Abstract
Objective:
We aimed to investigate sex-dependent alterations in resting-state relative cerebral blood flow, amplitude of low-frequency fluctuations and relative cerebral blood flow–amplitude of low-frequency fluctuations coupling in patients with schizophrenia.
Method:
Resting-state functional magnetic resonance imaging and three-dimensional pseudo-continuous arterial spin labeling imaging were performed to obtain resting-state amplitude of low-frequency fluctuations and relative cerebral blood flow in 95 schizophrenia patients and 99 healthy controls. Sex differences in relative cerebral blood flow and amplitude of low-frequency fluctuations were compared in both groups. Diagnostic group differences in relative cerebral blood flow, amplitude of low-frequency fluctuations and relative cerebral blood flow–amplitude of low-frequency fluctuations coupling were compared in male and female subjects, respectively.
Results:
In both healthy controls and schizophrenia patients, the males had higher relative cerebral blood flow in anterior brain regions and lower relative cerebral blood flow in posterior brain regions than did the females. Compared with multiple regions exhibiting sex differences in relative cerebral blood flow, only the left middle frontal gyrus had a significant sex difference in amplitude of low-frequency fluctuations. In the females, schizophrenia patients exhibited increased relative cerebral blood flow and amplitude of low-frequency fluctuations in the basal ganglia, thalamus and hippocampus and reduced relative cerebral blood flow and amplitude of low-frequency fluctuations in the frontal, parietal and occipital regions compared with those of healthy controls. However, there were fewer brain regions with diagnostic group differences in the males than in the females. Brain regions with diagnostic group differences in relative cerebral blood flow and amplitude of low-frequency fluctuations only partially overlapped. Only the female patients exhibited increased relative cerebral blood flow–amplitude of low-frequency fluctuations couplings compared with those of healthy females.
Conclusion:
The alterations in the relative cerebral blood flow and amplitude of low-frequency fluctuations in schizophrenia are sex-specific, which should be considered in future neuroimaging studies. The relative cerebral blood flow and amplitude of low-frequency fluctuations have different sensitivity in detecting changes in neuronal activity in schizophrenia and can provide complementary information.
Keywords
Introduction
Schizophrenia is a devastating neuropsychiatric disorder characterized by the deterioration of cognitive and emotional processing. Schizophrenia is associated with alterations in the resting-state brain activity, which can be measured by either regional cerebral blood flow (CBF) (Pinkham et al., 2011; Scheef et al., 2010) or amplitude of low-frequency fluctuations (ALFF) of the blood-oxygen-level-dependent (BOLD) signals (Hoptman et al., 2010; Turner et al., 2013). All of the previous studies have separately investigated regional CBF or ALFF changes in schizophrenia; however, none of these studies have assessed whether these two measures can provide complementary information in exploring the underlying mechanisms of schizophrenia.
The regional CBF has been traditionally measured by positron emission tomography (PET) and single photon emission computerized tomography (SPECT). Recently, some studies have used arterial spin labeling (ASL) to detect regional CBF changes in schizophrenia (Pinkham et al., 2011; Scheef et al., 2010). The ALFF developed by Zang et al. (2007) is a method to examine the amplitude of low-frequency (0.01–0.08 Hz) oscillations of BOLD signals of the resting-state functional magnetic resonance imaging (fMRI) data. The ALFF is an indirect measure of spontaneous neural activity (Logothetis et al., 2001; Zang et al., 2007), and it has been used to explore functional deficits in schizophrenia (Hoptman et al., 2010; Turner et al., 2013; Yu et al., 2014). The ALFF and CBF are supposed to be correlated with each other because both indices reflect hemodynamic changes. For example, as the most active network during task-free state, brain regions of the default mode network (DMN) exhibit both higher CBF and greater ALFF (Zou et al., 2009). Moreover, most brain regions have shown regional CBF–ALFF coupling in healthy subjects (Li et al., 2012). However, it remains unknown whether this regional CBF–ALFF coupling is altered in schizophrenia.
Sex differences in the CBF of the brain have been repeatedly revealed in both healthy subjects and patients with schizophrenia (Gur and Gur, 1990; Rodriguez et al., 1988). Although sex differences in ALFF have been reported in healthy subjects (Biswal et al., 2010), this difference has never been investigated in schizophrenia. In this study, we aimed to characterize sex differences in CBF and ALFF in both groups and to investigate sex-dependent alterations in CBF, ALFF and CBF–ALFF coupling in the same group of schizophrenia patients.
Materials and methods
Participants
A total of 105 patients with schizophrenia and 100 healthy controls were included in our study. Diagnoses of patients were confirmed using the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM-IV). Inclusion criteria included age (15–60 years) and right-handedness. Exclusion criteria for both patients and healthy controls included magnetic resonance imaging (MRI) contraindications, poor image quality and the presence of a systemic medical illness, a central nervous system (CNS) disorder, or a history of head trauma, or substance abuse within the last 3 months or lifetime history of substance abuse or dependence. Additional exclusion criteria for healthy controls were history of any Axis I or II disorders and a first-degree relative with a psychotic disorder. A total of 10 patients and 1 healthy subject were excluded due to poor image quality. The final sample included 95 schizophrenia patients and 99 healthy controls. The clinical data of 1 patient were lost except the age and gender. One patient could not remember her antipsychotic medication accurately. In all, 85 patients were receiving medications at the time of the MRI examinations and 8 patients had never received any medications. The antipsychotic dosages were reported as chlorpromazine equivalents and were calculated based on clinically equivalent dosing estimates (Gardner et al., 2010). For each schizophrenia patient, the chlorpromazine equivalent was estimated according to the antipsychotic drugs and dosages used in the latest week before the MRI scan. The Positive and Negative Syndrome Scale (PANSS) was used to quantify the clinical symptoms (Kay et al., 1987). This study was approved by the Medical Research Ethics Committee of Tianjin Medical University General Hospital, and all participants provided written informed consent.
Image data acquisition
MRI was performed using a 3.0-Tesla MR system (Discovery MR750; General Electric, Milwaukee, WI, USA). Tight but comfortable foam padding was used to minimize head motion, and ear plugs were used to reduce scanner noise. Sagittal three-dimensional (3D) T1-weighted images were acquired by a brain volume (BRAVO) sequence with the following parameters: repetition time (TR) = 8.2 ms, echo time (TE) = 3.2 ms, inversion time (TI) = 450 ms, flip angle (FA) = 12°, field of view (FOV) = 256 mm × 256 mm, matrix = 256 × 256, slice thickness = 1 mm, no gap, and 188 sagittal slices. The fMRI data were obtained using a gradient-echo single-shot echo planar imaging (EPI) with the following parameters: TR/TE = 2000/45 ms, FOV = 220 mm × 220 mm, matrix = 64 × 64, FA = 90°, slice thickness = 4 mm, gap = 0.5 mm, 32 interleaved transverse slices and 180 volumes. During fMRI scans, all patients were instructed to keep their eyes closed, to relax, to move as little as possible and not to fall asleep. The resting-state perfusion imaging was performed using a pseudo-continuous arterial spin labeling (pcASL) sequence with a 3D fast spin-echo acquisition and background suppression: TR = 4886 ms, TE = 10.5 ms, post-label delay = 2025 ms, spiral in readout of eight arms with 512 sample points, FA = 111°, FOV = 240 mm × 240 mm, reconstruction matrix = 128 × 128, slice thickness = 4 mm and 40 axial slices and 1.9 mm × 1.9 mm in-plane resolution. A pair of the label and control whole-brain image volumes required 16 TRs (Xu et al., 2010). A total of three pairs of label or control images were acquired and averaged to calculate ASL difference images.
Data processing
We calculated the relative CBF (rCBF) and ALFF from the pcASL images and resting-state fMRI images, respectively. The detailed steps are described in the supplementary data.
Statistical analyses
Two-way analysis of covariance (ANCOVA) controlling for age was used to estimate the sex differences in ALFF or rCBF in each diagnostic group and the diagnostic group differences in ALFF or rCBF in each sex group. These statistical analyses were performed in a voxel-wise manner using the SPM8 software. Multiple comparisons for these analyses were corrected using a false discovery rate (FDR) (p < 0.05). Beforehand, one-sample t-test was performed on ALFF to estimate the distribution of ALFF in eachdiagnostic group. Multiple comparisons for the analysis were corrected using a family-wise error (FWE) method (p < 0.05). For an individual diagnostic group, the rCBF–ALFF coupling was calculated using partial correlation analysis controlling for age, which was only performed for brain regions with significant intergroup differences in ALFF or rCBF. The correlation coefficients were further converted into z values using Fisher r–z transformation to increase the normality. A z-test was used to compare the differences in rCBF–ALFF coupling between schizophrenia and healthy controls groups. A significant threshold of p < 0.05 was used for this analysis.
To test whether the sex-specific changes in ALFF or rCBF were correlated with the clinical variables in schizophrenia, we extracted the ALFF and rCBF values of brain regions with significant group differences in the males and the females, respectively. Spearman’s correlation coefficients were used to test correlations between these ALFF or rCBF values and PANSS subscores in male or female schizophrenia patients. An uncorrected threshold of p < 0.05 was used for this analysis.
Results
Demographic and clinical characteristics of subjects
The demographic and clinical characteristics of schizophrenia patients and healthy controls are summarized in Table 1. Neither the males (t = 0.945; p = 0.347) nor the females (t = 0.600; p = 0.550) exhibited significant differences in age between schizophrenia patients and healthy controls. Sex differences in clinical variables in schizophrenia patients are shown in Table 2. Male and female patients did not differ in antipsychotic dosage (t = 0.281; p = 0.780), duration of illness (t = 1.7; p = 0.092) and PANSS scores (total score: t = 0.499; p = 0.619; positive score: t = 0.422; p = 0.674; negative score: t = 0.695; p = 0.489; general score: t = 0.190; p = 0.850).
Demographic and clinical characteristics of schizophrenia patients and healthy controls.
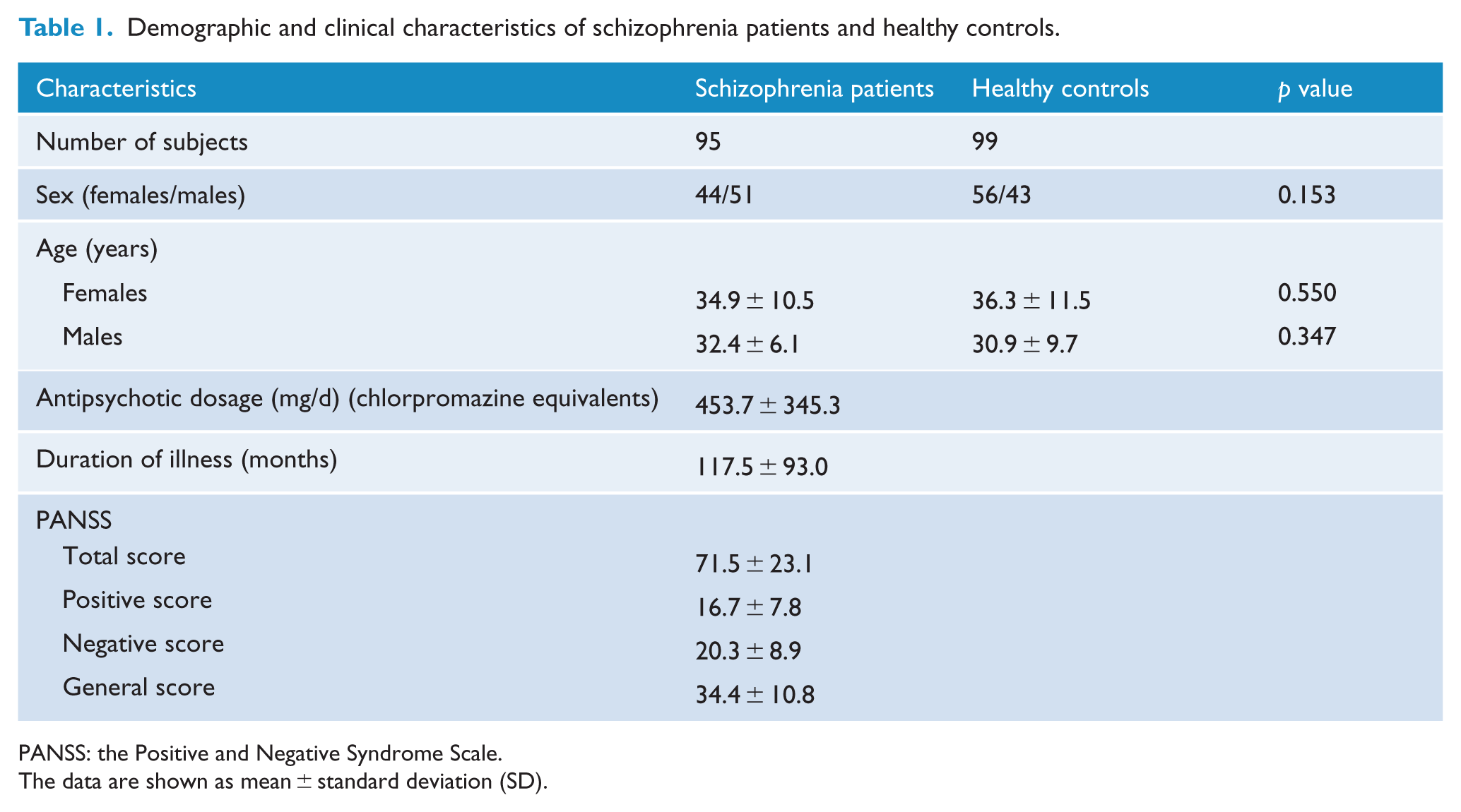
PANSS: the Positive and Negative Syndrome Scale.
The data are shown as mean ± standard deviation (SD).
Comparisons in clinical characteristics between male and female patients with schizophrenia.
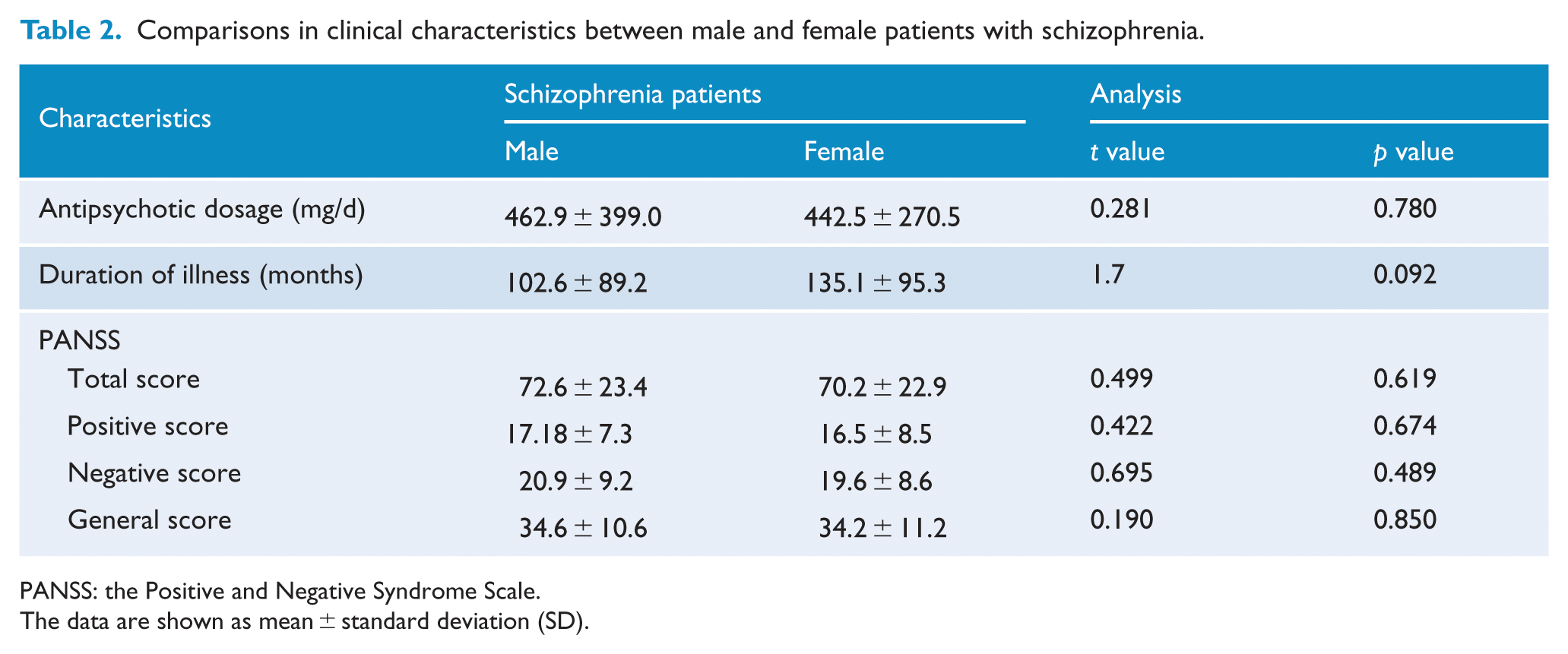
PANSS: the Positive and Negative Syndrome Scale.
The data are shown as mean ± standard deviation (SD).
Sex differences in rCBF and ALFF
The sex differences in rCBF (p < 0.05, FDR corrected) are shown in Figure 1. In both healthy controls and schizophrenia patients, male subjects had higher rCBF in anterior brain regions (i.e. frontal and temporal cortices and deep nuclei) and lower rCBF in posterior brain regions (i.e. occipital and parietal cortices) compared with female subjects. However, the spatial extent and the strength of sex differences in rCBF were less prominent in schizophrenia patients compared to healthy controls.
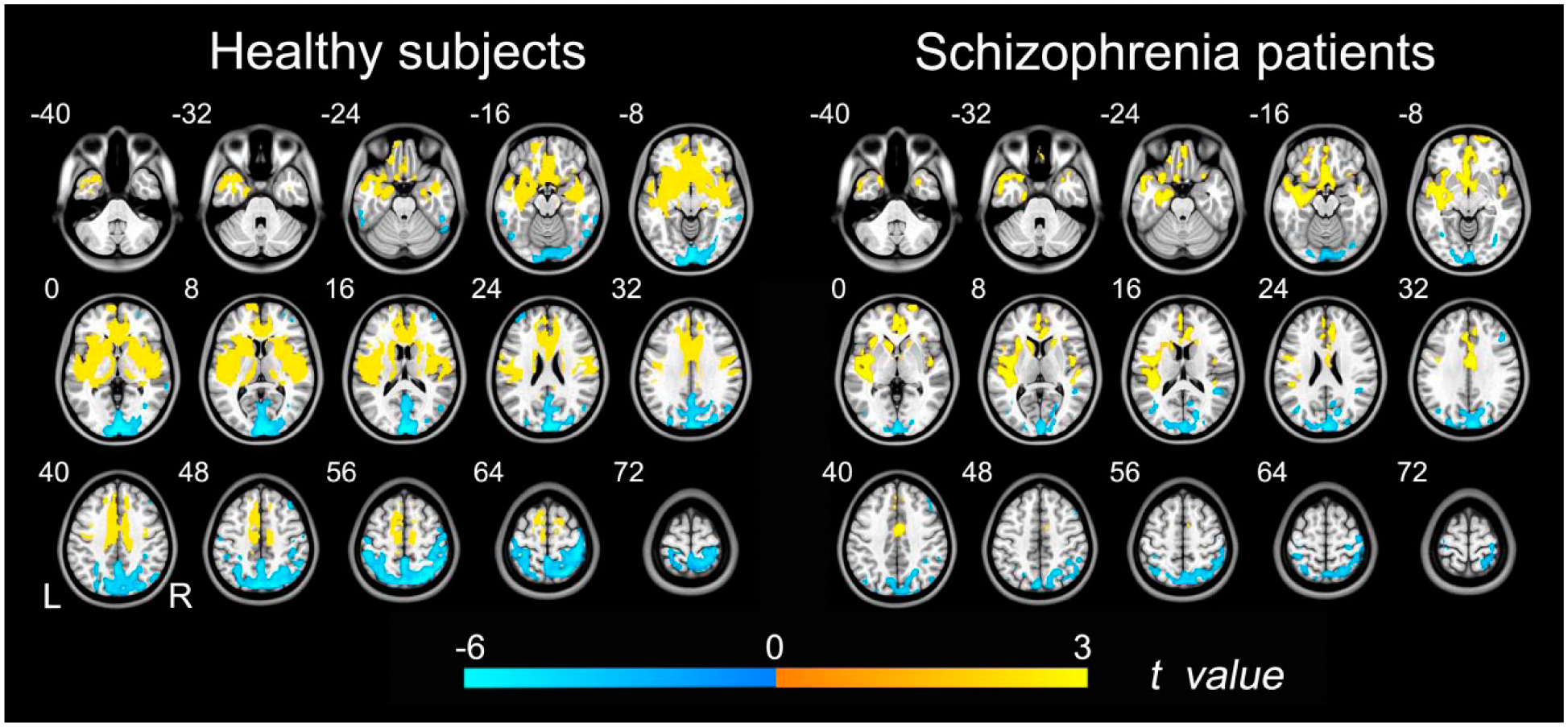
Brian regions with sex differences in rCBF. Left row shows brain regions with significant differences in rCBF between healthy male and female subjects (p < 0.05, FDR corrected). Right row shows brain regions with significant sex differences in rCBF in patients with schizophrenia (p < 0.05, FDR corrected). The warm color represents the significantly higher rCBF in males. The cold color represents the higher rCBF in females.
The distribution of ALFF in healthy controls and schizophrenia patients is presented in Supplementary Figure 1. We noted that brain regions with significant sex differences in ALFF were much less than those with significant sex differences in rCBF. Although the male patients had a higher ALFF (p < 0.05, FDR corrected) in the left middle frontal gyrus (MFG) than the female patients (Figure 2), we did not find any brain region that showed significant sex differences in ALFF in healthy controls.
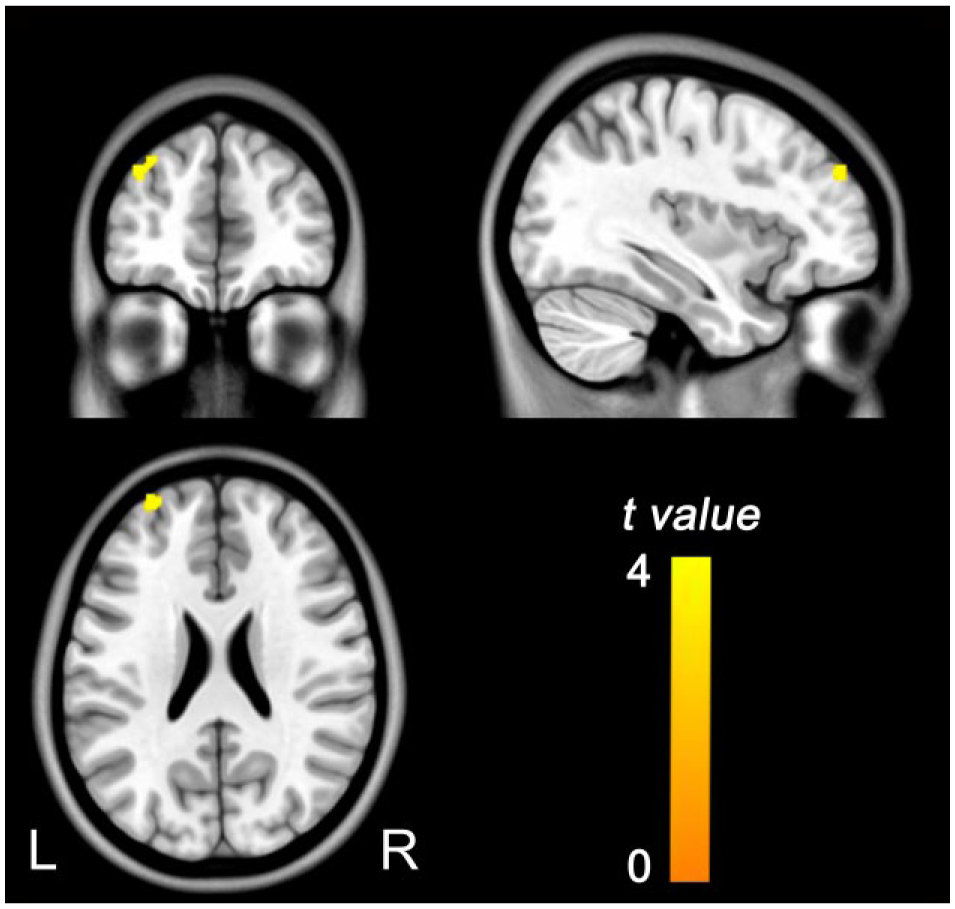
Brian regions with sex differences in ALFF in schizophrenia patients. The warm color represents the significantly higher ALFF in males (p < 0.05, FDR corrected).
Sex-dependent group differences in rCBF and ALFF
Although in the two-way ANCOVA model we did not find any brain regions that exhibited significant diagnostic group × gender interaction (p < 0.05, FDR corrected) in either rCBF or ALFF, we directly used different contrasts to further evaluate sex-dependent intergroup differences.
Group differences in rCBF between schizophrenia patients and healthy subjects (p < 0.05, FDR corrected) are shown in Figure 3 and Supplementary Table 1. Compared to healthy subjects, both male and female patients with schizophrenia exhibited significantly decreased rCBF in the medial prefrontal cortex (MPFC), anterior cingulate cortex (ACC), insula and dorsolateral prefrontal cortex (DLPFC), but the diagnostic group differences in rCBF were involved in a larger spatial extent in the females than in the males. Moreover, female patients also exhibited rCBF decreases in the occipital and parietal cortices compared with those of the female controls. Compared to the healthy female subjects, the female patients with schizophrenia showed increased rCBF in the bilateral temporal cortex, sensorimotor cortex, basal ganglia and thalamus. None of the brain regions had higher rCBF in the male patients compared with the male controls.
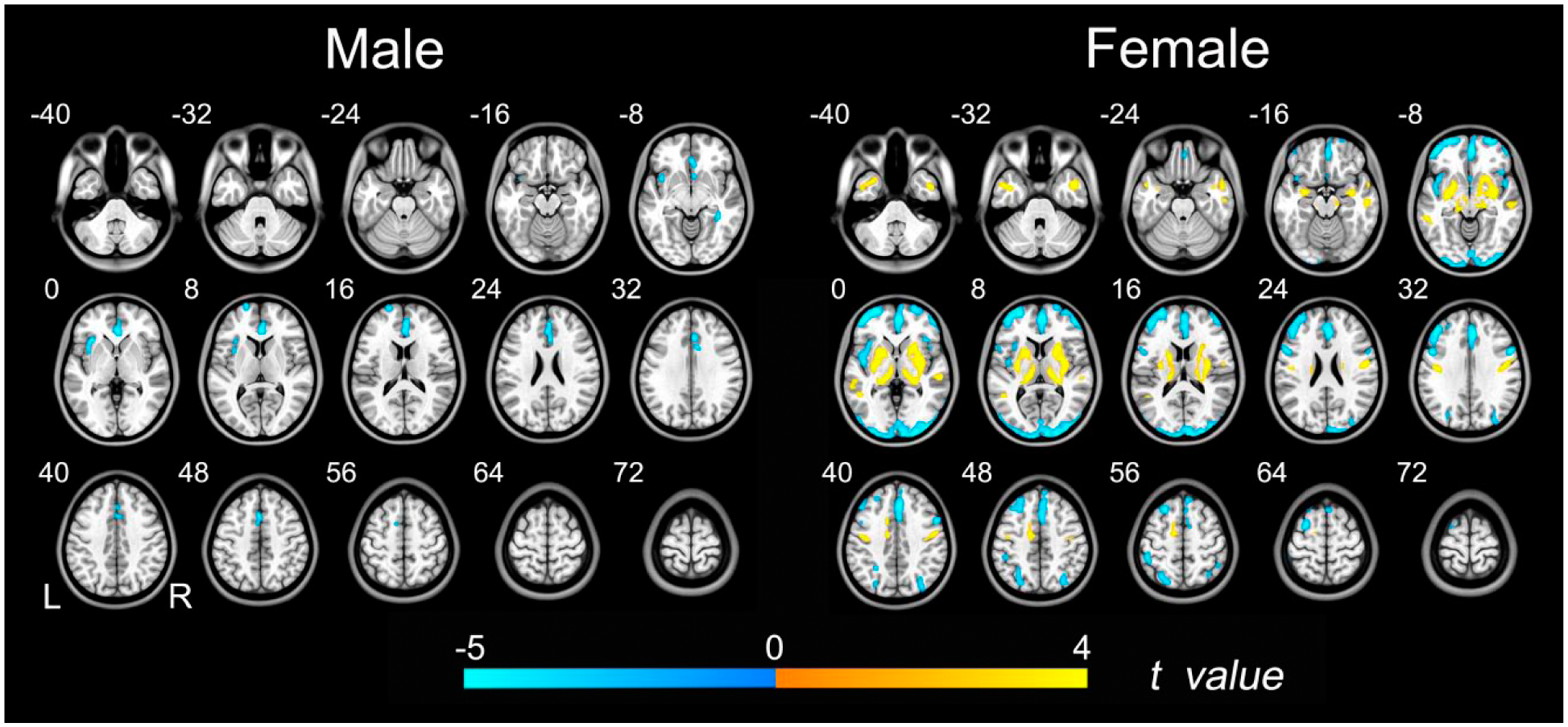
Group differences in rCBF between schizophrenia patients and healthy controls. Left row shows brain regions with significant group differences in males (p < 0.05, FDR corrected). Right row shows brain regions with significant group differences in females (p < 0.05, FDR corrected). The warm color represents the significantly increased rCBF in patients with schizophrenia. The cold color denotes that the rCBF was significantly decreased in schizophrenia patients.
Group differences in ALFF between the schizophrenia patients and the healthy subjects (p < 0.05, FDR corrected) are shown in Figure 4 and Supplementary Table 2. Generally, schizophrenia patients had increased ALFF in the anterior brain regions (deep nuclei and temporal cortices), but the distribution was stronger and wider in the female subjects. The female patients also had decreased ALFF in the posterior brain regions (occipital and parietal cortices).
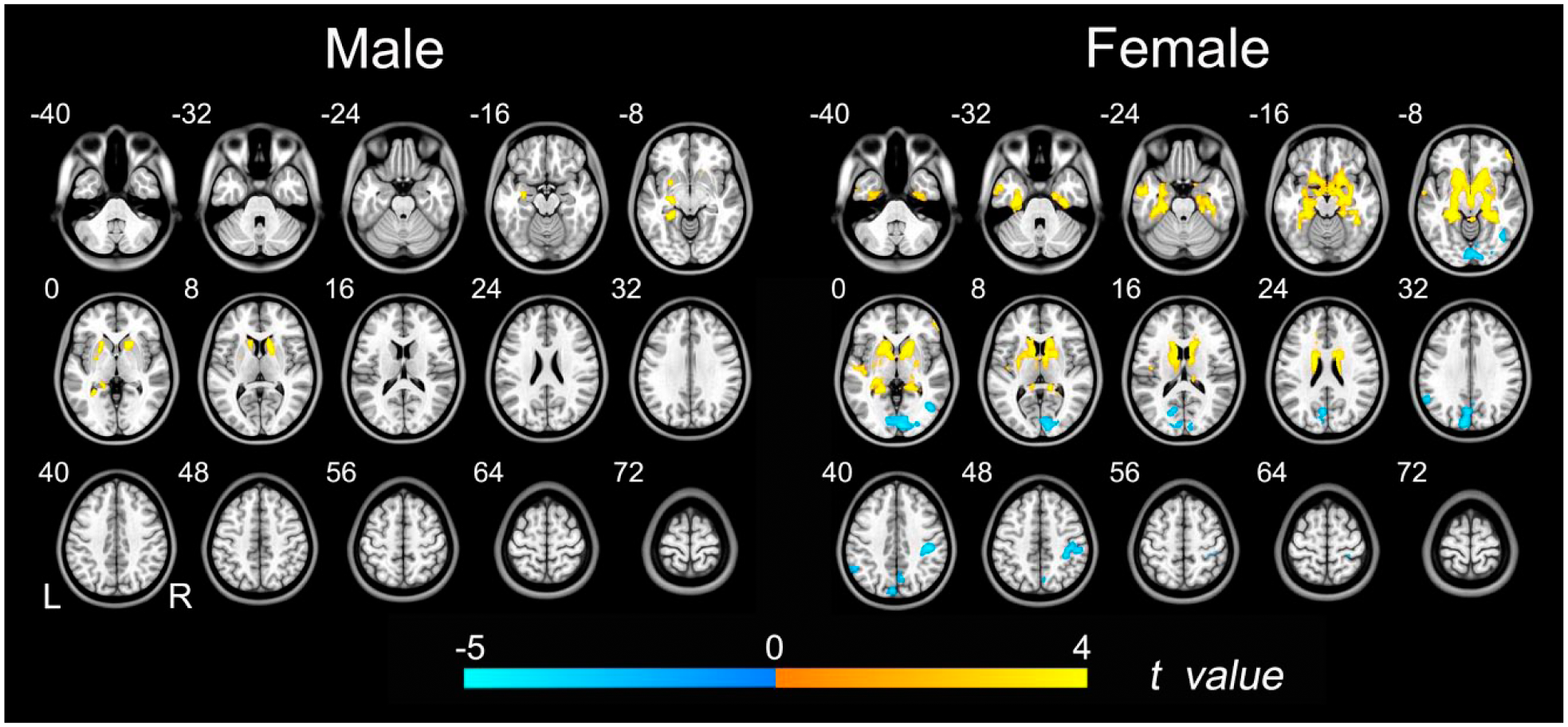
Group differences in ALFF between schizophrenia patients and healthy controls. Left row shows brain regions with significant group differences in males (p < 0.05, FDR corrected). Right row shows brain regions with significant group differences in females (p < 0.05, FDR corrected). The warm color represents the significantly increased ALFF in patients with schizophrenia. The cold color denotes that the ALFF was significantly decreased in schizophrenia patients.
Sex-dependent group differences in rCBF–ALFF coupling
No significant group differences were found in rCBF–ALFF coupling between the two diagnostic groups in the males. The diagnostic group differences in rCBF–ALFF coupling in the females are shown in Figure 5. Compared to the healthy female subjects, the female patients with schizophrenia exhibited significantly increased rCBF–ALFF coupling in the right hippocampus (z = −3.831, p < 0.001) and the right thalamus (z = −2.791, p = 0.005).
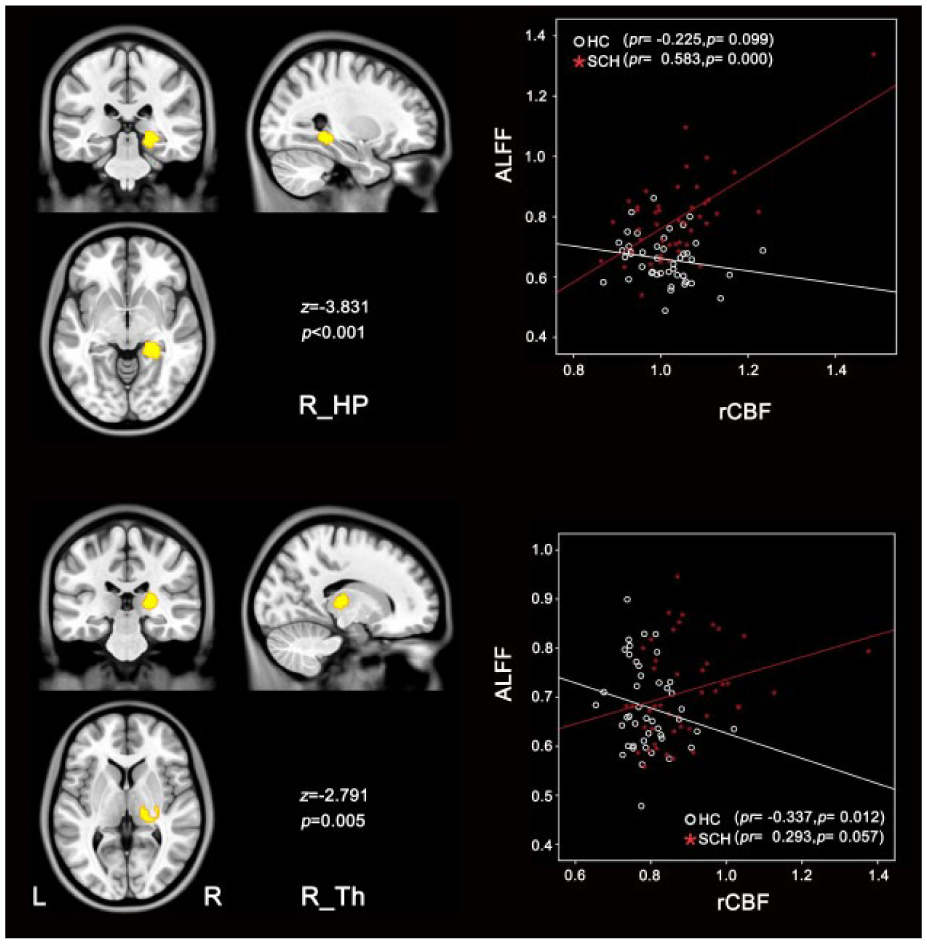
Group differences in rCBF–ALFF coupling in female subjects. Compared with healthy comparison subjects, schizophrenia patients exhibited increased rCBF–ALFF coupling in right hippocampus (R_HP), right thalamus (R_Th) (p < 0.05, uncorrected). Scatter plots demonstrate the corresponding correlationship between rCBF and ALFF in each group.
Sex-dependent correlations between rCBF or ALFF and clinical parameters
We performed partial correlation analyses between the altered ALFF or rCBF and PANSS subscores controlling for age in each sex subgroup (Figure 6, Supplementary Table 3). In the male patients, the PANSS negative subscores were positively correlated with the ALFF in the left hippocampus and parahippocampus (p < 0.05, uncorrected). In the female patients, the PANSS negative subscores were positively correlated with the ALFF in the right postcentral gyrus and negatively correlated with the ALFF in the left inferior parietal lobe (IPL) (p < 0.05, uncorrected). Additionally, the PANSS positive subscores were positively correlated with the ALFF in the left cuneus and the rCBF in the left middle temporal gyrus (MTG) (p < 0.05, uncorrected).
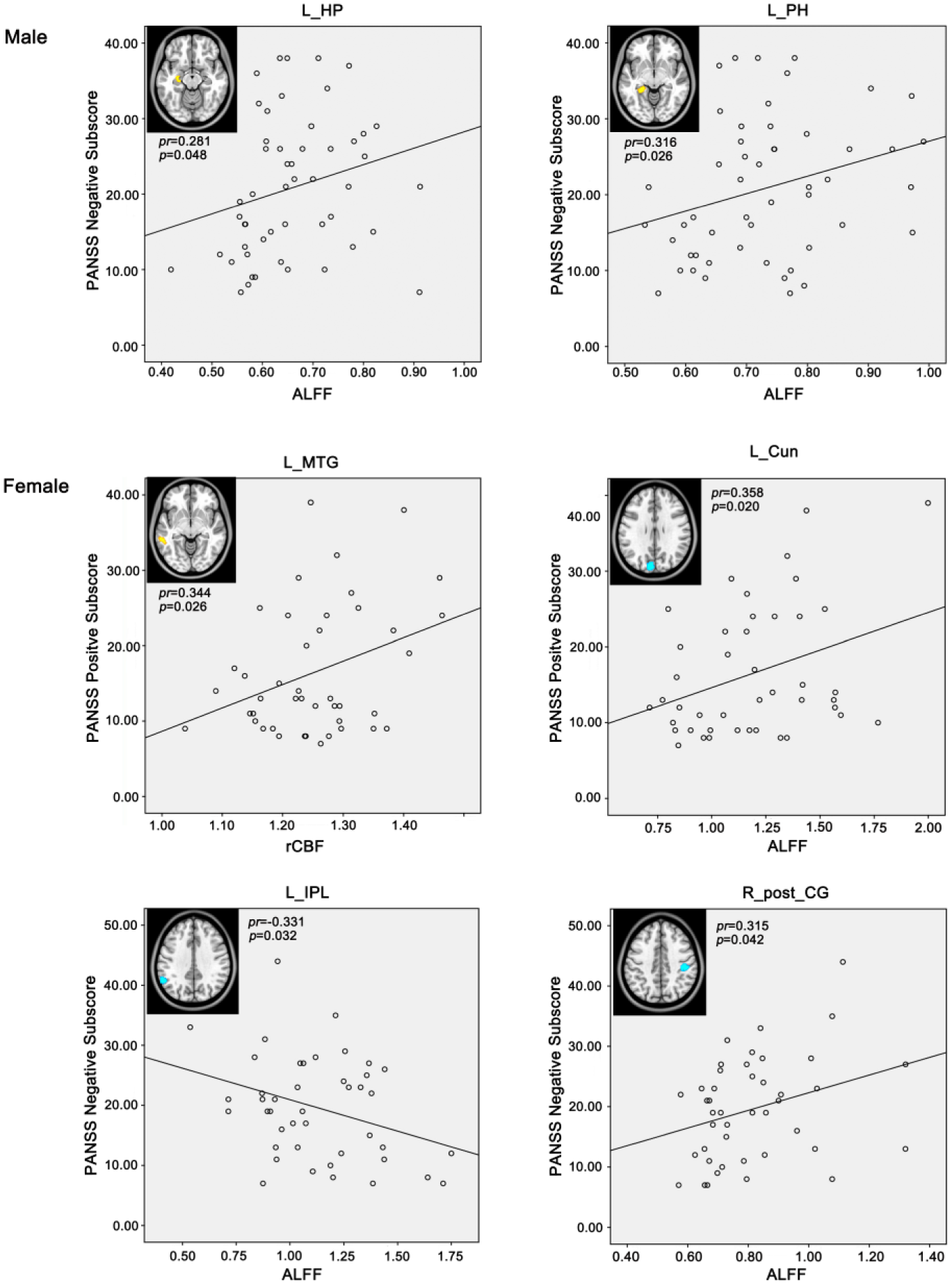
Correlations between rCBF or ALFF and clinical parameters in male or female patients with schizophrenia (p < 0.05, uncorrected).
Discussion
In terms of sex differences within each diagnostic group, both groups demonstrated that brain regions with significant sex differences in rCBF were much more than those with significant sex differences in ALFF. Moreover, the spatial extent and the strength of sex differences in rCBF were less in the patient group than in the control group. In terms of sex-dependent differences between the two diagnostic groups, we found that several intergroup differences in rCBF, ALFF and their coupling were only presented in males or females, representing sex-specific changes in schizophrenia. In terms of measure-dependent differences between the two diagnostic groups, we found that some intergroup differences were only presented in rCBF or ALFF, suggesting that these two measures may have different sensitivity in detecting changes in neuronal activity in schizophrenia.
Differences between rCBF and ALFF changes in schizophrenia
Although we observed several brain regions that exhibited consistent changes in ALFF and rCBF in schizophrenia, we found other brain regions that displayed inconsistent changes in ALFF and rCBF in this disorder. The latter phenomenon is consistent with previous findings of mismatches between changes in rCBF and BOLD response in some brain regions (Brown et al., 2003). Although both CBF and ALFF are used as measures to estimate neural activity, the physiological meanings of the two measures are not completely equivalent. The CBF delivers glucose and oxygen to the brain to maintain energy metabolism during neuronal activity (i.e. CBF changes are concomitant with changes in neuronal activity) (Borogovac and Asllani, 2012). The ALFF is calculated from BOLD signals, which examine local inhomogeneities in the magnetic field due to changes in the relative concentrations of deoxygenated hemoglobin accompanying brain activation (Ogawa et al., 1992). The BOLD signal is composite in nature and unable to pinpoint to a single correlate of neuronal activity because the BOLD effect originates from an intricate interplay between changes in CBF, CBV (cerebral blood volume) and oxygen consumption (Ogawa et al., 1993a, 1993b; Van Zijl et al., 1998). In contrast, the ASL signal is straightforward to interpret because it theoretically reflects a single physiological process (CBF) (Borogovac and Asllani, 2012). Thus, the inconsistent findings in ALFF and rCBF may be explained by inter-regional differences in physiological changes in schizophrenia.
ALFF and rCBF have different sensitivity in detecting changes in neuronal activity. In contrast to the frequency-dependency of the ALFF, the rCBF is frequency-independent and more sensitive to slow varying changes in the brain due to emotional changes (Borogovac and Asllani, 2012). BOLD acquired by the EPI sequence is prone to artifacts in the areas with high susceptibility such as the ACC and MPFC; however, ASL acquired by the spin-echo sequence is not sensitive to susceptibility artifacts, thus yielding greater sensitivity in detecting changes in these brain regions (Wang et al., 2004). The sensitivity differences between ALFF and rCBF may explain why abnormal neuronal activity in the frontal areas (related to emotional processing and high susceptibility) was only detected by rCBF. These findings suggest that rCBF and ALFF can provide complementary information in exploring alterations of brain activity in brain disorders including schizophrenia.
Sex-specific changes in rCBF and ALFF in schizophrenia
Sex differences have been documented in multiple aspects of schizophrenia, including age of onset, course of disease, clinical symptoms and cognitive impairments (Goldstein and Link, 1988; Gur et al., 1996; Hafner, 2003). For example, male patients express more negative symptoms, but female patients present more affective, depressive and anxiety symptoms (Ochoa et al., 2012). Female patients perform worse in spatial memory (Ragland et al., 1999); however, male patients perform worse in verbal memory and executive function (Bozikas et al., 2010; Goldstein et al., 1998; Vaskinn et al., 2011). The neural mechanisms underlying sex differences in schizophrenia have become a topic of interest.
In our study, although both the male and female patients exhibited decreased rCBF in the midline brain regions and increased ALFF in the deep nuclei, the spatial extent of abnormal brain regions was much broader in female patients. The consistent changes across sex may be considered to be the common changes in schizophrenia; however, the sex-specific changes in rCBF and ALFF may be associated with sex differences in clinical manifestations of schizophrenia. For example, only the female patients showed decreased rCBF and ALFF in the occipital and parietal cortices, which play an important role in visuospatial processing (Backus et al., 2001; Blankenburg et al., 2010; Thiyagesh et al., 2009). These female-specific functional impairments may explain why female patients of schizophrenia perform worse on visuospatial tasks than male patients (Gur et al., 2001; Halari et al., 2006).
Correlation analyses revealed that negative symptoms were correlated with the ALFF in the hippocampal structures in male patients but were correlated with the ALFF in the parietal cortices in female patients. The different neural correlates of negative symptoms in male and female patients with schizophrenia are consistent with a previous study reporting differences between male and female patients in the correlations between frontal lobe volume and clinical symptoms (Cowell et al., 1996). These findings may provide an explanation for why male and female patients exhibit different clinical symptoms of schizophrenia (Ochoa et al., 2012). We only found neural correlations of positive symptoms in female patients with schizophrenia, which is consistent with the finding that female patients expressed more positive symptoms than male patients (Goldstein and Link, 1988). The sex differences in rCBF and ALFF in schizophrenia may be related to the hormonal difference between the males and females (Hafner, 2003). This inference is supported by several independent studies demonstrating that modification of estrogen/testosterone improves psychopathology/cognition in schizophrenia (Kindler et al., 2015; Kulkarni et al., 2014; Weickert et al., 2015).
Diminishing sex differences in schizophrenia
Consistent with previous CBF findings (Pirson et al., 2006; Rodriguez et al., 1988; Van Laere et al., 2001), we also found significant sex differences in rCBF in healthy subjects. In healthy subjects, male subjects had higher rCBF in anterior brain regions (frontal and temporal regions) and lower rCBF in posterior brain regions (occipital and parietal regions) compared to female subjects. This finding is consistent with previous findings in CBF (Taki et al., 2011; Van Laere et al., 2001), glucose metabolism (Kawachi et al., 2002), gray matter volume (Chen et al., 2007) and cortical thickness (Sowell et al., 2007).
In schizophrenia patients, although the regional distribution of sex differences in rCBF is similar to that in healthy subjects, the spatial extent and strength of sex differences were largely reduced in schizophrenia (Figure 1). The diminished sex differences in schizophrenia have been reported in brain perfusion (Russell et al., 1997), cortical gray matter volume (Goldstein et al., 2002), activation (Jimenez et al., 2010) and structural covariance (Abbs et al., 2011). The diminished sex differences in rCBF may be explained by sex-specific changes in schizophrenia. For example, the diminished sex difference (male > female) in rCBF in the MPFC/ACC (Figure 1) may be explained by broadly decreased rCBF in this region in male compared to female patients (Figure 3). The diminished difference (male > female) in rCBF in the striatum and thalamus (Figure 1) may be associated with the significant increased rCBF in these regions only in female patients but not in male patients.
There are several limitations of our exploration. First, although we found sex differences in rCBF, ALFF and rCBF–ALFF coupling in schizophrenia, the underlying mechanisms of the sex effects remain unclear and need to be clarified in future studies. Second, most of our patients have chronic schizophrenia and have received medications for a long time, which may influence our interpretation.Investigations on medication-native first-episode schizophrenia patients may facilitate a more accurate characterization of pathogenesis of schizophrenia. Third, during the scanning, the patients were instructed to keep their eyes closed, but it is possible that our results may be affected if they failed to follow our rules. Fourth, none of our clinical correlations could pass the Bonferroni correction for multiple testing, suggesting that they should be validated in future studies.
Conclusion
We found sex-specific alterations in resting-state rCBF, ALFF and rCBF–ALFF coupling in patients with schizophrenia, indicating that sex differences should be routinely assessed in future neuroimaging studies in schizophrenia. We also found that rCBF and ALFF had different sensitivity in detecting group differences, suggesting that the rCBF and ALFF can provide complementary information on the neural mechanisms of schizophrenia.
Footnotes
Acknowledgements
The authors thank Drs Zhenyu Zhou and Ziheng Zhang of the GE Healthcare MR Research China for their support and assistance. Dr Yu designed the study and was pivotal in the project. Authors Ma and Wang contributed equally to the article, including data analysis, summarizing the findings and manuscript writing. Author Qin set up the magnetic resonance imaging (MRI) sequences and the parameter. He also wrote the statistical analysis section of the manuscript. Author Zhuo provided the patients and normal subjects for the study. Authors Ma, Wang, Zhou and Zhu were responsible for data collection. All authors contributed equally to and have approved the final manuscript.
Declaration of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Natural Science Foundation of China (91332113, 81301201 and 81271551), the Tianjin Key Technology R&D Program (14ZCZDSY00018) and the National Key Clinical Specialty Project.
