Abstract
Background:
Although clozapine is the most effective medication for treatment refractory schizophrenia, only 40% of people will meet response criteria. We therefore undertook a systematic review and meta-analysis of global literature on clozapine augmentation strategies.
Methods:
We systematically reviewed PubMed, PsycInfo, Embase, Cochrane Database, Chinese Biomedical Literature Service System and China Knowledge Resource Integrated Database for randomised control trials of augmentation strategies for clozapine resistant schizophrenia. We undertook pairwise meta-analyses of within-class interventions and, where possible, frequentist mixed treatment comparisons to differentiate treatment effectiveness
Results:
We identified 46 studies of 25 interventions. On pairwise meta-analyses, the most effective augmentation agents for total psychosis symptoms were aripiprazole (standardised mean difference: 0.48; 95% confidence interval: −0.89 to −0.07) fluoxetine (standardised mean difference: 0.73; 95% confidence interval: −0.97 to −0.50) and, sodium valproate (standardised mean difference: 2.36 95% confidence interval: −3.96 to −0.75). Memantine was effective for negative symptoms (standardised mean difference: −0.56 95% confidence interval: −0.93 to −0.20). However, many of these results included poor-quality studies. Single studies of certain antipsychotics (penfluridol), antidepressants (paroxetine, duloxetine), lithium and Ginkgo biloba showed potential, while electroconvulsive therapy was highly promising. Mixed treatment comparisons were only possible for antipsychotics, and these gave similar results to the pairwise meta-analyses.
Conclusions:
On the basis of the limited data available, the best evidence is for the use of aripiprazole, fluoxetine and sodium valproate as augmentation agents for total psychosis symptoms and memantine for negative symptoms. However, these conclusions are tempered by generally short follow-up periods and poor study quality.
Introduction
Approximately 20–33% of people with schizophrenia have treatment-resistant schizophrenia (TRS) (Agid et al., 2011). TRS is defined as ongoing symptoms and functional impairment despite two adequate and adherent trials of different antipsychotics (Howes et al., 2017). In this situation, clozapine has superior efficacy when compared to first- and second-generation antipsychotics particularly in the case of positive symptoms (Siskind et al., 2016) and hospitalisations (Land et al., 2017).
Even so, only 40% of people with TRS trialled on clozapine meet clinical response criteria (Siskind et al., 2017) and, as a result, several augmentation agents have been tried. These include mood stabilisers, antidepressants and other antipsychotics, as well as electroconvulsive therapy, transcranial magnetic stimulation and cognitive behavioural therapy.
Previous systematic reviews of clozapine augmentation agents have generally focussed on one class or agent (Srisurapanont et al., 2015; Zheng et al., 2017). Where more than one was considered, studies were conducted 5 years ago (Sommer et al., 2012), were not specific to clozapine (Correll et al., 2017), were limited to English language publications (Porcelli et al., 2012), or did not focus on clozapine-refractory schizophrenia (Sommer et al., 2012; Veerman et al., 2014a, 2014b).
We therefore conducted a systematic review of randomised controlled trials of clozapine augmentation for people who failed to respond to clozapine. Importantly, we did not limit our search to English language publications.
Methods
Protocol and registration
This study was registered with PROSPERO (registration number: CRD42016033279), an international database of prospectively registered systematic reviews (Booth et al., 2012). We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) recommendations for the background, search strategy, methods, results, discussion and conclusions (Higgins and Green, 2011) including the Extension Statement for Incorporating Network Meta-analyses (Hutton et al., 2015). Ethical approval was not required as all included data had been previously published with ethical approval.
Search Strategy
The following databases were searched from inception to 24 October 2017: PubMed, Embase, PsycINFO and the Cochrane Schizophrenia Group’s Trials Register. We also searched the Chinese Biomedical Literature Service System and China Knowledge Resource Integrated Database. Hand searches of references lists in included studies and other key publications were also conducted. Studies were limited to humans. Search terms included terms for clozapine, treatment resistance and augmentation agents. PubMed search terms are provided in Supplementary Table 1.
Eligibility criteria and study selection
We included any randomised control trials that compared the following: clozapine plus an augmentation agent versus clozapine plus placebo, or clozapine plus one augmentation agent versus clozapine plus another augmentation agent. Augmentation agents could include pharmacological interventions or non-pharmacological interventions. There were no language limitations. Published data in Chinese were screened by bilingual Chinese-speakers (E.M. and Q.Z.), in French by a bilingual French-speaker (S.K.), while all other languages were translated into English. Included studies were required to specifically report that participants had ongoing psychotic symptoms unresponsive to an adequate trial of clozapine.
All studies identified in PubMed, Embase, PsycINFO and/or the Cochrane Schizophrenia Group’s Trials Register were screened at the title and abstract level by two authors (A.R. and M.L.) while those in the Chinese databases were screened by two bilingual Chinese speaking authors (E.M. and Q.Z.). All authors met to discuss inclusion criteria used in the different language databases to ensure they were consistent. Studies that met the inclusion criteria on title and abstract review, or that could not be excluded on the basis of information provided in the abstract, were reviewed at full-text level. We also performed snowball searches of reference lists from key papers and the included studies. Narrative and systematic reviews, posters, conference abstracts, case reports, letters to editors and other articles that did not meet the inclusion criteria were cross-referenced for additional potential sources of randomised controlled trials (RCTs). Attempts were made to contact corresponding authors of included studies in cases where published data were not able to be integrated into the meta-analyses.
Data collection process
Data extraction from the English language studies was conducted by two independent researchers (A.R. and M.L.), while data from the Chinese database studies were extracted by two native Chinese-speaking authors (E.M. and X.C.). All discrepancies during all stages of study selection, data extraction, and quality assessment were resolved by re-checking source papers. Data analysis was conducted by three authors (M.L., D.S. and S.K.).
Data items
Data were extracted on the following outcomes of interest: total psychosis symptom scores, (Positive and Negative Symptom Scale [PANSS]) (Kay et al., 1987), Brief Psychiatric Rating Scale (BPRS) (Overall and Gorham, 1962)); negative symptoms (Scale for the Assessment of Negative Symptoms [SANS] (Andreasen and Olsen, 1982), PANSS negative symptom subscale) and positive symptoms (Scale for the Assessment of Positive Symptoms [SAPS] (Andreasen and Olsen, 1982) and PANSS negative symptom subscale). Endpoint scores were most common, however, if endpoint scores were not provided, change scores were included. Data were extracted on trial duration, and separated into endpoints before, and after, three months. We also collected information on country, setting, diagnostic tool, mean ages and clozapine doses for intervention and control groups, number of participants in both groups, percentage of males in each group, mean dose of the augmentation agent (Table 1) and clozapine levels (Supplementary Figure 3). Data on rates of reported adverse events were also collected.
Included studies.
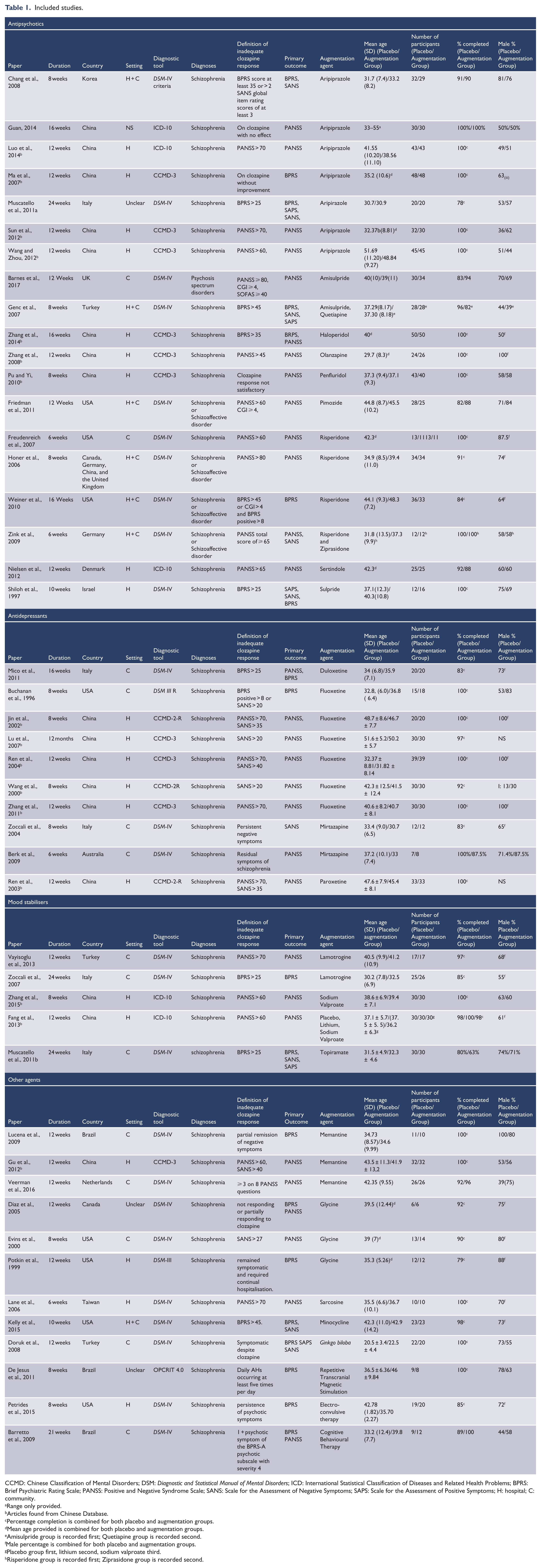
CCMD: Chinese Classification of Mental Disorders; DSM: Diagnostic and Statistical Manual of Mental Disorders; ICD: International Statistical Classification of Diseases and Related Health Problems; BPRS: Brief Psychiatric Rating Scale; PANSS: Positive and Negative Syndrome Scale; SANS: Scale for the Assessment of Negative Symptoms; SAPS: Scale for the Assessment of Positive Symptoms; H: hospital; C: community.
Range only provided.
Articles found from Chinese Database.
Percentage completion is combined for both placebo and augmentation groups.
Mean age provided is combined for both placebo and augmentation groups.
Amisulpride group is recorded first; Quetiapine group is recorded second.
Male percentage is combined for both placebo and augmentation groups.
Placebo group first, lithium second, sodium valproate third.
Risperidone group is recorded first; Ziprasidone group is recorded second.
Outcomes
Data from the last time point recorded in each study were used for all main analyses with subgroup analyses of short- and long-term studies (less or more than 3 months respectively). For cross-over studies, data from the first study phase were used. Studies reported either mean endpoint outcome and standard deviation (SD) or mean change in outcome and SD. The primary outcome was total psychotic symptoms, with secondary outcomes being positive and negative symptom subscales and adverse drug reactions (ADRs).
Study quality
This was assessed using the following criteria adapted from Cochrane Collaboration guidelines (Higgins and Green, 2011): (1) adequate generation of allocation sequence, (2) blinding of allocation to conditions to participant and/or assessor, (3) adequate random sequence generation, (4) pre-specified primary outcome measures, (5) appropriate reporting on missing data, (6) use of intention-to-treat analysis (ITT) and (7) other sources of potential bias including pharmaceutical company funding. Studies were coded low-quality if they had two or fewer elements with low bias risk, while those of high-quality had three or more elements with a low risk of bias.
Statistical analyses
We performed two types of meta-analyses. First, we used Review Manager version 5.3 for pair-wise comparisons of within-class interventions where the trials were placebo-controlled. Where possible, ITT were used. We used the standardised mean difference (SMD) for continuous data except where change and endpoint data for the same scale were combined, in which case mean differences were calculated. Risk ratios were calculated for categorical data. Subgroup and sensitivity analyses were conducted on study quality, database source, study duration (greater or less than 3 months), and whether clozapine-resistance was based on rating scales. We also compared clozapine levels, where provided (preferentially endpoint), between the intervention and control groups, with units converted to ug/L (Supplementary Figure 3).
Heterogeneity was assessed using the I2 statistic, a measure that does not depend on the number of studies in the meta-analysis and hence has greater power to detect heterogeneity when the number of studies is small. I2 provides an estimate of the percentage of variability due to heterogeneity rather than chance alone. An estimate of 50% or greater indicates possible heterogeneity, and scores of 75–100% indicate considerable heterogeneity. The I2 estimate is calculated using the chi-square statistic (Q) and its degrees of freedom. In addition to the previously described sensitivity analyses, we also explored heterogeneity through the effect of omitting each study in turn. For analyses with more than three studies, we undertook meta-regression on study quality, study duration and definition of inadequate response using Comprehensive Meta-Analysis to test for source of heterogeneity.
Second, we used MetaXL to combine direct and indirect evidence from the small number of studies where the comparison was with another active augmentation agent, not placebo. As treatment effects are fixed parameters from a frequentist perspective, they were ranked by effect size in a network forest plot rather than by the Bayesian equivalent of surface under the cumulative ranking curve (SUCRA) (Andreasen and Olsen, 1982). In practice, Bayesian and frequentist methods give similar results in network meta-analyses (Andreasen and Olsen, 1982). Inconsistency was measured using the weighted average of H, which is an adaptation of the chi-square statistic (Q) to meta-analyses that incorporate indirect comparisons. A value of less than three indicates minimal inconsistency.
Given the high rates of heterogeneity, the random effects model was used for all the analyses. We undertook comparisons between the intervention and placebo groups, comparing age, gender and dropouts. Where sufficient data existed, comparisons of ADRs between specific agents and placebo was conducted.
Publication bias
For meta-analyses that included 10 or more studies, publication bias was tested using funnel plot asymmetry where low p values suggest publication bias (Barendregt and Doi, 2016).
Results
Study selection
We identified 2228 unique articles (1718 in non-Chinese databases and 510 in Chinese databases), of which, 167 were included at title and abstract level. One additional study was identified by hand search. Forty-six full-text papers were included, of which 16 were identified in Chinese databases. These covered 25 different interventions. No studies in languages other than English and Chinese met inclusion criteria. Reasons for exclusion are listed in Figure 1 PRISMA Diagram and Supplementary Table 4 Excluded Studies. PRISMA checklist is provided in Supplementary Table 5.
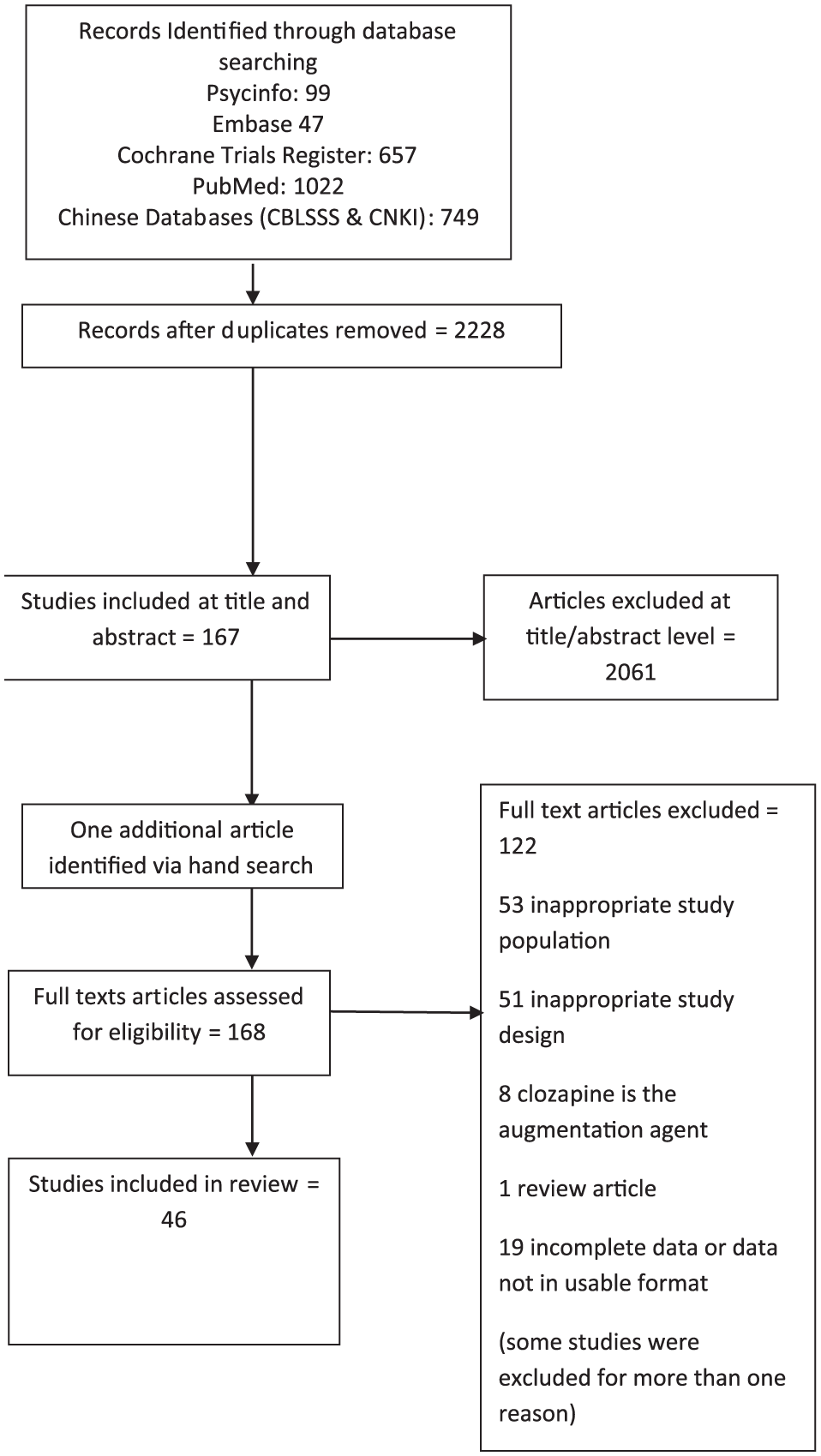
PRISMA diagram.
Study characteristics
Studies were conducted in the United States, Europe, Australia, Korea and China, predominantly in community settings (Table 1). Duration ranged from 3 weeks to 12 months while year of publication ranged from 1996 to 2017. There were 2114 study participants. Most studies were of antipsychotics (aripiprazole, risperidone, sulpride/amisulpride, sertindole, haloperidol, penfluridol, olanzapine, pimozide quetiapine and ziprasidone), followed by antidepressants (fluoxetine, paroxetine, duloxetine, mirtazepine) and mood stabilisers (sodium valproate, topiramate, lamotrigine and lithium). The remainder were of other agents (memantine, glycine, sarcosine, minocylcine and Ginkgo biloba). The three non-pharmacological interventions included cognitive behavioural therapy (CBT), electroconvulsive therapy (ECT) and transcranial magnetic stimulation (TMS). All participants had a diagnosis of clozapine-resistant schizophrenia and/or schizoaffective disorder, while psychotic symptoms were rated on one or more of the BPRS, PANSS, SANS and SAPS. Thirty-four studies used rating scales to define clozapine resistance, while the remainder reported ongoing psychotic symptoms without rating scales (Table 1).
In terms of risk of bias, all studies adequately reported randomisation, but not concealment method (Supplementary Table 2). All but one study from the non-Chinese databases were of high quality with low to moderate levels of bias. By contrast, all the studies identified in Chinese databases provided insufficient details with which to assess bias, and so were deemed to be of low-quality. Sensitivity analyses of study quality correlated strongly with a sensitivity analysis of database where study was identified.
In all the following, sensitivity analyses of the effect the effect of omitting each study in turn made little change on heterogeneity. Only aripiprazole and fluoxetine had sufficient studies to undertake meta-regression on study quality, study duration and definition of inadequate response. There were insufficient studies reporting clozapine levels to undertake a meta-regression on clozapine level.
Antipsychotic augmentation
A total of 19 studies with 1131 participants provided data on antipsychotic augmentation of clozapine. The following 10 antipsychotic agents were included in the analysis: aripiprazole (Chang et al., 2008; Guan, 2014; Luo et al., 2014; Ma et al., 2007; Muscatello et al., 2011a; Sun et al., 2012; Wang and Zhou, 2012); risperidone (Freudenreich et al., 2007; Honer et al., 2006; Weiner et al., 2010; Zink et al., 2009), haloperidol (Zhang et al., 2014); penfluridol (Pu and Yi, 2010); pimozide [Friedman et al., 2011 #136]; sulpride/amisulpride [Barnes et al., 2017; Genc et al., 2007; Shiloh et al., 1997]; sertindone (Nielsen et al., 2012), olanzapine (Zhang et al., 2008); quetiapine (Genc et al., 2007) and ziprasidone [Zink et al., 2009]). Only aripiprazole (7 studies) risperidone (3 studies) and sulpride/amisulpride (3 studies) featured in more than one study. Data were analysed across total psychosis scores and both positive and negative symptoms.
Two of the seven aripiprazole studies were of high quality (Chang et al., 2008; Muscatello et al., 2011a). Aripiprazole was statistically significantly superior to placebo for total psychosis scores but not positive or negative symptoms (Figure 2). The findings for total psychosis scores remained significant in both short- and long-term studies. However, the results were no longer significant for any of the psychosis outcomes when analyses were restricted to the higher quality studies and those that used rating scales to define clozapine resistance. Neither study quality, study duration, nor definition of inadequate response led to statistically significant results in individual variable or combined variable meta-regressions
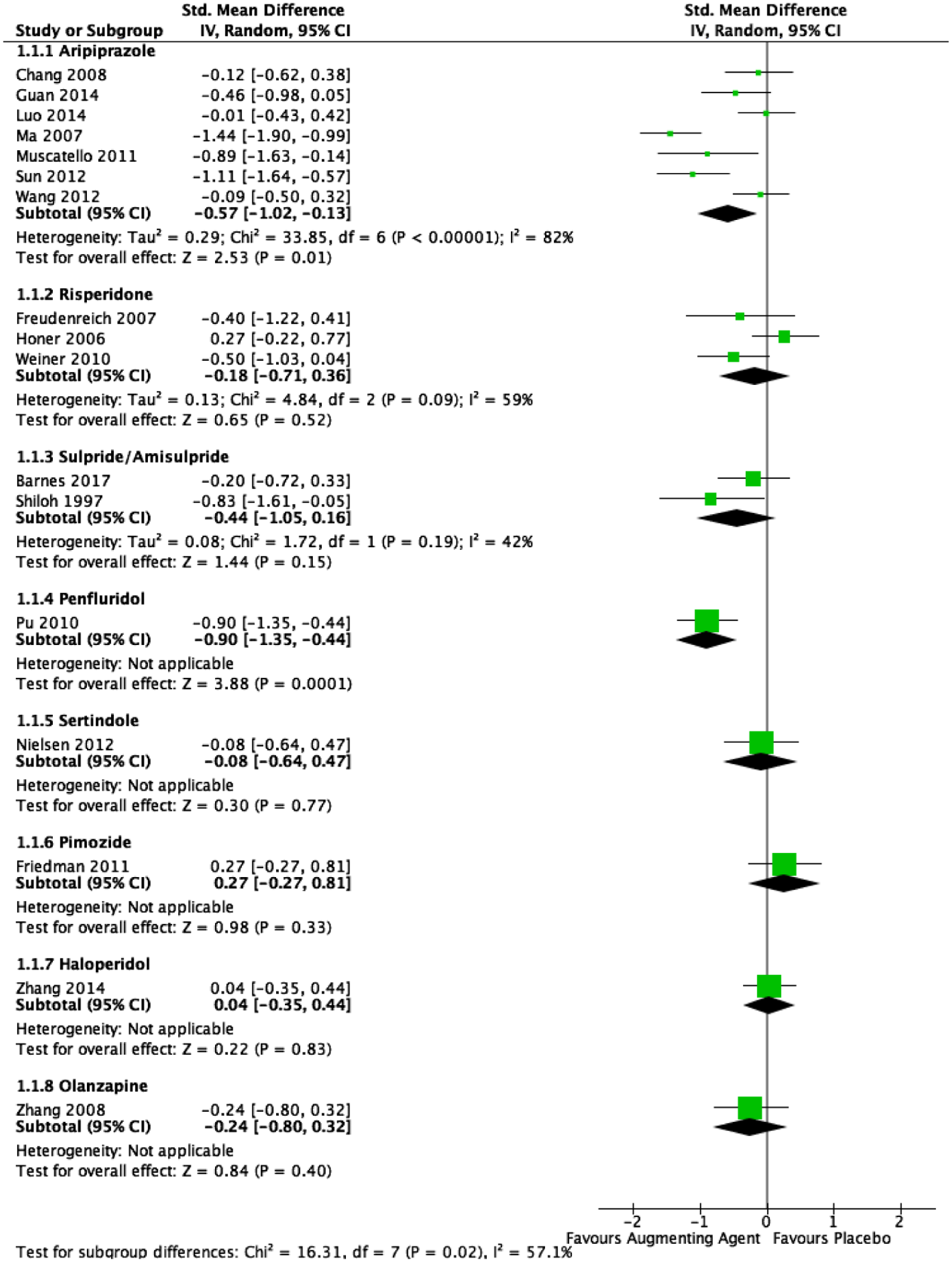
Forest plot of antipsychotics for total psychotic symptoms.
There were sufficient studies of aripiprazole to examine specific ADRs, with restlessness higher in the aripiprazole group (odds ratio [OR]: 0.05; confidence interval [CI]: 0.01 to 0.42, Z = 2.77, p = 0.006) and sedation higher in the control group (OR: 3.28; 95% CI: 1.31 to 8.19, Z = 2.55, p = 0.01), but no differences in dizziness, drooling, constipation, tachycardia, abnormal liver function tests, weight gain, hyperprolactinaemia, abnormal electrocardiography (ECG), dry mouth, insomnia or headache. Only one study provided data on clozapine levels, which were significantly higher for the aripiprazole group (167ug/L, 95% CI: 92 ug/L to 242 ug/L, p < 0.001) (Sun et al., 2012).
The three studies comparing risperidone to placebo were all of high quality (n = 144 participants) (Freudenreich et al., 2007; Honer et al., 2006; Weiner et al., 2010). There were no significant differences between risperidone and placebo across total psychosis and this was not altered in subgroup analyses of short-term or long-term studies (Figure 2). For the two studies (Honer et al., 2006; Weiner et al., 2010) that provided data on clozapine levels, there was no statistically significant difference between the risperidone and placebo groups.
Two high-quality studies of sulpride/amisulpride compared to placebo did not show any significant results (Barnes et al., 2017; Shiloh et al., 1997), nor did the one high-quality study of each of sertindole (Nielsen et al., 2012) or pimozide (Friedman et al., 2011). One low-quality study of penfluridol (n = 83) showed a significant reduction in total psychosis scores (Pu and Yi, 2010) (Figure 2). Augmentation with either haloperidol (Zhang et al., 2014), olanzapine (Zhang et al., 2008) did not significantly alter total and positive symptoms, although the one low-quality study of olanzapine showed a statistically significant improvement in negative symptom scores (Figure 2). There were no significant differences in clozapine levels between olanzapine and placebo groups (Supplementary Figure 3).
Antipsychotic medications were the only intervention class where indirect evidence was available. One low-quality study compared quetiapline with amisulpiride (Genc et al., 2007), while the other high-quality study compared ziprasidone with risperidone (Zink et al., 2009). Supplementary Figure 1 displays the geometric distribution of the mixed treatment comparisons while Supplementary Figure 2 ranks the treatments by efficacy. Only augmentation with aripiprazole or penfluridol was statistically superior to placebo for total psychosis scores, with non-significant results for the other medications including both ziprasidone and quetiapine (Supplementary Table 3). The weighted average of H was 1.
Antidepressant augmentation
There were 10 studies (n = 476 participants) of antidepressants as augmentation agents (Table 2) including six of fluoxetine (Buchanan et al., 1996; Jin et al., 2002; Lu et al., 2007; Ren et al., 2004; Wang et al., 2000; Zhang et al., 2011), and one each of paroxetine (Ren et al., 2003), duloxetine (Mico et al., 2011) and mirtazapine (Berk et al., 2009; Zoccali et al., 2004).
All agents and effect sizes.
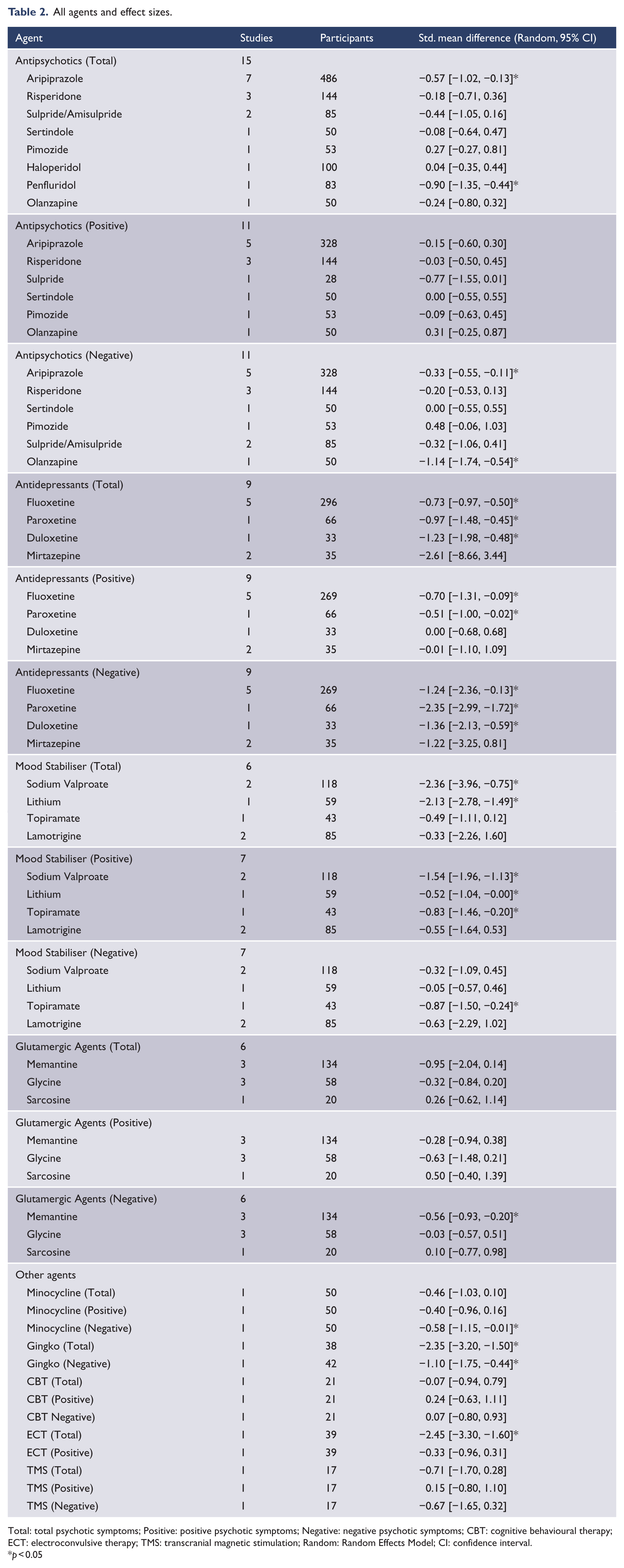
Total: total psychotic symptoms; Positive: positive psychotic symptoms; Negative: negative psychotic symptoms; CBT: cognitive behavioural therapy; ECT: electroconvulsive therapy; TMS: transcranial magnetic stimulation; Random: Random Effects Model; CI: confidence interval.
p < 0.05
Fluoxetine had one high (Buchanan et al., 1996), and five low-quality studies (Jin et al., 2002; Lu et al., 2007; Ren et al., 2004; Wang et al., 2000; Zhang et al., 2011) with 331 participants. This analysis showed a statistically significant improvement for total psychosis, positive and negative symptoms. However, when the low-quality studies were excluded, the results for positive and negative symptoms were non-significant. Three studies provided endpoint clozapine levels, with no statistically significant difference between the fluoxetine and placebo groups (Jin et al., 2002; Lu et al., 2007; Wang et al., 2000). There were insufficient differences between the studies to run meta-regressions on study quality, study duration or definition of inadequate response.
There were two high-quality studies for mirtazapine with 35 participants, but results for total psychosis, positive symptom and negative symptom scores were non-significant (Berk et al., 2009; Zoccali et al., 2004). One low-quality study of paroxetine showed a reduction in total psychosis, positive symptom and negative symptom scores, although the endpoint clozapine levels were significantly higher in the paroxetine group (129ug/L 95% CI 36 ug/L to 222 ug/L, p = 0.006) (Ren et al., 2004). For duloxetine, one high-quality study with 33 participants showed a reduction in total psychosis and negative scores but not positive symptoms (Mico et al., 2011).
Mood stabiliser augmentation
We found five studies of mood stabilisers as augmentation agents with a total of 278 participants (Table 2). These were of sodium valproate (Fang et al., 2013; Zhang et al., 2015), lithium (Fang et al., 2013), topiramate (Muscatello et al., 2011b) and lamotrigine (Vayisoglu et al., 2013; Zoccali et al., 2007). Two low-quality studies of sodium valproate with 118 participants showed a reduction in total psychosis and positive scores but not negative symptoms (Fang et al., 2013; Zhang et al., 2015). One low-quality study of lithium with 60 participants showed a reduction in total psychosis, positive symptom, but not negative symptom scores (Fang et al., 2013). The one high-quality study of topiramate showed improvements on positive and negative symptoms but not total scores (Muscatello et al., 2011b). The two high-quality studies of lamotrigine, showed no improvement for total psychosis, positive symptom nor negative symptom scores. One of the lamotrigine studies provided data on clozapine levels, with no difference between the lamotrigine and placebo groups (Vayisoglu et al., 2013).
Other augmentation agents
Seven studies included glutamatergic agents (Table 2): Three high-quality studies of glycine (Diaz et al., 2005; Evins et al., 2000; Potkin et al., 1999) with 58 participants showed no statistically significant difference in total, positive or negative symptoms. These results remained the same when restricted to the five studies that used rating scales to define clozapine resistance. One study provided clozapine levels, with no difference between the glycine and placebo groups (Diaz et al., 2005).
Three studies of memantine (n = 134 participants), two of high (Lucena et al., 2009; Veerman et al., 2016) and one of low-quality (Gu et al., 2012), with showed improvements in negative symptoms, but not total or positive scores (Table 2). These findings were unaltered in sensitivity analyses by study quality. However, none of these results were significant when restricted to the two studies that used rating scales to define clozapine resistance. One high-quality study provided data on clozapine levels, with levels higher in the memantine group (139u/L 95% CI: 19 to 259, p = 0.02) (Veerman et al., 2016), though, of note, this was the only study that did not show results favouring memantine.
One high-quality study of sarcosine (n = 20) found no statistically significant differences for any of the psychiatric outcomes (Table 2) (Lane et al., 2006). By contrast, another high-quality study of Gingko biloba (n = 38) showed a reduction in total psychosis and negative symptom scores (Table 2; Doruk et al., 2008).
Finally, one high-quality minocycline (n = 50) showed significant reductions in negative symptoms, but not total or positive scores (Table 2). There was no difference in clozapine levels between the groups (Kelly et al., 2015).
In terms of non-pharmacological interventions, one high-quality study of ECT (n = 39) reported a significant reduction in total psychosis scores (Petrides et al., 2015), but there were no significant differences in single studies of CBT (Barretto et al., 2009) or TMS (De Jesus et al., 2011) (Table 2).
Differences between groups
When comparisons were made between the intervention and placebo groups, there was no statistical difference in gender, age or dropouts. For specific ADRs, sufficient data existed only for aripiprazole, and are presented above.
Publication bias
We were unable to assess for publication bias as no analyses included 10 or more studies.
Discussion
This study provides the first comprehensive literature review of pharmacological and non-pharmacological augmentation in clozapine-refractory schizophrenia. A key strength was the inclusion of studies from China as well as the use of indirect comparisons that allowed the inclusion of two additional antipsychotic agents. We were therefore able to include 46 studies with 2223 participants examining 25 agents. Unlike previous meta-analyses, we focused on studies of clozapine-refractory schizophrenia, the clinical situation when augmentation would most likely to be considered. The only previous meta-analyses that considered all types of augmentation agents was published 5 years ago, not limited to participants with clozapine resistance and only included 29 studies with 15 different augmentation agents (Sommer et al., 2012).
The most effective augmentation agents for total psychosis symptoms were aripiprazole, fluoxetine and sodium valproate. Single studies of certain antipsychotics (penfluridol), antidepressants (paroxetine, duloxetine), lithium and Ginkgo biloba showed promising effects, although these preliminary findings require replication.
Aripiprazole, a partial 5-HT1A agonist with partial agonist effects on dopamine D2 S, D2 L and D3 receptors may have complementary neuro-receptor actions to clozapine (Chang et al., 2008; Muscatello et al., 2011a), although the favourable results for this agent were tempered by the poor quality of the majority of studies. When these poor-quality studies were excluded, the results were non-significant. Remaining studies that did not quantify clozapine-resistance had the same effect. Finally, only one study reporting clozapine levels confirmed that they were higher in the aripiprazole group.
In the case of antidepressants, the effects on negative symptoms could be partly explained by the nature of negative symptom rating scales. These may conflate primary negative symptoms including expressive deficits and avolition with secondary negative symptoms related to other factors including depressive symptoms (Carpenter and Buchanan, 2017). In addition, the effects of some antidepressants, such as fluoxetine and paroxetine, may be mediated by an increase in clozapine levels through inhibition of cytochrome CYP2D6. Support for this theory comes from the single study of paroxetine where there were higher clozapine levels in the paroxetine group. On the other hand, there were no differences in endpoint levels between fluoxetine and placebo groups in the three studies where this was reported. In addition, the favourable results for paroxetine and fluoxetine generally came from studies of low quality, while the negative findings for mirtazapine were from a high-quality study. All studies, irrespective of quality, found similar results for total psychosis and negative symptom scores.
Among mood stabilisers, only sodium valproate and lithium improved total psychosis symptoms, although all of these studies were of low-quality. One high-quality study of topiramate showed efficacy for positive and negative symptoms, while the high-quality studies of lamotrigine were non-significant.
Memantine was effective for negative symptoms. This is an NMDA receptor agonist, which when combined with clozapine, may act synergistically on NMDA receptors, thereby modulating glutamatergic activity with improvements in cognition and negative symptoms (Veerman et al., 2016). Similar effects have been reported with memantine augmentation of other antipsychotics (Zheng et al., 2018), although further studies of memantine for negative symptoms among people on clozapine are required to validate these results.
In relation to non-pharmacological treatments, the one small high-quality study of ECT showed very promising results for total psychosis scores, in keeping with evidence from non-randomised trials (Lally et al., 2016).
One strength of our search methodology was the inclusion of studies from Chinese databases (Cohen et al., 2015). This reduces the risk of selection bias. However, this is also a weakness as studies from Chinese databases were, overall, of low-quality. Generally, these studies provided limited information on methodology to allow assessment of bias with the possibility of an overestimate of the efficacy of the agents studied. As such, we would encourage clinicians to exercise caution in interpreting results, notably related to aripiprazole and fluoxetine. Studies were generally of short duration, with only nine running for longer than 12 weeks. Few studies reported clozapine levels at baseline and/or endpoint between the intervention and control groups. Differences between groups in terms of endpoint clozapine levels or change in clozapine levels could potentially confound the results (e.g. paroxetine).
Many of our results also showed heterogeneity. We attempted to explore heterogeneity through subgroup and sensitivity analyses and meta-regression. While we have used a random effects model throughout to incorporate heterogeneity into our analysis, our results should still be treated with caution.
Conclusion
Given that only 40% of people with TRS trialled on clozapine will meet response criteria (Siskind et al., 2017), effective augmentation strategies for clozapine are essential. The most effective strategies for clozapine augmentation appear to be aripiprazole, fluoxetine and sodium valproate, with promising results from ECT. Memantine may be beneficial for negative symptoms. To minimised polypharmacy, clozapine levels should be optimised by increasing clozapine dose prior to adding an augmentation agent, unless there are compelling reasons to do otherwise. However, if augmentation is ineffective, and quality of life, functional and psychosis symptoms are not better than with other antipsychotics used prior to commencement of clozapine, consideration should be given to cessation of clozapine and switching to an antipsychotic with fewer side-effects.
Supplemental Material
ANP772351_Supplementary_Figure_1 – Supplemental material for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis
Supplemental material, ANP772351_Supplementary_Figure_1 for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis by Dan J Siskind, Michael Lee, Arul Ravindran, Qichen Zhang, Evelyn Ma, Balaji Motamarri and Steve Kisely in Australian & New Zealand Journal of Psychiatry
Supplemental Material
ANZ772351_Supplementary_Figure_2 – Supplemental material for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis
Supplemental material, ANZ772351_Supplementary_Figure_2 for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis by Dan J Siskind, Michael Lee, Arul Ravindran, Qichen Zhang, Evelyn Ma, Balaji Motamarri and Steve Kisely in Australian & New Zealand Journal of Psychiatry
Supplemental Material
ANZ772351_Supplementary_Figure_3 – Supplemental material for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis
Supplemental material, ANZ772351_Supplementary_Figure_3 for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis by Dan J Siskind, Michael Lee, Arul Ravindran, Qichen Zhang, Evelyn Ma, Balaji Motamarri and Steve Kisely in Australian & New Zealand Journal of Psychiatry
Supplemental Material
ANZ772351_Supplementary_Table_1 – Supplemental material for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis
Supplemental material, ANZ772351_Supplementary_Table_1 for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis by Dan J Siskind, Michael Lee, Arul Ravindran, Qichen Zhang, Evelyn Ma, Balaji Motamarri and Steve Kisely in Australian & New Zealand Journal of Psychiatry
Supplemental Material
ANZ772351_Supplementary_Table_2 – Supplemental material for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis
Supplemental material, ANZ772351_Supplementary_Table_2 for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis by Dan J Siskind, Michael Lee, Arul Ravindran, Qichen Zhang, Evelyn Ma, Balaji Motamarri and Steve Kisely in Australian & New Zealand Journal of Psychiatry
Supplemental Material
ANZ772351_Supplementary_Table_3 – Supplemental material for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis
Supplemental material, ANZ772351_Supplementary_Table_3 for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis by Dan J Siskind, Michael Lee, Arul Ravindran, Qichen Zhang, Evelyn Ma, Balaji Motamarri and Steve Kisely in Australian & New Zealand Journal of Psychiatry
Supplemental Material
ANZ772351_Supplementary_Table_4 – Supplemental material for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis
Supplemental material, ANZ772351_Supplementary_Table_4 for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis by Dan J Siskind, Michael Lee, Arul Ravindran, Qichen Zhang, Evelyn Ma, Balaji Motamarri and Steve Kisely in Australian & New Zealand Journal of Psychiatry
Supplemental Material
ANZ772351_Supplementary_Table_5 – Supplemental material for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis
Supplemental material, ANZ772351_Supplementary_Table_5 for Augmentation strategies for clozapine refractory schizophrenia: A systematic review and meta-analysis by Dan J Siskind, Michael Lee, Arul Ravindran, Qichen Zhang, Evelyn Ma, Balaji Motamarri and Steve Kisely in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
We thank Michael Berk and Seetal Dodd for providing data for inclusion.
Declaration of Conflicting Interests
D.S. is partially supported by an NHMRC ECF APP1111136. The authors have no other declarations.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
