Abstract
Objective:
The brain functional alterations at regional and network levels in post-traumatic stress disorder patients are still unclear. This study explored brain functional alterations at regional and network levels in post-traumatic stress disorder patients with resting-state functional magnetic resonance imaging and evaluated the relationship between brain function and clinical indices in post-traumatic stress disorder.
Methods:
Amplitude of low-frequency fluctuation and seed-based functional connectivity analyses were conducted among typhoon survivors with (n = 27) and without post-traumatic stress disorder (n = 33) and healthy controls (n = 30) to assess the spontaneous brain activity and network-level brain function. Pearson correlation analyses were performed to examine the association of brain function with clinical symptom and social support.
Results:
Both the post-traumatic stress disorder group and the trauma-exposed control group showed decreased amplitude of low-frequency fluctuation in the dorsal anterior cingulate cortex relative to the healthy control group. The post-traumatic stress disorder group showed increased dorsal anterior cingulate cortex functional connectivity with the right paracentral lobule and bilateral precentral gyrus/postcentral gyrus relative to both control groups. Both traumatized groups exhibited decreased dorsal anterior cingulate cortex functional connectivity with the right hippocampus and left cerebellum relative to the healthy control group. More decreased dorsal anterior cingulate cortex functional connectivity with the right hippocampus was found in the post-traumatic stress disorder group. The Checklist-Civilian Version score positively correlated with functional connectivity between the dorsal anterior cingulate cortex and the right paracentral lobule as well as between the dorsal anterior cingulate cortex and the right precentral gyrus/postcentral gyrus. The social support was associated with functional connectivity between the dorsal anterior cingulate cortex and the bilateral precentral gyrus/postcentral gyrus as well as the dorsal anterior cingulate cortex and the left middle frontal gyrus.
Conclusion:
Trauma exposure may result in aberrant local and network-level functional connectivity in individuals with or without post-traumatic stress disorder. Altered amplitude of low-frequency fluctuation in the dorsal anterior cingulate cortex may be a predisposing risk factor for post-traumatic stress disorder development following trauma exposure. More prominent decreased dorsal anterior cingulate cortex functional connectivity with the right hippocampus might be specific in the post-traumatic stress disorder group. Improvement of social support might possibly be significant for post-traumatic stress disorder patients.
Keywords
Introduction
Post-traumatic stress disorder (PTSD) is a mental disorder characterized by four symptom clusters: re-experiencing, avoidance, negative alteration in cognitions and mood, and alterations in arousal. It usually develops after experiencing an emotionally traumatic event such as military combat, traffic accidents, rape, physical or sexual assault, or natural disasters. The life prevalence for PTSD is reported to be about 6.8% in a general population (Kessler et al., 2005). It is reported that 19% of PTSD patients will attempt suicide (Foa et al., 2006). In consideration of the high prevalence of PTSD and its substantial detrimental effects, it is of great significance to find reliable biomarkers to guarantee early diagnosis and effective therapeutic approaches.
In recent years, functional neuroimaging studies have tremendously advanced our understanding of the pathophysiological mechanism underlying PTSD. Abnormal functional changes have been identified in brain regions including the amygdala, hippocampus, middle cingulate cortex (MCC) and dorsal anterior cingulate cortex (dACC) and insula (Francati et al., 2007; Hayes et al., 2012; Nemeroff et al., 2006). However, most functional neuroimaging studies of PTSD have examined brain activity during symptom provocation or cognitive tasks (Etkin et al., 2004; Frewen et al., 2008), and the results are inconsistent. These inconsistencies may be due to the different cognitive tasks, medication, different sources of trauma and various analysis methods. Usually, PTSD subjects are compared to only one control group, the trauma-naive controls or trauma-exposed individuals without PTSD. Thus, whether the brain function abnormalities are specific to this disorder is unknown. Recently, the spontaneous low-frequency (0.01–0.08 Hz) fluctuation in blood oxygenation level–dependent signal during resting state has been utilized to probe the intrinsic brain activity (Mohamed et al., 2004; Raichle and Mintun, 2006). Free of task, without exposure to radiation and being easy to conduct, resting-state functional magnetic resonance imaging (rs-fMRI) may be a promising tool for further understanding of abnormalities in brain activity in participants with brain disorders.
Amplitude of low-frequency fluctuation (ALFF) is an effective method to measure the regional brain activity during resting state (Zang et al., 2007). ALFF can directly reflect the intensity or amplitude of spontaneous brain activity when compared with other algorithms such as a regional homogeneity algorithm (Zang et al., 2007) and is considered to be of physiological importance, and thus it can be used to evaluate brain functional changes (Mohamed et al., 2004). Previous studies have identified abnormal ALFF in many brain areas, such as the medial prefrontal cortex and ACC (Bing et al., 2013), middle occipital gyrus, insula, cerebellum, right medial and middle frontal gyri (Yin et al., 2011) in patients with PTSD. However, altered brain regional activities alone could not completely reflect the mechanism of PTSD. Functional connectivity (FC), which characterizes the spatially remote neurophysiological events (Van de Ven et al., 2004), could be further investigated to better understand the neural basis of PTSD. The combination of ALFF and FC analyses has been used as an effective tool to probe brain activity in healthy subjects and patients with various brain diseases such as depression, attention deficit hyperactivity disorder and irritable bowel syndrome (Lee et al., 2013; Li et al., 2014; Qi et al., 2016). However, the mechanism of alterations in ALFF and the relationship between ALFF and remote FC in PTSD patients are still unclear and need further exploration.
Social support refers to both the material support and spiritual help provided by others to be available in the context of both formal and informal helping relationships (Gottlieb and Bergen, 2010). It has been regarded as a potential mediator for an individual’s psychological health (Xu and Ou, 2014). In a recent meta-analysis, low social support was reported to be one of the most important risk factors for PTSD (Trickey et al., 2012). Feng et al. (2007) revealed that the occurrence of PTSD in flood victims was significantly related to social support. Particularly, individuals living in flood-affected areas with a higher degree of social support had a significantly lower PTSD rate. However, the relationship between social support and FC in PTSD caused by typhoon remains unknown and needs further investigation.
In this study, we aimed to explore brain functional alterations at regional and network levels with rs-fMRI in PTSD patients and to evaluate their association with clinical symptoms and social support. Based on previous findings, we hypothesized that PTSD patients would exhibit altered ALFF in regions which are suggested to be involved in PTSD, such as the dACC, hippocampus, insula, amygdala and parahippocampal cortex (Stark et al., 2015). In addition, in those who experienced traumatic event but without the diagnosis of PTSD, brain function changes may still occur. We also expected regions with ALFF abnormalities to show abnormal remote FC (hippocampus, etc.) and the connectivity measures to be associated with subjects’ symptomatology and social support.
Methods
Participants and clinical assessment
On July 18, 2014, Typhoon Rammasun, a category 5 super typhoon struck Wenchang city on the island province of China. People residing in this area were heavily affected by this typhoon, which caused at least 14 deaths. Particularly, in Luodou farm of Wenchang city, more than 1000 people were trapped and almost drown by the storm tide induced by this destructive typhoon. We recruited 70 typhoon-exposed subjects from this area, 36 with PTSD (9 males and 27 females) and 34 without PTSD (trauma-exposed control [TEC], 7 males and 27 females), who were all screened with the PTSD Checklist-Civilian Version (PCL). The PCL is a 17-item self-report questionnaire that measures the severity of Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV)-defined PTSD symptoms on a five-point ordinal scale ranging from 1 (not at all) to 5 (extremely). PTSD diagnosis was based on DSM-IV diagnostic criteria for current PTSD, and symptoms were assessed with the Clinician-Administered PTSD Scale (CAPS; Weathers et al., 2001). The CAPS for DSM-IV is a structured interview assessing the frequency and intensity of each PTSD symptom using behaviorally anchored rating (from 0 to 4). This scale assesses the 17 core PTSD symptoms listed in the DSM-IV and obtains information regarding symptom onset, duration and functional impact. The absence or presence of comorbid disorders was determined via the Structural Clinical Interview for DSM-IV. Furthermore, 32 healthy controls (HCs; 9 males and 23 females) who did not meet DSM-IV Criterion A1 for PTSD were recruited via advertisement from Haikou, a city about 35 km from Wenchang city. For all participants, Self-Rating Anxiety Scale (SAS; Zung, 1971) and Self-Rating Depression Scale (SDS; Zung, 1965) were administrated to assess anxiety and depression symptoms, respectively. Social Support Rating Scale (SSRS) developed by Shuiyuan Xiao (1994) was administrated to assess social support in our study. This scale includes subjective support, objective support and support utilization. Scoring methods of SSRS are as follows: (1) for items 1–4 and 8–10, only one choice is selected for each item. The items 1–4 are selected for 1, 2, 3 and 4 points, respectively; (2) the score of the fifth item contains the total score of A, B, C and D items. No to full support in each item is equal to the score of 1–4; (3) the sixth and seventh items: if the answer is ‘no source’, 0 points will be recorded. The number of sources is equal to the points. The analysis method of SSRS is as follows: (1) SSRS total scores—the sum score of all 10 entries; (2) objective support scores—the sum score of the second, sixth and seventh items; (3) subjective support scores—the sum score of the first, third, fourth and fifth items; (4) support utilization scores—the sum score of the eighth to tenth items. Objective support is visible or practical, including material direct assistance and the existence and participation of social networks and group relationships. The subjective support is subjective, experiential or emotional support, which refers to the emotional experience and satisfaction degree of individual being respected, supported and understood in society. All the above procedures took place between November 2014 and January 2015.
General exclusion criteria included age < 18 years or >65 years, left handedness, a history of head injury or loss of consciousness, significant medical and neurological conditions, comorbid lifetime or current psychiatric disorders other than depression and anxiety, alcohol or drug abuse/dependence, use of psychiatric medication and contraindications to MRI such as claustrophobia, pregnancy and ferromagnetic implants. In the PTSD group, complete imaging data were not available for three female subjects, and six were excluded for denture-related artifacts (one female and one male), brain infarction revealed by conventional MRI (one female), pregnancy (one female) and excessive movement during MRI scanning (translation > 1.5 mm or rotation > 1.5° in any direction; one male and one female). In addition, we excluded one female TEC for excessive movement and two male HCs for brain infarction. Thus, 27 PTSD patients, 33 TECs and 30 HCs were ultimately included in the statistical analysis. The study was in accordance with the Declaration of Helsinki and approved by the ethics committee of Hainan General Hospital and the Second Xiangya Hospital of Central South University. All participants provided written informed consent after a detailed description of this study.
MRI data acquisition
Magnetic resonance imaging scans were conducted at Hainan General Hospital using a 3-T MR scanner (Skyra; Siemens Medical Solutions, Erlangen, Germany) equipped with a 32-channel standard head coil. Subjects’ heads were immobilized using a foam pad and a Plexiglas head cradle. Whole brain resting-state functional images were obtained using an echo-planar imaging sequence using the following parameters: repetition time (TR)/echo time (TE) = 2000/30 ms, flip angle = 90°, field of view (FOV) = 230 × 230 mm2, matrix = 64 × 64, 35 slices, slice thickness = 3.6 mm, no intersection gap and total volume number = 250. The sections were placed approximately parallel to the anterior commissure–posterior commissure line. High-resolution T1-weighted three-dimensional (3D) anatomical images were also acquired with a sagittal magnetization-prepared rapid gradient echo sequence for later co-registration and normalization (TR/TE = 2300/1.97 ms, flip angle = 9°, FOV = 256 × 256 mm2, matrix = 256 × 256, 176 slices, slice thickness = 1 mm). Each functional magnetic resonance imaging (fMRI) scan lasted 500 seconds. During the functional scanning, subjects were instructed to lie quietly, keep eyes closed and let their mind wander without falling asleep.
Data preprocessing
The imaging was carried out using Statistical Parametric Mapping software (SPM8; www.fil.ion.ucl.ac.uk/spm/). The first 10 volumes of the functional images were discarded to ensure signal equilibrium. The remaining 240 volumes were slice time corrected, realigned and co-registered with the anatomical scan. The co-registered anatomical images were then segmented into gray matter (GM), white matter (WM) and cerebrospinal fluid (CSF) and normalized into standard Montreal Neurological Institute (MNI) space with a final size of 3 × 3 × 3 mm3 (Spati et al., 2015). The resulting normalization matrix was then applied to the functional data. After that, the functional images were smoothed by convolution with an isotropic Gaussian kernel (full width at half maximum [FWHW] = 8 mm). After smoothing, the imaging data were filtered (bandpass, 0.01–0.08 Hz) to remove the effects of low-frequency drift and high-frequency noise. Last, nuisance covariates including CSF signals, global mean signals, WM signals and head motion parameters were regressed out from the rs-fMRI data.
ALFF analysis
REST1.8 software (www.restfmri.net) was used to generate individual ALFF map, which was associated with regional spontaneous neural activity. After bandpass filtering and linear trend removal, the time series for a given voxel was first transformed to the frequency domain using fast Fourier transform. The square root of the power spectrum was computed and then averaged across 0.01–0.08 Hz at each voxel. This averaged square root was taken as the ALFF. The ALFF of each voxel was further divided by the global mean ALFF value of the individual to standardize data across subjects (Zang et al., 2007). REST1.8 software (www.restfmri.net) was also used to perform Monte Carlo simulation similar to AlphaSim in AFNI. Thus, we could identify the minimum number of contiguous voxels for additional cluster size correction of at least p < 0.05. Only activation clusters with that extent of voxels or greater were considered significant.
Seed-based FC analysis
A seed-based inter-regional FC analysis was conducted. Areas where altered ALFF difference was found among the three groups were selected as the seed regions. In detail, a mask of the ALFF effect was used to define the reference time series. A correlation map was produced by calculating the cross-correlation coefficient (r score) between the reference time series of each seed region compared to the rest of the brain. Fisher’s r-to-z transformation was performed to improve the normality of the correlation coefficient.
Statistical analysis
Chi-square test was used to analyze gender distribution, and one-way analysis of variance (ANOVA) was performed for all continuous variables except for PCL scores, for which an independent t test was used to examine differences between the PTSD and TEC groups. The above analyses were conducted with SPSS version 16.0 (SPSS Inc., Chicago, IL, USA), with the significance threshold set at p < 0.05. We used SPM8 and SPSS to analyze the ALFF and FC maps of the three groups, respectively. Within each group, a random-effect one-sample t test was used (p < 0.05, familywise error [few] corrected). ALFF and seed-based FC differences among the three groups were analyzed using ANOVA with education level and depression diagnosis as the covariates, followed by post hoc t tests to examine the between-group differences.
For the ALFF analysis, post hoc t tests were corrected (p < 0.05) for multiple comparisons for the number of voxels tested with the AlphaSim program (http://resting-fmrisourceforge.net; Song et al., 2011). For the seed-based remote FC analysis, the AlphaSim program was also used for multiple-comparison correction (Song et al., 2011). To investigate the association between PTSD symptom severity and brain measures, mean FC (Z values) from clusters with significant group differences were extracted and then correlated against the CAPS total scores, PCL scores and social support using Pearson correlation analysis. The correlation analysis was accomplished with SPSS, with a significant threshold of p < 0.05 (not corrected).
Results
Demographic and clinical variables
The demographic and clinical characteristics are summarized in Table 1. There is no significant difference in age (F = 0.317, p = 0.729) and gender distribution (p = 0.912) among the PTSD, TEC and HC groups. Significant difference was found in the education level among the three groups (F = 8.396, p < 0.001). Post hoc analyses revealed that the education level of the HC group was higher than that in the PTSD group (p < 0.001) and the TEC group (p = 0.001). No significant difference was found between the PTSD and TEC groups (p = 0.518). The mean CAPS total score of PTSD group was 78.2 ± 19.3, and the PCL scores were higher in this group as compared with the TEC group (p < 0.001). In total, 10 PTSD patients had current psychiatric co-morbidity: nine with depression (two males and seven females) and one with anxiety disorder (one female). Significant differences were also found among the three groups in the SAS (F = 81.864, p < 0.001) and SDS scores (F = 101.915, p < 0.001). Post hoc analyses revealed that the SAS (p = 0.025) and SDS (p = 0.003) scores in the TEC group were significantly higher than those in the HC group, but were significantly lower as compared with the PTSD group (all p < 0.001). Significant differences were also found among the three groups regarding the subjective support (F = 9.598, p < 0.001), objective support (F = 8.478, p < 0.001), support utilization (F = 4.411, p = 0.015) and SSRS total score (F = 12.159, p < 0.001). Post hoc analyses revealed that the PTSD group had significantly lower subjective support (p = 0.001; p < 0.001), objective support (p = 0.003; p < 0.001) and SSRS total scores (p = 0.002; p < 0.001) than the TEC and HC groups. No statistical difference was found in subjective support (p = 0.329), objective support (p = 0.294) and SSRS total score (p = 0.082) between the TEC and HC groups. Although no significant difference was found between the PTSD and TEC in the support utilization (p = 0.884), the PTSD and TEC groups had significantly lower support utilization scores than the HC group (all p = 0.012).
Demographic and clinical data of traumatized individuals and healthy controls.
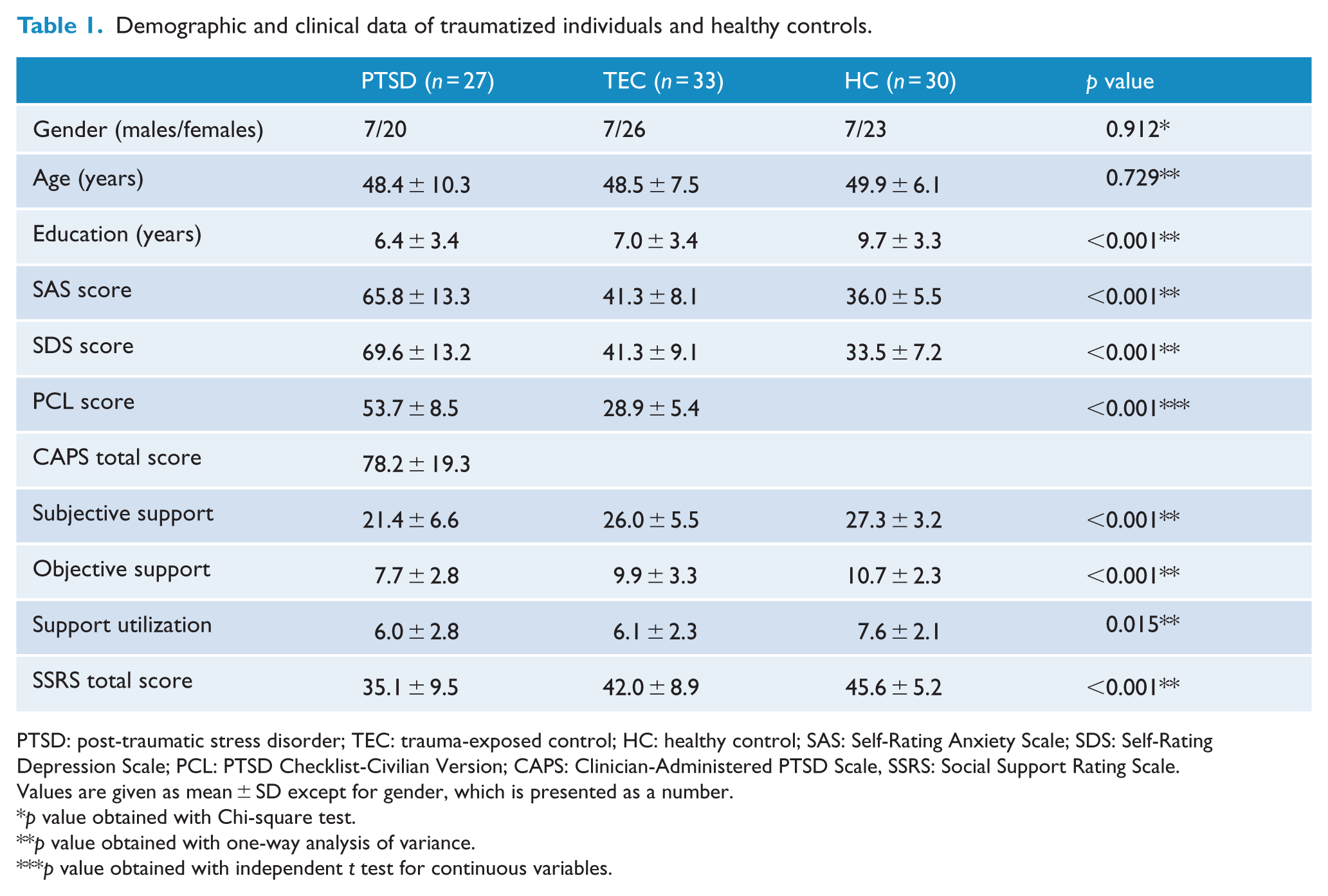
PTSD: post-traumatic stress disorder; TEC: trauma-exposed control; HC: healthy control; SAS: Self-Rating Anxiety Scale; SDS: Self-Rating Depression Scale; PCL: PTSD Checklist-Civilian Version; CAPS: Clinician-Administered PTSD Scale, SSRS: Social Support Rating Scale.
Values are given as mean ± SD except for gender, which is presented as a number.
p value obtained with Chi-square test.
p value obtained with one-way analysis of variance.
p value obtained with independent t test for continuous variables.
ALFF and seed-based FC
Analysis of variance showed that the dACC shows ALFF difference among the three groups (p < 0.05, AlphaSim corrected; equivalent to an individual voxel detection probability threshold of Pthr = 0.01 and a minimum cluster size threshold of CSthr = 14 voxels; Figure 1). Post hoc analyses revealed that both the PTSD and TEC groups showed decreased ALFF in the dACC relative to the HC group (Figure 1).
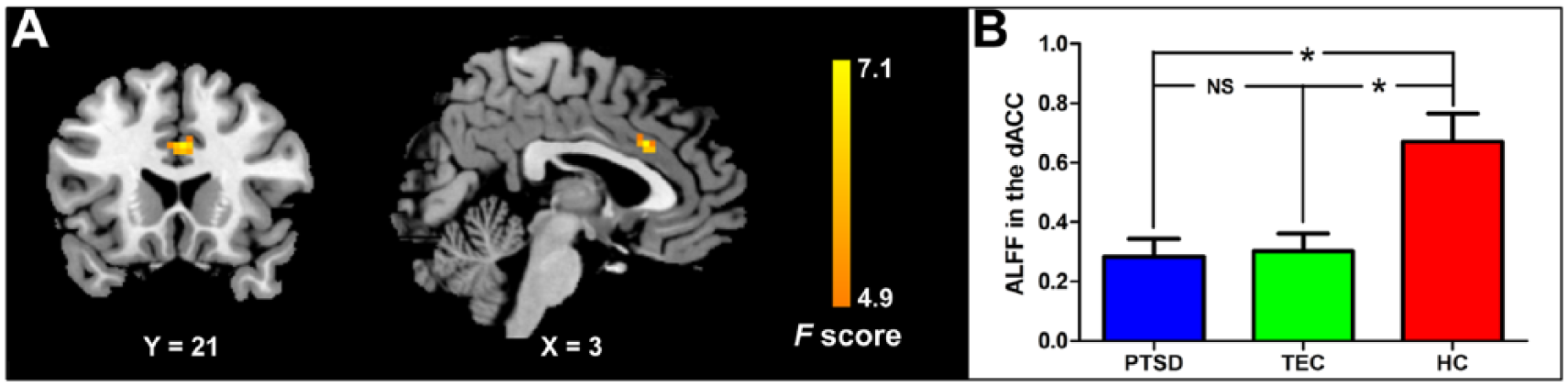
ALFF differences revealed by analysis of variance: (A) the dACC shows difference among the three groups (p < 0.05, AlphaSim corrected); (B) post hoc analyses reveal that both the PTSD and TEC groups show decreased ALFF in dACC relative to the HC group.
FC analysis of the dACC found that the dACC showed positive FC with bilateral paracentral lobule, supplementary motor area (SMA), MCC, middle frontal gyrus (MFG), inferior frontal gyrus (IFG), insula and superior temporal gyrus (STG), but negative FC with bilateral posterior cingulate cortex (PCC), precuneus, superior frontal gyrus (SFG), angular, ventromedial prefrontal gyrus (vmPFC), middle temporal gyrus (MTG), right hippocampus and parahippocampal gyrus in each group (p < 0.05, FWE corrected; Figure 2).
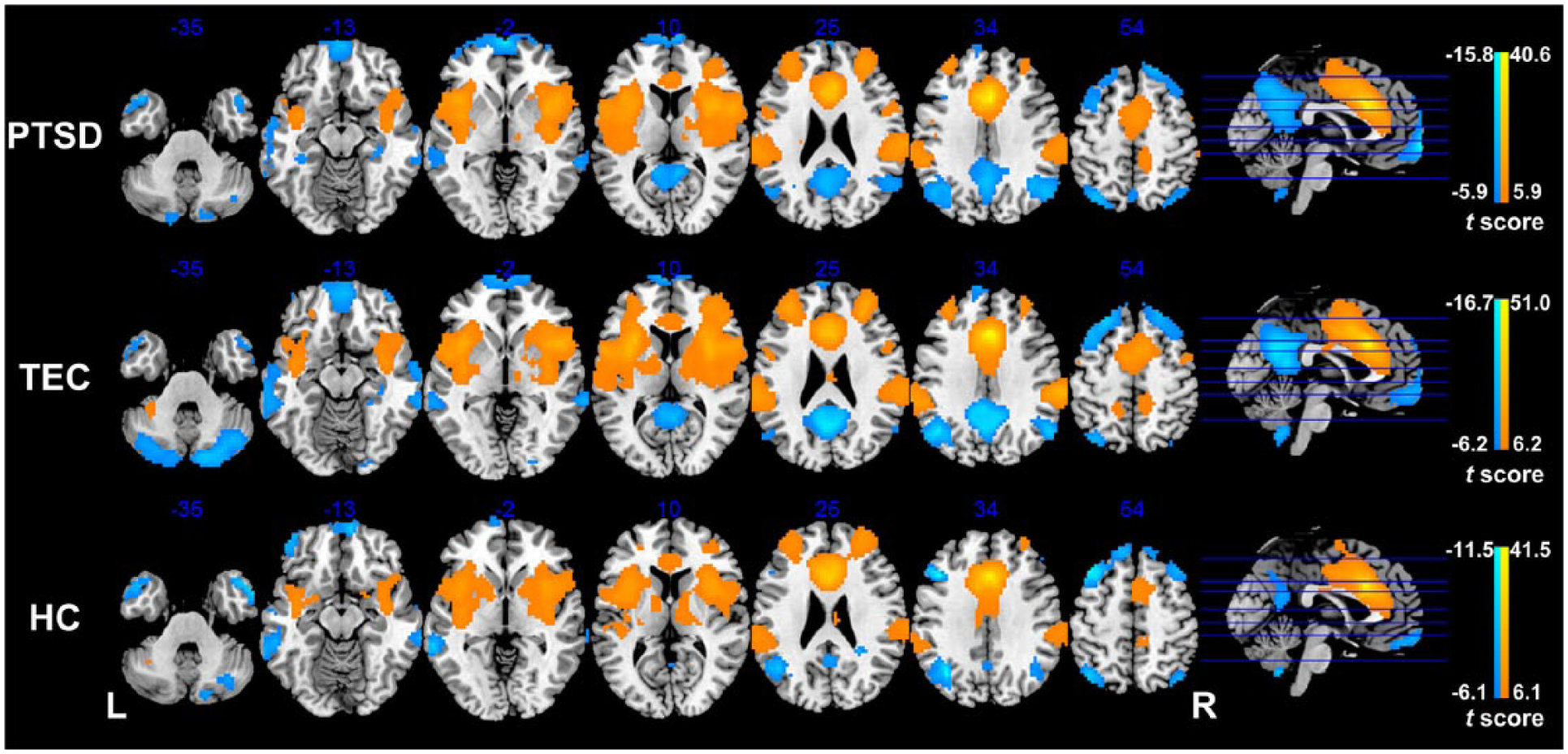
Functional connectivity of the dACC in the PTSD, TEC and HC groups. In each group, the dACC shows positive functional connectivity with the bilateral paracentral lobule, supplementary motor area, middle cingulate cortex, middle frontal gyrus, inferior frontal gyrus, insula and superior temporal gyrus, but negative functional connectivity with the bilateral posterior cingulate cortex, precuneus, superior frontal gyrus, angular, ventromedial prefrontal gyrus, middle temporal gyrus, right hippocampus and parahippocampal gyrus (p < 0.05, FWE corrected).
Significant differences were found in FC with the dACC among the PTSD, TEC and HC groups (p < 0.05, AlphaSim corrected; Pthr = 0.01, CSthr = 62 voxels; Figure 3 and Table 2). Post hoc t tests revealed that the PTSD group shows increased dACC FC with the right paracentral lobule and bilateral precentral gyrus (PreCG)/postcentral gyrus (PoCG) relative to both the TEC and HC groups (p < 0.05, AlphaSim corrected; Pthr = 0.01, CSthr = 10 voxels; Figure 3 and Tables 3 and 4). Both the PTSD and TEC groups showed increased dACC FC with the bilateral MFG/IFG and STG/insula (p < 0.05, AlphaSim corrected; Pthr = 0.01, CSthr = 10 voxels), but decreased dACC FC with the right hippocampus (p < 0.05, AlphaSim corrected; Pthr = 0.01, CSthr = 5 voxels) and the left cerebellum (p < 0.05, AlphaSim corrected; Pthr = 0.01, CSthr = 10 voxels) relative to the HC group (Figure 3 and Tables 4 and 5). In addition, the PTSD group also shows decreased dACC FC with bilateral MFG/IFG (p < 0.05, AlphaSim corrected; Pthr = 0.01, CSthr = 10 voxels) and the right hippocampus (p < 0.05, AlphaSim corrected; Pthr = 0.01, CSthr = 5 voxels) relative to the TEC group (Figure 3 and Table 3).
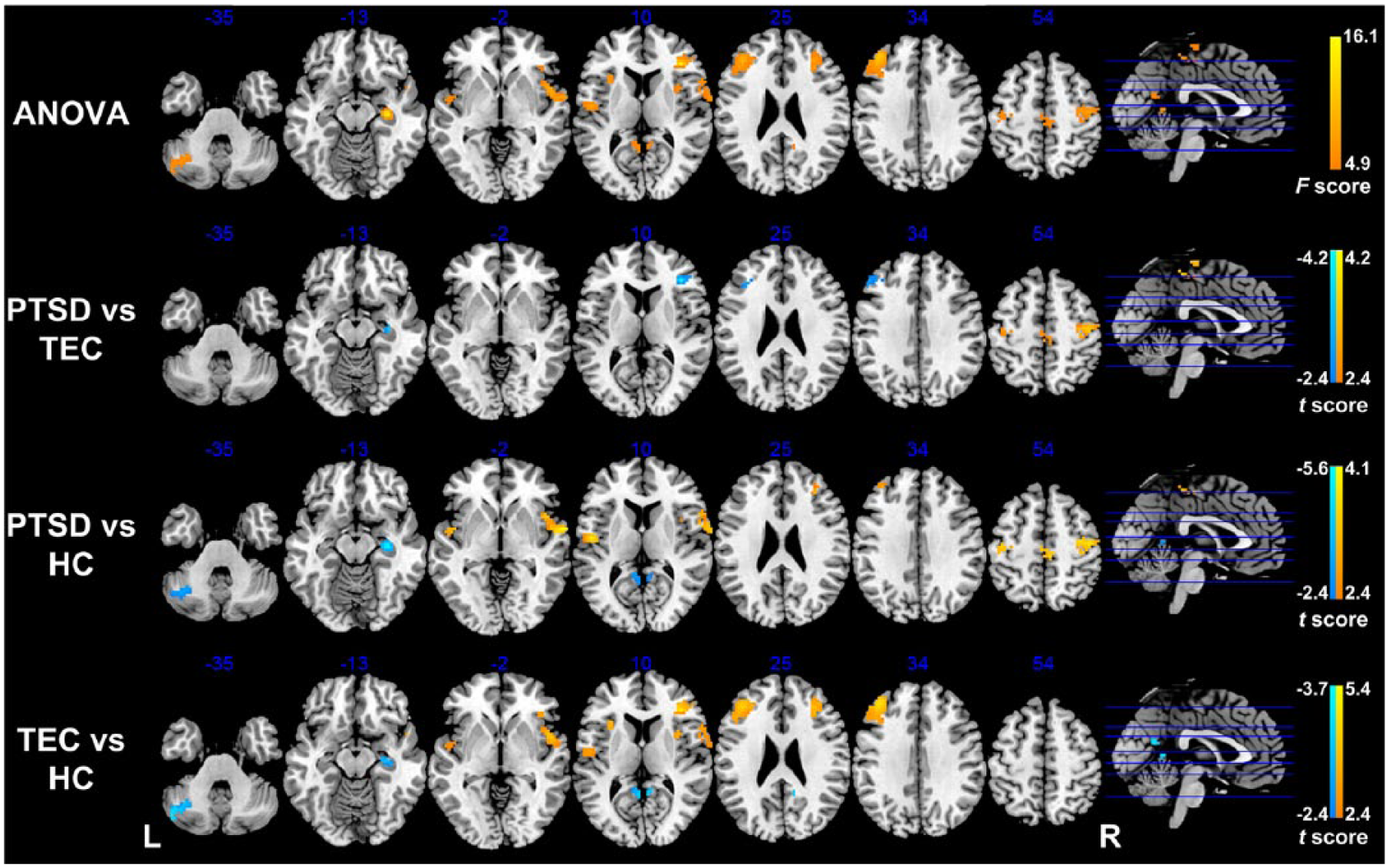
Group differences in dACC functional connectivity revealed by ANOVA and post hoc t tests. The PTSD group shows increased dACC functional connectivity with the right paracentral lobule and bilateral PreCG/PoCG relative to both the TEC and HC groups; both the PTSD and TEC groups show increased dACC functional connectivity with the bilateral MFG/IFG and STG/insula, but decreased dACC functional connectivity with the right hippocampus and left cerebellum relative to the HC group; the PTSD group also shows decreased dACC functional connectivity with bilateral MFG/IFG and right hippocampus relative to the TEC group (p < 0.05, AlphaSim corrected).
Brain regions showing differences in functional connectivity with the dACC among the PTSD, TEC and HC groups.
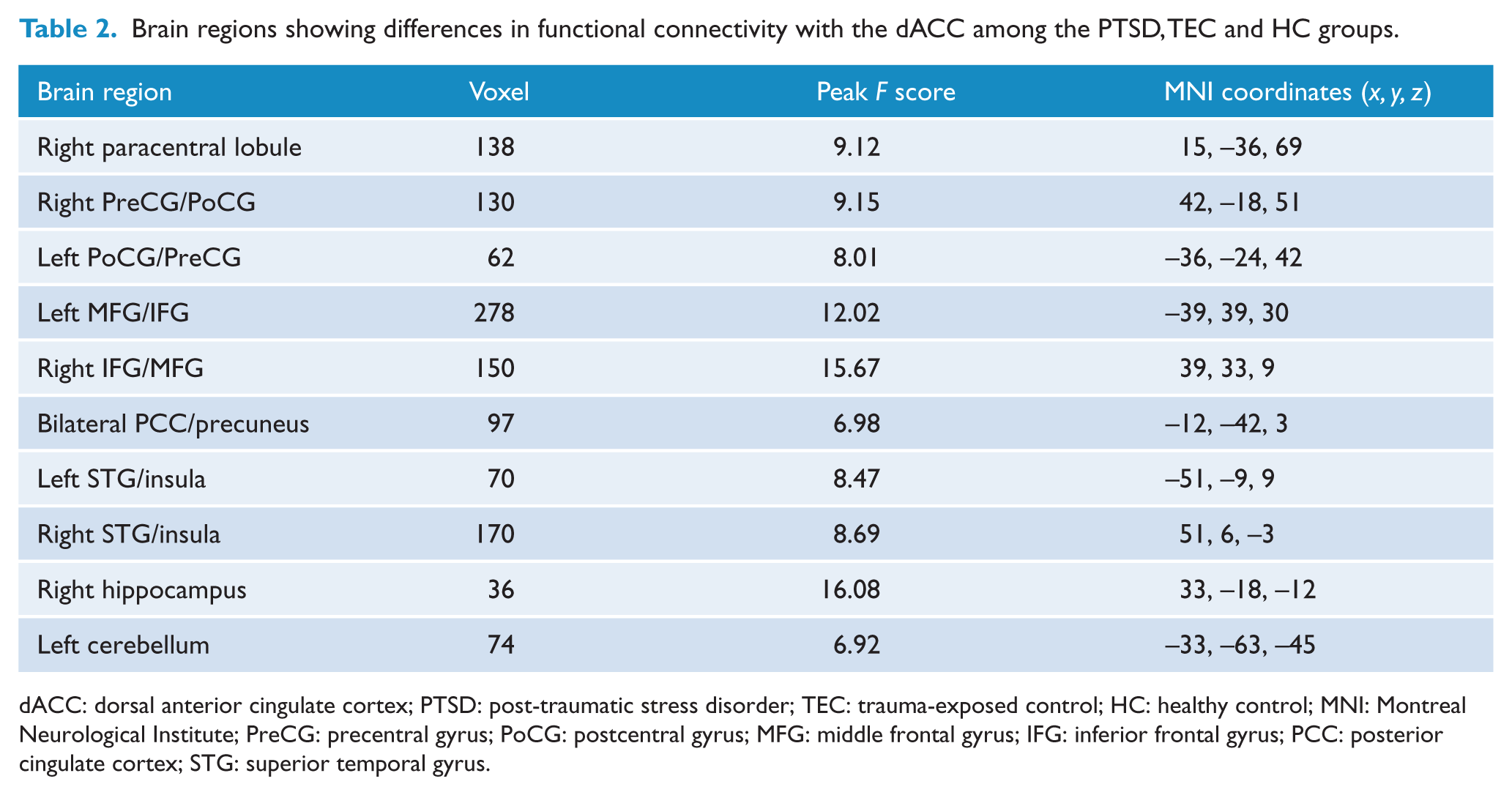
dACC: dorsal anterior cingulate cortex; PTSD: post-traumatic stress disorder; TEC: trauma-exposed control; HC: healthy control; MNI: Montreal Neurological Institute; PreCG: precentral gyrus; PoCG: postcentral gyrus; MFG: middle frontal gyrus; IFG: inferior frontal gyrus; PCC: posterior cingulate cortex; STG: superior temporal gyrus.
Brain regions showing dACC functional connectivity difference between the PTSD and TEC groups.

dACC: dorsal anterior cingulate cortex; PTSD: post-traumatic stress disorder; TEC: trauma-exposed control; MNI: Montreal Neurological Institute; PreCG: precentral gyrus; PoCG: postcentral gyrus; MFG: middle frontal gyrus; IFG: inferior frontal gyrus.
Brain regions showing dACC functional connectivity difference between the PTSD and HC groups.
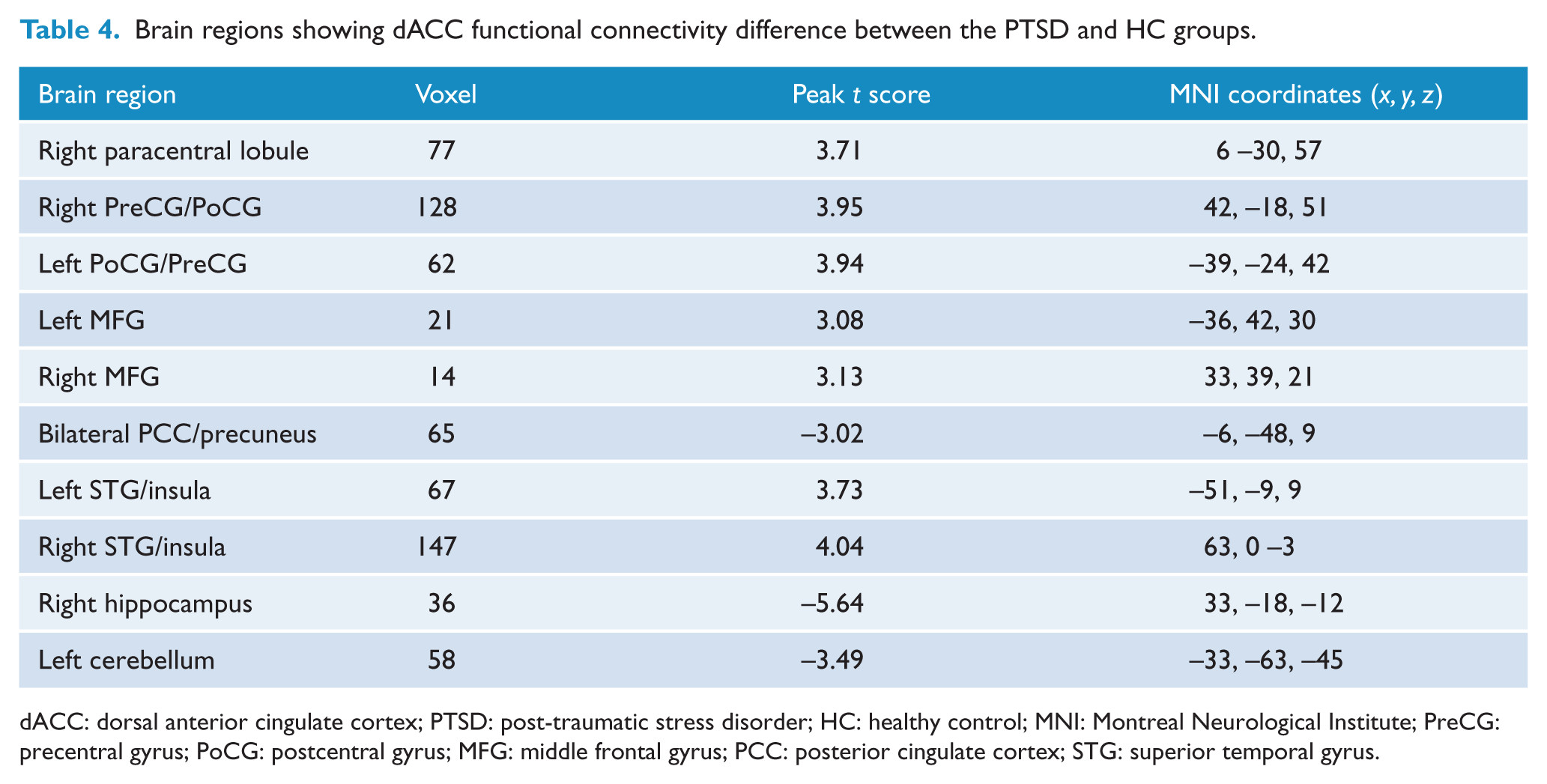
dACC: dorsal anterior cingulate cortex; PTSD: post-traumatic stress disorder; HC: healthy control; MNI: Montreal Neurological Institute; PreCG: precentral gyrus; PoCG: postcentral gyrus; MFG: middle frontal gyrus; PCC: posterior cingulate cortex; STG: superior temporal gyrus.
Brain regions showing dACC functional connectivity difference between the TEC and HC groups.
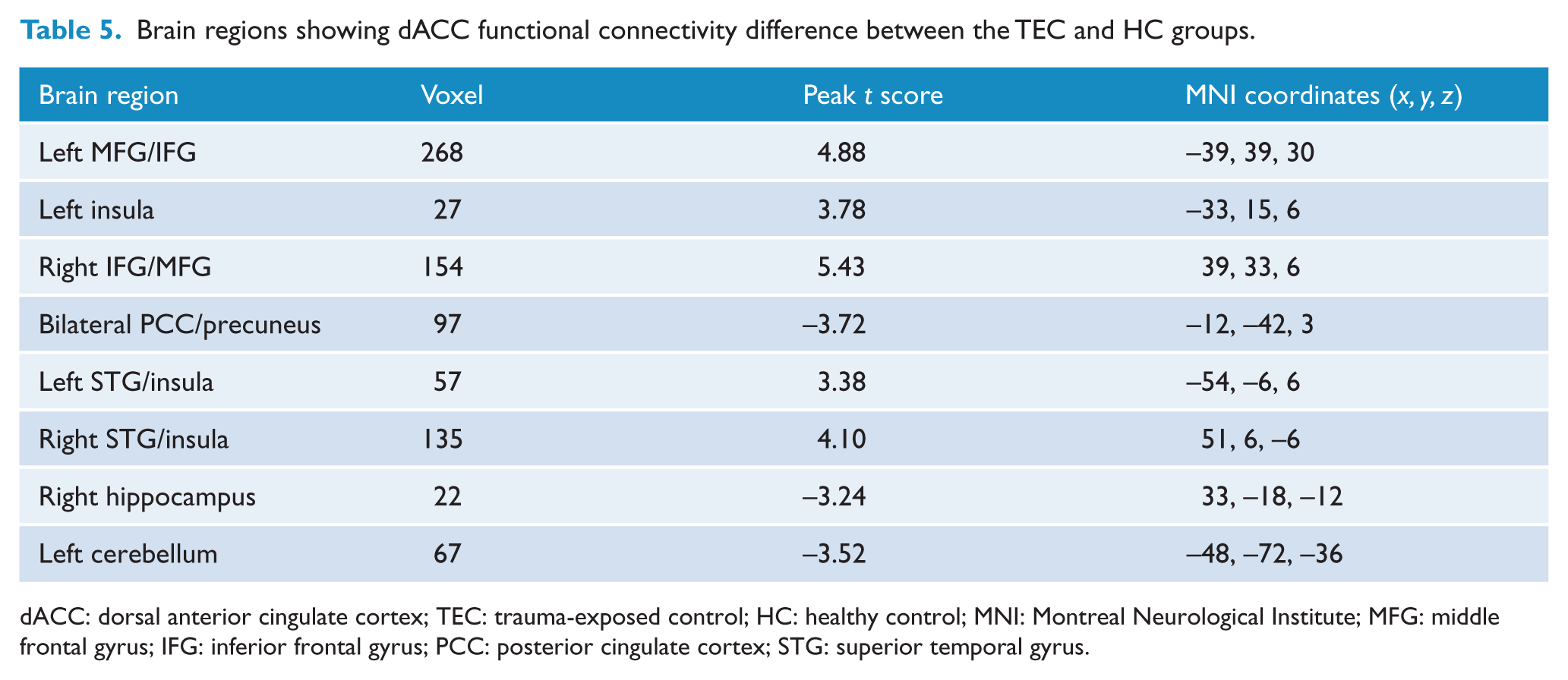
dACC: dorsal anterior cingulate cortex; TEC: trauma-exposed control; HC: healthy control; MNI: Montreal Neurological Institute; MFG: middle frontal gyrus; IFG: inferior frontal gyrus; PCC: posterior cingulate cortex; STG: superior temporal gyrus.
Across all traumatized subjects, the PCL score was positively correlated with FC between the dACC and right paracentral lobule as well as the dACC and right PreCG/PoCG, but is negatively correlated with FC between the dACC and left MFG (p < 0.05, not corrected; Figure 4). In the PTSD group, the objective support score was negatively correlated with FC between the dACC and the left (R = –0.434, p = 0.024) and right PreCG/PoCG (R = –0.455, p = 0.017; Figure 5). The SSRS total score (R = 0.419, p = 0.029) and subjective support score (R = 0.496, p = 0.008) were positively correlated with FC between the dACC and left MFG, respectively (Figure 5).
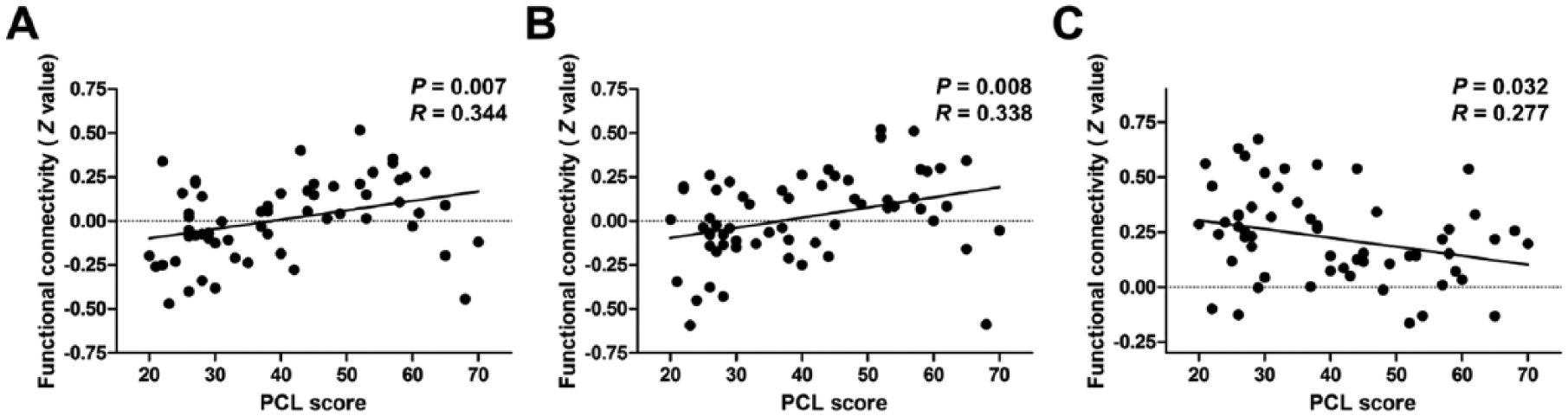
Results of correlation analyses between dACC functional connectivity and PTSD symptom severity. The PCL score is positively correlated with functional connectivity between (A) the dACC and right paracentral lobule, (B) the dACC and right PreCG/PoCG, but is negatively correlated with functional connectivity between (C) the dACC and left MFG (p < 0.05, not corrected).
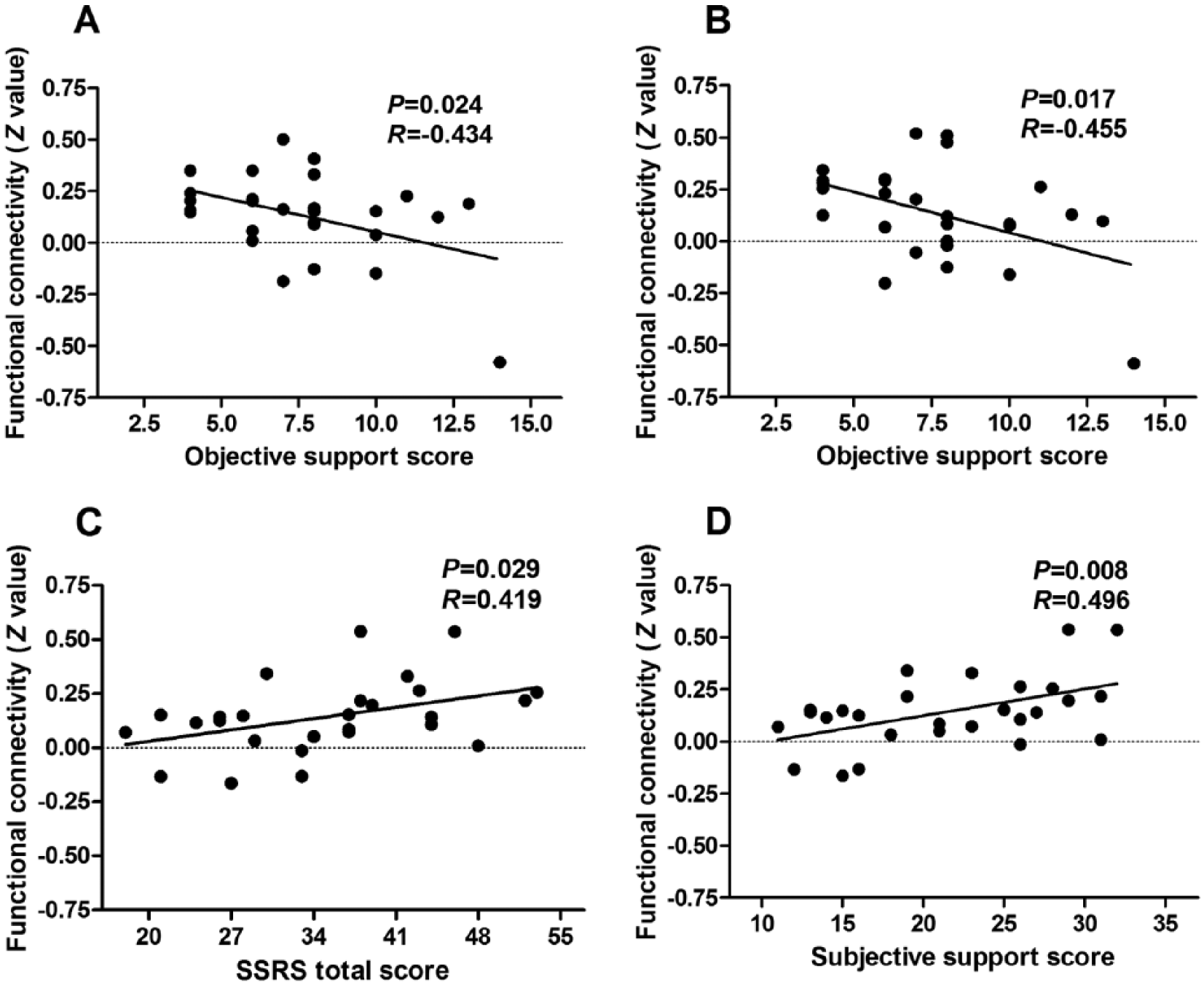
Results of correlation analyses between dACC functional connectivity and social support in the PTSD group: the objective support score is negatively correlated with functional connectivity between (A) the dACC and left PreCG/PoCG, (B) the dACC and right PreCG/PoCG (p < 0.05, not corrected); (C, D) the SSRS total score and subjective support score are positively correlated with functional connectivity between the dACC and left MFG (p < 0.05, not corrected).
Discussion
This study demonstrated that both the PTSD and TEC groups showed decreased ALFF in the dACC. The PTSD group showed increased dACC FC with the right paracentral lobule and bilateral PreCG/PoCG relative to two control groups. Both the PTSD and TEC groups demonstrated decreased dACC FC with the right hippocampus relative to the HC group. Importantly, the decreased dACC FC with the right hippocampus was more prominent in the PTSD group than the TEC group. In addition, abnormal FC may be associated with PTSD symptom severity.
The decreased ALFF in dACC in both the PTSD and TEC groups supported our hypothesis that in those who experienced traumatic event but without the diagnosis of PTSD, brain function changes may still occur. A recent meta-analysis (Stark et al., 2015) suggested that trauma has a measurable, enduring effect upon the functional dynamics of the brain, even in individuals who experience trauma but do not develop PTSD. Ke et al. (2017) also found that post-traumatic stress had an effect on local and remote FC in all trauma-exposed individuals, which fitted well with our findings. ACC has long been considered to play a pivotal role in emotional processing. It is generally considered that dACC is involved in negative emotion expression (Etkin et al., 2011). Importantly, the PTSD group had significantly lower subjective support, objective support and SSRS total scores than the TEC and HC groups. The low levels of social support in the PTSD group might be of significance since social support could reduce the harm caused by stressful events (Xu and Ou, 2014). One might adjust to crises more smoothly with higher levels of social support. This study also found that the objective support score, SSRS total score and subjective support score were correlated with alterations in the FC in the PTSD group (though lack of correction for multiple comparisons), indicating that social support might be associated with cerebral abnormalities in the PTSD patients. A previous study has found that low social support was associated with the onset of the PTSD (Feng et al., 2007), while strong and positive social support was beneficial for the recovery from prior PTSD (Dai et al., 2016). Relative absence of support creates an increased risk for PTSD, whereas positive support predicts lower risks for the onset of PTSD (Bryant, 2016; Charuvastra and Cloitre, 2008). Subjective support has always been reported to be a more powerful predictor of subsequent improvement in psychological disorders than objective measures of social support (Charuvastra and Cloitre, 2008; Dai et al., 2016; Feng et al., 2007). Surprisingly, objective and support scores were found to be correlated with alterations in the FC in the PTSD group in our study, indicating that both these two kinds of support were important for PTSD patients. The reason for this inconsistency might be lack of correction for multiple comparisons. Therefore, it is of great clinical implication that the improvement of social support might possibly be of great significance for PTSD patients. MFG is an important component of the prefrontal cortex, which is associated with the response to reward stimuli (Charuvastra and Cloitre, 2008). PTSD patients had been reported to have smaller signal changes in the prefrontal cortex (Charuvastra and Cloitre, 2008). Our study provides further evidence that this region may be implicated in the pathogenesis of PTSD. No one has ever reported the role of PreCG/PoCG in the PTSD patients. All in all, few studies have investigated the relationship between social support and brain functional alterations in PTSD patients. Thus, conducting more studies looking at the three different groups, trauma-exposed, trauma-naïve and PTSD is important to achieve better understanding of the neural mechanisms of PTSD. The association of social support with FCs should be further studied in the future.
Our results demonstrated that the PTSD group had increased dACC FC with the right paracentral lobule and bilateral PreCG/PoCG compared with both the TEC and HC groups. Paracentral lobule is thought to be involved in shock processing (Wiemer and Pauli, 2016). Pre- and postcentral gyri are important components of the sensorimotor network, which contributes to skill learning accompanied by FC changes within the primary motor cortex. dACC is a critical part of the salience network (SN) that processes autonomic, interoceptive, homeostatic and cognitive information of personal relevance (Seeley et al., 2007; Ziemann et al., 2004). Therefore, we speculated that enhanced FC of the motor cortex may be related to the preparation for coping with a physical threat (Bremner et al., 1999). This might underline the neural–psychobiological mechanisms of PTSD. Importantly, altered FC between the dACC and right paracentral lobule as well as the dACC and right PreCG/PoCG positively correlated with the PCL score (lack of correction for multiple comparisons). Taken together, these results provide important insight into the understanding of neuropathology of PTSD.
Interestingly, increased FC was identified in the bilateral insula/STG, MFG/IFG and decreased dACC FC with the right hippocampus and left cerebellum in both the PTSD and TEC groups. Greater insula activation has been linked to trauma recall (Britton et al., 2006) and viewing fearful faces (Fonzo et al., 2010) in individuals with PTSD. As a matter of fact, insula hyperresponsivity in patients with PTSD has previously been demonstrated to positively correlate with hyperarousal symptoms (Simmons et al., 2008) and re-experiencing severity (Hopper et al., 2007). Meanwhile, the insula, IFG and MFG are important components of the SN, which is associated with attention, cognition, control, working memory and switching (Seeley et al., 2007; Sridharan et al., 2008). The SN is believed to be involved in the recently proposed triple-network model (Menon, 2011; Patel et al., 2012) in PTSD. The lower dACC FC with bilateral MFG/IFG in PTSD suggested that more impairment occurred in these regions in this population. Our finding provides further support for this hypothesis. The cerebellum has also been suggested to be associated with the pathophysiology of PTSD (De Bellis and Kuchibhatla, 2006). The present results indicated that exposure to traumatic events may result in altered function in the SN, hippocampus and cerebellum in all trauma-exposed individuals, not just those with PTSD. Moreover, trauma survivors may exhibit distinct brain activity in these regions in a quantitative gradient. This was supported by the result that the PTSD group shows decreased dACC FC with bilateral MFG/IFG and right hippocampus relative to the TEC group.
The hippocampus plays a critical role in encoding emotional memory and modulating appropriate emotional responses to fear relevant stimuli. This study found that both the PTSD and TEC groups showed decreased dACC FC with the right hippocampus relative to the HC group. Importantly, a gradually decreasing trend was observed from the TEC group to the PTSD group. A previous study found reduced activity in the hippocampus in PTSD patients during successful encoding of trauma-related stimuli and the hippocampal activity was negatively associated with hyperarousal symptom severity (Hayes et al., 2011). Similarly, Astur et al. (2006) demonstrated that reduced hippocampal activity was associated with more severe PTSD symptoms. All these findings are in support of neurobiological theories positing reduced hippocampal activity under conditions of high stress and arousal. However, some studies found that increased activation in the hippocampus may be associated with negative response in PTSD patients (Brohawn et al., 2010; Thomaes et al., 2009). The inconsistent results may be related to the tasks selected. More studies are needed to clarify these inconsistencies.
This study had several limitations that merit consideration. First, the sample size was relatively small. Future studies with large cohort should be considered. Second, this was a cross-sectional study and we cannot ascertain that the functional alteration pattern we found in PTSD was definitively related to this disease rather than to previous risk factors for the disorder. Third, our study mainly focused on typhoon-related PTSD, and the findings could not be generalized to PTSD resulting from other etiology. Fourth, there was a lack of correction for multiple comparisons in the correlational analyses. Fifth, there was no Chinese version of a Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5) measure when we conducted the study. Last but not least, levels of subjective exposure to the typhoon and past trauma histories might have an effect on the results of the study. These two factors should be taken into consideration in the future.
In conclusion, aberrant local and network-level FC was found in all typhoon-exposed individuals. Altered ALFF in dACC may be a predisposing risk factor for PTSD development following trauma exposure. Increased dACC coupling with emotion production regions including the paracentral lobule, PreCG/PoCG and insula and reduced dACC coupling with emotion regulation and contextualization regions, including the hippocampus and the cerebellum, were found in patients with PTSD. The more prominent decreased dACC FC with the right hippocampus might be specific for the PTSD group. Improvement of social support might possibly be of great significance for PTSD patients.
Footnotes
Acknowledgements
H.J.C., L.Z. and J.K. have contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Nature Science Foundation of China (Grant nos. 814602161, 81760308, 81871346, 81801684), Key Science and Technology Project of Hainan Province (Grant nos. ZDYF2018141), Natural Science Foundation of Hainan Province (Grant nos. 818MS124).
