by researchers with appropriate training is also stated to be an essential component of these treatments. The memorandum further notes that these drugs are currently ‘illicit substances’ that cannot be prescribed or administered outside approved research trials (although potential access via the Special Access Scheme [SAS] is acknowledged).
In this Viewpoint, we reflect upon the RANZCP memorandum by providing an overview of three substances, namely psilocybin, ayahuasca and MDMA, framing this discussion within the context of a psychedelic-assisted psychotherapy model. We consider current evidence pertinent to potential clinical use, as well as unique regulatory challenges. These include access via the Therapeutic Goods Administration’s (TGA) SAS and the current application under consideration for down-scheduling of MDMA and psilocybin. We also make several recommendations to reduce risks associated with possible access via the SAS pathway. Microdosing of classical psychedelics such as psilocybin is beyond the scope of this paper, as is the use of ketamine for depression.
Current evidence
There have been numerous systematic reviews and meta-analyses summarising outcomes from clinical trials involving psychedelic compounds for psychiatric disorders. These report evidence of short- and long-term benefits relating to mood, depression, anxiety, PTSD and substance use, as well as a satisfactory safety profile; however, small sample sizes and other methodological limitations are also noted (see Table 1).
Recent meta-analyses and systematic reviews of psychedelic substances.
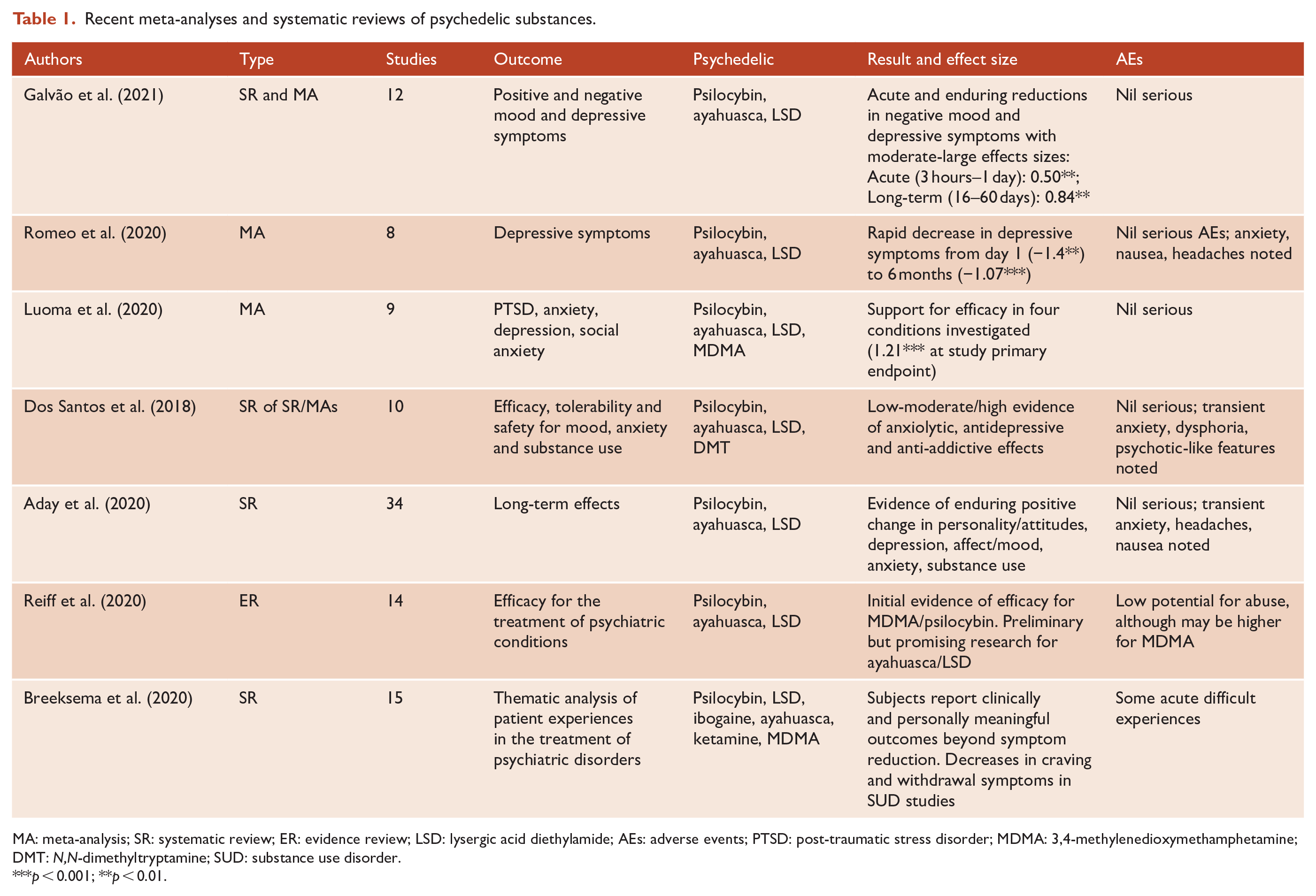
MA: meta-analysis; SR: systematic review; ER: evidence review; LSD: lysergic acid diethylamide; AEs: adverse events; PTSD: post-traumatic stress disorder; MDMA: 3,4-methylenedioxymethamphetamine; DMT: N,N-dimethyltryptamine; SUD: substance use disorder.
p < 0.001; **p < 0.01.
Psilocybin is the most researched of all of the classic psychedelics, with evidence for reduction of both depression and anxiety symptoms in patients (Nutt et al., 2020). A recent meta-analysis of three placebo-controlled studies (n = 117) of psilocybin for depression calculated large and statistically significant effects sizes (Gs = 0.82–0.83) (Goldberg et al., 2020). On the basis of such evidence, psilocybin has been granted Breakthrough Therapy status by the US Food and Drug Administration (FDA) for further investigation in treatment-resistant depression and MDD. Other recent studies using psilocybin have reported substantial abstinence rates when used for tobacco and alcohol addiction treatment (Aday et al., 2020).
Recent systematic reviews of ayahuasca (a traditional Amazonian decoction containing N,N-dimethyltryptamine [DMT] and harmala alkaloids) reported consistent evidence of anxiolytic, antidepressant and anti-addictive effects in human and animal studies, including open-label and randomised controlled trials for treatment-resistant depression (Dos Santos and Hallak, 2019). Insights into long-term effects are enabled by analysis of studies of the use of ayahuasca by various syncretic religious groups, which show no evidence of adverse neuropsychological impacts; apparent positive effects on mood and well-being; and reductions in problematic use of other substances of abuse (Dos Santos and Hallak, 2019).
MDMA-assisted psychotherapy for PTSD was granted Breakthrough Therapy status by the FDA in 2017, with Phase 3 trials slated for completion in 2022. Interim analysis of Phase 3 data conducted by an independent data monitoring committee suggests a greater than 90% probability of a statistically significant result. If confirmed, FDA approval of MDMA as a prescription drug may occur as early as 2023. On the strength of these results, the FDA recently approved early access for 50 patients outside clinical trials via an Expanded Access Program. A recent systematic review of the efficacy of MDMA-assisted psychotherapy for the treatment of PTSD rated the evidence as ‘moderate’ and concluded that further research is warranted (Varker et al., 2020).
In the context of careful screening, preparation, monitoring and follow-up, the studies discussed above have reported no serious adverse events over (typically) long follow-up periods. However, trials conducted to date were not powered for adverse event detection, given that serious adverse events such as psychosis remain relatively uncommon. It is acknowledged that transient adverse reactions including nausea, anxiety, panic and disorientation are not uncommon with classic psychedelics (Aday et al., 2020; Reiff et al., 2020). Reviews of the clinical use of MDMA report predominantly positive acute subjective drug effects and no evidence of neurotoxicity (Sessa et al., 2019). Hallucinogen-persisting perception disorder (HPPD), which has occasionally been associated with LSD use (estimated to occur in a chronic/invasive form in 1/50,000 users), is virtually unknown with psilocybin and ayahuasca (Aday et al., 2020).
Current evidence indicates that classic psychedelics are not associated with physical dependence, abuse or withdrawal (confirmed in animal studies) and have a low level of toxicity (Reiff et al., 2020). However, as noted in the RANZCP memorandum, potential psychiatric risks for people with premorbid schizoid and paranoid traits, or a family history of schizophrenia, have been identified (Dos Santos and Hallak, 2019; Reiff et al., 2020). While absence of evidence does not extrapolate to evidence of absence, population studies have established no links between lifetime classic psychedelic use and mental illness, psychological distress or suicidality (Aday et al., 2020). MDMA, by contrast, has been found to have some potential for abuse, although lower than amphetamine and methamphetamine. However, addiction is rare and long-term follow-up in studies to date have identified no patients subsequently obtaining and self-administering illicit ecstasy (Reiff et al., 2020; Sessa et al., 2019).
The Australian Drug Harms Ranking Study provides a useful assessment of relative risk, based on a comparison of 22 drugs. Alcohol was ranked as the most harmful, with a score of 77, fentanyl scored 51, prescription opioids 30 and cannabis 17, while ecstasy and LSD/mushrooms were in the lowest five ranked drugs, scoring 7 and 5 respectively, with very similar results obtained by an equivalent UK study (Bonomo et al., 2019; Reiff et al., 2020). Risks may be further reduced by use within a psychedelic treatment framework involving only one to three administrations under strict medical supervision, with psychotherapeutic support and exclusion of high-risk individuals.
Despite these overall promising results, we are in agreement with the RANZCP memorandum that there are several reasons to remain cautious. Many studies remain limited by highly selected cohorts, small sample sizes and lack of controls, while in those that are controlled, adequate blinding is difficult to achieve due to the obvious subjective and objective effects of the drug, further complicated by high expectation of benefit (introducing expectancy bias). Some studies have attempted to address this through the use of active controls such as antihistamines, amphetamines or high-dose niacin. Statistical power is a problem for adverse events, because psychotropic changes will be seen in the majority of users but adverse events only in a small minority.
Mechanisms of action
Understanding biological pathways of action for these substances is important in elucidating the potential mechanisms that underlie their beneficial effects, as well as for predicting potential adverse events and drug interactions.
Classical psychedelics are known to induce alterations in mood and perception via a modulation of the serotonergic, dopaminergic and glutamatergic systems (Dos Santos et al., 2018). Effects on 5-HT2A receptors expressed in frontal and paralimbic brain structures (involved in emotional processing and regulation, introspection and interoception) appear to be central to these effects. It is noteworthy that the 5-HT2A receptor plays a particular role in psychosis, which may explain identified risks for individuals with, or predisposed to, psychotic disorders (Dos Santos et al., 2018; Reiff et al., 2020). For note pimavanserin, a known novel antipsychotic with evidence of efficacy in psychosis associated with Parkinson’s disease, has the opposite effect, being an inverse agonist of this receptor.
Serotonin receptor agonism has also been shown to lead to a synthesis of brain-derived neurotrophic factor (BDNF), as well as reduced amygdala and default mode network (DMN) activity (Ly et al., 2018; Romeo et al., 2020). Such effects are proposed to alleviate the dysfunctional brain network connectivity that is evident in many psychiatric and substance use disorders (Zhang and Volkow, 2019).
Changes in resting-state functional connectivity (RSFC) in the DMN have been found in patients with depression taking part in psilocybin trials, and these changes have been shown to be predictive of treatment response, with a suggestion that this is reflective of a therapeutic ‘reset’ mechanism (Aday et al., 2020; Nutt et al., 2020). Such effects are also potentially relevant for substance dependence, where abnormal patterns of RSFC in the DMN have been reported across various classes of illicit drugs and are associated with craving and relapse via impaired self-awareness, negative emotions and rumination. DMN interactions with the salience and executive control networks have also been shown to be disrupted in drug-dependent individuals (Zhang and Volkow, 2019).
Other recent research has confirmed that serotonergic psychedelics can promote both structural and functional plasticity in the prefrontal cortex, robustly increasing neuritogenesis and spinogenesis, and leading to increased synapse numbers and function. These effects, reported to come about via TrkB, mTOR and 5-HT2A signalling pathways, are again highly relevant for the treatment of depression and addiction, as these conditions have been associated with prefrontal cortex dysfunction or neuronal atrophy (Ly et al., 2018). Finally, agonism at 5-HT2C receptors appears to be relevant for both the non-addictive nature of classic psychedelics themselves (vide lorcaserin) and their ability to counteract several core addiction-related behaviours (Higgins and Fletcher, 2015).
Several additional mechanisms of action have been identified for ayahuasca, positioning it as a potential ‘multi-target’ drug. These include the activation (by DMT) of both sigma-1 and trace amine-associated receptors (TAAR), suggested to play a key role in addiction behaviours as well as being involved in depression, anxiety and fear extinction (Rodrigues et al., 2019). There is also interest in potential therapeutic effects of ayahuasca’s harmala alkaloids, with preclinical studies of harmine, for example, demonstrating antidepressant-like effects (equivalent in one pilot study to fluoxetine), which are posited to come about via astrocyte-mediated restoration of BDNF protein levels and hippocampal neurogenesis, as well as anti-addictive effects, possibly via the glutamate transporter subtype 1 (GLT-1) (Brierley and Davidson, 2012).
In contrast to the serotonergic psychedelics, the primary mechanism of action of MDMA is as an indirect serotonergic agonist, increasing serotonin release. In addition, agonism at the 5-HT1A and 5-HT1B receptors is theorised to mediate anxiolytic effects, reduced amygdala fear responses, as well as increased self-confidence, compassion and empathy. Elevated levels of oxytocin attenuate stress response and support prosocial feelings, and it appears likely from animal models that increased BDNF in the amygdala may facilitate the safe recall of usually avoided traumatic memories (Sessa et al., 2019). Consistent with this mechanism is the rationale for the use of MDMA in alcohol dependence, based not on anti-addictive pharmacological effects but on facilitating psychotherapeutic processes to treat underlying psychological trauma (Sessa et al., 2019).
Psychotherapeutic mechanisms
A range of potential psychotherapeutic mechanisms have also been proposed for psychedelic treatments, and a recent systematic review of qualitative data identified a range of common mechanisms across substances (classic psychedelics and MDMA) and various psychiatric disorders. These included gaining insights (into self, their disorder/s and its origin), altered self-perception, increased feelings of connectedness and an expanded emotional spectrum, in addition to ‘mystical’ aspects of healing (Breeksema et al., 2020). Neurologically, such effects are hypothesised to reflect a revival of emotional responsiveness with medicinal psychedelics, paralleled by functional magnetic resonance imaging (fMRI) scans showing increased amygdala responsiveness to emotional stimuli, compared with a moderation or blunting of emotional responsiveness, to both negative stimuli and more broadly, with selective serotonin reuptake inhibitors (SSRIs) (Roseman et al., 2018).
Psychedelic treatment model
Although still evolving, evidence to date indicates that the optimal treatment model for the safe and effective therapeutic use of medicinal psychedelics involves several unique features. In addition to obvious differences in dosing (usually given only once or twice) and approach (to achieve profound altered states of consciousness), this also includes an expanded and somewhat unfamiliar collection of extra-pharmacological parameters that impact clinical outcomes. These include the patient’s mental state and psychological preparation prior to administration; the experience and context of consumption during the acute (dosing) phase; and quality of psychological support and integration post consumption (see Figure 1).
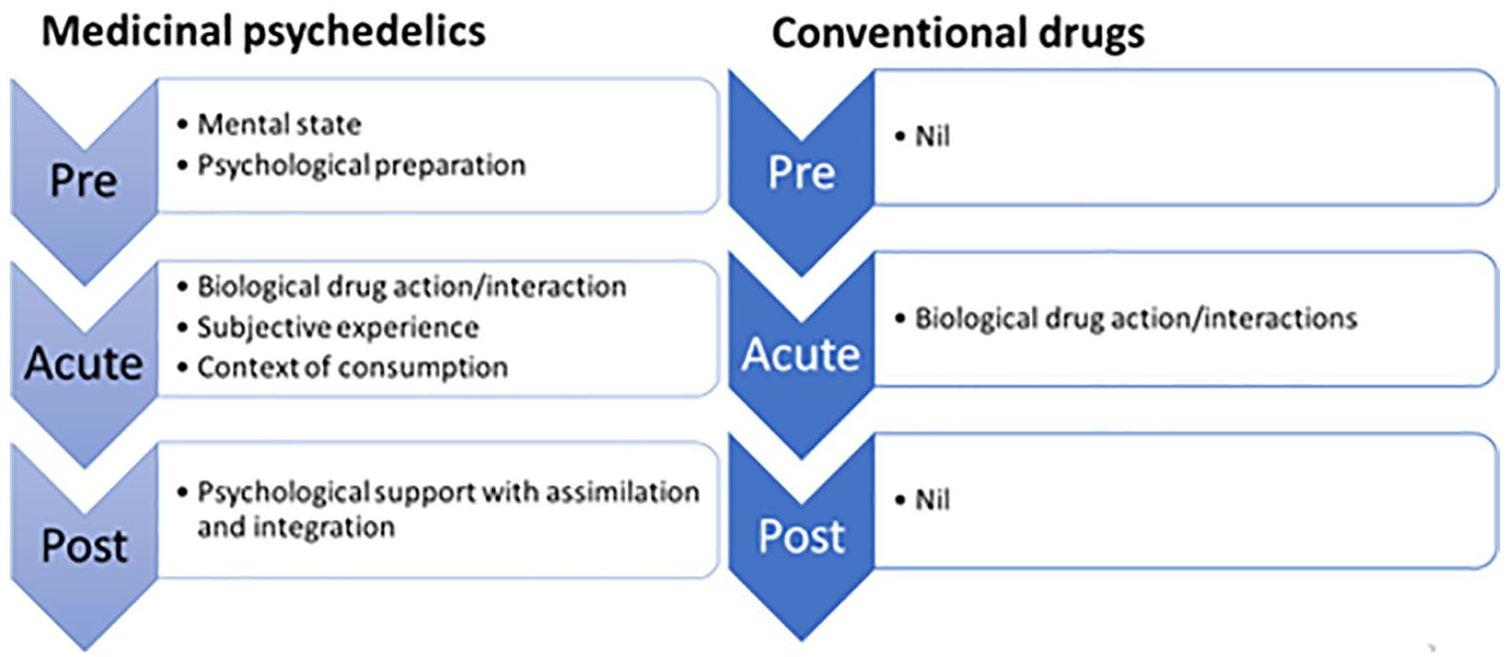
Factors affecting clinical outcomes for psychiatric conditions.
Subjective mystical experiences have been demonstrated across multiple studies to be predictive of clinical response to classic psychedelics, with one psilocybin study, for instance finding this to be predictive of therapeutic effect on depressive symptoms (Aday et al., 2020; Romeo et al., 2020). Russ et al. (2019) reported that a mental state of lower preoccupation (being occupied with daily concerns) and an increased sense of ‘surrendering’ (wilful release of goals, preferences) at the time of ingestion of psilocybin were predictive of a beneficial response. Moreover, in a challenge to some psychotherapeutic support models, it was found that exposure to words or conversation during the acute dosing session was a negative predictor of mystical experience, supporting the current focus on the use of music and controlled auditory stimuli during dosing sessions (Russ et al., 2019).
Other research has suggested negative effects associated with extremely challenging acute experiences, as well as the constructs of ‘emotional breakthrough’ (facing and resolving/releasing difficult feelings or memories) and peak experiences, which are reported to be positively associated with therapeutic outcomes (Roseman et al., 2019). Some differences in MDMA compared with classic psychedelics protocols include therapists tending to have greater engagement with the patient during the acute phase to facilitate the therapeutic process, and that the subjective experience appears to be less relevant (Sessa et al., 2019).
There remains much to be explored, but existing evidence supports the utilisation of appropriate treatment models involving suitably trained clinicians, pre-dosing preparation, acute session support and post-session therapy, to optimise clinical benefit and reduce risks of harm.
Regulation
At present, there are no psychedelic medicines that have been registered for use in Australia via the TGA’s Australian Register of Therapeutic Goods (ARTG). Indeed, psilocybin, DMT and MDMA are listed as controlled substances in Schedule 9 (S.9) of the Australian Poisons Standard. As noted in the RANZCP memorandum, it may still be possible to prescribe and administer these substances outside clinical trials via the TGA’s SAS Category B (SAS-B), and we understand that a number of such approvals have recently been granted. However, additional authorisation is also required from the relevant state or territory health department, and some jurisdictions cannot approve S.9 substances for use outside of research contexts. The SAS is intended to make treatments available on a case-by-case basis (following a medical review) where ARTG- listed medicines have not been successful, and evidence can be provided that expected clinical benefits outweigh potential risks. SAS-B can be used for medicines that may or may not be registered overseas.
While an access pathway such as SAS-B may be appropriate in judicious cases for use of medicinal psychedelics, we believe that any such approvals should include the following additional conditions: (1) that the administering clinician is a psychiatrist or addiction medicine specialist who has undertaken specific training in psychedelic-assisted therapy; (2) that the drug is administered according to a manualised treatment approach used within clinical trials (e.g. the MAPS Manual for MDMA-Assisted Psychotherapy for PTSD, or the Yale Manual for psilocybin-assisted therapy of depression); and (3) patients with a history or risk of psychosis are excluded. Previous supervised practice in psychedelic-assisted psychotherapy model would also be prudent, as would active ongoing surveillance of outcomes and caution when using these substances in patients with a prior addictive disorder (although they may sometimes be the target group).
The current application for down-scheduling of psilocybin and MDMA from Schedule 9 to Schedule 8 (declined in an interim decision) would position these substances alongside tetrahydrocannabinol (THC) containing medicines. As non-ARTG registered products, an SAS-B approval would still be required; however, state approvals for SAS-B would be easier or not necessary, depending on the jurisdiction. To note, down-scheduling these substances would also facilitate research by reducing approvals and security required for clinical research in several states.
Future directions
In sum, we concur with the RANZCP memorandum that evidence of benefit for psychedelic-assisted psychotherapy is emerging, but that further research is required. Acknowledging methodological limitations of extant studies, including small sample sizes, expectancy bias and difficulties with blinding, the promising results being obtained, with large effects sizes for benefit and a very low signal for adverse outcomes, make a strong case for further high-quality controlled studies.
However, numerous challenges exist with governments proving reluctant to fund research, and the psychedelic treatment model based on long-term remission of symptoms after one or two administrations, using existing compounds, being unattractive to big pharma. Venture capital may offer an alternative, but high levels of speculation and hype, as has been seen with medicinal cannabis, present other risks. In this context and with the growing interest in compassionate access pathways, establishing registry type data collection systems to advance knowledge relating to effectiveness and safety would be of particular benefit.
Should a body of robust research evidence demonstrate that psychedelic-assisted therapy is effective and safe, then a further crucial step will be the accreditation and regulation of clinical training and practice, to ensure provision by skilled expert clinicians. In the short term, and in the absence of ARTG-registered psychedelic medicines, it is important that any approvals for access include the additional conditions recommended above to minimise potential risks to patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: D.P., O.C., D.S. and D.H. have no conflicts to declare. N.L.G.-C. is supported by CAPES Foundation from Brazilian Ministry of Education (Research Fellowship 88887.466701/2019-00) and J.S. is supported by an NHMRC Clinical (Fellowship APP1125000). M.B. is supported by a NHMRC Senior Principal Research Fellowship (1059660 and 1156072). S.R. is supported by a NHMRC Senior Research Fellowship (GNT1154651). C.L. has received NHMRC funding for a clinical trial of ketamine in depression (1105089). C.D. is supported by an NHMRC Career Development Fellowship (1141738) and has received NHMRC funding for a clinical trial of ketamine in depression (1138736). M.W. is Executive Director of DGR-1 Health Promotion charity, Psychedelic Research in Science & Medicine Ltd. P.L. is a member of the Medical Advisory Board of Incannex Healthcare Limited.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
