Abstract
Objective:
Many people with psychotic experiences do not develop psychotic disorders, yet those who seek help demonstrate high clinical complexity and poor outcomes. In this systematic review and meta-analysis, we evaluated the effectiveness and cost-effectiveness of psychological interventions for people with psychotic experiences.
Method:
We searched 13 databases for studies of psychological interventions for adults with psychotic experiences, but not psychotic disorders. Our outcomes were the proportion of participants remitting from psychotic experiences (primary); changes in positive and negative psychotic symptoms, depression, anxiety, functioning, distress, and quality of life; and economic outcomes (secondary). We analysed results using multilevel random-effects meta-analysis and narrative synthesis.
Results:
A total of 27 reports met inclusion criteria. In general, there was no strong evidence for the superiority of any one intervention. Five studies reported on our primary outcome, though only two reports provided randomised controlled trial evidence that psychological intervention (specifically, cognitive behavioural therapy) promoted remission from psychotic experiences. For secondary outcomes, we could only meta-analyse trials of cognitive behavioural therapy. We found that cognitive behavioural therapy was more effective than treatment as usual for reducing distress (pooled standardised mean difference: −0.24; 95% confidence interval = [−0.37, −0.10]), but no more effective than the control treatment for improving any other outcome. Individual reports indicated that cognitive behavioural therapy, mindfulness-based cognitive therapy, sleep cognitive behavioural therapy, systemic therapy, cognitive remediation therapy, and supportive treatments improved at least one clinical or functional outcome. Four reports included economic evaluations, which suggested cognitive behavioural therapy may be cost-effective compared with treatment as usual.
Conclusion:
Our meta-analytic findings were primarily null, with the exception that cognitive behavioural therapy may reduce the distress associated with psychotic experiences. Our analyses were limited by scarcity of studies, small samples and variable study quality. Several intervention frameworks showed preliminary evidence of positive outcomes; however, the paucity of consistent evidence for clinical and functional improvement highlights a need for further research into psychological treatments for psychotic experiences.
PROSPERO protocol registration number:
CRD42016033869
Introduction
High-risk criteria for psychotic disorders (Broome et al., 2005; Cannon et al., 2008; Cornblatt et al., 2002; Miller et al., 2002; Yung et al., 1996, 2003) are predicated on the presence of sub-threshold psychotic symptoms, also called psychotic experiences (PEs), and the presumption that preventing or delaying transition to a full psychotic disorder syndrome is a primary therapeutic target. However, most people with PEs never develop a psychotic disorder (Hui et al., 2013; McGorry et al., 2018; Perez et al., 2018), but have high clinical complexity, poor response to treatment (Perlis et al., 2011; Valiji Bharmal et al., 2015; Wigman et al., 2014), sub-optimal clinical and functional outcomes, and increased risk of self-harm (Fusar-Poli et al., 2012; Granö et al., 2011; Hui et al., 2013; Hutton et al., 2011; Kelleher et al., 2012; Yates et al., 2019). Despite evidence of these poor outcomes, many people with PEs do not meet the increasingly high thresholds for secondary care mental health services, while in primary mental healthcare settings their PEs often go unnoticed or untreated even though their depression and anxiety scores are higher, on average, than those of individuals without PEs (Hui et al., 2013; Perez et al., 2018).
Research on psychological interventions for people with PEs has mainly focused on delaying or preventing transition to psychotic disorder. Despite this focus, a recent network meta-analysis of transition rates among people at high risk for psychosis found no evidence to support the effectiveness of needs-based interventions (NBIs), cognitive behavioural therapy (CBT), integrated psychological interventions, or family-focused therapy (FFT) in comparison with each other (Davies et al., 2018a). A subsequent network meta-analysis of intervention effects further found that no one specific intervention was more effective than others with regard to reducing attenuated positive psychotic symptoms (Davies et al., 2018b). Yet, Nelson et al. (2018) have proposed several limitations of these reviews, citing the omission of (1) trial evidence demonstrating positive group-level effects of these interventions and (2) key clinical (e.g. depression and general psychopathology) and functional outcomes that clearly have important implications for the treatment of people with PEs.
Recent meta-analyses have left a number of key gaps concerning interventions for people with PEs that must be filled in order to ensure that treatment decisions and clinical guidelines are based on the most relevant, accurate, and up-to-date evidence available. First, most reviews have limited their focus to ‘ultra-high risk’ or ‘clinical high risk’ populations, thereby omitting people with PEs who may not have these diagnoses. Second, there is presently no meta-analytic evidence addressing the question of which psychological interventions lead to remission from PEs and improvement in depression, anxiety, and general functioning, all of which are important features of at-risk states for psychosis that lead to disability (Byrne and Morrison, 2014; Fowler et al., 2018; Law and Morrison, 2014). Third, the psychological intervention that has been most investigated in the context of people with PEs is CBT, while the evidence concerning alternative approaches has yet to be collated (Nelson et al., 2009). Fourth, the cost-effectiveness of achieving therapeutic targets other than transition has received little attention. Fifth, no review has set limitations for the use of antipsychotics, despite the fact that international guidelines do not generally recommend their use for people at-risk for developing psychosis (Addington et al., 2017; Early Psychosis Guidelines Writing Group and EPPIC National Support Program, 2016; National Institute for Health and Care Excellence (NICE), 2014; Schmidt et al., 2015). Finally, no review has aimed to illuminate the key ingredients of effective psychological interventions for this population. To address these significant gaps in the literature and to inform the development of a new therapeutic framework, we conducted a systematic review and meta-analysis that aimed to (1) synthesise evidence about the effectiveness of and economic outcomes associated with psychological interventions for people with PEs and (2) identify common components of effective interventions.
Methods
This review was conducted as part of the Tailoring evidence-based psychological therapY for People with common mental disorder including Psychotic EXperiences (TYPPEX), a nationwide National Institute for Health Research (NIHR) Programme Grant for Applied Research (RP-PG-0616-20003) that aims to develop an effective therapeutic framework for service users with PEs in the UK Improving Access to Psychological Therapies (IAPT) primary mental healthcare setting (www.england.nhs.uk/mental-health/adults/iapt/). The programme focuses on clinical and functional outcomes other than transition to psychotic disorder, reflecting the low transition rate among individuals with PEs accessing primary mental healthcare services (Hui et al., 2013; Perez et al., 2018). The therapeutic framework will adhere to current international guidelines, which recommend psychological therapy – but not antipsychotic medication – for the treatment of individuals with PEs (NICE, 2014; Schmidt et al., 2015).
The protocol was registered with the International Prospective Register of Systematic Reviews (PROSPERO; www.crd.york.ac.uk/prospero), registration number: CRD42016033869 (22 May 2018 version), and a full protocol has been published prospectively elsewhere (Soneson et al., 2019). We follow the PRISMA (Liberati et al., 2009) reporting guidelines.
Data sources and searches
Two research assistants (E.S. and D.R.) collaborated with medical librarians at the University of Cambridge to create the search strategy (Supplemental Appendix A). The strategy combined terms for PEs, specific psychotic symptoms and psychological interventions, as well as database-specific subject headings. We searched MEDLINE, Embase and Health Management Information Consortium (HMIC) via Ovid; PsycINFO, Cumulative Index to Nursing and Allied Health Literature (CINAHL), Education Resources Information Center (ERIC) and EconLit via EBSCO; British Nursing Index (BNI) via ProQuest; and all Cochrane databases from 1 January 2000 (or the earliest publication date included in the database, if after 2000) to 15 December 2018 (when we ran all searches). We additionally searched the World Health Organization International Clinical Trials Registry Platform (WHO ICTRP) for relevant trials and Google Scholar, EThOS, and Open Grey for grey literature and dissertations. We collected additional citations through hand-searching reference lists of included publications.
Study selection
We included studies that examined any psychological intervention in adults with PEs but not psychotic disorders. To be included in our review, studies were required to have used the presence of PEs as the main study entry criterion. Due to the variety of terms used to represent PEs, we included populations with the following diagnoses: at-risk mental state, ultra-high risk/clinical high risk, attenuated psychosis, psychotic-like experiences, unusual experiences, sub-threshold psychosis, prodromal psychosis and schizotypal disorders. We restricted our studies to adults (operationalised as studies in which participants’ mean age was ⩾16 years) to reflect the age of people attending adult mental health services (e.g. UK IAPT services).
We included all frameworks of psychological interventions provided their effects were studied in people with PEs (i.e. interventions did not need to target PEs specifically). We did not restrict intervention setting (and included online interventions). We excluded studies that combined psychological and pharmacological interventions (i.e. where medication was provided as part of the intervention protocol). For medication prescribed external to the intervention, we placed no restriction regarding the proportion of participants taking medication for depressive or anxiety disorders, but included only studies in which less than 25% of participants were prescribed antipsychotic medication. The decision to limit the proportion of the study population using antipsychotic medication aligns with international guidelines’ cautions against prescribing antipsychotics for people at high-risk for developing psychosis (Addington et al., 2017; Early Psychosis Guidelines Writing Group and EPPIC National Support Program, 2016; NICE, 2014; Schmidt et al., 2015). This exclusion criterion further ensured the review was relevant to the UK IAPT setting, where psychological interventions are the only available treatment.
Our outcomes of interest were (1) the proportion of participants who remitted from PEs (primary outcome) and (2) changes in depression, anxiety, functioning, distress, quality of life or positive/negative psychotic symptoms (secondary outcomes). We placed no restriction on which tools were used to measure any of these outcomes, so long as they were valid and reliable. We did not set an a priori inclusion criterion for how to define remission from PEs (we include in our results how each study defined/measured this outcome). In addition, we included studies that reported any of the following economic outcomes: resource use, cost, partial economic evaluations, and full economic evaluations (where full economic evaluations are those that consider both the cost and outcomes of two or more interventions in a comparative analysis and partial economic evaluations focus only on cost description, cost-outcome description or comparative cost, analysis [Drummond et al., 2015]). Outcomes did not need to be the primary outcome of a study to be included in our review.
We placed no restriction on study design or comparator. We chose not to limit our review to controlled trials in order to ensure that newer intervention frameworks (which may be at pilot or earlier stages) could be represented.
We reviewed studies published in any language provided they had an English abstract (no foreign language articles advanced past the title/abstract screening stage). We excluded reports published before 2000 (when the at-risk mental state became widely adopted), reports where only an abstract was available, and secondary analyses of data from the same trial (to avoid including the same data from one individual multiple times within our results).
Two reviewers (E.S. and D.R.) independently screened titles and abstracts and excluded obviously irrelevant titles. We then reviewed the full texts of potentially relevant citations against our inclusion and exclusion criteria. Disagreements were resolved by discussion, with input from a third reviewer (C.K., J.P.) as necessary.
Data extraction and quality appraisal
Three reviewers (E.S., D.R., and M.H.) designed and piloted data extraction sheets. We extracted information on study/sample characteristics, intervention components and descriptions, data for outcomes related to our primary or secondary outcomes, and data required for quality assessment (see protocol for more detail (Soneson et al., 2019). Where information was not available, we consulted study protocols and contacted study authors by email. Two reviewers independently extracted data from a subset of four papers (17%) and one reviewer extracted the rest. Both reviewers reviewed all quantitative data for each included study.
We assessed risk of bias using the Effective Public Health Practice Project’s (EPHPP) Quality Assessment Tool for Quantitative Studies (Armijo-Olivo et al., 2012) for all reports and additionally used the Drummond Critical Appraisal of Economic Evaluations Checklist (Drummond and Jefferson, 1996) for economic reports. Two raters (E.S. and D.R.; M.H. and S.B. for economic studies) independently assessed quality, compared ratings and resolved disagreements by discussion.
Data synthesis and analysis
Meta-analysis
We analysed controlled studies through random-effects meta-analysis of standardised mean differences (SMDs) for our secondary clinical and functional outcomes (we did not have sufficient reports to perform meta-analysis for our primary outcome; see below). To combine outcomes from multiple follow-up points within individual reports, we fitted meta-analytic multilevel random-effects models via functions in the metafor package (Viechtbauer, 2010). A relatively new methodology, multilevel meta-analysis is becoming popular in the literature (Fernández-Castilla et al., 2019). The model overcomes the possibility of bias of overall effect by acknowledging that different time-points are not independent and correcting for this.
Ultimately, we conducted seven separate meta-analyses (one for each secondary outcome), separating each by the framework of the psychological intervention being investigated (as per protocol; Soneson et al., 2019). As CBT was the only intervention to be represented in more than one study, we were not able to conduct meta-analyses for the other intervention frameworks included in the review.
We separated results by comparator framework (supportive treatments [STs] vs treatment as usual [TAU]). We classified the following interventions as ST: supportive therapy, supportive counselling, non-directive reflective listening, NBI, and needs-focused intervention. The decision to group these interventions was based on similarities in their purpose and provision. We considered these interventions to have a common aim, namely, to act as non-specific active comparison groups. They further share several characteristics (e.g. warm, empathic listening and absence of active therapeutic techniques). This classification also facilitates comparison with related reviews that used similar groupings (Davies et al., 2018a, 2018b). In reporting our results, we provide separate pooled estimates for each comparator framework (i.e. ST and TAU separately) as well as an estimate for both comparators combined (i.e. ST and TAU combined). There are clinical and statistical reasons for this decision. First, the difference between TAU and ST is not well-defined; for example, ‘TAU’ sometimes consisted of CBT for depression or anxiety. Second, we found no statistical evidence to indicate any meaningful difference between outcomes for these comparators. As both interpretations are valid, and to ensure our results can adequately inform clinical practice, we include both estimates.
Sensitivity and sub-group analyses
We also conducted sensitivity analyses by including only those reports that received a global rating of ‘strong’ on the EPHPP tool.
No controlled clinical trials (CCTs) met inclusion criteria, and so our planned sensitivity analysis on the impact of CCTs was not possible. We had also intended to conduct sub-group analyses based on population (clinical vs non-clinical), but no studies of non-clinical populations were eligible for inclusion in the meta-analyses. Finally, we had intended to use sub-group analyses to quantitatively assess four a priori components of interest for cognitive therapy as previously highlighted in the literature: assessment of problems and goals, formulation, homework, and active change strategies (Flach et al., 2015; Morrison and Barratt, 2009). However, included reports did not meet our pre-specified criteria for sub-group analyses (see protocol for more detail; Soneson et al., 2019).
Assessment of heterogeneity and meta-biases
Although we aimed to assess heterogeneity of the meta-analytic results, our estimates were unreliable due to low numbers of included reports in each meta-analysis (Deeks et al., 2018). We still report Cochran’s Q for each meta-analysis, but interpretation needs to be cautious. For the same reason, it was not possible to perform the assessments of bias (e.g. publication bias, citation bias) specified in our protocol.
Narrative synthesis
We use narrative synthesis (Popay et al., 2006) to synthesise effectiveness findings and pre–post changes in our outcomes of interest from (1) controlled studies not eligible for inclusion in the meta-analyses and (2) uncontrolled studies. We furthermore narratively describe findings relating to common components of effective therapies.
Economic analysis
We present economic studies in tables containing study characteristics and results and use a narrative approach to synthesise findings as a result of the very small number of identified studies meeting inclusion criteria for the economic component of the review. We further discuss reports in terms of quality, using the Drummond checklist (Drummond et al., 2015).
Results
Search results
We identified 27 reports from 21 studies that met inclusion criteria (flowchart in Figure 1; summary of studies’ characteristics in Table 1; justifications for exclusion after full-text screening in Supplemental Appendix B; summary of baseline and outcome data in Supplemental Appendix C; intervention components in Supplemental Appendix D). Of these 27 reports, 4 reports using data from two randomised controlled trials (RCTs) included economic components that met our inclusion criteria. The interventions had diverse frameworks; while the vast majority of studies focused on variations on CBT or ST (with the latter always serving as the comparator), one study each represented strengths and mindfulness-based online social therapy, sleep CBT, mindfulness-based cognitive therapy, FFT, family psychoeducational intervention, cognitive remediation, and systemic therapy (each described below). The majority of these frameworks have been tested in the past 5 years, suggesting increased interest in new intervention frameworks for people with PEs.
Summary of studies included in the clinical effectiveness component of the review.
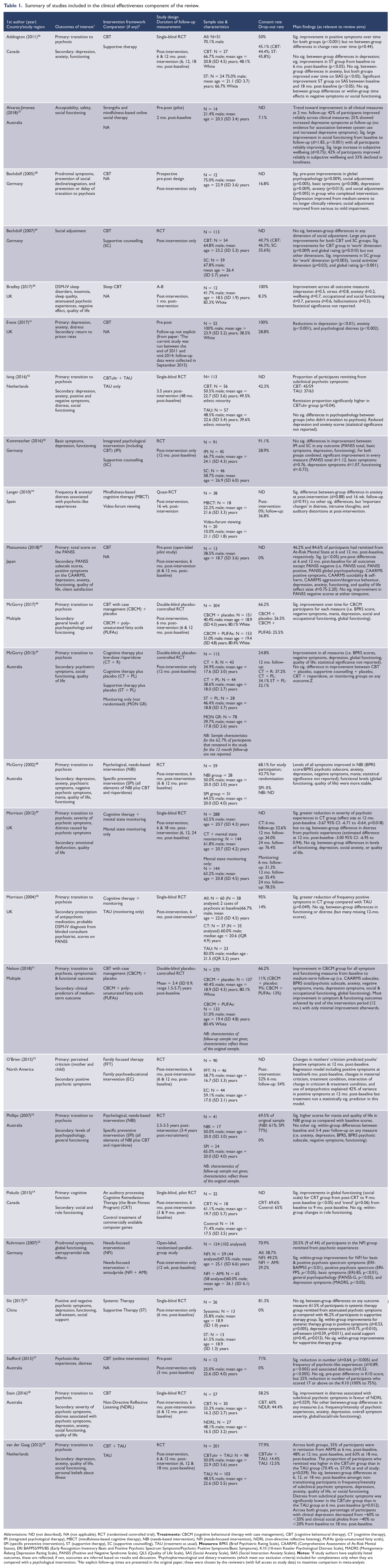
Abbreviations: ND (not described), NA (not applicable), RCT (randomised controlled trial);
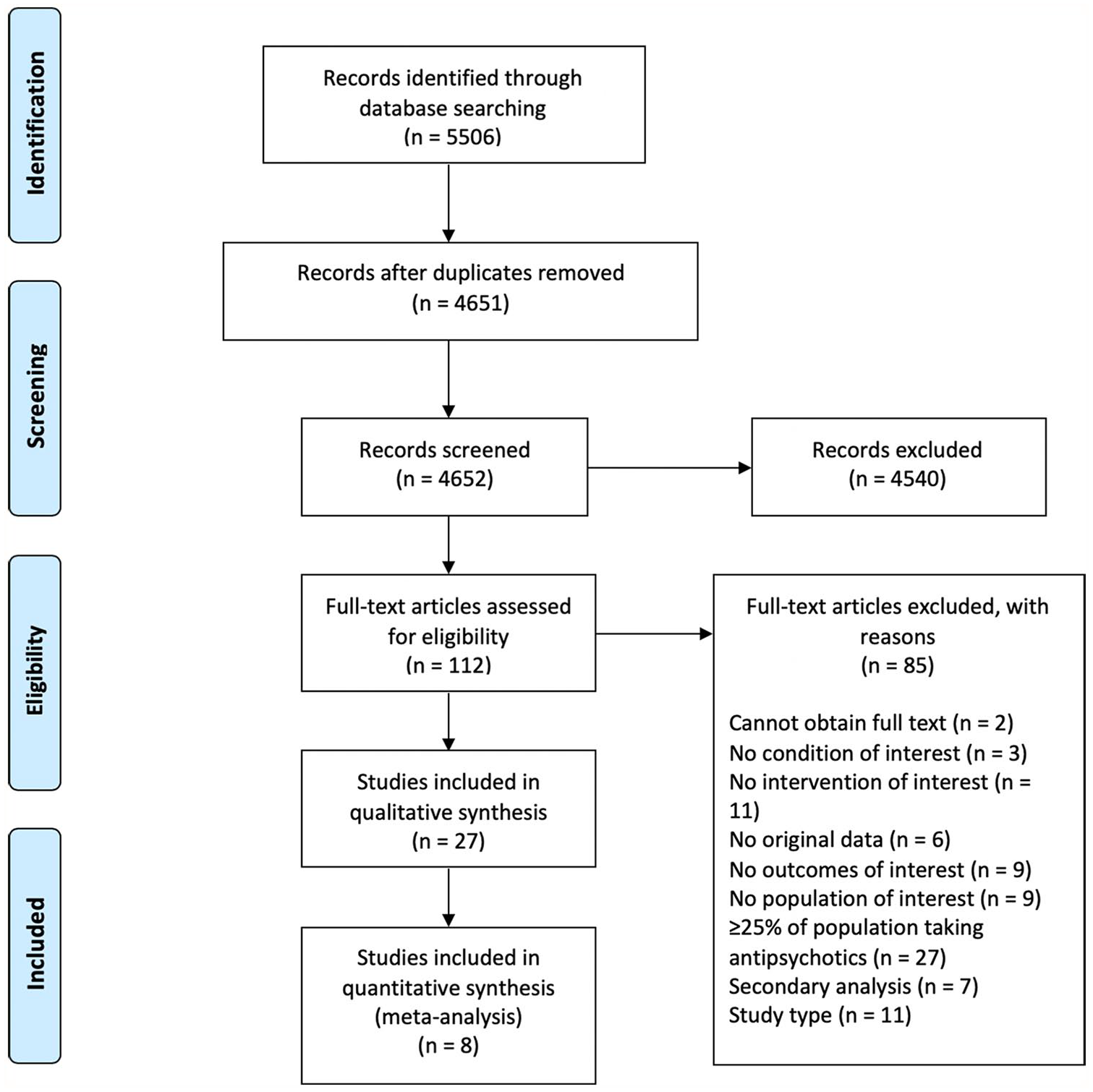
PRISMA flowchart (Liberati et al., 2009).
CBT
CBT for PEs (and other therapies where CBT is the key component, e.g. integrated psychological interventions) (Addington et al., 2011; Bechdolf et al., 2005, 2007; Evans et al., 2017; Ising et al., 2015, 2016, 2017; Kommescher et al., 2016; McGorry et al., 2013, 2017; Matsumoto et al., 2018; Morrison et al., 2004, 2012; Nelson et al., 2018b; Stafford et al., 2015; Stain et al., 2016; Van der Gaag et al., 2012) explores the links between thoughts, emotions and behaviour. The therapy is formulation-driven, problem-oriented, time-limited, and tailored to patients’ needs. The key components include patient engagement, creation of a mutually-agreed problem list, formulation, normalisation of PEs and patients’ interpretations of them, evaluation of alternative explanations, and behavioural experiments to challenge patients’ appraisals of PEs.
Cognitive remediation
Cognitive remediation refers to behavioural training aimed at improving cognitive processes (e.g. attention, memory and executive function) (Barlati et al., 2013). The cognitive remediation intervention included in this review focuses on improving auditory processing in people with PEs (Piskulic et al., 2015). It is computer-based and includes several different exercises to improve the diverse aspects of auditory processing.
Family-focused therapy
This therapy (O’Brien et al., 2015) treats people with PEs in the context of the family. The key components include psychoeducation around topics such as symptoms, daily stressors, coping strategies, the vulnerability–stress perspective, family support, and prevention action plans. Family members learn a structured approach to defining problems, breaking down complex problems, brainstorming solutions, analysing pros and cons of possible solutions, and selecting and implementing action plans.
Family psychoeducational intervention
The included family psychoeducational intervention (O’Brien et al., 2015) was a brief, three-session process of providing education and information. The content mirrored that of the psychoeducation aspect of the FFT described above.
Mindfulness-based cognitive therapy
Mindfulness-based cognitive therapy (MBCT; Langer et al., 2010) includes psychoeducation and exercises to demonstrate the links between thinking and feeling. Specific techniques include ‘Body Scan’ training, mindful breathing, breathing space, yoga, and sitting meditation. The intervention uses a group-based format.
Sleep CBT
The sleep CBT included this review (Bradley et al., 2017) used the ‘SleepWell’ treatment package, which utilises CBT techniques to address insomnia and circadian rhythm disruption to reduce sleep disturbances. Therapists use individualised formulation of sleep problems to identify treatment targets and actigraphy data to monitor changes in sleep patterns and highlight potential areas for change.
Strengths and mindfulness-based online social therapy
This intervention, set within a social media context, takes a strengths and mindfulness-based focus, and uses a self-determination theory of motivation to foster self-efficacy and increase positive emotions (Alvarez-Jimenez et al., 2018). The intervention provides social ‘online’ support moderated by expert and peer moderators. Modules addressed personal strengths, mindfulness, connecting with others, and group problem-solving to promote self-efficacy and interpersonal problem-solving.
Systemic therapy
Systemic therapy (Shi et al., 2017) is centred around systemic-constructivist and psychosocial resilience theories. The therapy focuses on solutions and resources, and encourages patients to reframe their problems and better understand their available resources in order to solve these problems.
Supportive treatments
As stated above, the category of STs includes supportive therapy, supportive counselling, non-directive reflective listening, NBI, and needs-focused intervention (Addington et al., 2011; Bechdolf et al., 2007; Kommescher et al., 2016; McGorry et al., 2013; Phillips et al., 2007; Ruhrmann et al., 2007; Shi et al., 2017; Stain et al., 2016). In general, these interventions use general counselling techniques, including warm, empathic, and non-judgmental face-to-face contact and supportive listening. They do not include active therapeutic techniques.
The quality of included studies was mixed (Table 2); 21 of the 27 reports used an RCT design, of which only 4 received a global rating of ‘strong’, 10 received a global rating of ‘moderate’, and 7 received a global rating of ‘weak’. Selection bias, confounding, and drop-out were the categories that most limited the global ratings (it should be noted that a rating of ‘strong’ in the selection bias category is not achievable when only help-seeking patients are included. Importantly, no study was excluded in the sensitivity analyses based solely on studying a help-seeking population). The remaining four studies used a pre–post design – relatively, a much weaker study design – but none of these received a ‘weak’ rating in any of the applicable categories.
Quality of included studies (EPHPP rating tool).
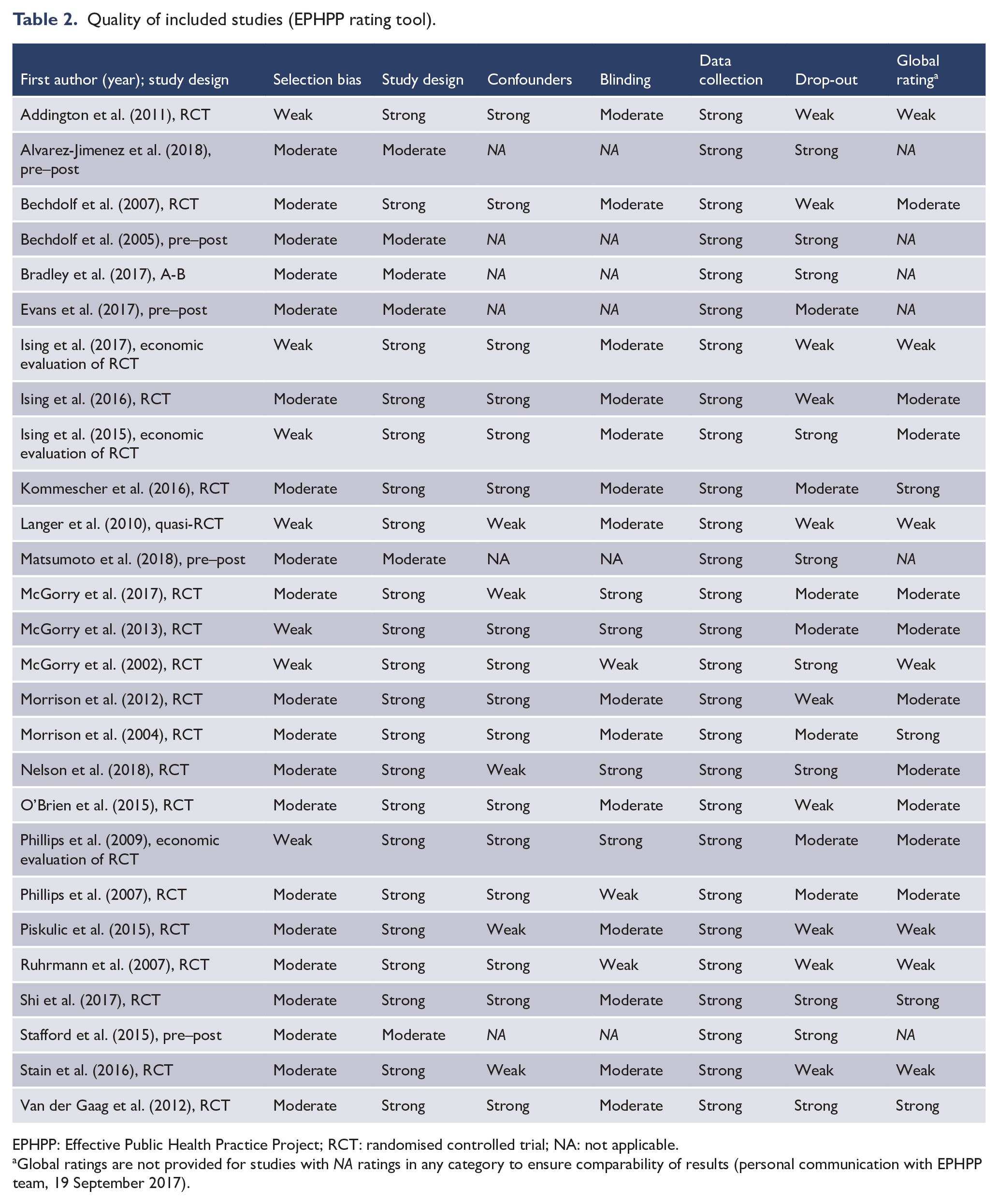
EPHPP: Effective Public Health Practice Project; RCT: randomised controlled trial; NA: not applicable.
Global ratings are not provided for studies with NA ratings in any category to ensure comparability of results (personal communication with EPHPP team, 19 September 2017).
Primary outcome
Five reports from four studies provided the proportion of participants that remitted from PEs following psychological intervention (Ising et al., 2016; Matsumoto et al., 2018; Ruhrmann et al., 2007; Shi et al., 2017; Van der Gaag et al., 2012). Meta-analysis was not possible for the primary outcome: only two reports had the same intervention framework and comparator category, and the more recent was a follow-up of the first (Ising et al., 2016; Van der Gaag et al., 2012).
CBT
Both studies of CBT used the Comprehensive Assessment of At-Risk Mental States (CAARMS; Yung et al., 2005) to determine remission status. In an RCT examining differences between CBT + TAU versus TAU, 70.4% of participants receiving CBT + TAU had remitted from at-risk mental state (ARMS) status by 12 months post-intervention, as compared with 57.0% of participants receiving TAU only (p = 0.039) (Van der Gaag et al., 2012). The difference remained significant at medium-term follow-up (approximately 3.5 years post-therapy), with 76.3% of the CBT + TAU group versus 58.7% of the TAU only group in remission (p = 0.04) (Ising et al., 2016). A pre–post study of CBT found ARMS remission rates of 46.2% at post-intervention and 84.6% 6 months post-intervention (Matsumoto et al., 2018).
Other frameworks
An RCT comparing systemic therapy with supportive therapy found greater remission from clinical high-risk status (measured using the Scale of Prodromal Symptoms; Miller et al., 2003) among those receiving systemic therapy (61.5% versus 46.2%), but the difference was not significant (p = 0.431) (Shi et al., 2017). Finally, a trial of a needs-focused intervention found a 20.5% remission rate from all psychotic symptoms (assessed with the Early Recognition Inventory – Positive Psychosis Spectrum [ERI-PPS]; Klosterkötter et al., 2001) at post-therapy (Ruhrmann et al., 2007).
Secondary outcomes
As mentioned above, we were only able to include studies of CBT in our meta-analyses, as CBT was the only framework examined in two or more studies.
CBT
We included eight reports from seven studies in our meta-analyses (Figures 2–8), four of which compared CBT (with or without TAU) with TAU only. CBT was superior to TAU in reducing distress (pooled SMD = −0.24 favouring CBT; 95% CI = [−0.37, −0.10]). No other statistically significant differences were found for positive psychotic symptoms (pooled SMD = −0.14 favouring CBT; 95% CI = [−0.32, 0.04]), depression (pooled SMD = −0.15 favouring CBT; 95% CI = [−0.35, 0.06]), anxiety (pooled SMD = −0.02 favouring CBT; 95% CI = [−0.22, 0.18]), functioning (pooled SMD = −0.09 favouring TAU; 95% CI = [−0.22, 0.04]), or quality of life (pooled SMD = −0.03 favouring TAU; 95% CI = [−0.24, 0.18]).
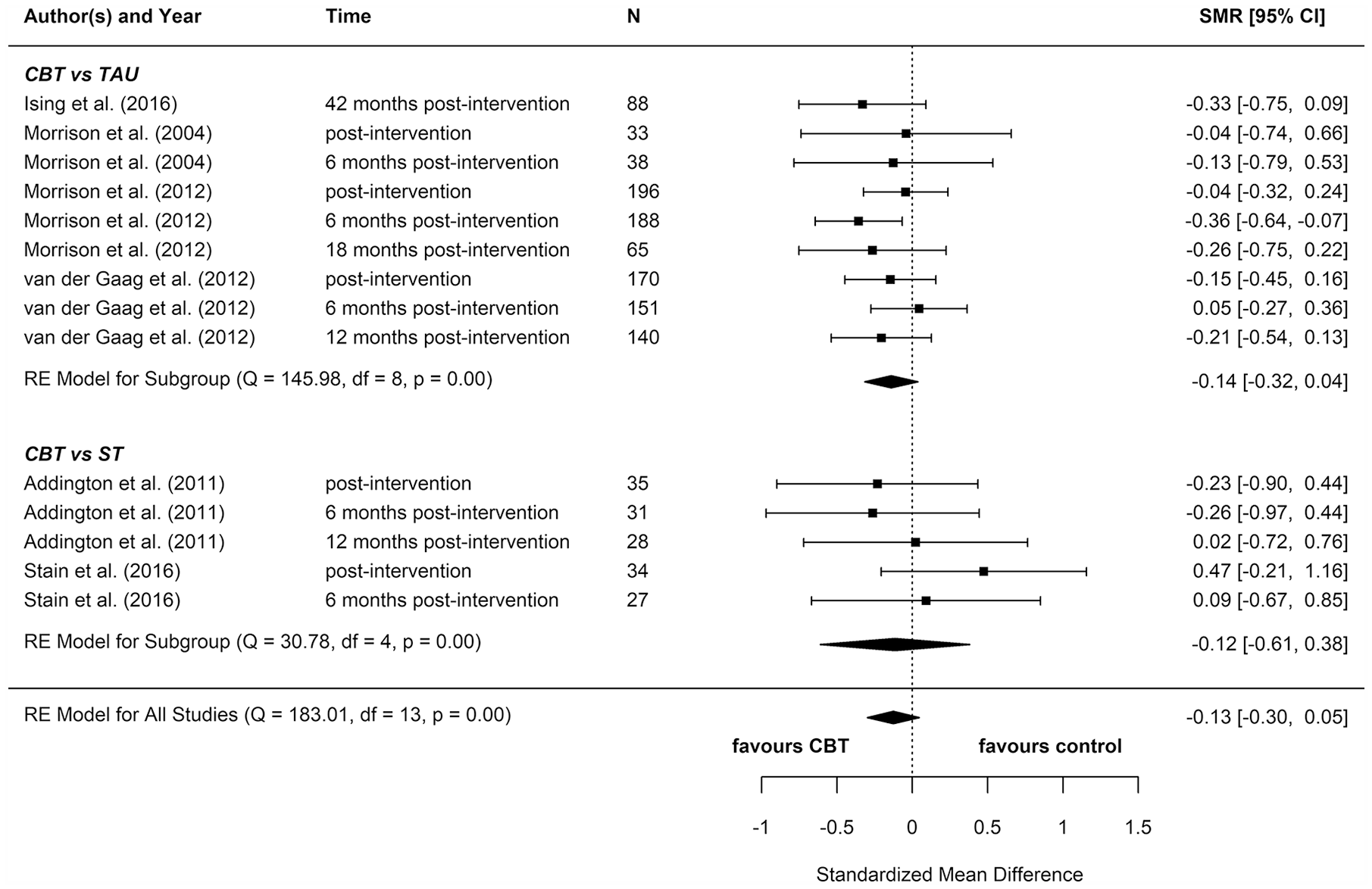
Positive psychotic symptoms: meta-analysis summary plot (NB: follow-up times are measured from the end of the intervention).
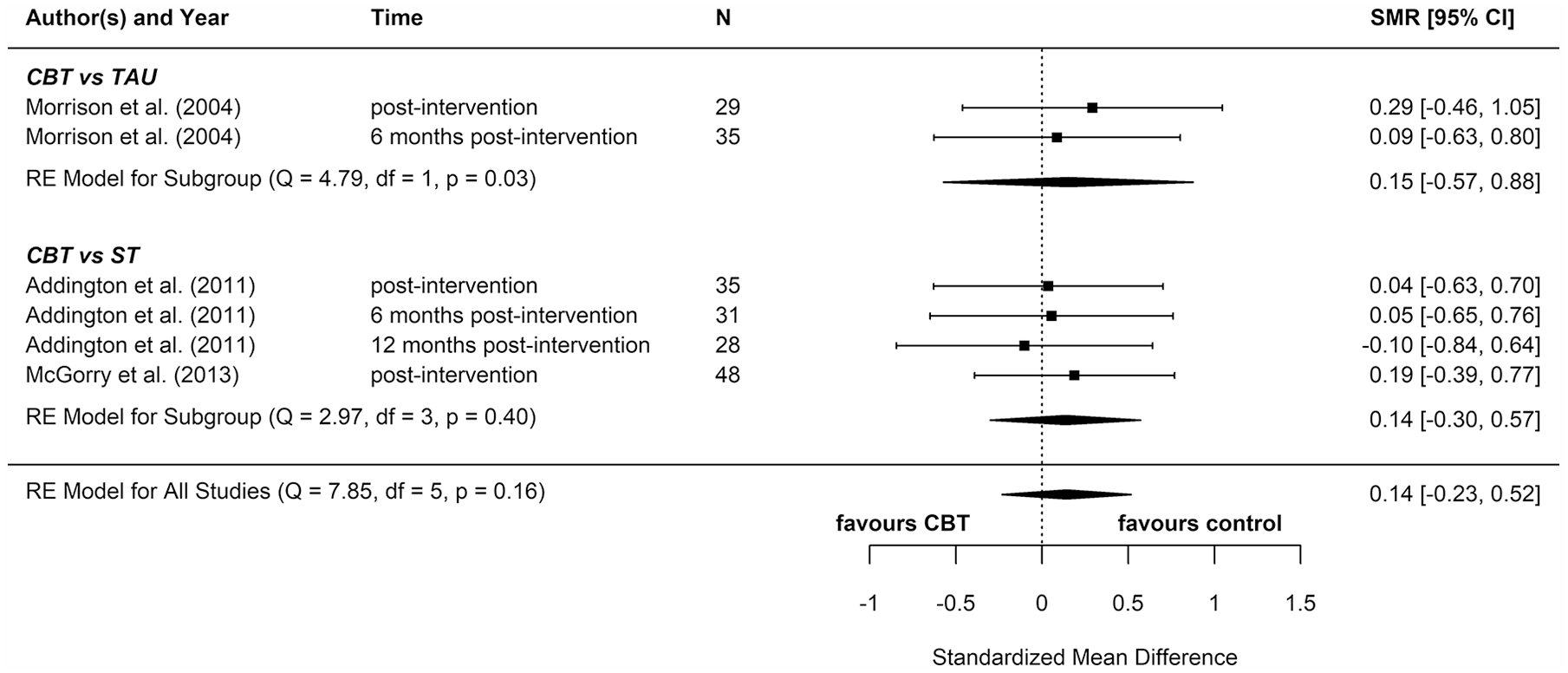
Negative psychotic symptoms: meta-analysis summary plot (NB: follow-up times are measured from the end of the intervention).
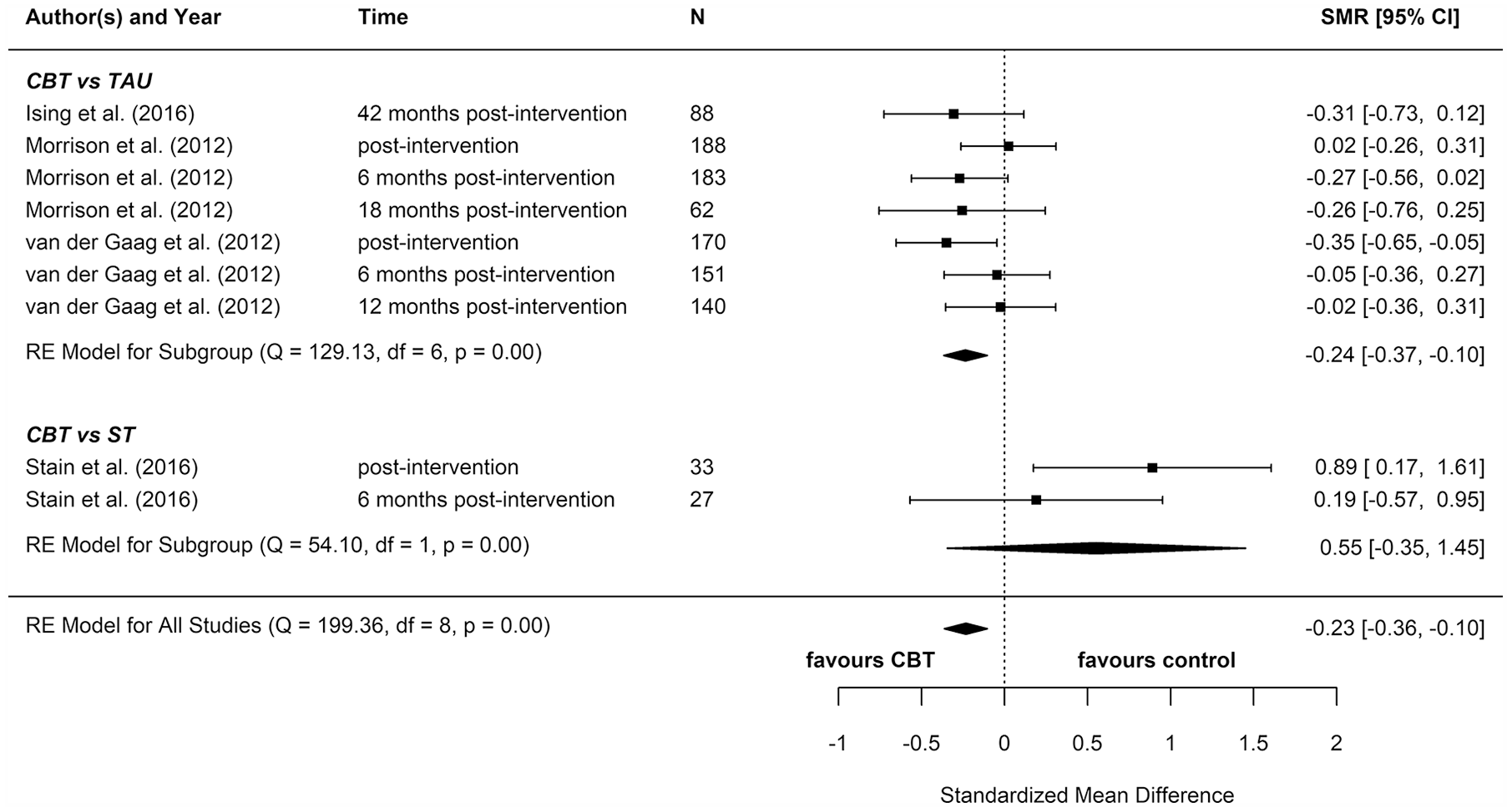
Distress: meta-analysis summary plot (NB: follow-up times are measured from the end of the intervention).
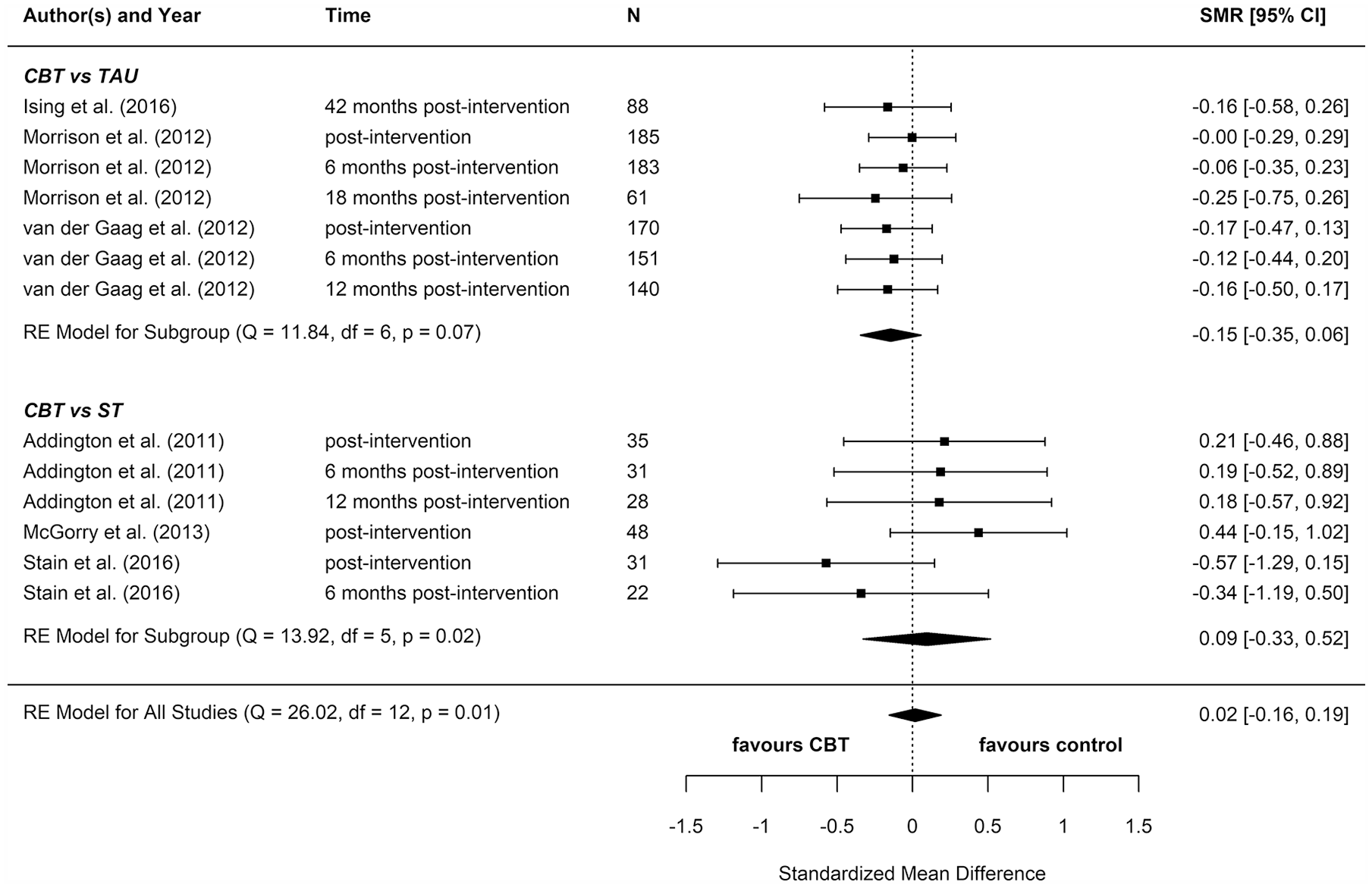
Depression: meta-analysis summary plot (NB: follow-up times are measured from the end of the intervention).
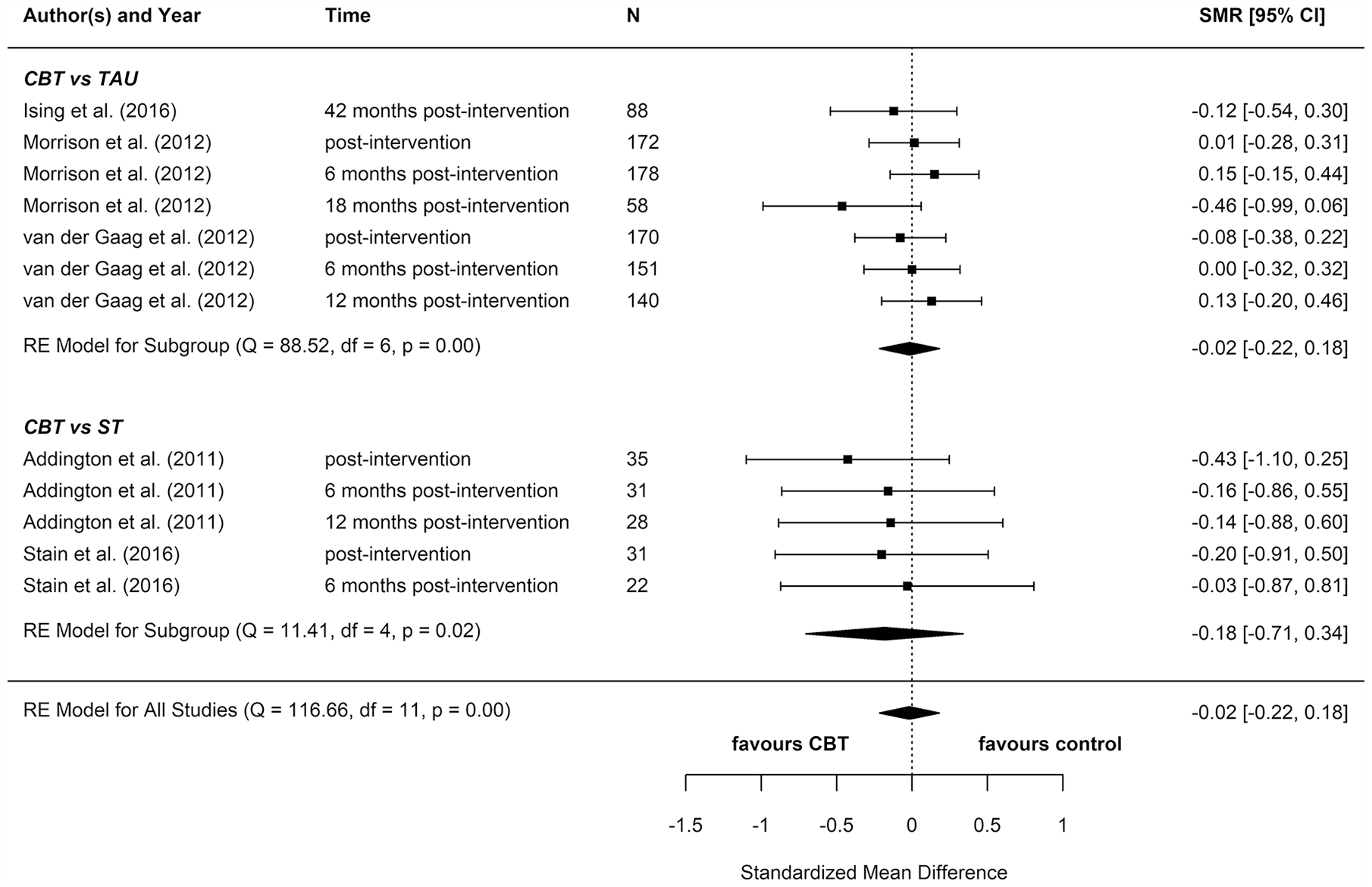
Anxiety: meta-analysis summary plot (NB: follow-up times are measured from the end of the intervention).
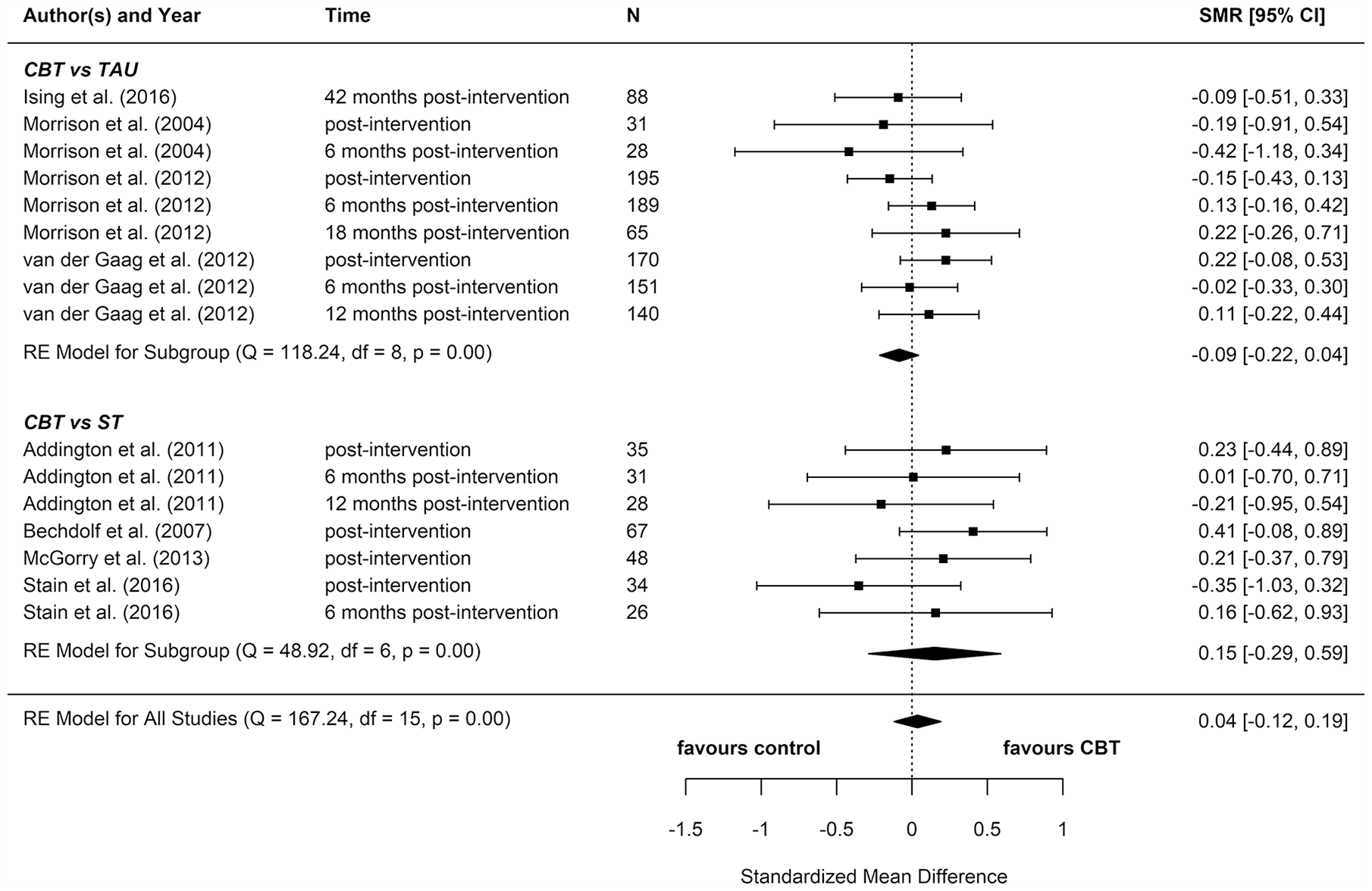
Functioning: meta-analysis summary plot (NB: follow-up times are measured from the end of the intervention).
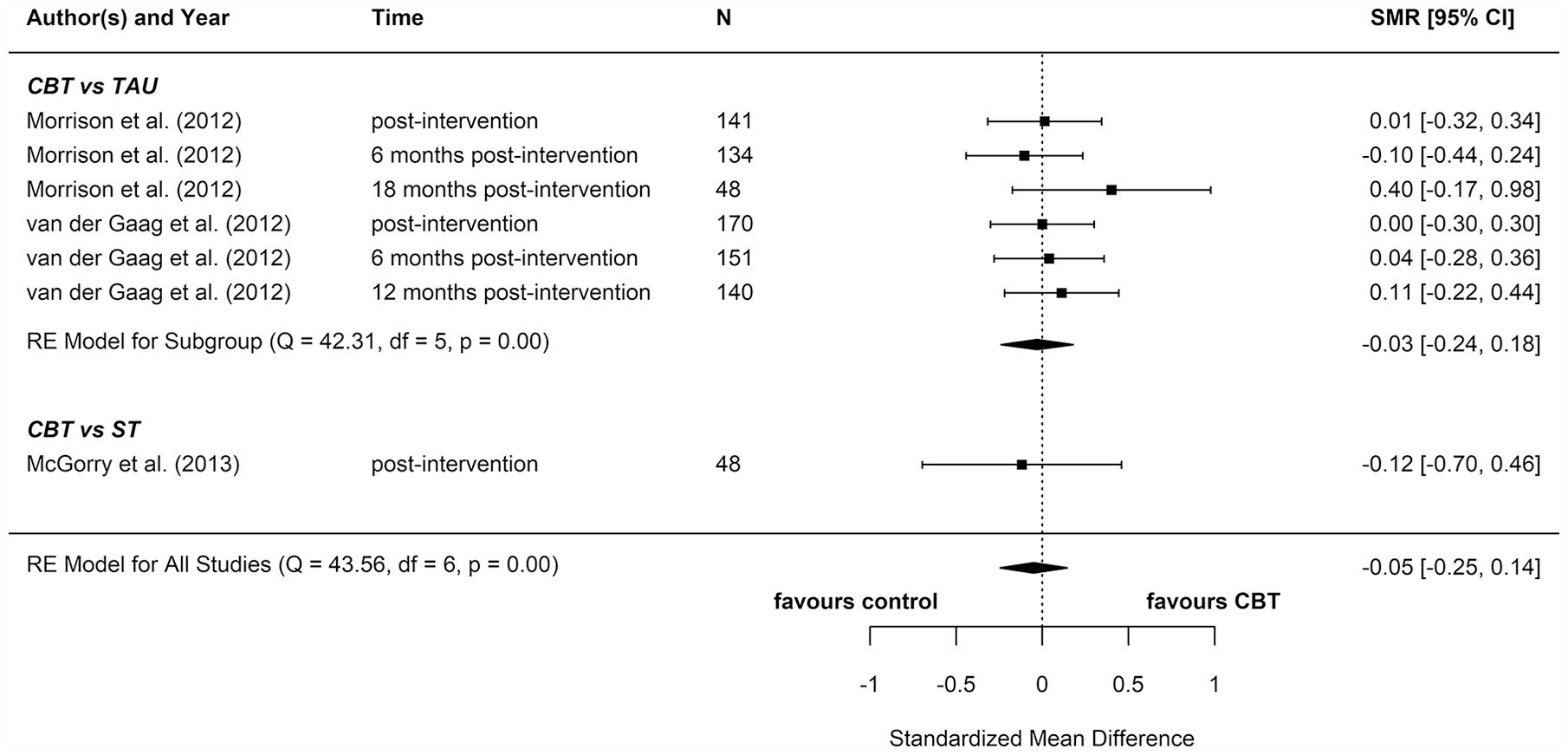
Quality of life: meta-analysis summary plot (NB: follow-up times are measured from the end of the intervention).
Four additional reports compared CBT with an ST (e.g. supportive therapy, supportive counselling or non-directive reflective listening). No statistically significant differences were found for positive psychotic symptoms (pooled SMD = −0.12 favouring CBT; 95% CI = [−0.61, 0.38]), negative psychotic symptoms (pooled SMD = 0.14 favouring ST; 95% CI = [−0.30, 0.57]), depression (pooled SMD = 0.09 favouring ST; 95% CI = [−0.33, 0.52]), anxiety (pooled SMD = −0.18 favouring CBT; 95% CI = [−0.71, 0.34]), or functioning (pooled SMD = −0.15 favouring CBT; 95% CI = [−0.29, 0.59]).
To determine whether there was a difference between the two different control groups, we included TAU and ST as predictors of SMD in a meta-regression model. Because there was not a statistically significant difference between the different control groups for any outcome, we also computed a pooled estimate for all reports regardless of comparator. When TAU and ST were collapsed into a single comparator group, CBT remained more effective than the combined TAU/ST comparison groups at reducing distress (pooled SMD: −0.23 favouring CBT; 95% CI = [−0.36, −0.10]). There were no other statistically significant differences between CBT and controls.
Most of these meta-analyses suffered from high heterogeneity (Cochran’s Q, p < 0.05). However, this measure is unreliable when the number of studies included is very low, so although heterogeneity cannot be ruled out, it is hard to ascertain its extent.
Two reports found significant between-group differences in severity of psychotic symptoms in two distinct trials, in each instance favouring cognitive therapy (p = 0.049 and p = 0.018, respectively) (Morrison et al., 2004, 2012). A further two reports found significant between-group differences in distress, but in opposite directions: while one found lower distress among participants in the CBT group (p = 0.012) (Van der Gaag et al., 2012), the other found lower distress among participants in the non-directive reflective listening (ST) group (p = 0.029) (Stain et al., 2016). No RCT found any statistically significant between-group differences for depression, anxiety, functioning, or quality of life.
In addition, reports from three controlled (Addington et al., 2011; Bechdolf et al., 2007; McGorry et al., 2017) and four uncontrolled studies (Bechdolf et al., 2005; Evans et al., 2017; Matsumoto et al., 2018; Stafford et al., 2015) provided results of significance tests for within-group pre–post changes for individuals receiving CBT (several more noted symptom improvement, but did not provide formal significance testing results). The three reports providing data on positive psychotic symptoms (Addington et al., 2011; Matsumoto et al., 2018; Stafford et al., 2015), and one of the three providing data on negative psychotic symptoms (McGorry et al., 2017) found significant improvement. Significant improvement was also noted in four of the five reports providing data on depression (Bechdolf et al., 2005; Evans et al., 2017; McGorry et al., 2017; Matsumoto et al., 2018) and functioning (Bechdolf et al., 2005, 2007; McGorry et al., 2017; Matsumoto et al., 2018), all four reports providing data on anxiety (Addington et al., 2011; Bechdolf et al., 2005; Evans et al., 2017; Matsumoto et al., 2018), one of the two providing data on distress (Evans et al., 2017), and in the one report that provided data on quality of life (Matsumoto et al., 2018). No study found statistically significant decline in any domain.
Supportive treatments
Reports from five controlled studies (Addington et al., 2011; Bechdolf et al., 2007; Phillips et al., 2007; Ruhrmann et al., 2007; Shi et al., 2017) provided results from significance testing for within-group pre–post changes for individuals receiving supportive or needs-focused treatments. Two of four reports providing data on positive psychotic symptoms (Addington et al., 2011; Ruhrmann et al., 2007), but none of the four providing data on negative psychotic symptoms, found significant improvement. Significant improvement was noted in two of four reports providing data on depression (Addington et al., 2011; Ruhrmann et al., 2007), one of two providing data on anxiety (Addington et al., 2011), one of five providing data on functioning (Bechdolf et al., 2007), and in the one report providing data on in quality of life (Phillips et al., 2007). No study found statistically significant decline in any domain.
Other intervention frameworks
Four additional RCTs focused on systemic therapy (Shi et al., 2017), MBCT (Langer et al., 2010), FFT (O’Brien et al., 2015), and cognitive remediation therapy (CRT) (Piskulic et al., 2015). Only the MBCT trial showed any between-group differences in our outcomes of interest. In this study, MBCT was more effective than the control condition (a video viewing forum) at reducing anxiety from baseline to post-therapy (d = 0.88, p = 0.012) as well as baseline to 12-week follow-up (d = 0.91, p = 0.048). However, they found no other significant between-group differences for psychotic symptoms or distress (Langer et al., 2010).
Systemic therapy, CRT and FFT were no more effective than their control treatments (supportive therapy, computer games, and family psychoeducation, respectively) (O’Brien et al., 2015; Piskulic et al., 2015; Shi et al., 2017). Although neither systemic therapy nor CRT was more effective than its control treatment, each showed within-group pre–post effects. Individuals who received systemic therapy showed significant reductions in positive symptoms (d = 0.53, p = 0.005) and depressive symptoms (d = 0.75, p = 0.010) from baseline to post-therapy, while no such changes were found for the supportive therapy group (Shi et al., 2017). Similarly, individuals assigned to CRT had significant improvements in social functioning (p < 0.05) from baseline to 6 months post-intervention, while those assigned to the computer games condition had no significant improvements (Piskulic et al., 2015).
A further two uncontrolled studies examined within-group pre–post effects of a strengths and mindfulness-based online social therapy (Alvarez-Jimenez et al., 2018) and a CBT intervention for sleep problems (Bradley et al., 2017). The former found significant improvements in social functioning (d = 1.83, p < 0.001) from baseline to post-intervention (Alvarez-Jimenez et al., 2018), and the latter found significant improvements in depression and quality of life (p < 0.05; exact values not given). These improvements were maintained at 1 month post-therapy, at which time improvement in paranoia and hallucinations also reached significance (p < 0.05; exact values not given) (Bradley et al., 2017).
Sub-group analyses
For sub-group analyses by quality, we were only able to perform two meta-analyses (for functioning and positive symptoms) due to the fact that in all other meta-analyses there was only one study without a high risk of bias in at least one category. We found no statistically significant difference between CBT and TAU in either sub-group analysis (see Supplemental Appendix E).
Components of effective interventions
We focused our components analysis on the five interventions that showed effectiveness for at least one outcome in controlled trials: three CBT (Morrison et al., 2004, 2012; Van der Gaag et al., 2012), one mindfulness-based cognitive therapy (Langer et al., 2010), and one non-directive reflective listening intervention (Stain et al., 2016) (intervention components in Supplemental Appendix D). Qualitative examination of the components of these five therapies revealed high heterogeneity: very few components were shared across the effective therapies, which is unsurprising given their differing frameworks. Furthermore, there were no ‘key ingredients’ that were particular to these five therapies: although there were some common components across the effective therapies (e.g. mode of delivery), these were also shared by therapies that did not demonstrate effectiveness.
Economic studies
Four reports met inclusion criteria for the economic component of the review (summary of studies’ characteristics in Supplemental Appendix F; quality assessment in Supplemental Appendix G; full economic analysis in Supplemental Appendix H). Two focused on CBT (Ising et al., 2015, 2017) and two on ST (Phillips et al., 2007, 2009).
CBT
Ising et al. (2015, 2017) reported the results of full economic evaluations in two reports, which were based on 18-month and 4-year post-baseline data, respectively, from a study conducted in the Netherlands between 2008 and 2010 comparing routine care plus CBT for the prevention of psychosis with routine care alone for individuals at ultra-high risk aged 14 to 35 years (Rietdijk et al., 2010). At 18 months post-baseline, the authors concluded that CBT proved to be cost-saving; however, differences in costs between groups were not tested statistically. When combined with outcome data, there was some evidence to suggest that CBT plus routine care may be cost-effective compared to routine care alone, but differences were small and no assessment of uncertainty was carried out. Results were clearer at 4 years post-baseline, with evidence to suggest a high probability (>80%) of the CBT group being cost-effective compared with routine care alone.
Supportive treatments
Phillips et al. (2007, 2009) explored resource use and cost-savings in two reports, both based on data from an RCT conducted in Australia between 1996 and 1999, which compared a NBI with NBI plus a specific preventive intervention (SPI) including psychotherapy and neuroleptic medication for individuals aged 14 to 30 years at ultra-high risk (McGorry et al., 2002). Phillips et al. (2007) explored resource use from a mental health service perspective between 12 and 36 months post-randomisation. Resource use was reported by group for some resource items and by those who did or did not develop psychosis for others. There was little difference in resource use with the exception of significantly higher mental health service use for those who did not develop psychosis in the control arm. However, sample sizes were small (total n = 41) and cost differences were not tested statistically. In Phillips et al. (2009), a cost-savings analysis was undertaken for the full 36-month post-baseline follow-up period. There were no significant differences in total cost between the groups over the full follow-up. In terms of outcomes (Phillips et al., 2007), no differences in the rate of transition to psychotic disorder, level of symptomatology, or functioning between the groups were identified, therefore indicating there may be no cost-effectiveness advantage of the intervention.
Discussion
This systematic review and meta-analysis included 27 reports concerning 21 studies of psychological interventions for PEs and aimed to determine their effectiveness and cost-effectiveness for improving a range of clinical and functional outcomes. In terms of the proportion of participants remitting from PEs, we found preliminary evidence from one RCT and one uncontrolled study for the potential effectiveness of CBT. We did not find meta-analytic evidence that CBT improved PEs on a continuous scale, though it is likely that our analyses were underpowered to detect small effects. While two individual RCTs favoured CBT over TAU for reducing the severity of psychotic symptoms, this effect was not consistent across all controlled studies. CBT, sleep CBT, and systemic therapy – but not ST – also showed promise in terms of within-group pre–post improvements in psychotic symptoms.
For our other non-psychotic secondary outcomes (depression, anxiety, functioning, distress and quality of life), only the meta-analysis of distress outcomes revealed evidence of comparative effectiveness, by which CBT was more effective than comparators. However, a high degree of heterogeneity cannot be ruled out in this meta-analysis, meaning that CBT may not reduce distress in all implementation scenarios in this patient population. Two individual trials showed a significant effect on distress, but in opposite directions. The only other RCT evidence of effectiveness was for mindfulness CBT, which significantly reduced participants’ anxiety symptoms. Low-quality evidence from uncontrolled studies showed that a number of therapies were effective for at least one non-psychotic clinical or functional outcome, including CBT, sleep CBT, systemic therapy, CRT, and mindfulness online social therapy. Supportive treatments were fairly effective at improving anxiety and depression, but not other outcomes.
The overall quality of studies included in the effectiveness component of the review was variable. While most reports (21 of the 27) focused on data from RCTs (the gold standard study design for investigating intervention effect), all but four of these received a rating indicating high risk of bias in at least one of the rating categories. High rates of attrition were the predominant reason for lower ratings, followed by high chance of selection bias. The six non-randomised, uncontrolled studies, although prone to the significant biases associated with low-quality study design, did not receive any rating indicating high risk of bias in any other applicable category (these were not rated in terms of blinding or confounders).
Economic data meeting the inclusion criteria were only available in four publications, which used data from two RCTs, one focusing on CBT and the other focusing on an intervention that included psychotherapy and antipsychotic medication. Both interventions were targeted at young adults at ultra-high risk of psychosis. The included economic studies were methodologically strong, meeting most of the Drummond checklist quality assessment criteria (Drummond and Jefferson, 1996). The economic studies focusing on CBT indicate that the addition of CBT to routine care has a high probability of being cost-effective compared to routine care alone in this ultra-high risk group.
Several previous systematic reviews and meta-analyses have examined the effectiveness of psychological, pharmacological, and nutritional interventions for people with PEs. Although most reviews focused primarily on transition (and four focused exclusively on transition), seven (Davies et al., 2018b; Devoe et al., 2019; Hutton and Taylor, 2014; Marshall and Rathbone, 2011; Okuzawa et al., 2014; Stafford et al., 2013; Van der Gaag et al., 2013) also reported selected secondary outcomes that do correspond with the current review’s focus, specifically psychotic symptoms (Davies et al., 2018b; Devoe et al., 2019; Marshall and Rathbone, 2011; Okuzawa et al., 2014; Stafford et al., 2013), distress (Hutton and Taylor, 2014; Okuzawa et al., 2014), depression (Marshall and Rathbone, 2011; Okuzawa et al., 2014; Stafford et al., 2013), anxiety (Marshall and Rathbone, 2011; Okuzawa et al., 2014), functioning (Hutton and Taylor, 2014; Marshall and Rathbone, 2011; Okuzawa et al., 2014; Van der Gaag et al., 2013), and quality of life (Hutton and Taylor, 2014; Marshall and Rathbone, 2011; Okuzawa et al., 2014; Stafford et al., 2013). Importantly, no prior review has included a consideration of remission from PEs. None of these reviews (including the review upon which current UK clinical guidelines are based) has found strong evidence to support the effectiveness of any particular psychological intervention for improving our outcomes of interest within this population. In general, these reviews reflect our own results. However, departing from previous findings, we found meta-analytic evidence that distress was significantly reduced after CBT as compared with control treatments (TAU/ST). It is possible that distress is a significant, under-measured, and under-reported outcome in the literature; indeed, only two previous reviews have reported distress as an outcome. Distress is an important factor to individuals with PEs as reductions can be interpreted as improvement, despite residual symptoms (Byrne and Morrison, 2014; Fowler et al., 2018; Law and Morrison, 2014); consequently, a broader consideration of this outcome is warranted.
Major treatment guidelines currently recommend CBT for the treatment of people at-risk for developing psychosis (Addington et al., 2017; Early Psychosis Guidelines Writing Group and EPPIC National Support Program, 2016; NICE, 2014; Schmidt et al., 2015). In the United Kingdom, NICE (2014) highlights the value of CBT for preventing transition to frank psychotic disorder. However, recent meta-analytic evidence published since the creation of these guidelines suggests that CBT for populations at-risk for developing psychosis may not be superior to other inventions in preventing transition (Davies et al., 2018a), although it is important to note that concerns have been raised about both the methodology and interpretation of results in this review (Nelson et al., 2018a). Our findings provide initial evidence that, while doubts remain about its effectiveness in terms of preventing transition to psychosis, CBT may nevertheless be more effective than other approaches at promoting remission from PEs and reduction of associated distress, and thus may still be considered as a potentially useful intervention for treating people with PEs. Conversely, when the aim of psychological intervention is to reduce other clinical symptoms (e.g. depression and anxiety) or functional impairment associated with PEs, CBT falls short in demonstrating effectiveness as compared with other treatments. This is an important shortcoming, as poor clinical and functional outcomes may serve to perpetuate mental ill health that may still require more than just monitoring for changes in post-CBT persistent symptoms, as currently recommended by NICE (2014).
Strengths and limitations
This review has a number of important strengths and addresses key gaps in the literature concerning psychological interventions for people with PEs. First, to our knowledge, we were the first to meta-analyse studies across such a broad range of clinical and functional outcomes. Second, we focus on remission from PEs, a new and important outcome that was developed in collaboration with our lived experience advisory panel. Third, we include economic outcomes, which again have not been reviewed previously. Fourth, we review a large number of studies not included in any other review, including, importantly, studies of newer, non-CBT frameworks.
These strengths notwithstanding, our review, and in particular our meta-analyses, has a number of limitations. First, each meta-analysis included a small number of reports, each of which had a limited number of participants (sometimes short of the recruitment target). This will have reduced our power to detect small, but potentially clinically meaningful, treatment effects. We aimed to increase power by including multiple study follow-up points within each meta-analysis. Although we could also have combined outcomes to reduce the total number of meta-analyses (and also the probability of type I error), we chose not to do this as (1) sometimes outcomes changed in different directions following intervention (e.g. see Langer et al., 2010), and (2) Cochrane warns against combining heterogeneous outcomes (see section 9.1.4) (Higgins and Green, 2011). Second, the high number of meta-analyses performed will have increased the probability of false positive results, which is particularly important in our analyses due to the fact that we found only one significant effect. Third, we could not rule out high heterogeneity within our meta-analyses. Fourth, our decision to group several therapy types under ‘supportive therapy’ was not without limitations; for example, patients under TAU conditions may well receive CBT for other mental health problems outside of PEs (e.g. depression or anxiety). Fifth, our exclusion criteria regarding age range and antipsychotic use may limit the generalisability of our findings to younger populations or patients prescribed antipsychotic medication as part of their treatment plan. Sixth, in terms of the studies themselves, while many utilised randomised controlled designs, the overall methodological quality was not high; only four studies received a global rating of ‘high’ on the quality rating tool. Finally, we acknowledge that we were not able to fulfil all a priori review aims. While the review was ambitious, we contend that it was not possible to predict which aims could and could not be accomplished. Furthermore, we believe that highlighting gaps in the literature is an important step in moving the field forward.
Conclusion
This review has clear clinical relevance and will be central in the development of a new therapeutic framework for IAPT, as well as for other programmes aiming to address PEs in primary mental healthcare settings internationally. The broad aims, comprehensive outcomes, and specific selection criteria all reflect this purpose. The review will ensure any decisions concerning treatment development and treatment selection for people with PEs within primary care are supported by the most recent and high-quality evidence. Overall, our findings indicate that clinicians must consider a wider range of clinical and functional outcomes as well as interventions for people with PEs that go beyond strategies for preventing transition to psychotic disorders. Our systematic review and meta-analysis suggest that, despite its limited effectiveness in preventing transitions, CBT may be useful to reduce the distress associated with PEs and cost-effective in comparison with treatment as usual. However, the scarcity of studies focusing on remission from PEs and improvement of other non-psychotic clinical and functional outcomes suggests a need for further research into psychological treatments for this population.
Supplemental Material
Soneson_Russo_et_al_appendices_R1 – Supplemental material for Psychological interventions for people with psychotic experiences: A systematic review and meta-analysis of controlled and uncontrolled effectiveness and economic studies
Supplemental material, Soneson_Russo_et_al_appendices_R1 for Psychological interventions for people with psychotic experiences: A systematic review and meta-analysis of controlled and uncontrolled effectiveness and economic studies by Emma Soneson, Debra Russo, Jan Stochl, Margaret Heslin, Julieta Galante, Clare Knight, Nick Grey, Joanne Hodgekins, Paul French, David Fowler, Louise Lafortune, Sarah Byford, Peter B Jones and Jesus Perez in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors are very grateful to medical librarians Veronica Phillips and Isla Kuhn at the University of Cambridge Clinical School, who helped design the search strategies for this review. The authors also thank the members of our Lived Experience Advisory Panel for their contributions to the design of this research.
Author Contributions
J.P., P.B.J., L.L., D.R., J.G., J.S. and E.S. conceived the review design. J.P. is the guarantor of the review. E.S., D.R., C.K., M.H., J.S., N.G., J.H., P.F., D.F., S.B., P.B.J and J.P. contributed to the design of the search strategy. E.S., D.R., J.S., M.H., S.B. and J.P. drafted the original manuscript draft. C.K., L.L., J.G., J.H., P.F., D.F., N.G., S.B. and P.B.J. contributed to the review of manuscript drafts. All authors approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.B., D.F., P.F., J.G., J.H., C.K., L.L., J.P., D.R. and E.S. declare no conflicts of interest. N.G. reports grants from Wellcome Trust and personal fees from book publishers and training events (outside the submitted work). P.B.J. reports personal fees from Lundbeck, Ricordati, and Janssen scientific advisory boards (outside the submitted work). J.S. reports personal fees from IESO Digital Health (outside the submitted work).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This paper presents independent research funded by the National Institute for Health Research (NIHR) under its Programme Grant for Applied Research Programme (reference number RP-PG-0616-20003). The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care. The funding body had no role in study design; collection, analysis, or interpretation of data; the writing of the report; or decision to submit the paper.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
