Abstract
Objective:
Vascular endothelial growth factor A is a growth factor with pro-angiogenic and neurotrophic properties. Anti-vascular endothelial growth factor A treatments, used to treat cancers and opthalmic diseases, are known to induce depressive symptoms. Thus, we hypothesized that vascular endothelial growth factor A plasma levels are low in patients experiencing a major depressive episode in the context of major depressive disorder, which consequently increase after antidepressant treatment. The aim of this study was to compare plasma vascular endothelial growth factor A levels in patients with major depressive episode-major depressive disorder before and after antidepressant treatment.
Methods:
Vascular endothelial growth factor A fasting plasma levels of 469 major depressive episode-major depressive disorder patients were compared with healthy controls. Depressed patients were assessed for remission after 3 and 6 months of antidepressant treatment. Bivariate and multivariate analyses adjusted for sex, age, body mass index and tobacco use were performed.
Results:
As compared to healthy controls, major depressive episode patients had lower vascular endothelial growth factor A, 66.0 (38.3) pg/mL (standard deviation) vs 83.2 (49.2) pg/mL,
Conclusion:
Depressed patients with major depressive disorder have lower plasma vascular endothelial growth factor A levels than healthy controls during their major depressive episode and after remission following antidepressant treatment. New strategies targeting enhancement of plasma vascular endothelial growth factor A could be promising for the prevention and treatment of major depressive disorder.
Keywords
Introduction
Major depressive disorder (MDD) is the second leading cause of disability worldwide (Mokdad et al., 2016). Identifying biomarkers associated with MDD could improve our understanding of the pathophysiological mechanisms underlying this disorder and could help to discover new treatments for MDD.
Vascular endothelial growth factor A (VEGF-A) is a growth factor with pro-angiogenic (Ferrara and Henzel, 1989) and neurotrophic properties (Fournier and Duman, 2012; Khaibullina et al., 2004; Newton et al., 2013; Sun et al., 2010; Warner-Schmidt and Duman, 2007). Humans have at least nine subtypes of VEGF-A derived from different splices of the same gene located on 6p21.3 (Takahashi and Shibuya, 2005). VEGF-A is produced by endothelial cells, macrophages and activated T cells; is found in the central nervous system (CNS); and crosses the blood-brain barrier (Fabel et al., 2003).
Anti-VEGF-A drugs can treat certain cancers such as metastatic colorectal cancers, metastatic breast cancers, glioblastomas and age-related macular degeneration (AMD). Some data show that these anti-VEGF-A drugs can induce depressive symptoms and major depressive episodes (MDEs) (Schmidinger, 2013). Sorafenib, an inhibitor of the VEGF-A VEGF-R2 receptor, prescribed to treat hepatocellular carcinomas, can induce MDEs in up to 20% of patients (Qu et al., 2012). Ranibizumab, an anti-VEGF antibody which inhibits VEGF-A binding with two of its receptors (VEGF-R1 and R2), prescribed to treat AMD, can also induce MDEs in 20–25% of patients (Senra et al., 2016)
In addition, a decrease in synaptic plasticity in the hippocampus of rats treated with bevacizumab was observed. This effect has also been described as a biological change in patients with MDD (Fathpour et al., 2014). Bevacizumab, another anti-VEGF antibody inhibiting the binding of VEGF-A with its receptors VEGF-R1 and R2, can also induce a decrease in plasma VEGF-A levels 7 days after a single intravitreal injection in patients with AMD and diabetic macular edema (Zehetner et al., 2013). Of note, hippocampal VEGF-A levels decrease in rat models of depression (Flinders Sensitive Line) (Elfving et al., 2010) and can increase after antidepressant treatment (Ferres-Coy et al., 2013; Lesemann et al., 2012; Warner-Schmidt and Duman, 2008). These results are in line with the neurotrophic hypothesis of MDD, whereby the decreased level of neurotrophins may cause atrophy of limbic structures that control mood, resulting in depressive symptoms (Clark-Raymond et al., 2014).
Conversely, a recent meta-analysis including five studies with 180 cases and 145 controls shows that VEGF-A plasma levels are increased in patients with MDD (Carvalho et al., 2015). However, relevant factors associated with VEGF-A levels (including age, sex, body mass index [BMI] and tobacco use) (Conklin et al., 2002; Loebig et al., 2010) were not taken into account in this meta-analysis.
In MDE patients, two underpowered studies (
However, despite these studies with ECT, the association between VEGF-A plasma level changes after antidepressant drug treatment and remission was not studied. Thus, VEGF-A plasma levels should be better studied in a large sample of patients with major depression before and after treatment and in HCs, while controlling for confounding factors.
The aim of this study was to compare plasma VEGF-A levels in MDD patients with a current MDE before and after antidepressant treatment (particularly in those in remission) and in healthy controls (HCs).
Materials and methods
Patients with major depression
In the METADAP study (Corruble et al., 2015), 469 consecutive inpatients or outpatients, aged 18–65 years, with a current MDE (according to Diagnostic and Statistical Manual of Mental Disorders-IV-TR criteria) in a context of MDD (with a minimum score of 18 at the HRDS-17 items) (Hamilton, 1960) were assessed for clinical and biochemical variables at the introduction of an index antidepressant treatment. All of them provided written informed consent. Patients with psychotic symptoms, bipolar disorders, psychotic disorders, eating disorders, current substance abuse or dependence, pregnancy, organic brain syndromes and severe unstable medical conditions or those receiving antipsychotics or mood stabilizers before inclusion and/or for 4 months or more during the last year could not be included. This study was registered by the French National Agency for Medicine and Health Products Safety (ANSM) and the Commission Nationale de l’Informatique et des Libertés (CNIL). It was approved by the Ethics Committee of Paris Boulogne, France, and conformed to international ethical standards. The METADAP study was funded by a national grant (PHRC, AOM06022) and sponsored by Assistance Publique-Hôpitaux de Paris (APHP) (ClinicalTrials.gov Identifier: NCT00526383).
Patients included were in naturalistic conditions, treated or untreated with antidepressants, before introduction of the index antidepressant. For patients previously treated with antidepressants, there was no systematic washout period before index of the new treatment. The index antidepressant treatment must belong to one of the four following classes: selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), imipraminics (IMI; tricyclic antidepressants ‘in general’) and other antidepressant treatments (others). A monotherapy of antidepressant was required. The drug and its dose were left to the patient’s psychiatrist, using ‘real world’ treatment options. When antidepressant treatment was changed, the patient was reassessed and removed from the study.
The HDRS (Hamilton, 1960) was rated by trained clinicians at baseline and after 3 and 6 months of antidepressant treatment.
Response and remission were assessed after 3 (M3) and 6 months (M6) of antidepressant treatment. Response was defined by a decrease in the HDRS total score of at least 50% from baseline to follow-up (Rush et al., 2006). Remission was defined by an HDRS total score of 7 or less at follow-up (Moller, 2008; Rush et al., 2006).
HCs
HCs were those of the VARIETE study, a population-based cross-section of the general population (Chanson et al., 2016). Subjects were recruited from 10 French university hospitals and enrolled between January 2011 and February 2012. To be included in the study, adult subjects (aged 18–89 years) had to be considered healthy based on medical history, clinical examination and standard biological tests performed after an overnight fast. The study included 893 subjects. All participants gave written informed consent before entering the study. This study was approved by the French National Agency for Medicine and Health Products Safety (ANSM) as well as by the Ethics Committee (Ile de France VII). The VARIETE study was funded by a national grant (PHRC, AOM09122) and sponsored by APHP (NCT01831648).
VEGF-A plasma levels
Plasma was preferred to serum to measure VEGF-A levels. This is because a significant and highly variable platelet-mediated secretion of VEGF-A during the clotting process invalidates the use of serum as an indicator of circulating VEGF-A levels (Webb et al., 1998).
Plasma samples were taken after an overnight fast and stored at −80°C. VEGF-A plasma levels were assessed using a commercial multiplex enzyme-linked immunosorbent assay (ELISA) kit assaying the VEGF-A using blind clinical data as a reference (Kit Reference: V-PLEX Angiogenesis Panel 1 [human], Meso Scale Discovery Kit, Rockville, USA). The measurement range was 40–1510 pg/mL. The mean intra-assay and interassay coefficients of variation (CV) were 2.7% and 6.0% for 16.7 pg/mL and 2.1% and 6.3% for 129 pg/mL, respectively. VEGF-A plasma levels were quantified in a central laboratory using controlled sampling and pre-analytical procedures.
Statistical analyses
The statistical analyses were performed using R 3.4.2 (r-project.org) and STATA MP (version 13). VEGF-A plasma levels were normally distributed (graphical assessment). Outlier values (higher than the VEGF-A third quartile value + three times the interquartile range) were excluded, corresponding to 23 patients and 20 controls at baseline.
Bivariate analyses were performed to compare MDE patients and HCs for socio-demographical and clinical characteristics (chi-square test and Student’s
Linear models were performed to control for potential confounders among demographic and clinical variables. Covariables of the multivariate models were selected based on a significant difference in bivariate analyses (
To assess VEGF-A plasma level changes after antidepressant treatment in MDE patients, mixed-effect multivariate models were used (including age, sex and BMI and current smoker status). They are a well-accepted method for analyzing longitudinal clinical data in which missing or mistimed observations are present.
The association between baseline VEGF-A plasma levels and response/remission after treatment were analyzed with Student’s
Results
Socio-demographic characteristics
Age, sex, BMI and smoker status differed between the 469 MDE patients and 893 HCs with a mean age of 45.9 (12.9; standard deviation) years vs 39.8 (18.6) years, 68.6% of women vs 49%, a mean BMI of 24.1 (4.9) kg/m2 vs 23.07 (2.4) kg/m2 and 37.1% of smokers vs 10.7% (
Two hundred fifteen (46.0%) patients were removed from the study before M3 (after 3 months of antidepressant treatment) and a further 70 (15.0%) later. Reasons for dropping out were the following: cessation of antidepressant medication studied,
VEGF-A plasma levels in MDE patients and HCs
VEGF-A plasma levels were associated with age (
As compared to HCs, at baseline, MDE patients had lower VEGF-A, 66.0 (38.3) pg/mL vs 83.2 (49.2) pg/mL, estimate = −16.7, bivariate:
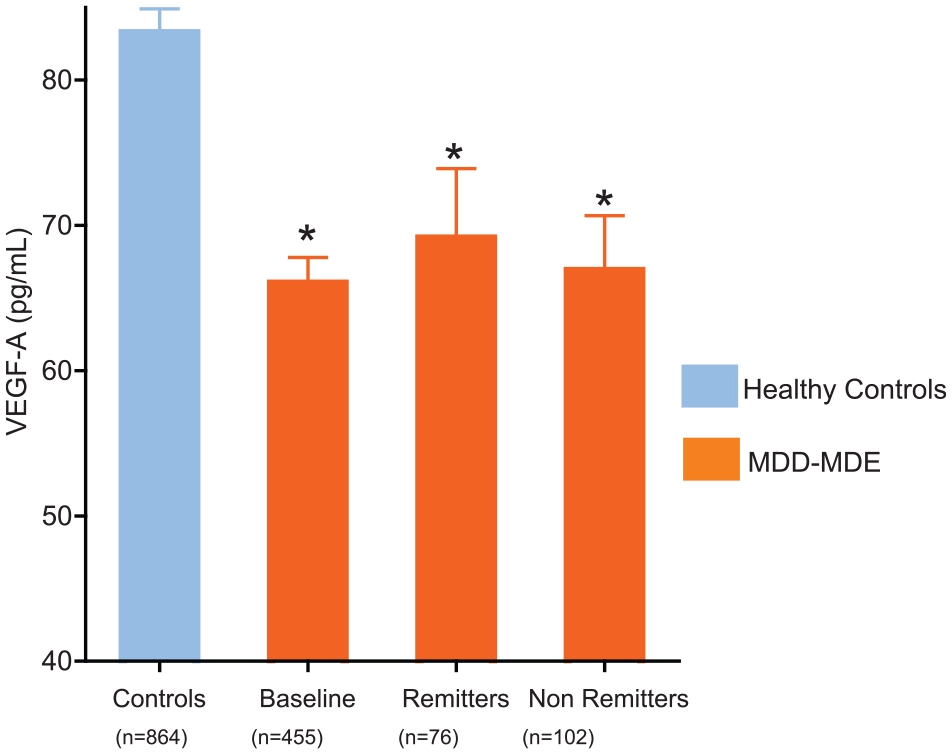
Plasma VEGF-A levels in MDD patients during MDEs and in remission or non-remission after antidepressant treatment compared to HCs.
These results remained significant after adjusting for sex, age, BMI and tobacco use (estimate = −27.8,
In MDE patients, we found no difference between antidepressant-naïve patients, VEGF-A 64.7 (38.1) pg/mL, and non-antidepressant drug-naïve patients at baseline, VEGF-A 66.4 (38.4) pg/mL, (
Plasma VEGF-A level changes after antidepressant treatment
Plasma VEGF-A levels did not change significantly over time in mixed multivariate models (M0 = 66.0 [38.3] pg/mL, M3 = 63.9 [33.9] pg/mL and M6 = 63.2 [29.6],
Plasma VEGF-A levels and response/remission after antidepressant treatment
There was no significant association between plasma VEGF-A levels at baseline and response/remission after 3 and 6 months of antidepressant treatment. The plasma VEGF-A level changes with time were not associated with response and remission (after 3 and 6 months) in multivariate mixed models (data not shown). As compared to HCs, remitters had lower VEGF-A levels, 65.98 (38.3) pg/mL vs 83.2 (49.2) pg/mL, bivariate:
Discussion
MDE patients had lower plasma VEGF-A levels than HCs. Their VEGF-A plasma levels did not change after antidepressant treatment and remained lower than those of HCs.
In line with preclinical results reporting lower hippocampal VEGF-A levels in rat models of depression (Flinders Sensitive Line) (Elfving et al., 2010), we observed lower plasma VEGF-A levels in MDE patients. This result fits the neurotrophic hypothesis of MDEs (Duman et al., 1997). It is also in line with the observed induced MDE and depressive symptoms after anti-VEGF-A treatment (Qu et al., 2012; Schmidinger, 2013; Senra et al., 2016). Altogether, these results suggest that the lower VEGF-A level could be a risk factor for MDEs. However, longitudinal studies are needed to test this hypothesis. Moreover, it would be valuable to measure plasma VEGF-A in patients before the onset of depression and thus detect patients at risk of a depressive episode.
The lower VEGF-A plasma levels shown in this sample of MDE patients contrast with the results of the previous meta-analysis (Carvalho et al., 2015), which reports an increase in plasma VEGF-A levels in 180 depressed patients compared to 145 controls. However, confounding factors were not taken into account in this meta-analysis (Carvalho et al., 2015). Indeed, four of five studies included did not present data on tobacco use (Fornaro et al., 2013; Lee and Kim, 2012), BMI (Fornaro et al., 2013; Takebayashi et al., 2010) or pooled unipolar and bipolar patients (Dome et al., 2009). Two studies included patients treated with mood stabilizers (Dome et al., 2009; Takebayashi et al., 2010). Our study focused on an homogeneous sample of patients with unipolar MDD, without treatment with mood stabilizers, and the results were adjusted for tobacco use and BMI. Moreover, in the previous meta-analysis, methods of the plasma VEGF-A level measurement were heterogeneous, and there was a large difference regarding VEGF-A level range between the meta-analyzed studies. VEGF-A levels above 400 pg/mL in MDEs and controls in one study (Fornaro et al., 2013) but lower than 10.5 pg/mL for MDEs and controls in another (Clark-Raymond et al., 2014). Among the measurement methods, some kits used (RayBio, RayBiotech Life, Peachtree Corners, USA; Fornaro et al., 2013) have not tested all the different types of VEGF in cross-reactivity. In our study, to measure plasma VEGF-A levels, an ELISA kit for plasma VEGF-A levels was used with no cross-reactivity between VEGFs. Indeed, different types of VEGFs have been described, which have different functions to VEGF-A (Takahashi and Shibuya, 2005). Altogether, our study provides the largest sample of MDE patients and HCs compared for VEGF-A plasma levels.
We observed no change in VEGF-A after antidepressant treatment. Three studies in rodents showed an increase in hippocampal VEGF-A after treatment with fluoxetine (Lesemann et al., 2012), other SSRIs (Ferres-Coy et al., 2013) and electroconvulsive seizure (Warner-Schmidt et al., 2008). Warner-Schmidt et al. (2008) found that expression of VEGF in the granule cell layer of the hippocampus is decreased by irradiation and that administration of electroconvulsive seizures can reverse this deficit. Of note, these studies assessed brain VEGF-A levels, whereas we assessed peripheral levels. Regarding depressed patients, our results are in line with those of the three prospective open studies including 24–34 patients reporting no plasma VEGF-A level changes after antidepressant treatments (Dome et al., 2012; Fornaro et al., 2013; Halmai et al., 2013). Our results in the largest sample of MDE patients go beyond these previous studies by showing that VEGF-A levels did not change in MDD-MDE patients with remission after treatment and remain lower than those of HCs.
Altogether, VEGF-A plasma levels were lower in MDE patients and remain lower in remitters after treatment, suggesting, first, that lower VEGF-A levels could be a trait marker of MDD and, second, that new strategies targeting enhancement of plasma VEGF-A could be promising for the prevention and treatment of MDD.
There are some limits which must be underlined. First, we did not control for potential confounding factors, such as the presence of cancers, which has previously been correlated with an increase in plasma VEGF-A (Hyodo et al., 1998). Nonetheless, cancers are likely to be very rare in the METADAP cohort since patients with unstable medical conditions were excluded and patients with cancer were excluded from the VARIETE study. In addition, we did not control for cardiovascular disorders that can also influence peripheral VEGF-A levels (Marks et al., 2018). Second, VEGF-A was only assessed at the peripheral level but not at the central level. To our knowledge, no clinical or preclinical data have been collected to assess the relationship between brain and plasma VEGF-A levels. One study (Elfving et al., 2010) presented a decrease in brain VEGF-A levels in a rat model of depression but not in serum. However, serum levels should be interpreted with caution as they are highly dependent on platelet-mediated VEGF-A secretion during clotting (Webb et al., 1998).
This study has several strengths. It provides the largest sample of MDE patients assessed for VEGF-A fasting plasma levels and compared with HCs. Furthermore, it provides the first quantification of VEGF-A plasma levels assessed in a central laboratory with controlled sampling and pre-analytical procedures. Hence, it provides a reliable picture of plasma levels of VEGF-A in MDE patients.
This study provides a new insight into the peripheral signature of MDEs. It highlights the need for new preclinical and clinical studies to understand the role of VEGF-A in MDEs and to explore mechanisms associated with plasma VEGF-A decrease in MDE patients, for example, an epidemiological study in healthy subjects with measurement of VEGF and assessment of the occurrence of MDEs. Further research should include other types of VEGFs such as soluble VEGF-C, VEGF-D and VEGF receptor R1, which are also involved in neuroangiogenic processes (Warner-Schmidt and Duman, 2007). Our results also suggest new antidepressant therapeutic strategies, raising the possibility that VEGF-A might be a relevant target for successful treatment of MDEs in patients.
Conclusion
MDD is associated with lower plasma VEGF-A levels. New strategies capable of increasing plasma VEGF-A could be a promising opportunity for prevention and treatment of MDD.
Footnotes
Acknowledgements
We thank James Bowers for English corrections.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: L.B. is an investigator for Antisense Therapeutics, Alnylam Pharmaceuticals, Alexion, Actelion, Auris Medical, Gilead Sciences, Ionis Pharmaceuticals, MedDay Pharma, Novartis, PregLem SA and Ultragenix pharmaceutical; received consulting fees from Sanofi-Aventis, Pfizer, Kyowa Kirin and Servier and lecture fees from Genzyme, GlaxoSmithKline, Bristol-Myers Squibb, Merck Sharp and Dohme; a close family memberworks at Sanofi France. P.C. has received unrestricted research and educational grants from Ipsen, Novartis, Novo-Nordisk and Pfizer as the Head of the Department of Endocrinology and Reproductive Diseases, Hôpitaux Universitaires Paris-Sud; has served as an investigator (principal or coordinator) for clinical trials funded by Novartis, Pfizer, Ipsen, Italpharmaco, Antisense, Prolor Biotech, Chiasma and Æterna Zentaris; P.C. has been a member of the Advisory Board of HypoCCS sponsored by Eli Lilly; and P.C. gave lectures for Ipsen, Novartis and Pfizer. All the fees and honoraria are paid to his institution. The other authors have no conflict of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: METADAP cohort was funded by a national Grant (PHRC, AOM06022) and sponsored by Assistance Publique-Hôpitaux de Paris (APHP) (ClinicalTrials.gov Identifier: NCT00526383). The VARIETE study was funded by a national Grant (PHRC, AOM09122) and sponsored by APHP (NCT01831648) and was hosted in the Biological Resource Center (BRC) of Paris South (numero BRIF: BB-0033-00089).
