Abstract
Objectives:
Bipolar disorder is associated with a decreased life expectancy of 8–12 years. Cardiovascular disease is the leading cause of excess mortality. For the first time, we investigated the Framingham 30-year risk score of cardiovascular disease in patients with newly diagnosed/first-episode bipolar disorder, their unaffected first-degree relatives and healthy individuals.
Methods:
In a cross-sectional study, we compared the Framingham 30-year risk score of cardiovascular disease in 221 patients with newly diagnosed/first-episode bipolar disorder, 50 of their unaffected first-degree relatives and 119 healthy age- and sex-matched individuals with no personal or first-degree family history of affective disorder. Among patients with bipolar disorder, we further investigated medication- and illness-related variables associated with cardiovascular risk.
Results:
The 30-year risk of cardiovascular disease was 98.5% higher in patients with bipolar disorder (p = 0.017) and 85.4% higher in unaffected first-degree relatives (p = 0.042) compared with healthy individuals in models adjusted for age and sex. When categorizing participants in low cardiovascular risk without considering age and sex distribution among participants, 81% of patients were at low risk, versus 92% of unaffected relatives and 89% of healthy individuals. Of the patients 209 (94.6%) were diagnosed within the preceding 2 years. Smoking was more prevalent among patients with bipolar disorder (45.2%) and their unaffected first-degree relatives (20.4%) compared with healthy individuals (12.8%). Similarly, dyslipidemia was more common among patients with bipolar disorder compared with healthy individuals. Treatment with psychotropic medication with metabolic adverse effects was associated with higher 30-year cardiovascular disease risk score, whereas we did not find illness-related variables associated with cardiovascular risk among patients with bipolar disorder.
Conclusion:
We found an enhanced cardiovascular disease risk score in patients with newly diagnosed bipolar disorder and their unaffected first-degree relatives, which points to a need for specific primary preventive interventions against smoking and dyslipidemia in these populations.
Introduction
Bipolar disorder (BD) has a lifetime prevalence of 1–2% and is one of the leading causes of disability worldwide (Alonso et al., 2011; Clemente et al., 2015; Kessler et al., 2007). The life expectancy is decreased 8–12 years in BD (Kessing et al., 2015a; Laursen et al., 2013), mainly due to physical illnesses even among younger patients aged 15–25 years (Kessing et al., 2015b). Cardiovascular disease (CVD) is the leading cause of excess mortality in BD (Osby et al., 2001; Weiner et al., 2011). Recently, the American Heart Association recognized BD in youth as a moderate risk condition for early CVD and accelerated atherosclerosis (Goldstein et al., 2015a). Specifically, CVD occurs up to 17 years earlier in patients with BD than in the general population (Goldstein et al., 2015b), and the mortality rate ratio of CVD is increased eightfold in patients with BD younger than 40 years of age (Westman et al., 2013). Emerging evidence indicates that the illness burden (e.g. episode frequency) in BD is predictive of CVD mortality exhibiting a dose–response relationship (Fiedorowicz et al., 2009, 2014; Goldstein et al., 2009). Furthermore, CVD risk factors are associated with greater illness severity, poorer treatment response and a chronic illness trajectory in patients with BD (Calkin et al., 2015; Kemp et al., 2010). As onset of BD typically occurs in young adulthood (Kessler et al., 2007), prediction of long-term CVD risk is critical as risk stratification can meaningfully inform primary and preventive interventions and resources.
The individual risk of CVD can be predicted using the Framingham 30-year risk score in young adults (Pencina et al., 2009b). The longitudinal risk estimate provided by the Framingham risk score is considered to more accurately predict overt and subclinical CVD than scores based on relatively shorter duration (Berry et al., 2009; Laing et al., 2012; Pencina et al., 2009b). Moreover, assessment of CVD risk factors in youth has been noted to be superior to later evaluations in predicting subclinical CVD during a 15-year time span (Loria et al., 2007). The Framingham 30-year risk score tool is derived from the Framingham Offspring cohort—a representative epidemiological sample from the general population—and estimates the risk of CVD defined as the outcomes of coronary death, myocardial infarction and stroke (‘hard CVD’) during a 30-year period. The Framingham risk factors largely overlap with the CVD risk factors clustered in the metabolic syndrome, but differ by additionally including age and smoking status and by including total cholesterol levels instead of triglyceride levels (Alberti et al., 2006; Pencina et al., 2009b).
Investigations of CVD risk factors at onset of BD are scarce despite the evidence for CVD being the primary source of mortality in patients with BD. Only one small study found lower fasting plasma high-density lipoprotein cholesterol (HDL) levels in young patients with newly diagnosed BD (n = 11) 2 years after psychotropic treatment initiation (Taylor et al., 2010). Findings of elevated CVD risk factors in adolescents with BD have also been reported (Naiberg et al., 2016). The risk of CVD has not previously been investigated in unaffected first-degree relatives of patients with BD except for a single small study of dyslipidemia, that is, lower HDL levels in the high-risk group compared with healthy controls (Sobczak et al., 2004). Consequently, the risk of CVD and the prevalence of CVD risk factors among newly diagnosed patients with BD and their unaffected first-degree relatives are largely unknown.
Aims of the study
The aims of this study were (a) to compare the 30-year risk score of CVD in patients with newly diagnosed/first-episode BD and their first-degree unaffected relatives with healthy individuals without personal or first-degree family history of affective disorder and (b) to determine to what extent illness and medication variables in patients with BD were associated with the 30-year risk score of CVD.
We hypothesized that the risk of CVD is elevated in patients with newly diagnosed/first-episode BD and—to a lesser degree—in their unaffected first-degree relatives compared with healthy individuals without a family history of psychiatric disorders.
Materials and methods
Study design
This study is a cross-sectional investigation of baseline data from the Bipolar Illness Onset Study (BIO), an ongoing longitudinal study, which aims to identify biomarkers for BD (Kessing et al., 2017). The recruitment took place from June 2015 to December 2017. The study protocol was approved by the Committee on Health Research Ethics of the Capital region of Denmark (protocol No. H-7-2014-007) and the Danish Data Protection Agency, Capital Region of Copenhagen (RHP-2015-023). All participants provided written informed consent. The study complied with the Declaration of Helsinki principles (Seoul, October 2008).
Participants
Patients with BD
Patients with BD were recruited from the Copenhagen Affective Disorder Clinic that covers the Copenhagen catchment area of 1.6 million inhabitants (The Capital Region). All patients referred to the Copenhagen Affective Disorder Clinic as newly diagnosed with BD or having a first episode of (hypo)mania were potentially eligible for the BIO study. These patients were routinely invited to participate in the study as part of their treatment in the clinic. Inclusion criteria were an International Classification of Diseases, 10th Revision (ICD-10) diagnosis of BD or a single manic episode and age 15–70 years. Exclusion criterion was having an organic BD secondary to brain injury. Patients with BD received treatment as usual in Copenhagen Affective Disorder Clinic without any involvement of study investigators.
Unaffected first-degree relatives
Siblings and children of the included patients with BD were invited to participate upon consent by the participating patient. Inclusion criteria were being a first-degree relative of an included patient with BD and aged 15–40 years. Exclusion criteria were an ICD-10 diagnosis lower than F34 including substance abuse, psychotic illnesses and a diagnosis of unipolar disorder or BD. We did not apply restrictions of number of relatives per patient with BD as we adjusted our analysis for the familial relationship.
Healthy individuals
Age- and sex-matched healthy individuals were recruited among blood donors from the Blood Bank at Rigshospitalet, Copenhagen, Denmark, by contacting blood donors in the waiting room at the Blood Bank on random days. Inclusion criterion was age 15–70 years. Exclusion criteria were a personal or first-degree family history of psychiatric disorders that had required treatment.
Notably, while our total study population encompassed individuals aged 15–70 years, only individuals aged 20–59 years were included in the current report, in accordance with the age range validated for use with the Framingham risk score.
Clinical assessments
The initial diagnostic assessment was completed by a specialist in the diagnosis of patients with BD according to ICD-10 and classifying patients with BD as type I or type II according to Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5) as part of daily practice in the Copenhagen Affective Disorder Clinic. All participants were assessed by a medical doctor or psychologist trained in diagnosing BD.
Following informed consent, the initial clinical diagnosis of BD was confirmed in a semi-structured research-based interview using the Schedules for Clinical Assessment in Neuropsychiatry (SCAN) (Wing et al., 1990). Exercise level was quantified using the International Physical Activity Questionnaire (IPAQ) (Craig et al., 2003) at the day of assessment. Diagnosis of the current affective state was based on ICD-10 criteria. Severity of depressive and manic symptoms was assessed using the Hamilton Depression Rating Scale-17 items (HAMD-17) (Hamilton, 1960) and the Young Mania Rating Scale (YMRS) (Young et al., 1978).
Absence of lifetime psychiatric morbidity defined by ICD-10 was confirmed for healthy individuals whereas psychiatric morbidity of F34 and higher (e.g. anxiety, phobias and personality disorders) according to ICD-10 were registered for unaffected first-degree relatives. Current medication and smoking habits were recorded.
Anthropometric assessment
Blood pressure was measured using a calibrated automatic sphygmomanometer (Microlife BP A3 plus) in a sitting position after a 10-minute rest. Height was measured lightly dressed and without shoes to the nearest millimeter on a rigid stadiometer. Weight was measured to the nearest 0.1 kg using a calibrated floor scale (Kern MPE PM).
Laboratory methods
At the same day as clinical assessments blood samples were collected in a fasting state, between 7.30 a.m. and 10 a.m., after participants had a period of minimum 15-minute rest. Five milliliters of blood was drawn by venepuncture into an EDTA-containing vacuum tube (Vacuette®) and within 30 minutes centrifuged at 1590 g and 4 °C for 15 minutes. Plasma was aliquoted into Eppendorf® tubes and kept frozen at −80°C until levels of insulin were assayed.
P-glucose, P-HbA1c, P-HDL and P-Cholesterol were measured as part of standard laboratory routine. Blood sampling and all aspects of laboratory processing were done at the Department of Clinical Biochemistry, Rigshospitalet by educated technicians blinded with respect to participant status.
Determination of 30-year risk score of CVD
Risk of CVD was calculated using a sex-specific multivariable algorithm, the Framingham 30-year risk score, which is validated for age 20–59 years (Pencina et al., 2009b) and available on the Framingham website (Pencina et al., 2009a). Eight risk factors are incorporated in the Framingham algorithm: age, sex, HDL, total cholesterol, systolic blood pressure, diabetes mellitus (DM), antihypertensive treatment and current smoking status. Body mass index (BMI) can be used instead of lipid levels if these are not available and both models are found to predict CVD with high accuracy, though the calculation based on lipids seems superior (Albertini et al., 2017; Pencina et al., 2009b).
The cardiovascular risk score was represented by the Framingham ‘hard CVD’ score, which is the risk of coronary death, myocardial infarction and stroke. First, we examined the absolute ‘hard CVD’ score; second, we investigated the normalized ‘hard CVD’ risk score, calculated as the difference between the individual risk score and the risk of an age- and sex-matched individual with the remaining risk factors at normal levels based on the Framingham cohort data (calculation available on the Framingham website) as an indicator of the individual CVD risk relative to the Framingham normal population.
Participants were considered having DM if they reported a diagnosis of type 1 or type 2 diabetes or met at least one of the following three criteria: in antidiabetic treatment, having a fasting blood glucose of 7 mmol/L or above (Framingham criteria) or a serum-HbA1C level of 48 mmol/mol or above. Participants were considered having CVD if they reported having been diagnosed with angina pectoris or myocardial infarction.
Statistical analyses
Descriptive data were analyzed by chi-square tests for categorical data and by Mann–Whitney U tests for two independent groups of continuous data, where assumptions of normal distribution were not met. Continuous data were presented as median and quartiles if assumptions of normal distribution were not met.
For our main analyses, we employed linear mixed effect models with the absolute and normalized 30-year risk of CVD as the dependent variable and group (patient with BD, unaffected relative, healthy control individual), age and sex as independent variables and familial relationship as random effect, to account for the correlation between family-related individuals. In an exploratory analysis, we additionally added the dichotomous variable ‘active smoking’ versus ‘non-smoking’.
In analyses among patients with BD, the associations between current medication and illness characteristics and the 30-year risk of CVD were explored in separate univariate models. In the first models, (a) current treatment with lithium (yes/no), (b) antipsychotics (yes/no), (c) antidepressants (yes/no) and (d) antiepileptics (yes/no) were entered as independent variables and then (e) repeating the analysis, substituting the four treatment variables with the categorical variable currently ‘receiving medication with metabolic adverse effects’ versus ‘not receiving psychotropic medication with metabolic adverse effects’. All daily psychotropic medication except for lamotrigine and aripiprazole were considered medication with metabolic adverse effects (Abosi et al., 2018). The category ‘not receiving psychotropic medication with metabolic adverse effects’ included patients not receiving psychotropic medication as well as patients receiving lamotrigine, aripiprazole or low-dose quetiapine (i.e. not exceeding 50 mg daily). In similar univariate analyses, ‘receiving more than one type of psychotropic medication’ versus ‘receiving one type of psychotropic medication’ (f), illness duration (g), the number of prior hypomanic and manic episodes (h), number of suicide attempts (i) prior psychotic episode (yes vs no; j) and prior hospitalization (yes vs no; k), respectively, was entered as the independent variable. Illness duration was defined as time from first episode (depressive, (hypo)manic or mixed episode) in years. In a last model (l), the BD type (I or II) was entered as independent variable. In this model, patients with a single manic episode (n = 3) were included in the BD type I group according to the DSM-5 classification.
For all parametric tests, Framingham risk scores were transformed by the natural logarithm. Results are presented as backtransformed values with a parameter estimate B, expressing the ratio between increments in independent variables. All model assumptions were met. The level of statistical significance was set at p < 0.05. Statistical analyses were performed using SPSS version 22.
Results
Demographic and clinical characteristics
We included 221 patients with BD, 50 of their unaffected first-degree relatives and 119 healthy individuals. Unaffected relatives of 41 patients with BD were included and nine of the patients with BD had two of their unaffected relatives included. Demographic and clinical variables of the study participants are presented in Table 1. The median [interquartile range] age was higher in patients with BD (30 [25–38.5] years vs 27 [24–35] years, p = 0.027) while education level was lower (15 [13–17] years vs 16 [15–17] years, p < 0.001) compared with healthy individuals, whereas sex distribution was similar in the two groups. There was no difference in age, sex and education level in unaffected first-degree relatives compared with healthy individuals. Patients with BD were less physically active than healthy individuals (p = 0.002) and had a larger waist circumference than healthy individuals (p < 0.001). Unaffected first-degree relatives did neither differ in level of physical activity from healthy individuals (p = 0.9) nor from patients with BD (p = 0.6). Waist circumference of unaffected first-degree relatives did not differ from healthy individuals (p = 0.7) but differed from patients with BD (p = 0.003). Among the patients with BD, 81% (n = 179) were diagnosed within the preceding year; additionally, 13.6% (n = 30) were diagnosed within the last 2 years, whereas the remaining 4.5% (n = 12) were diagnosed within the preceding 7 years. At least one previous suicide attempt was reported by 41 (18.5%) of the patients with BD, whereas 49 (22.2%) had experienced a psychotic episode and 103 (46.6%) had been hospitalized.
Demographic and clinical variables in patients with bipolar disorder (BD), their unaffected first-degree relatives (UR) and healthy individuals (HC).
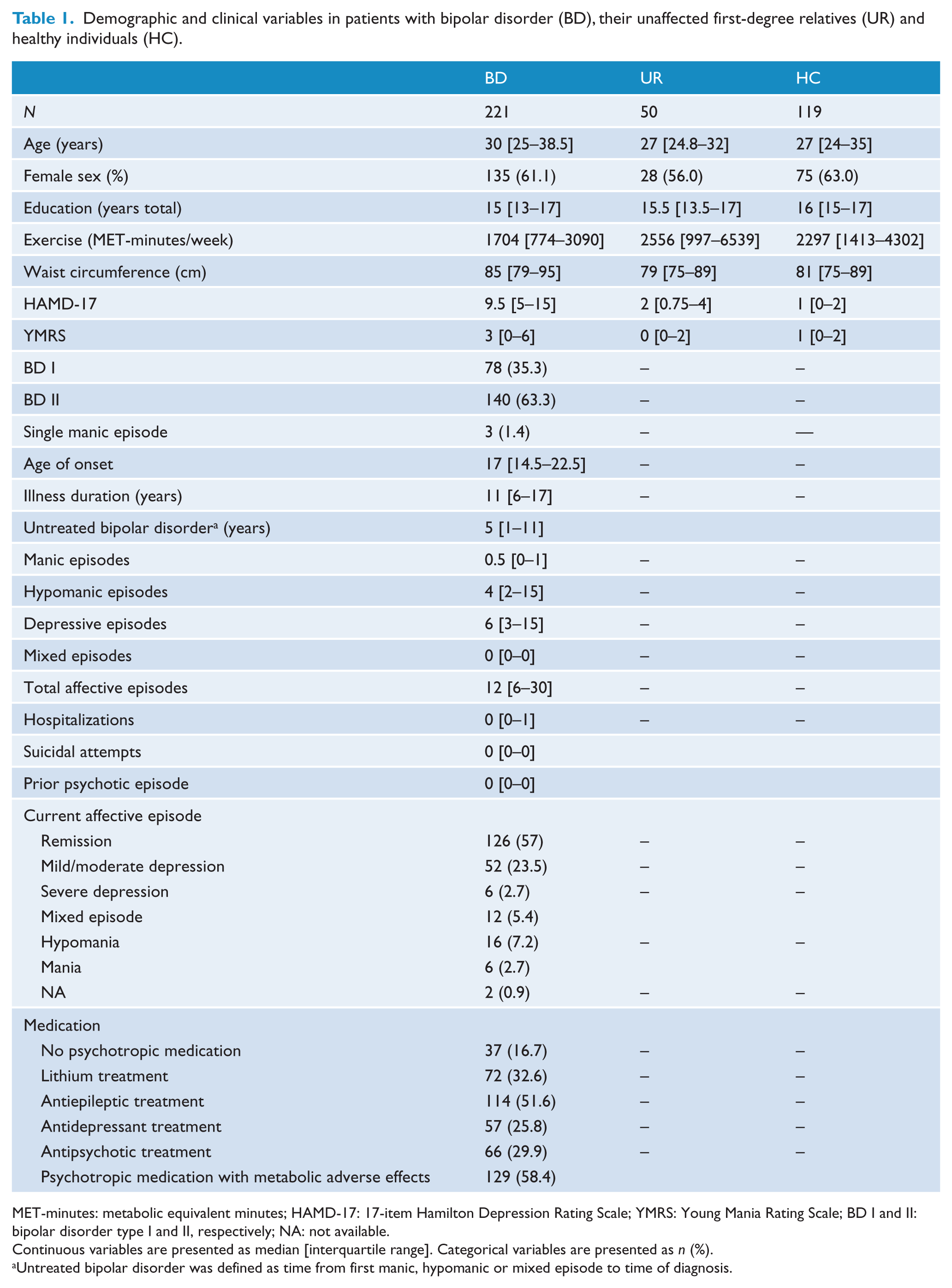
MET-minutes: metabolic equivalent minutes; HAMD-17: 17-item Hamilton Depression Rating Scale; YMRS: Young Mania Rating Scale; BD I and II: bipolar disorder type I and II, respectively; NA: not available.
Continuous variables are presented as median [interquartile range]. Categorical variables are presented as n (%).
Untreated bipolar disorder was defined as time from first manic, hypomanic or mixed episode to time of diagnosis.
Individual risk factors for CVD
The Framingham 30-year risk score variables are available in Table 2. Patients with BD had lower systolic blood pressure and lower HDL levels compared with healthy individuals. A larger proportion of patients with BD were smokers (45.2%) than unaffected first-degree relatives (20.4%) and healthy individuals (12.8%) (p < 0.001). BMI was higher in patients with BD compared with healthy individuals (p = 0.029); however, medians for all three groups were within the normal BMI range. Three participants, two patients with BD and one unaffected first-degree relative, fulfilled criteria for DM type 1 or 2, while three participants, all patients with BD, got antihypertensive treatment. No participant had manifested CVD. Levels of cholesterol and HDL were available for 220 patients with BD, 49 unaffected first-degree relatives and 118 healthy individuals. For the remaining three participants, BMI was used in the Framingham 30-year CVD risk calculation.
Framingham 30-year cardiovascular disease risk variables in patients with bipolar disorder (BD) and their unaffected first-degree relatives (UR) compared with healthy individuals (HC).
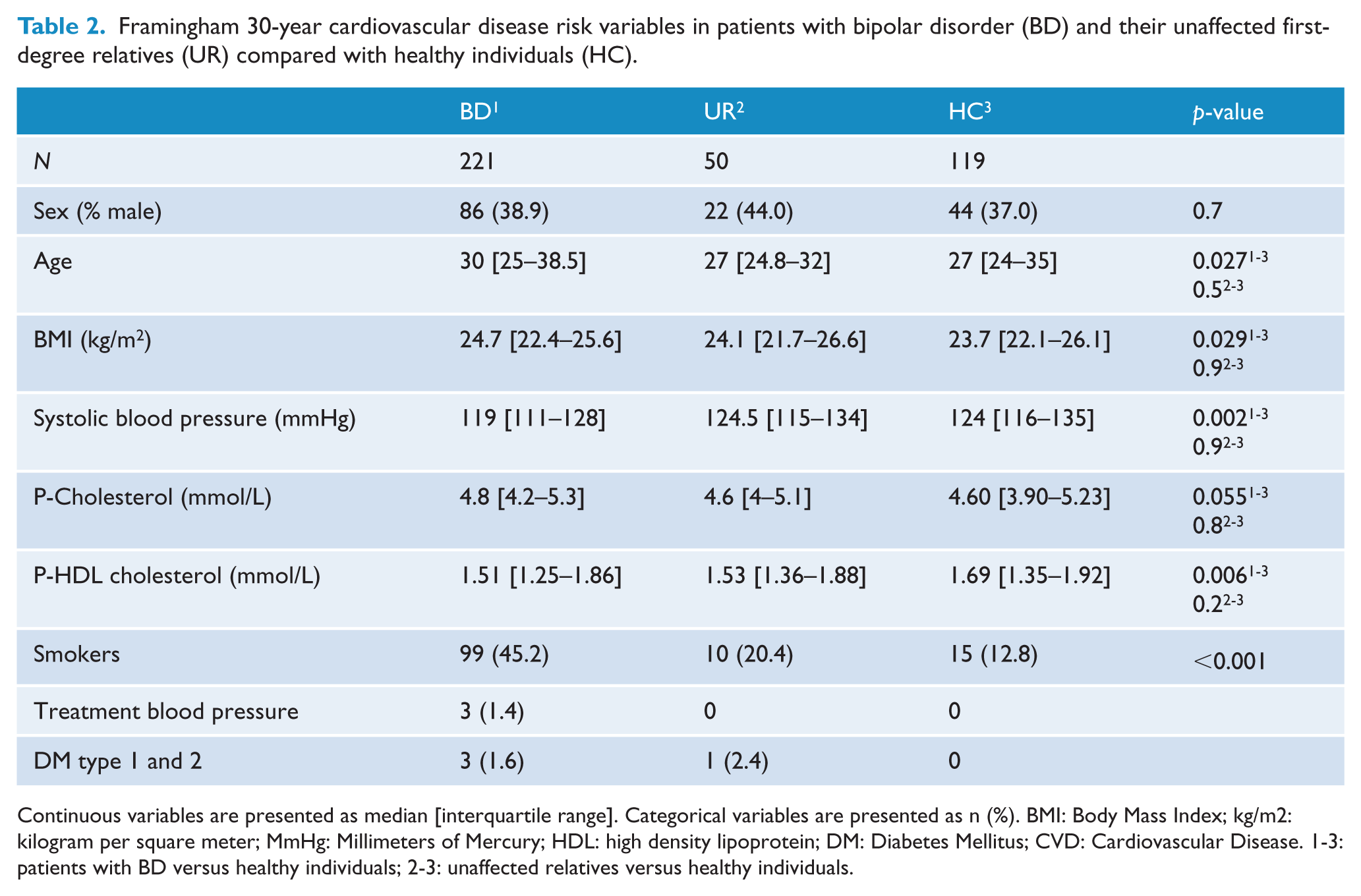
Continuous variables are presented as median [interquartile range]. Categorical variables are presented as n (%). BMI: Body Mass Index; kg/m2: kilogram per square meter; MmHg: Millimeters of Mercury; HDL: high density lipoprotein; DM: Diabetes Mellitus; CVD: Cardiovascular Disease. 1-3: patients with BD versus healthy individuals; 2-3: unaffected relatives versus healthy individuals.
The Framingham 30-year risk score of CVD in patients with BD, their unaffected first-degree relatives and healthy individuals
The median [interquartile range] absolute 30-year risk of CVD was 4% [2–9%] in patients with BD, 2% [1–5%] in their unaffected first-generation relatives and 2% [1–5%] in healthy individuals (Figure 1). The 30-year CVD risk was elevated by 98.5% in patients with BD (B = 1.985, 95% confidence interval (CI): [1.133, 3.476], p = 0.017) and by 85.4% in their first-degree relatives (B = 1.854, 95% CI: [1.024, 3.358], p = 0.042) compared with healthy individuals in analyses adjusted for group, sex and age (Table 3). In a comparable model, there was no statistically significant difference in CVD risk between patients with BD and their first-degree relatives (B = 1.070, 95% CI: [0.848, 1.357], p = 0.6).
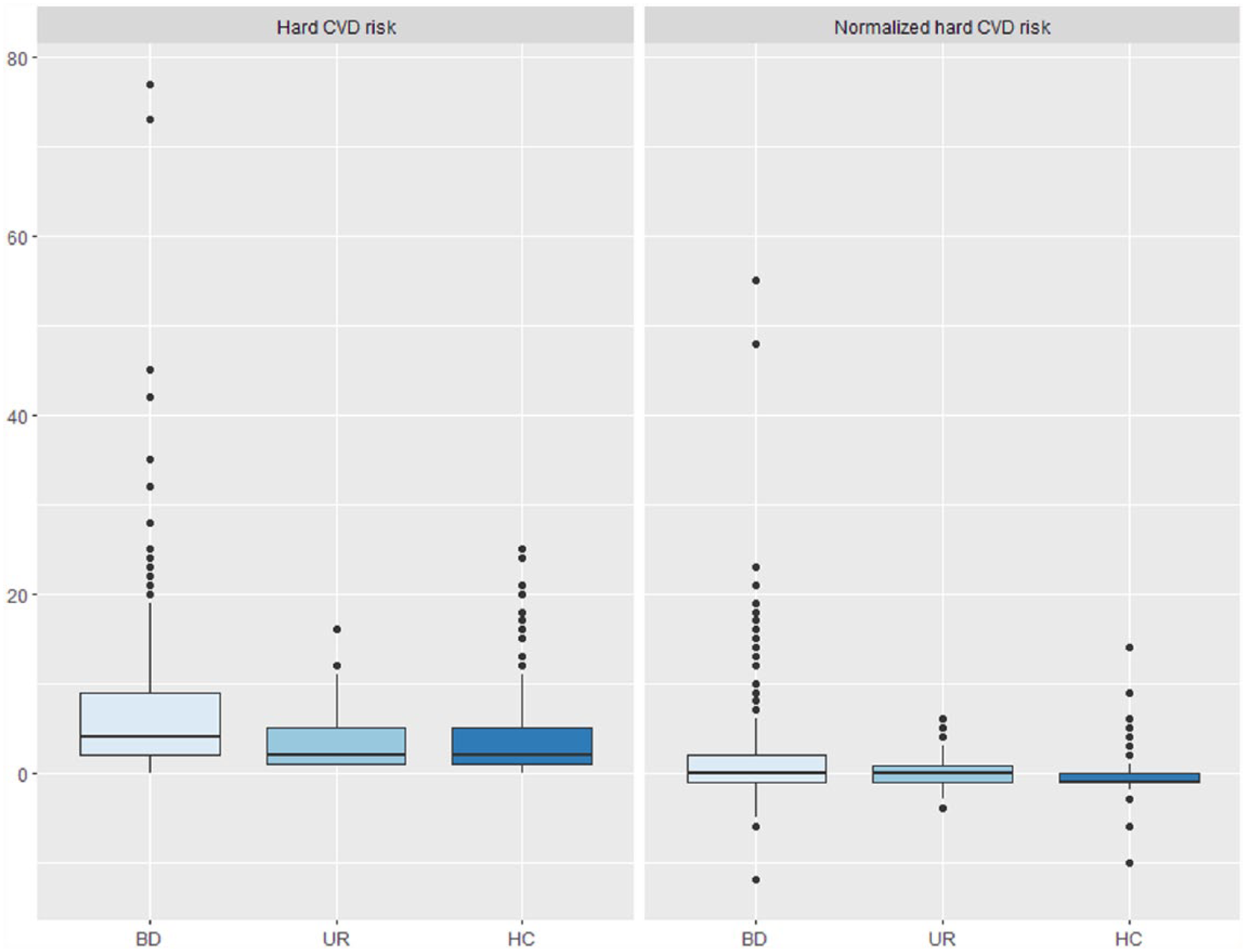
Box plot of absolute 30-year cardiovascular disease (CVD) risk and normalized CVD risk in patients with newly diagnosed bipolar disorder (BD), their unaffected first-degree relatives (UR) and healthy individuals (HC). The lower and upper hinges correspond to the first and third quartiles and the upper and lower whiskers extend from the hinge to the largest and lower value, respectively, no further than 1.5 times the interquartile range from the hinge. Data beyond the end of the whiskers are plotted individually.
Thirty-year risk of cardiovascular disease in patients with bipolar disorder (BD) and their unaffected first-degree relatives (UR) compared with healthy individuals (HC), and 30-year risk of CVD within the patients with BD.
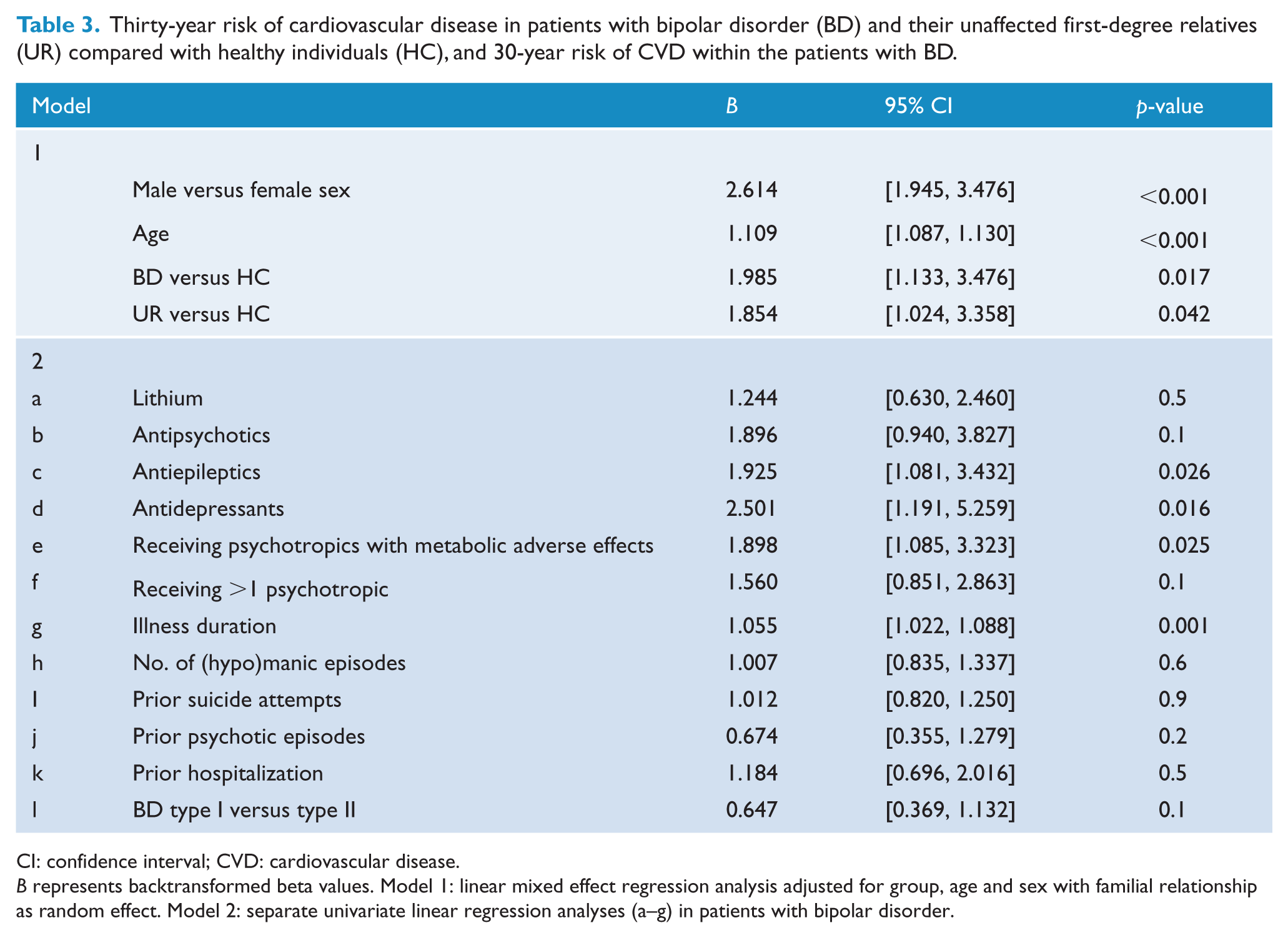
CI: confidence interval; CVD: cardiovascular disease.
B represents backtransformed beta values. Model 1: linear mixed effect regression analysis adjusted for group, age and sex with familial relationship as random effect. Model 2: separate univariate linear regression analyses (a–g) in patients with bipolar disorder.
Due to our naturalistic study design, patients with ‘newly diagnosed BD’ were diagnosed within the preceding 7 years. We repeated the analysis of 30-year CVD risk adjusted for sex and age, this time only including the 208 patients with BD (94.6%), who had been diagnosed within the preceding 2 years. In this model, we found similar elevated risks of 93.7% in patients with BD (B = 1.937, 95% CI: [1.092, 3.435], p = 0.024) and of 86.2% in their unaffected first-degree relatives (B = 1.862, 95% CI: [1.015, 3.417], p = 0.045). In a post hoc analysis of the 30-year CVD risk adjusted for sex, age and active smoking, the higher 30-year CVD risk score in patients with BD (B = 1.702, 95% CI: [0.956, 3.032], p = 0.07) and their first-degree relatives (B = 1.649, 95% CI: [0.904, 3.009], p = 0.1) compared with healthy individuals was no longer statistically significant. A final post hoc analysis was run substituting active smoking with exercise level, and the 30-year CVD risk score remained elevated in patients with BD (B = 2.098, 95% CI: [1.094, 4.022], p = 0.026) but not in their first-degree relatives (B = 1.946, 95% CI: [0.973, 3.891], p = 0.06) compared with healthy individuals.
When categorizing CVD risk into low (<12%), intermediate (12–40%) and high (>40%) CVD risk categories (Pencina et al., 2009b), 81% of patients with BD, 92% of unaffected first-degree relatives and 89% of healthy individuals, respectively, were stratified as low risk (Figure 2). Among patients with BD, 17.2% had intermediate risk, while the corresponding value was 8% in unaffected first-degree relatives and 10.9% in healthy individuals, and only four patients with BD (1.8%) were categorized as high risk. These differences were not statistically significant (chi-square = 7.6, p = 0.1).
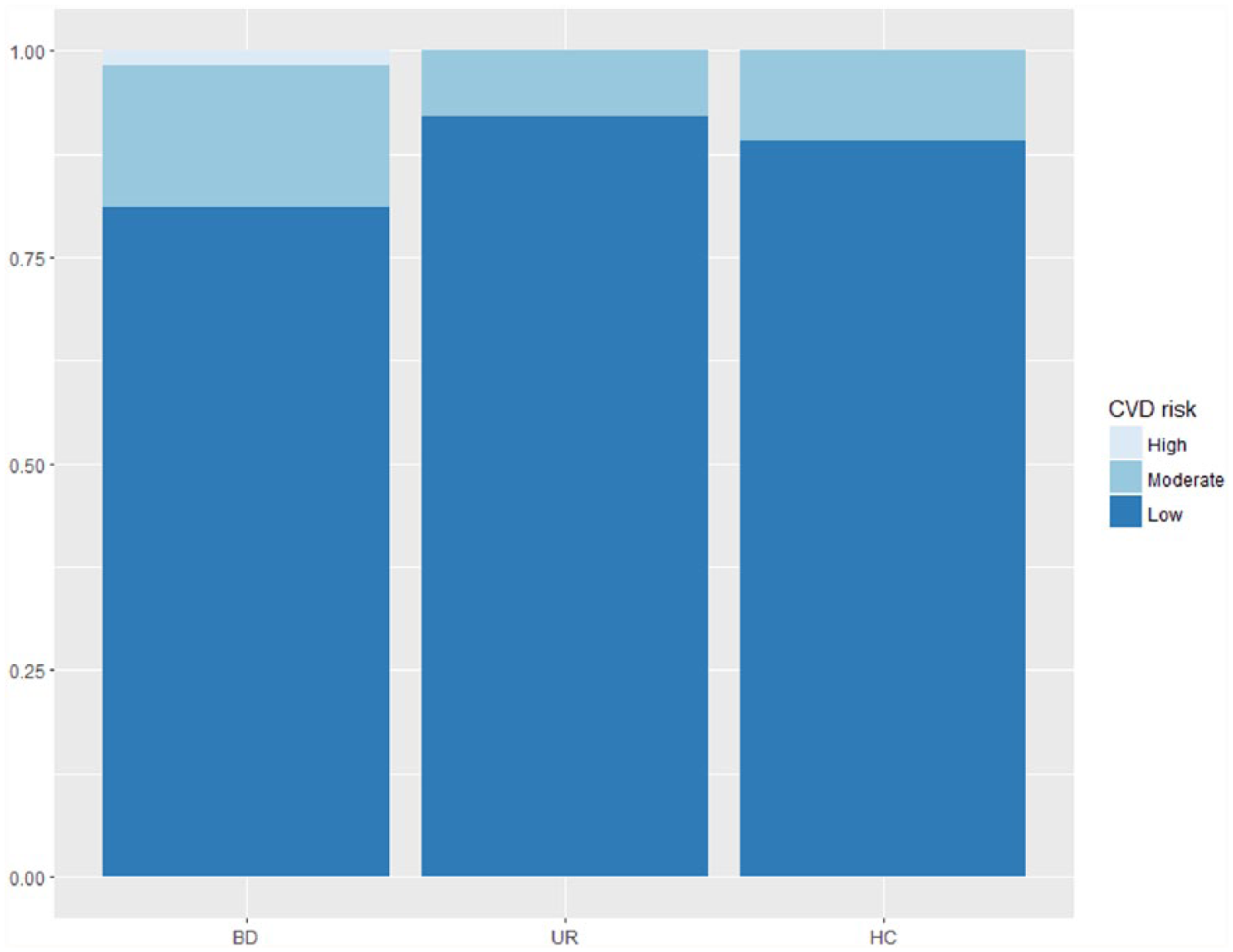
Stratification of cardiovascular disease (CVD) risk in low, intermediate and high risk in patients with newly diagnosed bipolar disorder (BD), their unaffected first-degree relatives (UR) and healthy individuals (HC).
The normalized 30-year risk score of CVD in patients with BD, their unaffected first-degree relatives and healthy individuals
The median [interquartile range] normalized 30-year risks of CVD, calculated as the difference between the 30-year ‘hard CVD’ score of the participants and the Framingham normal control group, were 0% in patients with BD, their unaffected first-generation relatives and in healthy individuals, however, with differing interquartile ranges in each group (Figure 1). There was a statistically significant increase of 8.2% in the normalized CVD risk in patients with BD (B = 1.082, 95% CI: [1.038, 1.127], p < 0.001) compared with healthy individuals, while there was no difference between unaffected first-degree relatives and healthy individuals (B = 1.033, 95% CI: [0.972, 1.099], p = 0.3). In a comparable model, there was no statistically significant difference in the normalized CVD risk between patients with BD and their unaffected first-degree relatives (B = 1.047, 95% CI: [0.989, 1.108], p = 0.1).
Association between medication and illness variables and CVD risk score in patients with newly diagnosed BD
Treatment with antiepileptics (B = 1.925, 95% CI: [1.081, 3.432], p = 0.026) and antidepressants (B = 2.501, 95% CI: [1.191, 5.259], p = 0.016) were associated with a higher CVD risk in patients with BD (Table 3). We observed a higher CVD risk score in patients with BD receiving psychotropic medication with metabolic adverse effects compared with patients not receiving psychotropic medication with metabolic adverse effects (B = 1.898, 95% CI: [1.085, 3.323], p = 0.025; Table 3). The CVD risk score did not differ between patients receiving one type of medication versus those receiving more than one type of medication (p = 0.1; Table 3).
In an unadjusted analysis, illness duration was positively associated with CVD risk (B = 1.055, 95% CI: [1.022, 1.088], p = 0.001, Table 3). To explore, whether this association was driven by age rather than illness duration, we repeated the analysis adding the covariate of age, and then, there was no longer an association between illness duration and CVD risk (p = 0.3). There was no difference in CVD risk score between patients with type I and patients with type II BD and CVD risk score was not associated with number of prior (hypo)manic episodes (p = 0.6), prior hospitalization (p = 0.5), number of suicide attempts (p = 0.9) or prior psychotic episode (p = 0.2).
Discussion
We applied, for the first time, the Framingham 30-year CVD risk tool at the initial cross-sectional assessment of a large cohort study including 221 newly diagnosed patients with BD, 50 of their unaffected first-degree relatives and 119 healthy individuals. We showed that patients with newly diagnosed BD and their unaffected first-degree relatives have increased 30-year risk of CVD. The three CVD risk factors smoking, elevated BMI and dyslipidemia were more common among patients with BD than among healthy individuals.
There are no prior studies of CVD risk in unaffected first-degree relatives of patients with BD, and CVD risk has been scarcely studied in patients with newly diagnosed BD. A small study found no difference in the Framingham 10-year risk in medication naïve young patients with newly diagnosed BD (n = 11) and healthy individuals (Taylor et al., 2010). However, in a young population, long-term risk prediction may be more useful as age and sex contribute substantially in the 10-year risk models, possibly underestimating the actual risk of CVD in younger individuals and in females (Pencina et al., 2009b), and it is possible that the calculation of a 30-year risk would have yielded findings similar to ours. Since our analyses were adjusted for age, the observed increase in CVD risk among patients with newly diagnosed/first-episode BD and in their unaffected first-degree relatives was not attributed to the age difference between groups. Furthermore, our observations of increased CVD risk in patients with newly diagnosed BD comport with results from a recent large-scale meta-analysis of CVD risk in patients with severe mental illness (Correll et al., 2017).
The normalized 30-year risk was elevated in patients with BD only. The normalized 30-year risk calculations indicated that the absolute CVD risk of our healthy individuals were comparable to the expected age- and sex-matched CVD risk of the control population in the Framingham offspring study. The latter finding may on one hand suggest that the healthy individuals recruited as blood donors from a blood bank were valid as a control group, but on the other hand the healthy individuals had higher education level, higher activity level and lower prevalence of smoking than our patients with BD and were likely healthier than the general population.
Waist circumference, a more accurate measurement of abdominal obesity than BMI (Cornier et al., 2011), was notably higher in patients with BD compared with healthy individuals. This contrasts with a recent smaller study where waist circumference in patients with newly diagnosed BD (n = 55) and healthy individuals (n = 25) did not differ (Guha et al., 2014); however, both groups had waist circumferences above the normal range. In line with prior findings of decreased levels of HDL in a study of psychotropic treated, patients with newly diagnosed BD (n = 11), we observed lower levels of HDL in patients with BD compared with healthy individuals (Taylor et al., 2010). Increased blood pressure in patients with newly diagnosed BD has previously been reported (Guha et al., 2014; Taylor et al., 2010), but surprisingly we observed lower systolic blood pressure in patients with BD relative to healthy individuals, even though our healthy individuals were blood donors and likely with a healthy lifestyle.
An American study found that 14.1% of youth with BD had an additional CVD diagnosis (Evans-Lacko et al., 2009). However, in our representative sample of participants none had manifested CVD, possibly due to regional differences in prevalence of obesity, which is higher in the United States than in Northern Europe (Afshin et al., 2017). Nonetheless, the presence of DM type 1 or 2 and hypertension in a few patients with BD and in a single unaffected first-degree relative did differ from healthy individuals, where none had any of these conditions.
Substantially more patients with BD and unaffected first-degree individuals were smokers than healthy individuals, which contributed to the differences in CVD risk scores between groups. In 2017, the smoking prevalence was 22% in the Danish general population according to the Danish Health and Medicine Authority (SST, 2017), and notably we found more than twice as many patients with BD being smokers (45.2%), whereas the proportion of smokers among unaffected relatives and healthy individuals were similar (20.4%) and lower (12.8%), respectively, than in the Danish population. When additionally including smoking as a covariate, the 30-year CVD risk score did not differ statistically significantly between patients with BD and their unaffected first-degree relatives compared with healthy individuals, indicating the role of smoking as a driver of the CVD risk in these groups.
We did not replicate the previously reported lower HDL levels in unaffected first-degree relatives (Sobczak et al., 2004). The finding of increased 30-year risk of CVD in unaffected first-degree relatives of patients with BD indicates that the CVD risk is present in individuals predisposed to BD. The co-occurrence of BD and risk factors for CVD (Amare et al., 2017) suggest overlapping genetic and/or environmental risk factors.
Strengths and limitations
Our study benefitted from including a relatively large group of well-characterized patients with newly diagnosed BD and from including a group of their unaffected first-degree relatives. Most of our participants (81%) were diagnosed within the preceding year and the maximum time with diagnosis was 7 years. In accordance with previous observations, the patients with BD in this study had a median age of illness onset of 17 years and a median delay in diagnosis of 5 years; hence, our study population appears representative of patients with newly diagnosed BD (Baldessarini et al., 2007; Kessler et al., 2007). These findings also illustrate the challenge of recruiting patients early in their course of illness due to the well-known diagnostic delay of BD (Baldessarini et al., 2007).
There are several limitations to the study. Our sample of unaffected first-degree relatives was modest, primarily caused by either first-degree relatives having a major psychiatric illness themselves or lacking consent from patients with BD to contact their relatives. Thus, low power may underlie the lack of statistically significant differences in individual risk factors between unaffected relatives and healthy individuals. Family history of CVD was not available, which would have been valuable given the known heritability of CVD (McPherson and Tybjaerg-Hansen, 2016).
The lack of association between illness duration and CVD risk in this study contrasts with previous findings of illness burden predicting CVD (Fiedorowicz et al., 2009; Goldstein et al., 2009) and may be explained by the relatively short illness duration in this cohort of patients with newly diagnosed BD. Burden of prior (hypo)manic episodes has also been associated with CVD mortality (Fiedorowicz et al., 2009); however, we did not find an association between CVD risk score and (hypo)manic load. Other illness characteristics associated with severity of illness such as prior hospitalization, suicide attempts or prior psychotic episode were not associated with CVD risk score in this study. Given the complexity in quantifying the specific individual illness burden and the correlation between age and any characteristics related to progression of the illness itself, inferences regarding the association between CVD risk and illness progression based on the present findings should be done cautiously.
Antipsychotics may increase CVD risk in patients with BD (Correll et al., 2008, 2017) and the aforementioned small study of 10-year CVD risk in newly diagnosed patients with BD found increased CVD risk scores 2 years after initiating psychopharmacological treatment (Taylor et al., 2010). In support of such findings, we found that patients using medication associated with metabolic adverse effects had a higher CVD risk score. However, while we found that use of antidepressants and antiepileptics that are both associated with weight gain was associated with a higher 30-year CVD risk score, we did not find such association for either antipsychotics or lithium, which are also associated with metabolic adverse effects. This lack of association between these medication types and CVD risk could be due to several factors. First, dichotomous treatment categories are not capturing the effect of duration of treatment. Second, patients with BD were treated with more than one type of medication in many cases, which may belie the true effect of the individual treatment categories. Therefore, our variable of ‘receiving psychotropic medication with metabolic adverse effects’ may possibly more accurately investigate the true effect of medication. Second, despite the pronounced cardiovascular adverse effects of second-generation antipsychotics (Abosi et al., 2018), treatment has been associated with lower mortality in patients with schizophrenia in two national database studies with 5-year follow-up from Sweden and Finland (Kiviniemi et al., 2013; Tiihonen et al., 2016), the latter including patients with first episode (Kiviniemi et al., 2013). Similarly, a recent study of clozapine treatment in treatment-resistant schizophrenia found that the hazard rate of all-cause mortality in patients treated with clozapine was almost halved compared with patients not treated with clozapine (Wimberley et al., 2017). Thus, the lack of association of CVD risk score and antipsychotics and lithium, respectively, may possibly reflect that these patients were well treated and possibly had a lower CVD risk than if not treated sufficiently.
The Framingham 30-year CVD risk tool was not tailored to psychiatric patients and evidence indicates that it may underestimate the CVD risk in patients with severe mental illness (Osborn et al., 2015). An alternative CVD risk prediction model in patients with severe mental illness has been proposed, integrating a range of additional factors, such as the patient’s diagnosis, social deprivation and prescriptions for antidepressants and antipsychotics (Osborn et al., 2015). Such efforts could hold promise and should be validated in independent cohorts. Finally, integrating markers of inflammation, oxidative stress, neurotrophic factors and microvascular function seems promising in understanding the link between CVD and BD (Goldstein, 2017) and will be further investigated in the ongoing longitudinal BIO study (Kessing et al., 2017).
In conclusion, we found that patients with newly diagnosed BD and their unaffected first-degree relatives have higher 30-year risks of CVD compared to healthy individuals. The findings highlight the need for interventions specifically targeting smoking and dyslipidemia in the efforts to reduce the risk of CVD in patients with BD as well as lifestyle interventions in their unaffected first-degree relatives. Further research is needed to investigate the effect of such early interventions against CVD risk factors in BD to prevent premature death and a disabling illness trajectory from the time of diagnosing BD.
Footnotes
Acknowledgements
A special thanks to Anne Præstegaard for her logistic and laboratory assistance throughout the study. L.V.K. and K.M. shared last authorship.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.C., M.F.-J., S.M., S.S., H.L.K. and K.M. declare no conflicts of interest. M.V. discloses within the last 3 years consultancy fees from Lundbeck. K.W.M. reports having received honoraria from Lundbeck and Allergan in the past 3 years. L.V.K. has within the preceding 3 years been a consultant for Sunovion. R.S.M. declares having received honoraria from Lundbeck, Pfizer, AstraZeneca, Elli-Lilly, JanssenOrtho, Purdue, Johnson & Johnson, Moksha8, Sunovion, Mitsubishi, Takeda, Forest, Otsuka, Bristoy-Myers Squibb, Shire and Allergan in the preceding 3 years.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Mental Health Services, Capital Region of Denmark (PhD student 164019 and the BIO study), The Danish Council for Independent Research, Medical Sciences (DFF-4183-00570), the A.P. Møller Foundation for the Advancement of Medical Science (15-55) and Beckett-Fonden (48282).
