Abstract
Objective:
Currently, pharmaceutical treatment options for autism spectrum disorder are limited. Brain glutaminergic dysregulation is observed in autism spectrum disorder. N-acetylcysteine, which can be converted to glutathione and subsequently release glutamate into the extracellular space, and thus reduce glutamatergic neurotransmission at synapses, is considered a potential drug for autism spectrum disorder treatment. Here, we analyzed the treatment effects of N-acetylcysteine on autism spectrum disorder in randomized controlled trials.
Study design:
Updated systematic review and meta-analysis.
Data sources:
By systematically searching the PubMed, Embase and Cochrane Library, we obtained five randomized controlled trials.
Study selection:
Meta-analyses were performed to examine the improvement in autistic behaviors as measured by the Aberrant Behavior Checklist, Social Responsiveness Scale and Repetitive Behavior Scale–Revised, using mean difference with a 95% confidence interval and a random-effects model.
Data Synthesis:
After 8–12 weeks of N-acetylcysteine supplementation, the pooled result of four trials revealed an improvement in Aberrant Behavior Checklist total score (mean difference = 1.31, 95% confidence interval = [0.42, 2.20]). When one trial was excluded, the sensitivity test result was stronger (mean difference = 1.88, 95% confidence interval = [0.92, 2.83]). The pooled results of three trials revealed significant improvements in hyperactivity (mean difference = 4.80, 95% confidence interval = [1.20, 8.40]) and irritability (mean difference = 4.07, 95% confidence interval = [1.13, 7.04]). Regarding Social Responsiveness Scale, the pooled result of two trials showed significant improvement in social awareness after 8–12 weeks of N-acetylcysteine supplementation (mean difference = 1.34, 95% confidence interval = [0.09, 2.59]). No differences were observed in the pooled results of two trials using Repetitive Behavior Scale, either in the total or the subscales.
Conclusion:
We concluded that N-acetylcysteine is safe and tolerable, reduces hyperactivity and irritability and enhances social awareness in children with autism spectrum disorder. However, further evidence should be sought before a general recommendation.
Keywords
Introduction
Autism, first described in the 1930s (Asperger, 1938) and 1940s (Kanner, 1943), is now considered one disorder named autism spectrum disorder (ASD). It is defined by two core symptoms, namely a deficit in social communication and/or interaction and the presence of repetitive behaviors and/or restricted interests (American Psychiatric Association, 2013). It was estimated to affect 7.6 per 1000 or 1 in 132 persons globally in 2010 (Baxter et al., 2015).
In their systematic review and meta-analysis, Zheng et al. (2016) reported that people with ASD had higher plasma glutamate levels than the healthy controls. A recent synthesis of several studies have shown a significant reduction of one of the most important intracellular defensive components - glutathione (GSH), and an increase of its oxidized form - glutathione disulfide, in the plasma of the patients with ASD compared with controls (Bjørklund et al., 2020). Increased glutamate concentration and subcortical GABAergic imbalance may cause neural excitotoxicity leading to insufficient inhibition of the prefrontal cortex and hyper-responsivity of the amygdala (Matte et al., 2010; Siever, 2008). Page et al. (2006) highlighted that the glutamate–glutamine ratio is high in the amygdala–hippocampus regions, whereas Horder et al. (2014) found that the subcortical glutamate–glutamine ratio was low in patients with ASD. Hence, ASD can be best described as brain glutaminergic dysregulation. Moreover, excessive oxidative stress in the brain contributes to ASD development, characterized by reduction in GSH levels (McBean, 2017) in the cerebellum and temporal cortex of patients with ASD (Chauhan et al., 2012). Consequently, researchers have begun to pay more attention to the tripartite synapse including the presynaptic and postsynaptic neurons as well as the ensheathing astrocytes that are involved in maintaining homeostasis through GSH, glutamate and glutamine regulation in ASD pathogenesis (McKenna, 2013; McKenna et al., 2012; Schousboe, 2017; Schousboe et al., 2014).
N-acetylcysteine (NAC), a derivative of the natural amino acid
Although the effectiveness of NAC in ASD has been examined in previous systematic reviews of clinical trials (Deepmala et al., 2015; Mechler et al., 2015), no meta-analysis has investigated the pooled effects of these trials. Moreover, the newly published randomized controlled trials (RCTs) have not been considered. Therefore, here, we performed an updated systematic review and meta-analysis to examine the treatment effects of NAC in ASD in RCTs.
Methods
Search strategy and study eligibility
In this study, we followed the Preferred Reporting Items for Systematic Reviews and Meta-Analysis guidelines. We performed a systematic literature search in PubMed, Embase and Cochrane Library for articles published until August 2019. This study adopted a broader search strategy using a combination of keywords including ‘acetylcysteine’ and ‘autism’ in the form of title words or medical subject headings. Clinicaltrials.gov, the official trial registration website, was also searched for any unpublished trial with available results, without language or publication period restrictions. The literature search was performed by two reviewers (T.-M.L. and K.-M.L.) independently with a third senior reviewer to resolve discrepancies through discussion and consultation (E.-W.L.). Reference lists of all retrieved articles were manually examined for possible articles meeting the inclusion criteria.
Inclusion and exclusion criteria
Our study focused on RCTs investigating the treatment effects of NAC on ASD. Therefore, we included RCTs on patients diagnosed as having ASD on the basis of Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, text revision (DSM-IV-TR) or other official diagnostic criteria. We used the following exclusion criteria: (1) ASD with a secondary diagnosis of schizophrenia, schizoaffective disorder or psychotic disorder not otherwise specified and (2) taking antioxidant agents, glutathione prodrugs or concomitant medications with glutamatergic effects before enrollment.
Assessment of risk of bias and data extraction
The risk of bias of each RCT was assessed using the Cochrane Risk of Bias tool version 2 (RoB 2.0, released on October 9, 2018). RoB 2.0 includes the following five aspects of bias: bias arising from the randomization process, deviation from intended interventions, missing outcome data, measurement of the outcomes and selection of the reported results. The same two reviewers conducted and compared the data extracted and disagreements were resolved in the manner as in the risk of bias assessment.
Outcomes of interest
Psychiatric symptoms and behavioral disturbances were the primary outcomes of this study, which were measured using the Aberrant Behavior Checklist (ABC) across the five domains of irritability, agitation and crying; lethargy/social withdrawal; stereotypic behavior; hyperactivity/noncompliance; and inappropriate speech. Social responsiveness and repetitive behavior were the secondary outcomes of this study, which were measured using the Social Responsiveness Scale (SRS) and Repetitive Behavior Scale–Revised (RBS-R). The SRS measures the domains of social awareness, cognition, communication, motivation and mannerisms, whereas the RBS-R consists of subscales measuring stereotypic, self-injurious, compulsive, ritualistic, sameness and restricted behaviors.
Data synthesis and statistical analysis
Mean difference (MD) based on the assumption of normal distribution was used to analyze continuous outcome data, and the precision of each effect size reported as a 95% confidence interval (CI). The DerSimonian and Laird random-effects model was used to compute the pooled estimate (DerSimonian and Laird, 1986). Cochran’s Q and I2 statistics were calculated to evaluate statistical heterogeneity across the included trials. Statistical significance was set at p < 0.10 for Cochran’s Q. For the ease of reporting, we tentatively classified I2 values of 25–50%, 51–75% and 76–100% as low, moderate and high, respectively.
The change of score reported by the included trials was used for meta-analyses in this study. When trials reported only the baseline and endpoints, we calculated change of score using the method recommended in the Cochrane Handbook (a correlation between baseline and endpoint measurement of 0.5 was used) (Higgins and Green, 2011). All analyses were performed on Review Manager (version 5.3; Cochrane Collaboration, Oxford, England).
Results
Literature search
Figure 1 illustrates the flow of the literature search. In the initial search, we identified 181 records totally. After duplicates were removed, 136 records remained. Of them, 91 with irrelevant contents were excluded. After close examination of the remaining records, 5 conference abstracts, 31 reviews and/or meta-analyses and 4 case reports were excluded. Finally, five RCTs were used for our meta-analysis and their characteristics are summarized in Table 1. The included trials had sample sizes ranging from 29 to 98 patients. None of them included adult patients.
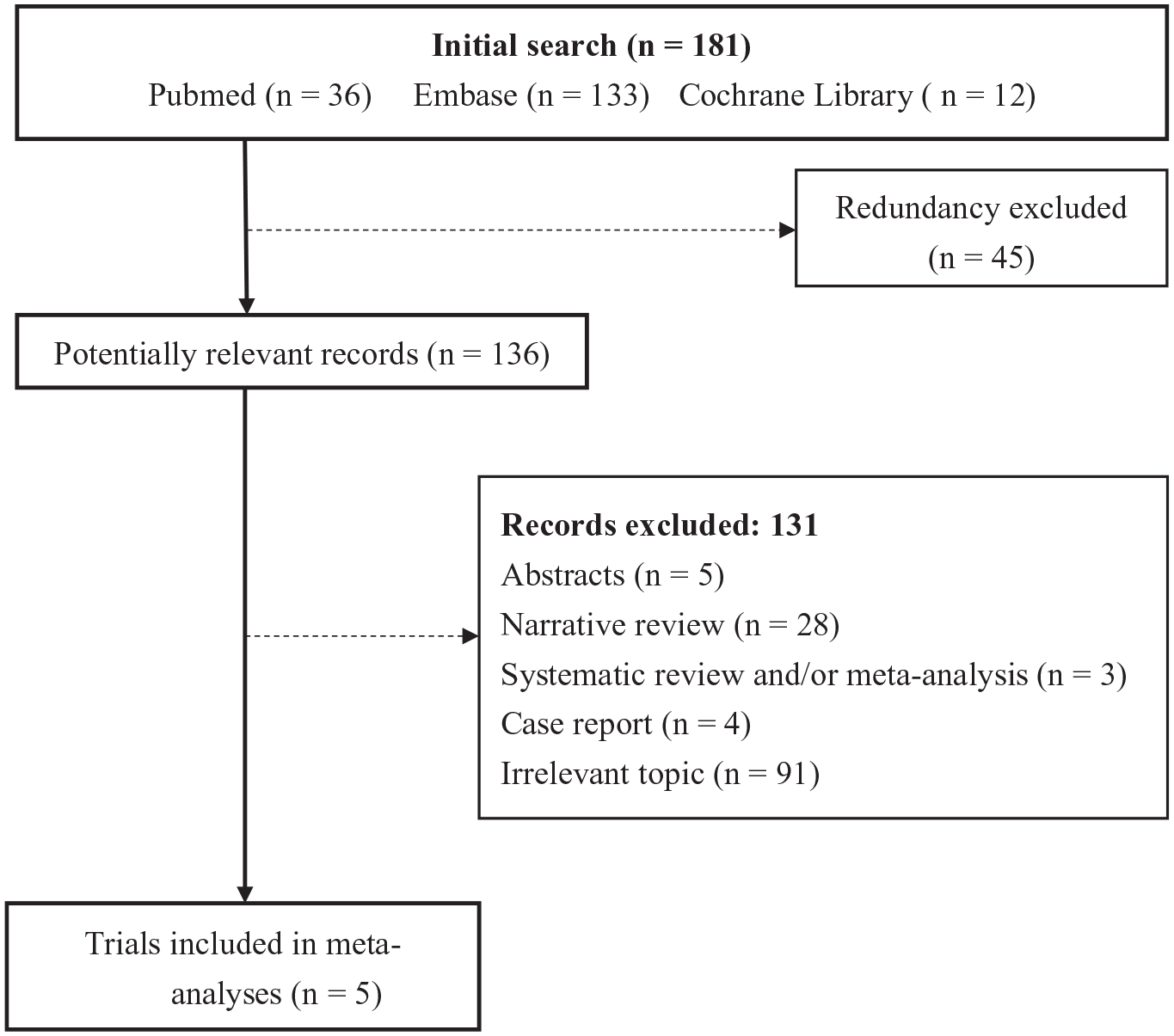
Flow of trial selection.
Characteristics of included trials.
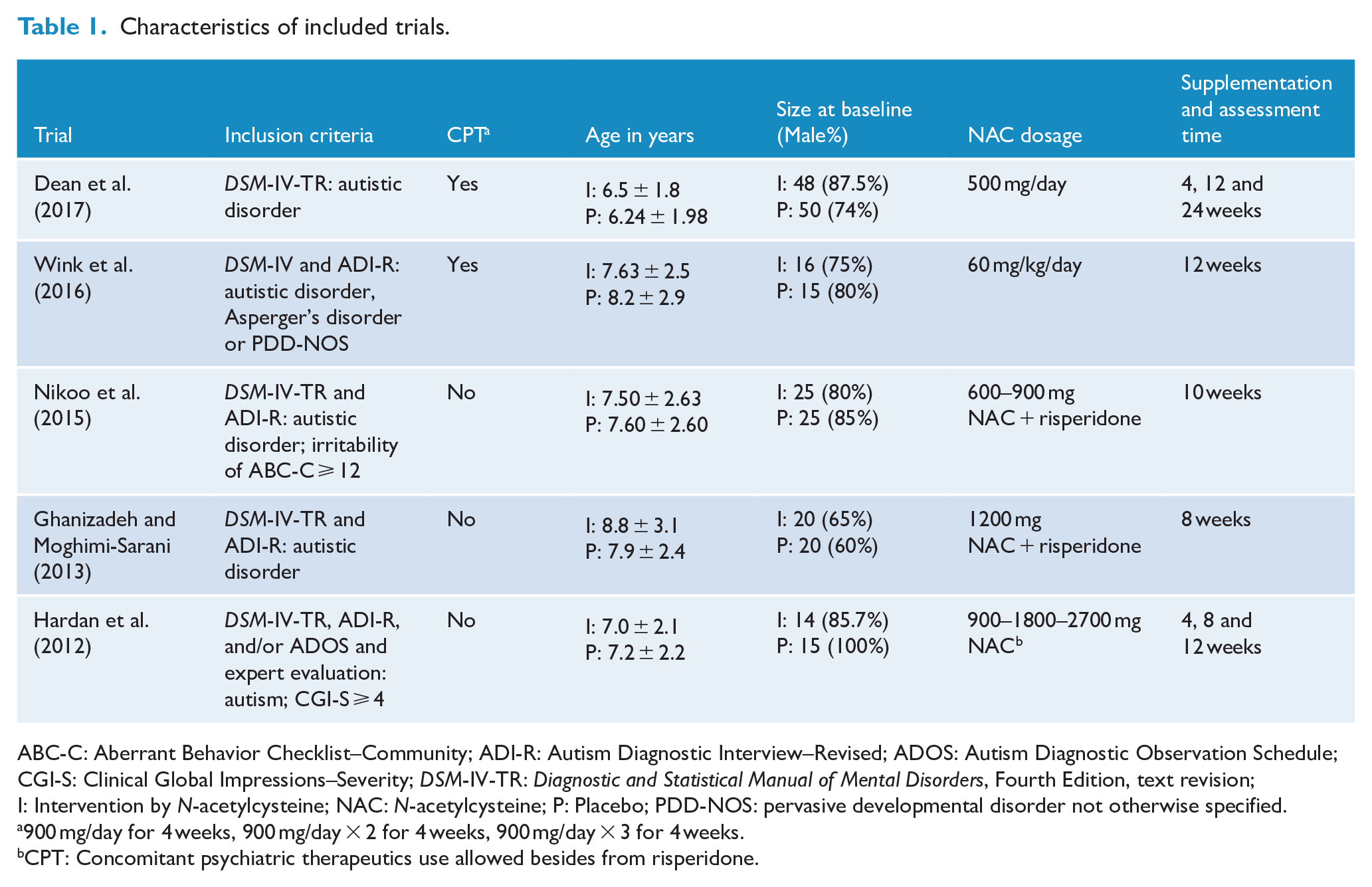
ABC-C: Aberrant Behavior Checklist–Community; ADI-R: Autism Diagnostic Interview–Revised; ADOS: Autism Diagnostic Observation Schedule; CGI-S: Clinical Global Impressions–Severity; DSM-IV-TR: Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, text revision; I: Intervention by N-acetylcysteine; NAC: N-acetylcysteine; P: Placebo; PDD-NOS: pervasive developmental disorder not otherwise specified.
900 mg/day for 4 weeks, 900 mg/day × 2 for 4 weeks, 900 mg/day × 3 for 4 weeks.
CPT: Concomitant psychiatric therapeutics use allowed besides from risperidone.
Risk of bias assessment
The results of the risk of bias assessments are summarized in Figure 2. All the included trials followed stringent research designs, including the use of proper randomization methods, concealment and blindness, and thus demonstrated a low risk of bias in the domain of randomization and the domain of deviation from the intended intervention. Although Dean et al. (2017) demonstrated a high risk of bias in missing outcome data, measurement of the outcome and some concerns in the selection of the reported result mainly because of the lack of information in the ABC, the four remaining trials demonstrated low risk in the same domains. The bias due to missing outcome data, measurement of the outcome and selection of the reported result in SRS had low risk of bias in three trials and high risk of bias in the remaining two, mainly because of the lack of information in the corresponding requirements. Three trials demonstrated a high risk in identical domains for RBS because of lack of information. Overall, of included trials, four demonstrated a high risk of bias, whereas one demonstrated a low risk of bias.

Risk of bias. Red: high risk; yellow: some concerns; green: low risk. The overall risk is graded according to the worst of the risk of bias in the domains assessed.
ABC
Four trials compared the improvement in hyperactivity, speech, irritability, lethargy and stereotypic behavior using the ABC after 8–12 weeks of NAC supplementation (Ghanizadeh and Moghimi-Sarani, 2013; Hardan et al., 2012; Nikoo et al., 2015; Wink et al., 2016). We performed two meta-analyses: one with four trials (Figure 3) and the other with three trials (Figure 4); in the three-trial meta-analysis, one trial was excluded because it recruited participants with different disease severities (Asperger’s disorder and pervasive developmental disorder-not otherwise specified) and better functions (Wink et al., 2016) and thus serves as a sensitivity test. The pooled results of the four trials demonstrated that hyperactivity (MD = 3.66, 95% CI = [−0.24, 7.57]) and irritability (MD = 3.06, 95% CI = [−0.64, 6.77]) improved after NAC supplementation, whereas speech (MD = 0.65, 95% CI = [−0.55, 1.84]), lethargy (MD = 0.72, 95% CI = [−2.06, 3.51]) and stereotypic behavior (MD = 1.27, 95% CI = [−0.55, 3.10]) (Figure 3) showed no significant improvement. Overall, the total score of ABC improved after 8–12 weeks of NAC supplementation (MD = 1.31, 95% CI = [0.42, 2.20]) (Figure 3). No significant heterogeneity was observed in hyperactivity (I2 = 21%, p = 0.29), speech (I2 = 0%, p = 0.50), irritability (I2 = 47%, p = 0.13), lethargy (I2 = 0%, p = 0.77) and stereotype (I2 = 0%, p = 0.74).
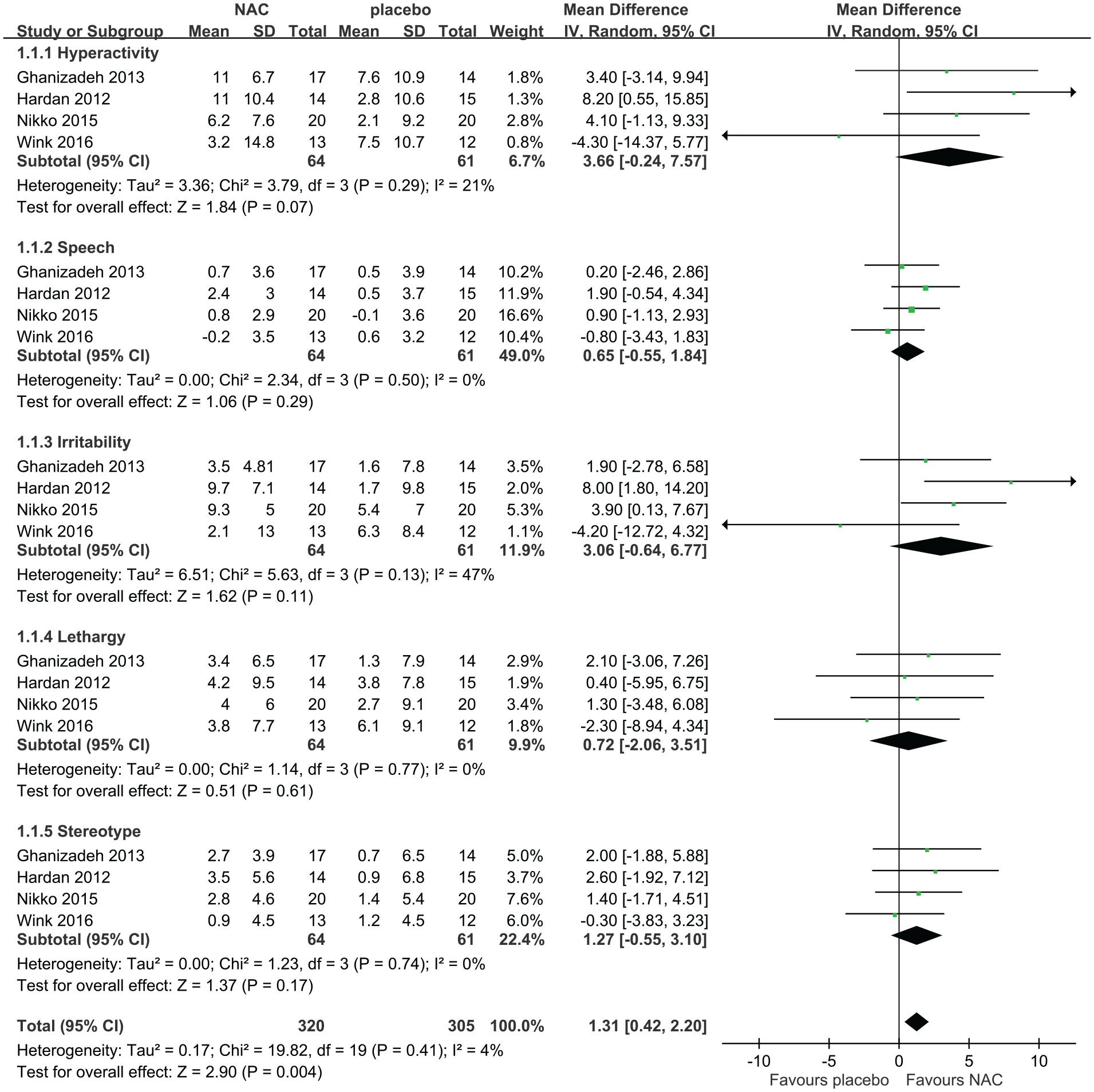
Forest plot of comparisons of four included trials in the change of score of the Aberrant Behavior Checklist after 8–12 weeks of N-acetylcysteine supplementation.
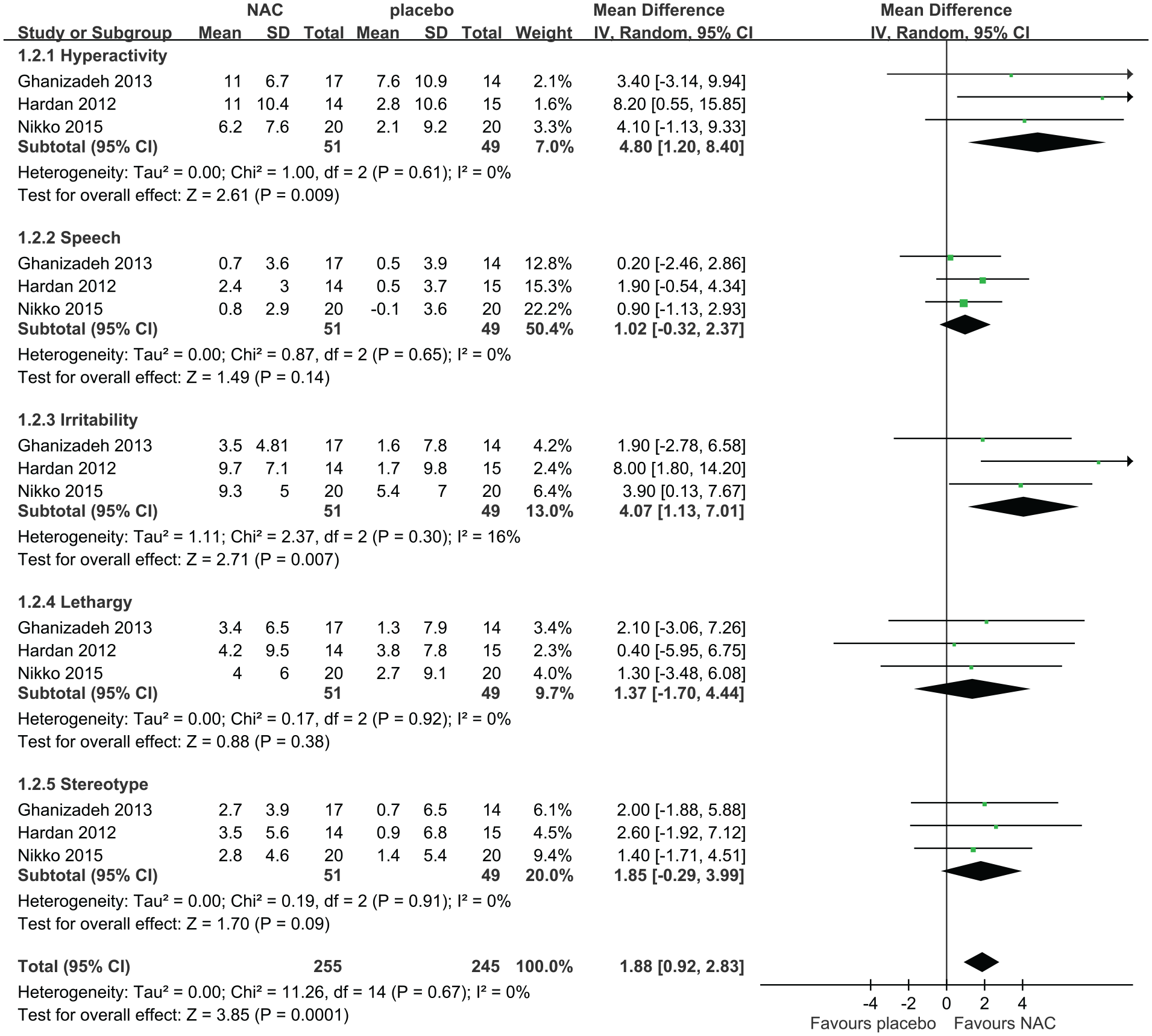
Forest plot of comparisons of three included trials (excluding one trial for sensitivity test) in the change of score of the Aberrant Behavior Checklist after 8–12 weeks of N-acetylcysteine supplementation.
After 8–12 weeks of NAC supplementation, the pooled results of three trials revealed a significant improvement in hyperactivity (MD = 4.80, 95% CI = [1.20, 8.40]) and irritability (MD = 4.07, 95% CI = [1.13, 7.01]) and a trend toward improvement in stereotype (MD = 1.85, 95% CI = [−0.29, 3.99]) but not in speech (MD = 1.02, 95% CI = [−0.32, 2.37]) and lethargy (MD = 1.37, 95% CI = [−1.70, 4.44]; Figure 4). No heterogeneity across trials was demonstrated across all subscales (hyperactivity: I2 = 0%, p = 0.61; speech: I2 = 0%, p = 0.65; irritability: I2 = 16%, p = 0.30; lethargy: I2 = 0%, p = 0.92; stereotype: I2 = 0%, p = 0.91). After 8–12 weeks of NAC supplementation, the total score of ABC improved, and the significance level was stronger than when four trials were pooled (MD = 1.88, 95% CI = [0.92, 2.83]).
SRS
All five included trials compared the effects of 8–12 weeks of NAC supplementation on SRS. Among them, three trials reported the comparison of total score, whereas two reported the comparisons of social awareness, cognition, communication, motivation and mannerism. A significant improvement in social awareness (MD = 1.34, 95% CI = [0.09, 2.59]) and a trend toward improvement in social communication (MD = 3.06, 95% CI: [−0.51, 6.62]) and in the total score (MD = 7.60, 95% CI = [−1.57, 16.77]) was observed in the pooled results. Social cognition (MD = 1.05, 95% CI = [−0.77, 2.87]), social motivation (MD = 0.25, 95% CI = [−1.76, 2.26]) and social mannerisms (MD = 2.24, 95% CI = [−1.38, 5.85]) demonstrated no significant improvement (Figure 5). Moreover, none of the analyses showed heterogeneity (total score: I2 = 0%, p = 0.82; awareness: I2 = 0%, p = 0.85; social cognition: I2 = 0%, p = 0.90; social communication: I2 = 0%, p = 0.69; social motivation: I2 = 0%, p = 0.64; social mannerisms: I2 = 48%, p = 0.17).
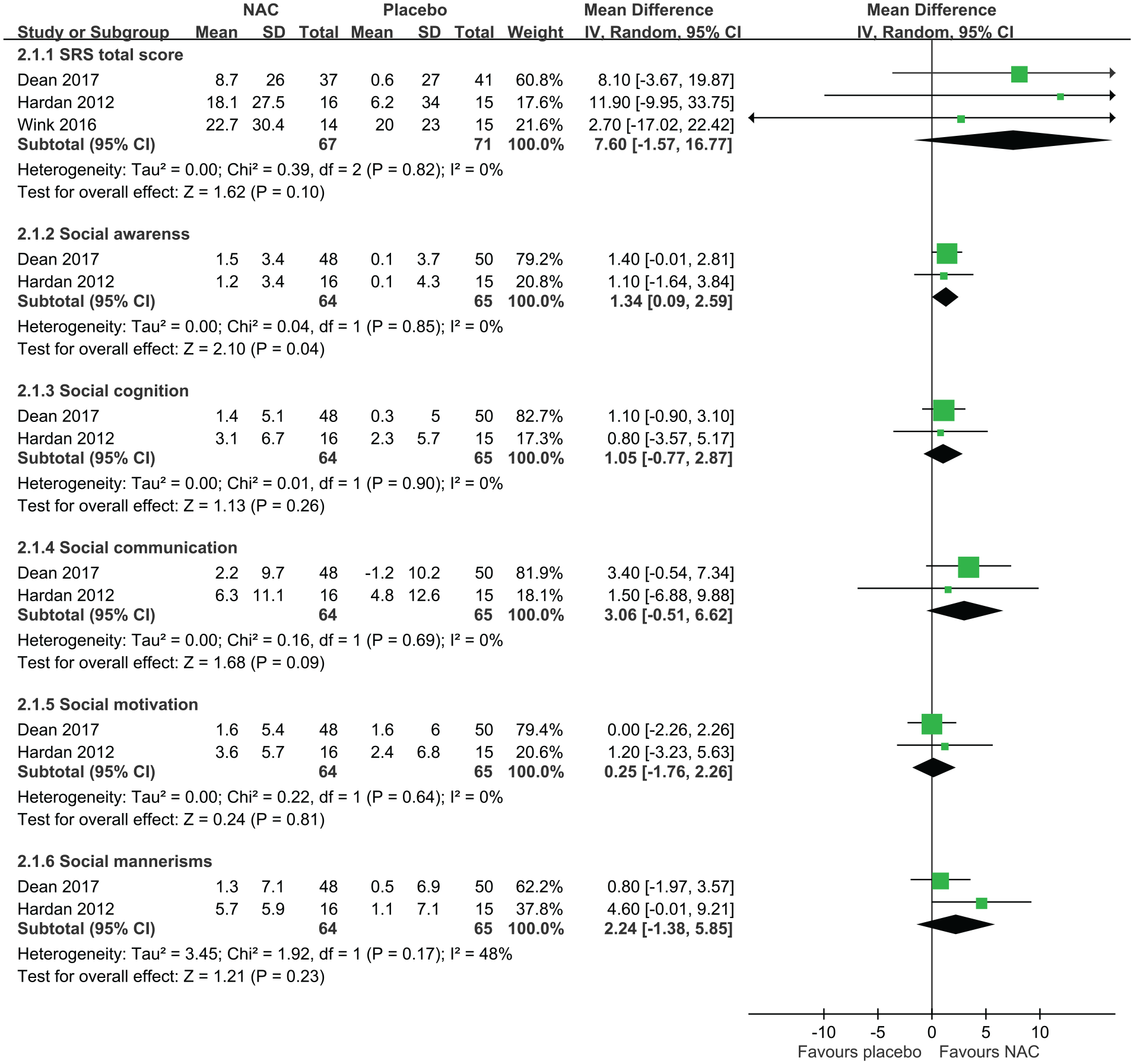
Forest plot of comparisons in the change of score of the Social Responsiveness Scale after 8–12 weeks of N-acetylcysteine supplementation.
RBS-R
The effect of 12 weeks of NAC supplementation on RBS-R was compared in two included trials. No significant improvement in stereotyped (MD = 0.08, 95% CI = [−1.30, 1.46]), self-injurious (MD = 0.72, 95% CI = [−0.41, 1.86]), compulsive (MD = 0.05, 95% CI = [−1.92, 2.01]), ritualistic (MD = −0.4, 95% CI = [−1.73, 0.92]), sameness (MD = −0.37, 95% CI = [−2.43, 1.69]) and restricted behaviors (MD = −0.26, 95% CI = [−1.31, 0.78]) was observed in the pooled results (Figure 6). Overall, no significant improvement was observed in RBS-R after 12 weeks of NAC supplementation (MD = −0.05, 95% CI = [−0.56, 0.47]). The subscales (stereotypic behaviors: I2 = 0%, p = 0.59; self-injurious behaviors: I2 = 0%, p = 0.65; compulsive behaviors: I2 = 42%, p = 0.19; ritualistic behaviors: I2 = 0%, p = 0.77; sameness behaviors: I2 = 0%, p = 0.49; restricted behaviors, p = 0.35, I2 = 0%) and the total scores (I2 = 0%, p = 0.86) demonstrated no heterogeneity.
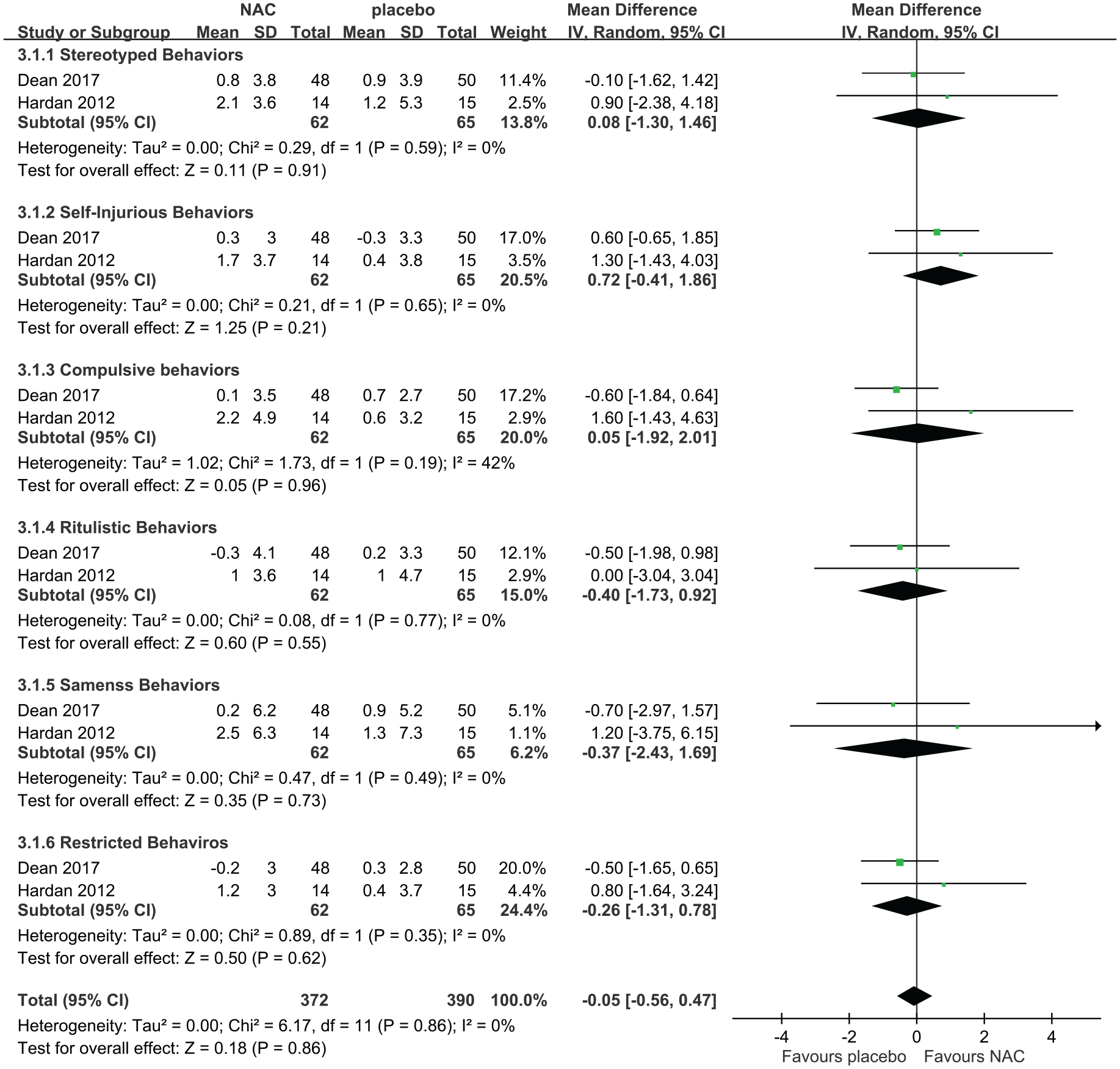
Forest plot of comparisons in the change of score of the Repetitive Behavior Scale–Revised after 8–12 weeks of N-acetylcysteine supplementation.
Other results: safety and tolerability
Adverse effects of NAC supplementation were observed in some of the patients in all the included trials. Gastrointestinal symptoms (Dean et al., 2017; Hardan et al., 2012), cold or cold-like symptoms (Dean et al., 2017), constipation and increased appetite (Ghanizadeh and Moghimi-Sarani, 2013), vomiting (Nikoo et al., 2015) and upper respiratory infection (Wink et al., 2016) were the most common adverse effects. However, none of these seemed to correlate across trials; no statistical difference in any adverse event was observed between the NAC and control groups and no fatal events were reported.
Discussion
Our study demonstrated that NAC supplementation alleviates ASD symptoms in ABC, particularly in hyperactivity and irritability. It also demonstrated a trend toward improvement in total score and social communication subscale of SRS, with a significant improvement in social awareness subscale: the ability to pick up social cues and sensory awareness of social reciprocity (Constantino and Gruber, 2005). No significant effects of NAC supplementation were observed in RBS-R. Moreover, current information suggests that NAC is safe and tolerable in children with ASD.
Although it may take long before the details of the whole picture of NAC in ASD can be clearly described, evidence from animal studies suggests that NAC helps improve ASD symptoms by (1) GSH replenishment, (2) normalization of glutaminergic activity and (3) direct action. Zhang et al. (2017) reported that NAC administration in male rats with valproate-induced autism increased GSH levels but reduced malondialdehyde levels and ameliorated the rats’ repetitive and stereotypic activity. Chen et al. (2014) showed that NAC supplementation over 10 days normalized the increased frequency of miniature excitatory postsynaptic currents and decreased the paired-pulse facilitation in the brains of rats with valproate-induced autism. Intra-amygdala infusion of mGluR2/3 antagonist blocked the effects of NAC on social interaction and anxiety-like behavior of the animals, suggesting that NAC exhibits its effects at least partially through mGluR2/3 activation. Neuronal activity governed by N-methyl-
A systematic review on the prevalence and patterns of psycho-pharmacotherapy in individuals with ASD, performed by Jobski et al. (2017), revealed the psycho-pharmacotherapy prevalence to be 2.7–80%, probably because of treatment of non-core ASD symptoms and psychiatric comorbidities. Moderate to severe irritability levels are observed in approximately 20% of patients with ASD and greater than 50% of them exhibit significant emotion dysregulation (Fung et al., 2016). Other atypical antipsychotics, such as lurasidone, may alleviate irritability in patients intolerant or refractory to risperidone and aripiprazole (McClellan et al., 2017). Other types of psychiatric therapeutics that are not approved by the US Food and Drug Administration for the purposes (e.g. methylphenidate and guanfacine) can be used in hyperactivity management in ASD; however, its adverse effects remain unclear (Stepanova et al., 2017). As per our current results, NAC may be considered an off-label drug to reduce hyperactivity and irritability and increase social awareness in children with ASD. In our study, the number of included trials was relatively small. However, a dose–response tendency was observed in the symptoms responding to the treatment; for instance, the study by Hardan et al. (2012) in which the largest dose was used reported the highest treatment effects on hyperactivity and irritability among the included trials. NAC is also far cheaper than the current standard psychiatric therapeutics. Compared with children without ASD, the mean annual age- and gender-adjusted total medical cost per child was more than threefold higher in children with ASD as demonstrated from the data analysis of the Kaiser Permanente Medical Care Program in Northern California (US$2757 vs US$892) (Croen et al., 2006). Similarly, a study using national data from the Medical Expenditure Panel Survey linked to the National Health Interview Survey in the United Kingdom demonstrated that ASD is associated with US$3020 higher health care costs and US$14,061 higher non–health care costs, including higher school costs of US$8610 (Lavelle et al., 2014). The effectiveness of NAC in ASD, albeit part of the symptoms, would be of great help to the patients, health professionals and caregivers. The low cost of this drug can be expected to lower the overall medical cost of caring for children with ASD.
Heterogeneity
This study observed no significant heterogeneity across trials in any comparison. However, underlying heterogeneity might exist in the trials because of the variation in clinical factors. First, although the main recruitment criteria for all trials were DSM-IV or DSM-IV-TR, their diagnoses and severity were inconsistent. Second, the use of concomitant psychiatric therapeutics was allowed in three trials while two trials specifically used risperidone and NAC combined in the intervention group. These might result in varied effects of treatment. Third, in each trial, the NAC dosage differed. Fourth, no trial documented whether the children with ASD received behavioral treatment during the research period.
Limitations
Our study has a few limitations. First, our meta-analysis did not have a large sample size. Second, the trials included in our study used children as participants; therefore, our findings may not be applicable to older patients of ASD. Third, the trials that we included had very short research periods. Thus, possible long-term effects remain to be elucidated.
Conclusion
NAC is safe, tolerable and effective in improving ASD’s comorbid symptoms, particularly in hyperactivity and irritability. Moreover, our study found some evidence that suggests NAC might ameliorate the core symptoms of social awareness and social communication. The design of future clinical trials is appropriately shaped with the help of our study’s summarized findings. Because of the high overall risk of bias of RCTs published thus far, additional trials with larger sample sizes, controlling for confounding effects, long-term follow-up and clearer information are warranted.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by a grant provided the Taipei Medical University, Taipei, Taiwan, for newly employed teaching staff (TMU107-AE1-B05).
