Abstract
Keywords
Introduction
The application of morphological and fMRI techniques has contributed notably to improve our understanding of the pathophysiology of different headache conditions (1). In patients with cluster headache (CH), functional imaging studies have suggested a central role of the hypothalamus in the genesis of the attacks by showing the activation of its posterior part during spontaneous and provoked attacks of the disease (2–5). Hypothalamic involvement has been supported further by the demonstration, using voxel-based morphometry, of its increased volume in CH patients (2), and by a beneficial effect of deep brain stimulation of this area (6). Other brain regions, known to be part of the pain processing brain network, such as the cingulum, the insula, the frontal cortex, and the thalamus, are also thought to be involved in the pathogenesis of CH (4); however, as is the case for the activation of the hypothalamus, recruitment of pain-related regions in CH patients has been detected only during headache attacks (4,5), since, outside the attack, only a generic cranial neurovascular activation has been observed (4). The recent demonstration of abnormalities of mid-latency and long-latency auditory-evoked responses in CH patients outside the acute phase of the attack pointed, however, towards a more wide spread abnormality of high-order neuronal processing in these patients (7). As a consequence, defining the pattern and extent of central nervous system (CNS) dysregulation in CH patients outside the attack phase might shed light on disease pathophysiology and, ultimately, guide future therapeutic strategies (i.e. local vs systemic treatment, surgical vs medical therapy).
The analysis of low-frequency (<0.1 Hz) fluctuations seen on fMRI scans at rest (i.e. in the absence of external stimulations) has demonstrated the presence of high temporal coherence between spatially distinct, functionally-related brain regions, resembling specific neuro-anatomical networks, such as the motor, the visual, and the dorsal and ventral attention systems, which characterise the resting-state networks (RSNs) of the human brain. Low-frequency resting-state (RS) fluctuations consist in synchronous variations of the BOLD signal, whose entity can be measured as a percentage signal change compared to the BOLD mean signal intensity over time (8–10). These RS fluctuations are thought to reflect an intrinsic property of brain functional organisation that serves to stabilise brain ensembles, consolidate the past and be prepared for the future (11). The importance of assessing intrinsic brain activity has been shown in normal ageing (12), psychiatric affections (13–15) and Alzheimer’s disease (16). More recently, RSN abnormalities have also been demonstrated in patients with chronic pain (17).
Against this background, we wished to assess whether brain RSN abnormalities are present in patients with episodic CH, outside the attack phase, with the ultimate aim to define if functional CNS abnormalities in this disease are limited to the hypothalamus and pain-related matrix or, conversely, tend to be more wide spread, as suggested by preliminary neurophysiological studies (7,18,19).
Patients and methods
The study was conducted with institutional review board approval. Written informed consent was obtained from each participant.
Patients
We studied 13 right-handed patients with episodic CH (20) (2 women, 11 men; mean age, 43.0 years; range, 29–64 years; mean disease duration, 17 years; range, 1–39 years; 8 with right-sided headache, 5 with left-sided headache). Patients with hypertension, hypercholesteraemia, diabetes mellitus, vascular/heart diseases, and other major systemic and neurological or psychiatric conditions were excluded. At the time magnetic resonance imaging (MRI) was performed, five patients had been taking a prophylactic treatment for CH for at least one year (verapamil in 3 patients and topiramate in 2 patients). Patients who had a disease re-exacerbation within one month from MRI acquisition were excluded. Fifteen right-handed (21), healthy volunteers, with no familial history of migraine, no history of neurological dysfunction (including migraine), and a normal neurological examination, served as controls (4 women and 11 men; mean age, 45.0 years; range, 25–63 years).
Image acquisition
On a 3.0 Tesla Intera scanner (Philips Medical Systems), RS fMRI scans were acquired using a T2*-weighted single-shot echo planar imaging (EPI) sequence (repetition time [TR], 3000 ms; echo time [TE], 35 ms; flip angle [FA], 90°; field of view [FOV], 240 mm2; matrix, 128 × 128; slice thickness, 4 mm). Two-hundred sets of 30 contiguous axial slices, parallel to the AC–PC plane and covering the entire brain, were acquired for each subject. The total acquisition time was 10 min. During scanning, subjects were instructed to remain motionless, to close their eyes and not to think anything in particular. After scanning, all subjects reported they had not fallen asleep during the RS fMRI scanning.
An axial T2-weighted turbo-spin echo and an axial fluid attenuated inversion recovery (FLAIR) sequence of the brain were also acquired.
Image analysis
T2-weighted and FLAIR scans were analysed for the presence of lesions. Lesion volumes (LVs) were measured using a local thresholding segmentation technique (Jim 4.0, Xinapse System, Leicester, UK).
RS fMRI data were first pre-processed using SPM2. Functional images were realigned to the first image to correct for subject motion, and spatially normalised into the standard SPM2 space, with a data subsampling to 3 × 3 × 4 mm of resolution (22). Then, fMRI data were smoothed using a 3D Gaussian kernel of 6 mm. Finally, fMRI time series from each subject were linear detrended and band-pass filtered between 0.01–0.08 Hz to remove low-frequency drifts and physiological high-frequency noise.
Resting-state independent component analysis
Independent component analysis (ICA) was performed using the GIFT (Group ICA of FMRI Toolbox) software (22). This toolbox uses a group approach involving an initial ICA estimation on the concatenated data from all subjects, followed by the computation of subject-specific components and time courses. First, individual subjects’ data were reduced to a lower dimensionality by using the principal component analysis to decrease computational workload involved with ICA estimation. Then, group spatial ICA was conducted using the infomax algorithm (23). At this stage, data were decomposed into 40 components using the GIFT software. Finally, spatial functional maps and their associated time courses were reconstructed for each subject (22).
A systematic process was applied to inspect and select the components of interest from the 40 estimated components. The association of each component spatial map with
The individual subject’s data of the ICs of interest detected at this stage were entered into SPM2 random-effect analysis, to assess the spatial extent of within- and between-group activations by using a one-sample
Resting-state seed correlation analysis
To test RS connectivity abnormalities in the hypothalamus and the pain-related matrix, an additional analysis of functional connectivity (FC) was performed, using a seed correlation approach, which consisted in the selection of a region of interest as the seed region for correlation, and in the performance of a correlation analysis between this seed region and all remaining brain regions. For the assessment of hypothalamic-related RS connectivity abnormalities, time series were extracted for each subject from a spherical volume (5-mm radius) within an
The correlation between RS abnormalities and disease duration was assessed using SPM2 and a linear regression analysis (
Results
Non-specific brain T2-hyperintense lesions were detected in seven CH patients and six controls. Mean T2 lesion volume was 0.7 ml (SD, 0.06 ml) in CH patients and 0.6 ml (SD, 0.07 ml) in controls (not significant).
In controls and CH patients, the analysis of RS data showed 11 networks with potential functional relevance: RSNs 1 and 2 included primary and secondary visual cortical areas; RSNs 3 and 4 included sensorimotor areas; RSNs 5, 6 and 7 included bilateral frontoparietal-temporal areas; RSN 8 included frontoparietal areas lateralised to the left hemisphere; RSN 9 included frontoparietal areas lateralised to the right hemisphere; RSN 10 included bilateral parietal areas and the posterior cingulum (default mode network [DMN]); and RSN 11 included primary and secondary auditory areas (Figure 1).
Potentially functionally relevant resting state networks (RSNs) in healthy volunteers and patients with cluster headache: RSN 1 (A) and RSN 2 (B) included primary and secondary visual cortical areas; RSN 3 (C) and RSN 4 (D) included sensorimotor areas; RSN 5 (E), RSN 6 (F) and RSN 7 (G) included bilateral frontoparietal-temporal areas; RSN 8 (H) included frontoparietal areas lateralised to the left hemisphere; RSN 9 (I) included frontoparietal areas lateralised to the right hemisphere; RSN 10 (J) included bilateral parietal areas and the posterior cingulum (default mode network); and RSN 11 (K) included primary and secondary auditory areas. See text for further details.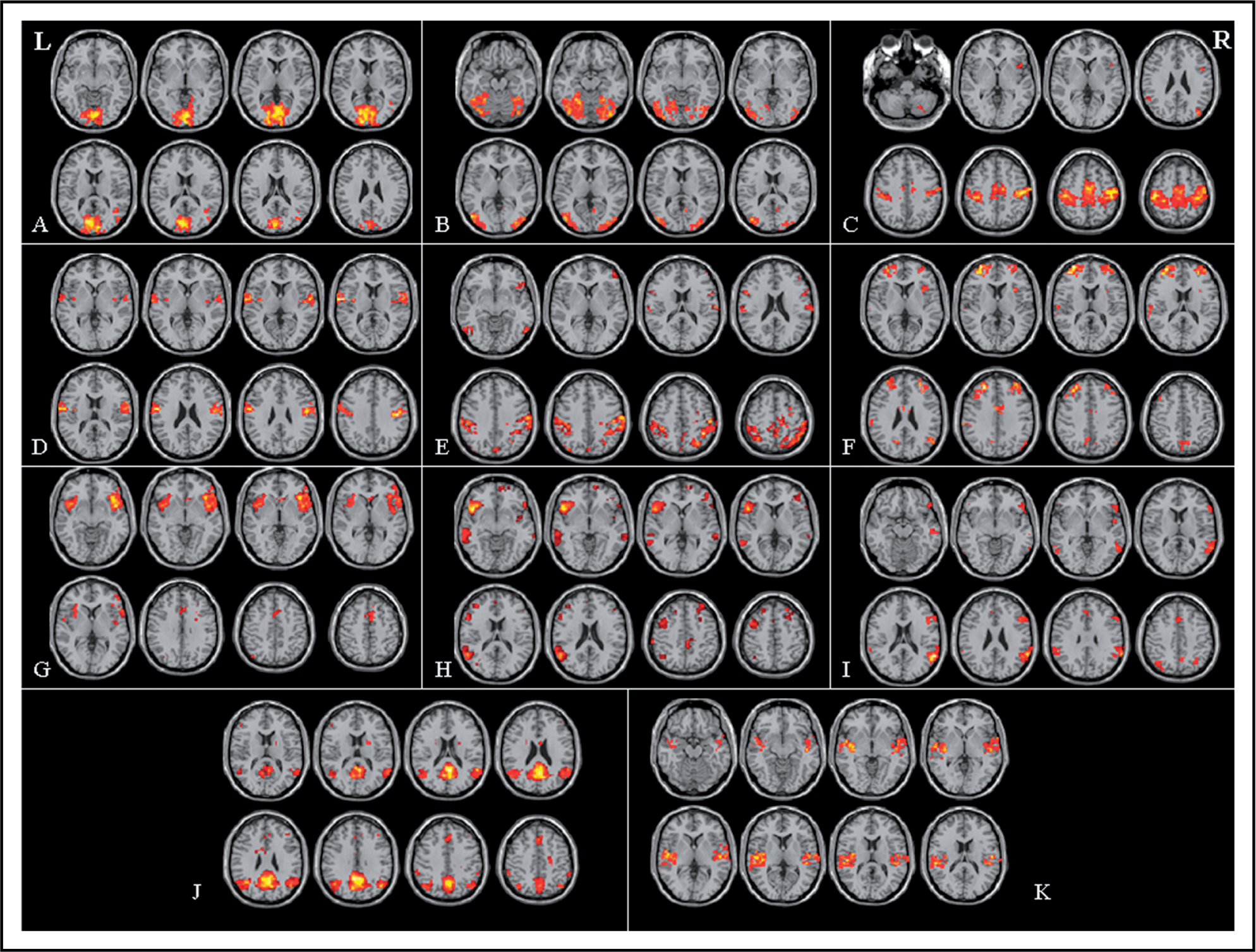
The spatial distribution of the detected RSNs did not differ between CH patients and controls. Significant between-group differences ( Top row: spatial pattern (A) and average percentage signal change of resting state fluctuations (B) detected in the sensorimotor network of healthy subjects (black) and CH patients (grey). Bottom row: Spatial pattern (C) and average percentage signal change of resting state fluctuations (D) detected in the primary visual network of healthy subjects (black) and CH patients (grey).
Average percentage signal change (and SD) of resting state fluctuations in the clusters showing a significant difference between healthy controls and patients with cluster headache

SPM, statistical parametric mapping; CH, cluster headache; L, left; R, right; B, bilateral; SMC, primary sensorimotor cortex; SMA, supplementary motor area; ACC, anterior cingulate cortex.
Disease duration was significantly correlated with the reduction of the average RS fluctuations in the left primary SMC of the RSN 3 ( Scatter plots of the correlation between disease duration and average percentage signal change in the left primary SMC of the sensorimotor network (
The seed correlation analysis showed that, in comparison with controls ( Statistical parametric map showing brain regions having significantly increased functional connectivity (FC) (
CH patients also had increased FC between RS fluctuations of the right thalamus and activity of the primary SMC, bilaterally, as well as between RS fluctuations of the left thalamus and activity of the bilateral primary SMC, SMA and ACC (Figure 4). Using an uncorrected
Discussion
In this study, we analysed RS fluctuations in CH patients to gain additional insight into the functional brain abnormalities associated with this condition. To achieve a comprehensive assessment of all the possibly involved RSNs, we studied only patients in the headache-free state, since the evaluation of patients during an attack is likely to lead to the preferential or exclusive detection of abnormalities in the pain-related network.
In line with previous studies of RS fluctuations in healthy individuals (10,12), we were able to identify 11 RSNs, which were consistently present in both controls and CH patients. These RSNs included the motor system, several sensory systems, as well as attention- and memory-related networks. The comparison between CH patients and controls demonstrated that percentage signal change fluctuations were significantly reduced in two of the 11 detected RSNs, i.e. the sensorimotor and the visual networks.
Previous neuro-imaging studies have supported the notion that frequent nociceptive input related to chronic painful conditions may induce impaired supraspinal modulation in the CNS (27,28), and have contributed to the identification of a distributed brain network recruited in the processing of nociceptive stimuli. Such a network includes sensory-discriminatory areas (e.g. primary and secondary somatosensory area, thalamus, posterior part of the insula) and affective-cognitive areas (e.g. anterior part of the insula, cingulum, prefrontal cortex). The majority of these areas are relay stations of the human motor network, whose activation is known to be related to that of all the principal brain sensory systems.
In the RS data assessment, we identified the IC resembling the motor network (as well as other sensory networks, such as the visual and the auditory), whereas we were unable to identify the previously described pain-related matrix using the ICA approach (28). Conversely, using a seed correlation approach for the analysis of RS abnormalities, starting from the hypothalamus and the thalamus, allowed us to identify abnormalities within such a network. This discrepancy was not unexpected. Indeed, ICA and seed-voxel correlation analysis of FC are two complementary ways for estimating networks of functionally related areas. ICA is a multivariate approach that tries to identify hidden factors from a set of fMRI data, by grouping brain activity into regions that share the same response pattern. The power of the ICA approach is that this grouping of brain regions by similarity of their time courses is done on all time series at once, and it is not biased by the choice of one seed region as a model for connectivity. Moreover, such an approach is able to find multiple networks of functionally connected brain regions in the same dataset. On the other hand, such a decomposition into independent components might result in difficulties for small brain areas (such as the hypothalamus and thalamus), whose echo-planar imaging time series can be seriously affected by noise; as a consequence, the attribution of these areas to the correct brain network can be hampered. In this case, a seed correlation approach might be useful to detect the network of brain regions connected with the areas of interest. This approach uses the average time series of a selected area as a reference time series, and correlates all remaining brain areas with this template. Since ICA is a multivariate data-driven technique and FC is a template-based univariate approach, these methods can give different and complementary results in terms of maps of correlated activity.
In the sensorimotor network, we found reduced RS fluctuations in the primary SMC, the SMA, and the ACC, bilaterally. The primary sensory cortex receives multiple ascending pathways transmitting painful sensation and it is interconnected with multiple cortical and subcortical structures for the processing of such stimuli, including the primary motor cortex, the SMA and the ACC (29,30). All these regions also contribute to motor response to unpleasant painful stimulation, which might result in an enhanced interaction in case of a chronic input. In this context, the primary motor cortex has been supposed to have a role in the affective-motivational system possibly involved in eliciting escape responses (30). Indeed, a seminal single photon emission tomography study (31) detected a reduced cerebral blood flow in the primary SMC in CH patients in the headache-free state in comparison with healthy controls, using an experimental model of tonic aching pain. The ACC exerts a descending modulation to gate pain (32) and its activation has been related to analgesia (33). This region is also thought to be involved in attention, conflict monitoring, and goal-directed behaviours, thus explaining its frequent recruitment in fMRI studies of motor and cognitive tasks (34,35). In addition, the ACC is supposed to play a complex role in the integration of attentional, sensory and affective components of painful stimulation (29,30). The notion that ACC activity might be impaired in CH patients is in line with the results of a few neurophysiological investigations (18,19), which have demonstrated increased event-related potential (P3) latencies in these patients compared to healthy controls. This suggests the presence in these patients of a distributed dysfunction of cortical information processing, particularly of visual modalities, in these patients. Remarkably, our analysis revealed abnormalities of the visual network restricted to V1. Although visual symptoms under the form of aura or aura-like warning have been reported in up to 14% of patients with CH (36,37), none of our patients had such a complaint. However, retro-orbital pain and photophobia are typically present in CH patients (36) and, as a consequence, the central processing of these sensations is likely to influence visual system rewiring. The demonstration of decreased P100 amplitudes of visual evoked potentials in these patients when stimulating the ‘painful’ side supports this hypothesis (38).
Once it had been established that RSN fluctuation abnormalities do occur in CH patients, we wished to gain some insight into the nature of such abnormalities. Although our results have to be considered with caution, given the relatively small number of subjects studied, the correlation we found between disease duration and RS fluctuation abnormalities in the primary SMC and V1 suggests that the observed abnormalities might be the consequence of a persistent CNS dysfunction. It is worth noting that, since the analysis of correlation was done on the whole brain, disease duration correlated with reduction of RS fluctuations in some of the regions that were significantly different between patients and controls, but not in others. fMRI studies in healthy individuals have demonstrated consistently incremental experience-dependent changes and shifts in activations, following learning (39,40). In this context, it is, therefore, tempting to speculate a rewiring of those cortical networks which usually cooperate with the pain-related matrix in processing the painful sensations in CH patients. This hypothesis is also supported by the results of our FC analysis, which showed, in CH patients, an increased interaction between RS fluctuations of the thalamus and those of areas which are part of the sensorimotor network, as well as between RS fluctuation of the hypothalamus and those of areas which are part of the pain-related matrix and the visual system.
We could not detect any difference in low-frequency fluctuations of the DMN, a set of regions known to be deactivated during task performance (10,12,41) and to be active during rest. DMN abnormalities have been shown to occur in patients with autism (15), Alzheimer’s disease (16), depression (14), and schizophrenia (42). More recently, a reduced deactivation within regions of the DMN has been shown in patients with chronic back pain, supporting the notion that a dysfunction of this network might underlie the cognitive and behavioural abnormalities observed in these patients (17). Differences in the experimental set up (only rest condition in our study vs active and rest conditions in Baliki et al.) (17) method of analysis (no
Conclusions
These findings show that CH patients have a diffuse dysfunction of brain functional connectivity, which extends beyond the antinoceptive system and involves preferentially brain networks, which are likely to interact with the pain-related matrix.
