Abstract
Objective:
Childhood trauma confers great risk for the development of multiple psychiatric disorders; however, the neural basis for this association is still unknown. The present resting-state functional magnetic resonance imaging study aimed to detect the effects of childhood trauma on brain function in a group of young healthy adults.
Methods:
In total, 24 healthy individuals with childhood trauma and 24 age- and sex-matched adults without childhood trauma were recruited. Each participant underwent resting-state functional magnetic resonance imaging scanning. Intra-regional brain activity was evaluated by regional homogeneity method and compared between groups. Areas with altered regional homogeneity were further selected as seeds in subsequent functional connectivity analysis. Statistical analyses were performed by setting current depression and anxiety as covariates.
Results:
Adults with childhood trauma showed decreased regional homogeneity in bilateral superior temporal gyrus and insula, and the right inferior parietal lobule, as well as increased regional homogeneity in the right cerebellum and left middle temporal gyrus. Regional homogeneity values in the left middle temporal gyrus, right insula and right cerebellum were correlated with childhood trauma severity. In addition, individuals with childhood trauma also exhibited altered default mode network, cerebellum-default mode network and insula-default mode network connectivity when the left middle temporal gyrus, right cerebellum and right insula were selected as seed area, respectively.
Conclusion:
The present outcomes suggest that childhood trauma is associated with disturbed intrinsic brain function, especially the default mode network, in adults even without psychiatric diagnoses, which may mediate the relationship between childhood trauma and psychiatric disorders in later life.
Introduction
Childhood trauma, a most common psychological stressor which has adverse effects on the development of children and adolescents, has now become a major social public-health problem worldwide (Gilbert et al., 2009). The high prevalence of childhood trauma has been reported both in Western countries (Perez-Fuentes et al., 2013) and in China (Zhang et al., 2013). Moreover, it has been demonstrated that childhood trauma is a documented predictor of developing multiple psychiatric illnesses in later life, such as post-traumatic stress disorder (PTSD) (Pederson and Wilson, 2009) and major depressive disorder (MDD) (Takizawa et al., 2014). In recent years, several lines of studies have been directed to elucidate the biological mechanisms underlying this association from neuroendocrinology (Lu et al., 2016), neuroinflammation (Baumeister et al., 2016) and neuroimaging (Duncan et al., 2015) in individuals with childhood trauma, even without psychiatric diagnoses. However, to date, the neural pathways from childhood trauma to psychiatric disorders remain unknown.
Over the last two decades, functional neuroimaging has enriched our understanding of brain circuits correlated with childhood trauma (Marusak et al., 2015). In particular, resting-state functional connectivity (rs-FC) has been increasingly utilized to investigate the characteristics of resting-state networks in subjects with childhood trauma, and one area that is estimated to be relevant to childhood trauma is the default mode network (DMN) (Philip et al., 2013b). Bluhm et al. (2009) first measured resting-state DMN connectivity in chronic PTSD patients related to childhood abuse and found that patients with PTSD showed decreased FC of posterior cingulate cortex (PCC) with medial prefrontal cortex (mPFC) in DMN. This study could be interpreted as that childhood abuse might be associated with decreased DMN connectivity among individuals who developed PTSD. In a recent study, altered FC between two core DMN regions, PCC and anterior mPFC, was observed in infants with higher interparental conflict since birth, and increased DMN connectivity might serve as a predictor of negative infant emotionality after trauma exposures (Graham et al., 2015). In addition to DMN dysfunction, other key findings included disruptions in executive and emotion-regulating networks in subjects with early life stress (Cisler et al., 2013), as well as altered intrinsic connectivity of the salience network (SN) in trauma-exposed youth (Marusak et al., 2015). Childhood trauma was also revealed to be associated with diminished FC between amygdala and precuneus, as well as with decreased FC of the insula with hippocampus in a previous study (Van der Werff et al., 2013).
Despite the growing body of evidence that effects of childhood trauma may be observable in rs-FC measures, the FC method is limited to the measurement of interregional temporal correlation only while ignoring intraregional brain activity (Van de Ven et al., 2004). On the contrary, regional homogeneity (ReHo) is a data-driven method which targets the identification of voxels where spontaneous activity is synchronous locally (Zang et al., 2004). ReHo abnormalities may indicate the changes of temporal neural activities in the regional brain (Zhong et al., 2011). The ReHo approach has been widely used to evaluate resting-state cortical activity in many psychiatric disorders (Cui et al., 2016; Lai and Wu, 2016; Yin et al., 2012); however, few studies have been designed toward childhood trauma using this method. The only study by Philip et al. (2013a) demonstrated that childhood trauma in medication-free healthy adults was associated with diminished ReHo in two DMN regions, the right inferior parietal lobule (IPL) and superior temporal gyrus (STG), but the sample size of the experimental group was relatively small.
Taken together, these findings indicate that there may be an association between childhood trauma and resting-state brain dysfunction. Actually, the majority of these studies performed FC analysis only; however, it is suggested that coupled ReHo and FC analyses may be more effective than either method alone in resting-state functional magnetic resonance imaging (rs-fMRI) study (Philip et al., 2013a). In addition, most studies did not exclude individuals with lifetime psychiatric diagnoses and the association between current mood state, and brain activity in subjects with childhood trauma was not well controlled. Therefore, the existing research outcome requires verification in further studies that will overcome these limitations in order to exactly describe the effects of childhood trauma on brain function. In this context, we present here a study trying to examine the impacts of childhood trauma on resting-state brain function in a group of young healthy adults by combining ReHo and FC on the basis of excluding the influence of current anxiety and depression.
Method
Participants
A total of 48 subjects (male/female, 18/30)—aged 18–33 years, including 24 subjects with childhood trauma experiences (CT group) and 24 age- and gender-matched subjects without childhood trauma exposures (non-CT group)—were recruited in this study. For assignment to the CT group, individuals must have had experienced chronic moderate–severe trauma exposures (abuse and/or neglect) before the age of 16. All participants were recruited from a survey that we had carried out to investigate the occurrence of childhood trauma in local communities and universities. Subjects responded with no direct reference to childhood trauma as a key variable in the study. All subjects were thoroughly interviewed by two professional psychiatrists and were free from any current or lifetime history of psychiatric disorders according to Diagnostic and Statistical Manual of Mental Disorders–Fourth Edition (DSM-IV) criteria, as screened with the Structured Clinical Interview for DSM-IV interview (SCID). The general exclusions were as follows: (1) left-handedness; (2) standard scores >50 on Zung’s Self-rating Depression Scale (SDS) (Zung et al., 1965) or >40 on Zung’s Self-rating Anxiety Scale (SAS) (Zung, 1971); (3) significant medical illness; (4) presence of major sensorimotor handicaps; (5) history of seizures, head trauma or unconsciousness; (6) intake of any psychotropic medication or hormone; (7) alcohol or substance abuse; (8) women with pregnancy/lactation or in their menstrual period; (9) contraindications to magnetic resonance imaging (MRI) scan, including metallic implants, retractors or braces, and claustrophobia; and (10) inability to keep still during MRI scanning. The demographic data were collected using a self-designed questionnaire from all the participants. This study was approved by the ethic committee of the Second Xiangya Hospital of Central South University. A complete description of the study was provided to every subject, after which written informed consent was obtained from each participant.
Assessment of childhood trauma
Childhood trauma was assessed using Childhood Trauma Questionnaire (CTQ) (Bernstein et al., 2003). This 28-item, reliable and valid self-reporting questionnaire can yield five sub-scales which evaluate five aspects of childhood trauma exposures: emotional abuse, emotional neglect, sexual abuse, physical abuse and physical neglect. Subjects who score higher than the threshold (moderate–severe) of a sub-scale are treated as existence of corresponding childhood trauma experience. The cutoffs of each sub-scale for moderate–severe exposure are (1) emotional abuse ⩾ 13, (2) emotional neglect ⩾ 15, (3) sexual abuse ⩾ 8, (4) physical abuse ⩾ 10 and (5) physical neglect ⩾ 10 (Bernstein and Fink, 1998). The Chinese version of CTQ was introduced in our study. It was translated into Chinese by Zhao et al. (2005); the CTQ has good internal consistency (Cronbach’s alpha) for the CTQ total score (0.77), and the five sub-scales range from 0.41 to 0.68 in a Chinese sample of 794 individuals. It has been widely used in many Chinese studies, although many researchers would like to revise the CTQ and hope its sub-scales to be more suitable for the Chinese population (Lu et al., 2013).
MRI acquisition
The imaging data were obtained on a 3.0T Philips Achieva scanner at the Second Xiangya Hospital, Central South University. Subjects were asked to lie on the scanner and keep eyes closed. A standard birdcage head coil was used, and the restraining foam pads were placed on two sides of the head to minimize head motion while cotton plug was used with the purpose of diminishing the noise. A total of 180 volumes of echo planar images were obtained axially; the parameters were as follows: repetition time (TR) = 3000 ms; echo time (TE) = 30 ms; slices = 36; thickness = 4 mm, no slice gap; field of view (FOV) = 240 × 240 mm2; resolution = 64 × 64; flip angle = 90°; scan time = 9′09″.
Data preprocessing
The MRIConvert program (http://lcni.uoregon.edu/~jolinda/MRIConvert/) was used to convert neuroimaging data to Neuroimaging Informatics Technology Initiative data format. All the rs-fMRI data were processed using Data Processing Assistant for Resting-State fMRI Advanced Edition V2.2 (DPARSFA, www.restfmri.net/forum/DPARSF). The preprocessing steps included slice timing to correct within-scan acquisition time differences between slices, realignment to the first volume to correct head motion (a six-parameter spatial transformation), spatial normalization to the Montreal Neurological Institute template using the participant-specific transformation parameters created by fitting mean functional images to the single reference EPI standard template in Statistical Parametric Mapping 8 (SPM8) and resampling images into a spatial resolution of 3 × 3 × 3 mm3. The functional images were subsequently smoothed by convolution with an isotropic Gaussian kernel (full width at half maximum [FWHM] = 4 mm). After smoothing, the imaging data were temporal band-pass filtered (0.01–0.08 Hz) to reduce the effects of low-frequency drift and physiological high-frequency noise, and the linear trend was removed.
The head motion may be a huge problem in rs-fMRI datasets; in this context, the mean framewise displacement (FD) was computed by averaging FD i from every time point for each subject. The mean FD ranged from 0.06 to 0.20 mm in our study samples, and there was no difference for the mean FD between two groups (t = 0.835, p = 0.408) (see Table 1).
Demographic and clinical characteristics of all subjects (N = 48).
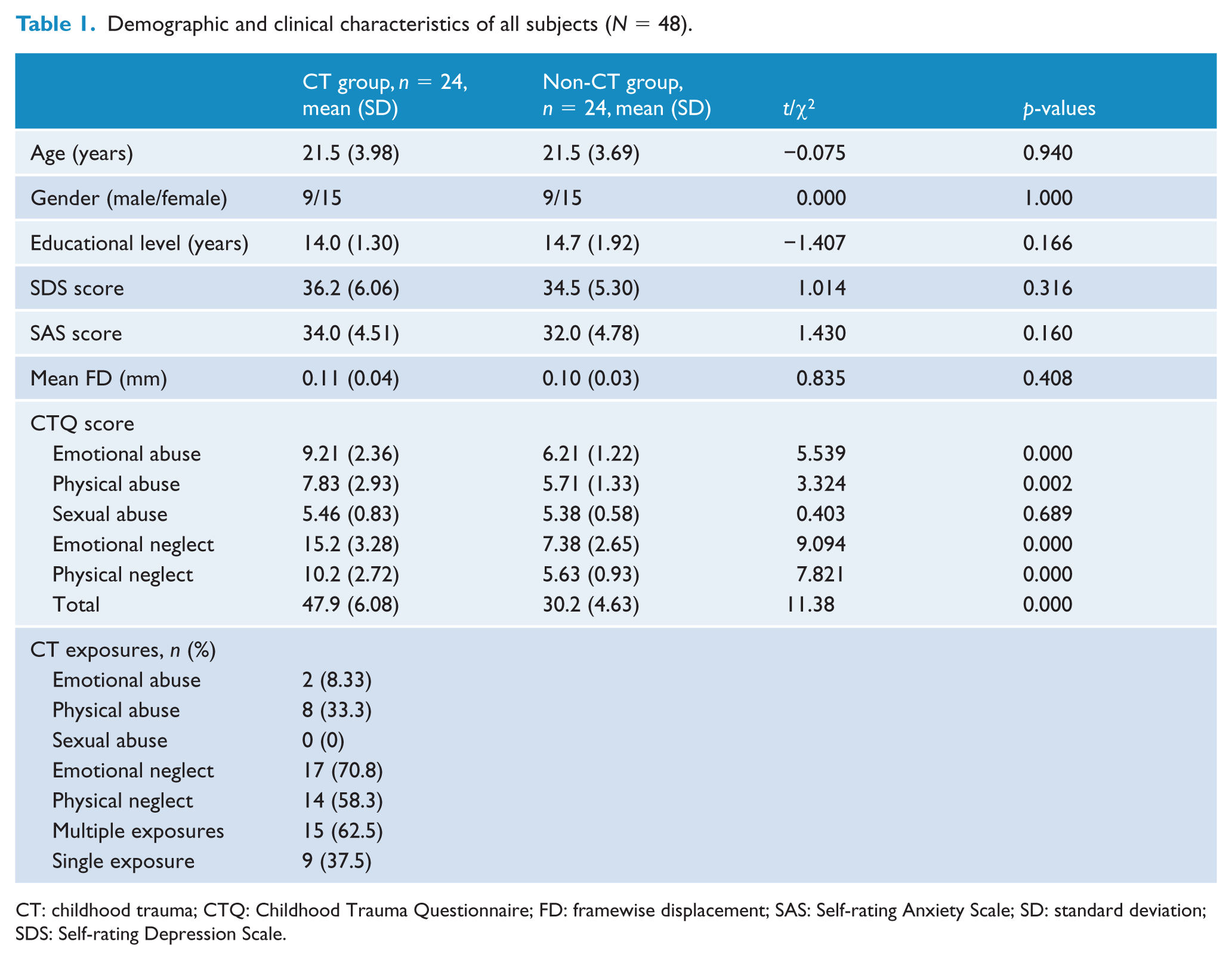
CT: childhood trauma; CTQ: Childhood Trauma Questionnaire; FD: framewise displacement; SAS: Self-rating Anxiety Scale; SD: standard deviation; SDS: Self-rating Depression Scale.
ReHo and FC calculation
Data without spatial smoothing were used for ReHo analysis with DPARSFA. The Kendall’s coefficient of concordance (KCC) was calculated to measure ReHo of the time series of a given voxel with its nearest neighbor (26 voxels) in a voxel-wise way that had been described by Zang et al. (2004). Then, a mask (made from the MNI template to assure matching with the normalization step), in the REST software, was used to remove non-brain tissue and for standardization purposes. Each individual ReHo map was divided by its own global mean KCC value within the mask (Wu et al., 2009). Finally, the data were smoothed with a Gaussian kernel of 4 mm FWHM.
A seed-based interregional FC analysis was used in this study. Areas where between-group ReHo differences correlated with childhood trauma were selected as seeds. The average time series data from seed regions were extracted and FC analysis was performed using the data resulting from preprocessing. The nuisance covariates, including cerebrospinal fluid signals, global mean signals, white matter signals, as well as six head motion parameters, were regressed out; then a voxel-wise correlation analysis was conducted between the seed region and the rest of the brain (r score); and finally, Fisher’s r to z transformation was performed to improve the normality of the correlation coefficient.
Statistical analysis
Demographic and clinical data were analyzed by using Statistical Package for the Social Sciences, version 16.0 (SPSS Inc., Chicago, IL, USA). Independent two-sample t-tests and Chi-square tests (χ2) were, respectively, used to test for the continuous variables and categorical variables between the two groups. Values are given as mean ± standard deviation. The level of two-tailed statistical significance was set at p < 0.05 for all tests.
Between-group differences of ReHo and FC were analyzed using two-sample t-tests on a voxel-by-voxel basis with SPM8 software. The potential confounders—such as age, sex, educational level, mean FD values, scores of SDS and SAS—were included as covariates of no interest in the comparison. The t-map was set at a corrected significance level of p < 0.05 (combined height threshold p < 0.001 [T > 3.32] and cluster >5). Threshold correction was performed using AlphaSim program (parameters were as follows: individual voxel p = 0.001, 1000 simulations, FWHM = 4 mm, with mask) in the REST software, which applied Monte Carlo simulation to calculate the probability of false-positive detection by taking into consideration both the individual voxel probability thresholding and cluster size (Poline et al., 1997).
Furthermore, to evaluate any correlations of between-group ReHo or FC differences with CTQ scores, whole-brain multiple regression analyses integrated in SPM basic models were performed at p < 0.05 (AlphaSim corrected) (combined height threshold p < 0.001 [T > 3.31] and cluster >5); the potential confounders described above were included as covariates as well. Then, the mean ReHo values of the survived clusters were extracted using region of interest (ROI) analyses. Correlations were conducted using Pearson’s product moment.
Results
Demographics and clinical measures
As indicated in Table 1, the two groups of subjects did not differ with respect to age, gender, educational level, SAS score and SDS score. As we would expect, the two experimental groups differed on levels of CTQ and its sub-scales except sexual abuse. In maltreated subjects, the most common aspect of childhood trauma experience was emotional neglect (17, 70.8%); a proportion of 62.5% (15) of traumatic subjects experienced at least two forms of childhood trauma exposures.
Between-group ReHo and FC differences
As compared with subjects without childhood trauma, individuals with adverse experiences in childhood shared decreased ReHo in bilateral STG and insula, and the right IPL, as well as increased ReHo in the right cerebellum and left middle temporal gyrus (MTG) at the defined threshold (see Table 2 and Figure 1). Although significant positive correlations of ReHo in the left MTG (x = −63, y = −15, z = −21, cluster = 6, T = 4.18) and right cerebellum (x = 45, y = −66, z = −30, cluster = 13, T = 4.32), while negative association of ReHo in the right insula (x = 42, y = −15, z = 9, cluster = 14, T = −4.69), with CTQ total scores were found in the whole sample, no dose-dependent effect of childhood trauma within the exposed group was observed. The mean ReHo values of the regions that showed significant correlations with CTQ total scores were extracted from each subject. The scatter plots are shown in Figure 2.
Brain regions showing altered ReHo values in CT individuals as compared with subjects without CT (AlphaSim-corrected p < 0.05).
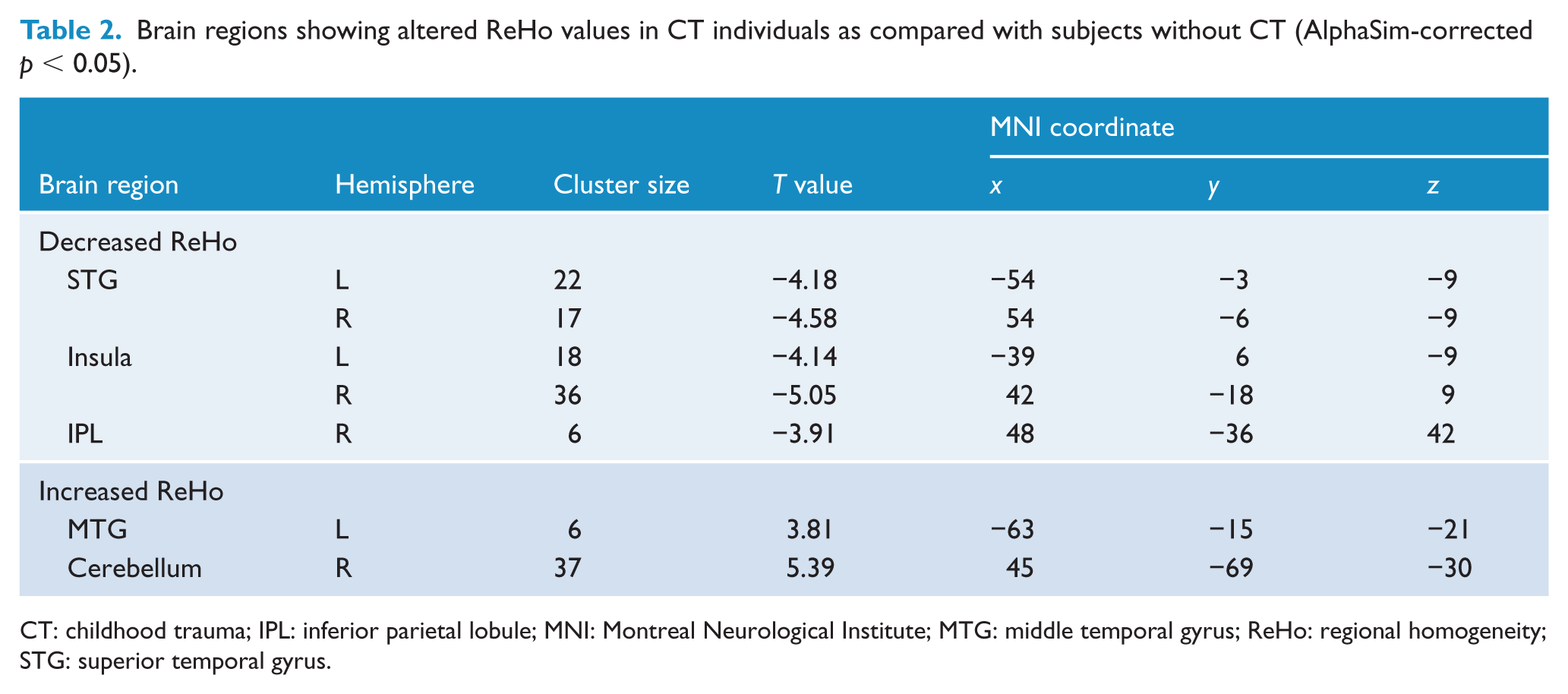
CT: childhood trauma; IPL: inferior parietal lobule; MNI: Montreal Neurological Institute; MTG: middle temporal gyrus; ReHo: regional homogeneity; STG: superior temporal gyrus.
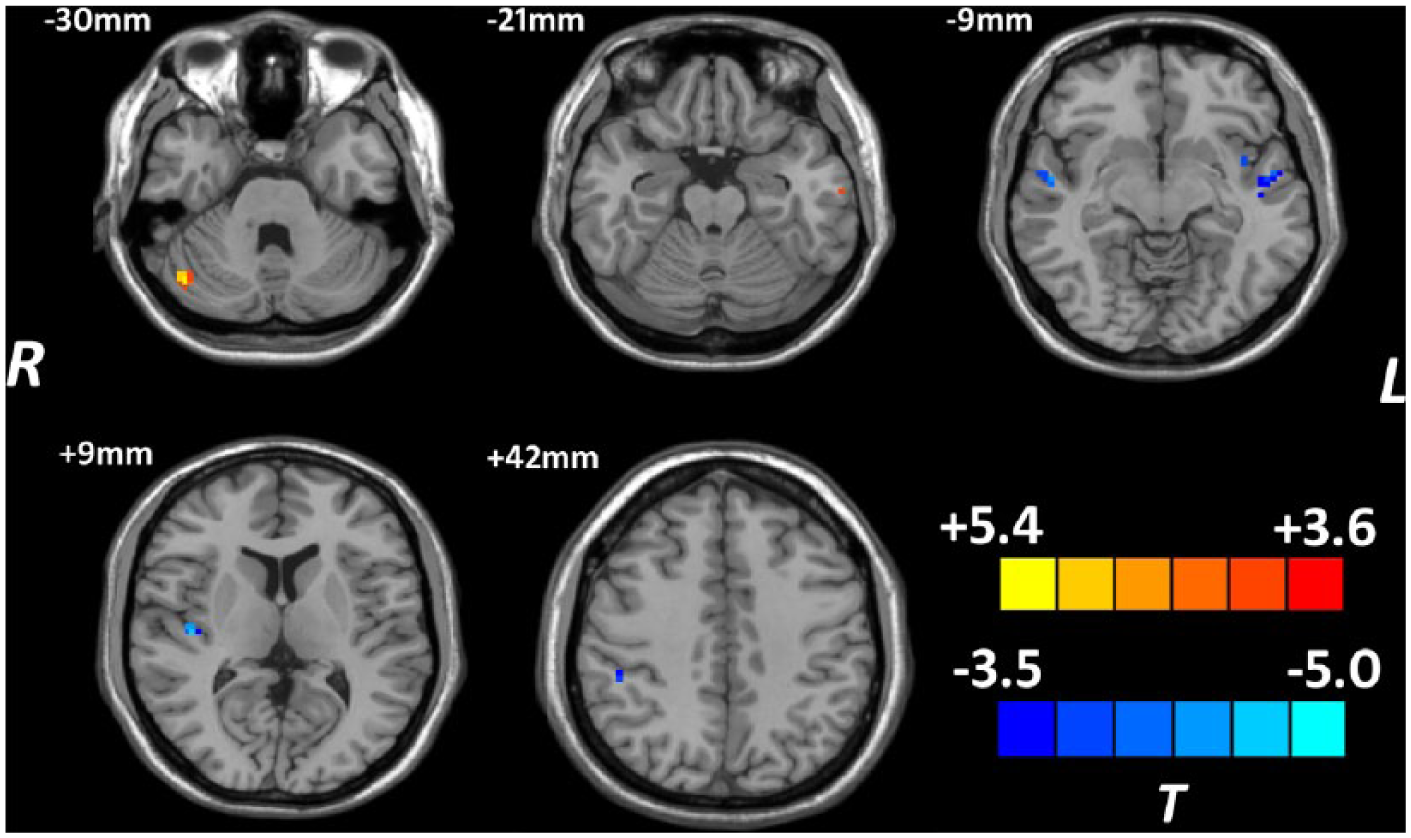
Brain regions showing different ReHo values between individuals with and without childhood trauma (AlphaSim-corrected p < 0.05). Hot and cold colors indicate increased and decreased ReHo in individuals with childhood trauma, respectively, compared with subjects without childhood trauma.
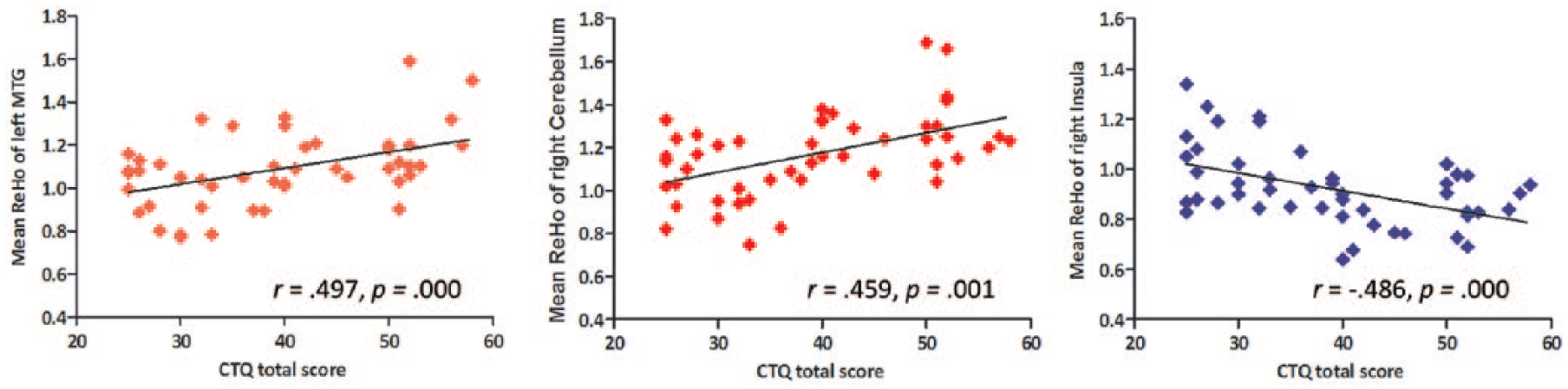
Correlation analyses between CTQ total scores and ReHo values in the whole sample.
The ReHo analysis revealed that the activity in the left MTG (6 voxels), right cerebellum (13 voxels) and right insula (14 voxels) exhibited significant between-group difference and correlated with CTQ total score in the whole sample. Therefore, these three regions were selected as seed regions for subsequent FC analysis. Subjects with childhood trauma showed decreased FC in the left STG and increased FC in the right inferior temporal gyrus, diminished FC in the right STG along with elevated FC in the mPFC and right anterior cingulate cortex, and decreased FC in the mPFC, right middle frontal gyrus and right MTG when the left MTG, right cerebellum and right insula were selected as seed area, respectively (see Table 3 and Figure 3). However, no significant correlation was found between FC alterations and CTQ scores in individuals with childhood trauma.
Brain regions showing altered functional connectivity with selected seeds in CT individuals as compared with subjects without CT (AlphaSim-corrected p < 0.05).
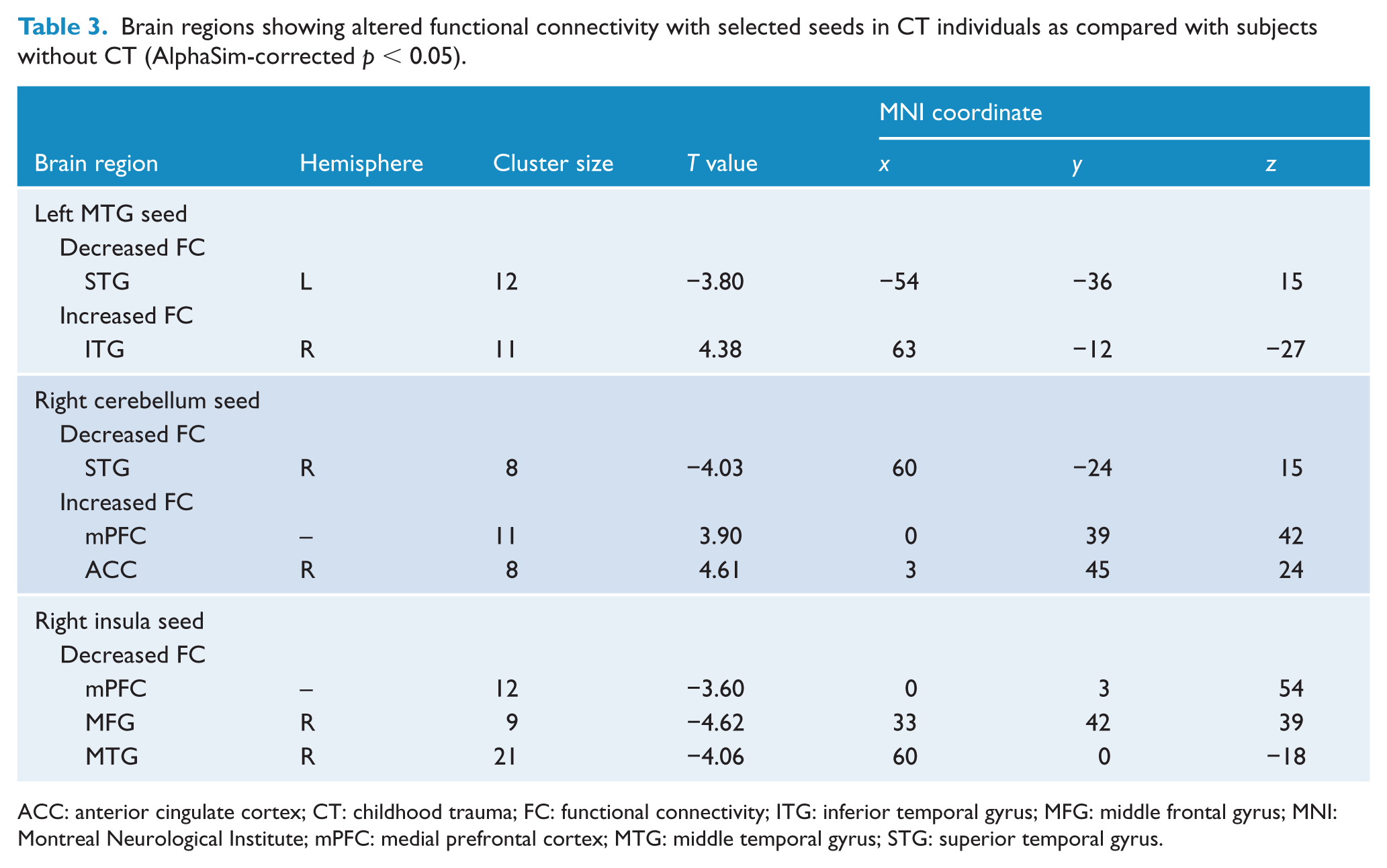
ACC: anterior cingulate cortex; CT: childhood trauma; FC: functional connectivity; ITG: inferior temporal gyrus; MFG: middle frontal gyrus; MNI: Montreal Neurological Institute; mPFC: medial prefrontal cortex; MTG: middle temporal gyrus; STG: superior temporal gyrus.
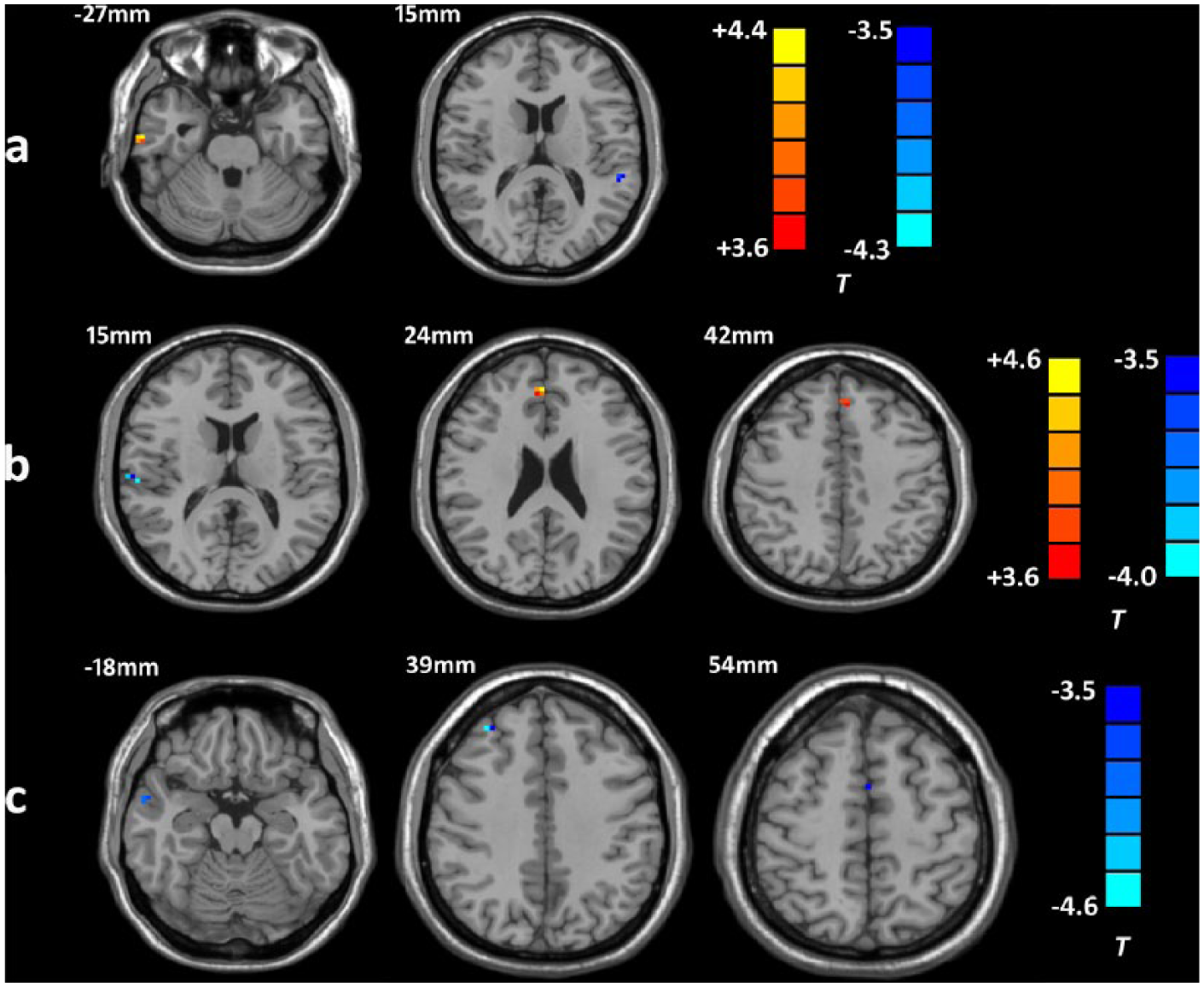
Brain regions showing altered functional connectivity with selected seeds in individuals reporting childhood trauma (AlphaSim-corrected p < 0.05). Hot and cold colors indicate increased and decreased FC in individuals with childhood trauma, respectively, compared with subjects without childhood trauma: (a) left MTG–based FC; (b) right cerebellum–based FC; (c) right insula–based FC.
Discussion
This study investigated the impacts of childhood trauma on intra- and interregional synchronized activity using ReHo and FC in a group of young healthy adults. The current results revealed altered regional brain activity in several DMN regions (IPL, STG and MTG), insula and cerebellum, as well as altered DMN, cerebellum-DMN and insula-DMN connectivity in individuals with self-reported childhood trauma history. These outcomes together with the previous findings suggest that childhood trauma is associated with disturbed intrinsic brain function, especially the DMN, in adults even without current psychiatric or medical symptoms (Philip et al., 2013a, 2013b).
The DMN, which is reported to be involved in stimulus-independent, internally focused thought, including spontaneous cognition, autobiographical memory, prospection and mind-wandering (Sripada et al., 2014), has rapidly become a central theme in contemporary cognitive and clinical neuroscience (Broyd et al., 2009). Previous studies have demonstrated that impairments in the activation or connectivity of DMN are linked to many psychiatric disorders—for example, schizophrenia, PTSD and MDD (Sripada et al., 2012; Tang et al., 2013)—which are closely correlated with childhood trauma. However, it remains unclear whether the DMN abnormalities are related to the disease or disease process. Recruiting subjects with childhood trauma who are at high risk of developing mental disorders may help to disentangle this puzzle.
We found that even without psychiatric symptoms, histories of childhood trauma alone still confirmed an elevated risk of intra- and interregional brain dysfunction within DMN. The present outcomes indicated that DMN abnormalities might be secondary to childhood adversity, likely representing psychobiological vulnerability for the development of psychiatric disorders in response to stress. This indication was supported by prior studies showing the association between early traumatization and alterations in DMN functioning (Philip et al., 2013b; Sripada et al., 2014). Interestingly, in a recent study, reduced DMN connectivity was also found in veterans exposed to combat trauma regardless of PTSD diagnosis when compared to healthy civilian controls (DiGangi et al., 2016). Taken together, these findings suggest that aberrant DMN function at rest may reflect stress exposure, as opposed to illness per se. They also prompt future study of the relevance of baseline brain dysfunctions as a consequence of stress and trauma.
A second noteworthy finding in this study was altered cerebellum activation, including increased ReHo and altered cerebellum-DMN connectivity in subjects with childhood trauma. The cerebellum is traditionally considered a brain structure involved in motor domain; a growing number of independent lines of research, however, points toward a significant role for the cerebellum in processes associated with cognition and emotion (Schutter, 2012). The cerebellum is sensitive to the effects of early maltreatment; previous structural MRI studies have accumulated the evidence that childhood trauma is associated with volume reductions in the cerebellum (Bauer et al., 2009). Our data were, at least in part, comparable with findings of a prior rs-fMRI study, which detected an abnormal activation of the cerebellum in MDD patients with childhood neglect (Wang et al., 2014). In line with this, childhood sexual abuse was also suggested to be able to produce a functional deficit in the cerebellum leading to diminished blood volume and neuronal activity in a resting-state blood flow study (Anderson et al., 2002).
In this study, increased cerebellum-DMN connectivity is a novel finding that has not yet reported in individuals with childhood trauma. On the contrast, altered cerebellum-DMN connectivity in subjects with ultra-high risk of psychosis (Wang et al., 2016), first-episode schizophrenia patients and their unaffected siblings (Guo et al., 2015a), drug-naive MDD (Guo et al., 2015b) and treatment-resistant MDD (Guo et al., 2013) has been reported in a series of studies. Based on these findings, it is proposed that aberrant cerebellum-DMN activity may contribute to disrupted motor control of emotional and cognitive processing (Coombes et al., 2009; Guo et al., 2015a). However, future research designs are needed for more focused characterization of the role of cerebellum-DMN connectivity in childhood trauma and related conditions.
The final region we found to show altered brain activity was the insula. The insula has been demonstrated to be involved in face recognition, tactile recognition and working memory in some studies (Reed and Means, 2004). In a previous study for patients with PTSD, volume reduction in bilateral insula was found to be associated with declarative memory deficits; meanwhile, decreased activation in the insula when performing word encoding and retrieval tasks was revealed as well in the same sample (Chen et al., 2009). More interestingly, childhood trauma alone has also been demonstrated to increase the activation of insula during a working memory task in healthy volunteers (Philip et al., 2016). Of note, a relationship between childhood trauma and cognitive impairments, including decreased memory, has been well documented outside of the neuroimaging literature (Tyrka et al., 2013). In that case, we speculate that the present finding of altered insula activation in subjects with childhood trauma may reflect impaired cognitive function, in particular, working memory deficits.
In recent studies, the insula has been increasingly known to be a core hub of the SN which plays a major role in monitoring the salience of external inputs and internal brain events (Manoliu et al., 2013). Prior studies have demonstrated that aberrant activity and connectivity of the SN may contribute to negative biases in attention and thought inherent in many psychiatric disorders (Etkin and Wager, 2007; Manoliu et al., 2013). Interestingly, in a study of Marusak et al. (2015), young subjects with childhood trauma also showed disrupted insula-based neural circuit organization within SN that is associated with diminished reward sensitivity, a critical risk/resilience trait following stress. In addition to altered intrinsic connectivity of the SN, they further observed reduced connectivity between the SN (insula) and DMN in the same participants, which was comparable with our findings. The right insula has been suggested to critically contribute to appropriate behavioral responses to salient stimuli via switching between DMN-related self-referential and Central Executive Network–related goal directed cognitive activity; moreover, aberrant switching may have relevance for mental health (Sridharan et al., 2008). Taken together, it is possible that functional changes in the SN and SN-DMN connectivity are a consequence of childhood trauma that also serve to increase psychiatric risk.
Limitations
This study had several important limitations. First, a question has arisen whether the self-reported childhood trauma history reflects the actual experience of participants in their childhood and early adolescence. In this context, we have carried on face-to-face interviews to confirm their childhood adverse experience in order to reduce information biases as possible. Second, since the cross-sectional design precludes causal inferences, the current results should be interpreted with caution. Third, patients with active psychiatric illness were not included in our study, and direct comparisons between these findings and, for instance, findings observed in samples with PTSD or MDD patients are not possible. Finally, the sample size in each group is relatively small which restricts us to discuss the different effects of five types of childhood trauma on brain functional changes.
Conclusion
In summary, the present findings suggest that childhood trauma is associated with disturbed intrinsic brain activity and interregional FC, especially the DMN. These effects are still evident even in the absence of psychiatric symptoms, which may mediate the relationship between childhood trauma and psychiatric disorders in later life.
Footnotes
Acknowledgements
The authors would like to thank all participants who took part in this study and the experts at the Magnetic Resonance Center of the Second Xiangya Hospital for providing scan time and technical assistant.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We sincerely thank the support of funds from the National Science and Technology Program (2015BAI13B02 to Lingjiang Li), the Medical Science and Technology Project of Zhejiang Province (2015KYA082 to Shaojia Lu) and the National Natural Science Foundation of China (81301158 to Manli Huang).
