Abstract
Background:
Abnormal functional connectivity has been observed in major depressive disorder. Anatomical distance may affect functional connectivity in patients with major depressive disorder. However, whether and how anatomical distance affects functional connectivity at rest remains unclear in drug-naive patients with major depressive disorder.
Methods:
Forty-four patients with major depressive disorder, as well as 44 age-, sex- and education-matched healthy controls, underwent resting-state functional magnetic resonance imaging scanning. Regional functional connectivity strength was calculated for each voxel in the whole brain, which was further divided into short- and long-range functional connectivity strength.
Results:
The patients showed decreased long-range positive functional connectivity strength in the right inferior parietal lobule, as well as decreased short-range positive functional connectivity strength in the right insula and right superior temporal gyrus relative to those of the controls. No significant correlations existed between abnormal functional connectivity strength and the clinical variables of the patients.
Conclusion:
The findings revealed that anatomical distance decreases long- and short-range functional connectivity strength in patients with major depressive disorder, which may underlie the neurobiology of major depressive disorder.
Keywords
Introduction
As a common mental illness, major depressive disorder (MDD) is the leading contributor to the global disease burden (Wang et al., 2012). Efforts are underway to develop new antidepressants; however, MDD continues to affect patients and their families at the individual and societal levels. The exact neurobiology underlying MDD remains poorly understood.
Functional neuroimaging studies have demonstrated that MDD is an illness characterized by disturbances of brain networks rather than dysfunction of a single brain region (Drevets et al., 2008; Sheline, 2000). Several brain networks such as the limbic-cortical-striatal-pallidal-thalamic network (Drevets et al., 2008; Marchand, 2010; Sheline, 2000), cortico-limbic-cerebellar network (including the fronto-limbic network) (Drevets et al., 2008; Pizzagalli, 2011) and default-mode network (DMN) (Graham et al., 2013; Guo et al., 2014) were advanced to explain the neurobiology of MDD.
DMN is among the most examined networks, and it encompasses large-scale brain regions, such as the medial prefrontal cortex (MPFC), posterior cingulate cortex (PCC)/precuneus, as well as the medial, lateral and inferior parietal cortices (Raichle et al., 2001). Abnormal functional connectivity (FC) of DMN has been observed in MDD with inconsistent results. Hamilton et al. (2011b), Sheline et al. (2010) and Zhou et al. (2010) found enhanced FC of DMN in MDD. A resting-state FC study using independent component analysis revealed increased FC in the thalamic and subgenual cingulate regions of the DMN of depressed patients (Greicius et al., 2007). By contrast, several studies detected decreased FC of DMN in MDD (Veer et al., 2010), such as reduced FC between bilateral caudate and PCC/precuneus (Bluhm et al., 2009). Interestingly, both increased and decreased FCs have been reported in first-episode (Zhu et al., 2011) and late-life depressed individuals (Wu et al., 2011). These abnormalities correlate with depressive symptoms (rumination), suggesting that these abnormalities exert clinical implications (Hamilton et al., 2011a; Zhu et al., 2011).
Other brain networks have also been implicated in the neurobiology of MDD. Decreased frontal and increased limbic FCs of the fronto-limbic network have been reported in functional neuroimaging studies (Wang et al., 2012). We recently observed decreased regional activity and network homogeneity in the frontal regions of the fronto-limbic network in MDD (Guo et al., 2015b). Abnormal insular connectivity with the brain regions of the fronto-limbic network, hate circuit and visual areas has also been found in MDD (Guo et al., 2015a; Perlman et al., 2012).
The above-mentioned studies provide substantial evidence that MDD is an illness characterized by disturbances of brain networks. Nevertheless, the effect of structural distance on FC in MDD remains unclear. An efficient human brain depends on both long- and short-range FC. Long-range FC runs at higher metabolic and time costs (Bullmore and Sporns, 2012; Liang et al., 2013), whereas short-range FC runs at lower metabolic and time costs and exhibits enhanced functional connectivity strength (FCS) (Salvador et al., 2005). However, the brain prefers endorsing long-range FCs to ensure efficient communication despite their high metabolic and time costs (Sepulcre et al., 2010).
In this study, we examined the whole-brain FCs in a group of drug-naive patients with MDD. Specifically, the FCs were divided into long- and short-range FCs according to their structural distance (Achard et al., 2006; He et al., 2007). We hypothesized that anatomical distance would affect FC in patients with MDD. Hence, the patients were expected to exhibit abnormal long- and short-range FCs. On the basis of the above-mentioned findings, the brain regions of the DMN were expected to be affected in MDD. We also investigated whether possible alterations in short- and long-range FCSs correlate with clinical variables.
Materials and methods
Participants
Forty-four right-handed outpatients with MDD were recruited from the Mental Health Center, the First Affiliated Hospital, Guangxi Medical University, China, and 44 right-handed healthy controls were recruited from the community. The patients and the controls were group-matched by age, sex and education. The patients were diagnosed using the Structured Clinical Interview of the DSM-IV (SCID), patient edition (First et al., 1997). The patients were identified as drug-naive with current episode duration of less than 6 months to exclude potential confounding effects from medication use and long illness duration of the current episode. The depressive symptom severity was assessed using the Hamilton Rating Scale for Depression (HRSD, 17 items) (Hamilton, 1967), and the patients scored more than 18 in this scale. The Automatic Thoughts Questionnaire (ATQ, 30 items) (Harrell and Ryon, 1983) was used to assess the severity of automatic thoughts in the patients, which has been validated in a Chinese population (Cao et al., 2001). Healthy controls were screened by the SCID, non-patient edition (First et al., 1997). The participants were screened using the following exclusion criteria: other Axis I disorders such as schizophrenia, bipolar disorder, substance-induced mood disorder, anxiety disorders, substance abuse or dependence, acute physical illness and a history of head injury resulting in loss of consciousness. The controls reported no psychiatric disorders among their first-degree relatives.
All participants provided their written informed consent to participate in the study. The local Ethics Committee of the First Affiliated Hospital of Guangxi Medical University approved this study.
Imaging acquisition
Images were acquired on a Siemens 3T scanner. Participants were directed to remain motionless and awake but with their eyes closed. Soft earplugs and foam pads were applied to attenuate scanner noise and head motion. The following parameters with a gradient-echo echo-planar imaging (EPI) sequence were used to acquire resting-state functional images: repetition time/echo time (TR/TE) = 2000/30 ms, 30 slices, 64 × 64 matrix, 90° flip angle, 24 cm field of view (FOV), 4 mm slice thickness, 0.4 mm gap and 250 volumes (500 s).
Data preprocessing
Data Processing Assistant for resting-state functional magnetic resonance imaging (fMRI) (Yan and Zang, 2010) was applied to preprocess the imaging data in MATLAB (Mathworks). The data were corrected for slice timing and head motion. No participants had more than 2 mm of maximal translation and more than 2° of maximal rotation. Several spurious covariates were removed, including 24 head motion parameters obtained by rigid body correction, the signal from a ventricular region-of-interest (ROI) and the signal from a region centered in the white matter. The global signal was saved in preprocessing the FC data as previously suggested (Hahamy et al., 2014). The data were then normalized to the standard Montreal Neurological Institute (MNI) EPI space in SPM8 and resampled to 3 mm × 3 mm × 3 mm. The obtained data were subsequently smoothed with a 4-mm full width at half maximum Gaussian kernel, bandpass-filtered (0.01–0.08 Hz) and linearly detrended.
Long- and short-range FCS analysis
The preprocessed data were analyzed using whole-brain FC analysis. Pearson’s correlation coefficients were calculated between the time series of one voxel and the other voxels within a gray matter mask, thereby obtaining a whole-brain FC matrix for each participant (Liu et al., 2015). The matrices were then z-score transformed using Fisher z-transformation to improve normality. The regional FCS for a given voxel was calculated as the sum of the connectivities (z values) between this voxel and all other voxels. The regional FCS was divided into long- and short-range FCS to examine the effects of structural distance on FC findings. The long-range FCS of a given voxel was defined as the sum of the connectivities (z values) between this voxel and other voxels with structural distances greater than 75 mm, whereas the short-range FCS of a voxel was defined as the sum of the connectivities (z values) with structural distances less than 75 mm (Achard et al., 2006; He et al., 2007). In this study, the structural distances between two voxels referred to the Euclidean distance between their MNI coordinates. Four categories of FCS were identified: long-range positive/negative FCS (lpFCS and lnFCS) and short-range positive/negative FCS (spFCS and snFCS). After testing for normality of data, voxel-wise two-sample t-tests were conducted to compare the FCS differences between the patients and the controls with the software REST (Song et al., 2011). Framewise displacement (FD) values were calculated for each participant (Power et al., 2012), and the mean FD was applied as a covariate in the group comparisons. The significance level was set at p < 0.001, which was corrected for multiple comparisons at the cluster level using the theory of Gaussian random fields (GRF) (min z > 3.2905, cluster significance: p < 0.001).
Correlation analysis
Voxel-based correlations were correspondingly performed between abnormal FCS values of each patient and the clinical variables. The obtained correlation maps were GRF-corrected at p < 0.05 (min z > 1.96, cluster significance: p < 0.05).
Results
Characteristics of the participants
The characteristics of the participants are presented in Table 1. No significant difference in FD values was indicated between the patients and the controls.
Characteristics of the participants.
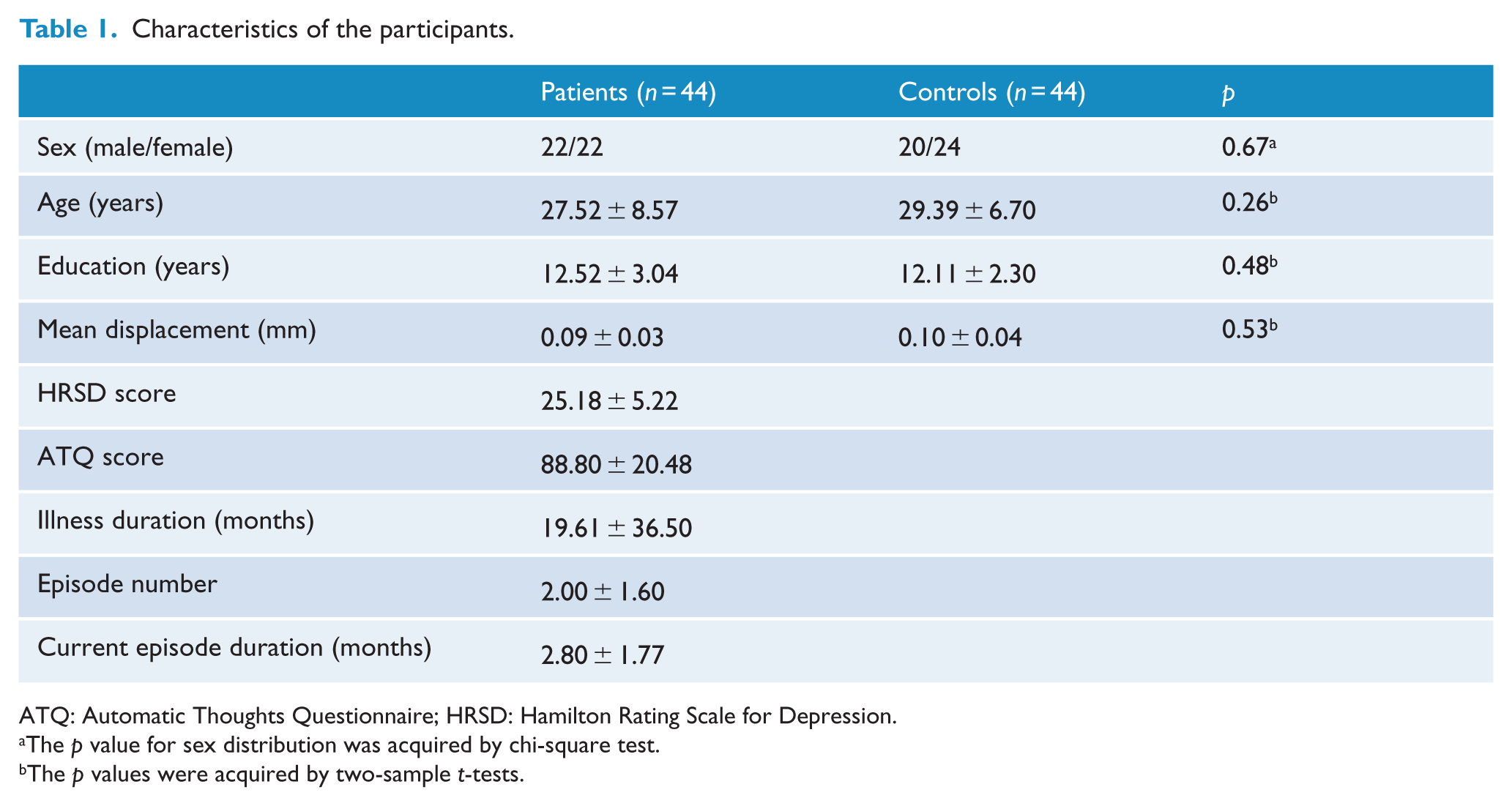
ATQ: Automatic Thoughts Questionnaire; HRSD: Hamilton Rating Scale for Depression.
The p value for sex distribution was acquired by chi-square test.
The p values were acquired by two-sample t-tests.
FCS differences between groups
Compared with the controls, the patients exhibited decreased lpFCS in the right inferior parietal lobule (IPL) (Table 2 and Figure 1) and decreased spFCS in the right insula and right superior temporal gyrus (STG) (Table 2 and Figure 2). No significant differences between the groups were identified in lnFCS and snFCS.
Regions with increased long-range and short-range connectivity in patients.
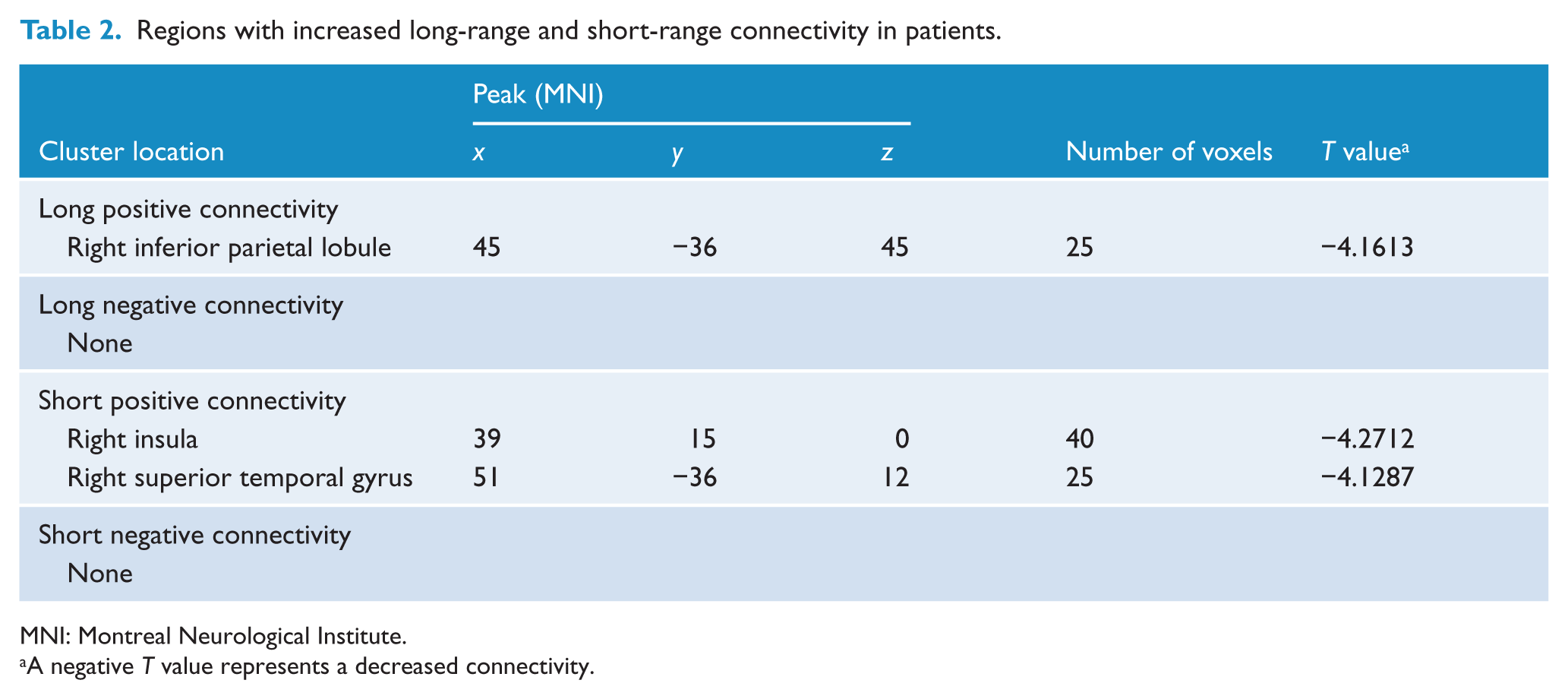
MNI: Montreal Neurological Institute.
A negative T value represents a decreased connectivity.
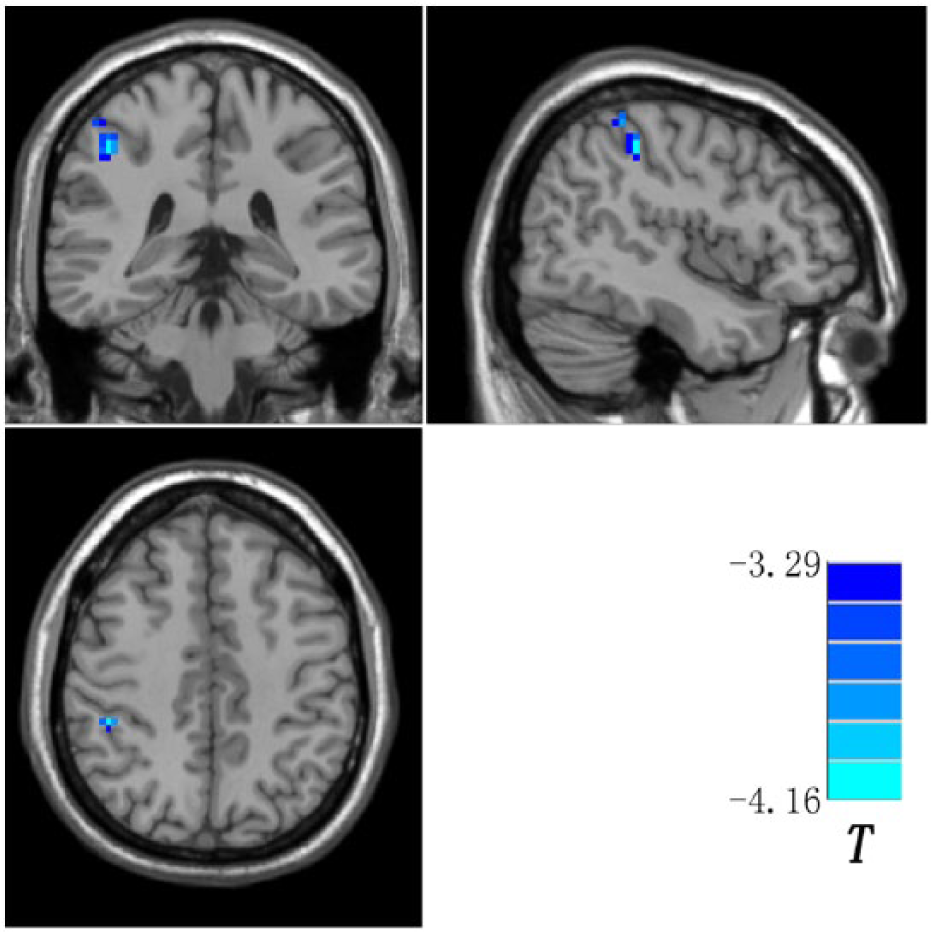
Brain region (right inferior parietal lobule) showing decreased long-range positive FCS in the patients compared with the controls. Blue denotes decreased long-range positive FCS in the patients. The color bar indicates the T values from two-sample t-tests. FCS: functional connectivity strength.
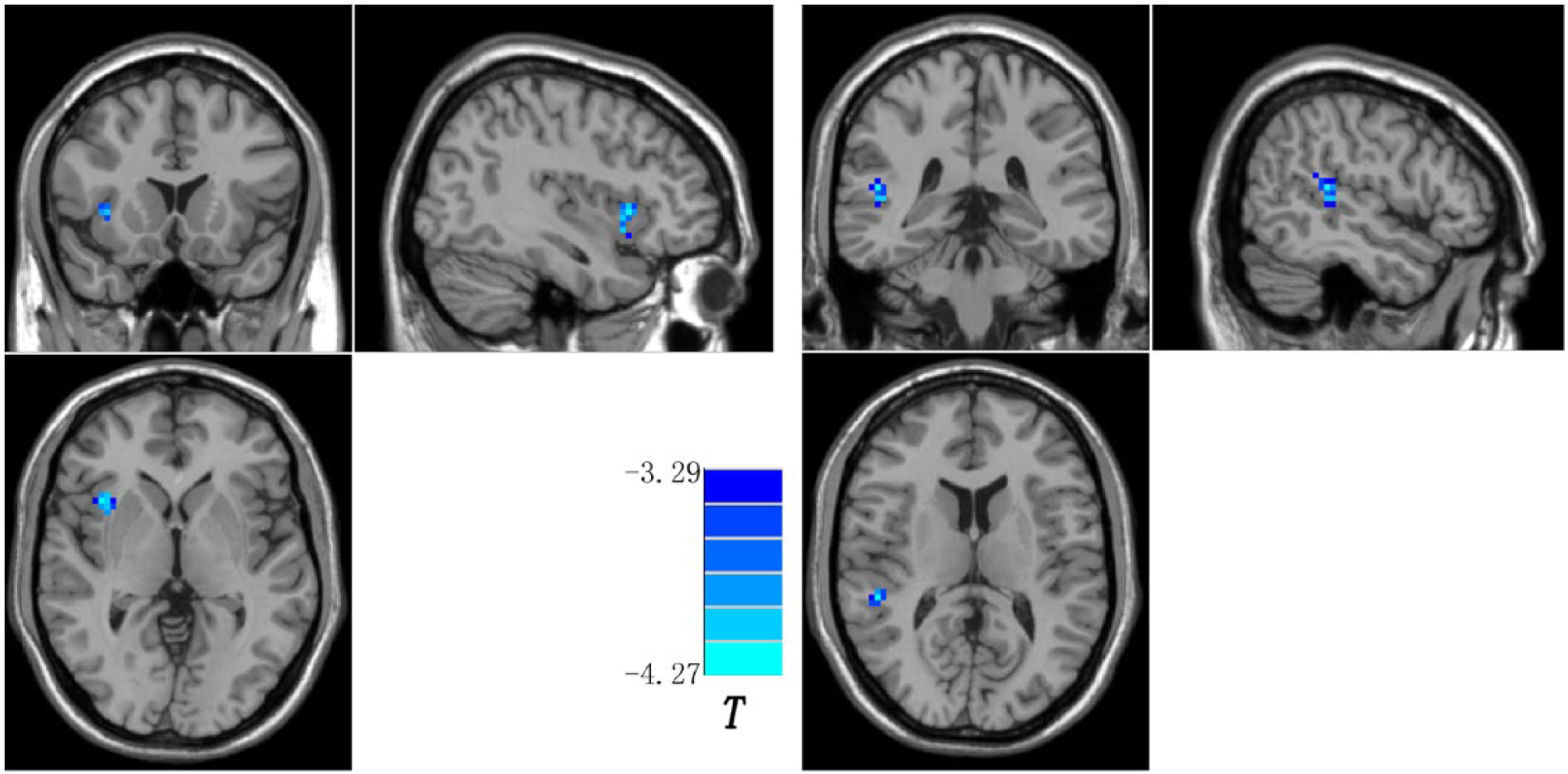
Brain regions (right insula and right superior temporal gyrus) showing decreased short-range positive FCS in the patients compared with the controls. Blue denotes decreased short-range positive FCS in the patients. The color bar indicates the T values from two-sample t-tests. FCS: functional connectivity strength.
Correlations between abnormal FCS and clinical variables in the patients
No correlation was indicated between abnormal FCS and HRSD scores or ATQ scores in the patients. No significant correlation was detected between abnormal FCS and age or education level in the patient group.
Discussion
In this study, regional FCS was divided into long- and short-range FCS. Compared with the controls, patients with MDD showed decreased lpFCS in the right IPL and decreased spFCS in the right insula and right STG. These abnormal FCSs exhibited no correlations with the clinical variables (HRSD and ATQ scores).
As a key player in DMN, IPL plays a critical role in emotional regulation and self-referential processing (Tao et al., 2013). The IPL demonstrates connections with the inferior frontal gyrus, which is linked to response inhibition (Aron et al., 2014; Swick et al., 2008). The IPL is also important for emotional perception at the presentation of facial stimuli (Peeters et al., 2009). These pathways are weakened in MDD (Macuga and Frey, 2011; Rizzolatti and Sinigaglia, 2010). Consistent with these studies, decreased lpFCS is observed in the right IPL in our patients. Decreased lpFCS may reflect reduced connections with DMN, such as the inferior frontal gyrus, leading to DMN dysfunction in the patients. Therefore, the patients would exhibit problems in emotional regulation and self-referential processing.
The insula presents extensive connections with the fronto-limbic network and related brain regions (Wang et al., 2012) and is considered an integration center for processing autonomic, emotional, interoceptive and visceromotor information (Sliz and Hayley, 2012). Structural and functional abnormalities in the insula have been reported in MDD. For instance, reduced gray matter volume in the right insula has been found in MDD (Kong et al., 2014). Decreased FC between the insula/prefrontal regions and amygdala has been observed in MDD when the patients were subjected to a task with negatively valenced images, indicating top-down emotional dysregulation in these patients (Perlman et al., 2012). Recently, reduced insular connectivity with the fronto-limbic network, hate circuit (comprising the superior frontal gyrus, insula and putamen) and visual regions has been observed in the present patient group (Guo et al., 2015a). In line with these findings, the decreased spFCS in the right insula suggests that the patients have decreased insular connections, mainly in the short-range positive connections. The decreased spFCS in the right insula may disrupt its integration role of emotional processing in the patients.
STG is one of the most identified brain regions linked to the neurobiology of MDD (Fitzgerald et al., 2008). STG has connections with the prefrontal cortex and amygdala, which is involved in social cognition and emotional processing (Jou et al., 2010). Decreased regional activity (Fitzgerald et al., 2008) and altered FC (Cullen et al., 2009) in STG have been reported in MDD, resulting in negative bias when interpreting interpersonal feedback (Cullen et al., 2009). As such, the patients may withdraw from daily social activity (Cullen et al., 2009). Consistent with these studies, reduced spFCS in the right STG in this study may disrupt the connections of this region, causing social withdrawal and emotional dysregulation commonly found in patients with MDD.
Abnormal FC in the frontal cortex in MDD has been reported in numerous neuroimaging studies (Anand et al., 2005; Cullen et al., 2009; Su et al., 2015). Therefore, it was unexpected to find no significant alterations in FCS in the frontal cortex in our study. A possible explanation is that the frontal cortex has efferent and afferent links to the temporoparietal regions (Zhang et al., 2009). As mentioned above, the IPL, insula and STG are connected with the frontal cortex. Moreover, brain networks are dynamic, and frontal FC alterations may be associated with a multitude of non-frontal disrupted links.
Contrary to our expectations and to what Zhang et al. (2011) reported previously, we observed no correlations between abnormal FC and HRSD. The possible reason for this lack of correlations is that the decreased lpFCS in the right IPL and the decreased spFCS in the right insula and the right STG are trait alterations in the patients independent of symptom severity. To test this possibility, we performed additional analyses with the HRSD scores as a covariate (the HRSD score for each control was set to 1). Similar results (Figures S1, S2 and Table S1) were obtained, suggesting that symptom severity rarely affected these FCSs in the patients.
This study has certain limitations. First, the definition of long- and short-range FCS with the structural distance of 75 mm is arbitrary to a certain extent. However, this method is considered conventional (Achard et al., 2006; He et al., 2007) and facilitates comparison between the present findings and those of other studies. Second, this study is cross-sectional, and a longitudinal study is required to observe the dynamic changes of the long- and short-range FCS in MDD. Finally, the present findings are acquired at rest and not with a specific task. Hence, the present findings are associated with depressive neurobiology in general.
Despite the limitations, this study is the first to examine long- and short-range FCS of the whole brain in drug-naive patients with MDD. The findings revealed decreased lpFCS in the right IPL, as well as decreased spFCS in the right insula and the right STG in the patients, suggesting that anatomical distance decreases long- and short-range FCS in patients with MDD, which may underlie the neurobiology of MDD.
Footnotes
Acknowledgements
The authors wish to thank all the participants.
Declaration of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Natural Science Foundation of China (Grant Nos 81571310 and 81260210), Natural Science Foundation of Guangxi Province for Distinguished Young Scientists (Grant No. 2014GXNSFGA118010) and Natural Science Foundation of Guangxi Province (Grant No. 2013GXNSFAA019107).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
