Abstract
Objectives:
Alterations in thalamocortical anatomical connectivity, specifically the connection between the orbitofrontal cortex and thalamus, have been frequently reported in schizophrenia and are suggested to contribute to the pathophysiology of schizophrenia. The connectivity of the thalamocortical white matter in unaffected relatives of schizophrenia patients was compared to that of healthy controls.
Methods:
The unaffected relative group was defined as asymptomatic family members who had at least one first-degree relative with schizophrenia and one or more other affected first- to third-degree relatives. A total of 35 unaffected relatives and 34 healthy controls underwent diffusion-weighted and T1-weighted magnetic resonance imaging to examine the white matter connectivity between the thalamus and orbitofrontal cortex using probabilistic tractography.
Results:
After controlling for age and sex, the unaffected relatives exhibited significantly reduced fractional anisotropy values for the left thalamo-orbitofrontal tract compared to that of healthy controls, F(1, 65) = 6.93, p = 0.011, effect size partial η2 = 0.10. However, there was no association between the Genetic Liability Score and fractional anisotropy in the left thalamo-orbitofrontal tracts.
Conclusion:
Our findings in the unaffected relatives of schizophrenia patients, which are in line with the alterations reported in schizophrenia, first-episode psychosis and clinical high risk for psychosis, highlight a possible genetic contribution to the proposed biomarker of altered thalamocortical connectivity.
Keywords
Introduction
The current diagnostic system for schizophrenia, based on clinical symptoms, is a huge hindrance to understanding its pathophysiology due to the heterogeneity of the disease. This issue has led to the pursuit of more entity-specific biological markers. Among potential biomarkers, anomalies in the thalamus have been fairly consistently identified, such as a reduced number of neurons (Byne et al., 2002; Pakkenberg, 1990), reduced volume (Haijma et al., 2012; Konick and Friedman, 2001) and altered glutamate receptors (Ibrahim et al., 2000) although there have also been some inconsistencies reported (for review of post-mortem findings, see Dorph-Petersen and Lewis [2017]). Also, our group recently reported abnormal microstructure in higher order nuclei of first-episode psychosis (FEP) patients using non-invasive diffusion kurtosis imaging (Cho et al., 2018). Andreasen et al. (1998) have proposed the cognitive dysmetria model which suggested an involvement of the cortico-thalamic-cerebellar circuitry in the pathophysiology. As the thalamus has extensive connections with the cortex, relaying sensory signals and modulating cortico-cortical information transfer, these findings and proposals seem plausible.
More recently, the alteration of the thalamocortical connection in schizophrenia has been highlighted. Marenco et al. (2011) showed a reduction in anatomical connections between the thalamus and prefrontal cortex (PFC), whereas Kubota et al. (2013) specifically highlighted the alteration in the connection between the thalamus and orbitofrontal cortex (OFC). We have previously found the same pattern of changes in FEP and, to a lesser extent, in subjects at clinical high risk (CHR) for psychosis (Cho et al., 2015), which suggests that altered connectivity is a putative biomarker for schizophrenia. Giraldo-Chica et al. (2018) also reported similar finding of reduced thalamo-prefrontal connectivity in schizophrenia. These reports are also consistent with the findings from thalamocortical functional connectivity investigations (Anticevic et al., 2014, 2015; Ferri et al., 2018; Woodward and Heckers, 2016; Woodward et al., 2012). (A schematic summary of the existing findings is shown in Figure 1.)
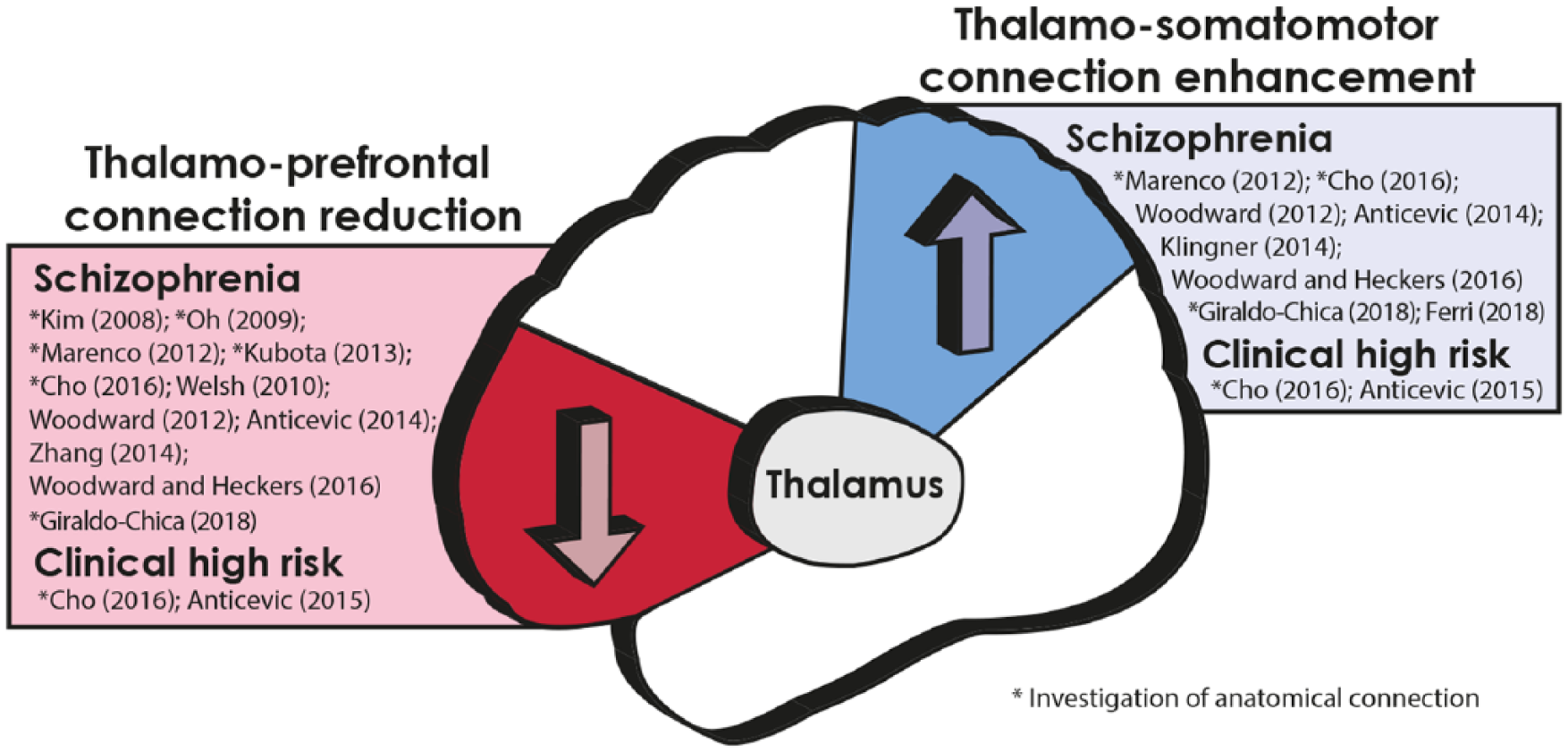
Summary of current reports of thalamocortical network anomalies in schizophrenia and high-risk groups. The schematic diagram represents the reduced thalamo-prefrontal connection and enhanced thalamo-somatomotor/parietal connection. Only direct investigations of the thalamocortical system are included in the reference list (Anticevic et al., 2014, 2015; Ferri et al., 2018; Giraldo-Chica et al., 2018; Kim et al., 2008; Klingner et al., 2014; Oh et al., 2009; Welsh et al., 2010; Woodward and Heckers, 2016; Zhang et al., 2014).
Although there is evidence of enhanced thalamo-somatomotor/parietal connectivity, the most frequently affected thalamocortical network in schizophrenia is the thalamo-PFC network. Specifically, the OFC within PFC is involved in sensory integration, feedback processing, reward and decision-making (Cavada, 2000), which indicates that this region plays a key role in goal-directed behavioural planning. These functions are fundamentally disrupted in schizophrenia (Gold et al., 2008; Javitt, 2009) and may lead to disturbed perception and impaired affective processing, which manifest as positive and negative symptoms, respectively (Millan et al., 2014). Consistent with this, reports of OFC alterations in schizophrenia have been accumulating (Kringelbach, 2005). Altogether, these highlight thalamo-OFC disconnection as a biomarker and emphasize the need for further investigation in this system.
Biomarkers are objective indications of a disease state which can be measured accurately and reproducibly (Strimbu and Tavel, 2010). Altered thalamo-cortical connectivity is a putative biomarker of genetic liability for schizophrenia. This hypothesis of altered thalamocortical connectivity as a biomarker could be strengthened if a genetic aspect of this alteration could be identified. Unaffected relatives of patients with schizophrenia are at genetic high risk (GHR) of the disorder due to the high proportion of shared genes. Various schizophrenia endophenotype candidates have been reported in twin and relative studies of brain structure and function (Hulshoff Pol et al., 2004; Seidman et al., 1999); these potential endophenotypes include changes in the PFC and the thalamus. In addition to focal findings, there are reports of functional and structural connectivity alterations (Bohlken et al., 2016; Munoz Maniega et al., 2008; Zalesky et al., 2015) in relatives of schizophrenia patients, which also highlight the genetic effect on the brain network. However, to our knowledge, there has not been a direct investigation of the anatomical thalamocortical connection in unaffected relatives of people with schizophrenia.
In this study, we sought to determine whether the change consistently reported in the thalamo-OFC anatomical connectivity is related to genetic risk for schizophrenia; for this purpose, we investigated anatomical thalamocortical connectivity using diffusion tensor tractography in unaffected relatives of schizophrenia patients, who have high genetic loading but no prodromal symptoms. Therefore, this group of unaffected subjects clearly differs from subjects at CHR, who present prodromal symptoms. We hypothesize that thalamo-OFC anatomical connectivity is related to genetic risk for schizophrenia and is evident as reduced thalamo-OFC connectivity in unaffected relatives of schizophrenia patients.
Methods
Unaffected relatives of people with schizophrenia were recruited from the Seoul Youth Clinic (Kwon et al., 2010), a centre for the prospective, longitudinal investigation of people at high risk for schizophrenia, and from the inpatient and outpatient clinics of the Department of Psychiatry at Seoul National University Hospital from 2010 in a cohort design. The unaffected relative group was defined as unaffected family members who had at least one first-degree relative with schizophrenia and one or more other affected first- to third-degree relatives. This criterion has advantage of ruling out sporadic cases of schizophrenia. The probands of unaffected relatives were diagnosed with schizophrenia according to the Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) by experienced psychiatrists. Upon admission into the study, the relatives were assessed using the Positive and Negative Syndrome Scale (PANSS), the Brief Psychiatric Rating Scale (BPRS) and the Global Assessment of Functioning (GAF) scale. Subjects with any lifetime history of major psychiatric illness, including psychosis, bipolar disorder and major depressive disorder, were excluded from this study. The age range for inclusion was 15–34 years. In addition, prodromal symptoms were assessed in the relatives using the Structured Interview for Prodromal Syndromes (SIPS). Relatives who met any criteria for CHR for psychosis with SIPS (any P1–P5 ⩾ 3) were excluded from the study. The genetic liability score (generalized least squares [GLS]) of each participating relative was estimated using his or her family tree according to the method described by McDonald (2008).
Healthy control (HC) subjects were recruited via advertisements placed on the Internet. The HC subjects were included if they reported no past or current Axis-I diagnoses and no first- to third-degree biological relatives with any major psychiatric disorder. The HC subjects were assessed for the presence of psychiatric disorders or symptoms by screening with the Structured Clinical Interview for DSM-IV – Non-Patient Edition (SCID-NP). The IQ of each participant was measured with the abbreviated form of the Korean Wechsler Adult Intelligence Scale (K-WAIS).
The exclusion criteria for all participants included a lifetime diagnosis of substance abuse or dependence, neurological disease or head injury; evidence of medical illness with documented cognitive sequelae; sensory impairments; or intellectual disability (IQ < 70). From the larger pool of the unaffected relative cohort and HCs, a propensity-matching R script was used to select groups with matching age, sex, handedness, IQ and years of education, resulting in a total of 35 unaffected relatives (from 31 families in total) and 34 HCs. GLS score was available in 32 unaffected relatives (the family tree was inaccessible for three unaffected relatives). The Institutional Review Board of Seoul National University Hospital approved the study, and written informed consent was obtained from all subjects and from the parents of subjects under 18 years of age.
Image acquisition
T1-weighted (T1) and diffusion-weighted imaging (DWI) data were acquired using a 3T scanner (MAGNETOM TrioTim Syngo MR B17; Siemens, Erlangen, Germany). For the T1 images, we used three-dimensional (3D) magnetization-prepared rapid-acquisition gradient echo (MPRAGE) and the following parameters: TR 1670 ms, TE 1.89 ms, voxel size of 1 × 0.98 × 0.98 mm3, field of view (FOV) 250 mm, 9° flip angle and 208 slices. The DWI data were acquired using echo-planar imaging with the following parameters: TR 11,400 ms, TE 88 ms, 128 × 128 matrix, FOV 240 mm and a voxel size of 1.9 × 1.9 × 3.5 mm3. Diffusion-sensitizing gradient echo encoding was applied in 64 directions using a diffusion-weighting factor (b) of 1000 s/mm2. One volume was acquired with a b-factor of 0 s/mm2 (B0).
To ensure the quality of the images, we visually checked them for any malformation of brain structures, as well as abnormally heightened or lowered intensity. The signal drops in the DWI were also visually checked, appearing as a blank dark line, and any individual dataset including more than five slices with signal drop was set to be removed. Three subjects had signal drop of one a slice in a volume of their DWI image, but none of the data had greater than one slice of signal drop.
Magnetic resonance imaging processing
T1
Left and right OFC and thalamus regions of interest (ROIs) in each individual’s T1 space were automatically extracted as binary masks using FreeSurfer (Reuter et al., 2012).
DWI
DWI data were preprocessed using eddy current correction with b vector correction and skull removal and motion correction using the FMRIB Software Library (FSL). Information about the head motion has been added to supplemental material. The tensor was fitted to the DWI data, which created a fractional anisotropy (FA) map. An individual B0 image was used to create the matrix that registered its own T1 image to DWI using FLIRT (Jenkinson et al., 2002). The quality of the registration was visually checked for any abnormal fitting of the data by overlaying the two images together, and none of the images showed abnormal registration. Then, for each side of the brain, FSL probabilistic tractography (Behrens et al., 2007) was applied with default options (including 5000 streams per voxel seed) using the individually registered thalamus ROI as a seed and the OFC ROI as the target. Exclusion masks of contralateral white matter, temporal, occipital and parietal white matter, as well as a coronal plane placed at the most posterior coordinate of the thalamus, were used to block the tracts projecting to unwanted areas. After thresholding the tractography map at the third percentile to remove aberrant connections, we estimated the mean FA in the tract.
Statistical analysis
All statistical analyses were performed using the StatsModels module (Seabold and Perktold, 2010) in Python. We tested the demographic data for differences using Student’s t-tests and tests for equality of proportions. Student’s t-test has been used to test group differences in the intracranial volume, which was estimated using FreeSurfer. Then, for each hemisphere, the group difference in the thalamo-OFC FA values was assessed with analysis of covariance (ANCOVA), with age and gender as the covariates, to reveal the group effect. ANCOVAs with motion, ROI volumes and intracranial volumes are presented in the supplemental material for readers’ reference. Partial Spearman’s correlation was used to investigate the correlation between the GLS and FA value of the tracts with significant group differences using age and gender as the covariates.
Results
There was no significant demographic difference between the unaffected relatives and HCs (Table 1). The individual tracts were successfully estimated by tractography as shown in Figure 2(A). There was no difference in the intracranial volume between groups, T(68) = −0.057, p = 0.955. There was a significant group effect on the FA value in the tract between the thalamus and OFC, F(1, 65) = 6.93, p = 0.011, partial η2 = 0.10, in the left hemisphere. This effect remained significant when multiple comparisons for both sides were corrected with Bonferroni methods. However, the group effect in the right hemisphere was not significant, F(1, 65) = 1.76, p = 0.189, partial η2 = 0.02. The tract and FA value in the left thalamo-OFC tract of each group are shown in Figure 2(B). Partial correlation analysis between the FA value in the left hemisphere and the GLS revealed no significance, R = 0.28, p = 0.14.
Demographic information and statistical comparisons of the groups.
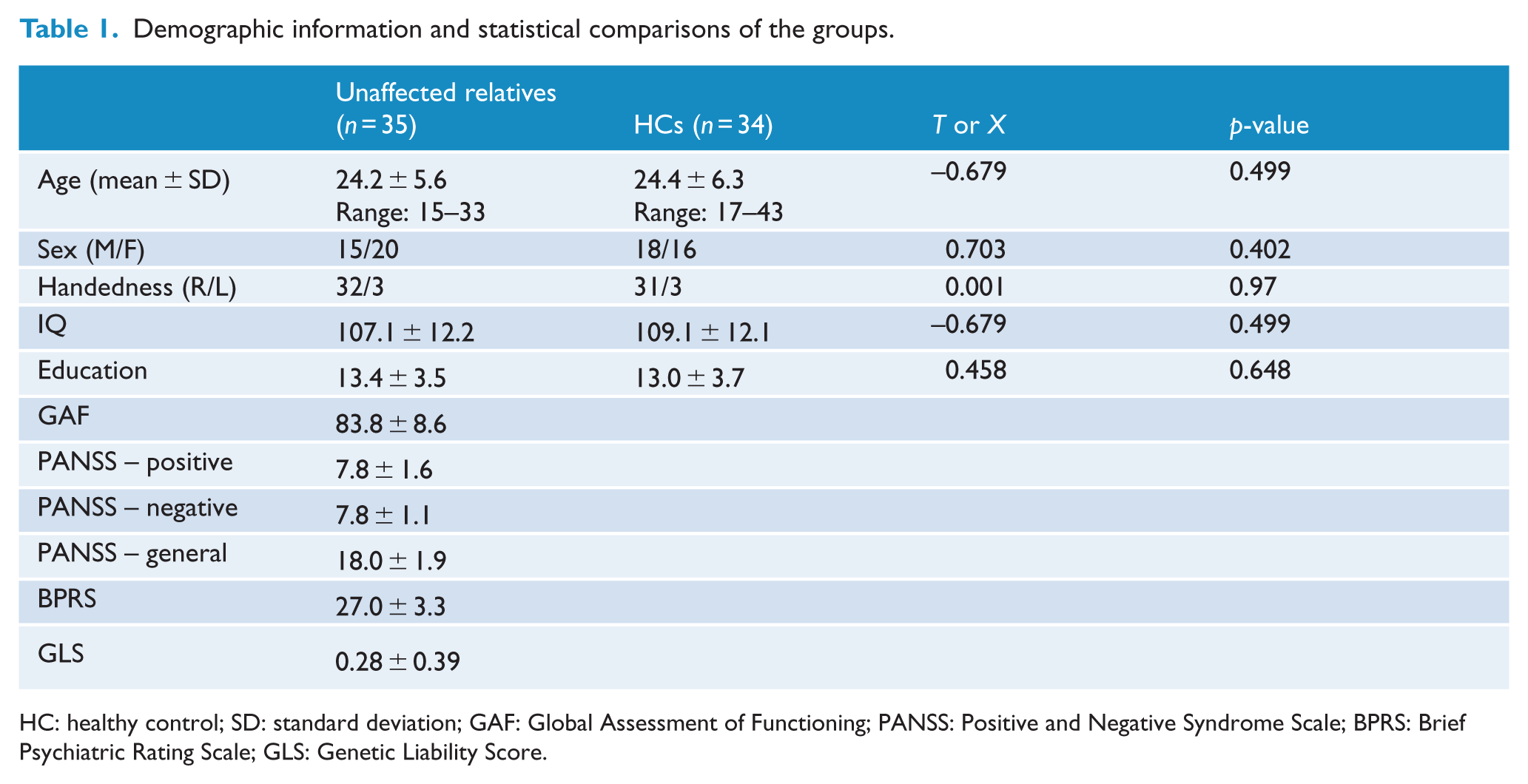
HC: healthy control; SD: standard deviation; GAF: Global Assessment of Functioning; PANSS: Positive and Negative Syndrome Scale; BPRS: Brief Psychiatric Rating Scale; GLS: Genetic Liability Score.
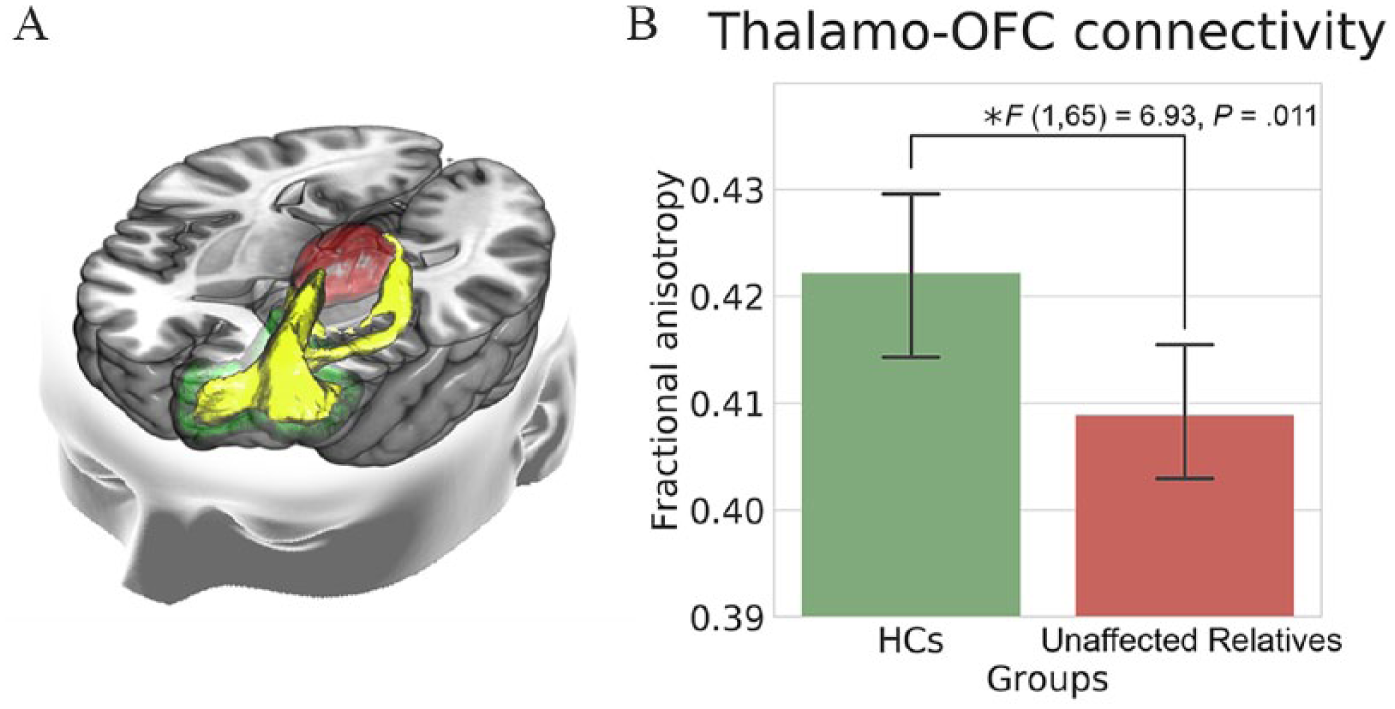
(A) Thalamo-orbitofrontal tracts according to probabilistic tractography. The thalamus is the red structure, orbitofrontal cortex is the green structure and the tracts are shown in yellow. (B) Bar graph of fractional anisotropy in the left thalamo-orbitofrontal tracts for each group.
Discussion
To our knowledge, this is the first study to investigate thalamocortical connectivity disruptions in unaffected relatives of schizophrenia, which could show that this consistently reported alteration also relates with schizophrenia genetics. In agreement with our hypothesis, the results show reduced FA in the thalamo-OFC connection in the unaffected relatives, which is consistent with the findings from chronic schizophrenia (Kubota et al., 2013), FEP and CHR (Cho et al., 2015). This result confirms that such hypoconnectivity is not only associated with illness but also supports the possibility that the proposed biomarker is also attributable to schizophrenia genetics.
This potential biomarker has gained support not only from thalamocortical tractography studies (Cho et al., 2015; Giraldo-Chica et al., 2018; Kubota et al., 2013; Marenco et al., 2011) but also from many studies showing focal changes in the white matter structures that link the thalamus with the PFC, such as the internal capsule (Kubicki et al., 2005; Rosenberger et al., 2012) and the anterior thalamic projections (Clark et al., 2011). These studies have highlighted the role of thalamocortical circuitry in the pathophysiology of schizophrenia and have been supported by reports of functional connectivity changes in the circuit (Anticevic et al., 2014, 2015; Ferri et al., 2018; Woodward and Heckers, 2016; Woodward et al., 2012). The findings in the thalamocortical circuit are in agreement with Friston’s (1999) disconnection hypothesis, and since the connection involves the major relay centre of the brain, the thalamus, it is no exaggeration to state that there is a possible link between the thalamocortical system and functional asynchronies between distant brain regions (Crossley et al., 2009; Yoon et al., 2015). In addition to the accumulated evidence, the alteration found in unaffected relatives in our study further strengthens the plausibility of the findings and suggests that this alteration also has genetic aspects. Therefore, this potential biomarker may serve as an endophenotype to identify individuals with psychosis as well as those at clinical or genetic risk of psychosis on biological basis. Establishing more biomarkers such as this aberrant brain connection may contribute to early detection and prevention of schizophrenia.
The previously reported alterations of thalamocortical connections in patients with schizophrenia were complicated by various confounding factors that affect the circuitry, including unhealthy lifestyles, medications and onset-related brain changes. These confounding factors make it difficult to define altered connectivity as a true biological marker. Our investigation of unaffected family members allows the identification of biomarkers that are free from the aforementioned confounding factors, making it possible to focus on the effect of shared genes.
While this is the first study to investigate alterations in the thalamo-OFC anatomical connectivity in unaffected relatives, there are several previous findings that seem to suggest abnormalities in the thalamo-OFC connections in relatives of schizophrenia patients. In addition to reports of reduced orbitofrontal volumes in relatives (Bhojraj et al., 2011; McIntosh et al., 2011), there are reports of white matter anomalies in the anterior limb of the internal capsule and in the inferior fronto-occipital fasciculus (Clark et al., 2011; Kubicki et al., 2011), which significantly overlap with the thalamo-prefrontal connection paths. Although the authors did not specifically describe these changes in terms of the thalamocortical circuits, the changes in these regions are consistent with our finding.
In addition to the accumulating studies of the thalamocortical system, a potential link between the symptoms of schizophrenia and the thalamic role of information transfer and modulation has gained more attention. As it is the core relay system in the brain, the altered thalamocortical system might affect signalling in other systems, which may then lead to symptoms. The unaffected family members also manifest this common alteration, but perhaps to a lesser degree. Although these potential factors need to be investigated further, our study confirms that the alteration in the thalamocortical connection is a common feature of schizophrenia that arises partly from shared genetic factors. We hope that future studies of this common variable will lead to a deconstructed diagnosis or deconstructed diagnosis of schizophrenia at a more biological level using this abnormal connectivity in combination with other different types of measures.
Limitations
From our analysis, the correlation between the reduced FA in the thalamo-OFC tract and the GLS was not significant. It is possible that the FA measure may not have been sensitive enough to show linear correlation with the genetic loading estimated using GLS. Effects other than genetic loading, such as a shared rearing environment, may have complicated the relationship. However, it is also possible that the GLS may not have been sensitive enough to adequately represent genetic loading, as it is predisposed to major bias on the basis of family size. More precise genetic loading estimated from blood samples, examining the number of single-nucleotide polymorphisms shared with a large sample of schizophrenia patients, such as the data published by Schizophrenia Working Group of the Psychiatric Genomics Consortium, (Schizophrenia Working Group of the Psychiatric Genomics Consortium, 2014) may help reveal a direct relationship between abnormal thalamo-OFC connectivity and genetic loading.
Probabilistic tractography method was used to estimate the structure of the white matter tracts from the thalamus to OFC. These tracts pass through a major fibre bundle, the internal capsule; however, other thalamo-prefrontal tracts also pass through this area. Currently, the resolution of DWI is limited; it is difficult to distinguish between fibres with different targets within the same bundle. Therefore, the thalamo-OFC hypoconnectivity identified in our study does not fully exclude the effects of changes in other thalamo-prefrontal connections and might convey overall disconnection between the thalamus and the PFC. Also, more advanced method of bi-compartmental models such as free water elimination could be used to investigate tissues in more detail (Pasternak et al., 2009). In addition, the diffusion imaging protocol used in this study could have been optimized further by setting the voxel size to isotropic dimensions, obtaining more B0 images and using dual encoding directions, which could significantly help correct the susceptibility-induced distortions.
Conclusion
Reduced thalamo-OFC white matter connectivity was evident in the unaffected relative group, which indicates that white matter changes occur even in the unaffected family members of patients with schizophrenia. This finding suggests that thalamocortical dysconnectivity, especially the thalamo-OFC white matter connection, is related to the genetic risk of schizophrenia. It strengthens the possibility of the alteration in the thalamo-OFC connection as a putative biomarker for psychosis that may contribute to the understanding of the pathophysiology of schizophrenia.
Supplemental Material
Supplementary – Supplemental material for Disturbed thalamocortical connectivity in unaffected relatives of schizophrenia patients with a high genetic loading
Supplemental material, Supplementary for Disturbed thalamocortical connectivity in unaffected relatives of schizophrenia patients with a high genetic loading by Kang Ik K Cho, Minah Kim, Youngwoo Bryan Yoon, Junhee Lee, Tae Young Lee and Jun Soo Kwon in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
K.I.K.C. contributed to analysis and interpretation of the neuroimaging data and writing of the manuscript; M.K., Y.B.Y., J.H.L., T.Y.L. and J.S.K. contributed to design of the cohort, analysis and interpretation of the data and writing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This research was supported by the Brain Research Programme through the National Research Foundation of Korea (NRF), funded by the Ministry of Science, ICT & Future Planning (grant nos 2017M3C7A1029610 and 2016R1E1A1A02921618).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
