Abstract
Background:
Bipolar disorder is associated with a high risk of suicide. Routine neuroimaging examination exhibited that bipolar disorder with suicidality was associated with brain structural and functional changes. However, the alterations of brain dynamics have still remained elusive.
Purpose:
To investigate the alterations of brain dynamics in unmedicated bipolar disorder II depression with suicidality and predict the severity of suicidality.
Materials and Methods:
This prospective study included 106 bipolar disorder II participants (20 with suicidal attempt, 35 with suicidal ideation, 51 without suicidal ideation) and 50 healthy controls who underwent resting-state functional magnetic resonance imaging between February 2016 and December 2017. We first used sliding window analysis to evaluate the dynamic amplitude of low-frequency fluctuations. Then, we predicted the severity of suicidality using a multivariate regression model.
Results:
One-way analysis of covariance revealed that the dynamic amplitude of low-frequency fluctuations in the right temporal pole, inferior temporal gyrus, superior temporal gyrus and the bilateral precuneus/posterior cingulate cortex was significantly different among the four groups. Post hoc pairwise comparisons revealed that dynamic amplitude of low-frequency fluctuations was remarkably decreased in the bilateral precuneus/posterior cingulate cortex in the three bipolar disorder II groups compared with that in healthy controls group. Increased dynamic amplitude of low-frequency fluctuations was found in the right superior temporal gyrus and inferior temporal gyrus in the suicidal attempt group compared with that in the other groups, and in the right temporal pole in the suicidal attempt group compared with that in the suicidal ideation and healthy controls groups. Importantly, these temporal variabilities could be used to predict the severity of suicidality (r = 0.330, p = 0.036), whereas static amplitude of low-frequency fluctuations couldn’t (r = –0.050, p = 0.532).
Conclusion:
Our findings indicated that alterations of temporal variability in the precuneus/posterior cingulate cortex are such a common feature of bipolar disorder patients. Besides, the severity of suicidality could be predicted by the dynamic amplitude of low-frequency fluctuations abnormalities rather than static amplitude of low-frequency fluctuations abnormalities, which is the first evidence of dynamic brain alterations in bipolar disorder patients with suicidality. The proposed predictive model may be advantageous for clinical applications.
Introduction
Bipolar disorder (BD) is a chronic psychiatric disorder characterized by high suicide risk. About 25–50% of BD adults make at least one suicidal attempt (SA) in their lifetime, and 8–19% of individuals with BD may die due to suicide (Goldstein, 2009). Suicide is one of the leading causes of death from BD, especially during depressive episodes. Due to the difficulties in predicting and preventing suicide, it has remarkably attracted scholars’ attention in the field of psychiatry and public health.
It is important to explicate that suicide is a ‘process’ that builds and develops in stages before the final step, suicide, is completed. This process is influenced by the interaction of genetic, neurobiological, neuropsychological and environmental factors and is thus a culmination of a complex set of intrinsic and extrinsic forces (Malhi et al., 2018). Evidence of an association between a vulnerability to suicidal behavior and neurobiological abnormalities is accumulating. Among functional and structural characteristics of suicidal brains, most researches have focused on major depressive disorder (MDD) (Bani-Fatemi et al., 2018). The main damaged brain areas are the structural and functional abnormalities in the frontal lobe, temporal lobe and limbic system (Gosnell et al., 2016; Kang et al., 2017; Olie et al., 2017). A previous functional magnetic resonance imaging (fMRI) study on MDD found greater amplitude of low-frequency fluctuations (ALFF) in the right superior temporal gyrus (STG) in MDD with SA compared to without SA and healthy controls (HCs) (Fan et al., 2013). As for BD, several structural magnetic resonance imaging (MRI) studies reported widespread abnormality including reduction of gray matter volumes in the STG, prefrontal cortex, fronto-limbic regions and parieto-occipital cortex, reduced fractional anisotropy within the orbital frontal white matter and corpus callosum in BD patients with SA compared to patients without a history of SA (Johnston et al., 2017; Mahon et al., 2012). Task-based fMRI revealed that the intensity of past suicidal ideation (SI) was associated with higher control-related activation in the ventrolateral prefrontal cortex and orbitofrontal cortex, insula and striatum, in addition to reduced activation in the cuneus and precuneus in BD patients, suggesting SA and SI histories related to prefrontal cortex–based circuit function in support of cognitive control (Minzenberg et al., 2015). Malhi et al. (2019a) found that patients with mood disorders with higher scores of suicidal activity had decreased default mode network (DMN) activity for Happy face-‘Sad’ word manipulation and increased basal ganglia network activity for Sad face-‘Happy’ word manipulation. Resting-state fMRI (rs-fMRI) correlated of the BD with suicidality mainly focused on functional connectivity, involving stronger posterior cingulate cortex (PCC) functional connectivity with parietal/insular region (Marchand et al., 2013), and greater posterior DMN activity, while less activity in the basal ganglia network (Malhi et al., 2019b). Only one study has used task-based fMRI to compare the brain activity of BD with and without suicidality (Minzenberg et al., 2015), while application of rs-fMRI, especially ALFF, has not been previously reported.
However, these convergent structural and functional MRI findings relied on the assumption of the ‘static’ of the brain and did not explore the dynamic brain alterations of brain injury over time in psychiatric patients with suicidality. Recently, a number of scholars have focused on exploring dynamic functional connectivity or networks that provide data about alterations of brain strength or spatial dynamic organization. Numerous researchers have successfully carried out dynamic inter-regional functional connectivity at resting state in a variety of mental and neurological diseases, such as schizophrenia (Damaraju et al., 2014), epilepsy (Li et al., 2018), MDD (Wu et al., 2019) and BD (Nguyen et al., 2017); however, knowledge about the temporal variability (dynamics) of local intrinsic brain activity quantified using the variance of the ALFF over time is still limited. Traditional studies about intrinsic brain activity amplitude assume that brain activity is stable throughout rs-fMRI scans, while recently conducted studies on brain activity have reported that blood oxygenation-level-dependent (BOLD) activity might be highly fluctuating over time during the resting state. This time-varying BOLD activity can be quantified by measuring the temporal variability of intrinsic brain activity amplitude among voxels using a sliding window approach to decompose full-length BOLD fMRI time series into a series of small data segments (Fu et al., 2018; Liao et al., 2019). The temporal variability of dynamic amplitude of low-frequency fluctuations (dALFF) was defined as the variance of dALFF maps across sliding windows (Liao et al., 2019). Alterations of temporal variability in regions involved in executive and emotional processing are associated with SI in MDD patients (Li et al., 2019) and new evidence on aberrant time-varying brain activity and its associations with brain connectivity in schizophrenia (Fu et al., 2018). To date, no study has concentrated on the dynamics of local brain activity of ALFF in BD patients and its correlation with suicidality.
Besides, genetic (Lee et al., 2011) and metabolic (Nikolaus et al., 2017) studies have found that BD I and BD II are different in pathophysiological and neurobiological mechanisms. BD II patients are characterized by more depressive episodes, longer time spent on a state of depression and more comorbidities of anxiety disorders compared with BD I (Judd et al., 2003). Moreover, neuroimaging studies have reported differences in structure (Abe et al., 2016) and task-based function (Caseras et al., 2013) between BD I and BD II. However, the majority of previously conducted studies have focused on patients with BD I and BD II combined indiscriminately, while a limited number of studies have investigated patients with BD II depression.
In this study, we employed temporal variability of intrinsic brain activity using dALFF on rs-fMRI in a homogeneous, large sample of unmedicated BD II depression participants with SA, with SI, without suicidal ideation (NSI) and HCs. The presented approach is supposed to be a reflection of neurobiological activity, which may cause high time-varying and fluctuating intrinsic brain activity (Fan et al., 2013). According to previous studies (Fan et al., 2013), we hypothesized that BD participants with suicidality, especially with SA, would show abnormal intrinsic brain activity in the temporal lobes in temporal variability. Furthermore, we used a multivariate regression model to indicate whether abnormal intrinsic brain dynamic activity can predict the severity of BD suicidality.
Materials and methods
Participants and clinical assessments
This retrospective study was approved by the Ethics Committee of the First Affiliated Hospital of Jinan University, Guangzhou, China. All subjects were right-handed, volunteered to participate in the study without compulsion and signed a written informed consent after a complete written and oral interpretation of the study. A total of 110 participants with BD II were recruited from the psychiatry department in- and out-patients of our hospital between February 2016 and December 2017. The participants were aged from 18 to 55 years.
According to the diagnostic assessment by the Structured Clinical Interview for DSM-IV Patient Edition (SCID-P) (First et al., 2002), all participants met the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (known as DSM-IV) criteria (First et al., 1997) for BD II depression. The clinical state was assessed using the 24-item Hamilton Depression Rating Scale (HDRS) (Hamilton, 1960) and the Young Mania Rating Scale (YMRS) (Young et al., 1978) by two experienced clinical psychiatrists (Y.J. and S.Z., with 21 and 6 years of experience in clinical psychiatry, respectively). All scales were finished during the 3-day period prior to the imaging session. All participants with BD II were diagnosed with total YMRS score < 7 and total HDRS-24 score > 21. The presence/absence and seriousness of current SI, and SA in participants with BD were evaluated using the Beck Scale for Suicide Ideation–Chinese Version (BSI-CV) (Li et al., 2011). SI referred to the desire to kill oneself without taking any practical action. SA is defined as a self-inflicted, potentially injurious behavior with a non-fatal outcome, in the presence of implicit or explicit evidence of intent to die, although the degree of intent may vary. There is sometimes more emphasis on deliberation rather than intention, where a suicidal attempt has to be deliberate and excludes ambiguous, interrupted or aborted attempts (Malhi et al., 2018). The measurements of SA were based on both self-report and participant’s medical records. It measures the medical lethality of any history of suicide attempt for one of nine possible methods (sedative drugs, taking poisonous substances, creating traffic accidents, cutting, jumping, drowning, shooting, immolation, hanging) with a clear or implicit intention of killing oneself based on SI but did not result in death. BSI is a self-reported scale with 19 items that can be used as a tool to detect and measure the severity of SI. The first five BSI items are often used to screen for the presence of SI, while the remaining 14 are often used to assess the risk of participants with SI. Score 1–3 for each item on the BSI. In our study, participants with any score >1 of item 4 or 5, indicating active SI, required completion of all the remaining 14 items. If the patient states a response to items 4 and 5 with zero score, indicating that there is no active SI, then the remaining 14 items can be skipped. The exclusion criteria were participants with other Axis-I mental disorders or Axis-II personality disorder, pregnancy, history of electroconvulsive therapy, organic brain disorder, neurological disorders, mental retardation, alcohol/drug abuse, cardiovascular diseases or any concurrent major physical illness. At the time of the test, all participants were either drug naive or unmedicated for at least 6 months.
In addition, 54 age- and gender-matched HCs were recruited through local advertisements. They were carefully screened by a diagnostic interview, the Structured Clinical Interview for DSM-IV Nonpatient Edition (SCID-NP), to rule out any current or past mental illness history. Further exclusion criteria for HCs were any history of psychiatric illness in first-degree relatives, current or past major medical or neurological illness.
MRI data acquisition and preprocessing
All MRI data were obtained on a GE Discovery MR750 3.0 T System, which uses an 8-channel phased array head coil. Subjects were scanned in a supine, head-first position with symmetrical cushions placed on both sides of the head to reduce movement. During the scanning, participants were asked to relax with their eyes closed, not to fall asleep; after the experiment, only the participants confirmed that they were not asleep were included. The gradient-echo echo-planar imaging sequence was used for rs-fMRI data with the following parameters: time repetition (TR)/time echo (TE) = 2000/25 ms, flip angle = 90°, voxel size = 3.75 mm × 3.75 mm × 3 mm, field of view (FOV) = 240 mm × 240 mm, matrix = 64 × 64 and slice thickness/gap = 3.0/1.0 mm. A total of 210 TRs were collected for each subject. Additionally, structural data acquisition was acquired using a three-dimensional brain volume imaging (3D-BRAVO) sequence covering the whole brain with: TR/TE = 8.2/3.2 ms, flip angle = 12°, bandwidth = 31.25 Hz, slice thickness/gap = 1.0/0 mm, matrix = 256 × 256, FOV = 240 mm × 240 mm and NEX = 1. Anatomical MRI images were also collected to exclude anatomical abnormalities. All participants were discovered by two experienced radiologists to confirm that there were no brain structural abnormalities.
The preprocessing was performed using Data Processing & Analysis of Brain Imaging (DPABI_V3.0, http://rfmri.org/DPABI) (Yan et al., 2016) based on Statistical Parametric Mapping (SPM12, www.fil.ion.ucl.ac.uk/spm/). The first 10 images of each subject’s rs-fMRI dataset were discarded to ensure longitudinally stable magnetization. The remaining 200 images were first corrected for slicing time and then realigned to the first image to correct for inter-TR head motion. This rearrangement adjustment provided a record of the head motion within the rs-fMRI scan. All subjects should not exceed 2 mm maximum displacement in any plane and 2° of angular motion, and 0.2 mm mean frame-wise displacement (FD) (Jenkinson et al., 2002). Individual T1 structural image segmentation (gray matter, white matter and cerebrospinal fluid) used a Segmentation toolbox. Then, the DARTEL toolbox was used to create a study-specific template for the accurate normalization. Then, the resting-state functional images were co-registered to the structural images and transformed into standard Montreal Neurological Institute (MNI) space with a resliced voxel size of 3 mm × 3 mm × 3 mm resolution. Then, several false covariates and their time derivatives were regressed out from the time course of each voxel, including the brain’s global mean signal, white matter, cerebrospinal fluid and the Friston-24 head motion parameters (including the 6 head motion parameters, 6 head motion parameters one time point before and the 12 corresponding squared items) (Friston et al., 1996).
Dynamic ALFF computation
Based on the temporal dynamic analysis (TDA) toolkits integrated in DPABI (http://rfmri.org/DPABI), we calculated the dALFF variability. The characteristics of dALFF variability were examined using the sliding window approach. First, the hamming window was chosen to slide the whole brain BOLD time series. Previous study has shown that the sliding window size between 10 and 180 s was reasonable to capture the dynamic characteristics of the whole brain activities (Gonzalez-Castillo et al., 2015). In order to rapidly capture dynamic shifting (with shorter windows) and reliable estimate of the correlations between regions (with longer windows), the sliding window size of 50 TR and shifting step of 1 TR were selected to examine the dALFF variability of the whole brain activities (Liao et al., 2019). We also calculated the dALFF with other window sizes (30 and 70 TRs), and the results were presented in the Supplementary Materials (Figure S1). For each individual, after removing the first 10 time points of the rs-fMRI dataset, the remaining 200 time points were segmented into 151 windows. In each sliding window, for a given voxel, the conversion of time series to the frequency domain was undertaken using a fast Fourier transform. The square root of the power spectrum was computed and then averaged across a predefined frequency interval (0.01–0.1 Hz). This averaged square root was termed ALFF at the given voxel (Zang et al., 2007). Subsequently, we calculated the standard deviation of dALFF values across all 151 windows to quantitative depict the temporal dynamic characteristics of ALFF. Then, z-standardization (subtract global mean dALFF and then divide the standard deviation dALFF) was applied within a gray mask to reduce the global activities variability effects among subjects. Finally, the dALFF maps were smoothed with a 4-mm full width at half maximum (FWHM) Gaussian kernel.
Statistical analysis
One-way analysis of covariance (ANCOVA) and post hoc analysis were performed to compare the demographic and clinical data among the four groups using SPSS 17.0 software (SPSS, Chicago, IL, USA). A chi-square test was used to compare the gender differences among the four groups. And p < 0.05 was considered statistically significant for all comparisons, two tailed (Bonferroni corrected for post hoc analysis). To examine the differences in dALFF variability among the four groups (the sliding window size of 50 TR and shifting step of 1 TR), one-way ANCOVA was carried out to compare differences on the standard deviation in Z value at each voxel within the gray matter mask with age, gender, education and the frame-wise displacement values as nuisance covariates. The brain regions showing significant differences based on the results of one-way ANCOVA were defined as masks for further post hoc analysis for the comparison of each of the two groups (Bonferroni corrected, p < 0.05). Multiple comparisons were corrected using Gaussian random field (GRF) theory (minimum Z > 2.3, cluster significant p < 0.05, corrected, and the cluster extent threshold at κ > 25 voxels).
Once statistically significant differences were observed, we calculated the Pearson correlation coefficients between the clinical variables and dALFF values for each region in the three BD participant groups. The clinical variables included age of onset, number of episodes, BSI score, 24-item HDRS score, YMRS score and duration of illness.
BSI symptom prediction
To assess the relationship between altered dALFF values and the BSI symptom, we predicted the BSI score for the BD participants in both SA and SI groups using a multivariate regression model. We used altered dALFF values (voxel level) in the SA and SI groups (compared with the HCs group, respectively) as features. The BSI scores in the SA and SI groups were specified as regression targets to perform regression analysis. The leave-one-out cross-validation (LOOCV) method was applied to establish a robust and reliable model that is an unbiased strategy and was applied in previous studies (Li et al., 2019). Leaving one single subject’s dALFF values out as a test dataset, training dataset included the remaining subjects’ dALFF values in each cross-validation iteration, so that the BSI score for the testing dataset was predicted according to the predicted score for the training dataset. Finally, we calculated the Pearson’s correlation coefficient between the predicted BSI score and the observed BSI score to indicate whether the predicted BSI score could be correlated with the observed BSI score. The dALFF values could predict the BSI scores if the statistical significance level of p < 0.05. We also applied multivariate regression model and the LOOCV scheme to predict the BSI score by static ALFF values, in order to determine whether the dALFF values could be more advantageous than static ALFF values in predicting the BSI scores.
Validation analysis
To detect the reliability of dALFF with the window length of 50 TR (100 seconds), we applied two additional window lengths (30 TR and 70 TR) to validate the main results (see Supplementary materials).
Results
Demographic data and clinical comparisons
Table 1 shows the demographic and clinical data for all subjects. In this study, four participants with BD II and four controls were excluded from further analyses because of excessive head motion during the image acquisition. Finally, 106 participants with BD II (20 with SA, 35 with SI and 51 NSI) and 50 HCs were enrolled in this study. Among the four groups, there were no significant differences in age and gender (p > 0.05), while there were significant differences in BSI score (p = 0.036) and 24-item HDRS score (p = 0.032), and there were subtle differences in years of education (p = 0.043).
Demographic and clinical data and standard deviations by group.
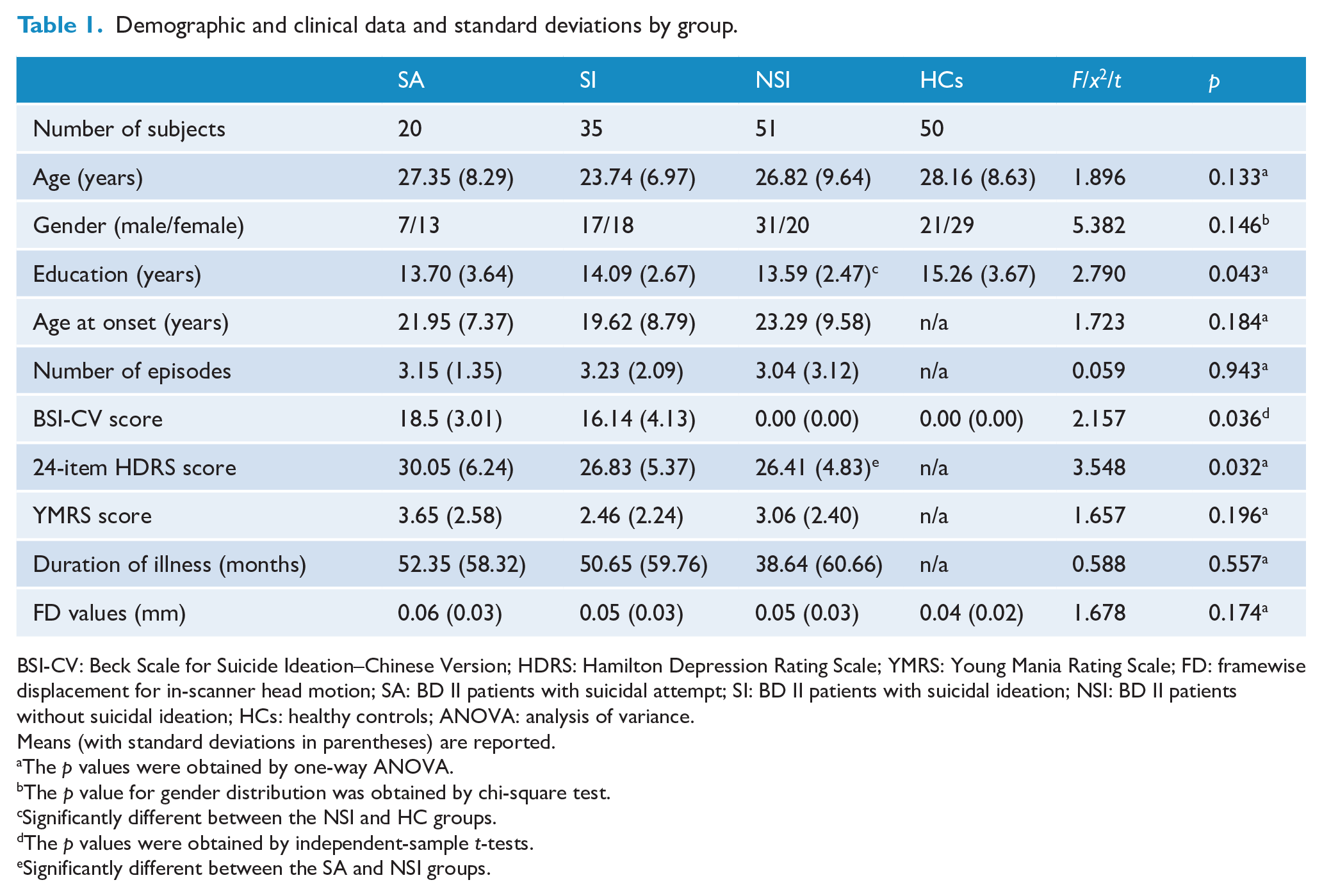
BSI-CV: Beck Scale for Suicide Ideation–Chinese Version; HDRS: Hamilton Depression Rating Scale; YMRS: Young Mania Rating Scale; FD: framewise displacement for in-scanner head motion; SA: BD II patients with suicidal attempt; SI: BD II patients with suicidal ideation; NSI: BD II patients without suicidal ideation; HCs: healthy controls; ANOVA: analysis of variance.
Means (with standard deviations in parentheses) are reported.
The p values were obtained by one-way ANOVA.
The p value for gender distribution was obtained by chi-square test.
Significantly different between the NSI and HC groups.
The p values were obtained by independent-sample t-tests.
Significantly different between the SA and NSI groups.
Dynamic ALFF variability differences among the four groups
According to one-way ANCOVA, we found that the dALFF variability in the right temporal pole (TP), the right inferior temporal gyrus (ITG), the right STG and the bilateral precuneus/PCC was significantly different among the four groups (Figure 1). Post hoc pairwise comparisons revealed that dALFF value was significantly decreased in the bilateral precuneus/PCC in all the three BD II groups compared with that in HCs group (p < 0.05). Remarkably increased dALFF value was noted in the right STG and ITG in the SA group compared with that in the other groups (p < 0.05). Additionally, noticeably elevated dALFF value was found in the right TP in the SA group compared with that in the SI and HCs groups (p < 0.05) (Figure 2, Table 2). For the static ALFF differences, see Supplementary Material (Figure S2).
Areas of dALFF differences between groups.
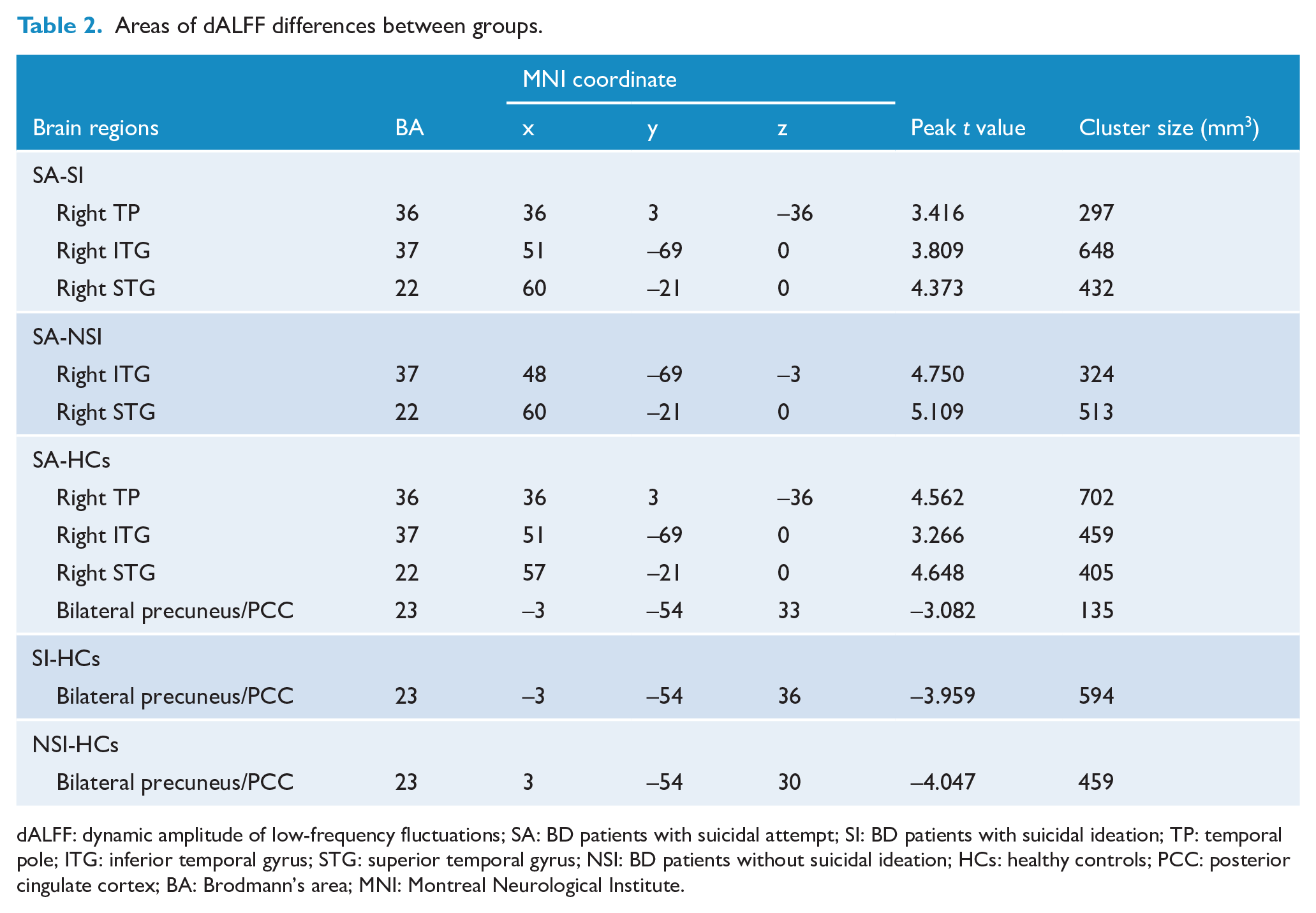
dALFF: dynamic amplitude of low-frequency fluctuations; SA: BD patients with suicidal attempt; SI: BD patients with suicidal ideation; TP: temporal pole; ITG: inferior temporal gyrus; STG: superior temporal gyrus; NSI: BD patients without suicidal ideation; HCs: healthy controls; PCC: posterior cingulate cortex; BA: Brodmann’s area; MNI: Montreal Neurological Institute.
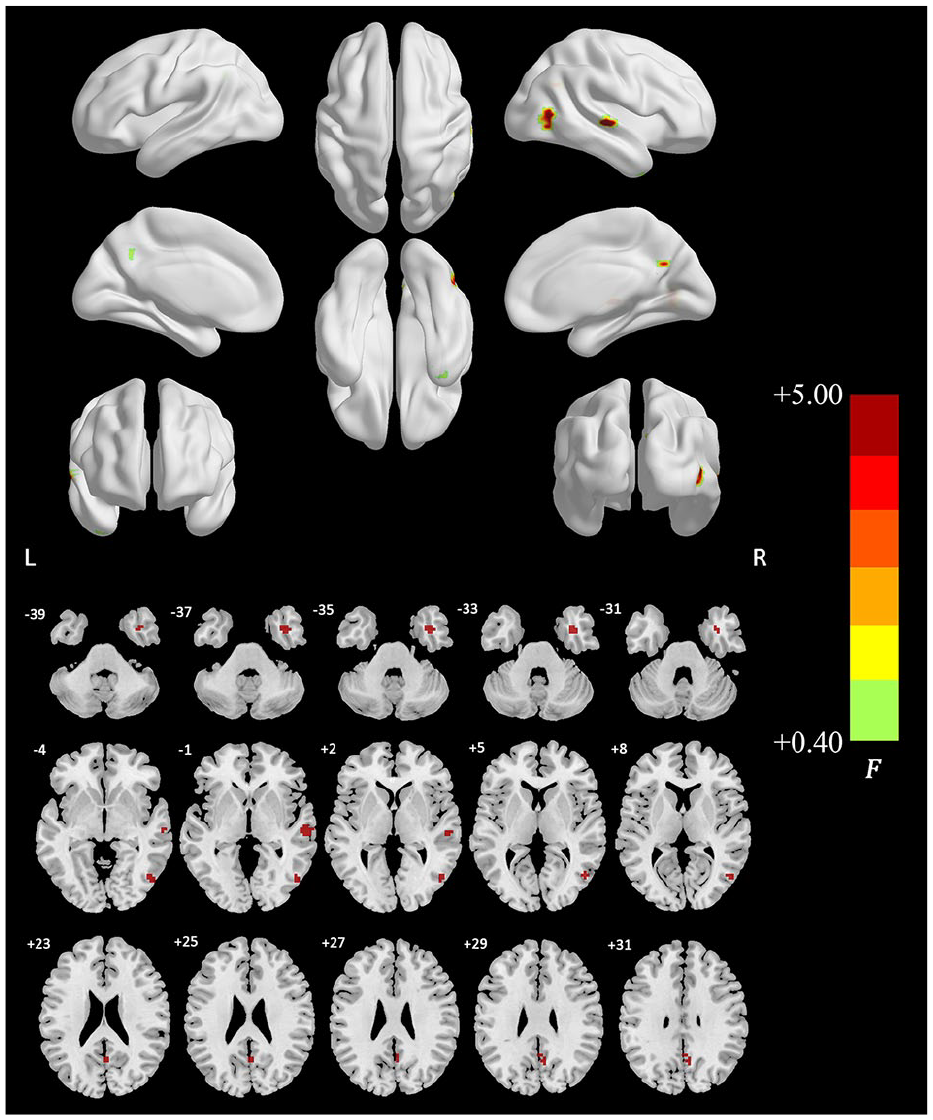
Group differences of temporal variability of the dALFF among the SA, SI, NSI and HCs group by one-way ANCOVA (minimum z > 2.3, cluster significant p < 0.05, GRF corrected, and the cluster extent threshold at κ > 25 voxels).
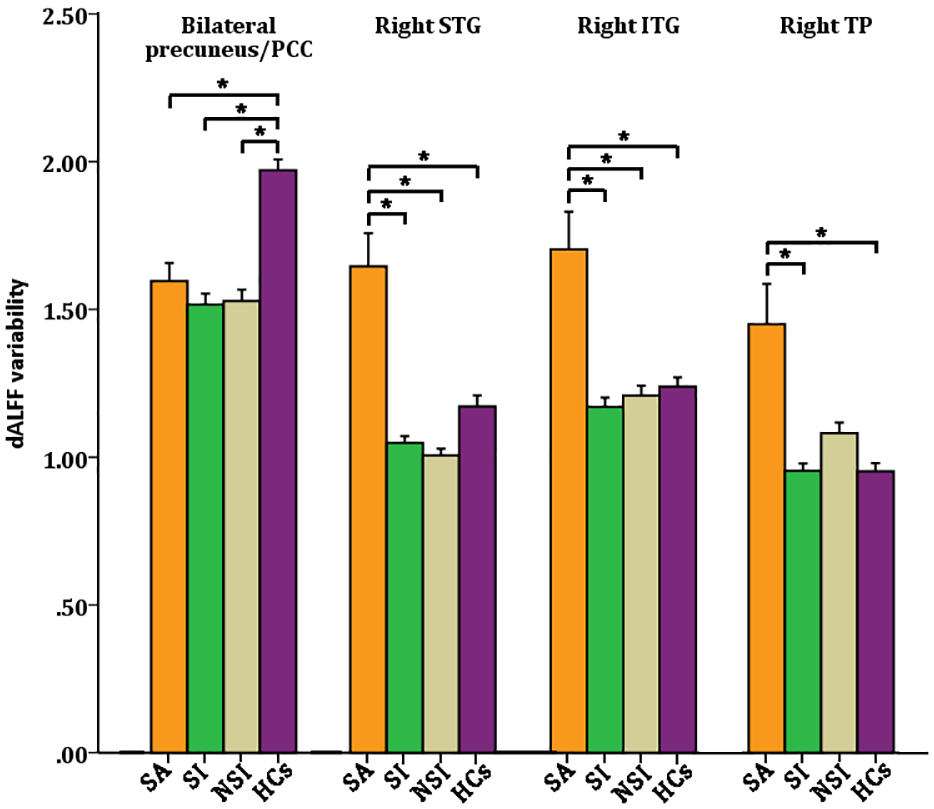
Group differences of temporal variability of the dALFF among the SA, SI, NSI and HCs group by post hoc two-sample t tests (minimum z > 2.3, cluster significant p < 0.05, GRF corrected).
Correlational analysis
Bonferroni correction was used for multiple comparisons in Pearson correlation analysis. There were no significant correlations between dALFF and any clinical variables (age of onset, number of episodes, BSI score, 24-item HDRS score, YMRS score and duration of illness) among the three BD groups.
Prediction of BSI score from temporal variability of ALFF
We found that the dALFF values in the SA and SI groups could be utilized to predict the severity of suicidality (r = 0.330, p = 0.036), while static ALFF values were not appropriate for this purpose (r = –0.050, p = 0.532) (Figure 3).
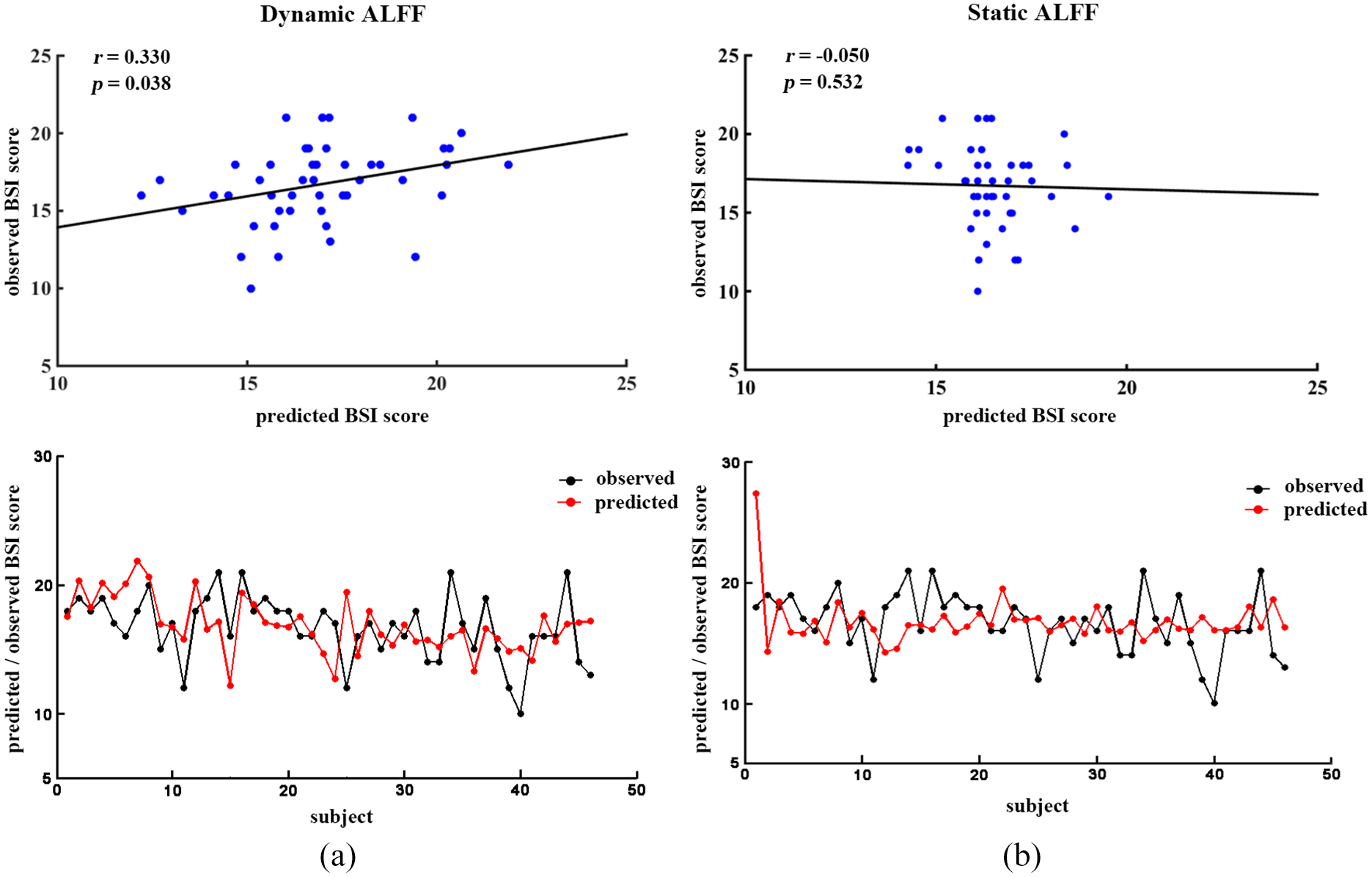
The altered dALFF values in the SA and SI groups predict the BSI scores: (A) the results of dALFF values as features to predict the BSI score of both SA and SI and (B) the results of static ALFF values as features to predict the BSI score of both SA and SI.
Validation results
The results of different sliding window lengths (30 TR and 70 TR) supported our main findings (50 TR) (Figure S1 in the Supplementary Materials).
Discussion
We applied rs-fMRI and temporal variability of ALFF to explore brain dynamics in a relatively large sample of participants with unmedicated BD II with SA, SI and without SI. The primary findings of this study indicated decreased dynamic local activity in the bilateral precuneus/PCC in the three groups of SA, SI and NSI. Notably increased dALFF was found in the right STG and ITG in the SA group compared with other groups. Dramatically elevated dALFF was noted in the right TP in the SA group compared with that in the SI group. Specifically, our results showed that the severity of suicidality could be predicted by the dALFF abnormalities rather than static ALFF abnormalities. To the best of our knowledge, this is the first study that used a novel approach to explore brain dynamics in unmedicated BD II depression with suicidality.
We found increased brain dynamics (more temporal variability of dALFF) in the right temporal cortex (including the STG, ITG and TP) in BD II depression with SA. The STG is a part of a complex facial and auditory processing system that participates in emotional perception and cognitive regulation (Campbell et al., 1990). The ITG plays a significant role in cognitive learning behavior, visual perception, object memory and visceral functions associated with emotions (Kuroki et al., 2006). The TP covers the anterior-most part of the temporal lobe, for example, a cap bridging the orbital frontal cortex and the amygdala, which receives and sends signals to both regions, and it is highly vital for the ability of socioemotional processing (Olson et al., 2007). In a diffusion tensor imaging study, reduced fractional anisotropy was found in the uncinate fasciculus, which is a white matter association tract in the human brain, connecting parts of the temporal lobe with frontal ones, indicating decreased structural connectivity in a ventral fronto-limbic neural system subserving emotion regulation in BD with SA (Johnston et al., 2017). Structural MRI study suggested that patients with psychotic disorders with SA had less gray matter volume in the STG and/or ITG when compared to those without attempts (McLellan et al., 2018). In addition, STG abnormalities are associated with the existence of auditory hallucinations, a painful experience that can lead to suicidality (Fialko et al., 2006). A rs-fMRI study unveiled differential activation of the STG in depressed patients with SA compared to patients without SA and HCs (Weng et al., 2019). A recent task-based (completed a version of the Cyberball peer interaction) fMRI study demonstrated that altered brain activity in the STG in depressed adolescents with high suicide ideation was significantly correlated with negative emotionality, social functioning and cognitive control (Harms et al., 2019). Moreover, previous task-based MRI studies in borderline personality disorder found that the STG mediated a ‘reflexive’ response to visual social inputs (Buchheim et al., 2008), especially negative visual stimuli caused by perceived rejection and social disappointment has been thought to be the most common precipitant of suicidal behavior (Soloff et al., 2012). A previous research revealed that offenders with psychotic disorders with SA had lower empathic accuracy and smaller TP volume, which suggested that the brain-behavioral correlation of social cognition may increase the significance of suicide risk models (Harenski et al., 2017). Magnetoencephalography uncovered that sustained activity of auditory cortex was strongly positively correlated with impulsivity (Han et al., 2015). Taken together, the mentioned findings indicated that increased dynamics of the temporal cortex may result in abnormal impulsivity and social and emotional processing related to BD patients with SA.
In the current research, decreased brain dynamics (less temporal variability of dALFF) in the bilateral precuneus/PCC was found in the three depressed BD II groups (SA, SI and NSI) compared with that in HCs group. The precuneus/PCC, as a part of the DMN, plays an important role in retrieval of autobiographical episodic memory and self-referential tasks (Buckner and Vincent, 2007). Our previous study on rs-fMRI demonstrated functional abnormities in the precuneus/PCC in BD patients (Zhong et al., 2019). Besides, cognitive control task–based fMRI reported relatively lower control-related activation in the precuneus/PCC among patients with history of SI and SA (Minzenberg et al., 2015). Alterations in the precuneus/PCC have been found to be associated with the presence of rumination (Zhu et al., 2017), a mode characterized by responding to distress that involves repetitively and passively focusing on negative self-relevant information which can be involved in the tendency toward SI or SA (Chase et al., 2017). Thus, findings of the above-mentioned studies combined with our results suggested that the decreased dALFF variability in the precuneus/PCC may be related to instability in regulating unbidden, intrusive thoughts motivating suicidality in BD patients.
More importantly, the severity of suicidality can be predicted by the temporal variability of dALFF. Although number of previous prior studies have reported a correlation between the neuroimaging characteristics and the clinical variables of SI in BD (Bani-Fatemi et al., 2018), the use of dynamic values to predict the severity of suicidality in BD has still remained elusive. In the present research, we employed dynamic values to predict the severity of suicidality in BD II patients, and we found that the dALFF value can successfully predict the severity of suicidality in BD, while the static ALFF value cannot. A recently conducted study on rs-fMRI demonstrated that temporal dynamics of intrinsic brain activity could be used to predict the SI in MDD patients (Li et al., 2019). Therefore, these findings suggest that the dALFF value can provide more valuable information about the intrinsic brain activity than static ALFF value and may be a more reliable biomarker for the pathophysiology of suicidality in individuals with BD.
The current research involves a number of limitations. First, the sample size of this study in SA and SI groups was relatively small, and results will require replication in a study with larger sample size. Second, we used a sliding window approach with the window size of 50 TRs (100 seconds) as a reasonable choice for estimation of dALFF value. Previous studies have shown that changes of functional connectivity are not particularly sensitive to a window size in the range of 20 seconds to 1 minute (Fu et al., 2018). However, since in the present research we only concentrated on dALFF, it remained unclear whether such a window size would be optimal for capturing the dynamics of brain activity. We have tested the reliability of the analysis with different window sizes, ranging from 30 to 70 TRs, and the overall results were similar. Therefore, we assumed that window size of 50 TRs could provide an appropriate trade-off between the ability to solve brain dynamics in the current sample. Third, because the BSI relies on being self-report, a possible explanation of our results is that the observed changes in function may be related to self-reported suicidal tendencies, or differences in the intentionality of self-harming behavior. Finally, we only conducted the rs-fMRI study, and cognitive evaluation and task-based fMRI study were not carried out. Hence, combination of task-based fMRI and cognitive evaluation will be advantageous to verify the existing data.
Conclusion
In summary, our results demonstrated that all the patients with BD II depression including SA, SI and NSI showed decreased dynamic local activity in the bilateral precuneus/PCC. Specifically, we noted that the BD II depressive patients with SA had significantly increased dynamic local activity in widespread areas of the right temporal cortex compared with the other three groups. Furthermore, the severity of suicidality could be predicted by the dALFF abnormalities rather than static ALFF abnormalities, which is the first evidence of dynamic brain alterations in BD patients with suicidality. Our findings suggest that the proposed innovative predictive model may help to develop a more powerful evidence for clinical applications.
Supplemental Material
Supplementary_material – Supplemental material for Characteristics of temporal dynamics of intrinsic brain activity in unmedicated bipolar disorder with suicidality
Supplemental material, Supplementary_material for Characteristics of temporal dynamics of intrinsic brain activity in unmedicated bipolar disorder with suicidality by Jiaying Gong, Guanmao Chen, Mengyao Zhou, Yanbin Jia, Shuming Zhong, Feng Chen, Shunkai Lai, Zhenye Luo, Jurong Wang, Hao Xu, Lu Wang, Li Huang and Ying Wang in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was supported by grants from the National Natural Science Foundation of China (81971597, 81671670 and 81501456); Project in Basic Research and Applied Basic Research in General Colleges and Universities of Guangdong, China (2018KZDXM009); Planned Science and Technology Project of Guangzhou, China (20160402007 and 201604020184). The funding organizations play no further role in study design, data collection, analysis and interpretation, and paper writing.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
