Abstract
Objective:
Preliminary evidence supports the safety and efficacy of subanesthetic ketamine as an experimental antidepressant, although its effects are often not sustained beyond one week. Studies are lacking that have examined the sustained effects of escalating ketamine doses as augmentation in outpatients with treatment-resistant depression. Therefore, the aims of this study were twofold: (1) to assess the safety and antidepressant efficacy of two-step, repeated-dose ketamine augmentation and (2) to assess the duration of ketamine’s antidepressant efficacy as augmentation to ongoing antidepressant pharmacotherapy for 3 months after the final infusion.
Methods:
Fourteen patients with treatment-resistant depression were eligible to receive augmentation with six open-label intravenous ketamine infusions over 3 weeks. For the first three infusions, ketamine was administered at a dose of 0.5 mg/kg over 45 minutes; the dose was increased to 0.75 mg/kg over 45 minutes for the subsequent three infusions. The primary outcome measure was response (as measured on Hamilton Depression Rating Scale–28 items).
Results:
After the completion of three ketamine infusions, 7.1% (1/14) responded; after all six ketamine infusions, 41.7% (5/12) completers responded and 16.7% (2/12) remitted. Intent-to-treat response and remission rates at the end of the final infusion were 35.7% (5/14) and 14.3% (2/14), respectively. However, all but one responder relapsed within 2 weeks after the final infusion.
Conclusion:
Repeated, escalating doses of intravenous ketamine augmentation were preliminarily found to be feasible, efficacious and well tolerated. Interaction with concomitant medications and elevated level of treatment resistance are possible factors for non-response.
Introduction
Major depressive disorder (MDD) is a leading cause of disability worldwide (Kessler et al., 2003). Unfortunately, treatment-resistant depression (TRD) accounts for 12–20% of all depressed patients, costing society US$29–US$48 billion annually (Mrazek et al., 2014; Rush et al., 2009). Perhaps most strikingly, patients with TRD are at a much greater risk for suicide attempts than those with treatment-responsive depression (Crown et al., 2002). Because of the high morbidity of patients with TRD, the discovery of novel, rapidly acting antidepressants is essential for this patient population.
Toward this end, ketamine (a noncompetitive N-methyl-
Ketamine’s antidepressant properties are promising; however, prolonging its effect beyond a week is an important topic. One recent open-label ketamine study showed that repeated infusions (six infusions over 2 weeks) of subanesthetic ketamine (0.5 mg/kg over 40 minutes) in known ketamine responders with TRD (n = 10) were safe and efficacious, with a mean time-to-relapse of 19 days (aan het Rot et al., 2010)—longer than the ⩽7 days reported in previous studies (Berman et al., 2000; Zarate et al., 2006). Of note, the antidepressant response rate was 9/10 (90%) at day 1 after the first infusion; all responders continued to receive ketamine. Throughout the repeated infusions, the reduction in depression scores from baseline did not correlate significantly with patient expectancies (averaged across their infusion visits; r = 0.37, p = 0.4, controlling for baseline severity). There was also no significant difference between patient expectancies before the first infusion and before the last infusion (p = 0.6). This sample was extended to 24 patients, with a median time-to-relapse of 18 days in responders (Murrough et al., 2013b). Here, responders to the first administration of ketamine continued to improve slightly—but significantly—after the initial 2-hour improvement (average daily decrease in Montgomery–Asberg Depression Rating Scale (MADRS) score of 0.35 ± 0.10, p = 0.004), whereas non-responders tended to worsen over time (average daily increase in MADRS score of 0.78 ± 0.40, p = 0.096). The separation of responders from non-responders was identified at 4 hours post-infusion and reached a peak at 24 hours (total MADRS scores 8.35 ± 4.2 vs 18.8 ± 5.5, p = 0.002). By 4 hours, 94% of study responders had responded.
In addition, certain clinical characteristics of patients with depression have been shown to predict a significantly longer response time (following a single infusion of ketamine), including anxious depression (Ionescu et al., 2014), with a median time-to-relapse of 19 days (vs 1 day in nonanxious depression). One case report suggests melancholic depression as a possible predictor for ketamine’s antidepressant effects (Galvez et al., 2014). A pharmacologic attempt to prolong the duration of ketamine effects with riluzole, a glutamatergic modulator, has not been shown to significantly extend ketamine’s antidepressant effects compared to placebo when given for 28 days after a single ketamine infusion (Ibrahim et al., 2012). These findings suggest efficacy and safety of subanesthetic ketamine’s rapidly acting antidepressant properties (although larger studies are needed to confirm and expand these conclusions); these studies also highlight the need for identifying agents or strategies to prolong ketamine’s antidepressant effects.
Ketamine’s rapidly acting antidepressant effects have been demonstrated mostly in medication-free patients with MDD (Murrough et al., 2013a; Zarate et al., 2006). Because most studies excluded medicated patients, these data may not be easily translatable to the practice of general outpatient clinical psychiatry (where patients are on various antidepressant regimens). There are several exceptions that deserve discussion. In one small open-labeled study, patients with TRD (n = 10) were treated with up to four infusions over 2 weeks, with a very slow infusion rate (0.5 mg/kg over 100 minutes) while maintained on their antidepressant medications (Rasmussen et al., 2013). In all, 80% (8/10) of the sample met criteria for response, and five of them (50%) met criteria for remission, although only two sustained remission during the 4-week follow-up period. Another study demonstrated the efficacy and safety of repeated-dose ketamine (six infusions over a 12-day period) in outpatients who were maintained on stable antidepressants for at least 2 months (n = 12) at the typically studied ketamine infusion dose of 0.5 mg/kg over 40 minutes (Shiroma et al., 2014). The mean time-to-relapse in this study was 16 days (although 5 of the 11 total responders sustained response at the end of the study). However, the patients in these studies were not followed for more than 4 weeks after the final infusion. Finally, Diamond and colleagues administered open-label ketamine (0.5 mg/kg over 40 minutes) once a week for 3 weeks (n = 15) or twice a week for 3 weeks (n = 13) to patients with unipolar and bipolar depression maintained on antidepressants (Diamond et al., 2014). The response rates were slightly delayed compared to other ketamine studies, with only three patients responding after the first infusion. Patients were followed for up to 6 months when possible, and the median duration of response was 70 days (range: 25–168 days). Also of note, Sos et al. (2013) examined ketamine’s antidepressant effects, as they relate to psychotomimetic side effects, in inpatients maintained on medications. Finally, two National Institute of Mental Health (NIMH) studies were conducted in patients with bipolar depression maintained on lithium or valproic acid (Diazgranados et al., 2010; Zarate et al., 2012). However, the ketamine doses were not escalated in any of these studies that maintained patients on medications.
Building on this, our study aims to address the following critical gaps in the literature: (1) to assess the clinical antidepressant safety and efficacy of two-step repeated intravenous dose ketamine augmentation in outpatients with TRD and (2) to assess the duration of ketamine’s antidepressant efficacy as augmentation to ongoing antidepressants for 3 months after the final infusion. Since preliminary evidence of a single intravenous infusion of ketamine appears effective and safe for treating depression in well-characterized, medication-free patients with TRD (Murrough et al., 2013a; Zarate et al., 2006) and preliminary evidence from repeated doses of ketamine appears effective and safe for treating depression in outpatients maintained on their antidepressants (Diamond et al., 2014; Rasmussen et al., 2013; Shiroma et al., 2014), we hypothesized that escalating doses (from 0.5 mg/kg over 45 minutes to 0.75 mg/kg over 45 minutes) in non-responders may increase the efficacy, while remaining safe, for treating depression in outpatients with TRD maintained on antidepressants. We further hypothesized that some responders would continue to experience response beyond 1 month post-infusion.
Methods
Patient selection
This study was approved by the Partners Human Research Committee (Institutional Review Board [IRB]) and was conducted in accordance with the ethical principles of the Declaration of Helsinki. Patients were referred to the study by their treating psychiatrists. Inclusion criteria were (1) age 18–65 years; (2) primary diagnosis of MDD, based on the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders–Fourth Edition (DSM-IV) Diagnoses (SCID) (First et al., 1997) as administered by a trained psychiatrist or psychologist, in a current major depressive episode, regardless of depression specifier (i.e. atypical, melancholic); (3) Hamilton Depression Rating Scale–28 items (HAM-D28) score ⩾20 at screening; (4) history of three or more failed antidepressant treatment trials of adequate dose and duration during the current episode (including the current regimen), as measured by the Massachusetts General Hospital (MGH) Antidepressant Treatment History Questionnaire (Chandler et al., 2010); (5) suicidal ideation (SI) for more than 3 months, as measured by the Columbia-Suicide Severity Rating Scale (C-SSRS), without the requirement for immediate hospitalization; and (6) a score on the HAM-D28 suicide item ⩾2 (current SI, thoughts of own death). Exclusion criteria were as follows: (1) pregnancy, (2) unstable medical illness, (3) bipolar disorder, (4) past multiple adverse drug reactions, (5) psychotic illness, (6) substance use disorder within 1 year, (7) positive urine toxicology, (8) past history of ketamine abuse and (9) SI requiring immediate hospitalization or immediate risk. In addition, although patients were maintained on their stable outpatient medication regimens prior to the start of the study and during infusions, certain medications were exclusionary: St. John’s wort, theophylline, tramadol and any narcotic or barbiturate use within the previous 6 months.
All patients were physically healthy as determined by physical examination, blood laboratory testing, electrocardiogram and medical history obtained by a board-certified psychiatrist. Ethical concerns prevented the tapering of current antidepressant regimens, as participants were outpatients with a certain degree of SI; the risks versus benefits of tapering patients off medications for research were deemed too great by the IRB. Therefore, patients were required to maintain their current antidepressant medication regimen for 4 weeks prior to the start of the study and for the duration of the ketamine infusions. Duration of 4 weeks was chosen as sufficient time for their medications to reach steady-state.
Study design and treatment
This open-label ketamine study was conducted between April 2012 and October 2013. Patients were referred to the study by their primary psychiatrists, and no compensation was provided for participation. After a 2-week lead-in period consisting of baseline testing and confirmation of a stable antidepressant medication regimen, patients were admitted to the Clinical Research Center (CRC) at MGH for the infusions. Patients received six intravenous infusions of ketamine over 3 weeks, with two infusions per week. Infusions were generally scheduled to start at the same time of day and on the same 2 days each week in an attempt to keep the experiments as consistent as possible. In addition, although other repeated-dose ketamine trials administered infusions three times a week (Diamond et al., 2014; Shiroma et al., 2014), this was not possible in our sample due to scheduling conflicts. The initial dose of ketamine was 0.5 mg/kg, administered over 45 minutes via a MedFusion 3500 syringe pump. In the initial design, if the patient did not experience an improvement (defined as 30% or higher on the primary outcome measure [HAM-D28]) after Infusion 3, the dose was escalated to 0.75 mg/kg for the final three infusions (30% was chosen as a cut-off point to determine dose escalation as this represents a clinically reasonable cut-off point at which clinicians decide to optimize, switch, augment or combine therapies in the practice of general psychiatry). All but one patient met criteria for a dose increase after the third infusion. However, due to human error, all patients received a dose increase after the third infusion. Although most depression studies administered ketamine over 40 minutes, there are no current data to confirm this as the ‘standard’ time frame for dosing; a dose-finding study is currently underway (ClinicalTrials.gov Identifier: NCT01920555). Therefore, we chose to infuse ketamine for 45 minutes. The infusion pump was programmed by a board-certified anesthesiologist. An anesthesiologist and a psychiatrist were present for the entire infusion.
During the infusion, vital signs (heart rate, blood pressure, respirations, pulse oximetry and electrocardiogram) and the patient’s clinical status were monitored and recorded by a nurse every 5 minutes. Any concerning or intolerable treatment-emergent side effects (e.g. hemodynamic instability, severe dissociation, worsening depression or anxiety) would prompt discontinuation of the infusion. Side effects and vital signs were monitored 30 minutes prior to and during the infusion (every 5 minutes) and for 2 hours after the end of the infusion. At the end of each infusion, patients were monitored for an additional 2 hours by the nursing staff in the CRC. Patients were then escorted to the outpatient psychiatry clinic for further evaluation by a doctoral-level psychiatrist or psychologist and were then discharged home with a responsible adult, as witnessed by research staff. Following the completion of the six infusions, patients were followed every other week for 3 months. During this time, necessary medication adjustments were allowed according to the clinical judgment of the treating psychiatrist and were recorded.
Outcome measures
The primary outcome measure for efficacy was a ⩾50% improvement on the HAM-D28 (Hamilton, 1960), indicating response, while a HAM-D28 score ⩽ 7 was considered remission. The HAM-D28 was used as it captures symptoms of atypical depression (e.g. hypersomnia, hyperphagia) not present in shorter versions. Secondary outcome measures included the Clinical Global Improvement Scale–Severity and Improvement (CGI-S/I) (Guy, 1976); the Quick Inventory of Depressive Symptoms, Self-Report version (QIDS-SR) (Trivedi et al., 2004); the Beck Depression Inventory (BDI) (Beck et al., 1997); the Brief Psychiatric Rating Scale (BPRS) (Overall and Gorham, 1962); and the C-SSRS (Posner et al., 2011). Tolerability was assessed with the Systematic Assessment for Treatment Emergent Events (SAFTEE) (Rabkin et al., 1992), the MGH Cognitive and Physical Functioning Questionnaire (CPFQ) (Fava et al., 2009) and the Quality of Life Enjoyment and Satisfaction Questionnaire (QLES-Q) (Endicott et al., 1993). Scales were administered at the end of each study visit, which occurred approximately 3 hours after the infusion. Specifically at each assessment, patients were asked to rate their symptoms based on the time period since the last visit/infusion. The Clinician Administered Dissociative States Scale (CADSS) (Bremner et al., 1998) was administered immediately prior to and after the ketamine infusion at 60 and 120 minutes.
Inter-rater reliability
The assessment of inter-rater reliability among clinicians at the Depression Clinical and Research Program at MGH for diagnosing MDD and measuring the severity of depression has yielded k > 0.75, indicating satisfactory concordance. This was established through live, independent interviews of patients with the use of HAM-D and SCID assessments.
Statistical analysis
Mean imputation was used to correct for the two subjects who did not complete all visits (Subject 5, Infusions 3–6; Subject 11, Infusions 5–6). We then computed the average HAM-D28 scores for each of the Baseline 0, 1 and 2 and Infusion visits 1–6 (Figure 1). Two baseline assessments were recorded to ensure that patients continued to meet eligibility criteria for the study throughout the pre-treatment phase. The non-parametric Friedman’s test was used to detect changes in scores across study visits (p < 0.0001). A post hoc analysis was completed using the Wilcoxon rank-sum test, with Bonferroni-adjusted p < 0.0014 required for significance. All statistical analyses were completed using MATLAB 8.0 (MathWorks, Natick, MA).
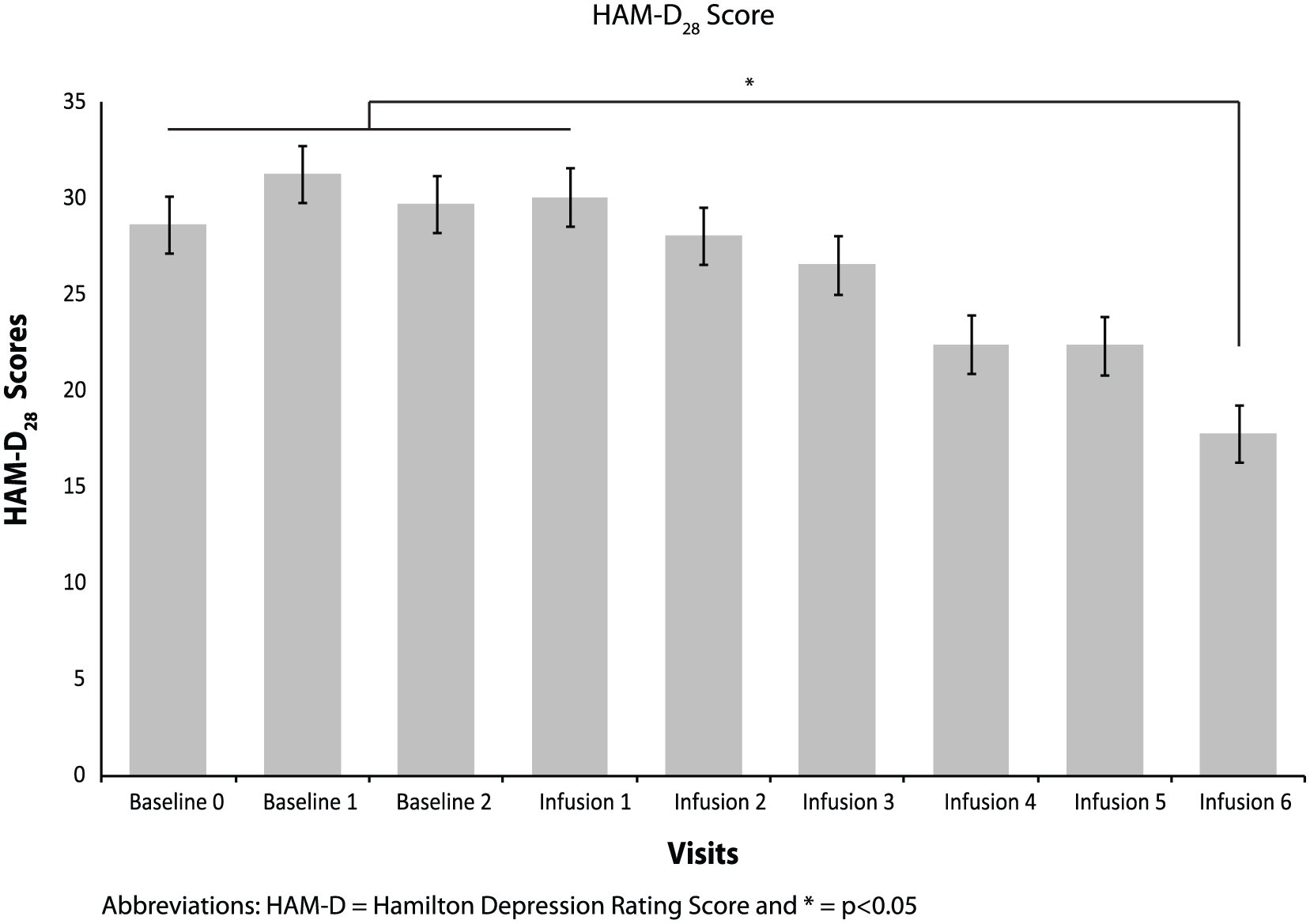
Average HAM-D28 scores for each visit. Infusion 6 HAM-D28 scores were significantly improved compared to scores at Baseline 0, Baseline 1, Baseline 2 and Infusion 1 (all p-values ⩽ 0.001), suggesting a significant improvement during the last infusion of ketamine.
A paired t-test was also used to compare HAM-D28 scores from Baseline 0 to Infusion 3 and from Infusion 3 to Infusion 6 to assess the effect of the increased doses of ketamine. The standardized mean difference (Cohen’s d) from Baseline 0 to Infusion 3 and Infusion 3 to Infusion 6 was computed. Outcomes were analyzed on the basis of the intent-to-treat (ITT) group of all participants.
Results
Study participants
In total, 17 patients were screened, 14 of which met the inclusion criteria for participation and were enrolled in the study. Reasons for exclusion were ongoing alcohol dependence (n = 1), not meeting the suicidality threshold (n = 1) and exposure to an investigational drug in the previous 6 months (n = 1). Of the 14 participants, 12 completed all six infusions; one patient discontinued after Infusion 2 because of intolerable side effects (e.g. unpleasant feelings and mild dissociative symptoms during the infusions), and the other patient discontinued after Infusion 4 due to difficulty in finding a responsible escort home at the end of the study visits. Therefore, 14 participants received at least one infusion of 0.5 mg/kg of ketamine, and 13 participants received at least one infusion of ketamine at 0.75 mg/kg. Demographics, clinical characteristics and concomitant antidepressant medication regimens are presented in Table 1.
Patient characteristics.
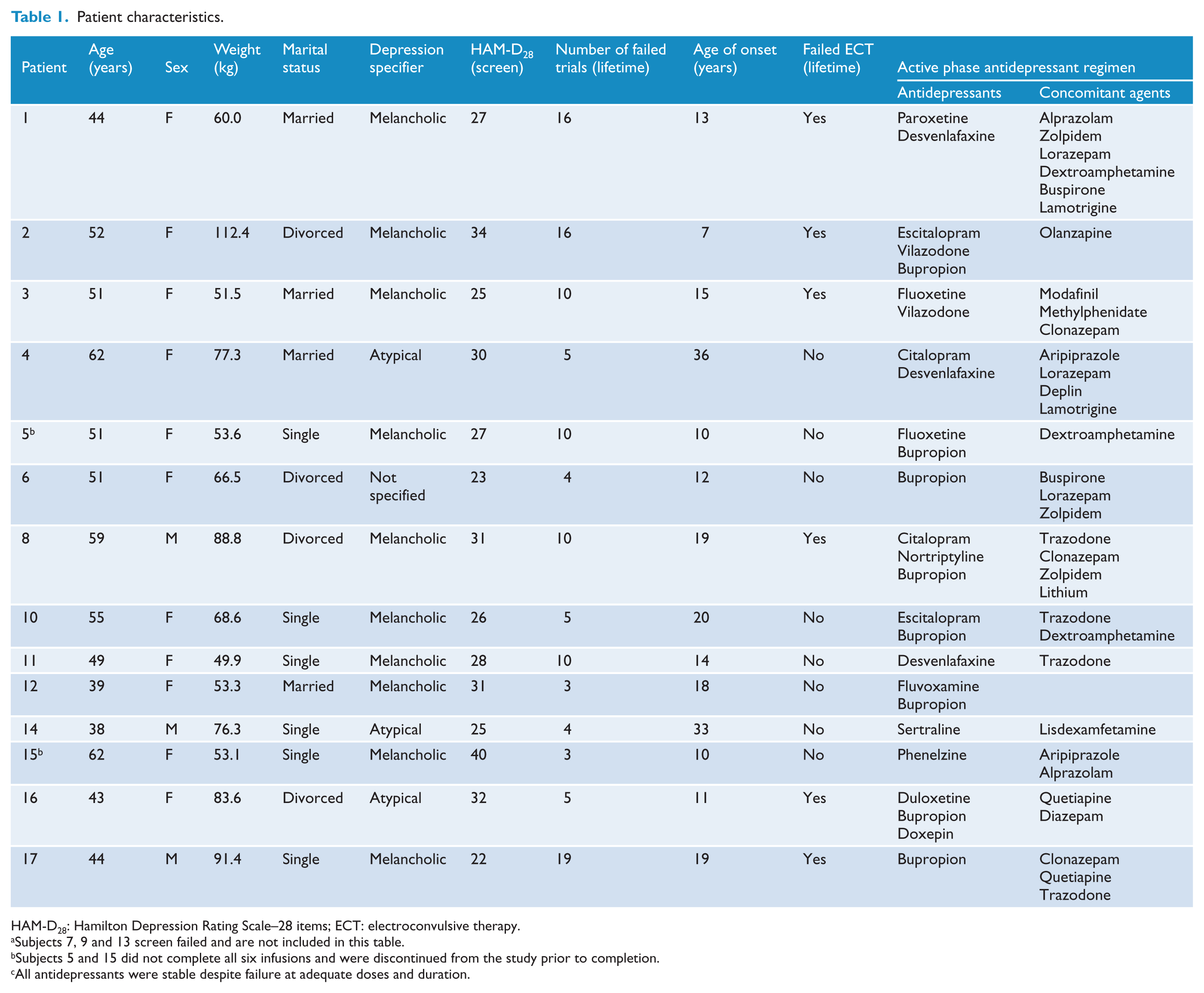
HAM-D28: Hamilton Depression Rating Scale–28 items; ECT: electroconvulsive therapy.
Subjects 7, 9 and 13 screen failed and are not included in this table.
Subjects 5 and 15 did not complete all six infusions and were discontinued from the study prior to completion.
All antidepressants were stable despite failure at adequate doses and duration.
Of the 14 enrolled subjects, 7 (50%) had one or more comorbid anxiety disorders, 3 (21%) had comorbid posttraumatic stress disorder PTSD, 2 (14%) had past history of eating disorders and 2 (14%) had past history of alcohol dependence. Regarding depression subtypes, 10/14 (71%) patients met criteria for melancholic depression, 3 (21%) for atypical depression and 1 (7%) did not meet criteria for melancholic or atypical depression (‘not specified’).
The mean HAM-D28 score at Baseline 0 was 28.6 ± 4.8 (mean ± standard deviation) and the mean CGI-S was 5.3 ± 0.6, indicating moderate-to-severe depression. Patients were taking an average of 1.9 ± 1.0 antidepressants and 1.9 ± 1.7 other psychotropic medications (i.e. mood stabilizers, atypical antipsychotics, benzodiazepines). On average, subjects had failed 8.3 ± 5.7 previous antidepressant trials in the current episode and 6/14 (42.9%) had failed an adequate course of electroconvulsive therapy (ECT) either in the current episode or lifetime. The average dose of ketamine administered during the first three infusions was 29.0 ± 16.2 and 43.5 ± 24.3 mg during the subsequent three infusions.
Primary outcome
After completing three infusions of ketamine at 0.5 mg/kg, the dose of ketamine was increased to 0.75 mg/kg for the following three infusions. After Infusion 6, 5/12 completers met criteria for response (41.7%) and 2/12 (16.7%) met criteria for remission. Of the responders, 3/5 (60%) met criteria for melancholic depression and 2 (40%) met criteria for atypical depression; both remitters had melancholic depression. ITT response and remission rates were 5/14 (35.7%) and 2/14 (14.3%), respectively.
Figure 1 shows the average HAM-D28 scores for each visit. Infusion 6 HAM-D28 scores were significantly improved compared to scores at Baseline 0, Baseline 1, Baseline 2 and Infusion 1 (all ps ⩽ 0.001), suggesting a significant improvement during the last infusion of ketamine.
From Baseline 0 to Infusion 3, there was a decrease in HAM-D28 scores that approached statistical significance (t(13) = 2.14, p = 0.052) with a medium effect size (Cohen’s d = 0.48). In contrast, from Infusion 3 to Infusion 6, there was a statistically significant difference in HAM-D28 scores (t(13) = 3.79, p = 0.002) and a large effect size (d = 1.01).
Additionally, the average change from Infusion 3 to Infusion 6 was 8.6 points on the HAM-D28 scale (31.8% improvement), compared to an average change from Baseline 0 to Infusion 3 of 2.6 points (8.7% improvement). A paired t-test comparing the average improvement for each individual across these two specified intervals (Baseline 0 to Infusion 3 vs Infusion 3 to Infusion 6) was statistically significant (t(13) = 2.57, p = 0.02). This result indicates that the improvement during treatment with 0.75 mg/kg of ketamine was significantly greater than the improvement during treatment with 0.5 mg/kg of ketamine.
Efficacy: 3-month follow-up
Patients were evaluated every 2 weeks during the 3-month follow-up. One of the five responders experienced a sustained response for 6 weeks after the final infusions, while the other four responders relapsed by 2 weeks. One non-responder at the final infusion continued to improve during the 3-month follow-up and approached the cut-off for response in the absence of changes in medication regimen. These data suggest that the combination of antidepressants, mood stabilizers and/or atypical antipsychotics did not appear to be helpful in maintaining the response to ketamine.
Tolerability
Percentages for specific physical, dissociative and sympathomimetic side effects for 0.5 and 0.75 mg/kg dosages are shown in Table 2. No serious adverse events were reported during the course of the study.
Side effects for lower versus higher dose of ketamine.
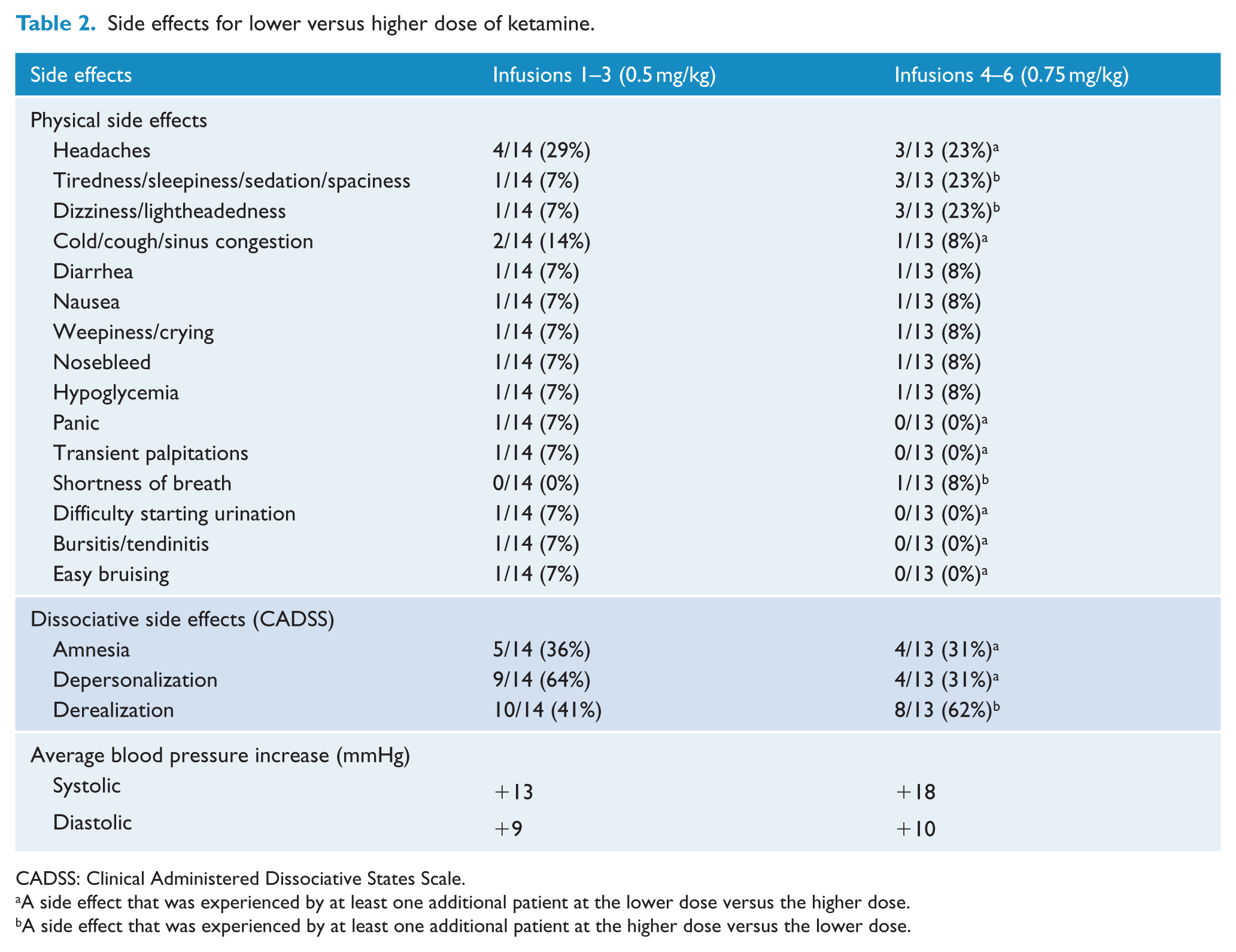
CADSS: Clinical Administered Dissociative States Scale.
A side effect that was experienced by at least one additional patient at the lower dose versus the higher dose.
A side effect that was experienced by at least one additional patient at the higher dose versus the lower dose.
Across both doses, side effects associated with ketamine infusion included mild visual disturbances, moderate auditory disturbances (‘buzzing sounds’) and mild dissociative symptoms based on the CADSS ratings. Ketamine administration was also associated with mild drowsiness or sedation and mild physical adverse events such as headache and nausea. The majority of these effects dissipated within 60–120 minutes upon the completion of ketamine administration.
In no case did side effects persist beyond 60 minutes after the infusion (based on CADSS ratings). Vital signs showed mild transient increases in systolic and diastolic blood pressure during the infusion, with all patients experiencing at least a 10-mmHg elevation in systolic blood pressure during the infusion. No significant changes in pulse rate or oxygen saturation were observed before, during or after the infusion.
Discussion
This is the first study to demonstrate that the administration of repeated, flexible-dose intravenous ketamine augmentation significantly improved depression symptoms in medicated outpatients with TRD. Specifically, patients experienced significant improvements between Infusion 1 and Infusion 6, with more pronounced improvement occurring during the escalated dose (0.75 mg/kg) phase compared to lower dose (0.5 mg/kg) phase. Both the repetition and increased dose of ketamine were well tolerated, with no serious adverse side effects.
Our findings in moderately to severely depressed outpatients are relevant to the practice of general clinical psychiatry as our sample represents typical patients often seen in TRD clinics. The high average number of treatment failures (8.3 ± 5.7) in the current episode of our cohort indicates a high level of resistance to antidepressant treatment; indeed, 42.9% had failed ECT (in the current episode or lifetime). While many patients with TRD are severely depressed and chronically suicidal, hospitalization is not indicated for the vast majority as they preserve sufficient functioning and they are not at imminent risk of self-harm. This underscores the need for treatment for this specific cohort; indeed, our data confirm the work by Ibrahim et al. (2011), who reported rapid decreases in depressive symptoms post-ketamine in patients resistant to ECT.
Of note, our cohort had a surprisingly low response rate (7.1%) to the first three ketamine doses (0.5 mg/kg over 40 minutes) compared to previous research. For example, response rates as high as 64% have been observed at the 4-hour mark in single-infusion studies (Zarate et al., 2006, 2012). The ‘reverse placebo’ effect may explain this discrepancy; in our study, patients knew that they would be receiving a higher open-label ketamine dose from Infusions 4 to 6. Therefore, they may have been predisposed to have less of a response to the initial (‘lower’) doses. Of note, our outpatient sample had a high number of psychiatric co-morbidities. We must consider the possibility that the significant heterogeneity of our sample contributed to our overall lower response and remission rates, compared to the results of previous ketamine studies for depression. Ultimately, our sample may provide a window into the issues that may occur if the widespread use of ketamine for depression becomes a part of clinical practice.
The incidence of subjective side effects was comparable to that in prior studies of ketamine for MDD. Overall, ketamine at both doses was well tolerated. For the higher dose of ketamine compared to the lower dose, an increase by at least one patient was observed for the following side effects: tiredness/sleepiness/sedation/spaciness, dizziness/lightheadedness, shortness of breath and derealization; additionally, there was a 5-mmHg additional increase in systolic blood pressure with the higher ketamine dose compared to the lower. (Of note, patients taking immediate-release stimulants [2/14] had higher average increases in blood pressure, which was not observed in patients taking sustained-release stimulants [2/14].) Compared to the higher ketamine dose, the following symptoms were observed in at least one additional patient at the lower dose: headaches, cold/cough/sinus congestion, panic, transient palpitations, difficulty starting urination, bursitis/tendonitis, easy bruising, amnesia and depersonalization. Most notably, the percentage of patients who experienced depersonalization during the higher ketamine dose (4/13; 31%) was much lower than the percentage experiencing depersonalization at the lower dose (9/14; 64%). Finally, retention was remarkably good (despite lengthy, bi-weekly study visits).
This study has several important limitations. First, the open-label design did not include a placebo arm. Given the robust but short-lived response, we cannot exclude the possibility that this improvement was due to a placebo effect. However, severe TRD has historically been considered a disorder with very low placebo response rate (Nelson and Papakostas, 2009). Second, we acknowledge that the heterogeneity of concomitant antidepressant regimens is a possible confounding factor. However, patients were maintained on an optimized regimen (i.e. adequate dose and duration of antidepressant medications) for at least 4 weeks prior to the first infusion, thereby limiting the influence of changing drug levels on ketamine’s efficacy. In addition, the use of ketamine as an augmentation strategy (as opposed to monotherapy) may be more clinically useful for ‘real-world’ psychiatric clinics, at which patients are often unable to be safely tapered off of their medications. Regardless, further a priori research is necessary to explore the influence of concomitant medications on the antidepressant effects of ketamine.
Third, aside from CADSS and vital signs, ratings were not obtained on the morning prior to the infusion. Ketamine’s antidepressant effects have been observed as early as 110 minutes post-infusion (Zarate et al., 2006); therefore, we administered rating scales at 3 hours post-infusion. However, since a single dose of ketamine may be significantly effective as late as 24 hours post-infusion (Murrough et al., 2013a), it is possible that scores obtained 3 hours post-infusion reflected the state of the patient following the current infusion only. Fourth, the extent to which the improvements between Infusion 1 and Infusion 6 were related to the increased ketamine dose versus the cumulative effect of ketamine administration over time (regardless of dose) remains unknown within the design of this study. Fifth, we did not use a rating scale to prospectively assess substance dependence risk. Although we excluded patients with active substance misuse disorders within the past year or positive urine toxicology, and no patients met criteria for dependence throughout the follow-up period based on clinical assessment, we acknowledge the need for more objective measures when assessing substance misuse with repeated ketamine dosing. In addition, the sample size of this study is small, and we did not assess for long-term side effects that have been associated with ketamine abuse (e.g. neurotoxicity, bladder dysfunction), underscoring the need for larger, confirmatory trials.
Finally, the time interval between infusions varied between and within patients, due to hospital and patient schedules, making the ‘standardization’ of the infusions difficult. However, these variable times are likely to be more representative of scenarios experienced in ‘real-world’ settings, when scheduling conflicts arise and visits are not always on the same day/time. Typically, the time range between infusions was within 1–2 days. Furthermore, the optimal dosing, frequency and route of administration of ketamine for depression remain unknown, making it somewhat difficult to translate research findings into a feasibly clinical practice model. Toward this end, a multi-site dose-finding study is underway and actively recruiting (NCT01920555).
In conclusion, this report demonstrates the tolerability and potential efficacy of two-step escalating ketamine augmentation for treatment-resistant depressed outpatients. Our cohort showed improvement from repeated ketamine infusions, despite general lack of response to the first infusion. Larger, controlled studies are needed to confirm these preliminary findings.
Footnotes
Acknowledgements
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by funds from the Department of Psychiatry, Massachusetts General Hospital, Boston, MA; from Harvard Clinical and Translational Science Center (Grant Number: 8UL1TR000170-05) and from the National Center for Advancing Translational Science. The funding source had no involvement in study design, collection, analysis or interpretation of data; in writing the report; or in the decision to submit the article for publication.
