Abstract
Objective:
There is accumulating evidence that adjunctive treatment with N-acetylcysteine may be effective for schizophrenia. This study aimed to conduct a comprehensive meta-analysis examining the efficacy of randomised control trials investigating N-acetylcysteine as an adjunct treatment for schizophrenia and the first to investigate cognition as an outcome.
Methods:
We systematically reviewed Medline, EmCare, PsycINFO, Embase, CINAHL Complete, China Knowledge Resource Integrated Database and the Cochrane Clinical Trials online registry for randomised control trials of N-acetylcysteine for schizophrenia. We undertook pairwise meta-analyses of N-acetylcysteine vs placebo for psychosis symptoms and cognition.
Results:
Seven studies, including n = 220 receiving N-acetylcysteine and n = 220 receiving placebo, met inclusion criteria for the pairwise meta-analyses. Positive and Negative Syndrome Scale negative and total scores were significantly improved in the N-acetylcysteine group after 24 weeks of treatment. The cognitive domain of working memory improved with N-acetylcysteine supplementation.
Conclusion:
Evidence supports the notion that N-acetylcysteine may be a useful adjunct to standard treatment for the improvement of schizophrenia symptoms, as well as the cognitive domain of working memory. Treatment effects were observed at the later time point (⩾24 weeks), suggesting that longer interventions are required for the success of N-acetylcysteine treatment.
Introduction
Schizophrenia is a debilitating illness characterised by positive symptoms (e.g. hallucinations/delusions), negative symptoms (e.g. low motivation and drive) along with symptoms of cognitive impairment. While there has been modest success in treating the positive symptoms with antipsychotic medications (Siskind et al., 2017), no robustly efficacious pharmacological treatment for the negative or cognitive symptoms exists at this stage (Remington et al., 2016; Swartz et al., 2008). However, emerging evidence suggests that adjunct antioxidant treatment may improve these symptoms.
It has been suggested that redox dysregulation may be central to the pathophysiology of schizophrenia (Steullet et al., 2016). Schizophrenia has been associated with oxidative stress reflected in decreased concentrations of the antioxidant glutathione (GSH) in the brain (Yao et al., 2006). A novel treatment option which has found some promise in treating symptoms is a nutraceutical called N-acetylcysteine (NAC), which is a precursor to GSH (Berk et al., 2013). NAC has shown promise as an adjunct treatment for various psychiatric illnesses, and there have been numerous reviews of NAC as a treatment for psychiatry generally (Berk et al., 2013; Dean et al., 2011; Deepmala et al., 2015; Minarini et al., 2017; Sansone and Sansone, 2011; Zheng et al., 2018). In particular, Zheng et al. (2018) conducted a small meta-analytic investigation into NAC for schizophrenia in the context of psychopathology generally and found initially evidence that NAC shows promise for negative symptoms. NAC also has neurobiological effects on inflammation, glutamate signalling, mitochondrial energy generation and apoptosis, all pathways additionally dysregulated in schizophrenia (Davis et al., 2014a; Robertson et al., 2019). However, to date, there remains limited research conducted to investigate the efficacy of NAC as an adjunct treatment for schizophrenia, specifically.
Evidence also suggests a link between decreased GSH concentrations and cognitive impairment (Hajjar et al., 2018; Newton et al., 2015), and it has been proposed that NAC may improve certain cognitive domains through the increased synthesis of GSH. A recent review of the impact of NAC on cognition concluded that NAC may provide improvement, but emphasised the need for more controlled studies (Skvarc et al., 2017).
Whether NAC has a global impact on cognition based on a general reduction of oxidative stress, versus a more specific action (such as the modulation of N-methyl-
While there has been research suggesting a link between oxidative stress and schizophrenia since the 1930s (Emiliani et al., 2014), it is only in the last decade that NAC has been investigated as a possible adjunct treatment for this disorder. While there have been meta-analyses examining the impact of a range of anti-inflammatories on schizophrenia (Çakici et al., 2019), until recently, there has not been enough research in the field to conduct a systematic meta-analysis focusing solely on the impact of NAC on positive, negative and cognitive symptoms of schizophrenia. This study aimed to conduct a rigorous meta-analysis focussed exclusively on randomised control trials (RCTs) of NAC as an adjunct treatment for schizophrenia, and the first to investigate cognition as an outcome.
We hypothesised that augmentation with NAC would reduce total psychotic symptoms in longer trials (⩾24 weeks), but may not show a significant effect in shorter trials, in line with Berk et al. (2008a). We further hypothesised that NAC would improve cognitive symptoms. These hypotheses were examined with a systematic review and meta-analysis of RCTs examining NAC versus placebo for people with schizophrenia, with a primary outcome of change in negative, positive, general and total psychotic symptoms and measures of cognitive function.
Methods
Protocol and registration
This study was registered with PROSPERO, an international database of prospectively registered systematic reviews (registration number: CRD42018092097) (Booth et al., 2012). We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) recommendations for the background, search strategy, methods, results, discussion and conclusion (Higgins and Green, 2011).
Search strategy
The following databases were searched to the 14 June 2018: Medline, EmCare, PsycINFO, Embase, CINAHL Complete, the Cochrane Clinical Trials online registry and the China Knowledge Resource Integrated (CKRI) Database. Further searches were conducted of the reference lists of identified studies. Search terms included variations for the spelling of N-acetylcysteine and schizophrenia spectrum disorders. Medline search terms are provided in Supplementary Table 1.
Eligibility criteria and study selection
All randomised controlled trials comparing adjunctive NAC to a placebo for the treatment of schizophrenia or first-episode psychosis were included in the present synthesis. Other clinical conditions, such as affective disorders with psychotic features, were excluded from the synthesis. There were no restrictions on main treatment provided for the present inclusion criteria. There were no language restrictions on included studies. Studies were excluded from the search if they did not involve humans.
All studies identified in non-Chinese databases were screened at the title and abstract level, and full-text level by two authors (D.H. and C.O.Y.). E.J.T. searched the Chinese language database. Conflicts around inclusion were resolved by consulting co-authors (E.N., S.L.R. and D.S.)
We also conducted snowball searches of reference lists from key papers and the included studies. Reviews, posters, conference abstracts, case reports, letters to editors and other sources of information that did not meet the inclusion/exclusion criteria were cross-referenced for additional potential sources of RCTs. In cases where published data were missing key variables, an attempt was made to contact corresponding authors.
Data collection
Data extraction was conducted by two independent researchers (C.O.Y. and D.H.). The Chinese language paper was translated into English by E.J.T. (Zhang et al., 2015). Any discrepancies identified at any stage of the data selection process were resolved by rechecking the source papers.
Data items
Data were extracted for the following outcomes of interest: Positive and Negative Syndrome Scale (PANSS) negative, positive, general or total scores (Kay et al., 1987), and the cognitive outcomes of working memory and processing speed as data were only available for these cognitive domains. Data were extracted for two time points, first at ⩽8 weeks of treatment (six out of seven provided data for 8 weeks, the remaining paper reported at 4 weeks), and at ⩾24 weeks of treatment (one study concluded at 24 weeks, the remaining two at 52 weeks). We also collected information where possible on country, setting, diagnosis, mean age and gender balance in each group, as well as NAC daily dose, length of illness and sample size (see Table 1, section ‘Study selection’).
Included studies.
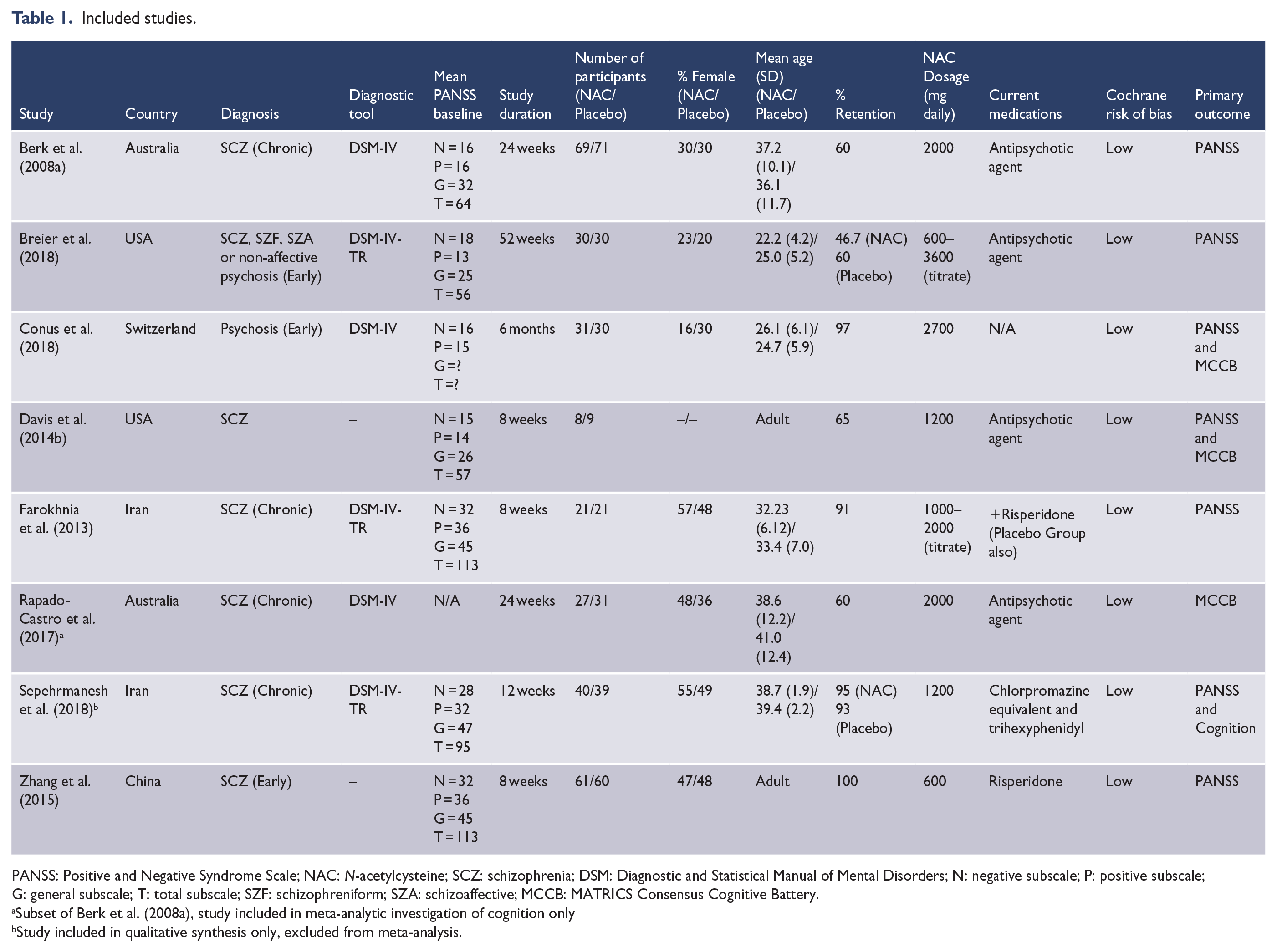
PANSS: Positive and Negative Syndrome Scale; NAC: N-acetylcysteine; SCZ: schizophrenia; DSM: Diagnostic and Statistical Manual of Mental Disorders; N: negative subscale; P: positive subscale; G: general subscale; T: total subscale; SZF: schizophreniform; SZA: schizoaffective; MCCB: MATRICS Consensus Cognitive Battery.
Subset of Berk et al. (2008a), study included in meta-analytic investigation of cognition only
Study included in qualitative synthesis only, excluded from meta-analysis.
Outcomes
The outcomes of interest were clinical measures of psychotic symptoms (including negative, positive, general and total PANSS) and cognitive performance. Studies reported either mean endpoint outcome and standard deviation, or mean change in outcome and standard deviation. Endpoint scores were preferred; however, if endpoint scores were not available, change scores were used.
Study quality and publication bias
Study quality was assessed using the following criteria adapted from the Cochrane Collaboration guidelines (Higgins and Green, 2011): (1) random sequence generation, (2) allocation concealment, (3) blinding of participants and personnel, (4) blinding of outcome assessment, (5) incomplete outcome data/appropriate reporting on missing data, (6) selective reporting and (7) other sources of potential bias including pharmaceutical company funding. Studies were deemed to be low-quality if they had three or fewer elements with low bias risk, while those of high-quality had four or more elements with a low-risk of bias.
Standards suggest that where more than 10 studies provide data for analysis, publication bias should be assessed using funnel plot asymmetry (Higgins and Green, 2011). As there were fewer than 10 studies included, this analysis was not conducted.
Statistical analysis
We performed pairwise meta-analyses using Review Manager version 5.3 for comparison between NAC and placebo groups. We used the standardised mean difference (SMD) for continuous data. Random effects models were used. Where standard error scores were provided, these were converted to standard deviation for quantitative synthesis. Sensitivity analysis was conducted on study quality and database source (English or non-English source), and subgroup analysis was conducted on study duration (greater or less than 8 weeks). Heterogeneity was assessed using the I2 statistic, a measure that does not depend on the number of studies in the meta-analysis and hence has greater power to detect heterogeneity when the number of studies is small. I2 provides an estimate of the proportion of variability due to heterogeneity rather than chance alone. An estimate of 50% or greater indicates possible heterogeneity, and scores of 75–100% indicate considerable heterogeneity. In addition to the previously described sensitivity analyses, we also explored heterogeneity through the effect of omitting each study in turn.
Results
Study selection
We identified 1005 unique articles (1004 in non-Chinese databases, 1 Chinese article acquired through hand search, no articles in the Chinese database). Of these, 53 studies were included at title and abstract level, with 8 being included for qualitative synthesis; 7 of these were appropriate for quantitative synthesis (meta-analysis). Sepehrmanesh et al. (2018) was excluded from quantitative synthesis due to missing data and having significantly different scores between the NAC and placebo groups at baseline. An attempt was made to contact the authors, but no further data were provided. No studies in languages other than English and Chinese met inclusion criteria. Reasons for exclusion are listed in Figure 1 (PRISMA diagram) and Supplementary Table 2 (Excluded studies). The PRISMA checklist is provided in Supplementary Table 3. Table 1 outlines the included studies, including means baseline PANSS scores for each study. The authors of one of the included studies (Davis et al., 2014b) provided us with their unpublished data.
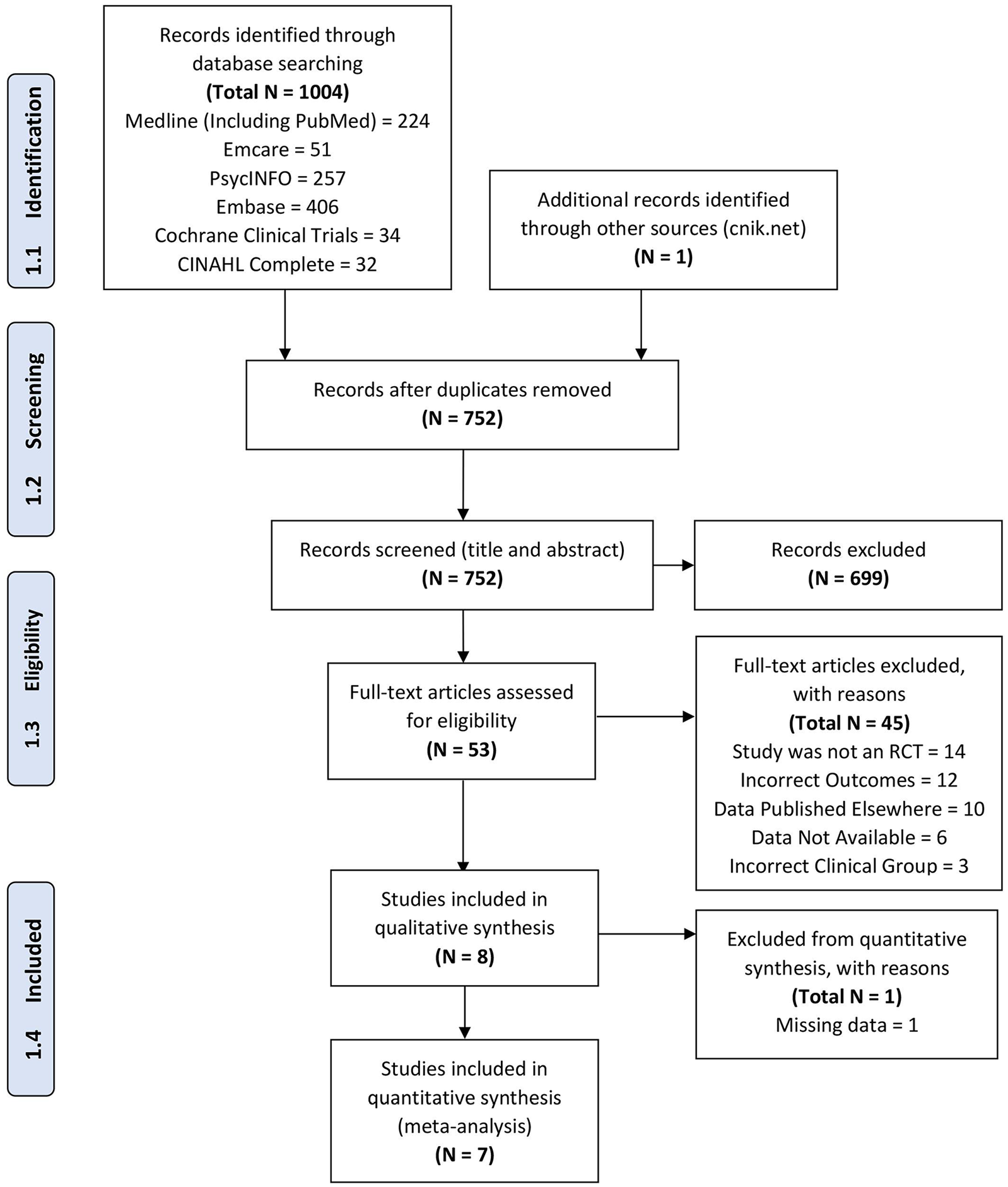
PRISMA diagram.
Study characteristics
Included studies were conducted in community settings in Australia, the United States, Switzerland, Iran and China (Table 1). Total duration of NAC intervention ranged from 8 to 52 weeks, and year of publication ranged from 2008 to 2018. There were 220 study participants in the NAC intervention groups and 220 in the placebo groups (n = 440 total). Numbers for Rapado-Castro et al. (2017) were not included in the overall count as they were a subset of Berk et al. (2008a) and included only in the cognitive component. Daily dosage of NAC ranged from 600 to 3600 mg, with two studies choosing to titrate from lower dosages. Overall, the mean illness duration across all included studies was 6.4 years (SD = 4.5), ranging from early psychosis (M = 1.3 years, SD = 1.2) (Breier et al., 2018) through to chronic schizophrenia (M = 12.1 years, SD = 9.6) (Berk et al., 2008a).
In terms of risk of bias, all studies adequately reported randomisation, but not concealment method (Supplementary Table 4). Overall, the risk of bias was low for all studies but one (Zhang et al., 2015). Sensitivity analyses of the effect of omitting each study, in turn, made little change in terms of heterogeneity estimates.
Qualitative summary of individual study outcomes
Berk et al. (2008a) was the first randomised placebo-controlled trial investigating NAC as a potential adjunct treatment for schizophrenia. This study adopted a 24-week intervention for chronic schizophrenia patients with 2000 mg/day of NAC, and reported improvement in PANSS total, negative and general subscales. A subset of this data was later analysed with specific interest in cognitive outcomes (Rapado-Castro et al., 2017). Rapado-Castro et al. (2017) found that NAC treatment resulted in improved working memory, but not attention or executive function.
Farokhnia et al. (2013) also investigated 2000 mg/day of NAC for chronic schizophrenia patients. The intervention was 8 weeks and resulted in improvement in PANSS negative and total subscales. A further smaller 8-week trial examined the effect of 1200 mg/day of NAC and reported no significant improvement for either PANSS or cognition (Davis et al., 2014b). Zhang et al. (2015) also conducted an 8-week trial, administering 600 mg/day for first-episode schizophrenia patients. The study found NAC improved both PANSS Total and Negative subscales (Zhang et al., 2015).
More recently, Conus et al. (2018) investigated early psychosis (with the exclusion of affective psychoses). Over the course of 24 weeks, participants were given 2700 mg/day of NAC. There was a significant improvement in the cognitive domain of processing speed, but not for PANSS subscales or other cognitive measures. Another study explored the effect of 3600 mg of NAC for schizophrenia patients over 52 weeks (Breier et al., 2018). This study also used the Brief Assessment of Cognition in Schizophrenia (BACS) to assess general cognitive function (Keefe et al., 2004). Improvements in PANSS total, negative and a disorganised subscale were observed in response to NAC treatment, but not cognition (Breier et al., 2018).
Finally, Sepehrmanesh et al. (2018) administered 1200 mg/day of NAC to schizophrenia patients over 12 weeks. The authors reported improvement in PANSS positive, negative, general and total subscales, and also in the cognitive domains of processing speed, working memory and executive function. However, there were several methodological issues with this study, in that PANSS scores were significantly different between the NAC and placebo groups at baseline, and the method of controlling for this difference was unclear. Furthermore, standard deviations were not provided for post-intervention time points. For these reasons, this study was not eligible for inclusion in the meta-analytic investigation below. Table 2 provides a visual summary of the outcomes of each study in response to NAC treatment.
Qualitative summary of individual study outcomes.
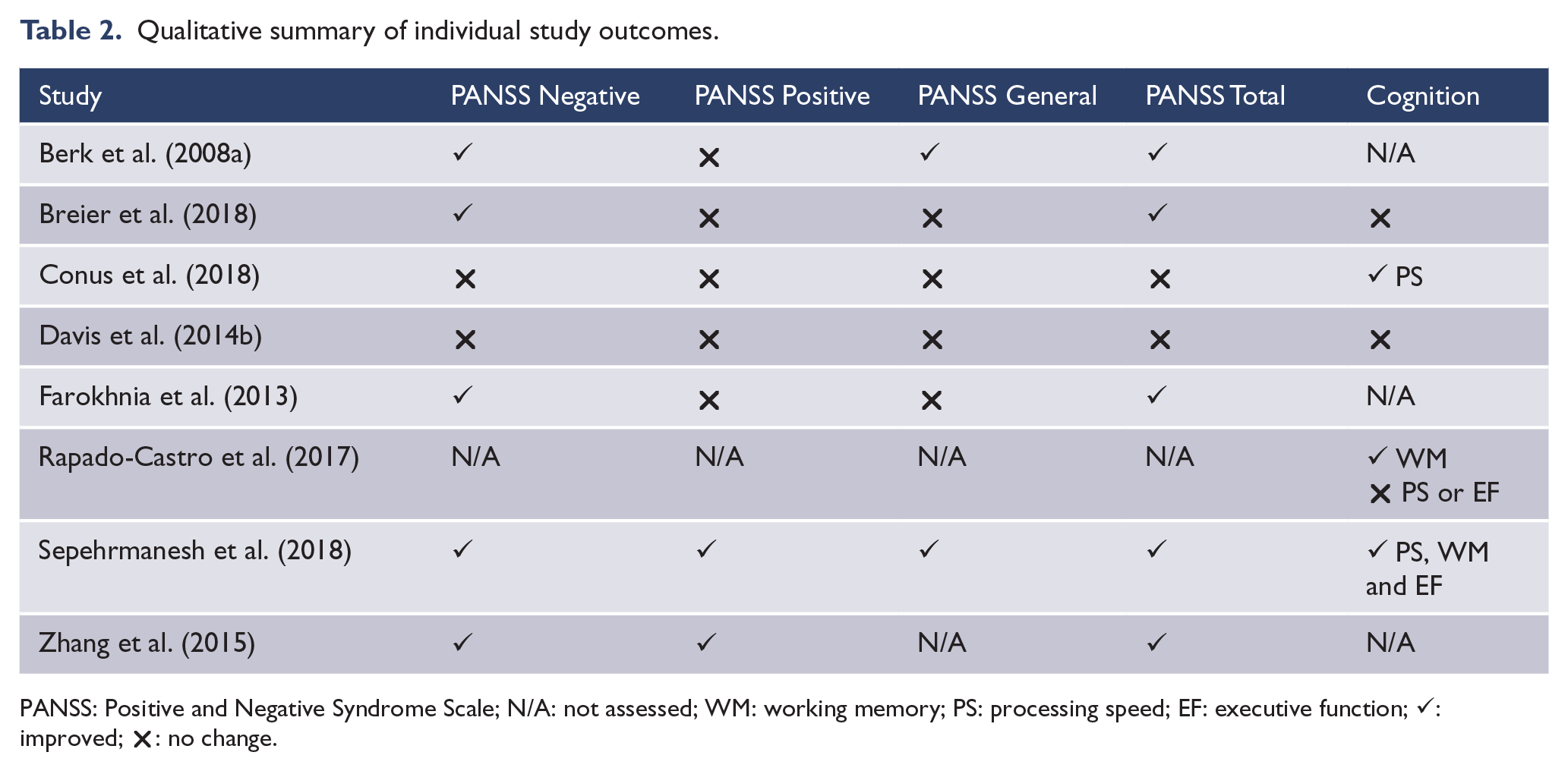
PANSS: Positive and Negative Syndrome Scale; N/A: not assessed; WM: working memory; PS: processing speed; EF: executive function; ✓: improved; ✗: no change.
Overall, from a qualitative perspective, NAC treatment seems to generally result in improvement in negative and total PANSS. Given the above, there is no immediately obvious effect of NAC dosage, illness chronicity or intervention length. The meta-analysis allows for a more in-depth quantitative investigation of the potential effect of length of intervention on study outcomes.
Meta analytic quantitative findings
Table 3 summarises the key meta-analytic findings. Improvement on symptom measures are indicated by a reduced score (i.e. negative SMD values), whereas improvement on cognitive measures are indicated by a higher score (positive SMD values).
Results of meta-analytic investigation comparing NAC vs Placebo.
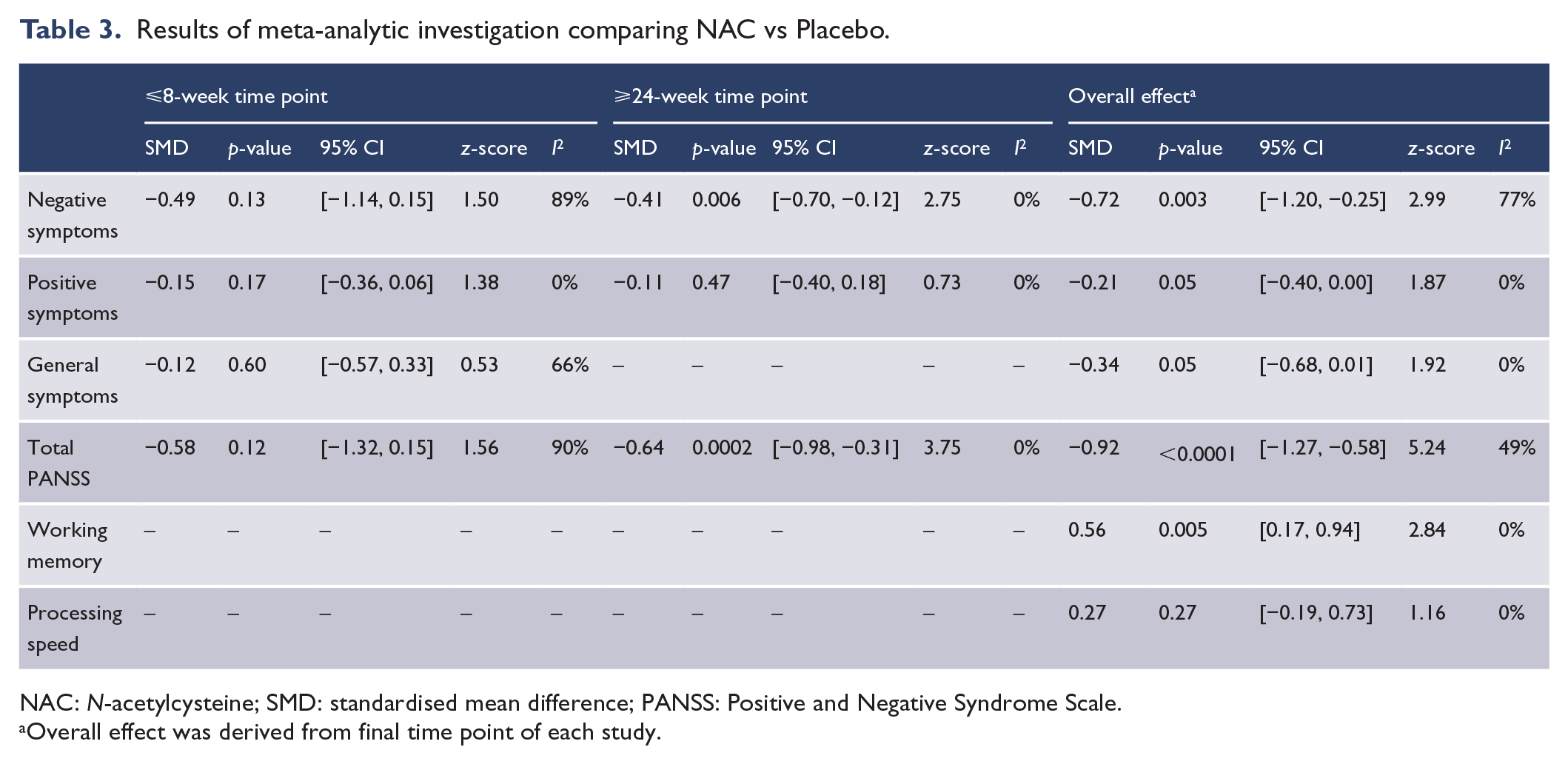
NAC: N-acetylcysteine; SMD: standardised mean difference; PANSS: Positive and Negative Syndrome Scale.
Overall effect was derived from final time point of each study.
Effect of NAC treatment: PANSS negative symptoms
There were six studies that provided data on negative symptoms as measured by the PANSS at ⩽8 weeks of NAC treatment (Berk et al., 2008a; Breier et al., 2018; Conus et al., 2018; Davis et al., 2014b; Farokhnia et al., 2013; Zhang et al., 2015), and three of these studies also provided data for ⩾24 weeks of treatment (Berk et al., 2013; Breier et al., 2018; Conus et al., 2018). There was no significant difference between NAC and placebo groups in negative symptoms at the ⩽8-week time point (Figure 2). However, at the ⩾24-week time point, NAC did significantly improve negative symptoms in comparison to placebo, with a moderate effect size (SMD = −0.41, p = 0.006; Cohen, 1988) (Table 3). When combining data to investigate the overall effect of NAC, we included data from the final time point available from each study only. For PANSS negative, a statistically significant large overall effect was observed (SMD = −0.72, p = 0.003).
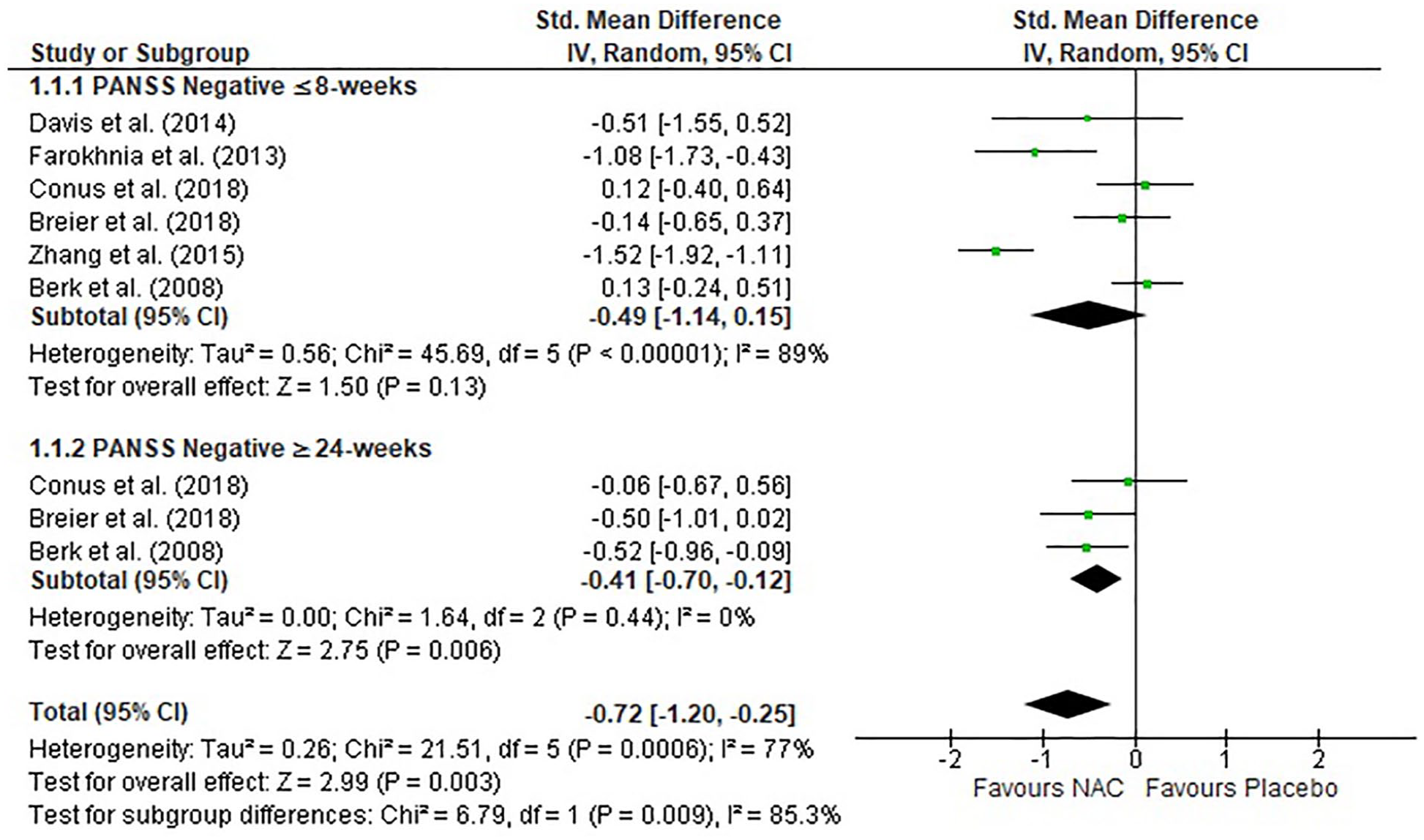
Forest plot of NAC treatment for negative symptoms as measured by the PANSS.
Effect of NAC treatment: PANSS positive symptoms
There were five studies that explored positive symptoms at ⩽8 weeks of NAC treatment (Berk et al., 2008a; Breier et al., 2018; Davis et al., 2014b; Farokhnia et al., 2013; Zhang et al., 2015), and three of these at ⩾24 weeks of treatment (Berk et al., 2008a; Breier et al., 2018; Conus et al., 2018). There was no significant difference between the NAC and placebo groups in positive symptoms at the ⩽8-week time point or the ⩾24-week time point (Figure 3). However, when combining data from final time points, the small overall effect did reach the significance threshold of p = 0.05 (SMD = −0.21).
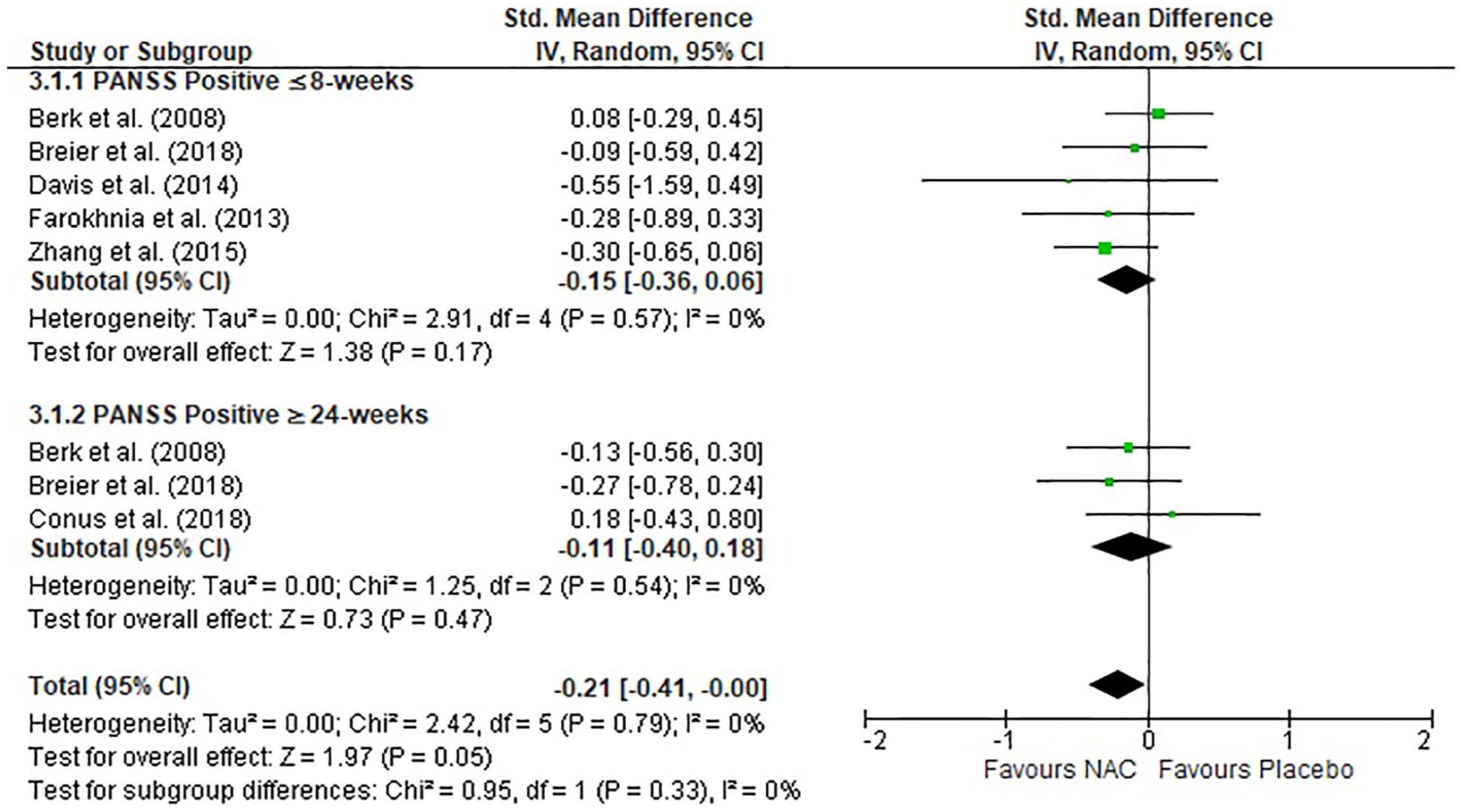
Forest plot of NAC treatment for positive symptoms as measured by the PANSS.
Effect of NAC treatment: PANSS general symptoms
Four studies reported general symptoms at ⩽8 weeks of NAC treatment (Berk et al., 2008a; Davis et al., 2014b; Farokhnia et al., 2013; Zhang et al., 2015). Only one study (Berk et al., 2008a) reported the effect of NAC on general symptoms after ⩾24 weeks of treatment and thus we could not conduct meta-analytic investigation of this time point. There was no significant difference between the NAC and placebo groups in general symptoms at the ⩽8-week time point (Figure 4). However, when combining data from final time points, the overall small effect did reach the significance threshold (SMD = −0.34, p = 0.05).
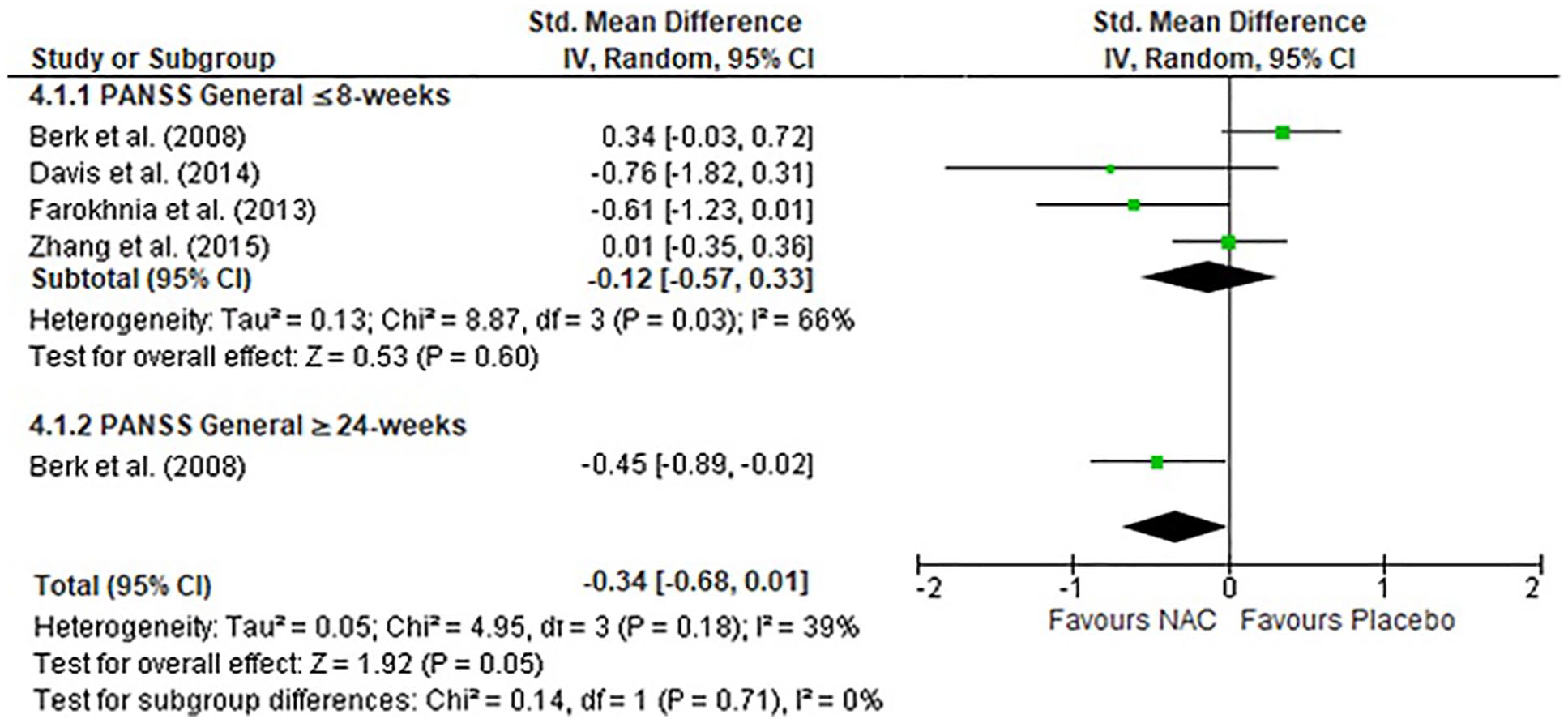
Forest plot of NAC treatment for general symptoms as measured by the PANSS.
Effect of NAC treatment: PANSS total symptoms
Five studies reported total PANSS score after ⩽8 weeks of NAC treatment, and only two studies at ⩾24 weeks of treatment. There was no significant difference between the NAC and placebo groups in total PANSS score at the ⩽8-week time point (Figure 5). However, there was a moderate significant improvement in PANSS total score after ⩾24 weeks (SMD = −0.64, p < 0.001). When combining data from final time points, there was a large significant overall effect of NAC for total PANSS scores (SMD = −0.92, p < 0.001).
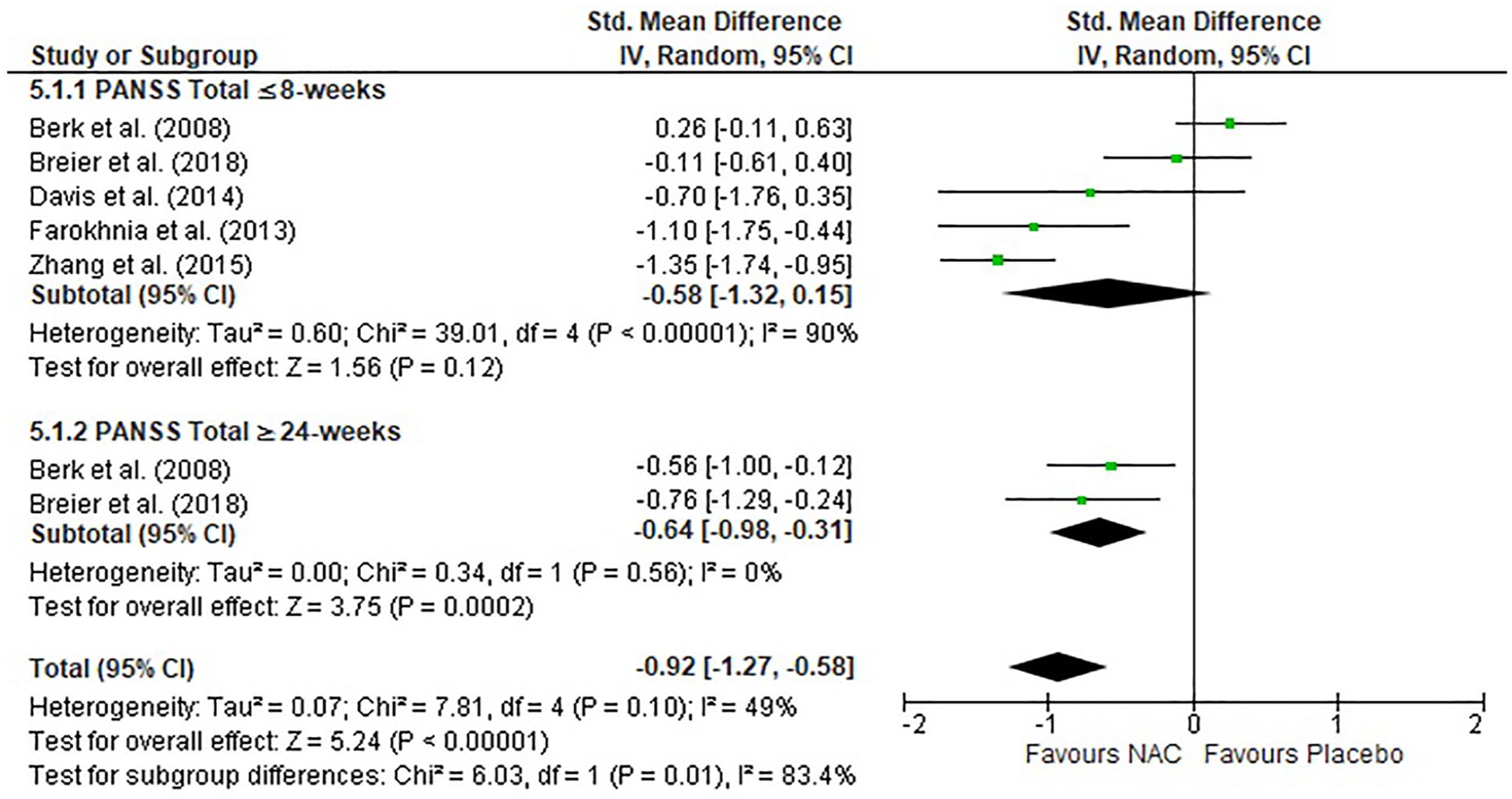
Forest plot of NAC treatment for total score as measured by the PANSS.
Effect of NAC treatment: cognition
We were not able to conduct meta-analysis on overall composite cognitive scores as only one study provided this data (Breier et al., 2018). In terms of specific cognitive domains, data were available to perform meta-analysis on processing speed and working memory, with three studies providing the requisite information for meta-analysis (Conus et al., 2018; Davis et al., 2014b; Rapado-Castro et al., 2017). For an outline of the measures used to determine processing speed and working memory score, see Supplementary Appendix 1.
Regarding processing speed, there was no significant difference in processing speed between the NAC and placebo groups. In terms of working memory, there was a moderate significant improvement in working memory in response to NAC treatment across the three studies (SMD = 0.56, p = 0.005; Figure 6). There were insufficient data to explore the effect of different time points on cognition subscales.
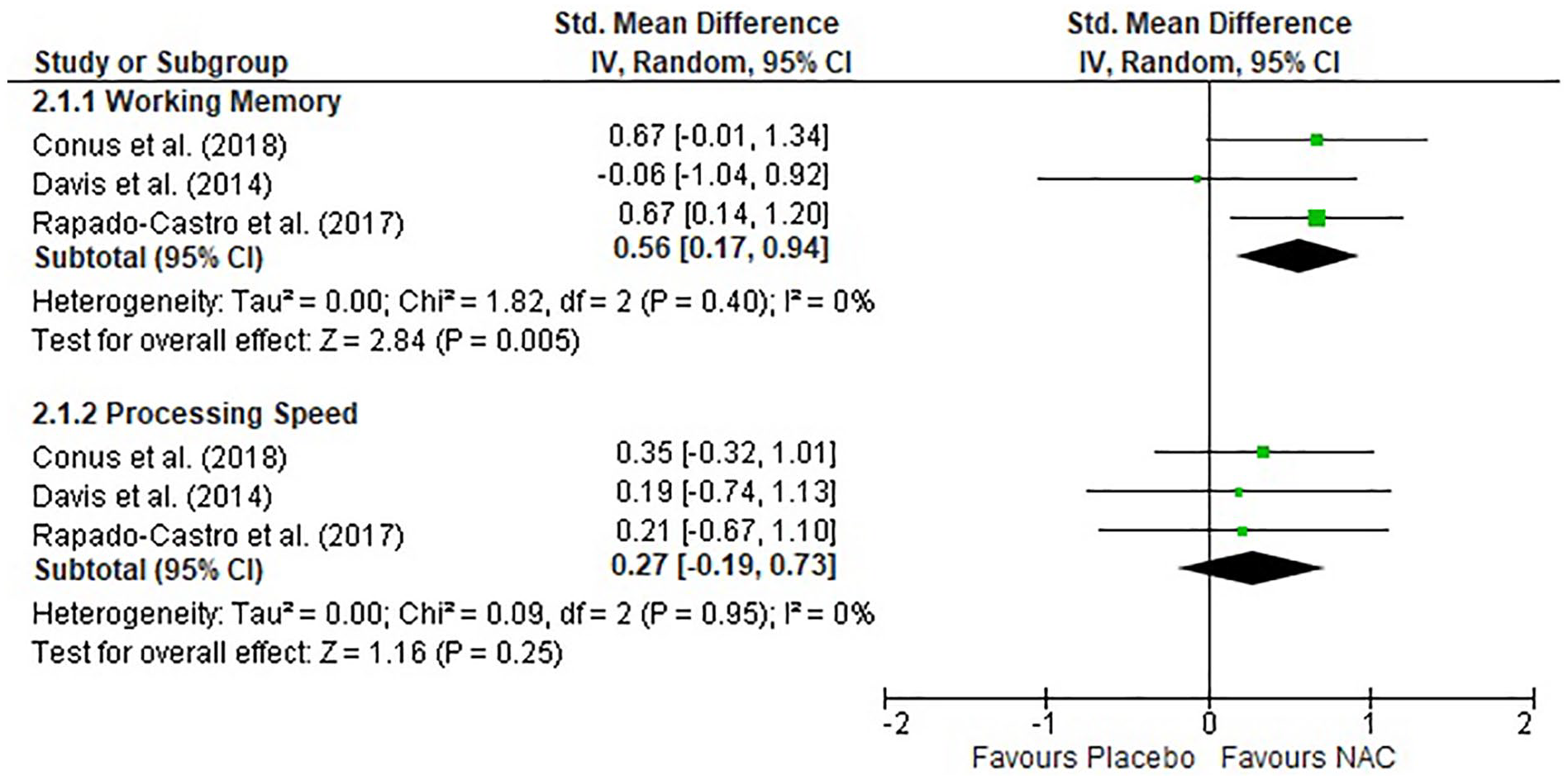
Forest plot of NAC treatment for working memory and processing speed.
Differences between groups
When comparisons were made between the NAC and placebo groups across all studies, there were no statistical differences in demographic variables, including gender and age.
Publication bias
We were unable to assess for publication bias, as there were fewer than 10 studies for any individual outcome assessed (Higgins and Green, 2011).
Sensitivity analysis
Sensitivity analysis was conducted by systematically excluding the single study that involved high risk of bias and was identified through the Chinese database source. The analysis revealed that excluding this study had no significant impact on the results from the ⩽8-week time point or the overall effect for PANSS negative, positive, total or general. The study did not contribute to our analysis for the ⩾24-week time point or cognition.
Discussion
This study revealed that NAC as an adjunct to antipsychotic medication may be effective for improving negative and total PANSS scores in schizophrenia after 24 weeks (or more) of treatment. No statistical improvement was observed at ⩽8 weeks of treatment in PANSS total or any of the PANSS subscales. PANSS positive and general symptoms were not significantly improved at either time points in isolation, but reached significance overall. Finally, NAC was found to improve cognition in terms of working memory, but not processing speed.
This study provides the first systematic meta-analysis investigating NAC as an adjunct treatment for first-episode psychosis and schizophrenia, excluding those with mood disorders. This was also the first meta-analytic study to investigate cognition as a key outcome for NAC treatment in schizophrenia.
A strength of our analysis was the inclusion of a range of international data (Zhang et al., 2015), and as yet unpublished data from Davis et al. (2014b). It is also the first study to investigate treatment effect by time point for most of the PANSS measures, in addition to overall effect. This is particularly meaningful as it has been proposed that longer interventions are required to receive the full benefit of NAC treatment (Berk et al., 2008a).
We found a significant difference between the NAC and placebo groups for both negative and total PANSS symptoms at the ⩾24-week time point and overall. However, there was no significant difference between groups on any symptom measures after ⩽8 weeks of NAC treatment, consistent with the aforementioned finding that longer exposure to NAC is required (Berk et al., 2008a).
The finding for positive symptoms overall was notable, as the theoretical link between NAC treatment and positive symptoms is less clear than for negative symptoms. Conus et al. (2018) found a significant association between improvement in positive symptoms and low baseline levels of GSH, which requires replication. As such, while general group data does not support the use of NAC for the treatment for positive symptoms, targeting those with low baseline GSH may result in some benefit from NAC.
There were no significant outcomes for PANSS general symptoms at the ⩽8-week time point. Berk et al. (2008a) were the only group to report on the PANSS general subscale at the ⩾24-week time point and found a significant improvement in response to NAC treatment. Similar to the finding for PANSS positive above, a significant overall effect was observed when investigating the final time point only for the PANSS general. The PANSS general factor encompasses elements of positive, negative, cognitive and mood domains and some combinations therein (van der Gaag et al., 2006). Given that NAC has been found to improve negative and some cognitive symptoms, it is possible that the significant finding by Berk et al. (2008a) at 24 weeks reflects an improvement on items relating to specific domains within the general subscale. Future studies might consider examining those items of this scale that relate to these items specifically, such as mood symptoms.
The effect of NAC on PANSS total score showed a similar trend to negative symptoms, in that there was a significant effect for both the ⩾24-week time point and overall, but not for ⩽8 weeks of treatment. There is substantial heterogeneity within the PANSS in regard to the constituent items and hence future studies might explore individual item effects on Marder factors (Giesbrecht et al., 2016; van der Gaag et al., 2006).
Duration of treatment as a critical issue is reflected in the data from bipolar studies. A trial with a 6-month duration separated from placebo (Berk et al., 2008b; Magalhães et al., 2011), but failed to separate earlier, and replication studies with shorter time frames have been negative (Berk et al., 2019; Ellegaard et al., 2019). It is likely that a neuroprotective agent like NAC may be working on neuroregenerative pathways in neuroprogressive disorders including neurogenesis, which is likely to be a very slow-moving process.
With respect to cognition, the present meta-analysis revealed a significant improvement in working memory in response to NAC treatment. In other disorders such as bipolar disorder, working memory also seems to be a domain specifically improved by pharmacotherapy (Daglas et al., 2016; Miskowiak et al., 2016). This relationship was not observed for the cognitive domain of processing speed. In contrast to this, Conus et al. (2018) found a significant correlation between improvement in processing speed and a decrease in negative symptoms in NAC-treated patients. This suggests that there may be a mediating effect of cognitive function on negative symptom improvement (or vice versa). As it stands, there is emerging evidence that NAC may be helpful in the treatment of cognitive impairment in schizophrenia. However, there are very few studies that have used cognition as a key outcome measure and more consistent reporting of specific cognitive domain scores. Ideally, future studies could use the MATRICS Consensus Cognitive Battery (MCCB), which is the gold standard measure for clinical trials (Nuechterlein and Green, 2006). It is highly linked to functional outcome (August et al., 2012) and would allow for great cross-study comparison. Indeed, a large-scale RCT investigating NAC in schizophrenia is currently being conducted utilising the MCCB (Rossell et al., 2016).
There are other studies of NAC in schizophrenia that did not meet inclusion criteria for the present meta-analysis but nevertheless support the notion that NAC may be an effective adjunct treatment for schizophrenia (Di Rosa et al., 2010; Lavoie et al., 2008; Tharoor et al., 2018). Lavoie et al. (2008) conducted a study on a subset of data from Berk et al.’s (2008a) study and found that NAC treatment significantly improved mismatched negativity (MMN) in comparison with placebo. MMN is an auditory evoked potential produced by a deviant stimulus, the amplitude of which is significantly decreased in schizophrenia patients (Lavoie et al., 2008; Michie et al., 2016). This low-level sensory processing deficit is considered a potential contributor to the higher order neurocognitive impairment observed in schizophrenia (Todd et al., 2013). It has been suggested that NAC may improve MMN through modulation of the glutamatergic receptor pathways via the NMDA receptor system (Lavoie et al., 2008; Michie et al., 2016). The relationship between sensory processing deficits and higher order cognitive processing such as executive function is as yet unclear (Neill and Rossell, 2013). This highlights an interesting theory regarding the possible mechanism of NAC, as improvement in sensory processing deficits potentially leading to higher order cognitive improvement.
Finally, an important additional finding from the included study of Zhang et al. (2015) was the secondary outcome measure of lipid profile in response to NAC treatment. The mortality risk due to cardiovascular disease is significantly increased in individuals with schizophrenia (Ringen et al., 2014). In addition, both atypical and conventional antipsychotic medications have been shown to increase lipids in people with schizophrenia (Roohafza et al., 2013). Zhang et al. (2015) found that indicators of harmful cholesterol and fats were lower in the NAC group after 8 weeks of treatment (significantly lower triglycerides, total cholesterol and low-density lipids). This is consistent with previous studies that suggest NAC may have a beneficial effect on lipid profiles (Korou et al., 2010). This provides an important further outcome measure of interest, as this recovery of lipid profile in response to NAC treatment may be extremely valuable for improvement of cardiovascular health in schizophrenia.
The present meta-analysis was restricted by the limited number of studies that have been performed in this field. There were only six independent studies (not including Rapado-Castro et al., 2017, which was a subset of Berk et al., 2008a) investigating symptoms, which included 440 participants. The studies included also had substantial variations in protocol, with differences in length of intervention, illness chronicity and daily dosage of NAC. As such, there was substantial heterogeneity between the included studies, and results should therefore be interpreted with caution. In addition, this study was unable to assess the effect of NAC on general well-being, as there have not yet been enough studies reporting these results to conduct a meta-analytic investigation at this time. It would be beneficial for future studies to report general well-being, utilising measures such as the Clinical Global Impression–Schizophrenia scale (CGI-SCH; Haro et al., 2003) or the Global Assessment of Functioning (GAF; Hall, 1995). Finally, while the PANSS was consistently used to calculate clinical symptoms, not all subscales were consistently reported. Similarly, there were differences in the cognitive outcomes reported. However, given the negative and cognitive symptoms are particularly pervasive in schizophrenia, the large effect size observed for negative symptom improvement is clinically meaningful. Similarly, a moderate improvement in working memory provides an encouraging initial finding.
Recommendations
Future research should endeavour to adopt longer interventions, as ⩽8 weeks of treatment does not appear to be sufficient for revealing the benefits of NAC supplementation. Furthermore, consistent reporting of cognitive outcomes, and ideally the use of the MCCB, would allow for more in-depth investigation of the effect of NAC on cognition.
It would be advisable for future studies to investigate the impact of illness chronicity and baseline GSH on response to NAC treatment (Conus et al., 2018; Girgis et al., 2019; Rapado-Castro et al., 2015), as is currently being undertaken by Rossell et al. (2016). Finally, making data available at the item level for the PANSS General scale would allow for a more in-depth exploration of this complex component of schizophrenia symptomology.
Conclusion
In summary, this study provided a rigorous meta-analysis investigating NAC as an adjunct treatment for schizophrenia. NAC may be effective in reducing negative and total symptoms in schizophrenia when longer intervention periods are employed (e.g. ⩾24 weeks of treatment). There is also evidence to suggest that NAC may improve specific domains of cognition, in particular working memory, but further research is needed. These findings may be used to aid in the design of future RCTs investigating NAC as an adjunct treatment for schizophrenia.
Supplemental Material
Supplementary_Material – Supplemental material for Meta-analysis of randomised controlled trials with N-acetylcysteine in the treatment of schizophrenia
Supplemental material, Supplementary_Material for Meta-analysis of randomised controlled trials with N-acetylcysteine in the treatment of schizophrenia by Caitlin OB Yolland, Donal Hanratty, Erica Neill, Susan L Rossell, Michael Berk, Olivia M Dean, David J Castle, Eric J Tan, Andrea Phillipou, Anthony WF Harris, Ana Rita Barreiros, Abigail Hansen and Dan Siskind in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors thank Yvonne Yang who, on behalf of Davis et al. (2014b), provided us with the necessary data to include this study in the present meta-analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Health and Medical Research Council Project Grant (GNT1098442). S.L.R. holds an NHMRC Senior Research Fellowship (GNT1154651). M.B. is supported by an NHMRC Senior Principal Research Fellowship (GNT1059660 and GNT1156072). E.J.T. holds an NHMRC Early Career Research Fellowship (GNT1142424).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
