Abstract
Objective:
Delirium is an acute brain failure related to uncertain problems in neural connectivity, including aberrant functional interactions between remote cortical regions. This study aimed to elucidate the underlying neural mechanisms of delirium by clarifying the changes in resting-state functional connectivity induced by postoperative delirium using imaging data scanned before and after surgery.
Method:
Fifty-eight patients with a femoral neck fracture were preoperatively scanned using resting-state functional magnetic resonance imaging. Twenty-five patients developed postoperative delirium, and 14 of those had follow-up scans during delirium. Eighteen patients without delirium completed follow-up scans 5 or 6 days after surgery. We assessed group differences in voxel-based connectivity, in which the seeds were the posterior cingulate cortex, medial prefrontal cortex and 11 subcortical regions. Connections between the subcortical regions were also examined.
Results:
The results showed four major findings during delirium. Both the posterior cingulate cortex and medial prefrontal cortex were strongly connected to the dorsolateral prefrontal cortex. The posterior cingulate cortex had hyperconnectivity with the inferior parietal lobule, whereas the medial prefrontal cortex had hyperconnectivity with the frontopolar cortex and hypoconnectivity with the superior frontal gyrus. Connectivity of the striatum with the anterior cingulate cortex and insula was increased. Disconnections were found between the lower subcortical regions including the neurotransmitter origins and the striatum/thalamus in the upper level.
Conclusions:
Our findings suggest that cortical dysfunction during delirium is characterized by a diminution of the anticorrelation between the default mode network and task-positive regions, excessive internal connections in the posterior default mode network and a complex imbalance of internal connectivity in the anterior default mode network. These dysfunctions can be attributed to the loss of reciprocity between the default mode network and central executive network associated with defective function in the salience network, which might be closely linked to aberrant subcortical neurotransmission-related connectivity and striato-cortical connectivity.
Keywords
Introduction
Delirium is clinically important in general hospitals because it is closely related to longer hospital stays, increased mortality rates and increased costs (Marcantonio, 2017; Stevens et al., 1998). A few studies have suggested that both structural and functional brain abnormalities can occur during delirium (Nitchingham et al., 2018; Soiza et al., 2008). In particular, our group has reported that delirium is related to aberrant functional interactions between remote cortical regions, including a loss of the hub functions of the posterior cingulate cortex (PCC) in the default mode network (DMN) (Choi et al., 2012; van Montfort et al., 2018) and decreased functional connectivity among acetylcholine/dopamine-related subcortical regions (Choi et al., 2012).
It should be noted that these abnormalities were revealed through comparisons with the post-delirium state. Because cognitive dysfunctions can remain after recovery from delirium (Saczynski et al., 2012), the functional neural changes during delirium might not be completely recovered in the post-delirium state. Therefore, a comparison with the brain state before the development of delirium is necessary to elucidate the delirium-related abnormalities more clearly. The present study was designed to longitudinally investigate connectivity using resting-state functional magnetic resonance imaging (rsfMRI) scans before and after surgery in patients with femoral neck fractures. This study has the additional advantage of few confounding factors because the sample was homogeneous, whereas our previous studies included heterogeneous diagnostic groups (Choi et al., 2012; van Montfort et al., 2018).
Moreover, the present study uses a different methodology from previous research. First, we added the medial prefrontal cortex (mPFC) to analysis of the DMN. Because most neurotransmitter pathways involve prefrontal regions before reaching posterior cortical regions (Ballinger et al., 2016; Gorelova et al., 2012), the mPFC is as important a key region of the DMN as the PCC. Previous studies for other brain diseases showed that the hub functions of the PCC could be preserved for longer than the functions of other DMN hubs, including the mPFC (Miao et al., 2011; van Eimeren et al., 2009). Because delirium is an acute brain failure, mPFC dysfunction could be a sensitive indicator of DMN dysfunction in delirious patients. Second, we added analysis of subcortico-cortical connectivity, which could be crucial to investigating the mechanisms of delirium because it is closely related to neurotransmitter circuits that connect the subcortex and cortex (Maldonado, 2018). One typical subcortico-cortical circuit is the striato-cortical circuit, which has been implicated in the pathophysiology of psychosis. For example, improvement in psychosis is related to increased functional connectivity of the striatum with the anterior cingulate cortex (ACC), dorsolateral prefrontal cortex (DLPFC) and insula (Sarpal et al., 2015). Given the similarity between delirious symptoms and psychotic symptoms, an investigation of striato-cortical connectivity could help to elucidate the mechanism of delirium.
This study thus aimed to clarify the changes in resting-state functional connectivity induced by postoperative delirium. All participants were patients with femoral neck fractures, and rsfMRI scans were performed before and after surgery. Functional connectivity analysis was conducted to verify the following three hypotheses: Compared with healthy brain states (the preoperative state and postoperative non-delirium), postoperative delirium would be associated with (1) disrupted DMN hub functions, including both the PCC and mPFC; (2) aberrant subcortico-cortical connectivity and (3) decreased connectivity among the subcortical regions.
Methods
Participants
This study was conducted for 11 months on patients older than 70 who underwent orthopedic surgery at Yonsei University Gangnam Severance Hospital due to a femoral neck fracture. Before the surgery, the procedure was fully explained to both patients and their surrogates, and we obtained written informed consent. Of the 98 patients who met the criteria, 58 completed an initial preoperative magnetic resonance imaging (MRI) scan 1 day before surgery. Prior to surgery, demographic and clinical information was acquired. After surgery, patients were visited daily by a trained psychiatrist to diagnose delirium. If delirium occurred, follow-up MRI scanning was performed within 24 hours of delirium onset. Delirium severity on the date of delirium onset was assessed using the Korean version of the Delirium Rating Scale (K-DRS) (Trzepacz et al., 2001). Patients who did not develop delirium by the fifth postoperative day were classified as the non-delirium group, and the follow-up MRI was performed 5 or 6 days postoperatively. This study was approved by the institutional review board of Yonsei University Gangnam Severance Hospital.
Image acquisition and preprocessing
Preoperative rsfMRI, preoperative high resolution T1-weighted imaging and postoperative follow-up rsfMRI scans were obtained with a Signa EXITE 3.0 Tesla MR system (GE, Milwaukee, WI, USA). Functional images were scanned for 5 minutes using gradient-echo-planar imaging sequences (matrix = 64 × 64, echo time = 17.6 ms, repetition time = 2000 ms, field of view = 240 mm, slice thickness = 3 mm, flip angle = 90° and number of slices = 50). Participants were instructed to rest with their eyes closed during the scan. A T1-weighted image was scanned using a spoiled gradient-echo sequence (matrix = 256 × 256, echo time = 3.2 ms, repetition time = 8.2 ms, field of view = 240 mm, slice thickness = 1.2 mm, flip angle = 12° and number of slices = 136) to serve as an anatomical underlay for brain activity.
The time series data for the first 10 seconds were discarded to eliminate any signal decay related to the magnetization reaching equilibrium. The preprocessing step for the functional data included slice-timing correction, head motion correction, co-registration to the T1-weighted image for each participant, spatial normalization to the Montreal Neurological Institute template with the resolution of 2 mm isotropic voxel size and smoothing with a 6 mm full-width at half-maximum Gaussian filter using the standard pipeline provided in the functional connectivity toolbox (CONN) v.18a (www.nitrc.org/projects/conn) and Statistical Parametric Mapping (SPM) 12 software. Considering the importance of motion artifacts in patients with delirium, between-group differences in the number of invalid scans (outlier threshold for scan-to-scan motions: 0.5 mm), maximum displacement and mean displacement among the preoperative states, postoperative delirium states and postoperative non-delirium states were examined using the one-way analysis of variance (ANOVA). To address artifacts including spiking and motion, the aCompCor method was used as implemented in the CONN toolbox (Behzadi et al., 2007; Muschelli et al., 2014). The white matter, cerebrospinal fluid and realignment parameters were considered to be confounds in a first-level analysis, and a band-pass filter was applied from 0.008 to 0.09 Hz. This denoising step addressed the confounding effects of participant movement without regressing the global signal and without affecting intrinsic functional connectivity (Chai et al., 2012).
Regions of interest (ROIs)
The DMN and subcortical regions associated with acetylcholine, dopamine and the reticular activating system were considered to be seed regions for the imaging analysis. In terms of a seed in the DMN, two ROIs were defined as a 10 mm sphere centered at previously defined coordinates: the PCC (centered at coordinates:
Functional connectivity analysis
The analyses were performed using a seed-based correlation approach. Using the CONN toolbox, seed-to-voxel functional connectivity maps for each participant were generated. Voxel-wise correlations were calculated between the time series of seed regions and whole brain. Correlation coefficients were converted to
Results
Participant characteristics
Among the 58 participants who underwent a preoperative MRI scan, 25 suffered from postoperative delirium, and the remaining 33 did not. We found no statistical differences in age, gender or education level between the two groups (Table 1). Among the 25 patients with delirium, 11 patients did not undergo a follow-up scan because of surrogate refusal (
Demographics and clinical characteristics of participants.
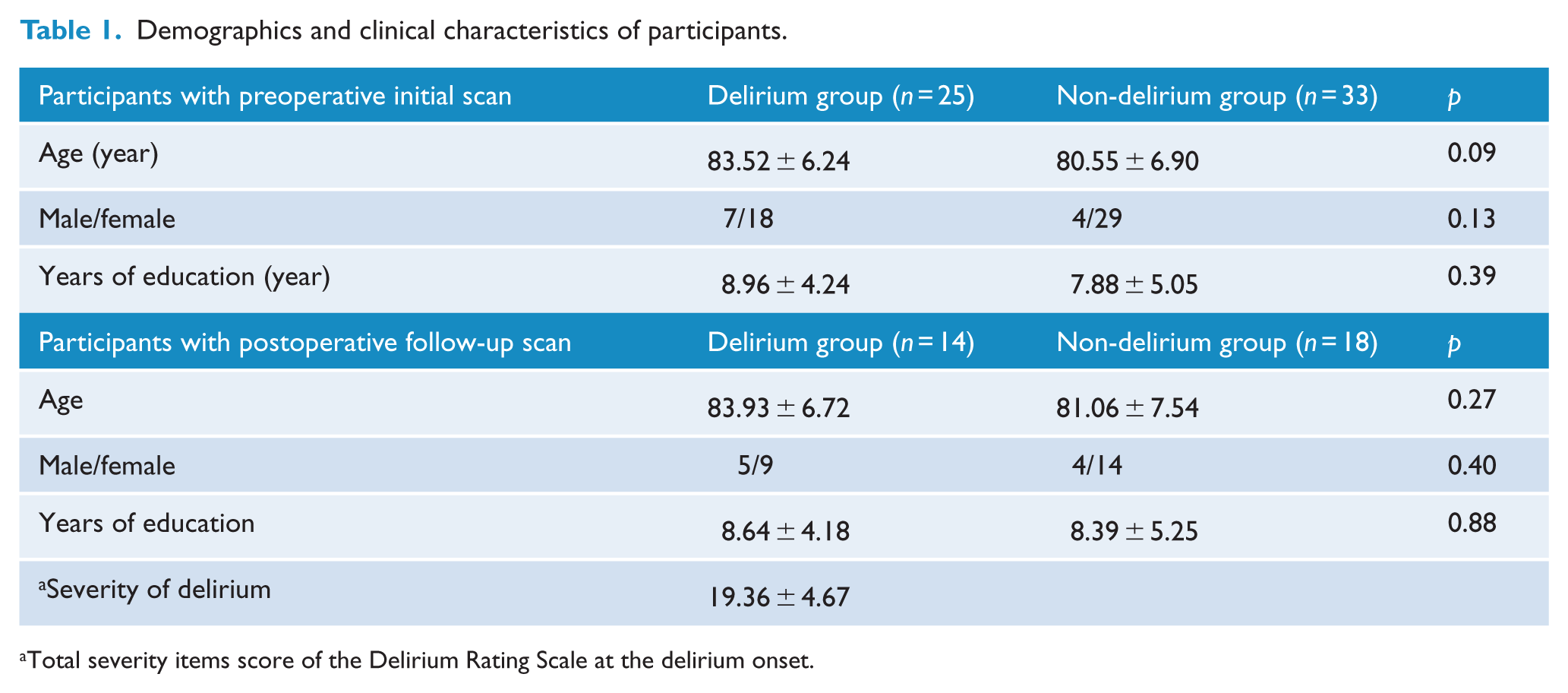
Total severity items score of the Delirium Rating Scale at the delirium onset.
Functional connectivity associated with the PCC and mPFC
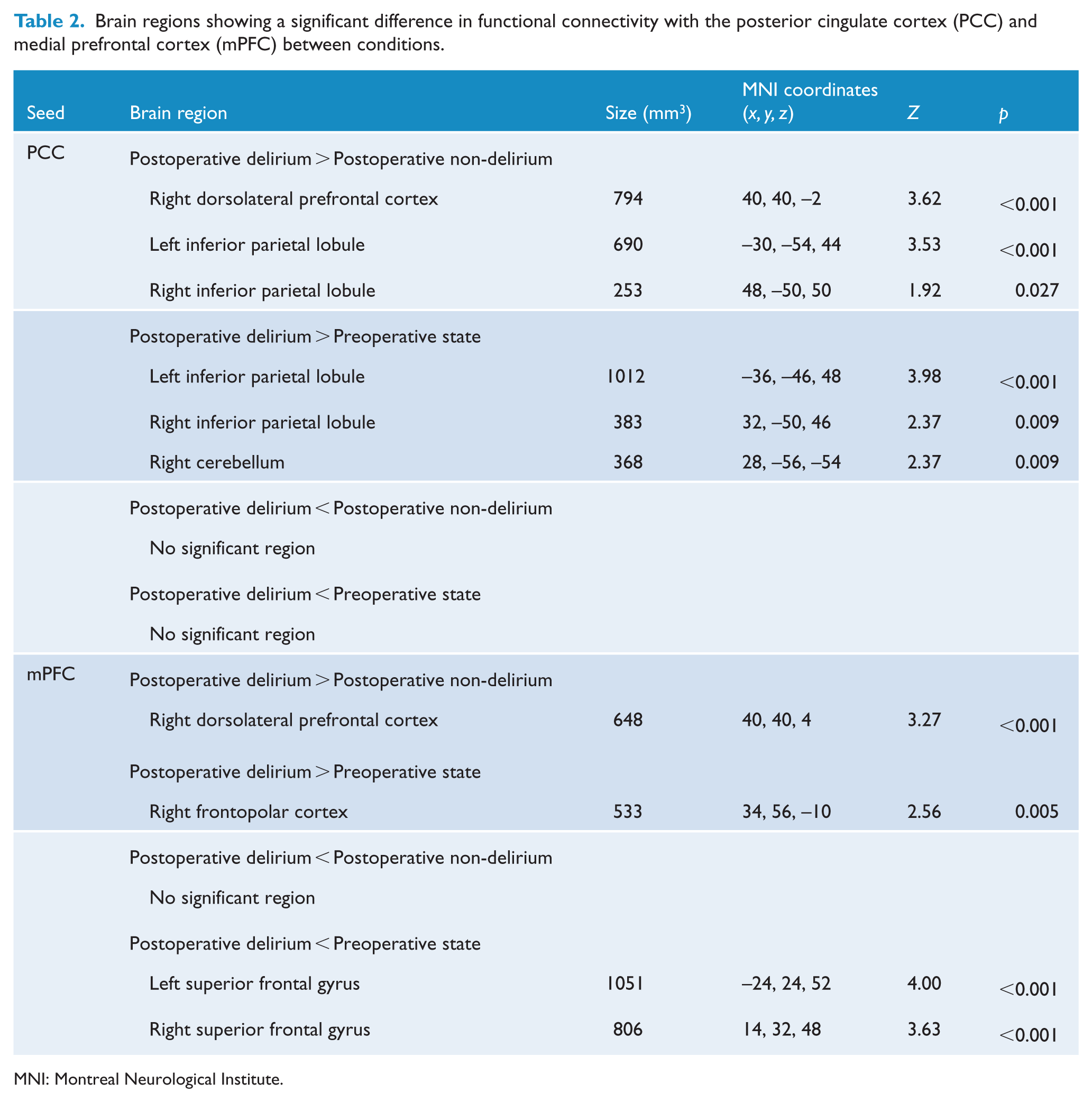
Figure 1 shows the DMN in the preoperative, postoperative non-delirium and postoperative delirium states. The obvious difference in a visual inspection was in the dorsal frontal regions; they were negatively correlated with the PCC in the preoperative state, but the negative correlations decreased in the postoperative non-delirium state and disappeared in the postoperative delirium state. Statistical differences between these states are listed in Table 2. A common result between PCC-based connectivity and mPFC-based connectivity was found in the DLPFC, which showed increased connectivity in the postoperative delirium state compared to the postoperative non-delirium state. As shown in Figure 2, several different results were found between PCC-based connectivity and mPFC-based connectivity. For instance, increased connectivity with the bilateral inferior parietal lobules in the postoperative delirium state compared with both the postoperative non-delirium and preoperative states was identified only in the PCC-based connectivity analysis. On the contrary, increased connectivity with the frontopolar cortex and decreased connectivity with the bilateral superior frontal gyrus in the postoperative delirium state compared with the preoperative state were found only in the mPFC-based connectivity analysis. Meanwhile, we found no significant difference in PCC-based connectivity or mPFC-based connectivity between the postoperative non-delirium state and preoperative state.
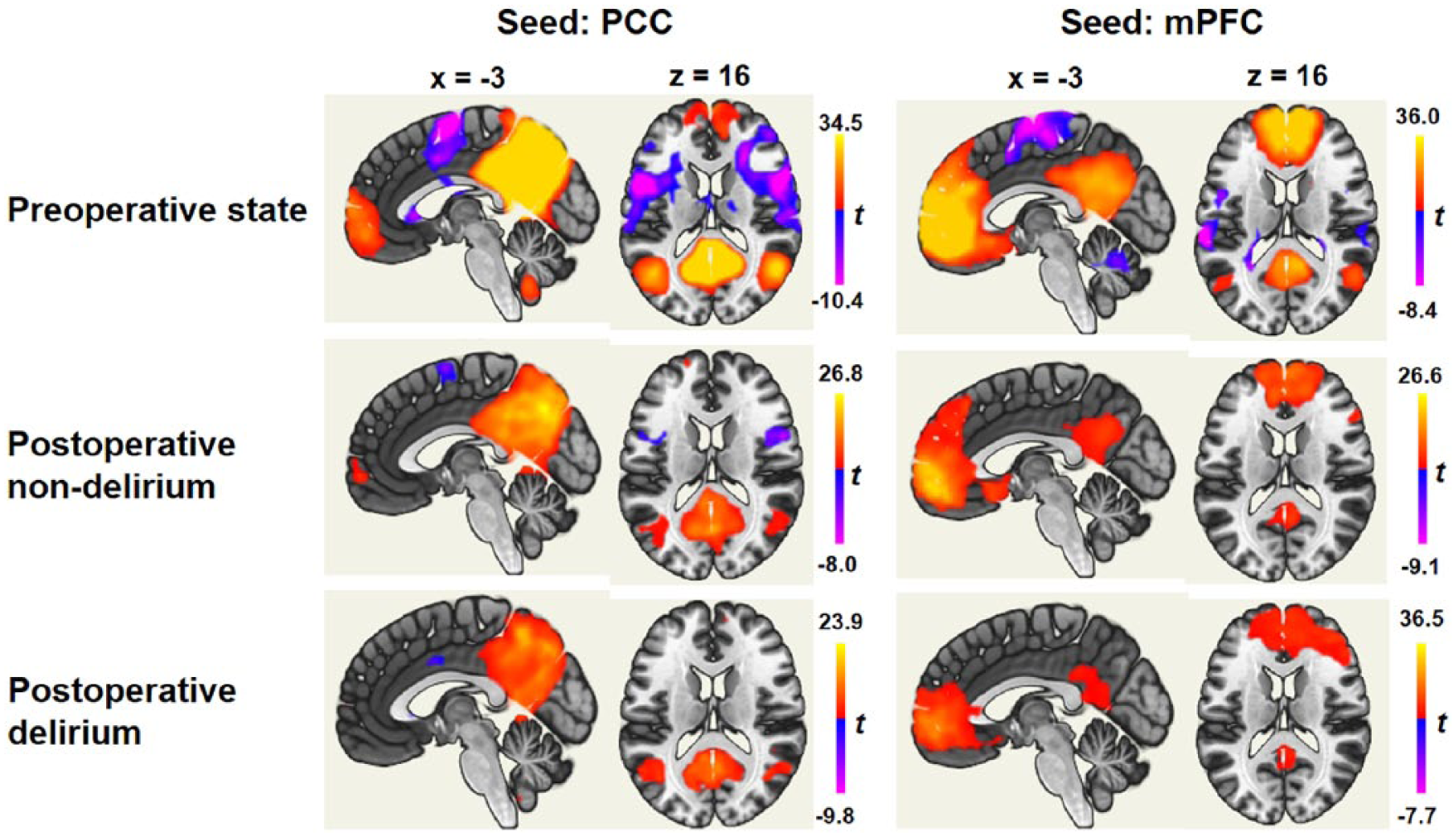
Functional connectivity with the posterior cingulate cortex (PCC) and medial prefrontal cortex (mPFC) in each of the preoperative, postoperative non-delirium and postoperative delirium states.
Brain regions showing a significant difference in functional connectivity with the posterior cingulate cortex (PCC) and medial prefrontal cortex (mPFC) between conditions.
MNI: Montreal Neurological Institute.
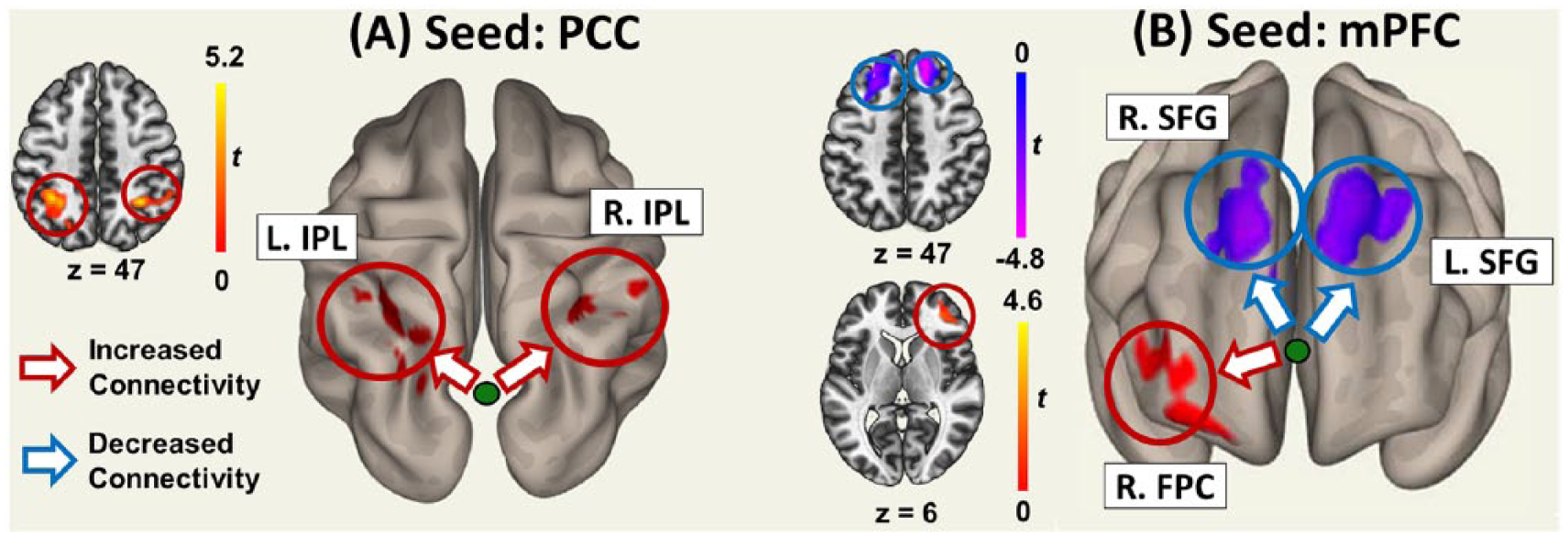
Increases or decreases of the posterior cingulate cortex (PCC)–based connectivity and medial prefrontal cortex (mPFC)–based connectivity during postoperative delirium when compared to the preoperative state: (A) PCC-based functional connectivity was increased with the bilateral inferior parietal lobule (IPL) (red circle); (B) mPFC-based functional connectivity was increased with the right frontopolar cortex (FPC) (red circle) and decreased with the bilateral superior frontal gyrus (SFG) (blue circle). L: left; R: right.
In the ANOVA of PCC-based connectivity, a main group effect was found only in the left inferior parietal lobule (
Subcortico-cortical functional connectivity
Table 3 shows only the significant results in subcortical seed-based functional connectivity. In the postoperative delirium state, compared with the preoperative state, increased connectivity with the striatum was shown in the ACC, left DLPFC, left temporal pole and left insula. Among these regions, the ACC and left insula also showed increased connectivity in the postoperative delirium state, compared with the postoperative non-delirium state. Decreased connectivity with the striatum in the postoperative delirium state was found in the right temporal pole compared with the postoperative non-delirium state and the right frontal eye field compared with the preoperative state. In connectivity with the right intralaminar thalamic nucleus, the left orbitofrontal cortex showed decreased connectivity in the postoperative non-delirium state, compared with the preoperative state. In connectivity with the right nucleus basalis, the bilateral orbitofrontal cortex showed decreased connectivity in the postoperative delirium state, compared with the postoperative non-delirium state.
Brain regions exhibiting a significant difference in functional connectivity with the subcortical seed regions between conditions.
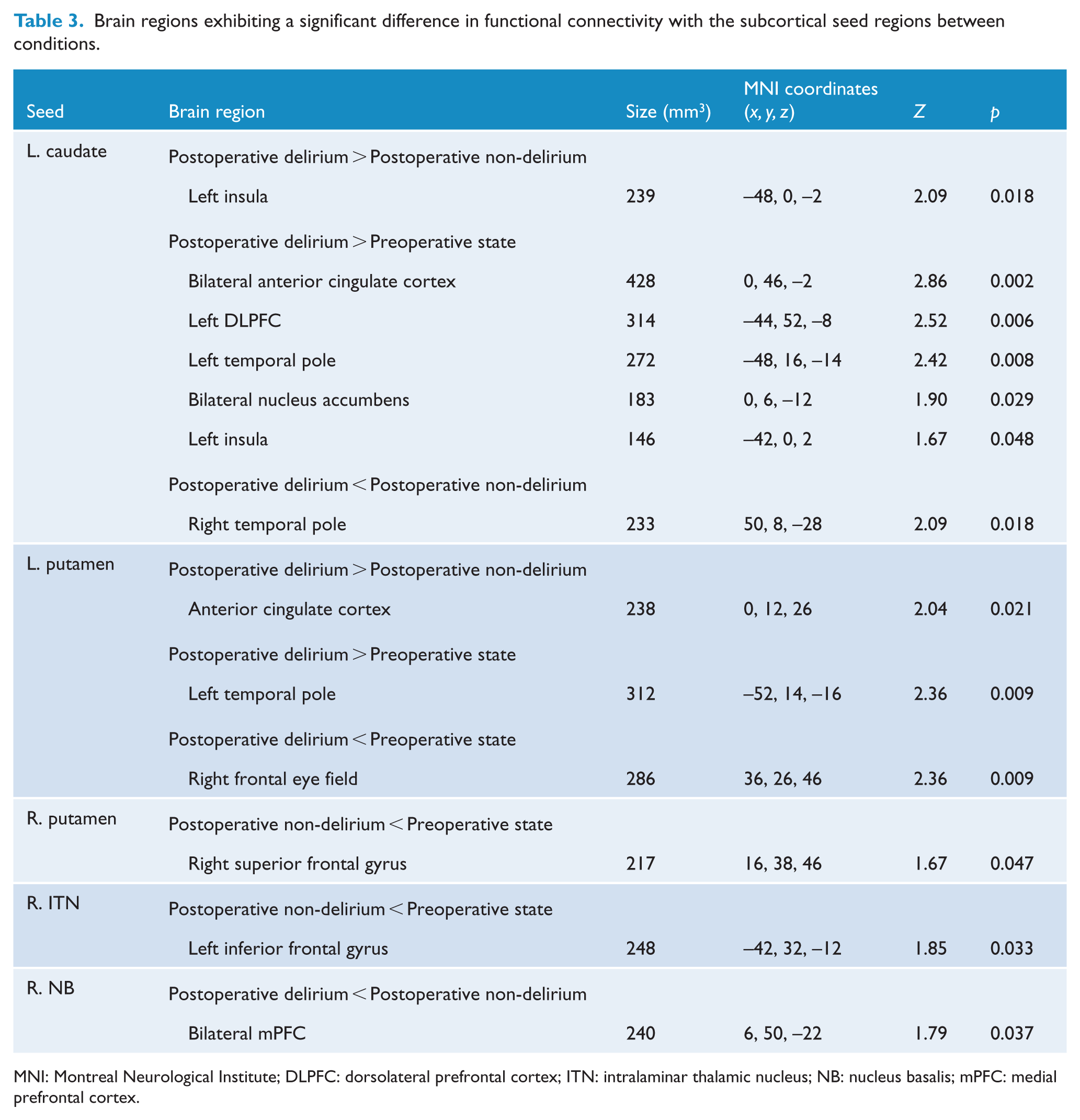
MNI: Montreal Neurological Institute; DLPFC: dorsolateral prefrontal cortex; ITN: intralaminar thalamic nucleus; NB: nucleus basalis; mPFC: medial prefrontal cortex.
In the ANOVA of subcortex-based functional connectivity, no group effect or group × time interaction effects were found, but a time effect was identified in two connections. In connectivity with the ventral tegmental area and right mesencephalic tegmentum, the time effect was found in the lateral occipital cortex (
Functional connectivity among the subcortical regions
Functional connectivity among the 11 subcortical regions was investigated using ROI-to-ROI analyses. As shown in Figure 3, these regions were well connected with one another before surgery, and those connections were maintained in the postoperative non-delirium state. However, many of those connections were severed in the postoperative delirium state. In detail, mutual connections that were significant in the preoperative and postoperative non-delirium states but not in the postoperative delirium state were found in five subcortical combinations: (1) the caudate and intralaminar thalamic nucleus, (2) the caudate and nucleus basalis, (3) the caudate and mesencephalic tegmentum, (4) the putamen and nucleus basalis and (5) the nucleus basalis and mesencephalic tegmentum. In particular, the connections between the lower regions (the nucleus basalis, ventral tegmental area and mesencephalic tegmentum) and upper regions (the caudate, putamen and intralaminar thalamic nucleus) were not noticeable during delirium. Detailed statistical values of these ROI-to-ROI connections are provided in Supplemental Table S1.
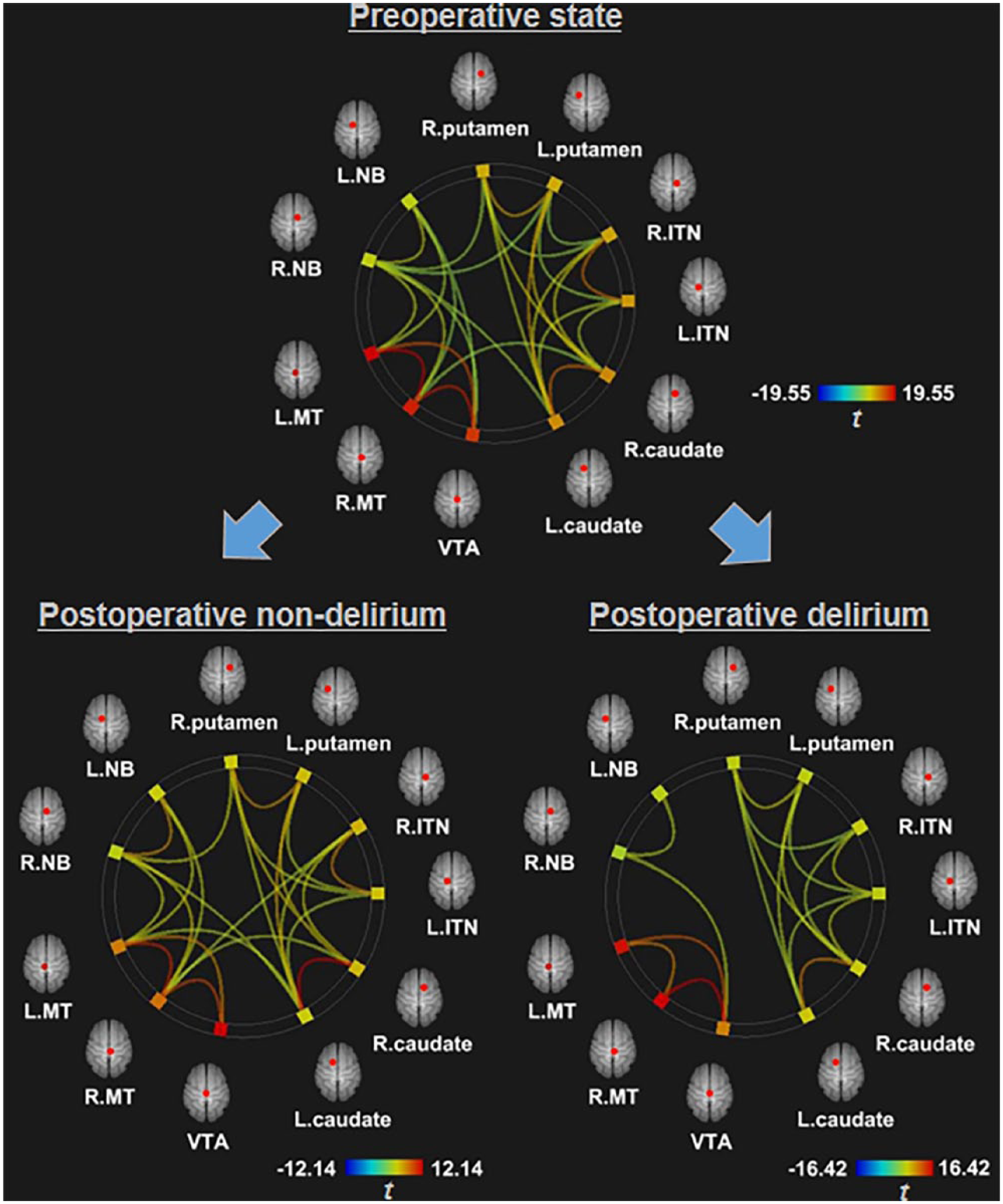
Resting-state functional connectivity among subcortical regions, showing that the connections were intact in the preoperative and postoperative non-delirium states, but disrupted in the postoperative state.
Discussion
In this study, we obtained serial rsfMRI scans to elucidate differences in the resting-state brain network before and during an episode of delirium. Compared with our previous study (Choi et al., 2012), in which we conducted comparisons between delirium and the post-recovery state, the present study revealed some replicative findings. First, increased connectivity between the PCC and DLPFC during delirium was equally evident in both studies, suggesting that delirium is characterized by diminished anticorrelation between the DMN and task-positive regions. Reduced connectivity between subcortical regions was another replicative finding, suggesting that stable connectivity related to cholinergic/dopaminergic neurotransmission and the proper function of the ascending reticular activating system are important in maintaining a healthy consciousness. It is worth mentioning that these replicative findings could be related to the general connectivity features of delirium regardless of homogeneity or heterogeneity in the sample or comparison with the preoperative or post-recovery state.
In this study, we also report novel findings about PCC-based functional connectivity: the bilateral inferior parietal lobules showed increased connectivity during delirium. In our previous study, we found a strong link between the PCC and precuneus during an episode of delirium (Choi et al., 2012). Given that both the precuneus and inferior parietal lobule are core nodes of the posterior DMN (Utevsky et al., 2014) and the inferior parietal lobule is farther from the PCC than the precuneus, the present result could support the idea of enhanced connectivity within a wider range of the posterior DMN during delirium. Because a course of delirium is reversible, this increased connectivity within the posterior DMN seems to be a transient compensatory mechanism for the disrupted anticorrelation between the task-negative and task-positive regions. Compensatory hyperconnectivity responses after the disruption of a network have been similarly observed in various brain disorders, including Parkinson’s disease (Simioni et al., 2015) and traumatic brain injury (Hillary et al., 2014). In particular, these responses usually occur in the early phase of each disorder, suggesting that chronic overloading of neural hyperconnectivity caused by excessive use of brain resources could lead to increased local amyloid beta deposition and resultant synaptic loss (Buckner et al., 2005). In light of this hyperconnectivity in the early phase of degenerative disorders, it is clear that the transient compensatory hyperconnectivity in the acute delirium state is an inevitable operation of the brain. The result from this overuse could be linked to previous evidence of incomplete recovery and long-term cognitive decline after an episode of delirium (Saczynski et al., 2012).
Another possible meaning of hyperconnectivity within the posterior DMN is that an excessive increase in interconnections could paradoxically lead to excessive error information delivery, which causes delirious symptoms. It has been documented in schizophrenia that hyperconnectivity of the DMN can blur the boundary between internal thought and external perception and misdirect attentional resources (Whitfield-Gabrieli et al., 2009). Hyperconnectivity of the posterior DMN has also been found in subjects at high risk for depression and interpreted as the reflection of an excessive focus on one’s emotional state instead of the external environment (Posner et al., 2016). These aspects of schizophrenia and depression could be the same psychopathology that occurs in delirium. Therefore, hyperconnectivity of the posterior DMN could be the channel through which error information is transmitted during the compensatory process.
By adding the mPFC as a seed, we further found an increased connection between the mPFC and DLPFC during delirium, suggesting that the anterior DMN also shows diminished anticorrelation with task-positive regions, as in the posterior DMN. Aberrant coupling of the two regions and the resultant loss of reciprocity have been implicated in aging-related cognitive decline (Keller et al., 2015) and psychopathology, including psychosis and impulsivity (Posner et al., 2016; Wotruba et al., 2014). Given that the DLPFC is part of the task-positive region or the central executive network (CEN) (Chen et al., 2013), the loss of reciprocity between the anterior DMN and CEN during delirium could be responsible for various delirious symptoms, including disorganized and impulsive behavior.
In addition, seeding the mPFC allowed us to observe increased connectivity with the frontopolar cortex and decreased connectivity with the superior frontal gyrus in the postoperative delirium state compared with the preoperative state. The frontopolar cortex has high connectivity with the DMN (Liu et al., 2013), and its medial part constitutes the anterior DMN (Davey et al., 2016). Because this region has a gateway function, playing a role in the attendance to one among various competing sensory representations (Burgess et al., 2007; Davey et al., 2016), hyperconnectivity with the mPFC during delirium may suggest abnormally strengthening processes for filtering information of internal sources. The superior frontal gyrus is also a component of the DMN (Andrews-Hanna et al., 2010), and hypoconnectivity with the mPFC could reflect a defective function of the region, which represents reflective thinking about oneself in relation to others (Moran et al., 2013). Although significance was borderline, the connectivity strength between the mPFC and this region was negatively correlated with delirium severity, suggesting the importance of the anterior DMN in the pathology of delirium. These anterior DMN structures are the fundamental regions for self-related processing (Qin and Northoff, 2011). The coexistence of an increase and decrease in one region could indicate an imbalance of functional circuits within that region. Unlike the simple compensatory changes within the posterior DMN, the anterior DMN exhibits a complex imbalance associated with aberrant self-related processing during delirium.
The results in subcortical seed-based functional connectivity showed increased connectivity of the caudate with the ACC and insula and increased connectivity of the putamen with the ACC. The ACC and insula constitute the salience network (SN) (Manoliu et al., 2014), and thus, our result shows that the SN might receive increased signals from the striatum. The SN is responsible for switching between the DMN and CEN and modulates the interactions between the two networks (Goulden et al., 2014; Sridharan et al., 2008). The DMN is more oriented toward internal mental events, whereas the CEN is more related to events from external sources, including planning, decision making and working memory (Menon, 2011). A failure in the modulatory function of the SN toward the DMN and CEN has been reported in various psychopathologies, including hallucination and anxiety (Beucke et al., 2012; Manoliu et al., 2014). Functional disturbances in frontostriatal loop circuits, including the SN, are important in abnormal self-regulation (Marsh et al., 2009; Peters et al., 2016). Hyperconnectivity between the striatum and ACC has been linked to the anticipation of a potential, negative emotional event (Beucke et al., 2012), which is similar to the anxiety or paranoid symptoms of delirious patients. Normalization of connectivity in the loop circuits of the SN was associated with recovery from pathological mental states (Sale et al., 2015; Veit et al., 2012), as in the reversibility of delirium. Taken together, our findings suggest that immoderate striatal information transmission to the SN may lead to various delirious symptoms because of a dysfunction in the modulatory role of the SN between the DMN and CEN.
Our connectivity analysis among the subcortical regions found that they were well connected with each other in the preoperative and postoperative non-delirium states, but many subcortical connections were severed in the postoperative delirium state. These findings are in line with our previous study, which showed a reduction in connectivity among the subcortical regions during delirium (Choi et al., 2012). Specifically, both studies found disconnections between the caudate and nucleus basalis, the caudate and mesencephalic tegmentum, the intralaminar thalamic nucleus and nucleus basalis and the intralaminar thalamic nucleus and ventral tegmental area during delirium. However, there is a notable difference between the two studies. Connections between the subcortical regions of the upper level (the caudate, putamen and intralaminar thalamic nucleus) and those of the lower level (the nucleus basalis, ventral tegmental area and mesencephalic tegmentum) during delirium were found in several combinations in the previous study, but not in the present study. In particular, this study exhibited a complete disconnection between the cholinergic/dopaminergic origins and the striatum and between the mesencephalic tegmentum and intralaminar thalamic nucleus. Given that altered neurotransmission is important in the development of delirium (Sanders, 2013) and that rsfMRI can read neurotransmission changes (Shah et al., 2015), the functional disconnection between the origin of each neurotransmitter (nucleus basalis for acetylcholine and ventral tegmental area for dopamine) and the thalamic/striatal regions could reflect the pathogenic process of delirium. In addition, the mesencephalic tegmentum and intralaminar thalamic nucleus, which are both part of the ascending reticular activating system and play a role in the regulation of arousal (Kinomura et al., 1996), were found to be disconnected during delirium, suggesting that disruption of the arousal mechanism on the subcortical level could be important in the development of delirium.
Based on our results, we propose a subcortical origin model for delirium, as summarized in Figure 4. We have provided several lines of evidence supporting the model in the present study. The starting point of the problem is the disconnection between the lower subcortical regions, including the neurotransmitter origins, and the striatum/thalamus in the upper level, which can be created by various medical/surgical causes. Initial subcortical disconnection could lead to aberrant hyperconnectivity between the striatum and SN, which can cause problems in the regulatory function of the SN. Dysfunction of the SN could in turn induce a loss of reciprocity between the DMN and CEN, and lead to a disturbance in task-positive responses in the CEN and a compensatory mechanism in the DMN. In particular, hyperconnectivity within the posterior DMN and unbalanced connectivity within the anterior DMN could deliver error information and cause confusion. The main argument of this model is that the serial changes initiated from the lower subcortical origins may induce the entire disorganization of brain connectivity associated with delirium.
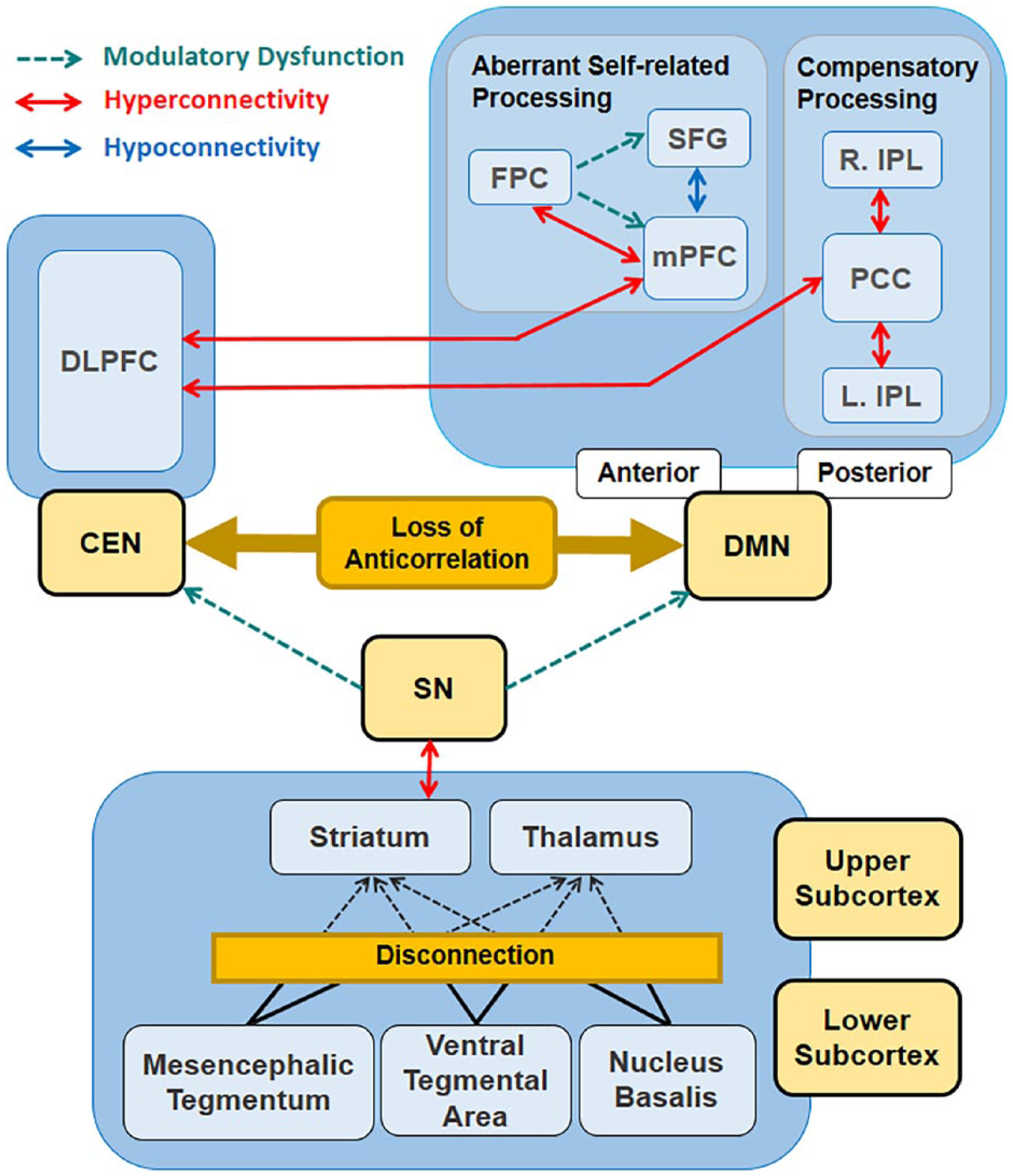
Subcortical origin model for delirium.
The significance of our study is our longitudinal investigation of functional connectivity through repeated rsfMRI scanning before and after surgery. Although we assumed that all participants’ brain states were normal before surgery, subjects’ brain states might have been impaired before delirium occurred. In fact, in a previous paper, we found that the amplitude of low-frequency fluctuation in the DLPFC and striatal gray matter density were predisposing factors for postoperative delirium (Kyeong et al., 2018). Other studies have also suggested factors contributing to postoperative delirium, such as cerebral atrophy (Brown et al., 2015). In fact, our sample showed a subtle group difference in the preoperative Mini-Mental State Examination (MMSE) score (postoperative delirium, 16.79 ± 7.14; postoperative non-delirium, 21.17 ± 6.45), although it was not statistically significant (
The current study has some other limitations. Due to the delirious state, motion artifacts could have a lot of influence on our data. We tried to minimize this effect by letting a researcher stand next to the patient during scanning and using an artifact correction tool (the aCompCor method) in the preprocessing step. Another limitation is that our postoperative sample size became relatively small due to the high drop-out rate. This might be why we found no significant correlations between the regional connectivity strengths and delirium severity. We included only patients with femoral neck fractures to control for the possible confounding effects of a heterogeneous sample. Although this homogeneity is a merit of this study, it could also be a disadvantage because it makes our results difficult to generalize to postoperative delirium patients after other types of surgery or delirium are caused by other medical problems.
In summary, patients with femoral neck fractures underwent repeated rsfMRI scans before and after surgery, and analyses were applied to elucidate the changes in functional connectivity during postoperative delirium. The posterior DMN had excessive internal connections, whereas the anterior DMN had a complex imbalance in internal connectivity, and anticorrelation between the DMN and task-positive regions was diminished. The striatum and SN were excessively connected. The lower subcortical neurotransmitter regions and upper striatum/thalamus were completely disconnected. Taking all these changes into account, we propose a subcortical origin model of delirium that provides new insights into how the overall connectivity changes in the whole brain begin and propagate during an event of postoperative delirium. Our proposed model highlights the aberrant neurotransmission-related connectivity and disrupted modulatory control between critical brain networks as important mechanisms of delirium.
Supplemental Material
Supplemental__table_1 – Supplemental material for Cortical and subcortical changes in resting-state functional connectivity before and during an episode of postoperative delirium
Supplemental material, Supplemental__table_1 for Cortical and subcortical changes in resting-state functional connectivity before and during an episode of postoperative delirium by Jooyoung Oh, Jung Eun Shin, Kyu Hyun Yang, Sunghyon Kyeong, Woo Suk Lee, Tae-Sub Chung and Jae-Jin Kim in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI16C0132).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
