Abstract
Objective:
There is growing evidence confirming increased inflammation in a subset of adults with depression. The impact of this relationship has mostly been considered in biologically based interventions; however, it also has potential implications for psychological therapies. Cognitive behaviour therapy is the most commonly used psychological intervention for the treatment of depression with theories around its efficacy primarily based on psychological mechanisms. However, cognitive behaviour therapy may have an effect on, and its efficacy influenced by, physiological processes associated with depression. Accordingly, the purpose of this systematic review was to examine the relationship between cognitive behaviour therapy and inflammation.
Method:
Studies examining the anti-inflammatory effects of cognitive behaviour therapy in people with depression and other medical conditions (e.g. cancer, diabetes and heart disease) were examined. In addition, the relationship between change in inflammatory markers and change in depressive symptoms following cognitive behaviour therapy, and the influence of pre-treatment inflammation on cognitive behaviour therapy treatment response were reviewed.
Results:
A total of 23 studies investigating the anti-inflammatory effects of cognitive behaviour therapy were identified. In 14 of these studies, at least one reduction in an inflammatory marker was reported, increases were identified in three studies and no change was found in six studies. Three studies examined the relationship between change in inflammation and change in depressive symptoms following cognitive behaviour therapy. In two of these studies, change in depressive symptoms was associated with a change in at least one inflammatory marker. Finally, three studies examined the influence of pre-treatment inflammation on treatment outcome from cognitive behaviour therapy, and all indicated a poorer treatment response in people with higher premorbid inflammation.
Conclusion:
Preliminary evidence suggests inflammation should be considered within the context of cognitive behaviour therapy, although robust studies examining the relationship are sparse, and heterogeneity between studies and populations examined was high. The potential treatment implications of the bi-directional relationship between inflammation and cognitive behaviour therapy are discussed, and recommendations for future research are proposed.
Introduction
Cognitive behaviour therapy (CBT) is the most commonly implemented and investigated psychological treatment for major depressive disorder. It is an effective treatment for mild-to-moderate depression, with outcomes often comparable to antidepressant medications (Cuijpers et al., 2013; Malhi et al., 2015). CBT is based on the premise that negative thoughts and unhelpful belief systems are a major cause of depression. Therefore, through the use of several cognitive and behavioural techniques, clients are taught to refute or modify unhelpful thoughts and coping patterns. Although CBT is an effective intervention, cognitive changes are not necessarily the primary reason for symptomatic improvements in depression. For example, the degree of symptom reduction among patients participating in CBT was found to be related to change in patients’ acquisition of coping skills rather than reduced endorsement of implicitly assessed maladaptive beliefs (Adler et al., 2015). In a recent meta-analysis, it was also confirmed that while CBT had an influence on dysfunctional thoughts, its effects on dysfunctional thinking were not significantly different compared to other psychotherapies or pharmacotherapy (Cristea et al., 2015). A common element across all therapies is the development of a therapeutic alliance, the strength of which is positively associated with treatment outcome, and possible cognitive changes across all therapies (Ardito and Rabellino, 2011; Malhi et al., 2015). CBT, despite being classified as psychological intervention, may also influence physiological processes relevant to depression. This has received limited attention, but could potentially be an additional mechanism behind its efficacy.
Increasing research has confirmed that there is a relationship between depression and inflammation. Findings from several meta-analyses have confirmed that compared to healthy counterparts, depression is associated with chronic low-grade inflammation as indicated by greater concentrations of C-reactive protein (CRP), interleukin-6 (IL-6) and tumour necrosis factor-α (TNF-α) (Haapakoski et al., 2015; Howren et al., 2009; Liu et al., 2012; Valkanova et al., 2013). These findings have contributed to the development of the cytokine/inflammatory hypothesis of depression whereby chronic, low-grade inflammation is speculated as playing a role in the establishment, progression and maintenance of depression (Raison et al., 2006; Schiepers et al., 2005). While this subclinical inflammation is not a characteristic of all depressed patients, it may be pertinent for a subset of patients (Raison and Miller, 2011). Inflammation has particular relevance as it can influence many physiological disturbances implicated in depression such as neurotransmitter activity, hypothalamus–pituitary–adrenal (HPA) axis activity, oxidative and nitrosative stress, mitochondrial activity and neuroprogression (Maes et al., 2011; Moylan et al., 2013).
The causes of this subclinical inflammation are considered multifactorial, comprising biological, psychological, lifestyle and social factors. From a psychological perspective, current and past stress and trauma can influence the immune response. For example, a history of childhood abuse and trauma is a risk factor for higher inflammation (Rooks et al., 2012). Acute and chronic stressors also influence the inflammatory response (Calcia et al., 2016). There are several physiological mechanistic effects of stress on inflammation, as stress and corresponding elevated cortisol can impair central and peripheral nervous system feedback leading to a state of ‘glucocorticoid resistance’ (Rodriguez et al., 2016), may impair mitochondrial function (Gong et al., 2011) and can influence the gut microbiome (Rogers et al., 2016), which are all able to influence the immune response. Eating an unhealthy diet, reduced physical activity and poor sleep is also associated with elevated inflammation (Berk et al., 2013; Lopresti et al., 2013). From a biological standpoint, obesity and suffering from medical conditions such as diabetes, cardiovascular disease, digestive disorders and autoimmune diseases are also associated with increased inflammation (Petra et al., 2015; Ruparelia et al., 2016; Schwartz et al., 2016). Depression has a high comorbidity with these medical conditions (Kang et al., 2015).
The aim of this article is to provide a comprehensive, systematic review of the relationship between CBT and inflammation. In particular, evidence of the anti-inflammatory effects of CBT in people with depression and other medical conditions will be examined. In addition, the relationship between change in inflammatory markers and change in depressive symptoms following CBT will be reviewed. As there have been findings suggesting reduced therapeutic efficacy from pharmaceutical antidepressants in people with higher inflammation (Strawbridge et al., 2015), the influence of pre-treatment inflammation on CBT outcomes will also be examined.
Methods
Search strategy
Information for this review was compiled by searching PubMed, Google Scholar, PsycINFO and The Cochrane Library databases, and by examining reference lists of relevant papers to locate additional studies that were not identified by the database searches. Databases were scanned from all years of study until December 2016.
Search strategy and eligibility criteria
A systematic search of trials using the terms ‘cognitive-behaviour therapy’, ‘cognitive-behavior therapy’, ‘CBT’, ‘psychotherapy’, ‘inflammation’, ‘CRP’, ‘cytokine’, ‘interleukin’ and ‘immunity’ was completed. Specific inclusion criteria included the following: (1) published in English, (2) human populations, (3) investigated the relationship between CBT and markers associated with inflammation and immune response and (4) published in peer-reviewed journals.
Results
Search results
Based on the systematic search of studies, a total of 159 studies were identified. In all, 82 studies were excluded because there was no pre- or post-measurement of inflammatory biomarkers. A total of 52 studies were review articles, did not examine the specific application of CBT or did not investigate the relationship between CBT and inflammatory markers. In all, 25 studies met all inclusion criteria and were included in this review. In Figure 1, the process of inclusion of studies in this review is detailed.
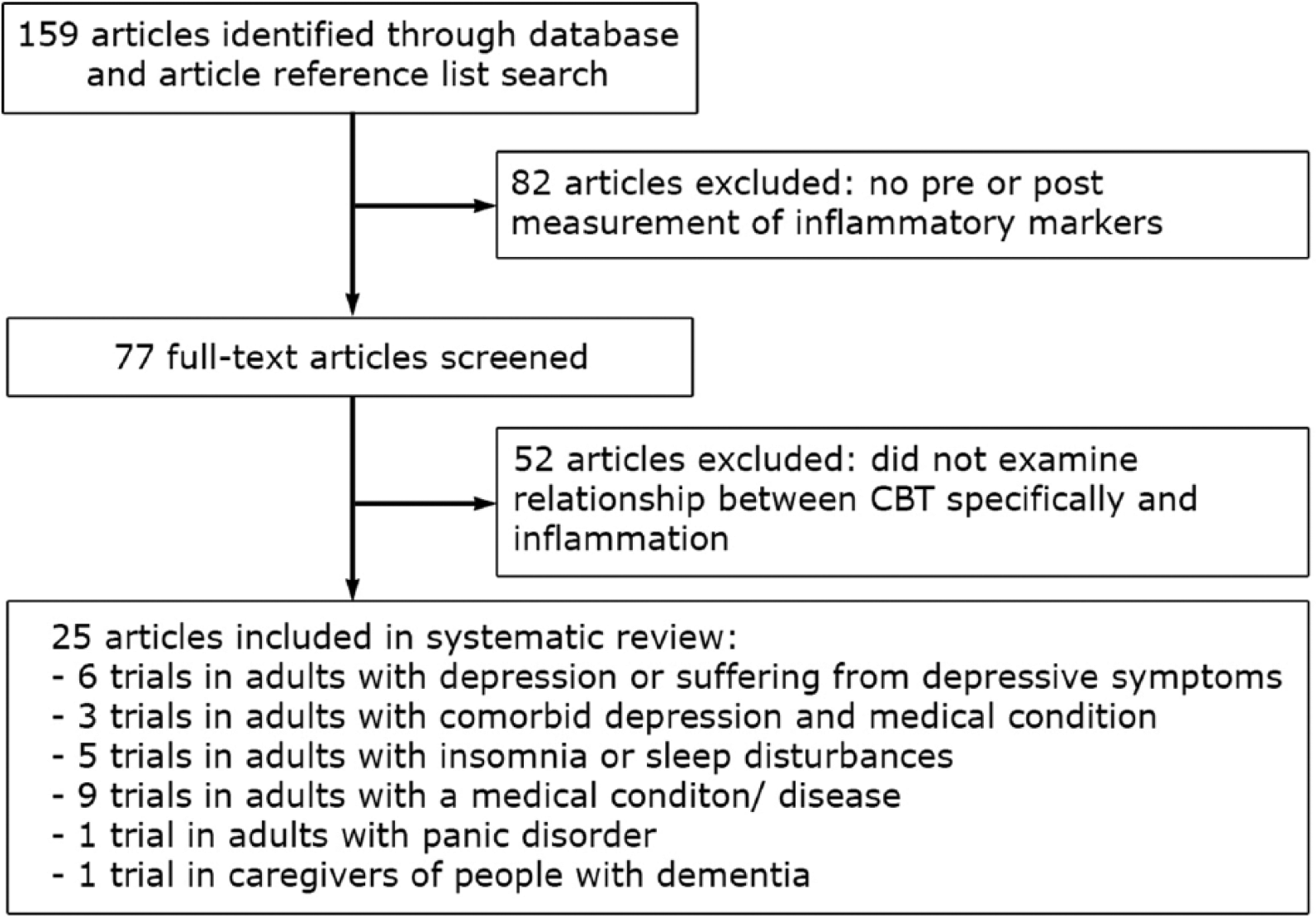
Process of study inclusion in review.
A meta-analytic analysis was not conducted as there was significant variability in study designs, many not including a control condition. There was also significant variability in the inflammatory markers measured and sample populations examined.
CBT and its effect on inflammation
As summarised in Table 1, 23 studies were identified examining changes in inflammatory or immune markers following CBT. Populations examined included four studies on people suffering from major depressive disorder or depressive symptoms, three studies on adults with depression or depressive symptoms and a comorbid medical condition (breast cancer, coronary artery bypass and unspecified chronic medical condition), five studies on adults with insomnia or sleep disturbances (three included populations with a comorbid medical disease comprising one with breast cancer and two with kidney disease), nine studies on adults suffering from a medical condition/disease (comprising ischaemic heart disease, rheumatoid arthritis, breast cancer, diabetes, osteoarthritis, chronic pain, irritable bowel disease), one study on adults with panic disorder and one study on caregivers of adults with dementia.
Summary of studies examining CBT and its effect on inflammation.
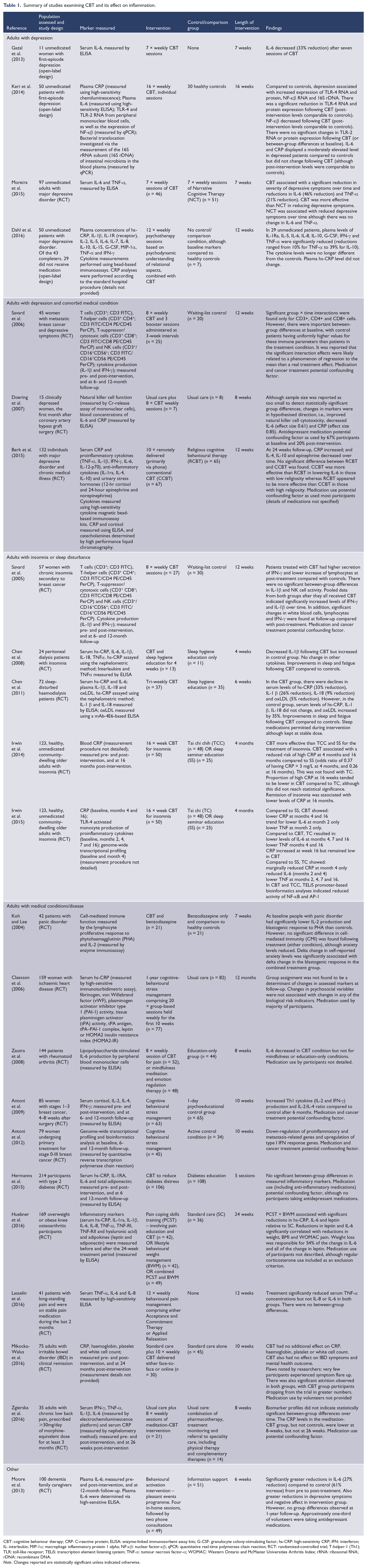
CBT: cognitive behaviour therapy; CRP: C-reactive protein; ELISA: enzyme-linked immunosorbent assay kits; G-CSF: granulocyte colony-stimulating factor; hs-CRP: high-sensitivity CRP; IFN: interferon; IL: interleukin; MIP-1α: macrophage inflammatory protein 1 alpha; NF-κβ: nuclear factor-κβ; qPCR: quantitative real-time polymerase chain reaction; RCT: randomised-controlled trial; T-helper-1 (Th1); TLR: toll-like receptor; TELiS: transcription element listening system; TNF-α: tumour necrosis factor-α; WOMAC: Western Ontario and McMaster Universities Arthritis Index; rRNA: ribosomal RNA; rDNA: recombinant DNA.
Note. Changes reported are statistically significant unless indicated otherwise.
The robustness of findings varied significantly as some studies were not randomised, blinded and/or did not compare inflammatory changes to a control or placebo group. In fact, three of the four studies conducted on depressed only populations utilised open-label designs. CBT duration also varied from 6 weeks to 12 months, and oftentimes, medication use was a potential confounding factor. Moreover, inflammatory markers examined varied across studies, with CRP, IL-6 and TNF-α the most common markers assessed.
From the identified 23 studies, 14 reported at least one reduction in an inflammatory marker. Increases were reported in three studies (two studies in women with breast cancer), and no changes were found in six studies. In the four studies that examined the effects of CBT on unmedicated adults with depression, reductions in at least one inflammatory marker were found in all of them. For example, women with first-episode depression experienced reductions in IL-6 after seven sessions of CBT (Gazal et al., 2013); however, changes were not compared to a control condition. CBT provided to adults with depression was also associated with reductions in IL-6 and TNF-α after seven weekly sessions. In this study, CBT was more effective than narrative cognitive therapy (NCT) in reducing depressive symptoms, and no change in IL-6 or TNF-α was found following NCT (Moreira et al., 2015). Participation in 16 weekly, individual CBT sessions in adults with first-episode depression was associated with reductions in toll-like receptor-4 (TLR-4) signalling, but no change in TLR-2 signalling, IL-6 and CRP levels (Keri et al., 2014). An examination of changes in immune markers in unmedicated adults with depression following 12 weeks of CBT revealed reductions in plasma levels of IL-1Ra, IL-5, IL-6, IL-8, IL-10, granulocyte-colony stimulating factor, interferon-γ (IFN-γ) and TNF-α (Dahl et al., 2016). However, changes were not compared to a control. In adults with depression and a comorbid medical disease (i.e. breast cancer, cardiovascular disease or unspecified chronic medical illness), changes in inflammatory markers were more variable as no change was found in one study, increased immune response in another and decreases in a third. Medication use was a potential confounding factor in all three of these studies, as a significant portion of participants were taking some form of medication (including antidepressants) during the study.
The relationship between clinical improvement from CBT and change in inflammation
Although the bulk of evidence suggests that reduced inflammation can occur following CBT, this does not confirm that reduced inflammation is necessary for the alleviation of depressive symptoms. This has received little attention in research, and only three studies (detailed in Table 2) were identified examining the relationship between change in inflammation and change in depressive symptoms. In unmedicated women suffering from first-episode depression, Gazal et al. (2013) found that after 7 weeks of CBT, a reduction in IL-6 was positively correlated with a reduction in depressive symptoms. Similar findings were revealed after 16 weeks of CBT in unmedicated adults with first-episode depression. More pronounced reductions in TLR-4 and nuclear factor-κβ RNA expression were associated with better clinical improvement (Keri et al., 2014). However, in a final study, although 7 weeks of CBT in depressed, unmedicated adults was associated with a reduction in depressive symptoms, IL-6 and TNF-α, there was no significant correlation between remission of depressive symptoms and change in the inflammatory markers (Moreira et al., 2015).
Summary of studies examining relationship between clinical improvement following CBT and change in inflammation.
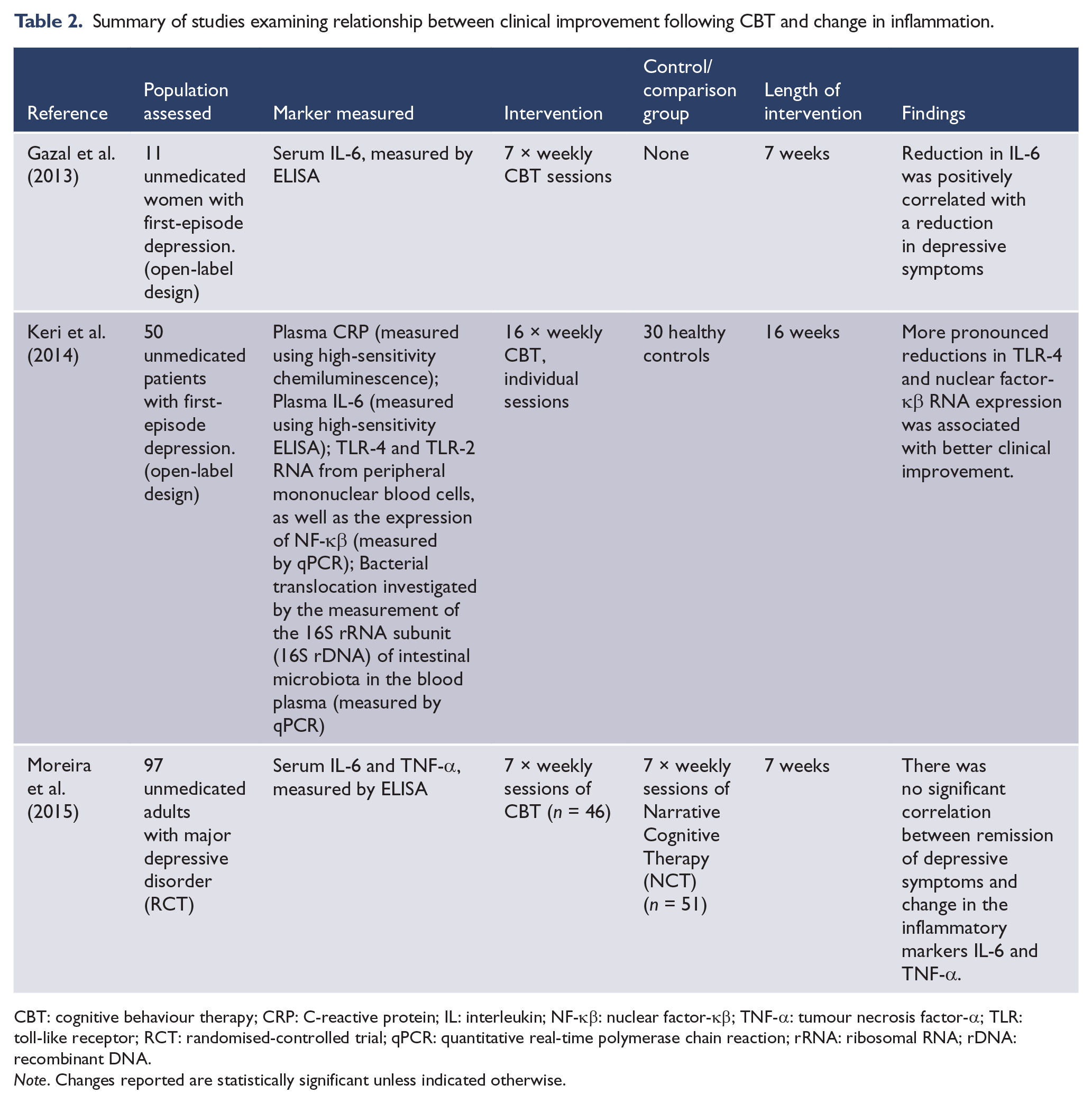
CBT: cognitive behaviour therapy; CRP: C-reactive protein; IL: interleukin; NF-κβ: nuclear factor-κβ; TNF-α: tumour necrosis factor-α; TLR: toll-like receptor; RCT: randomised-controlled trial; qPCR: quantitative real-time polymerase chain reaction; rRNA: ribosomal RNA; rDNA: recombinant DNA.
Note. Changes reported are statistically significant unless indicated otherwise.
Inflammation and treatment response from CBT
Despite the generally positive influence of CBT on inflammation in people with depression, three studies were identified confirming poorer treatment response in people with higher premorbid inflammation (Table 3). Harley et al. (2010) treated depressed adults with 8–19 sessions of either interpersonal therapy or CBT. They found that patients with elevated pre-treatment CRP (n = 21) experienced poorer symptomatic improvement than people with lower CRP (n = 147). Patients with a baseline CRP ⩾ 10 mg/L experienced an average 36.3% reduction in depressive symptoms compared to 55.1% in people with lower baseline CRP. In another study, patients with long-standing pain and on stable pain medication were provided 12 weekly sessions of behavioural pain management comprising either Acceptance and Commitment Therapy or applied relaxation. Although treatment was associated with reductions in TNF-α, it was revealed that higher pre-treatment levels of TNF-α and IL-6 were associated with less improvement in pain intensity, psychological inflexibility and mental health-related quality of life (Lasselin et al., 2016). Finally, in patients with type 1 diabetes and major depression, 12 weeks of diabetes-specific group CBT was less effective in reducing depressive symptoms in patients with higher CRP concentrations. However, no such influence was identified in people with type 2 diabetes (Zahn et al., 2016).
Summary of studies examining relationship between inflammation and CBT treatment response.
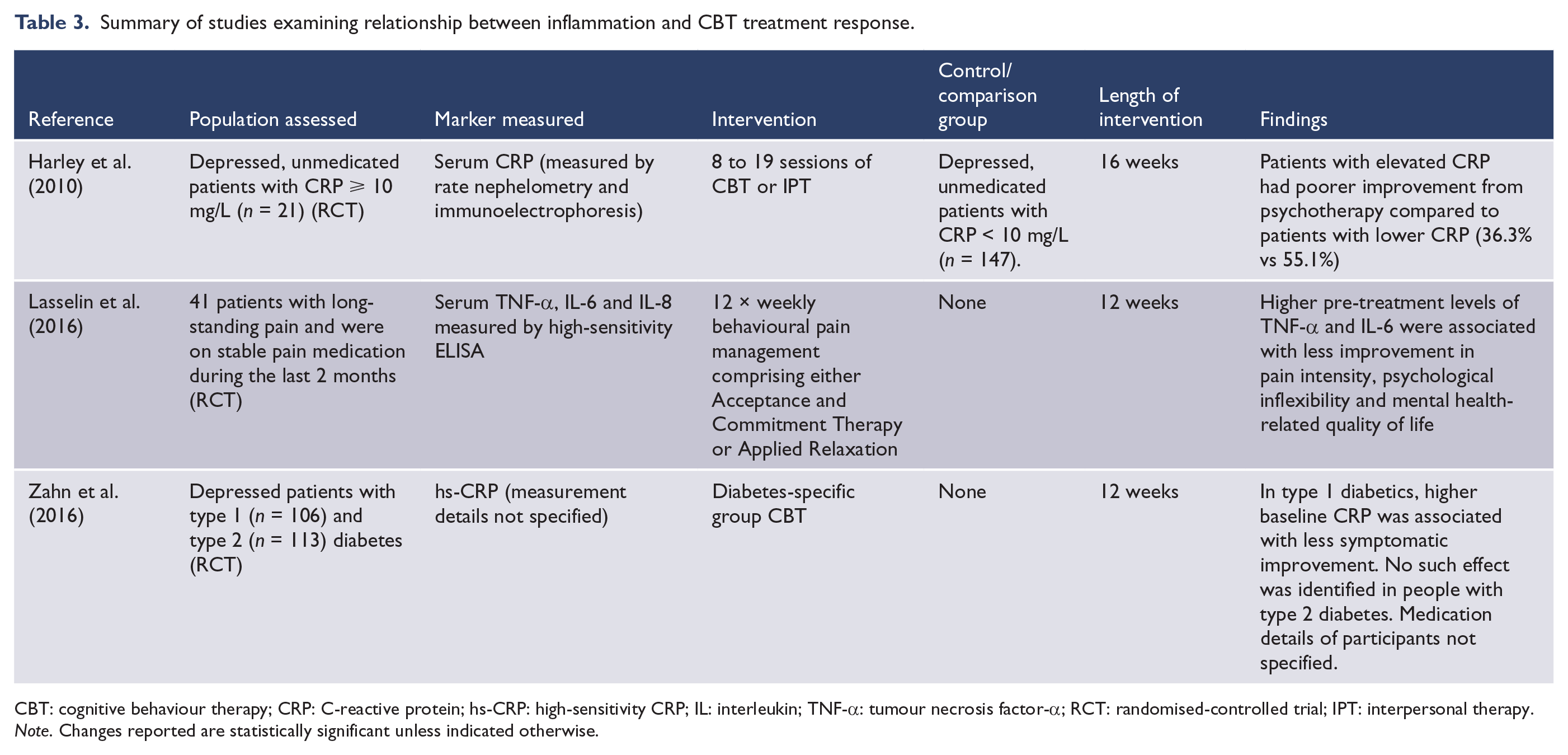
CBT: cognitive behaviour therapy; CRP: C-reactive protein; hs-CRP: high-sensitivity CRP; IL: interleukin; TNF-α: tumour necrosis factor-α; RCT: randomised-controlled trial; IPT: interpersonal therapy.
Note. Changes reported are statistically significant unless indicated otherwise.
Discussion
Anti-inflammatory effects of CBT
From this systematic review, the bulk of evidence suggests that CBT may influence inflammatory processes. Although findings were not entirely consistent, results from 14 out of 23 studies demonstrated that CBT was associated with a reduction in inflammation over time. Pertaining to the four trials examining the effects of CBT in adults with depression, there was confirmation in all these studies that CBT was associated with a reduction in at least one inflammatory marker over time (Dahl et al., 2016; Gazal et al., 2013; Keri et al., 2014; Moreira et al., 2015). These reductions are likely considered clinically meaningful as reductions in biomarkers ranged from 10% to 39%. However, interpretation of these findings is moderated by several important factors.
In the reviewed CBT studies, change in inflammation was examined over time; however, study designs did not always include a placebo condition or comparison group. Therefore, it is plausible that reduced inflammation may simply be an artefact of the passage of time and/or may also occur with non-CBT-based psychological interventions. It is also possible that improvements in mood lower inflammation rather than vice versa. Controlling for or analysing the influence of potential confounding variables is, therefore, essential. These include medication use (particularly ones that can influence inflammation), obesity, exercise, sleep, diet, blood sampling collection procedure (e.g. fasting), levels of baseline inflammation and medical illness progression over time.
The bulk of studies included in this review examined the effects of CBT in people with medical diseases comprising breast cancer, heart disease, rheumatoid arthritis, diabetes, irritable bowel disease and chronic pain. These diseases are all associated with an increased inflammatory/immune response, and at levels typically much higher than the subclinical elevations found in patients with depression. Consequently, the lack of anti-inflammatory effect from CBT found in some of the studies may be a characteristic of the population examined. The anti-inflammatory influences of CBT could be more potent in people with mildly elevated inflammation rather than clinical levels that occur with many of these medical conditions. This possibility is supported by the positive anti-inflammatory findings following CBT in all seven studies that only included populations without an existing medical disease (i.e. depression, insomnia and caregivers of adults with dementia).
Moreover, there was also a lack of consistency in inflammatory markers measured across studies. Identifying the most pertinent inflammatory markers remains uncertain, although CRP, IL-6 and TNF-α were the most commonly analysed peripheral markers. The markers measured to assess anti-inflammatory changes following CBT also need to be cautiously interpreted as some were proinflammatory cytokines which may have a direct and causal effect on depression (e.g. IL-6, TNF-α), while others were anti-inflammatory and may be upregulated to reflect a response to (or to counter-regulate) proinflammatory or other stimuli.
Finally, the treatment length and crucial elements of CBT for greatest anti-inflammatory effects have not yet been examined. The length of CBT treatments varied considerably, and there was an array of strategies covered in CBT that could account for its potential anti-inflammatory influences. It is possible that CBT instigates positive lifestyle changes that, in turn, lower inflammation. For example, improved sleep which is confirmed to have anti-inflammatory effects is one plausible explanation for the changes (Irwin et al., 2016). The coaching of several relaxation techniques and engagement in pleasurable activities are also encouraged in CBT. Relaxation practice is confirmed to have anti-inflammatory effects (Kang et al., 2011; Laudenslager et al., 2016), and in one study, greater daily participation in positive activities was protective against inflammation (Sin et al., 2015). CBT can also reduce rumination, which is a common problem in depression (Manicavasagar et al., 2012). Interestingly, it was demonstrated in one study that engaging in rumination after a laboratory speech stressor increased concentrations of CRP compared to distraction (Zoccola et al., 2014). Reappraisal (i.e. altering how to think about an emotionally provoking situation in order to change its emotional impact) was also associated with significantly lower CRP, while emotional suppression was associated with significantly higher CRP (Appleton et al., 2013). These skills are regularly targeted in CBT. Longer term CBT interventions may also be associated with reduced body weight and increased glycaemic control, particularly in patients with diabetes and obesity. Such changes are associated with inflammatory reductions and therefore could account for the reduced inflammation in patients over time (Nicklas et al., 2005; Nowlin et al., 2012). In order to enhance our understanding of the impact of CBT on inflammation, further randomised, robust studies are essential. In particular, they need to control for the aforementioned factors.
Reduced inflammation and symptomatic improvement
Three studies were identified examining the relationship between change in inflammation and alleviation of depressive symptoms following CBT. In two studies, a reduction in inflammation was associated with improvements in depressive symptoms (Gazal et al., 2013; Keri et al., 2014), while in the third, no such relationship was identified (Moreira et al., 2015). The significance of inflammatory changes on symptomatic improvement following CBT requires further investigation. In particular, if inflammation is lowered by CBT, it is unknown whether it is simply an artefact of the intervention, having no major influence on symptomatic relief, or is an essential element for recovery from depression. If the latter is the case, then measurement of inflammatory biomarkers present as potential, objective markers of treatment outcome. Davis et al. (2015) have defined such a biomarker as a ‘state or acuity biomarker’. Currently, treatment efficacy for depression is primarily assessed via clinician-rated and self-report instruments, and there is no validated, objective, state biomarker of treatment outcome or disease process associated with psychological interventions.
Pharmaceutical antidepressants have been demonstrated to have anti-inflammatory effects, with a meta-analysis confirming their ability to lower levels of IL-1β and IL-6 (Hannestad et al., 2011). This has prompted some researchers to postulate that reduced inflammation may be a potential mechanism behind their antidepressant influence, and therefore may be important for symptomatic relief. However, it is possible that reduced inflammation is simply a result of treatment with an anti-inflammatory agent and has little to do with treatment response. CBT poses as a better tool to study state-related changes in biomarkers and the value of inflammation as a prognostic marker than antidepressant medications where it is difficult to tease out drug effects on illness processes from the direct effects of medications on inflammation.
Inflammation and CBT treatment response
Although CBT is an effective treatment for depression, there remain a significant portion of patients who either receive no benefit or only achieve a partial response. Of additional concern is the finding from a recent meta-analysis that showed modern CBT clinical trials were providing less relief from depressive symptoms compared with seminal trials (Johnsen and Friborg, 2015). Tools to identify CBT treatment-responders prior to treatment allocation would be invaluable. This may enhance patient-treatment matching or could support decision-making around the need for adjunct or more intensive interventions. Only three studies were identified that examined the influence of premorbid inflammation on treatment outcome (defined as a ‘prognostic biomarker’ by Davis et al., 2015). In all of these studies, higher pre-treatment inflammation was associated with a reduced treatment response from CBT (Harley et al., 2010; Lasselin et al., 2016; Zahn et al., 2016). This is an area that requires further investigation and could be a relatively simple addition to CBT and depression research. However, identifying the most valuable inflammatory prognostic biomarker, or collection of markers, requires examination. CRP, IL-6 and TNF-α present as promising peripheral options. Moreover, genetic variants involved in neurobiological pathways associated with both immune activation and depression hold some promise, such as functional allelic variants of genes for IL-1β, TNF-α and CRP, as well as single nucleotide polymorphisms in the IL-1β, IL-6 and IL-11 genes (Bufalino et al., 2013).
Causes for this reduced treatment efficacy in people with higher inflammation are unknown although several possibilities are speculated. First, inflammation can have detrimental effects on memory, attention and learning (Talarowska et al., 2014). These may be important cognitive skills necessary for the effective adoption of CBT techniques. However, this hypothesis is not supported by previous findings which confirmed cognitive ability has little influence on treatment outcome in CBT (Spangler et al., 1997). Second, a common symptom of inflammation is fatigue (Karshikoff et al., 2017). It is possible that people with higher inflammation and therefore greater fatigue may obtain reduced benefit from CBT, particularly as strategies covered in CBT require practice, and regular homework assignment is common. A third possibility is demonstrated by a group of studies where healthy participants were exposed to the inflammatory endotoxin, Escherichia coli. This endotoxin increased blood concentrations of IL-6 and TNF-α, and also heightened feelings of social disconnection (Eisenberger et al., 2010), impaired social cognitive processing (Moieni et al., 2015) and increased neural sensitivity to social rejection (Eisenberger et al., 2009). Moreover, in a study by Muscatell et al. (2016), it was demonstrated that exposure to the inflammatory endotoxin heightened neural activity (as measured by functional magnetic resonance imaging) to both negative and positive feedback in areas of the brain associated with threat (i.e. bilateral amygdala, dorsal anterior cingulate cortex) and reward (i.e. ventral striatum and ventromedial prefrontal cortex). Although these experimental procedures (which trigger acute, clinical inflammation) do not replicate the subclinical, chronic inflammation seen in depression, they potentially provide an indication of the possible effects of inflammation on cognitive and neural activity. It has been argued that inflammation, and its corresponding cognitive changes, may have an evolutionary survival function (Miller and Raison, 2015); however, these changes in cognitive sensitivities could reduce the therapeutic potency of CBT. To determine the influence of subclinical inflammation on cognitive processes, experimental manipulations comprising low-grade inflammation require investigation. Fourth, it is possible that inflammation per se may not necessarily be a cause of poorer CBT treatment response but is simply an artefact of factors that influence inflammation. These include poor sleep, an unhealthy diet, reduced physical activity, obesity, chronic stress or suffering from a comorbid medical condition (Berk et al., 2013; Lopresti et al., 2013).
If higher inflammation is a prognostic marker of poorer response to CBT, then investigations into the comparative efficacy of alternative or adjunct interventions need examination. It is possible that the use of both CBT and pharmacological treatments may be required for patients with elevated inflammation. Combined interventions may be especially pertinent as it was also demonstrated that elevated inflammation was associated with reduced clinical response to antidepressant medications (Strawbridge et al., 2015). By combining the potential stress-reducing skills covered in CBT, the neurotransmitter effects of antidepressants and the anti-inflammatory influences of both treatments, a greater treatment response may be realised in this subgroup of patients. Other options to consider include lifestyle-based changes targeting sleep, diet and exercise, which all have anti-inflammatory effects (Berk et al., 2013; Lopresti et al., 2013). Psychological treatments, including relaxation training, meditation/mindfulness, yoga and even imagery can have anti-inflammatory influences (Black and Slavich, 2016; Bower and Irwin, 2016; Eremin et al., 2009; Laudenslager et al., 2016). Pharmaceutical anti-inflammatories such as nonsteroidal anti-inflammatory drugs and cytokine antagonists present as other additional adjunct options (Raison, 2016), although their long-term administration is cautioned against given their potential for gastrointestinal and cardiovascular adverse effects (Scarpignato et al., 2015). Natural supplements with antidepressant, immune-modulating and other potential therapeutic mechanisms of action (e.g. via reducing oxidative stress, supporting mitochondrial function or influencing neurotransmitter activity) such as omega-3 fatty acids (Mazza et al., 2015), curcumin (Lopresti et al., 2012), saffron (Lopresti and Drummond, 2014), n-acetylcysteine (Fernandes et al., 2016) and probiotics (Pirbaglou et al., 2016) could also be considered for CBT treatment resistant patients, and/or those presenting with greater pre-treatment inflammation. Such adjunct options are particularly pertinent in patients presenting with ‘pro-inflammatory’ lifestyle, dietary, environmental and medical risk factors. For example, sleep interventions for people with insomnia, dietary changes for patients with poor dietary habits, exercise for physically inactive individuals and supplementation such as probiotics for patients with a history of extensive antibiotic use or presenting with digestive disturbances or diseases.
Finally, although the purpose of this systematic review was to examine the effects of CBT on inflammation, there remain several other potential physiological mechanisms of actions. These include effects on HPA axis activity, neurotransmitter activity, neuroprotection and neurotropic action, and even oxidative stress. All these mechanisms can be influenced by inflammation, and thereby a reduction in inflammation can have a positive influence on these biological processes (Moylan et al., 2013). However, it is also plausible that CBT can have direct, independent effects on these mechanisms.
Given the paucity of studies conducted to date, examining the effects of other psychological therapies (e.g. behaviour therapy, client-centred therapy, psychodynamic therapy) on inflammation and other physiological mechanisms, and comparing their anti-inflammatory effects to CBT could not be undertaken. Further investigation into the relationship of CBT and other therapies on inflammation would be beneficial to determine whether changes are unique to CBT or occur across all therapies where a therapeutic alliance is a crucial ingredient to treatment success.
In closing, findings from this systematic review suggest there may be a bi-directional influence between CBT and inflammation, whereby CBT has anti-inflammatory effects, and inflammation, particularly in a subset of patients with elevated inflammation, influences the treatment response to CBT. However, there are several mediating variables that could account for this relationship and further research in this area is certainly warranted. The significant heterogeneity of studies reviewed and populations examined also significantly temper any conclusions that can be made about the relationship between CBT and inflammation. However, it is hoped that this review highlights the potential benefits of examining the inter-relationship between CBT (and other psychological therapies) on physiological processes implicated in depression.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
