Abstract
Background:
Depressive disorders are significantly more common in the medically ill compared to the general population. Depression is associated with worsening of physical symptoms, greater healthcare utilization and poorer treatment adherence. The present paper provides a critical review on the assessment and management of depression in the medically ill.
Methods:
Relevant articles pertaining to depression in the medically ill were identified, reviewed and synthesized qualitatively. A systematic review was not performed due to the large breadth of this topic, making a meaningful summary of all published and unpublished studies not feasible. Notable studies were reviewed and synthesized by a diverse set of experts to provide a balanced summary.
Results:
Depression is frequently under-recognized in medical settings. Differential diagnoses include delirium, personality disorders and depressive disorders secondary to substances, medications or another medical condition. Depressive symptoms in the context of an adjustment disorder should be initially managed by supportive psychological approaches. Once a mild to moderate major depressive episode is identified, a stepped care approach should be implemented, starting with general psychoeducation, psychosocial interventions and ongoing monitoring. For moderate to severe symptoms, or mild symptoms that are not responding to low-intensity interventions, the use of antidepressants or higher intensity psychotherapeutic interventions should be considered. Psychotherapeutic interventions have demonstrated benefits with small to moderate effect sizes. Antidepressant medications have also demonstrated benefits with moderate effect sizes; however, special caution is needed in evaluating side effects, drug–drug interactions as well as dose adjustments due to impairment in hepatic metabolism and/or renal clearance. Novel interventions for the treatment of depression and other illness-related psychological symptoms (e.g. death anxiety, loss of dignity) are under investigation.
Limitations:
Non-systematic review of the literature.
Conclusion:
Replicated evidence has demonstrated a bidirectional interaction between depression and medical illness. Screening and stepped care using pharmacological and non-pharmacological interventions is merited.
Keywords
Introduction
Major depressive disorder (MDD) is a common and disabling illness, with an annual prevalence of 2–4% in the general population (Global Burden of Disease (GBD) DALYs and Hale Collaborators et al., 2015). The prevalence of MDD is approximately doubled in the medically ill with an estimated annual prevalence of 5–10% (Read et al., 2017). The interaction is likely bidirectional as MDD is also independently associated with an increased prevalence of chronic medical disorders. Comorbid medical illness and MDD are associated with reciprocally poorer psychiatric and medical outcomes (Barth et al., 2004; Darwish et al., 2018; Penninx, 2017; Rutledge et al., 2006). Depression is associated with greater physical symptom burden, poorer treatment adherence and quality of life, increased health care utilization and increased all-cause mortality in individuals with chronic medical illnesses (Barber et al., 2016; Barefoot et al., 1996; Barth et al., 2004; Bhattarai et al., 2013; Fitzgerald et al., 2013; Gold et al., 2016; Mausbach and Irwin, 2017). Furthermore, Gallo et al. (2013) demonstrated a reduction in all-cause mortality in primary care in elderly patients given evidence-based treatments for MDD, as compared with treatment as usual. Notably, however, a recent umbrella review strongly suggests that the association between depression and all-cause mortality is not necessarily causal (Machado et al., 2018).
The assessment and management of MDD in the medically ill has several important differences and special considerations compared to MDD in the general population (Ramasubbu et al., 2012). Thus, the aim of the present review is to provide an up-to-date critical appraisal of available evidence on this issue. The following topics will be discussed: (1) epidemiology and pathogenesis, (2) evidence-based screening practices, (3) the accurate diagnosis of MDD in the medically ill and (4) prevention and management strategies. Unanswered questions and high-priority areas for future research will also be discussed. Of note, a systematic review was not performed due to the large breadth of this topic, making a meaningful summary of all published and unpublished studies not feasible. Notable studies were reviewed and synthesized by a diverse set of experts to provide a balanced summary.
Epidemiology and pathogenesis
Several observational and epidemiological studies have demonstrated an increased incidence of both MDD and depressive symptoms in people with chronic medical illnesses. However, the accuracy of prevalence estimates of MDD in the medically ill is often questioned, given significant limitations in study design, such as inaccurate case identification (Thombs et al., 2018). Most studies have relied on self-report questionnaires, such as the Patient Health Questionnaire (PHQ-9) (Kroenke et al., 2001). Validated structured diagnostic interviews are considered to be the ‘gold standard’ for the diagnosis of MDD in research (Levis et al., 2017). However, even with a structured interview, the diagnosis of MDD in medically ill populations is complex with no agreed upon ‘gold standard’ for confirming the diagnosis, leading to high inter-rater variability (Galeazzi et al., 2004; Koenig et al., 1995). In addition, sampling methods are often biased using convenient samples from clinics or academic tertiary care centers (Bornstein et al., 2013; Matte et al., 2016; Mikocka-Walus et al., 2016; Wells et al., 1988). More recently, the use of healthcare administrative databases has facilitated acquisition of more generalizable samples to decrease the inherent bias of convenient sampling (i.e. Berkson’s bias), although such databases may have limited reliability as the diagnosis is typically clinical and not standardized (Lo et al., 2013; Ratnasingham et al., 2013; Videbech and Deleuran, 2016).
While acknowledging these limitations, estimates of MDD in chronic medical illness have been consistently reported to be greater than twofold higher than the general population, in addition to the high prevalence of clinically relevant subthreshold depressive symptoms in this population (Li et al., 2019). Non-communicable medical and common psychiatric disorders may share many individual, social and biological pathways (O’Neil et al., 2015). While the magnitude of the association may be questioned, the presence of a statistically and clinically significant association between medical illness and depression is now rarely debated, given the consistent replication of this finding (Li et al., 2019). Furthermore, the presence of multiple medical conditions (e.g. ‘multimorbidity’) appears to be associated with even higher rates of depression and poorer medical and psychiatric outcomes (Gallo et al., 2016; Read et al., 2017).
Neurological disorders are strongly linked with increased rates of MDD, with an estimated lifetime prevalence of 20–40% across stroke, epilepsy, multiple sclerosis (MS), traumatic brain injury (TBI), Alzheimer’s disease (AD) and Parkinson’s disease (PD) (Bhattarai et al., 2013; Diniz et al., 2013; Elger et al., 2017; Rickards, 2005). Depression is associated with poorer medical outcomes and increased suicidality in neurological disorders (Bahraini et al., 2013; Bartoli et al., 2017; Christensen et al., 2007; Go et al., 2013; Pan et al., 2011; Pompili et al., 2012, 2006; Rickards, 2005; Robinson and Jorge, 2016). Of note, the diagnosis of comorbid MDD in AD and PD is controversial, with some investigators and clinicians advocating that depressive symptoms are part of the core features of these neurological disorders, rather than a comorbid syndrome on its own right, even when meeting full criteria for MDD (Caraci et al., 2010; Dauer and Przedborski, 2003).
Non-neurological disorders are also associated with significantly elevated rates of MDD, including cancer (lifetime prevalence of MDD of 10–20%), diabetes (6–20%), cardiovascular diseases (CVD; 15–25%), HIV (25–40%), chronic obstructive pulmonary disease (COPD; 20–30%) and autoimmune disorders (10–30%), such as irritable bowel disease (IBD), lupus, psoriasis and rheumatoid arthritis (RA) (Ali et al., 2006; Ciechanowski et al., 2000; Eshun-Wilson et al., 2018; Geng et al., 2018; Han et al., 2011; Palagini et al., 2013; Penninx, 2017; Pérez-Stable et al., 1990; Thom et al., 2019; Wang et al., 2016). In these chronic medical illnesses, MDD is also associated with poorer medical outcomes, increased health care utilization, decreased treatment adherence and poorer health-related quality of life. Increased all-cause mortality and disease progression has been associated with comorbid MDD specifically in cancer, HIV, CVD and diabetes; however, a causal relationship has never been clearly established (Cuijpers et al., 2014; Machado et al., 2018).
The bidirectional interaction between depression and medical illness is mediated by common biological, iatrogenic, psychological, social and lifestyle risk factors, as shown in Table 1. Common lifestyle risks include physical inactivity, smoking and poor diet (Berk and Jacka, 2019; Pasco et al., 2008; Strawbridge et al., 2002). Psychosocial factors associated with depression that are common to many chronic medical illnesses include the impact of physical limitations, disability, role transitions and loss of independence/autonomy (Thom et al., 2019). Many illnesses, such as HIV, continue to have significant stigma, often leading to social isolation (Green et al., 2012; Van Luenen et al., 2018). Biological disease-specific factors may also increase the incidence of depressive symptoms (Table 1). For example, immune activation and increased oxidative and nitrosative stress pathways in peripheral blood may explain the comorbidity between depression and peripheral autoimmune and immune disorders, including CVD, IBD and lupus erythematosus, as well as increased morbidity and mortality due to depression in these peripheral immune disorders (Berk et al., 2013b; Maes et al., 2011). Increased neuroinflammation coupled with oxidative stress may explain the comorbidity between depression and neurodegenerative/neuroinflammatory disorders including MS, PD, stroke and AD (Berk et al., 2013b; Maes et al., 2011). These factors may also mediate the accelerated cellular aging associated with depression, as shown by shortened leukocyte telomere length (LTL), which may also increase the risk of several medical illnesses, such as cancer, CVD and dementia (Muneer and Minhas, 2019).
Formulation of depression in the medically ill using the biopsychosocial model.
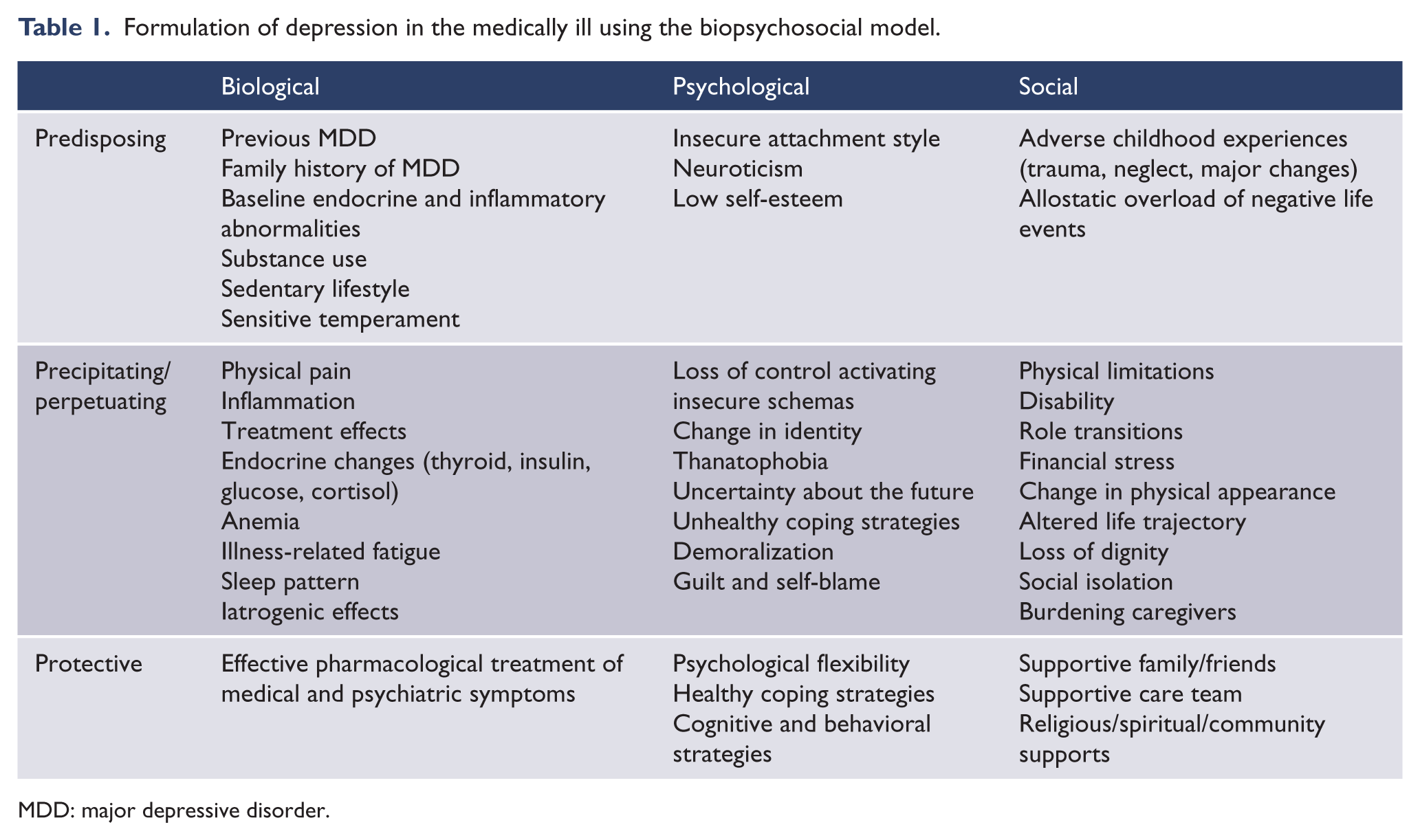
MDD: major depressive disorder.
Inflammation associated with autoimmune disorders, chronic infections, CVD, stroke and many cancers often causes anergic depressive symptoms (e.g. ‘sickness behavior’) mediated by pro-inflammatory cytokines causing downstream effects, including decreased monoamine levels, microglial over-activity and impaired neuroplasticity (Dantzer et al., 2008; Rosenblat et al., 2014). Similarly, the hypodopaminergic effects of PD, neurotoxic effects of insulin resistance, sleep apnea-related hypoxemia of the brain, and the direct effects of hypothyroidism on energy, mood and cognitive function represent other disease-specific biological pathways mediating the high rates of comorbidity between medical illness and depressive symptoms (Banks et al., 2012; Dauer and Przedborski, 2003; Irwin et al., 2016; Loas et al., 2012; Ramasubbu et al., 2012). The iatrogenic depressant effects of medications used to treat chronic medical illness (e.g. barbiturates, vigabatrin, topiramate, flunarizine, corticosteroids, mefloquine, beta blockers [association more recently questioned], efavirenz, interferon-α) may also have a relevant role (Agustini and Berk, 2018; Celano et al., 2011; Machado et al., 2017).
Screening for depression
Effective identification of depression in the medically ill is of great importance, given its high prevalence and the deleterious effects on quality of life, treatment adherence, healthcare utilization and medical outcomes (Ciechanowski et al., 2000). Depression is often underdiagnosed in the medically ill (Hermanns et al., 2013; Pérez-Stable et al., 1990). The propensity to ‘miss’ the diagnosis of MDD is multifactorial and partly relating to time pressures, competing clinical demands and patient and clinician discomfort with discussing psychological distress (Chen et al., 2016; Saver et al., 2007). In addition, diagnostic overshadowing may cause attribution of physical symptoms (e.g. insomnia, fatigue, decreased appetite) exclusively to the underlying medical condition. Notably, diagnostic overshadowing with MDD commonly also has the opposite effects, with clinicians at times misattributing some physical symptoms as ‘psychosomatic’ when a diagnosis of MDD is known, thus delaying investigations of potential reversible underlying medical causes (Byrne, 2018).
Paradoxically, although depressive disorders are often undetected in many medical settings, they may also be over-diagnosed, at times resulting in inappropriate treatment with antidepressants. The factors that contribute to these symptoms should be identified to ensure appropriate intervention, which include attention to pain or other physical distress, social or psychological factors (Grover et al., 2017; Iaboni et al., 2015; Kose et al., 2018). To prevent the over-diagnosis of MDD in the medically ill, an understanding of the spectrum of sadness is required. Indeed, upon receiving and living with a diagnosis of a chronic medical illness, an expression of sadness may be a normal and expected adaptive reaction. However, symptoms that exceed the normative, expected reactions and are causing significant distress and/or functional impairment may represent a comorbid depressive or adjustment disorder (APA, 2013). A careful assessment is required to determine where along the spectrum these depressive symptoms lie. Furthermore, psychological and physical symptom burden may change over the course of the illness, meriting regular reassessment, rather than one-time screening. Systematic depression screening practices may therefore be of value in the medically ill (Caruso et al., 2017b; Li et al., 2019).
International guidelines for most chronic medical conditions recommend routine screening for depression, as part of the standard of care (Butow et al., 2015; McIntyre et al., 2012; Reusch and Manson, 2017; Siu et al., 2016). Optimal screening methods should have the following key attributes: (1) high sensitivity and specificity, (2) ease of use by providers and patients, (3) low cost and (4) use of available and actionable approaches to confirm the diagnosis and implement evidence-based treatments (McAlpine and Wilson, 2004; Siu et al., 2016). Use of screening tools should always be part of a two-stage assessment process, with positive screens followed by further clinical assessment. A positive screen should lead to a clinical assessment to determine whether further psychological, social or biological interventions are required. Screening tools are not diagnostic for a depressive disorder and should never replace clinical assessment. Items 1–3 are related to the psychometric properties of screening tools and cut-off values selected, whereas item 4 relates to the algorithm implemented with a positive screen. Systematic reviews have demonstrated that screening for depression on its own does not improve outcomes (Thombs et al., 2014); however, systematically combining screening with actionable interventions and monitoring has demonstrated benefits for clinical outcomes (Gilbody et al., 2006, 2008; Meijer et al., 2011; Thombs et al., 2013).
Numerous depression screening tools have been assessed in people with chronic medical conditions, with variable clinical utility, given differences in specificity, sensitivity and patient/provider burden (e.g. time required to complete scale). Low sensitivity may lead to under-diagnosis, whereas low specificity may lead to inappropriately high rates of positive screens with unsustainable over-referral to specialized psychiatric/psychological services (Chen et al., 2016). Depression screening tools can be broadly categorized into very brief screening tools (e.g. single-item or two-item questionnaires) or full-length screening tools, typically with 10–20 self-report items. Very brief screening tools have a main advantage of low patient burden, whereas some longer screening tools have the advantage of increased sensitivity and specificity.
The most commonly used single-item tool is the Distress Thermometer, a 10-point Likert-type scale measuring level of distress in the past week. While easy to use, the clinical utility is limited as it has low sensitivity and specificity with poor positive predictive value, regardless of time-frame or cut-off scores selected (Grassi et al., 2009; Pantilat et al., 2012; Zwahlen et al., 2008). Other single-item screening practices use single questions with ‘yes’ or ‘no’ response options, such as ‘Are you depressed?’ or ‘Do you often feel sad or depressed?’ These single-question screening tools have been evaluated in medical populations with improved sensitivity compared to the Distress Thermometer, but they still have poor specificity, yielding high false positive rates (Frazier et al., 2014; Siu et al., 2016). The two-item questionnaire (Patient Health Questionnaire—2-item depression module [PHQ-2], asking about low mood and anhedonia) has demonstrated good sensitivity but poor specificity (Arroll et al., 2010). Therefore, the PHQ-2 is sometimes used as a pre-screener to trigger completion of a PHQ-9, which has shown good sensitivity and specificity in medical populations (80–90%) (Manea et al., 2012; Van Steenbergen-Weijenburg et al., 2010). Notably, however, cut-off scores used for a ‘positive screen’ are often higher than those in the general population (PHQ-9 > 10), since baseline somatic symptoms in medical illness elevate scores on most screening tools (Van Steenbergen-Weijenburg et al., 2010; Wasteson et al., 2009).
The four most commonly used full-length depression screening tools validated in chronic medical illness include the PHQ-9, the Center for Epidemiologic Studies Depression Scale (CES-D) (Cosco et al., 2019), the Beck Depression Inventory-II (BDI-II) (Steer et al., 1999) and the Hospital Anxiety and Depression Scale (HADS) (Cosco et al., 2012). All four scales have been translated and validated in numerous languages and in a variety of chronic medical illnesses (Siu et al., 2016). The CES-D and PHQ-9 are freely available, whereas the HADS and BDI-II are under copyright and must be purchased. The PHQ-9 and BDI-II are more specific to depressive symptoms, whereas the HADS and CES-D also capture non-specific psychological distress, with poorer specificity for MDD (Siu et al., 2016). Given that the PHQ-9 broadly maps onto the Diagnostic and Statistical Manual of Mental Disorders (5th edition; DSM-5) criteria, has been widely validated, is freely available and is relatively brief, its effective use as a depression screening tool continues to grow worldwide, with validated screening cut-offs in specific sub-populations (Manea et al., 2012). The properties of these commonly used screening tools are further summarized in Table 2.
Commonly used screening tools validated in the medically ill.
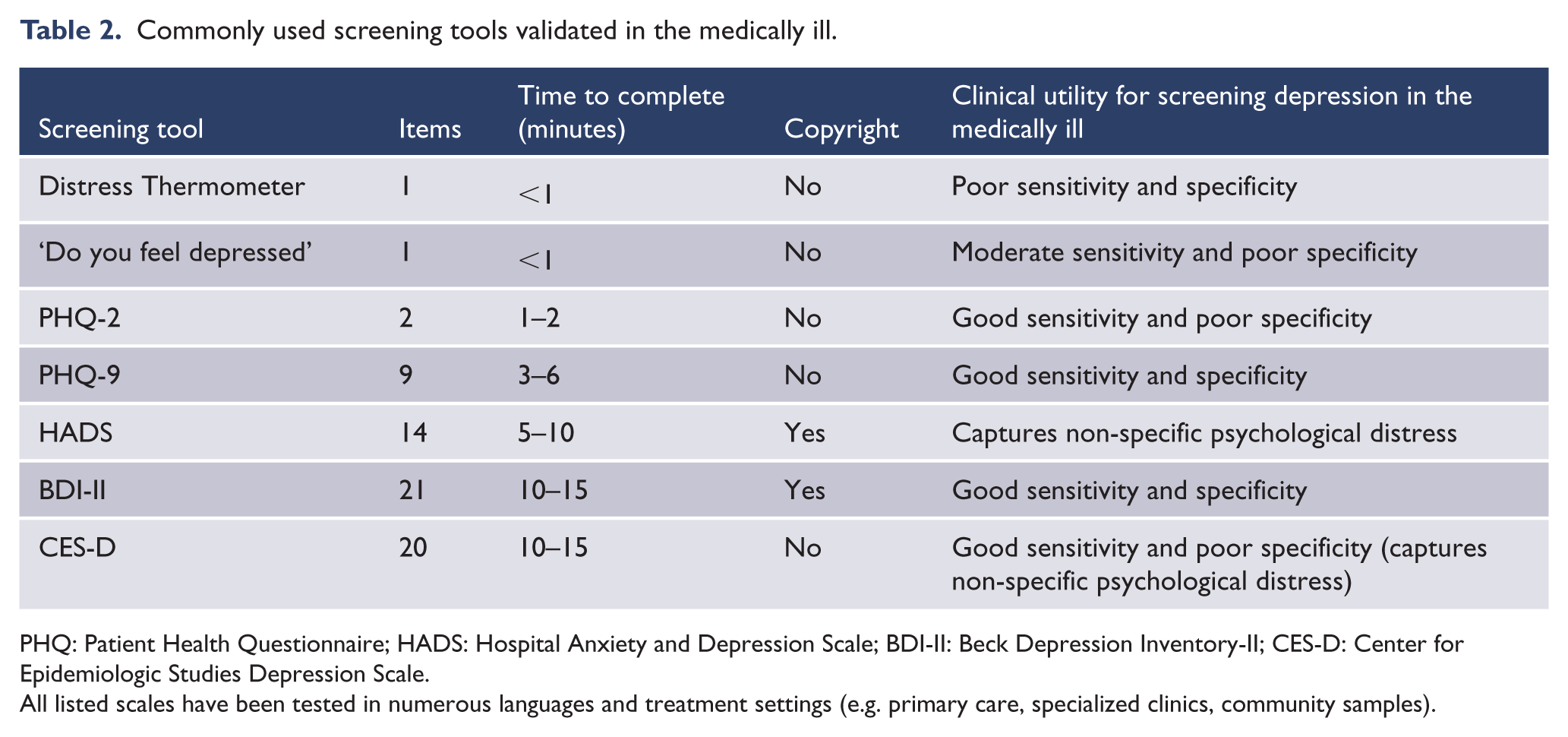
PHQ: Patient Health Questionnaire; HADS: Hospital Anxiety and Depression Scale; BDI-II: Beck Depression Inventory-II; CES-D: Center for Epidemiologic Studies Depression Scale.
All listed scales have been tested in numerous languages and treatment settings (e.g. primary care, specialized clinics, community samples).
Notably, a brief version of the BDI, the BDI-6, has shown good psychometric properties and has been recommended as a second step in the some screening procedures, since it has been validated to capture the severity of depressed mood (Blom et al., 2012). In addition, screening tools for sub-domains of depression, such as cognitive dysfunction, are being validated in the medically ill, given the importance of specific symptoms domains (e.g. cognition, anhedonia, energy) which may differentially respond to treatment (McIntyre et al., 2019; Siu et al., 2016).
In summary, numerous screening tools are available, with variable sensitivity, specificity, cost and acceptability. The use of a systematic screening method in chronic medical illness may increase the detection of depression. In addition, the use of tools should be made under the concept of incremental validity (Derogatis, 1987). A critical component of the screening process is an appreciation that a positive screen is not diagnostic, but rather, should prompt the screening provider to conduct a clinical assessment to better understand the depressive symptoms, prior to planning or initiating treatment. Harm may occur when screening tests are used to substitute a diagnostic assessment, as false positive screens may result in unnecessary initiation of antidepressant medications with associated risks to the patient including the emergence of ‘iatrogenic comorbidities’ (Carvalho et al., 2016; Fava et al., 2016). The reporting of depressive symptoms on a screening measure should trigger a clinic-based conversation with the patient to determine the most appropriate intervention, rather than an automatic referral for specialized psychiatric care. While assessing distress, conversations with primary and secondary health care providers, treatment of poorly controlled physical symptoms and engagement of social work, spiritual care or other support services may all be of value in this circumstance to address factors that contribute to depressive symptoms as a final common pathway of distress (Lo et al., 2010; Thombs et al., 2018).
Assessing depressive symptoms: the spectrum from normal sadness to subthreshold depressive symptoms to MDD
In assessing depressive symptoms, a comprehensive differential diagnosis of potential psychological, social, medical and psychiatric causes should be considered (Box 1), along with an appreciation for the spectrum of depressive symptoms (Figure 1). Sadness is a normal adaptive reaction to a new diagnosis of a chronic medical illness, to a recurrence of a medical condition or to a chronic illness (Chochinov, 2001). With milder levels of depression, patients may still be able to maintain closeness with their family and friends and to still enjoy activities, even if these activities are now different given their new physical and functional limitations (Caruso et al., 2017a). Maintaining some hope for the future and motivation to engage with the treating team would also be expected with normal sadness. Depressive symptoms that progress and cause distress and functional impairment may represent threshold (i.e. MDD) or subthreshold depressive symptoms. At this point, patients may begin to withdraw, potentially losing hope and motivation to receive treatment, even when the prognosis is favorable (Caruso et al., 2017a; Tecuta et al., 2015). As depressive symptoms progress further, the patient may meet criteria for a depressive disorder, with characteristic neurovegetative symptoms and negative cognitions distorting their views of themselves, the people around them and what the future will hold (APA, 2013). More significant social withdrawal, further worsening of low mood, anhedonia and worsening of physical symptoms would also be expected with an underlying depressive disorder.
Key differential diagnoses for depressive symptoms in medically ill.

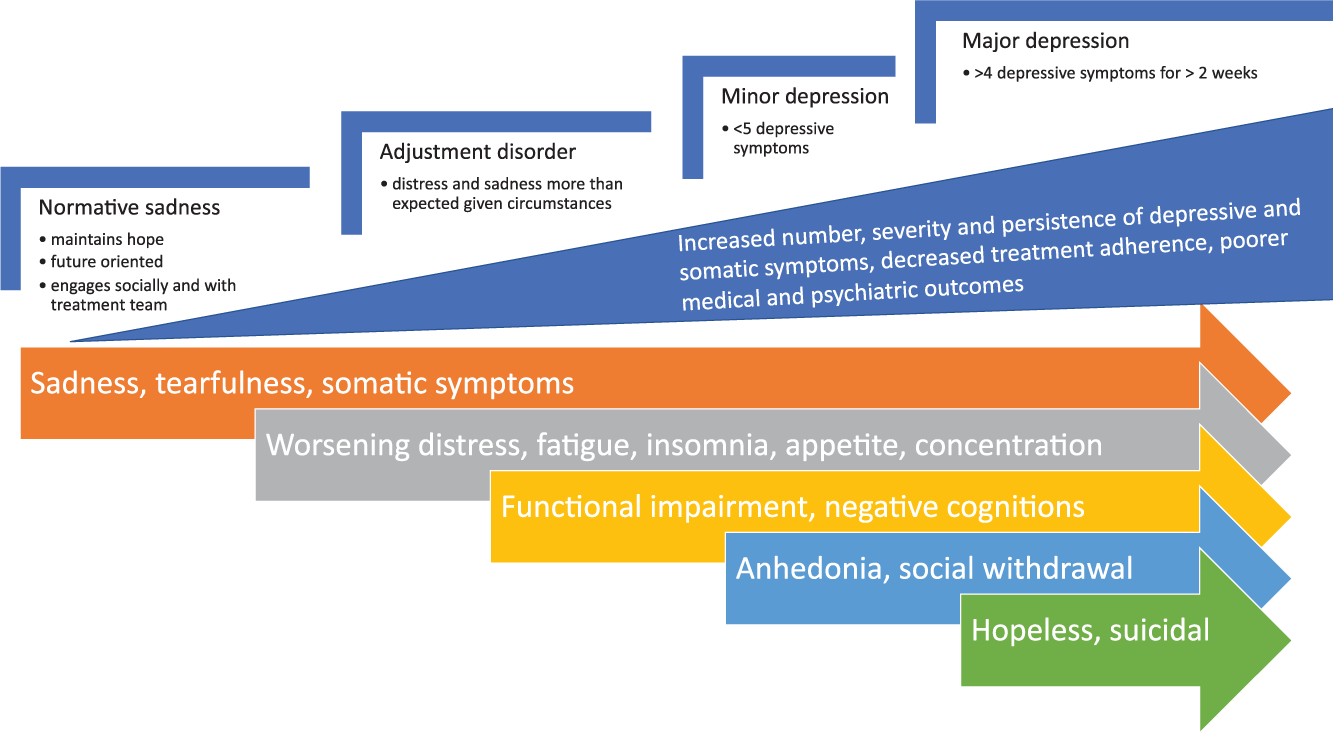
Spectrum of depressive symptoms from normal sadness to major depression.
In more severe cases, profound hopelessness and suicidal ideation may emerge. At times, suicidal ideation and behaviors secondary to depression may be difficult to differentiate from desire for hastened death (DHD), relating to their medical illness (Bellido-Pérez et al., 2017; Wilson et al., 2016). Differentiating suicidal thoughts and behaviors from DHD may be particularly important in regions where assisted dying is legalized for terminal medical illness (Sheehan et al., 2017). Notably, several medical illnesses, including cancer (Calati et al., 2017), end-stage renal disease (ESRD) (Pompili et al., 2013), HIV (Green et al., 2012), epilepsy (Christensen et al., 2007; Pompili et al., 2006), MS (Pompili et al., 2012), stroke (Bartoli et al., 2017) and TBI (Bahraini et al., 2013), are associated with increased rates of suicidal ideation, suicide attempts and death by suicide, meriting a detailed risk assessment in the medically ill with depressive symptoms. Of note, earlier research suggested that diabetes was also associated with increased suicide risk, but more recent meta-analytic level evidence has suggested there is no increased relative risk compared to appropriately matched controls (Wang et al., 2016). Added caution is needed regarding prescription dispensing intervals with suicidal patients, given the frequent large number of potentially lethal medications being prescribed and increased risk of overdose in some medical illnesses, such as HIV (Green et al., 2012).
Understanding and appreciating where depressive symptoms lie on this spectrum of severity is essential for accurately determining the diagnosis of depression in the medically ill. An initial assessment of the number of symptoms, severity, distress level and degree of functional impairment is needed to determine if depressive symptoms are subthreshold (e.g. less than five depressive symptoms) or above the threshold for a major depressive episode (MDE) (e.g. five or more symptoms with functional impairment occurring for greater than 2 weeks) (APA, 2013). In the medically ill, if symptoms are subthreshold and not better explained by the direct effects of medications, substances or a medical condition, the most likely diagnosis is an adjustment disorder (Mitchell et al., 2011). Adjustment disorders are commonly diagnosed conditions in the medically ill (15–30% lifetime prevalence) when presenting with symptoms of depression or anxiety (Mitchell et al., 2011). The diagnosis of an adjustment disorder requires symptoms to be disproportionate to what would be expected under the circumstances (e.g. diagnosis of medical illness), be functional impairing and/or subjectively distressing, although such distinctions may be difficult or impossible to determine. Importantly, while many providers may feel the diagnosis of adjustment disorder is less stigmatizing and more palatable for medical patients, if the criteria for MDD are met, this diagnosis takes priority (APA, 2013). This distinction is more relevant with regard to more severe depressive disorders that require antidepressant medication.
In establishing the diagnosis of MDD, several other important potential causes for symptoms should be ruled out (Box 1). Clearly establishing the chronology of symptoms is often helpful in refining the differential diagnosis. Symptoms starting acutely with a new medical illness may often relate to: (1) direct physiologic effects of the illness, (2) direct effects of a new medication being prescribed or (3) psychological distress relating to receiving a new diagnosis, along with adjusting to new physical limitations and role changes. Individual symptoms, such as tearfulness or apathy, can be associated with specific neurological disorders and are not necessarily indicative of an underlying, evolving depressive episode. In patients with a history of MDD, a new medical diagnosis is a significant risk factor for relapse or recurrence of depression in someone who may have previously been fully remitted (Rabiee et al., 2016). As in the general population, a thorough history of previous mood episodes (depressive, manic, hypomanic or mixed) and personal history is of great significance for an accurate diagnosis and biopsychosocial formulation that will directly impact the management plan.
People with personality difficulties are at particular risk of adjustment difficulties in the face of stressors, such as medical illness, that characteristically manifest as depressive symptoms. While beyond the scope of this paper, other factors such as personality disorder can be a powerful mediator of the relationship between medical and psychiatric comorbidity. Personality disorder is a risk for medical multimorbidity and independently is associated with a greater risk for depression as well as poorer outcomes in both groups of disorder (Quirk et al., 2015, 2016, 2017).
In establishing an MDD diagnosis, special attention should be paid to assessing the underlying etiology of specific symptoms, to differentiate symptoms primarily due to a depressive disorder, to a medical condition, or to both, or are compounded by comorbid personality difficulties. If a symptom is clearly secondary to a medical condition (e.g. fatigue in untreated hypothyroidism), the symptom should not be ‘counted’ toward a diagnosis of MDD when determining if greater than or equal to five symptoms of depression are present. However, it is often difficult to determine if somatic symptoms are entirely driven by a medical condition, as medically ill patients with depression report higher somatic symptom burden (Fitzgerald et al., 2013). Nevertheless, DSM-5 suggests that ‘clinician judgment’ should determine if specific symptoms are ‘counted’ toward the criteria of MDD (APA, 2013). As such, the clinician’s judgment is the current gold standard for determining if a patient meets specific somatic symptom criteria as part of a current MDE (Fava et al., 2015).
Given the complexity of deciding which somatic symptoms ‘count’ toward the diagnosis of an MDE, some investigators and clinicians have advocated for either an ‘exclusive’ approach (e.g. removing somatic symptoms completely in the medically ill) or a ‘substitutive approach’ where somatic symptoms are replaced by other non-somatic symptoms of depression that are not directly affected by medical illness (Huey et al., 2018). For example, in a study of outpatients at a large cancer center (n = 611) participants completed a modified PHQ-9 with additional items covering the Endicott and Cavanaugh substitutive criteria. Depression prevalence varied significantly by diagnostic approach, with the inclusive approach identifying the largest proportion as depressed (9.3%), followed by the Endicott-substitutive (6.2%), exclusive (4.6%) and Cavanaugh-substitutive approach (1.8%) (Saracino et al., 2018). In screening, the inclusive approach (e.g. including somatic symptoms) is generally recommended, but there continues to be debate and minimal consensus on which approach is best when making the diagnosis of MDD in the medically ill (Mitchell et al., 2012). Further research is needed to establish the construct and face validity of different criteria for MDD in the subgroup of the medically ill (Huey et al., 2018). The HADS, designed for depression in medically ill people, eliminates vegetative symptoms for this reason although its validity has been questioned (Stern, 2014).
Delirium is an important differential diagnosis as well, especially in inpatient and nursing home settings where rates of delirium are >25% (Ahmed et al., 2014). A large proportion of referrals to psychiatry services for depression in inpatient medical settings are identified as delirium upon further assessment (Grover et al., 2017). The presentation of delirium includes acute onset of fluctuating cognitive function, inattention and sleep-wake disturbances secondary to an underlying organic cause (APA, 2013). These signs and symptoms may be mistaken as ‘depressive symptoms’ of poor concentration, insomnia, fatigue, anhedonia and social withdrawal. However, the time course, fluctuating nature, medical status and lack of negative cognitions or subjective low mood may help differentiate delirium from depressive symptoms. Delirium is more common in people with dementia and early mild cognitive impairment, which is also an important differential diagnosis. Ruling out delirium is of particular importance given the high rate of mortality associated with this neuropsychiatric condition (Witlox et al., 2010).
Collaborative care models for the treatment of depression in the medically ill
Extensive evidence indicates that collaborative care (CC) interventions are effective for the management of depression in the context of co-occurring medical conditions (Bruce et al., 2004; Huffman et al., 2014; Katon et al., 2010; Patel et al., 2010; Unützer et al., 2002). Furthermore, CC interventions for depression are both cost-effective and improve outcomes for both depression and comorbid medical illness (Reiss-Brennan et al., 2016; Van Eck van der Sluijs et al., 2018). Typically, CC interventions are team-based interventions that are based on the principles of measurement-based care. For example, in a CC intervention for depression in primary care, patients are routinely assessed with a validated screening tool (e.g. the PHQ-9) in regular clinical visits. Second, these models use a non-physician case manager, usually a nurse, who is located in the medical setting who can further assess patients with a positive-screen (e.g. two-stage screening). If a patient meets criteria for entry into a CC program, the care manager can continue assessment and monitoring of symptoms over time (e.g. with repeated PHQ-9s); coordinate pharmacotherapy recommendations to general practitioners (GPs); and provide support, education and evidence-based psychotherapy to patients as appropriate. Finally, CC programs include specialist-provided stepped care recommendations, often from a psychiatrist, who reviews the case with the case manager and provides specific recommendations prompted by results of longitudinal assessments. A stepped care approach tailors the intensity of treatment to the patient’s initial depression severity and subsequent treatment response (Figure 2) (Bockting et al., 2018; Guidi et al., 2016).
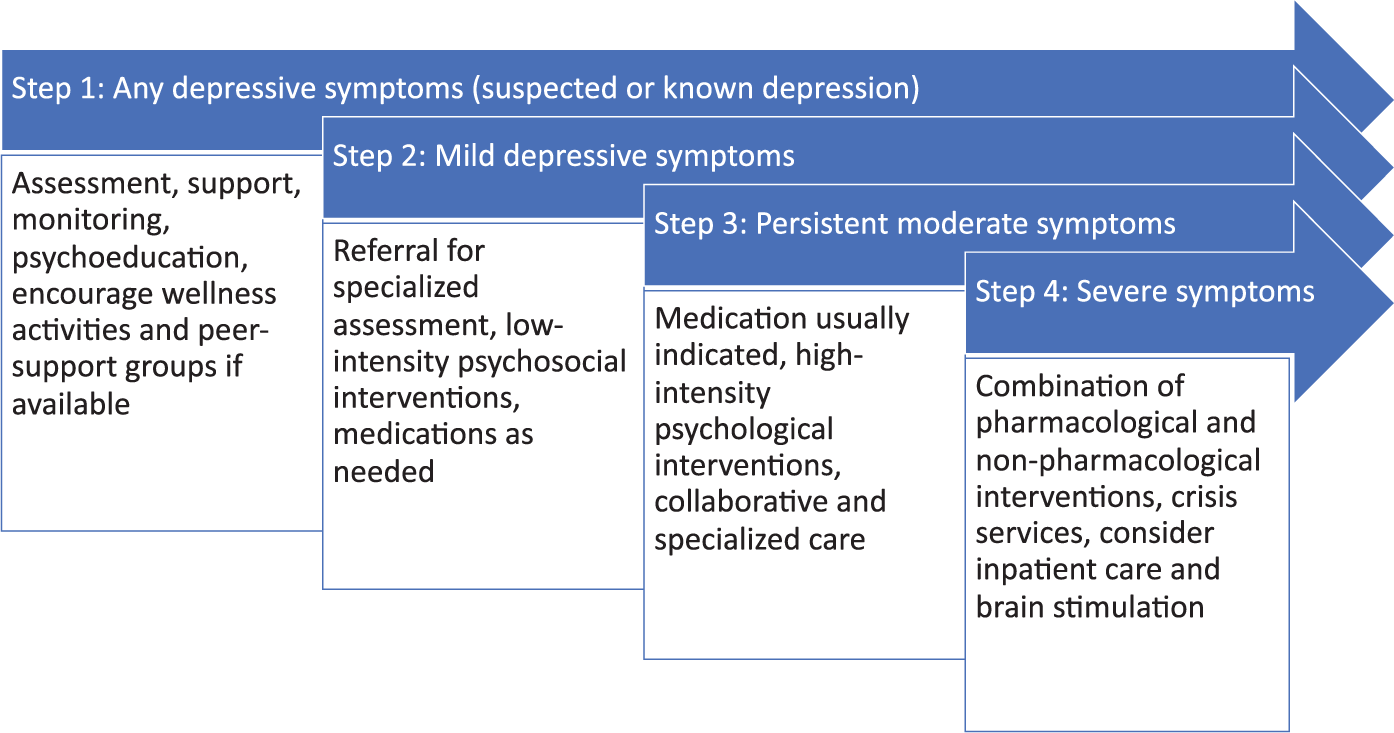
Stepped Care model of depression in the medically ill based on recommendations from National Institute for Health and Care Excellence (NICE) guidelines for treatment of depression with chronic physical illness.
With a stepped care approach, wellness strategies (e.g. exercise, peer support) are recommended for any subthreshold depressive symptoms (step 1). With mild to moderate symptoms, or persistent subthreshold symptoms that did not improve with step 1 interventions, low-intensity psychosocial interventions should be considered (e.g. individual or group psychotherapy), using different approaches according to the stage of the illness (Guidi et al., 2017). Depending on the clinical formulation, patient preference and severity of symptoms, antidepressant medications may also be considered at step 2 (Macneil et al., 2012). However, psychosocial interventions are usually preferable at this stage, given the high burden of polypharmacy with potential for harmful drug–drug and drug–disorder interactions (Mausbach and Irwin, 2017; Scott et al., 2015) and the potential iatrogenic adverse effects of psychopharmacological treatments (Carvalho et al., 2016; Fava et al., 2013, 2014; Offidani et al., 2014). Individual conceptualization of illness and personal treatment preference also predicts treatment response and adherence.
With moderate to severe symptoms or mild to moderate symptoms unresponsive to steps 1 and 2 interventions, a trial of medication or high-intensity psychotherapy should be considered. Medications and high-intensity psychotherapy may be used sequentially, although, in special circumstances, a combination of both may be required to achieve remission of symptoms (Guidi et al., 2016). Using a collaborative care approach to optimize both psychiatric and medical outcomes may also be beneficial and an efficient use of limited healthcare resources (Gilbody et al., 2006; Li et al., 2016). Finally, for severe and treatment-resistant depression (TRD), more intensive treatments, such as inpatient admission and brain stimulation (e.g. repetitive transcranial magnetic stimulation [rTMS] or electroconvulsive therapy [ECT]) should be considered (NICE, n.d.; Perera et al., 2016).
Psychotherapy in the medically ill
Given the high prevalence of polypharmacy and propensity for drug–drug and drug–disease interactions, as well as the phenomenon of iatrogenic comorbidity, psychological treatments are often preferable, when feasible and available, in this population (Scott et al., 2015). In addition, in some medical illnesses, such as heart failure, psychotherapy might be more effective than antidepressant medications (Das et al., 2019). In the general population and in the medically ill, psychotherapy is the preferred treatment for patients with subthreshold depressive symptoms and mild MDD, whereas moderate to severe MDD often requires pharmacological interventions (Kennedy et al., 2016). Clinical formulation should also guide treatment; factors such as family history, childhood trauma, comorbid personality and other disorders and prior history of response are factors that predict differential treatment utility. Notably, however, in the medically ill, there is a very limited number of studies directly comparing pharmacological and non-pharmacological treatments in subgroups of interest. In the general population, level 1 evidence is available to support the efficacy of the following psychological interventions in the acute treatment of an MDE: cognitive behavioral therapy (CBT), interpersonal therapy (IPT) and behavioral activation (BA) (Parikh et al., 2016). Tailored psychotherapies have also been shown to relieve depressive symptoms in some medical populations (Rodin et al., 2018). For maintenance treatment (e.g. relapse prevention) in the general population, level 1 evidence is available for CBT and mindfulness-based cognitive therapy (MBCT). In addition, staging may allow to apply a psychotherapeutic intervention to specific phases of development of depression: well-being therapy and MBCT appear to be uniquely suited for addressing the residual symptoms of depression, whereas IPT has been mainly tested in the acute phase. CBT appears to be suitable for all phases, but with persistent depressive disorder (PDD) its modifications (e.g. cognitive behavioral analysis system of psychotherapy) may be required.
These modalities, especially CBT, have also been shown to be efficacious in several chronic disease populations, but with a lower level of evidence (levels 2–3), given the limited number of studies and smaller sample sizes (Parikh et al., 2016). Structured and semi-structured psychotherapeutic interventions (e.g. CBT, IPT) have demonstrated antidepressant effects with small to moderate effect sizes in patients with cancer (Chong Guan et al., 2016; Okuyama et al., 2017), diabetes (Baumeister et al., 2014), coronary heart disease (Richards et al., 2018), heart failure (Das et al., 2019), HIV (Eshun-Wilson et al., 2018; Van Luenen et al., 2018), epilepsy (Elger et al., 2017) and MS (Fiest et al., 2016; Sesel et al., 2018). CBT has not been shown to be efficacious in advanced disease, although meaning-based and supportive-expressive therapies have been shown to relieve depressive symptoms in this population (Li et al., 2019; Rodin et al., 2018). Limited and mixed evidence also supports the use of psychotherapeutic interventions for post-stroke depression and in ESRD, but further study is still needed to more definitely determine the potential benefits in these populations (Cohen et al., 2007; Hadidi et al., 2017; King-Wing Ma and Kam-Tao Li, 2016).
Growing evidence has also suggested a role for focused psychotherapy, such as problem-solving therapy (PST), for prevention of depression in the medically ill. In a randomized controlled trial (RCT) of escitalopram and PST for prevention of post-stroke depression, the use of escitalopram or PST resulted in a significantly lower incidence of depression over 12 months of treatment compared with placebo (Robinson et al., 2008). Similarly, in a large RCT comparing PST (n = 105) to treatment as usual (n = 100), PST prevented depressive disorders and loss of valued activities in patients with age-related macular degeneration (Rovner et al., 2007). More recently, in a large RCT with older adults with subsyndromal depressive symptoms (n = 181) at rural and urban primary care clinics in India, Dias et al. (2019) demonstrated that lay counselors providing PST, brief behavioral treatment for insomnia and education in self-care of common medical disorders were effective for preventing major depressive episodes in this population. Taken together, these studies suggest a potential role for psychotherapeutic interventions for prevention of depression in these high-risk, medically ill groups.
There is minimal evidence to indicate which sub-populations (e.g. based on demographics, illness stage) are more likely to benefit from psychological interventions. Across all medical illnesses, illness-related physical symptoms such as fatigue and pain, and cognitive dysfunction may limit the effectiveness of psychotherapeutic interventions by directly impairing the ability of the patient to effectively engage in the therapy process and complete necessary tasks (e.g. homework for CBT) (Kok and Reynolds, 2017). The potential to treat these symptoms, which are strong risk factors for depression, should be carefully considered. Psychotherapeutic approaches also need to be adjusted based on the ability of the patient to tolerate and adequately engage in psychotherapy. This assessment may be an ongoing process over the course of treatment, as the physical status, symptom burden and cognitive function may change with progression of disease. Notably, however, cognitive impairment is not a contraindication to psychotherapy, as some with such impairment may still benefit from modified psychological interventions (Gitlin and Aravena, 2017; Orgeta et al., 2015).
Given the association of depression with poorer medical outcomes, numerous studies have evaluated the effects of psychotherapy on medical outcomes. Psychological interventions have been shown to improve depressive symptoms in diabetes, but did not have an effect on glycemic control (Baumeister et al., 2014). For coronary heart disease, meta-analytic level data suggest that psychological interventions may reduce cardiovascular mortality, while having no effect on all-cause mortality, future myocardial infarction (MI) or revascularization (Barth et al., 2004; Richards et al., 2018). For cancer, a recent large-scale RCT found that a collaborative care intervention referred to as Depression Care for People with Cancer (DCPC) that had both psychosocial and pharmacological components significantly improved depression and quality of life in patients with cancer compared to treatment as usual with no significant effects on survival (Mulick et al., 2018). A tailored supportive-expressive therapy, referred to as Managing Cancer and Living Meaningfully (CALM), has been shown in a large RCT to reduce depressive symptoms and death anxiety in patients with advanced cancer (Rodin et al., 2018). There is only weak evidence with low effect sizes for psychological interventions for primary prevention of depression (Forsman et al., 2011).
Taken together, moderate evidence supports the use of psychological interventions in the medically ill; however, effect sizes are generally lower in the medically ill than in the general population, which may also be partly driven by decreased response in older adults (Dodd et al., 2014). While significant effects are observed in psychiatric outcomes, the benefits of psychological interventions for medical outcomes remain unclear with mixed results to date. Several additional psychological interventions are currently under investigation for treatment of depression and psychological distress in specific medical sub-populations. For example, in cancer and palliative care, several new modalities have shown promising results, such as meaning-centered psychotherapy (Breitbart et al., 2012; Dose and Rhudy, 2018; Martínez et al., 2017) and CALM therapy (Rodin et al., 2018). Specifically in migraine patients, well-being therapy has been proposed (Mansueto et al., 2018). Future research may build further on determining subgroups that may benefit from specific interventions and tailoring psychological interventions to the unique needs of the medically ill.
Pharmacological interventions in the medically ill
Similar to psychotherapeutic interventions, antidepressants with efficacy in the general population have demonstrated efficacy in the medically ill, but with fewer supporting studies, smaller samples sizes and lower antidepressant effect sizes (Kennedy et al., 2016; Ostuzzi et al., 2018). Meta-analytic level data has demonstrated small-to-moderate antidepressant effect sizes of antidepressants for MDD in cancer (Ostuzzi et al., 2018), diabetes (Baumeister et al., 2014; Darwish et al., 2018), coronary heart disease (Ostuzzi et al., 2019), post-stroke (Robinson and Jorge, 2016; Sun et al., 2017), HIV (Eshun-Wilson et al., 2018), epilepsy (Elger et al., 2017; Maguire et al., 2014) and MS (Fiest et al., 2016). The benefits of antidepressants in heart failure and major neurocognitive disorder are unclear with mixed results (Das et al., 2019; Dudas et al., 2018). Potential benefits of antidepressants for the primary prevention of depression have been demonstrated in post-stroke and post-MI populations, but due to study limitations and risk of polypharmacy, routine antidepressant prophylaxis is not recommended (Ha and Wong, 2011; Ostuzzi et al., 2019; Towfighi et al., 2017). Use of antidepressants has mostly failed to demonstrate improvements in all-cause mortality in the medically ill (Almeida et al., 2010; Barth et al., 2004).
Available evidence is limited regarding the comparative efficacy of specific antidepressants in specific medical populations. In the general population, comparative antidepressant effects of different antidepressant classes and agents have been largely equivalent (Cipriani et al., 2018). Clinically, such group equivalence is not easily translatable to the individual, as many people will respond to one antidepressant class or agent and not another. As such, the pragmatic selection of pharmacological treatments is based on prior and family history of response, variability in potential behavioral toxicity, symptom-domain specific effects and side effects. As summarized in Table 3, the propensity for specific behavioral toxicity and side effects of common antidepressants and augmenting agents vary widely, but they all reduce depressive symptom severity overall (Carvalho et al., 2016; Cipriani et al., 2018; Fava et al., 2016). Special attention to these effects in the medically ill is of great importance, as off-target effects may improve, or worsen, some physical symptoms of medical illness. For example, sedating antidepressants may be of benefit for patients with prominent insomnia, which may be secondary to both depression and many chronic medical illnesses. Conversely, added caution about side effects in the medically ill is warranted to prevent worsening of already problematic physical symptoms as well as the emergence of burdensome iatrogenic comorbidities (Fava et al., 2016). For example, in a patient with cancer-related fatigue (CRF) and hypersomnia, use of antidepressants with sedating effects should be avoided. Rather, antidepressants and augmenting agents with wake-promoting effects should be preferentially prescribed in this circumstance. Within a single patient, sedation may be desirable or deleterious at different phases of illness. Consideration of this ‘behavioral toxicity’ is important and may change depending on phase of medical illness and depression (Fava et al., 2016). Similarly, in cardiac patients with arrhythmias and a prolonged QT interval, citalopram, lithium, tricyclic antidepressants (TCAs) and antipsychotics should be avoided if possible, given the arrhythmogenic potential of these agents (Vandael et al., 2017).
Side effect and biobehavioral toxicity considerations of antidepressants and common augmenting agents.
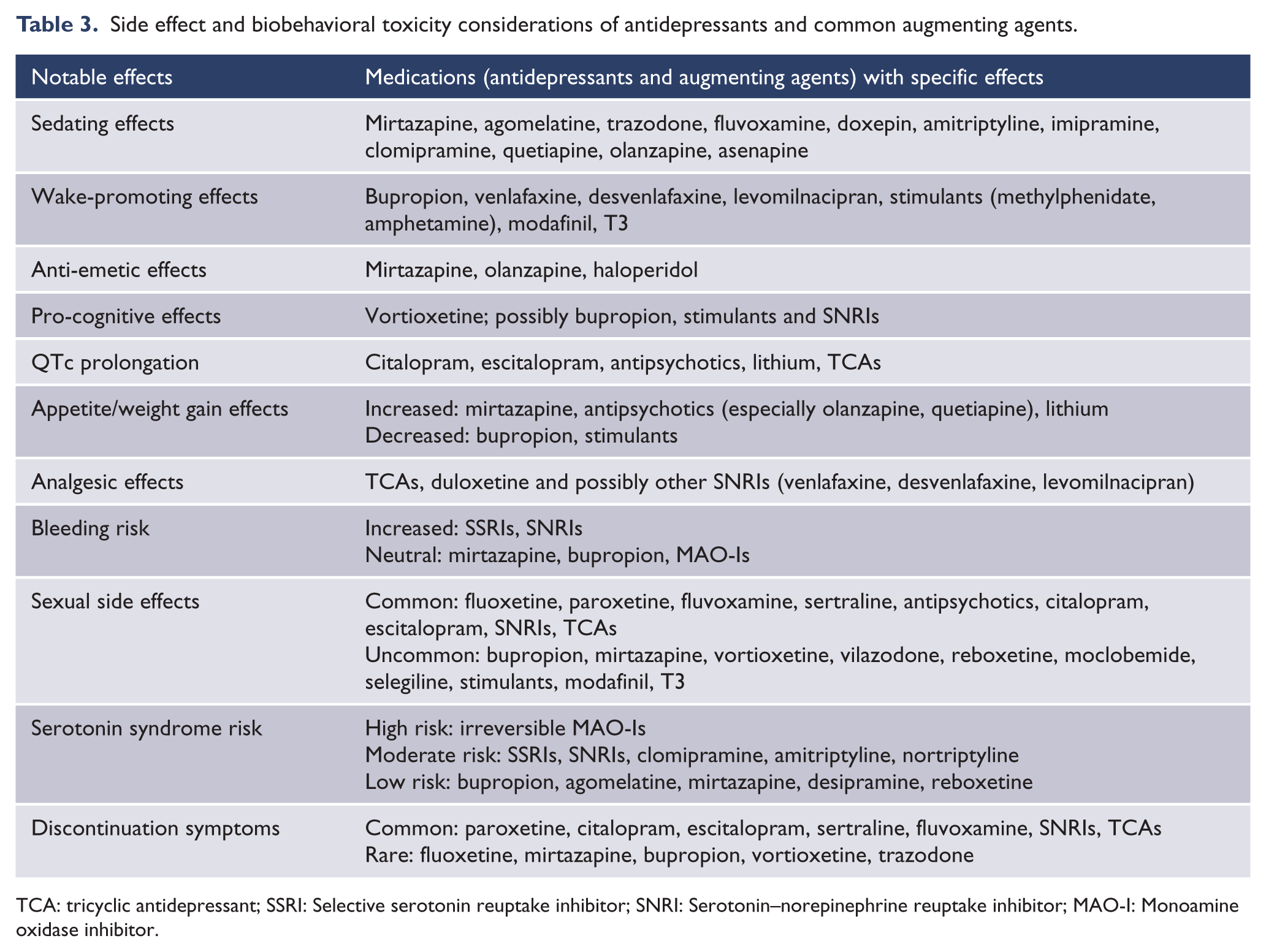
TCA: tricyclic antidepressant; SSRI: Selective serotonin reuptake inhibitor; SNRI: Serotonin–norepinephrine reuptake inhibitor; MAO-I: Monoamine oxidase inhibitor.
In addition to considering side effects, potential drug–drug interactions and drug–disease interactions must be critically evaluated. Given the rapidly growing number of medications used in chronic disorders (e.g. chemotherapeutics, biologics, anti-retrovirals), most published interaction guides are out-dated. The use of up-to-date drug interaction software packages is recommended when adding a psychotropic medication or making any dose adjustments in medically complex patients (e.g. Lexicomp). For medical disorders affecting liver and/or renal function, dose adjustments are often required, as summarized in Table 4, with some agents being contraindicated in hepatic and renal impairment. Given the propensity for both known and unknown interactions in this population, the general approach of ‘start low and go slow’ is advisable to minimize the risk of toxic effects. Most antidepressants have not been extensively tested in hepatic or renal impairment so product monographs often lack explicit, evidence-based direction for dose adjustment (Hedayati et al., 2012; Mauri et al., 2014). Therefore, added caution is needed, even in the absence of explicit dose adjustment recommendations from the product monograph.
Dose adjustment in renal and hepatic impairment based on product monographs.
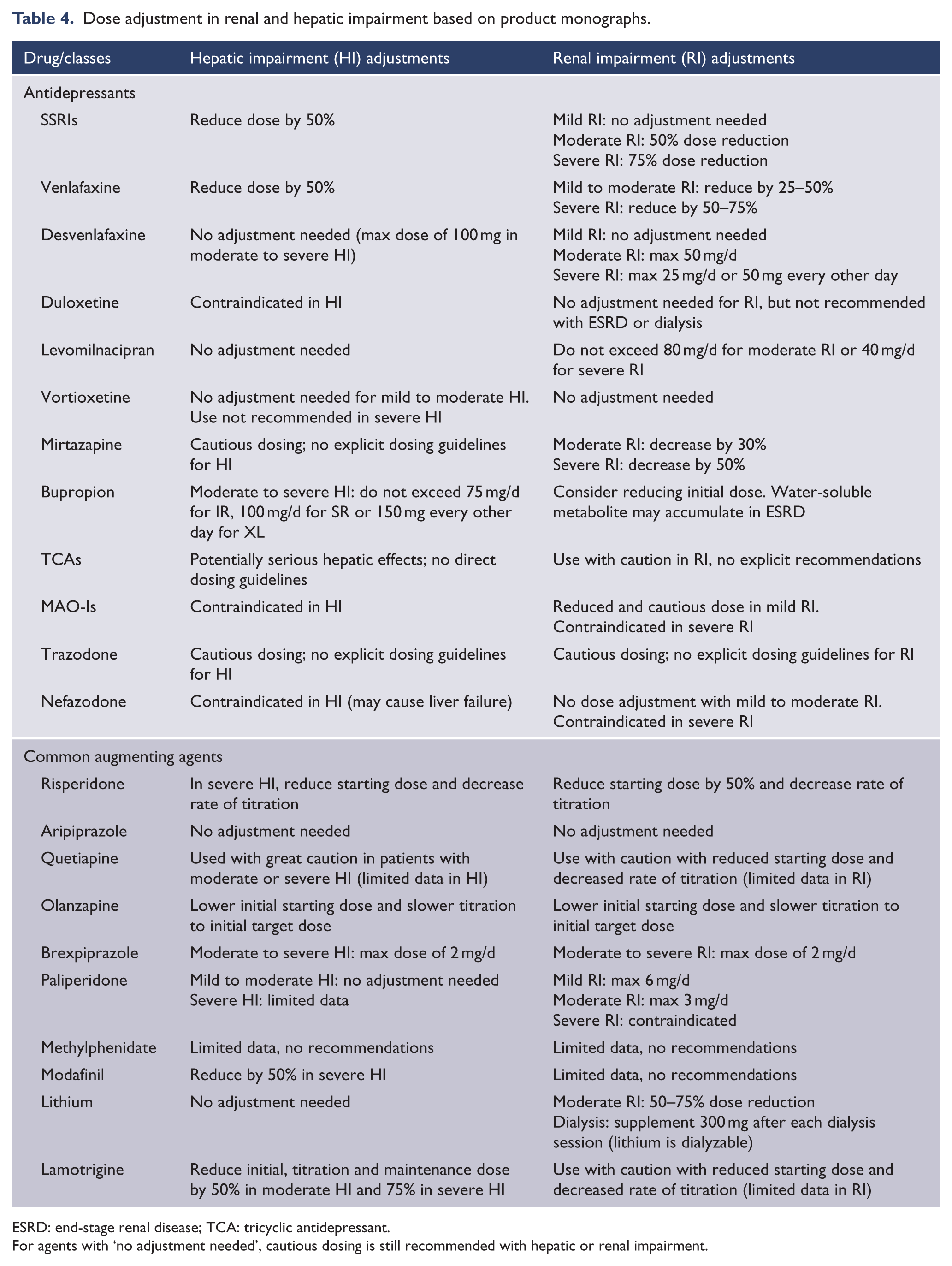
ESRD: end-stage renal disease; TCA: tricyclic antidepressant.
For agents with ‘no adjustment needed’, cautious dosing is still recommended with hepatic or renal impairment.
Several novel interventions primarily acting outside of the monoaminergic system are under investigation (Carvalho et al., 2014; Rosenblat et al., 2015). For example, the use of immune-regulatory, anti-inflammatory and anti-oxidant agents, with neuroprotective and mitochondria-protective properties, including curcumin, zinc, omega-3 polyunsaturated acids and N-acetylcysteine (NAC) have proven beneficial in the treatment of depression (Grosso et al., 2014, 2016; Li et al., 2017; Maes et al., 2012; Swardfager et al., 2013; Zheng et al., 2018). Results of RCTs are regarded as proof of concept supporting the neuro-immune and neuro-oxidative stress theory of depression (Leonard and Maes, 2012). These agents have also been investigated in the general population (Kohler et al., 2014). Notably, the potential role of anti-inflammatories in primary prevention of depression is also currently underway (Berk et al., 2013a, 2016).
In palliative care, growing focus has been placed on ‘rapid acting’ antidepressants, when life expectancy is limited, requiring a more immediate effect, to allow for improved quality of life in the final weeks to months of living (Irwin and Iglewicz, 2010; Ng et al., 2014). Stimulants, such as methylphenidate, initially were reported to demonstrate rapid reduction in depressive symptom, but further analysis revealed that the effects were primarily related to improved energy (Andrew et al., 2018; Hardy, 2009; Lavretsky et al., 2015; McIntyre et al., 2017; Ng et al., 2014). Several new and emerging medications are currently under investigation in the treatment of depression in the medically ill. Recent studies have demonstrated promising preliminary results for rapid antidepressant effects with psilocybin in patients receiving palliative care (Ross, 2018; Ross et al., 2016). Ketamine and esketamine have demonstrated rapid and robust antidepressant effects in the general population and are currently under investigation in patients with terminal cancer as well (Caddy et al., 2015; Irwin et al., 2013; Irwin and Iglewicz, 2010). Ketamine may have the added benefit of improvements in pain control, which is commonly problematic in palliative care (Bell et al., 2012; Fallon et al., 2018). However, the known risk of ketamine dependence will be a challenge to manage in clinical rollout, especially in non-terminal phases of the illness (Berk et al., 2018).
Brain stimulation and convulsive therapies in the medically ill
ECT is one of the oldest and most effective treatments for depression in the general population (Milev et al., 2016). The technique is based on the induction of controlled seizures in patients under general anesthesia via depolarization of a larger number of neurons using a stimulus of alternating polarity pulses with amplitude of 800–900 mA via two electrodes placed over one’s head (Milev et al., 2016). ECT is not widely disseminated as a first-line treatment due to issues such as costs, stigma, tolerability (e.g. memory deficits and untoward cognitive effects) and requirement for general anesthesia. Nevertheless, for acute and severe suicidality, treatment-resistance or severe depression, ECT should be considered (Milev et al., 2016). In the medically ill and the elderly, ECT is relatively safe and well-tolerated, with the added advantage of avoiding additional augmenting antidepressant medications when polypharmacy may already be high (Rasmussen et al., 2002). ECT has been tested in numerous populations including MS, PD, various endocrinological disorders and renal failure, demonstrating similar efficacy compared to the general population. It may be particularly beneficial for PD with comorbid MDD, as ECT is effective to treat depressive symptoms as well as the core symptoms and functional outcomes of PD (Borisovskaya et al., 2016; Williams et al., 2017). While there are no absolute contraindications for ECT, caution should be exercised with a history of stroke or MI in the past 6 months or with pre-morbid significant cognitive impairment. Cardiovascular risk factors might present contraindications for ECT, due to high blood pressure peaks immediately following the seizure. Magnetic seizure therapy (MST) has most recently been developed with encouraging preliminary results, potentially with less cognitive effects than ECT. However, MST still requires general anesthesia as a seizure is induced and MST has not been adequately studied in the medically ill (Cretaz et al., 2015).
In contrast, non-invasive brain stimulation (NIBS) therapies are relatively recent techniques that are generally less effective than ECT (Brunoni et al., 2019; Mutz et al., 2019). NIBS techniques include transcranial magnetic and direct current stimulation (TMS and tDCS, respectively), which use magnetic or electric fields to promote changes in cortical excitability (Brunoni et al., 2019). TMS is based on the principle of electromagnetic induction via an electric current passing through a coil, whereas for tDCS an electric current of low intensity (usually ⩽3 mA) is injected in the brain via electrodes placed over the scalp (Brunoni et al., 2019). These approaches do not require sedation or anesthesia and evidence indicates they have excellent safety and tolerability profiles (Bikson et al., 2016; Rossi et al., 2009). For tDCS, additional advantages include its affordability, ease of use, and portability and lack of serious adverse events, including the small risk of seizures that has been reported with TMS (Aparício et al., 2016). For depressed patients with medical conditions who are at risk for pharmacological interactions and toxicity, NIBS might present as interesting alternative modalities (Valiengo et al., 2013).
Several rTMS protocols are effective for the treatment of unipolar depression (Brunoni et al., 2017a; Mutz et al., 2018). In post-stroke depression, rTMS has replicated RCT level evidence of moderate quality demonstrating efficacy with comparable antidepressant effect sizes to medications (Deng et al., 2017; Shen et al., 2017). Conversely, in depression with PD, results have been mixed with rTMS (Zhou et al., 2018). Trials that investigated the efficacy of tDCS with or combined with antidepressant drugs have shown that tDCS is inferior to full-dose escitalopram (Brunoni et al., 2017b), although its effects are enhanced when combined with sertraline (Brunoni et al., 2013). tDCS is also effective in patients with post-stroke depression (Valiengo et al., 2017).
Limitations
The current review had several limitations, most notably, the approach to reviewing the current topic and the limits of the available evidence. The current review was not a systematic review or meta-analysis as the authors believed that an exhaustive review of the literature on this topic would be disadvantageous and elected to synthesize and summarize the evidence instead of reviewing all articles published on this large topic. This approach may lead to presenting the evidence in a biased fashion by potentially missing evidence that would have been otherwise reported in a systematic review. Moreover, as emphasized throughout the article, the evidence base on this topic is still limited, preventing conclusive answers on the topics discussed.
Concluding remarks and outlook
Replicated evidence has demonstrated a bidirectional interaction between depression and medical illness with high rates of comorbidity. MDD comorbid with medical disease is associated with poorer outcomes of both conditions. Epidemiological studies have demonstrated increased rates of subthreshold depressive symptoms and threshold MDD in almost all disorders, compared to healthy controls. However, prevalence estimates vary greatly, with heterogeneity in study designs, screening/diagnostic methods and populations of interest making it difficult to determine the absolute risk increase of MDD in the medically ill compared to medically well. Common disorders with particularly high risk of MDD include cancer, CVD, HIV, COPD and neurological disorders (MS, epilepsy, stroke) with estimated lifetime MDD prevalence rates of 20–40%. Risk of suicide is also increased in medically ill populations requiring careful assessment.
Preventive efforts for the management of MDD in the medically ill may be a relevant research direction. It was proposed that antidepressants may prevent the emergence of depression in patients with stroke or hepatitis C virus treated with interferon (Ehret and Sobieraj, 2014); however, evidence for such preventive effects in the medically ill remains limited. People with a medical illness and subthreshold depressive symptoms may also be candidates for targeted preventive strategies (Lee et al., 2019).
Accumulating evidence indicates that patients with depression and comorbid medical illness benefit from CC interventions with stepped care that may include both pharmacological and psychosocial treatments that have been proven effective in these populations. CC interventions may be challenging to deliver to remote and rural areas, although an increasing number of studies have attempted to assess the usefulness of technology-facilitated (e.g. digital) CC interventions for the management of depression in the medically ill with encouraging initial outcomes (Rojas et al., 2018; Wu et al., 2018). This research direction clearly deserves further investigation to improve access to the optimal care in rural communities.
Time-limited structured and semi-structured psychological interventions have also demonstrated antidepressant efficacy in these populations, with replicated studies using CBT, Mindfulness-based stress reduction therapy (MBSRT), IPT and supportive-expressive therapy. Antidepressant selection and dosing may need to be modified in the context of hepatic or renal impairment, or with drug-drug interactions, as polypharmacy is common in these populations. While evidence is limited, some studies have suggested symptom-targeted treatments to be of benefit. Brain stimulation is safe in most medical illnesses and is particularly beneficial in the elderly and some neurological disorders, but requires further study in the medically ill.
Key areas for future research include further evaluations of algorithms for screening and treatment of depression that are more customized and validated for specific chronic medical disorders (Table 5). New psychotherapy modalities being developed specifically for the medically ill hold hope for improved outcomes in both depressive symptoms and other psychological distress associated with medical illness. Novel modes of delivery (e.g. smartphone-based interventions; Firth et al., 2017) may improve the availability of evidence-based psychological treatments in remote and underserved areas where trained psychotherapists are not readily available, although few studies to date have been conducted in medically ill populations. Furthermore, novel pharmacological interventions are being investigated that target neurotransmission systems other than the conventionally targeted monoamine system. Understanding subgroups that are more likely to respond to specific interventions is also of great importance to allow for more personalized treatments rather than trial-and-error algorithms within the emerging framework of precision psychiatry (Fernandes et al., 2017; Insel, 2014).
Future direction of study for depression in the medically ill.

MDD: major depressive disorder.
Footnotes
Author Contributions
J.D.R. and A.F.C. developed the outline of the paper. J.D.R. wrote the initial manuscript draft. P.K., F.C., M.L., G.R., A.R.B. and R.S.M. provided significant revisions with a substantial intellectual contribution. J.D.R. and A.F.C. integrated all revisions into the final draft. All authors approved the final submitted manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: J.D.R., P.K., F.C., M.L., G.R. and A.F.C. have no conflicts of interest to declare. R.S.M. declares that he has been on advisory boards and/or received honoraria for educational activities and/or research grants from AstraZeneca, Bristol-Myers Squibb, Janssen-Ortho, Eli Lily, Forest, Lundbeck, Pfizer, Shire, Merck, Sepracor and Otsuk.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
