Abstract
Objective:
The fronto-limbic network is implicated in the neurobiology of major depressive disorder. However, no studies are designed to assess directly the abnormalities of regional activity and network homogeneity of this network in major depressive disorder.
Methods:
A total of 44 drug-naive major depressive disorder patients and 44 healthy controls participated in the study, and resting-state functional magnetic resonance imaging data were obtained. The fractional amplitude of low-frequency fluctuations and network homogeneity methods were employed to analyze the data.
Results:
Compared with the controls, the patients exhibited reduced fractional amplitude of low-frequency fluctuations in the right middle frontal gyrus (orbital part) and decreased network homogeneity in the left middle frontal gyrus. There was no correlation between abnormal fractional amplitude of low-frequency fluctuations/network homogeneity and clinical variables.
Conclusions:
Our findings suggest that decreased regional activity and network homogeneity in the frontal cortex may be the key impairment of the fronto-limbic network in major depressive disorder, and thus highlight the importance of the fronto-limbic network in the neurobiology of major depressive disorder.
Keywords
Introduction
Patients with major depressive disorder (MDD) typically show pervasive depressed mood, cognitive symptoms and changes of social behavior (First et al., 1997). Increasing evidence demonstrates that the apparent heterogeneity of depressive symptoms, such as symptom domains of mood, cognition and vegetative symptoms, is unlikely interpreted by the disturbances of a single brain region (Damasio, 1997). It is thus proposed that MDD is a disorder with the dysregulation of brain networks (Drevets et al., 2008; Mayberg, 2003).
Based on data from neuroimaging studies, several brain networks have been postulated to act as a key role in the neurobiology of MDD. For example, Mayberg et al. (2003) proposed a depressive model, the fronto-limbic network, encompassing dorsal and ventral subnetworks which were differentially affected in MDD. Meanwhile, other brain networks, such as the cortico-subcortical network (including the limbic–cortical–striatal–pallidal–thalamic network) (Drevets et al., 2008; Marchand, 2010; Sheline, 2000) and the default-mode network (DMN) (Graham et al., 2013; Guo et al., 2014), have been emphasized in the neurobiology of MDD.
In Mayberg’s classic neurobiological model (Drevets, 2001; Mayberg, 2003; Seminowicz et al., 2004), MDD was regarded as a disease with the fronto-limbic network dysregulation in seven key brain regions: the lateral prefrontal cortex (PFC), medial PFC, orbitofrontal cortex, rostral anterior cingulate cortex (ACC), subgeneual ACC, hippocampus and anterior thalamus (Seminowicz et al., 2004). In this model, patients with MDD were observed to have decreased frontal cortex function and increased limbic system function (Mayberg et al., 1999). Indeed, both structural and functional alterations of the fronto-limbic network have been noted in MDD. For example, structural deficits in the frontal cortex (Ma et al., 2012; Salvadore et al., 2011; Scheuerecker et al., 2010; Wagner et al., 2008), ACC (Salvadore et al., 2011; Tang et al., 2007; Wagner et al., 2008) and hippocampus (Arnone et al., 2013; Wagner et al., 2008; Zou et al., 2010) have been reported in MDD. Meanwhile, decreased frontal cortex function and increased limbic system activity have been revealed by functional findings (for a review, see Wang et al., 2012). Recently, we reported that brain regions within the cortico–limbic–cerebellar circuit exhibited unidirectionally affected causal connectivities disrupted by structural deficits in MDD (Guo et al., 2015). Although the above-mentioned studies contribute a lot to elucidate the neurobiology of MDD, the detailed brain regions are inconsistently reported in the resting-state functional Magnetic Resonance Imaging (fMRI) studies. For example, Peng et al. (2011) observed decreased regional homogeneity (ReHo) in the left thalamus, whereas Liu et al. (2010) reported no ReHo changes in this brain region. The inconsistency may result from the heterogeneity of study samples (i.e. sample size, depression severity, medication use and long illness duration) and analysis methods. Another possibility is that the brain network is dynamic, and dysfunction of one brain region may contribute to dysfunction of another brain region from the brain network perspective. Therefore, it is meaningful to conduct a study in drug-naive patients with short illness duration of current episode to minimize the possible effects from such confounding factors as medication use and long illness duration. Moreover, the disturbances in MDD cannot be explained by dysfunction of a single brain region. It is meaningful to examine the biological abnormalities in MDD at the brain network level.
In the present study, we designed to simultaneously examine regional activity and network homogeneity (NH) of the fronto-limbic network in a group of drug-naive patients with MDD. Fractional amplitude of low-frequency fluctuations (fALFF), measuring the ratio of power spectrum of low-frequency range to that of the entire frequency range (Zou et al., 2008), was used to measure the regional activity of the fronto-limbic network in MDD. The fALFF method is designed to detect the regional intensity of spontaneous fluctuations of blood oxygen level–dependent (BOLD) signals. The regional intensity of BOLD signals varies with the spontaneous fluctuations of blood flow to the area. Therefore, increased fALFF may be associated with a neural hyperactivity in the area or vice versa (Biswal et al., 1995). NH is a voxel-wise measure that is used to evaluate the correlations of a given voxel with the other voxels of a predefined network (Uddin et al., 2008). Homogeneity is defined as the mean correlation of any given time series of a voxel with the time series of every other voxel in the predefined network. Therefore, the NH method can be used to conduct a hypothesis-driven interrogation of long-range networks according to clinical applications. Previously, we applied the NH method to examine the NH of the DMN in a group of first-episode, drug-naive patients with MDD (Guo et al., 2014), and thus validated the utilization of the NH method in MDD. As we mentioned above, patients with MDD exhibited decreased frontal cortex function and increased limbic system activity (Wang et al., 2012). It would be interesting to simultaneously examine regional activity and NH of the fronto-limbic network in MDD in order to obtain the two types of complementary information (regional activity and functional connectivity) of the fronto-limbic network. We expected that the current patient group would show decreased frontal cortex activity and increased limbic system activity in the fronto-limbic network by using the fALFF method. The alteration of regional activity of a certain brain region would affect the correlations between this region and the other brain regions within the fronto-limbic network, and thus, we hypothesized that patients with MDD would show decreased NH in the frontal cortex and increased NH in the limbic system. We also hypothesized that abnormal fALFF/NH would be correlated with clinical variables (such as depression severity, illness duration and episode number) in patients with MDD.
Methods and materials
Subjects
Patients with MDD were recruited from Mental Health Center, the First Affiliated Hospital, Guangxi Medical University, China. Part of the patients was from our previous studies (Guo et al., 2013a, 2013b). Closely matched healthy controls were recruited from the community. All subjects were informed of the purpose of the study and signed a written informed consent before participating in the study. This study was approved by the ethics committee of the First Affiliated Hospital, Guangxi Medical University, China. Finally, 44 right-handed, drug-naive patients with MDD and 44 right-handed healthy controls were recruited. Patients were diagnosed according to the Diagnostic and Statistical Manual of Mental Disorders–Fourth Edition (DSM-IV) criteria (First et al., 1997). Hamilton Rating Scale for Depression (HRSD, 17 items) was used to assess the depression severity, and the patients had a score of more than 18. The shared exclusion criteria for all subjects were other Axis I disorders, such as schizophrenia, bipolar disorder, substance-induced mood disorder, substance abuse or dependence, acute physical illness and a history of head injury resulting in loss of consciousness. Healthy controls had no psychiatric disorders in their first-degree relatives.
Image acquisition
All images were acquired with a Siemens 3T scanner. Subjects were required to lie motionless with their eyes closed and keep awake. Soft earplugs and foam pads were used to limit scanner noise and head motion. Resting-state functional images were acquired using a gradient-echo echo-planar imaging (EPI) sequence with the following parameters: repetition time/echo time = 2000 ms/30 ms, 30 slices, 64 × 64 matrix, 90° flip angle, 24 cm field of view, 4 mm slice thickness, 0.4 mm gap and 250 volumes (500 s). Each subject was asked some questions to confirm the cooperation after the image acquisition.
Data preprocessing
Images were preprocessed with the Data Processing Assistant for Resting-State fMRI (DPARSF) software (Yan and Zang, 2010) in MATLAB. The first 10 volumes of each subject were discarded to account for T1 equilibrium effects and the subjects adapting to the scanning noise. Slice timing and head motion were first corrected, and no subject had maximal translation or maximal rotation exceeding ±2 mm or ±2°. The obtained images were normalized to the standard Montreal Neurological Institute (MNI) EPI template in SPM8 and resampled to 3 × 3 × 3 mm3. After that, the images were smoothed with an 8 mm full width at half maximum (FWHM) Gaussian kernel, bandpass filtered (0.01–0.08 Hz) and linearly detrended. Several spurious covariates along with their temporal derivatives, including 24 head motion parameters, the signal from a ventricular region-of-interest (ROI) and the signal from a region centered in the white matter, were regressed out (Power et al., 2014). The global signal is commonly regressed out from resting-state data, as it is regarded to be related to physiological noise. However, it is still debatable to remove the global signal in processing resting-state functional connectivity data (Fox et al., 2005; Murphy et al., 2009; Saad et al., 2012). The global signal may reflect important neuronal components, and its removal may introduce artifacts into the data (Hahamy et al., 2014; Yang et al., 2014). Hence, we did not remove this signal in the present study.
Making the fronto-limbic network mask
The fronto-limbic network was defined to include seven key brain regions from Mayberg’s classic neurobiological model for MDD (Mayberg, 2003) and amygdala. The amygdala was included because it was a well-known emotional regulation center and was implicated to be a part of the fronto-limbic network (Wang et al., 2012). The Anatomical Automatic Labeling (AAL) templates of these brain regions were used to make the fronto-limbic network mask, including the superior frontal gyrus, superior medial frontal gyrus, middle frontal gyrus, inferior frontal gyrus, medial orbital frontal gyrus, ACC, hippocampus, thalamus and amygdala (Figure S1).
fALFF analysis
The fALFF analysis was conducted with the software REST (Song et al., 2011) after linear trend removal. The process steps for fALFF (Zou et al., 2008) were as follows. First, the time series of each voxel was converted to the frequency domain without bandpass filtering to obtain the power spectrum by using a Fast Fourier Transform (FFT). Then, the square root was computed at each frequency of the power spectrum to obtain the mean square root across 0.01–0.08 Hz at each voxel. Finally, the sum of amplitude across 0.01–0.08 Hz was divided by that across the whole frequency range within a whole-brain mask for standardization purpose, and fALFF was acquired.
NH analysis
The NH analysis was calculated by using an in-house script in MATLAB. For each subject, voxel-wise NH was computed with the equation as described in a previous study (Uddin et al., 2008). The mean NH of each voxel in the fronto-limbic network mask was acquired. For standardization purpose, the NH values were z-transformed.
Statistical analysis
When appropriate, demographic and clinical variables were compared with two-sample t-tests and Chi-square tests. Two-sample t-tests were conducted to determine the fALFF and NH differences between groups with the software REST (Song et al., 2011). Age and sex were used as covariates to reduce the possible effect of these two variables. Since micromovements could significantly affect measures and results from resting-state fMRI data (Power et al., 2012; Saad et al., 2012), we computed the framewise displacement (FD) values of each subject. The mean FD was also used as a covariate in the group comparisons of the fALFF and NH results. The significance level was set at p < 0.005 corrected for multiple comparisons based on the Gaussian Random Field (GRF) theory (min z > 2.807, cluster significance: p < 0.005, corrected). A random field involving Gaussian probability density functions of the variables was defined as a GRF, and the GRF-correction method was provided by the software REST (Song et al., 2011).
Correlation analysis
To determine the correlations between abnormal fALFF/NH values and clinical variables, mean z values of fALFF/NH were extracted from brain regions with abnormal fALFF/NH. After assessing the normality of the data, Pearson correlations were performed between these variables (p < 0.05).
Results
Characteristics of the subjects
The patients and the controls exhibit no significant group differences in age (patients, 27.52 ± 8.57 years; controls, 29.39 ± 6.70 years), t(86) = −1.14, p = 0.26; sex ratio (22 female patients and 24 female controls), χ2(1) = 0.18, p = 0.67; education level (patients, 12.52 ± 3.04 years; controls, 12.11 ± 2.30 years), t(86) = 0.71, p = 0.48; and the mean FD values (patients, 0.09 ± 0.03 mm; controls, 0.10 ± 0.04 mm), t(86) = −0.65, p = 0.53). The patients have a mean HRSD score of 25.18 ± 5.22, the mean illness duration of 19.61 ± 36.50 months with the illness duration of current episode of 2.80 ± 1.77 months and the mean episode number of 2.00 ± 1.60 times.
fALFF: group differences within the fronto-limbic network
Compared with the controls, the patients showed reduced fALFF in the right middle frontal gyrus (orbital part) (Figure 1 and Table 1). No increased fALFF was observed in the patient group.
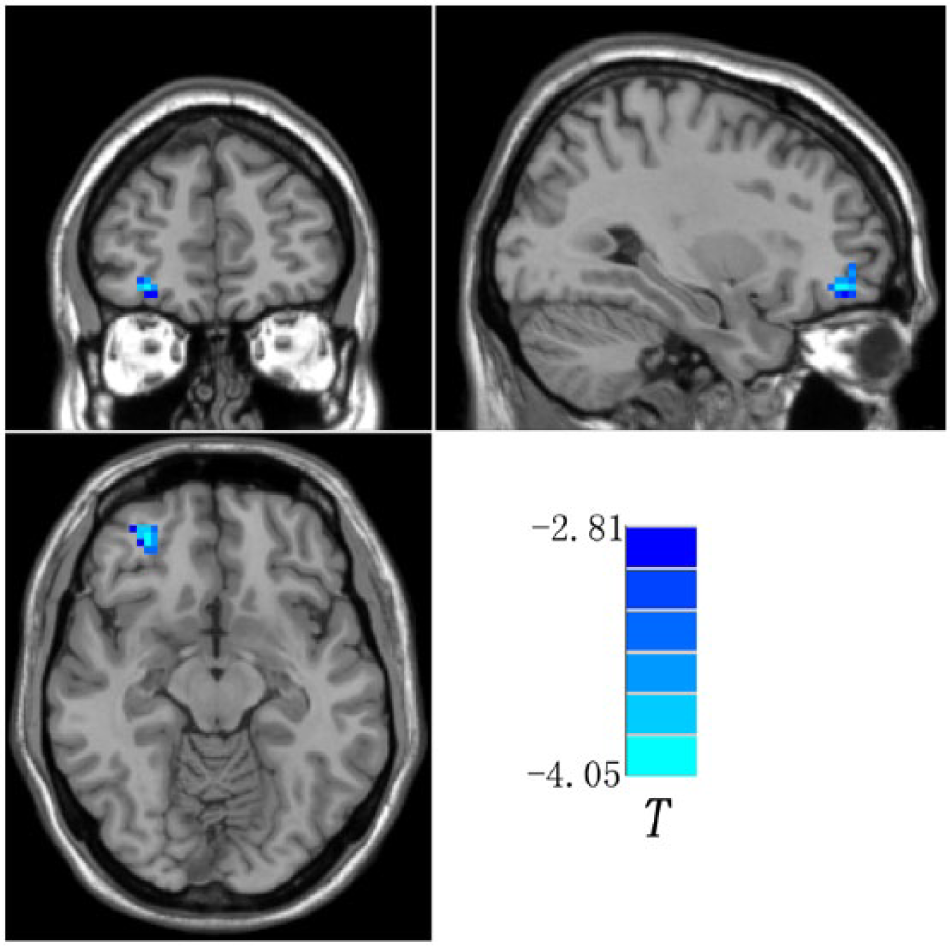
Statistical maps showing fALFF differences between groups (the right middle frontal gyrus, orbital part). Blue denotes lower fALFF in the patient group, and the color bar indicates the t values from two-sample t-tests.
Brain regions with decreased fALFF/NH in patients with major depressive disorder.
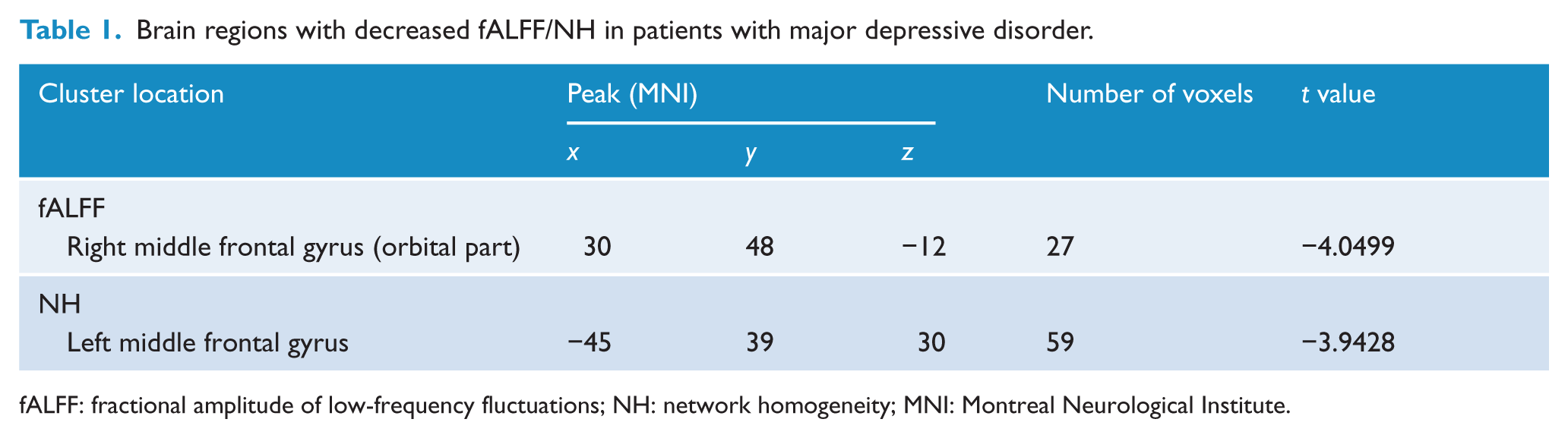
fALFF: fractional amplitude of low-frequency fluctuations; NH: network homogeneity; MNI: Montreal Neurological Institute.
NH: group differences within the fronto-limbic network
The patients exhibited decreased NH in the left middle frontal gyrus compared to the controls (Figure 2 and Table 1). No increased NH was detected in the patient group.
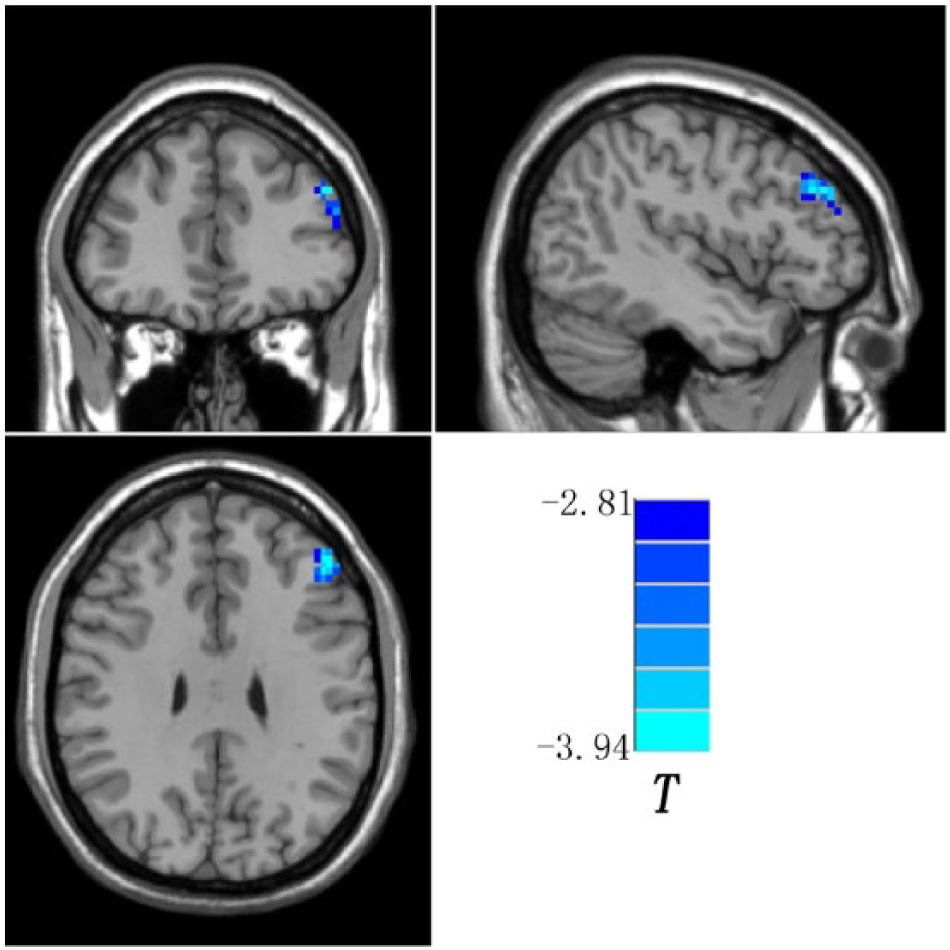
Statistical maps showing NH differences between groups (the left middle frontal gyrus). Blue denotes lower NH in the patient group, and the color bar indicates the t values from two-sample t-tests.
Correlations between abnormal fALFF/NH and clinical variables in the patients
No correlations were observed between abnormal fALFF/NH and clinical variables (i.e. HRSD scores, illness duration, illness duration of current episode and episode number). There was also no correlation between abnormal fALFF/NH and age, education level and FD values.
Discussion
In the present study, the fALFF and NH methods are applied to explore the regional activity and NH of the fronto-limbic network in drug-naive patients with MDD. The main findings are reduced fALFF in the right middle frontal gyrus (orbital part) and decreased NH in the left middle frontal gyrus in MDD. There is no correlation between abnormal fALFF/NH and clinical variables.
As a network disorder (Bluhm et al., 2009; Chen et al., 2012; Liu et al., 2012; Lui et al., 2011; Tao et al., 2011; Zhu et al., 2011), MDD is postulated to be a result from a failure in the coordinated interaction among brain regions of the fronto-limbic network. In this theoretical model, the ventral components of the fronto-limbic network (including the subcortical, limbic and paralimbic regions) are thought to regulate vegetative and somatic features of depression (i.e. appetite, sleep and endocrine alterations), and the dorsal components (including the neocortical and superior limbic regions) are hypothesized to mediate attention and cognitive function (i.e. apathy and attention impairment). A breakdown of this network could interpret the evolvement of depressive symptoms such as somatic complains and negative bias to interpersonal feedback (Cullen et al., 2009). Indeed, a bulk of studies have reported abnormal functional connectivity between the frontal and limbic regions (Anand et al., 2005a, 2005b; Cullen et al., 2009; Horn et al., 2010; Liu et al., 2010; Su et al., 2015). Previous evidence from resting-state fMRI studies has supported that dysregulation of the fronto-limbic network is attributable to symptomatic mood disturbances in MDD (Wang et al., 2012). Based on the neuroimaging data, Mayberg et al. proposed the classic neurobiological model for MDD (Drevets, 2001; Mayberg, 2003; Seminowicz et al., 2004). In this fronto-limbic model, MDD was observed to show decreased frontal cortex function and increased limbic system function. Consistent with the fronto-limbic model, we found reduced regional activity in the right middle frontal gyrus (orbital part) in MDD. Reduced fALFF in the right middle frontal gyrus was also identified in our previous study with 22 first-episode, medication-naive patients with MDD by a GE 1.5T scanner (Liu et al., 2013). Furthermore, we examined the fronto-limbic network at the network level by the NH method, and found decreased NH in the left middle frontal gyrus. The finding of decreased fALFF and NH in the frontal cortex could underpin impaired coordination between the frontal and limbic regions, and resulted in the abnormalities of the fronto-limbic network.
However, the affected brain regions are not the same regions by using the fALFF and NH methods in the present study. There are at least two possibilities to explain the inconsistency. First, the inconsistency may result from the different analysis methods. The fALFF method is employed to measure brain regional activity by detecting regional signal alterations of spontaneous activity (Zou et al., 2008), while the NH method is designed to estimate the homogeneity of a specific network at the network level (Uddin et al., 2008). Different aspects are measured by the two methods, and thus, it is not surprised that the affected frontal regions are not located in the same brain regions. Second, brain networks are dynamic, and impairment in one brain region may attribute to dysfunction of another brain region within the fronto-limbic network.
Limbic regions such as ACC are thought to act as a regulatory role over the limbic system that process emotional information (Anand et al., 2005b). Other limbic regions, including the medial thalamus (considered to play a role in emotional perception) and the amygdala (thought to be involved in the neural response to negative information), are also the key brain regions of the fronto-limbic network (Anand et al., 2005a). According to Mayberg’s neurobiological model for MDD (Mayberg, 2003), these limbic regions are expected to exhibit enhanced regional activity and NH in MDD. Therefore, it is surprised that no limbic findings are found in the present study. There are at least two likely interpretations for the absence of limbic findings. First, we recruited drug-naive patients with relatively short illness duration of current episode (about 2.80 months). Some confounding factors such as medication use and long illness duration of current episode can be excluded from the present findings with this recruitment criterion. Our results suggest that limbic changes are not necessarily present in the acute stage of depression episode. Of course, this speculation needs to be warranted or refuted by future studies. Another alterative explanation is that the frontal cortex has afferent and efferent connections with the limbic system (Zhang et al., 2009). As we mentioned above, the fronto-limbic network is dynamic, and limbic abnormalities may be related to a multitude of non-limbic impaired connections.
Previously, clinical variables, such as depression severity and illness duration, were observed to have correlations with abnormal neural activity in MDD (Zhang et al., 2011), and we hypothesized that there were some correlations between abnormal fALFF/NH and clinical variables in MDD. Thus, it is unexpected that there are no correlations between these variables in this study. One possibility for no correlations is that abnormal fALFF/NH in the frontal cortex are the trait changes for MDD independently of depression severity and illness duration. To test this possibility, we conducted additional analyses with depression severity as a covariate (the HAMD score for each control was set to 1) and got similar results (Figures S2 and S3 and Table S1) as the original findings, indicating that depression severity had little effect on the fALFF/NH abnormalities in the frontal cortex. Another possibility is that the ratings of the clinical parameters are concentrated, and the concentration of the clinical parameters will lead to no correlations with abnormal fALFF/NH in the frontal cortex. For example, the illness duration of current depression episode is around 2.80 months with a range from 1 to 6 months for the patients.
This study has certain limitations. First, we made the fronto-limbic network mask using the AAL templates. This mask only included the key regions of the fronto-limbic network. Other regions of the fronto-limbic network (e.g. parahippocampus and posterior cingulate cortex) were not included in the mask, which might have biased the present findings. Second, psychological measurements were not conducted in the present study, and thus, we could not know the correlations between abnormal fALFF/NH and psychological parameters. Finally, this study was performed at rest. Therefore, the present findings are related to depressive neurobiology in general, and not with a particular model of depressive tasks.
Despite the limitations, this is the first to examine both regional activity and NH of the fronto-limbic network in drug-naive patients with MDD. Our findings indicate that decreased regional activity and NH in the frontal cortex may be the key impairment of the fronto-limbic network in MDD, and thus advance the understanding of the important role of the fronto-limbic network in the neurobiology of MDD.
Footnotes
Acknowledgements
The authors thank all individuals who served as the research participants.
Declaration of interest
The authors declare no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Funding
This study was supported by grants from the National Natural Science Foundation of China (grant no. 81260210), the Natural Science Foundation of Guangxi Province for Distinguished Young Scientists (grant no. 2014GXNSFGA118010) and the Natural Science Foundation of Guangxi Province (grant no. 2013GXNSFAA019107).
