Abstract
Objective:
Bipolar disorder in the depressive phase (BDd) may be misdiagnosed as major depressive disorder (MDD), resulting in poor treatment outcomes. To identify biomarkers distinguishing BDd from MDD is of substantial clinical significance. This study aimed to characterize specific alterations in intrinsic functional connectivity (FC) patterns in BDd and MDD by combining whole-brain static and dynamic FC.
Methods:
A total of 40 MDD and 38 BDd patients, and 50 age-, sex-, education-, and handedness-matched healthy controls (HCs) were included in this study. Static and dynamic FC strengths (FCSs) were analyzed using complete time-series correlations and sliding window correlations, respectively. One-way analysis of variance was performed to test group effects. The combined static and dynamic FCSs were then used to distinguish BDd from MDD and to predict clinical symptom severity.
Results:
Compared with HCs, BDd patients showed lower static FCS in the medial orbitofrontal cortex and greater static FCS in the caudate, while MDD patients exhibited greater static FCS in the medial orbitofrontal cortex. BDd patients also demonstrated greater static and dynamic FCSs in the thalamus compared with both MDD patients and HCs, while MDD patients exhibited greater dynamic FCS in the precentral gyrus compared with both BDd patients and HCs. Combined static and dynamic FCSs yielded higher accuracy than either static or dynamic FCS analysis alone, and also predicted anhedonia severity in BDd patients and negative mood severity in MDD patients.
Conclusion:
Altered FC within frontal–striatal–thalamic circuits of BDd patients and within the default mode network/sensorimotor network of MDD patients accurately distinguishes between these disorders. These unique FC patterns may serve as biomarkers for differential diagnosis and provide clues to the pathogenesis of mood disorders.
Keywords
Introduction
Depression is a common mental disorder characterized by negative affect, anhedonia and cognitive dysfunction such as intrusive internally focused thoughts (rumination) and psychomotor retardation (Bennabi et al., 2013). Both major depressive disorder (MDD) and bipolar disorder (BD) patients experience major depressive episodes (Post, 2005). Although BD also includes manic episodes, the similar clinical presentation and diagnostic criteria result in patients with BD during a depressive episode (BDd) frequently being misdiagnosed with MDD (Smith and Craddock, 2011), which leads to inadequate treatment and poor outcome. Accordingly, it is necessary to identify distinguishing features both for improved differential diagnosis and for a better understanding of the pathological mechanisms underlying these mood disorders (Korgaonkar et al., 2019a).
Resting-state functional connectivity (FC) describes the correlation in spontaneous neuronal fluctuations among anatomically separated brain regions, reflecting the level of functional coupling between regions (van den Heuvel and Pol, 2010). Based on assumed stability throughout the whole measurement period, static FC analysis has revealed aberrant neural circuits associated with dysfunctional dopamine and serotoninneurotransmission in depressive patients (Belujon and Grace, 2017). Specifically, BDd patients exhibited more dysfunctional connectivity than MDD patients. BDd was characterized by abnormal static FC among frontoparietal network, and temporal and subcortical regions (Chen et al., 2015), whereas MDD was characterized by aberrant static FC in the default mode network (DMN) and between DMN and other regions (Korgaonkar et al., 2019b; Wang et al., 2018). The DMN is a constellation of brain regions activated during rest and deactivated during goal-oriented behaviors (Raichle et al., 2001). The DMN is implicated in self-related processes, and increased DMN connectivity in MDD is associated with rumination (Posner et al., 2016). In addition to stable changes in FC, emerging analytic methods, such as sliding window correlation analysis, have revealed dynamic changes in FC by measuring the temporal variability of FC (Liao et al., 2014, 2019). Dynamic FC is not only sensitive to human behavior (Shirer et al., 2012) and development (Marusak et al., 2017) in healthy populations, but is also physiologically relevant in neuropsychiatric diseases (Cui et al., 2020; Li et al., 2018; Liao et al., 2014). Recent dynamic FC studies of depression have focused mainly on the DMN, and have revealed increased FC variabilities of the DMN are related to a ruminative thinking style in MDD patients (Kaiser et al., 2016; Wise et al., 2017), whereas decreased FC variability in the DMN is associated with disrupted executive processing in BD patients (Nguyen et al., 2017). However, whole-brain dynamic FC analysis rather than biased analysis of predefined brain regions or networks may be required to reveal etiologically and clinically relevant differences in FC patterns.
There have been numerous dynamic FC studies in recent years, and dynamic FC has been widely considered a powerful supplement to static FC (Han et al., 2018; Kaiser et al., 2016). Indeed, combined static and dynamic FC has yielded greater overall accuracy for mental disorder classification than either static or dynamic FC alone (Liao et al., 2018; Rashid et al., 2016). Herein, static and dynamic voxel-wise FC strengths (FCSs) in gray matter were investigated to fully assess the unbiased neural circuit mechanisms of MDD and BDd. Static FCS analysis of the full-length time series was first conducted to estimate the strength of connections between a given voxel and all other voxels in the brain, which has a close relationship with neurophysiological measures such as regional cerebral blood flow and cerebral glucose metabolic rates (Liang et al., 2013). Sliding window correlation analysis was then employed to evaluate dynamic FCS, which was quantified as the standard deviation of FCS values across sliding windows. Based on previous literature, we predicted that FCS alterations in BDd patients would involve the frontal–subcortical system, whereas alterations in MDD patients would be associated with the DMN–related systems. We also predicted that combined static and dynamic FCSs would improve diagnostic accuracy for discriminating between BDd from MDD, compared with static or dynamic FCS measures alone. In addition, we examined whether altered static and dynamic FCSs in BDd and MDD patients correlate with two primary depressive symptoms, anhedonia and negative mood.
Materials and methods
Participants
Forty patients with BDd and 40 patients with MDD were recruited from the Clinical Hospital of Chengdu Brain Science Institute. All patients were interviewed by two experienced psychiatrists using the Structured Clinical Interview for DSM-IV-TR–Patient Edition (SCID-P, 2/2001 version) to evaluate the presence of axis I disorder. The patients were also interviewed according to the SCID-II instrument to evaluate the presence of personality disorders using the Chinese version of the Personality Disorder subscale (Pfohl et al., 1997). All patients were diagnosed according to DSM-IV criteria. Exclusion criteria included anxiety disorders, schizophrenia, mental retardation, personality disorders and any history of consciousness loss, substance abuse and serious medical or neurological illnesses.
The clinical characteristic information of all depressive patients were collected including the age at first onset, duration of depressive episodes, duration of single episode and number of depressive episodes. The 24-item Hamilton Depression Scale (HAMD) was used to evaluate current depression severity. Depressive patients were treated with antidepressants, mood stabilizers and antipsychotics at enrolment and regimens were not changed. Medication was evaluated using the medication load index described in detail in previous studies (Almeida et al., 2009; Pang et al., 2018). In addition, the 14-item Snaith–Hamilton Pleasure Scale (SHAPS) was used to assess the hedonic capacity of subjects (Snaith et al., 1995). The 9-item Negative Affect Subscale of the Positive and Negative Affect Scale (PANAS-N) (Watson et al., 1988) was conducted to assess the severity of negative affective arousal of subjects (Qiu et al., 2008). The SHAPS was completed by 31 BDd patients, 36 MDD patients and 49 HCs, and the PANAS-N was completed by 28 BDd patients, 32 MDD patients and 49 HCs.
Fifty healthy controls (HCs) of comparable age, sex ratio, education level and handedness ratio were recruited through advertisements. The SCID (non-patient edition) was employed to ensure lifetime absence of psychiatric illnesses. Written informed consents were obtained from all participants before the experiment. This study was approved by the research ethics committee of the University of Electronic Science and Technology of China in compliance with the latest revision of the Declaration of Helsinki and registered at ClinicalTrials.gov (Identifier: NCT02888509; https://www.clinicaltrials.gov/ct2/show/NCT02888509?term=NCT02888509&rank=1).
Image acquisition and preprocessing
The resting-state functional magnetic resonance imaging data were obtained on a 3T GE DISCOVERY MR750 scanner (General Electric, Fairfield Connecticut, USA) equipped with a high-speed gradient and an 8 channel head coil. Foam pads and headphones were used to minimize head movement and scanner noise. Functional images were scanned using an echo-planar imaging sequence with the following parameters: repetition time (TR) = 2000 ms, echo time (TE) = 30 ms, slices = 43, matrix size = 64 × 64, flip angle = 90°, field of view = 240 × 240 mm2, voxel size = 3.75 × 3.75 ×3.2 mm3, thickness = 3.2 mm, no gap, and a total of 255 volumes.
Image preprocessing was performed using the DPARSF toolbox (http://rfmri.org/dpabi) based on SPM12 (http://www.fil.ion.ucl.ac.uk/spm/software/spm12/). The first five volumes were deleted before subsequent processing. The remaining 250 volumes were first slice-timing and head motion correction. Two BDd patients were excluded under the head motion criteria, that is, translation > 2.5 mm or rotation > 2.5°, in any direction. The data were then spatially normalized to the standard Montreal Neurological Institute space with a voxel size of 3 × 3 × 3 mm3. Nuisance covariates included the Friston-24 parameters of head motion, white matter, cerebrospinal fluid and whole-brain signal were regressed out from each voxel’s time course. The linear detrend and filtering (0.01–0.08 Hz) were performed to reduce the influence of low-frequency drift and high-frequency physiological noise. In addition, the framewise displacement (FD) across time points for each subject was calculated to assess head motion. Finally, using the ‘scrubbing’ method (Power et al., 2013) at an FD threshold of 0.5 mm, the ‘bad’ time points and their 1-back and 2-forward neighbors were identified and estimated through cubic spline interpolation. The number of these identified time points showed no difference among three groups (p = 0.91, one-way analysis of variance [ANOVA]). In detail, the mean number in BDd patients, MDD patients and HCs were 5.58 ± 11.12, 6.15 ± 11.79 and 6.66 ± 11.36, respectively.
Voxel-wise FCS
For each participant, we calculated the static voxel-wise FCS according to the following steps. First, Pearson’s correlation coefficients (r values) were computed between each voxel and all other voxels within the gray matter mask (probability > 20%) across a complete time series. Second, a threshold p < 0.001 (uncorrected) was set to eliminate weak correlations caused by noise, and the insignificant r values were set to zero. Third, these correlation r values were then transformed into z values using Fisher’s z-transformation to improve normality. Finally, the sum of absolute z values was obtained for each voxel as an index of static FCS.
Temporal variability of FCS
The sliding window correlation approach was utilized to measure the time-varying dynamic of FCS. According to our previous dynamic FC study (Pang et al., 2018), the sliding window length was set to 50 TRs (100 s) and the shift step size was 3 TRs (6 s), resulting in 66 windows across the scan time. For a given voxel, the FCS value was calculated in each window. Thus, a set of FCS values of the voxel was obtained. Finally, the temporal variance of FCS (i.e. dynamic FCS) was estimated by calculating the standard deviation of the FCS values across sliding windows.
Statistical analyses
For each subject, static and dynamic FCS maps were first standardized through division by mean values and then smoothed with a Gaussian kernel of full-width at half maximum = 8 mm. Statistical tests were performed on each FCS map across the three groups using one-way ANOVA, controlling for mean FD, age, sex and years of education. Multiple comparison correction was performed using AlphaSim program at cluster-level threshold p < 0.05 with voxel-wise threshold of uncorrected p < 0.005, and minimum cluster sizes of 54 and 41 connected voxels for static and dynamic FCS, respectively. Clusters that survived multiple comparison correction were selected as regions of interest (ROIs) for subsequent post hoc analysis. A two-tailed, two-sample t-test was conducted on these ROIs to determine the changes among the three groups (BDd vs MDD, BDd vs HC and MDD vs HC). A p < 0.0167 (0.05/3) (Bonferroni corrected) was considered statistically significant. Finally, to test the effect of medication, the Spearman correlation between the changed FCS and medication load was calculated (p < 0.05, uncorrected) (see Supplementary Material S1 for details).
Classification analysis
Classification analysis was implemented to test whether the combined static and dynamic FCSs achieve better classification performance than dynamic or static FCS alone for distinguishing BDd patients from HCs, MDD patients from HCs and BDd patients from MDD patients. Based on one-way ANOVA analysis, the static and dynamic FCS values in the clusters showing significant group differences were used as classification features. The sigmoid kernel function in the support vector machine classifier was employed in the LIBSVM library (Chang and Lin, 2011). Optimal parameters were selected using the grid search algorithm. To test the stability of these results, classifications were repeated using other popular methods such as linear discriminant analysis and decision tree analysis (see Supplementary Material S2). The leave-one-out cross-validation technique was used to obtain stable and reliable performance of the proposed model and prevent possible overfitting (Rashid et al., 2016). Finally, the accuracy, specificity and sensitivity were calculated from the average performance results.
A nonparametric permutation test was used to evaluate the statistical significance of classification accuracy. Briefly, the labels of the three groups were randomly permuted 5000 times and classification accuracy scores were recalculated each time. The p-value was defined as the proportion of estimated accuracy exceeding the null distribution values. A p < 0.05 was used as the cutoff for significance.
Predictive analysis
The relationships between clinical measures (i.e. SHAPS scores for anhedonia and PANAS-N scores for negative mood) and altered static and dynamic FCSs in the BDd and MDD groups were investigated using support vector regression with the radial basis function as the kernel function. Abnormal static and dynamic FCSs in clusters differing significantly between the BDd and HC groups were used as features to predict the clinical symptom severity in BDd patients. Similarly, the abnormal static and dynamic FCSs in clusters differing significantly between the MDD and HC groups were used as features to predict the clinical symptom severity in MDD patients. The leave-one-out cross-validation method was utilized to develop the prediction model. Correlation coefficients between real and predicted clinical scores were then calculated. Candidate features were ranked according to a previously proposed method (Chang and Lin, 2008) that considers how predictive performance is influenced without a feature. If removing a feature deteriorated the predictive performance, the feature was considered important, and the degree of importance was ranked according to the degree of deterioration. We used the correlation between real and predicted clinical scores as the performance measure, and each feature was ranked according to the correlation difference in the absence and presences of that feature.
Finally, the nonparametric permutation test was used to evaluate the statistical significance of the correlations. Clinical scores were randomly permuted 5000 times and the predicted scores obtained were used to calculate the correlation with real clinical scores. We then assigned p-value by calculating the proportion of estimated accuracy exceeding the null distribution values. A p < 0.05 was used as the cutoff for significance.
Results
Demographic and clinical data comparisons
Table 1 summarizes the detailed demographic and clinical characteristics of the participants. Three groups were matched in age, sex, years of education, handedness and mean FD. There were no significant differences between patients groups in clinical measures, that is, duration of depressive episodes, age of first onset, number of depression episode and HAMD scores. However, the total medication load in BDd patients was higher than MDD patients. As expected, both BDd and MDD patients had higher SHAPS scores (pBDd vs HCs = 0.0009; pMDD vs HCs = 0.001) and PANAS-N scores (pBDd vs HCs = 0.0002; pMDD vs HCs < 0.0001) than HCs, but there was no difference in SHAPS (p = 0.97) and PANAS-N (p = 0.25) scores between BDd and MDD groups.
Demographic characteristics and clinical variables of study participants.
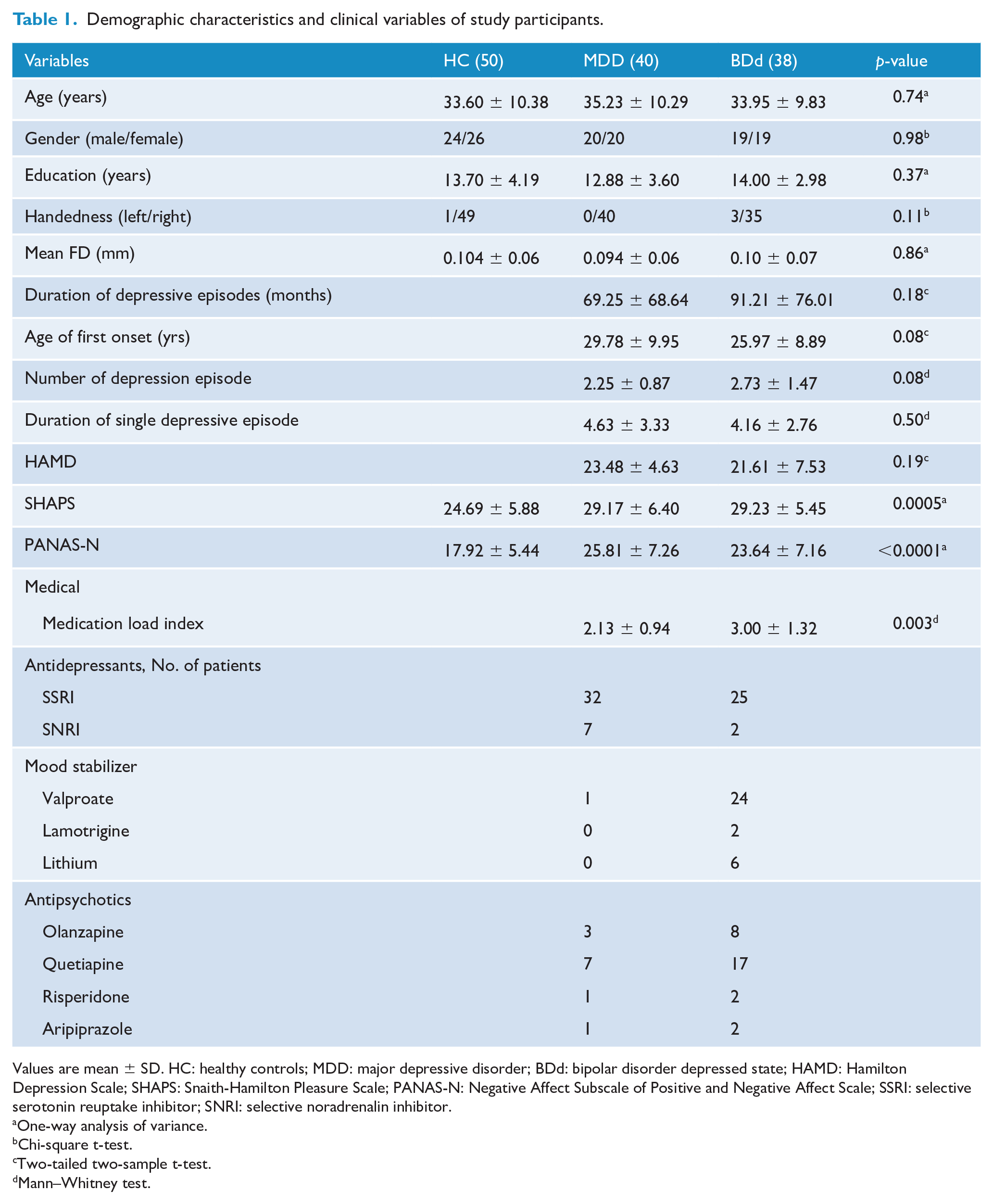
Values are mean ± SD. HC: healthy controls; MDD: major depressive disorder; BDd: bipolar disorder depressed state; HAMD: Hamilton Depression Scale; SHAPS: Snaith-Hamilton Pleasure Scale; PANAS-N: Negative Affect Subscale of Positive and Negative Affect Scale; SSRI: selective serotonin reuptake inhibitor; SNRI: selective noradrenalin inhibitor.
One-way analysis of variance.
Chi-square t-test.
Two-tailed two-sample t-test.
Mann–Whitney test.
FCS maps
Figure 1 presents the mean spatial distribution of the dynamic and static FCSs for all three groups. Regions in the bilateral posterior cingulate gyrus, precuneus, prefrontal cortex, visual cortex and posterior parietal cortex exhibited high FCS value and temporal change. The bilateral cerebellum (Crus I/II) also showed a high temporal change in FCS.
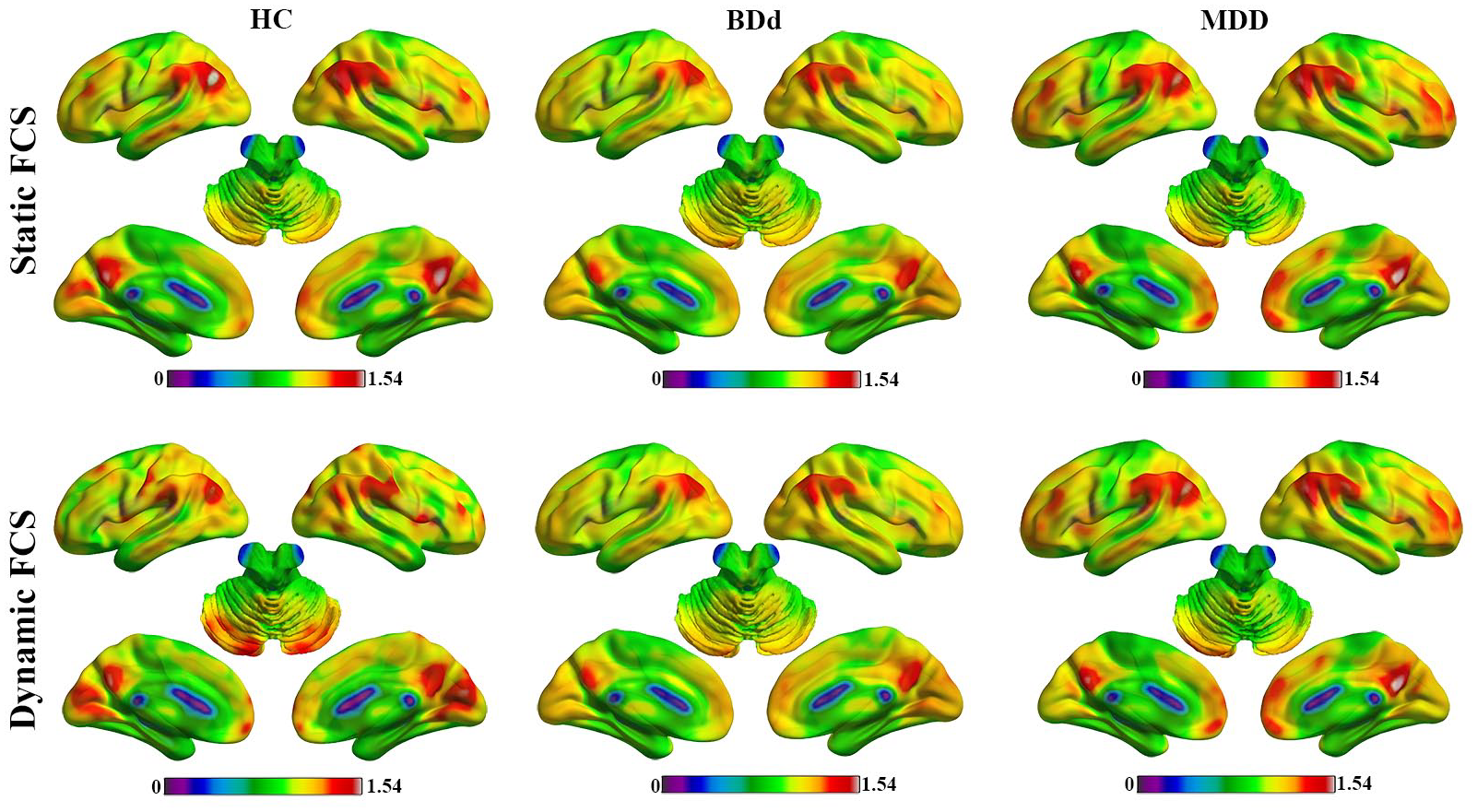
Mean static and dynamic FCS of HC, BDd and MDD groups.
Group differences in static FCS
One-way ANOVA revealed a significant group effect in several brain clusters, including the left medial orbitofrontal cortex (OFC), right caudate and thalamus (Figure 2). Post hoc t-tests revealed lower static FCS in left medial OFC of BDd patients and greater static FCS in left medial OFC of MDD patients, compared with HCs. The BDd patients also exhibited greater static FCS in the right caudate than HCs. In addition, BDd patients exhibited greater static FCS in the right thalamus compared with both MDD patients and HCs. These abnormal static FCSs in the left medial OFC, right caudate and thalamus were not significantly correlated with medication load (Supplementary Tables S2 and S3).
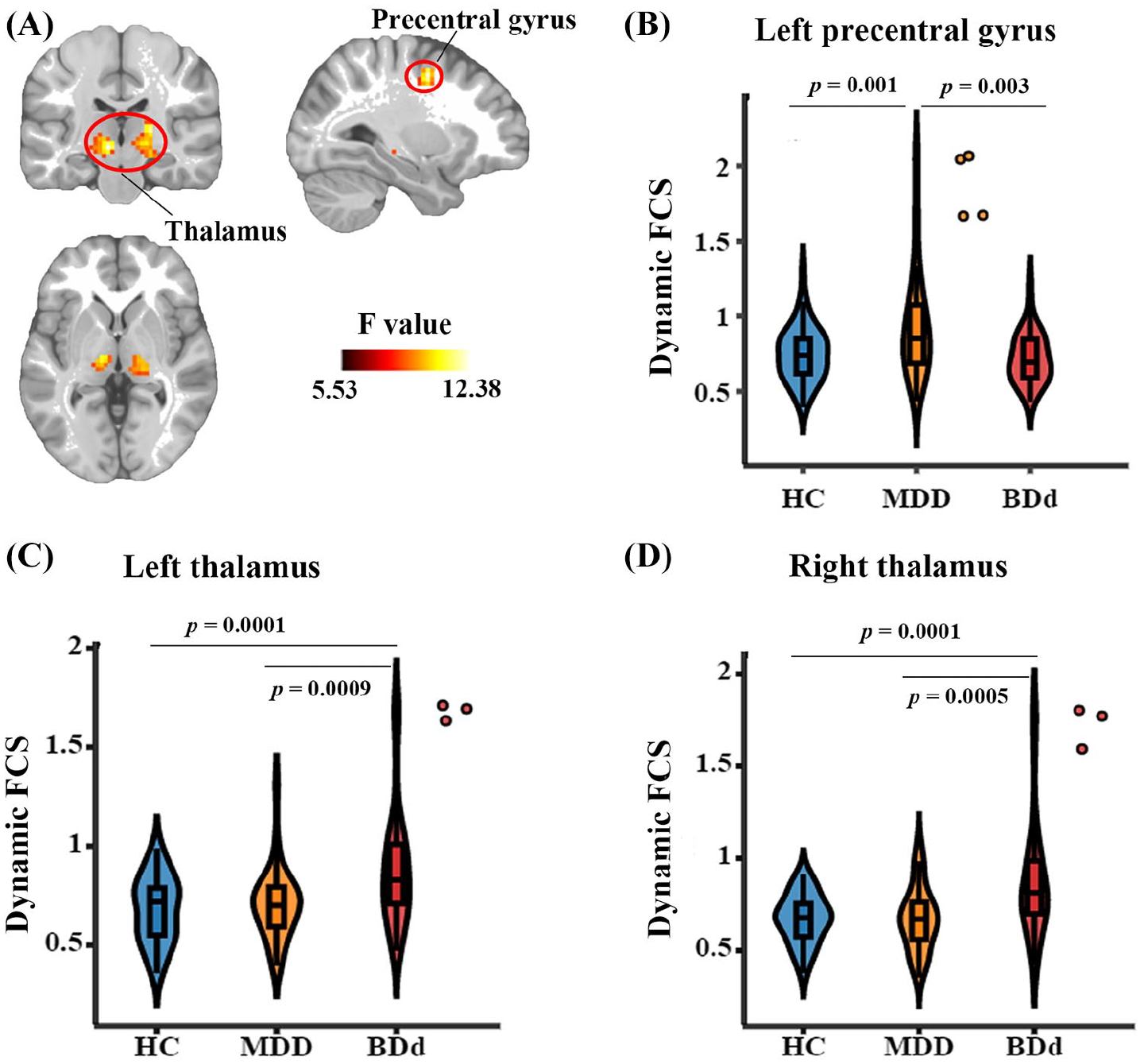
Differences in static FCS among the HC, BDd and MDD groups. (A) Left medial OFC (MNI coordinate: [–3, 45, –12]; cluster size = 64 voxels), right caudate (MNI coordinate: [15, 24, 3]; cluster size = 78 voxels) and right thalamus (MNI coordinate: [21, –24, 6]; cluster size = 196 voxels) showed significant group effects (voxel level p < 0.005, cluster level p < 0.05, AlphaSim corrected, one-way ANOVA). Color bar represents the F values. The brain map was visualized using MRIcroN. Post hoc analyses revealed greater FCS in the caudate (B) of BDd patients versus HCs. FCS in the medial OFC (C) was stronger in MDD patients and weaker in BDd patients, compared with HCs. Furthermore, FCS in the thalamus (D) was greater in BDd patients than in either the HC or MDD groups.
Group differences in dynamic FCS
One-way ANOVA revealed a significant group effect in the left precentral gyrus and bilateral thalamus (Figure 3), and post hoc t-tests indicated greater dynamic FCS in the left precentral gyrus of MDD patients, compared with BDd patients and HCs. Increased dynamic FCSs in the bilateral thalamus were found in the BDd patients, compared with MDD patients and HCs. These abnormal dynamic FCSs were not significantly correlated with medication load (Supplementary Tables S2 and S3).
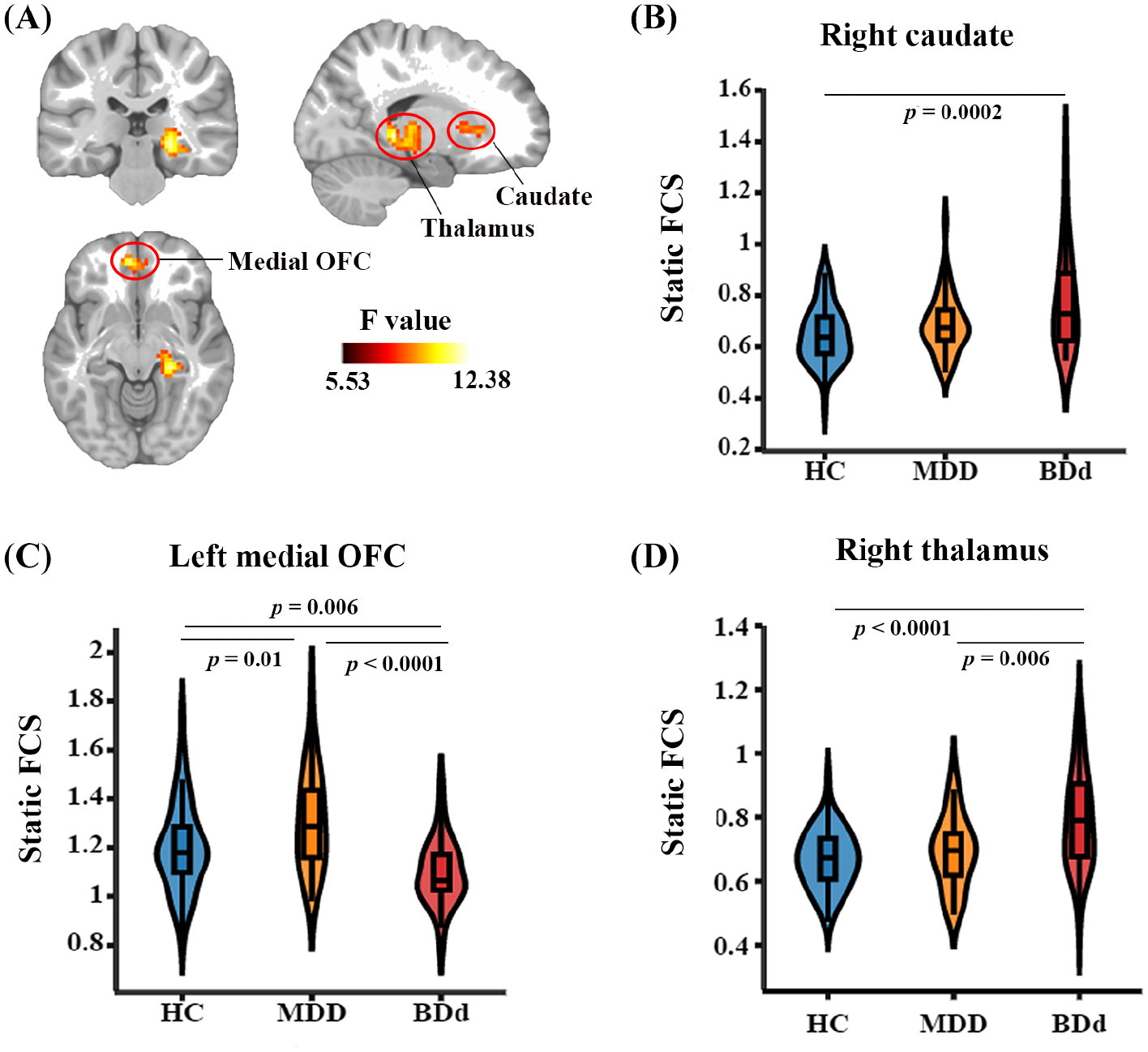
Differences in dynamic FCS among the HC, BDd and MDD groups. (A) Left precentral gyrus (MNI coordinate: [–30, –3, 48]; cluster size = 52 voxels), left thalamus (MNI coordinate: [–12, –18, 0]; cluster size = 82 voxels) and right thalamus (MNI coordinate: [21, –21, 6]; cluster size = 131 voxels) showed significant group effects (voxel level p < 0.005, cluster level p < 0.05, AlphaSim corrected, one-way ANOVA). Color bar represents the F values. The brain map was visualized using MRIcroN. Post hoc analyses revealed greater FCS variability in the left precentral gyrus (B) of MDD patients versus HCs and BDd patients, as well as greater FCS variability in the bilateral thalamus (C/D) of BDd patients versus HCs and MDD patients. The violin plots are described in Figure 2.
Classification result
As shown in Table 2, for each classification analysis between BDd and MDD patients, BDd patients and HCs, and MDD patients and HCs, the combination of dynamic and static FCSs in clusters with significant group differences exhibited higher accuracy than either dynamic or static FCS alone.
Performance evaluation of classifier using different FCS measures.
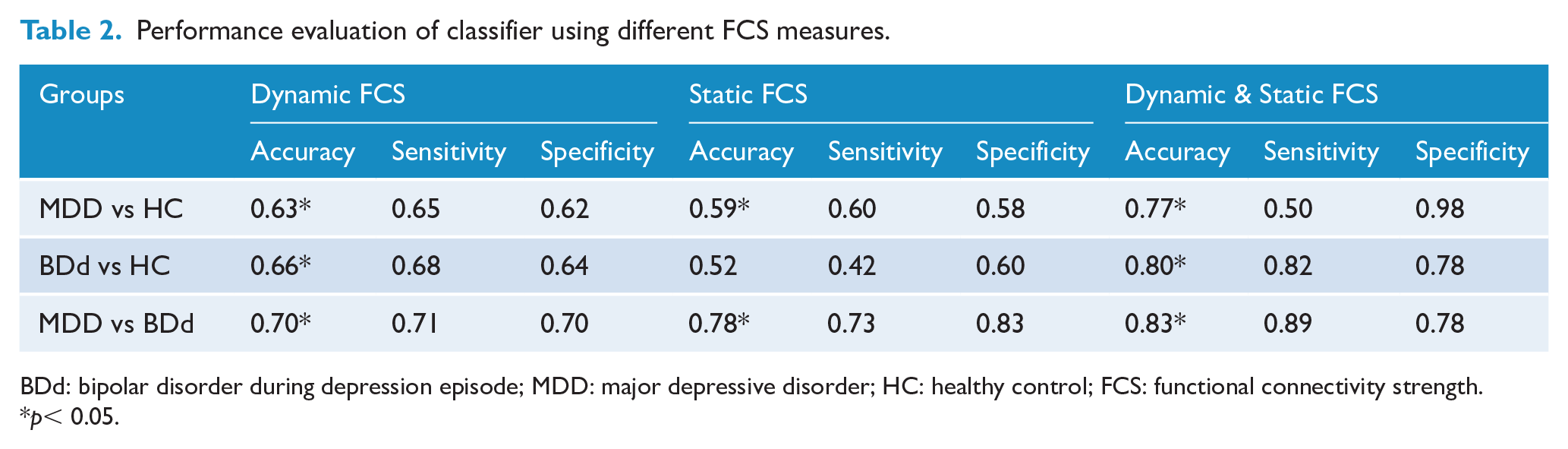
BDd: bipolar disorder during depression episode; MDD: major depressive disorder; HC: healthy control; FCS: functional connectivity strength.
p< 0.05.
Prediction of symptom severity
In BDd patients, the abnormal static and dynamic FCSs in the frontal–striatum–thalamic (FST) circuit collectively predicted SHAPS scores, a measure of anhedonia severity, and there was a strong correlation between real SHAPS scores and predicted SHAPS scores (r = 0.56, p = 0.001) (Figure 4[A]). Altered static FCS in the caudate and altered dynamic FCS in the thalamus made greater contributions to SHAPS score prediction than static FCSs in the thalamus and frontal regions. In MDD patients, abnormal static FCS in the DMN and dynamic FCS in the sensorimotor network (SMN) predicted PANAS-N scores, a measure of negative mood, and there was a strong correlation between real PANAS-N scores and predicted PANAS -N scores (r = 0.53, p = 0.002) (Figure 4[B]). The dynamic FCS in the SMN region made a greater contribution to PANAS-N score prediction than static FCS in the DMN.
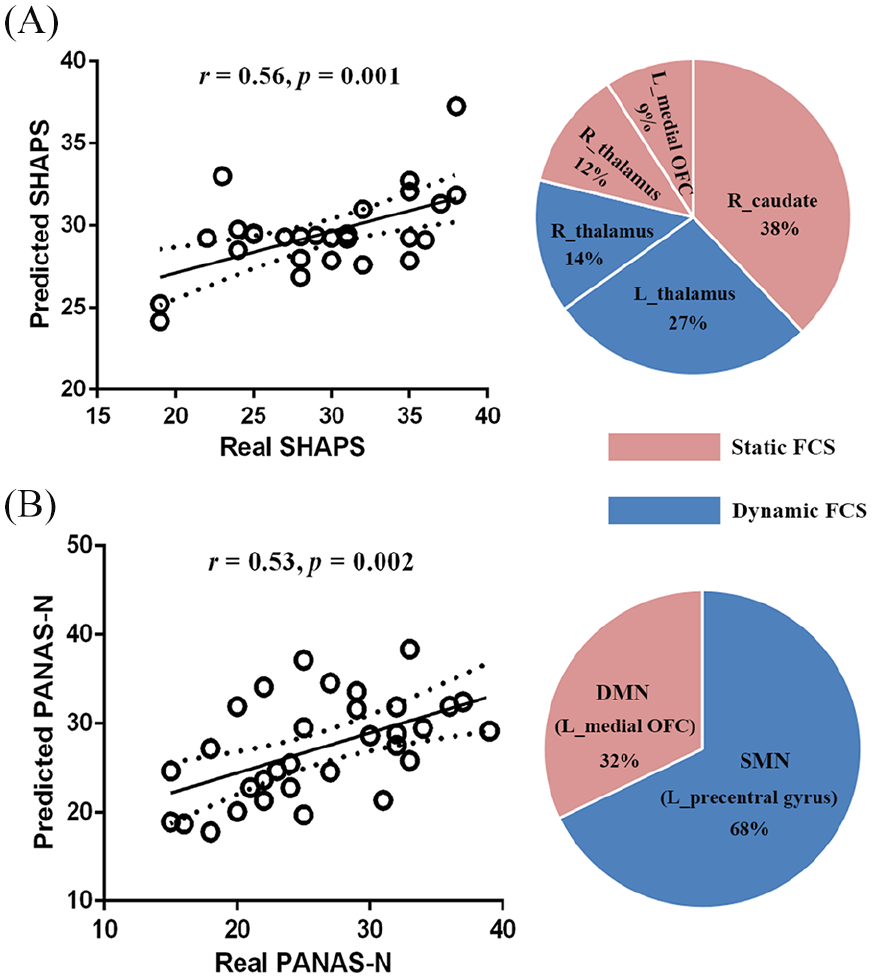
Combined static and dynamic FCSs in FST circuits predict the severity of anhedonia (SHAPS score) among BDd patients (A), while combined static and dynamic FCSs in the DMN and SMN predict the severity of negative mood (PANAS-N score) among MDD patients (B).
Discussion
This is the first study to examine altered whole-brain FC in BDd and MDD patients by combined static and dynamic measurements. These analyses revealed abnormal static and dynamic FCSs in the FST circuit of BDd patients and in the DMN/SMN of MDD patients. Consistent with our hypothesis, the static and dynamic FCSs provided complementary information for characterizing brain communication in these patient groups. Indeed, combined static and dynamic FCS measures improved the ability to differentiate BDd from MDD than static or dynamic FCS measure alone, and also predicted anhedonia in BDd and negative mood in MDD. These results provide evidence for disorder-specific abnormal connectivity patterns in MDD and BDd patients, suggesting distinct underlying pathophysiological mechanisms.
BDd-specific FC pattern
The BDd group exhibited greater static and dynamic FCSs in the thalamus, greater static FCS in the caudate and reduced static FCS in the medial OFC, suggesting disturbed integrity of FST connectivity in the resting state. The thalamus participates in the FST circuit by relaying striatal inputs to the frontal regions and providing feedback to the striatum (Metzger et al., 2013). In addition, dynamic FC of the thalamus promotes integration of information across the FST loop (Preti and Ville, 2017). Elevated temporal variation in thalamic connectivity is consistent with the notion that primary abnormalities in the FST circuit are related to abnormal emotional and cognitive processing in BDd (Teng et al., 2014). The FST circuit is a major target of dopaminergic neurotransmission (Ashok et al., 2017). The alterations in this circuit are related to dopaminergic dysfunction and influence the goal-directed actions and behavioral flexibility (Kesby et al., 2018). The medial OFC–striatum–thalamus circuit, in particular, is critical for processing of reward (Der-Avakian and Markou, 2012), and abnormalities within this circuit may lead to anhedonia in BDd (Satterthwaite et al., 2015). This association is supported by the significant correlation between abnormal connectomics in the FST circuit and anhedonia scale scores in BDd patients. Collectively our findings provide further evidence for atypical FST connectivity underlying BDd symptomatology, and thus, identify this circuit as a potential target for BDd treatment. Additional studies are required to examine whether this circuit disruption is also present in other psychiatric disorders such as schizophrenia that shares clinical symptoms with bipolar disorder and both disorders are associated with dysconnectivity (Rashid et al., 2014).
MDD-specific FC pattern
In contrast to BDd patients and HCs, MDD patients exhibited greater static FCS in the DMN and dynamic FCS in the SMN. The DMN is implicated in self-related processing (Buckner et al., 2008). Increased DMN connectivity likely leads to rumination and increased self-focus in MDD (Zhu et al., 2012). At the same time, MDD patients also exhibit psychomotor retardation clinically (Buyukdura et al., 2011), which is related to abnormal structure and function in the SMN (Bracht et al., 2012; Yin et al., 2018). The increased overall connectivity of the DMN and increased dynamic connectivity of the SMN might suggest an imbalance between the DMN and SMN activity in MDD, which may be caused by abnormal serotonergic signaling (Conio et al., 2020). This imbalance may also result in excessive self-focus and psychomotor retardation in depressive patients (Martino et al., 2016). Moreover, the increased FC in the DMN, especially the increased dynamic FC in the SMN also predicted the severity of negative mood in the MDD group. Greater dynamic FC of the SMN indicated higher temporal instability of SMN connectivity in MDD. In fact, abnormal connectivity in the SMN has been identified in different connectivity states in MDD (Zhi et al., 2018). Alternatively, decreased dynamic connectivity in the SMN regions was found in post-traumatic stress disorder patients (Jin et al., 2017). Therefore, the dynamic property of SMN connectivity may be a valuable biomarker to differentiate MDD from other mood disorders such as BDd and post-traumatic stress disorder. Taken together, we speculate that the abnormal static and dynamic FC in the DMN and SMN of MDD patients may reflect compensatory or antagonistic processes leading to excessive self-focus and psychomotor retardation, both of which likely contribute to negative mood.
Static and dynamic FC patterns
Our findings also indicate that the combination of static and dynamic FCS provides more useful information for discriminating BDd from MDD than static or dynamic FCS alone, in agreement with previous studies demonstrating that static and dynamic FCS methods provide complementary information about brain communication (Kaiser et al., 2016; Pang et al., 2018). While static FC represents overall mean FC, dynamic FC provides information about how FC changes over time. Therefore, combining static and dynamic FC is recommended for future neuroimaging studies.
Limitations
Our findings must be considered in light of several limitations. First, our sample size is relatively small, limiting the power to detect subtle group differences and significant correlations between brain connectivity and clinical variables. Second, most depressive patients in our study were taking psychotropic medications, and the medication load index differed between BDd and MDD groups. This is inevitable as these patients require disorder-specific treatment regimens for depression. Although we found no correlations with medication load index (Supplementary Material S1), it is difficult to completely exclude the effects of medication on resting-state FC. Thus, well-designed studies with larger numbers of medication-free patients are needed to confirm the presence of these distinct FC abnormalities in MDD and BDd.
Conclusion
The present study demonstrates that combined whole-brain static and dynamic FCS analyses are able to generate diagnostic and predictive models. The distinct FC patterns in BDd and MDD patients were revealed to be associated with symptom severity, strongly suggesting direct relevance to disorder development and expression. Abnormal FC in FST circuit was associated with anhedonia in BDd patients, whereas abnormal FC in the DMN and SMN was associated with negative mood in MDD patients. Our current findings highlight the potential of combined static and dynamic FC for identifying reliable diagnostic biomarkers and unique neuropathological mechanisms underlying MDD and BDd.
Supplemental Material
S1 – Supplemental material for Combined static and dynamic functional connectivity signatures differentiating bipolar depression from major depressive disorder
Supplemental material, S1 for Combined static and dynamic functional connectivity signatures differentiating bipolar depression from major depressive disorder by Yajing Pang, Huangbin Zhang, Qian Cui, Qi Yang, Fengmei Lu, Heng Chen, Zongling He, Yifeng Wang, Jiaojian Wang and Huafu Chen in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This work was supported by the Key Project of Research and Development of Ministry of Science and Technology (2018AAA0100705), the Natural Science Foundation of China (61533006, U1808204 and 81771919), the funded by Guizhou University (702570183301) and the China Postdoctoral Science Foundation Grant (2019M653383).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
