Abstract
Objective:
To examine the effects of 18 months of intensive stabilisation with medication management and Interpersonal and Social Rhythm Therapy or Non-specific Supportive Clinical Management on cognitive function in young people with bipolar disorder. Determinants of change in cognitive function over the 18 months of the trial were also examined.
Method:
Patients aged 15–36 years with Bipolar I Disorder, Bipolar II Disorder and Bipolar Not Otherwise Specified were recruited. From a battery of cognitive tests, change scores for pre-defined domains of cognitive function were created based on performance at baseline and follow-up. Change was compared between the two therapy groups. Regression analysis was used to determine the impact of a range of clinical variables on change in cognitive performance between baseline and follow-up.
Results:
One hundred participants were randomised to Interpersonal and Social Rhythm Therapy (n = 49) or Non-specific Supportive Clinical Management (n = 51). Seventy-eight patients underwent cognitive testing at baseline and 18 months. Across both groups, there were significant improvements in a Global Cognitive Composite score, Executive Function and Psychomotor Speed domains from baseline to 18 months. Lower scores at baseline on all domains were associated with greater improvement over 18 months. Overall, there was no difference between therapies in change in cognitive function, either in a global composite score or change in domains.
Conclusion:
While there was no difference between therapy groups, intensive stabilisation with psychological therapy was associated with improved cognitive function, particularly in those patients with poorer cognitive function at baseline. However, this was not compared with treatment as usual so cannot be attributed necessarily to the therapies.
Keywords
Introduction
Bipolar disorder (BD) is a chronic, recurring disorder (Judd et al., 2002, 2003). While most patients with BD recover from acute episodes of mania and acute episodes of depression, up to 60% of patients do not return to their pre-morbid level of functioning (Bauer et al., 2001; Judd et al., 2002, 2003) even during periods of sustained and substantial remission (Fagiolini et al., 2005). Earlier age of onset has been associated with a greater risk of recurrence, increased chronicity of mood symptoms and impairment (Perlis et al., 2009). It appears treatment is more effective if it occurs early in the course of the illness (Berk et al., 2009), indicating the importance of early access to interventions in reducing the chronicity and impairment associated with the disorder (Miklowitz, 2008).
One factor which may contribute to poor outcome in BD, in a proportion of patients, is impaired cognitive function. Studies in euthymic BD suggest that depending on the exact nature of the group studied, there are deficits of effect size 0.5–1.0 across cognitive domains (see Porter et al., 2015, for review of meta-analyses). However, not all patients are impaired and indeed there is probably a group of patients who are cognitively normal (Burdick et al., 2014). The percentage who are significantly impaired depends on the clinical characteristics of the group and on the way in which impairment is classified, with rates of cognitive impairment in euthymic BD between 20% and 80% depending on the definition (Douglas et al., 2018). The degree of cognitive impairment correlates to an extent with aspects of social and occupational functioning (Baune and Malhi, 2015) and with degree of ongoing mood morbidity (Martinez-Aran et al., 2004). Studies examining this issue often include a high percentage of more chronic and older patients (Porter et al., 2015). However, studies in first episode BD also suggest significant impairment (Lee et al., 2014) and there is even the suggestion of significant impairment prior to the first episode of mania or hypomania (Bora and Ozerdem, 2017b).
A number of previous studies in BD have examined cognitive function at baseline and then after a variable period of follow-up. Neither shorter term (mean 1.5 years) nor longer term (5.5 years) studies have suggested cognitive decline over time and, in fact, have shown modest improvements in tasks testing memory and working memory at follow-up (Bora and Ozerdem, 2017b). A number of these studies are in older patients and in patients around the age of 40 years, who are therefore some years after onset (Bora and Ozerdem, 2017b). Studies examining the longitudinal course of cognitive function in groups of younger patients have been over 1–2 years and have either shown no change in cognitive function (Bombin et al., 2013; Leany, 2010; Yucel et al., 2007) or improvements in aspects of cognitive function (Lee et al., 2015; Lera-Miguel et al., 2015; Torres et al., 2014).
Several studies have examined pharmacological agents and cognitive remediation strategies which have been hypothesised to improve cognitive function in BD. To date, the effects have been relatively limited (Miskowiak et al., 2016a), perhaps related, in part, to methodological issues (Miskowiak et al., 2017). No studies have examined the effects of more traditional psychological therapies on cognitive function in BD.
This randomised controlled psychological therapy study was designed to compare the efficacy of two 18-month psychotherapies: Interpersonal and Social Rhythm Therapy (IPSRT) and a ‘control’ psychological intervention, Non-specific Supportive Clinical (SSC) management, in younger people (aged 15–36 years) with BD. Patients in both groups also received psychopharmacological treatment. We hypothesised, primarily, that IPSRT would be superior to SSC in reducing depressive symptom burden, as measured by the Longitudinal Interval Follow-up Evaluation (LIFE; Keller et al., 1987), over 6–18 months from baseline, in young people with BD. The results of the primary analysis and of two secondary analyses (change in social functioning and cumulative manic/hypomanic burden) have been reported (Inder et al., 2015). Originally, we had two main hypotheses regarding cognitive function. First, a reduction in symptom burden, occurring in the process of stabilisation with psychological therapy and systematic pharmacotherapy, would result in an improvement in cognitive function between baseline and 18 months in both groups. Second, a greater reduction in symptom burden associated with IPSRT compared with SSC would result in greater improvement in cognitive function in the group of patients receiving IPSRT. The primary analysis, however, revealed a reduction in symptom burden but no difference between treatment groups. We therefore revised our second hypothesis to a null hypothesis – that there would be no difference in change in cognitive function between treatment groups. A further aim of the study is to determine which clinical and demographic factors other than treatment predicted change in cognitive function over a period of 18 months of stabilisation.
Method
Trial design
Further details of methodology are given in the original trial paper (Inder et al., 2015). This was a randomised controlled trial (RCT) of IPSRT versus SSC over 18 months. Ethical consent was obtained from the Canterbury Ethics Committee. The trial was registered with the Australian and New Zealand Clinical Trials Registry (ACTRN12605000722695).
Participants
The study recruited 100 adolescents and young adults aged 15–36 years with BD I, BD II and BD Not Otherwise Specified (BD NOS), with an adequate understanding of English, to participate in psychotherapy. Participants were recruited from a range of services including mental health services and General Practitioners. Exclusion criteria were schizophrenia or schizoaffective disorder and severe alcohol or drug dependence.
Clinical assessment
After giving consent and prior to randomisation, the treating psychiatrist completed an Axis I diagnostic assessment using the Structured Clinical Interview for DSM-IV (SCID; Spitzer et al., 1992). Mood symptom severity was rated using the Montgomery–Asberg Depression Rating Scale (MADRS; Montgomery and Asberg, 1979) and the Young Mania Rating Scale (YMRS; Young et al., 1978). The LIFE (Keller et al., 1987) was completed by an experienced research nurse, who was blind to treatment allocation, and covered the 6 months leading up to recruitment into the study.
Randomisation
At the time of consent, patients were allocated a research identification number. Randomisation for therapy modality was computer generated following a block design set up by a biostatistician and allocated after completion of the initial assessment period.
Cognitive assessment
Cognitive function was not originally reported as an outcome measure in the Clinical Trial Register but was planned prior to initiation of the study. All patients were tested at baseline, prior to the commencement of therapy, and at the end of the 18-month treatment period. Participants commenced cognitive testing at 1 p.m. following a light lunch. Testing took approximately 100 minutes. Pencil-and-paper tests were administered according to standardised instructions (Lezak et al., 2004). Computerised tests from the Cambridge Neuropsychological Test Automated Battery (CANTAB®) were administered according to CANTAB® manual protocols on a laptop fitted with a colour touch-screen monitor. The tests used are briefly summarised in the following.
Rey Auditory Verbal Learning Test
The Rey Auditory Verbal Learning Test (RAVLT; Rey, 1964) assesses verbal learning and memory. For this study, the recognition component of the RAVLT was presented as a computerised test using SuperLab Pro® version 2.0.
Spatial Recognition Memory
Participants learnt the spatial location of five squares presented sequentially at different on-screen locations in the Spatial Recognition Memory (SRM) test (CANTAB). Pairs of squares were presented and participants identified which of the two squares was in the index location by touching the correct square. Accuracy and reaction time were reported for the four trials.
Controlled Oral Word Association Test
In the Controlled Oral Word Association Test (COWAT; Benton and Hamsher, 1976), participants generated words beginning with the letters F, A and S over a 1-minute period for each letter. Rules included exclusion of proper nouns, place names or the same word with a different ending. Retesting used the letters P, R and W.
Digit Span Forwards and Backwards
The Digit Span Test involved participants reciting increasing lengths of digit series, either forwards or backwards, until a pair of sequences was incorrect or a nine-digit sequence was completed correctly (Wechsler, 1997).
Trail Making Test – Parts A and B
In Part A, the participant was asked to draw a line to connect circled numbers in a sequential order as quickly as possible. In Part B, the participant connected circled numbers and letters in an alternating sequence, again as quickly as possible.
Spatial Span
Participants remembered the order of white squares on-screen that changed colour one-by-one on the Spatial Span (SS) test (CANTAB). Trials progressed from two to nine squares and the test self-terminated after three successive trial failures on a given number of squares. The longest span length correctly recalled was reported.
Spatial Working Memory
During the Spatial Working Memory (SWM) task (CANTAB), participants searched through on-screen boxes to find a blue token, beginning with four trials of four boxes, progressing to four trials of six and then eight boxes. Repetitious search errors were reported and an efficiency index for search strategy was generated.
Delayed Matching to Sample
The task involved the participant being shown a complex visual pattern. The participant then selected the original pattern from four options which were shown either immediately or after a 4 or 12 second delay.
Motor Screening
The Motor Screening test (CANTAB) screens for motor and visual disorders and served as training for CANTAB® (use of the touch screen). Participants touched a series of pink and green crosses as they appeared on the screen. Mean reaction time (milliseconds) to correctly respond to each of 10 crosses was recorded.
Rapid Visual Information Processing
The Rapid Visual Information Processing task (RVIP; CANTAB) is used to measure visual sustained attention. A white box was presented to participants on a computer screen, within which numbers that ranged from 1 to 9 appeared one at a time in a pseudo-random order; these numbers were presented at a rate of 100 per minute. Participants were instructed to detect target sequences of digits (e.g. 3-5-7) and to respond by pressing a button on the response pad at the end of the target sequence. Primary outcome measures were mean correct latency and the total number of hits.
Psychopharmacological Treatment
Psychiatrists adjusted medication using clinical judgement and guided by a pharmacological decision tree, consistent with the American Psychiatric Association (APA, 1994) guidelines for the treatment of patients with BD (see Inder et al., 2015, for further details). The psychiatrist saw the patient fortnightly for 2 months, monthly for 4 months and subsequently every 2 months or earlier if indicated by the patient’s mood state.
Psychological Treatment
After randomisation, the therapist was provided with a sealed envelope advising of the therapy-type allocation. Therapists met with patients weekly for 3 months, fortnightly for up to 6 months and then fortnightly to monthly from 6 to 18 months. Variations in therapy frequency were tailored to the patient’s mental state.
IPSRT for BD was delivered according to a manual devised by Frank et al. (2001). SSC was designed as a ‘control’ psychological therapy by the researchers based on APA (1994) guidelines for the management of BD as well as the core features of supportive psychotherapy (for further details of both therapies see Inder et al., 2015).
Statistical analyses
Analyses were conducted using the Statistical Package for the Social Sciences (SPSS) version 19 for Windows (SPSS, 2004).
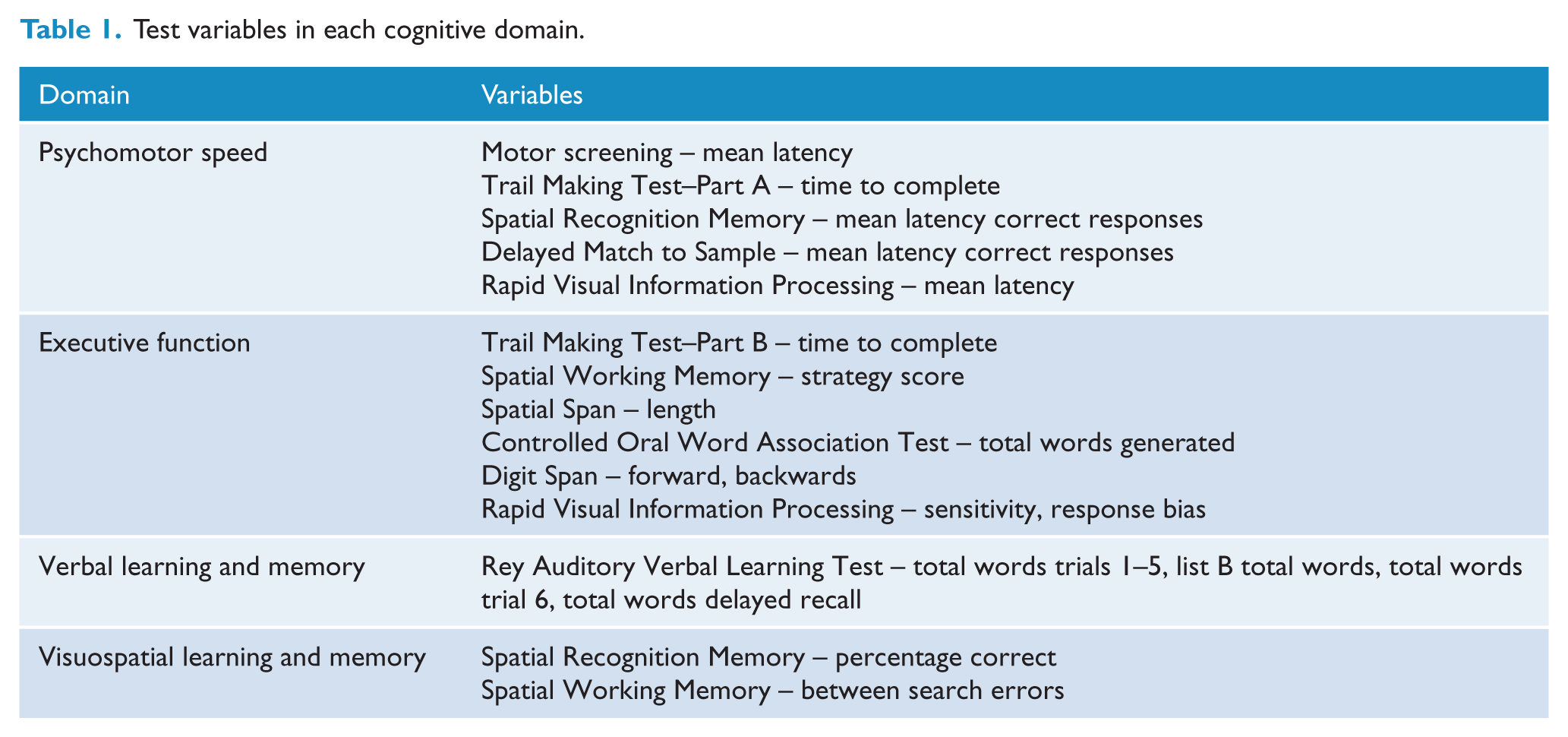
Scores on cognitive test variables at baseline were converted to z-scores, calculated as the difference from the mean of the whole group at baseline, divided by the group standard deviation at baseline. Change scores were calculated as change from group mean at baseline divided by the group standard deviation at baseline. In each case, this was computed so that a positive score indicated an improvement from baseline. Variables were grouped into domains according to an a priori decision, as shown in Table 1. Z-scores for each domain were computed as the mean of the z-scores of the individual variables in that domain. This then gave a baseline score and a change score for each patient in each domain. A Global Cognitive Composite score was computed based on the mean of all variables for each patient.
Test variables in each cognitive domain.
Change in cognition
Significance of change in Global Cognition and in each domain was calculated using one-sample t-tests on the domain change scores. Analysis of covariance (ANCOVA) was conducted with therapy type as the between-participants factor and baseline z-score for each domain as a covariate.
Planned regression analysis
The relationship between demographic and clinical variables and change in cognitive function in each domain was examined using Pearson correlation coefficients or for non-continuous data, independent t-tests. Where a test was significant at the level of p < 0.1, it was planned that this variable would subsequently be entered into a multivariate analysis in order to determine predictors of change in cognitive function. Variables examined were therapy type (IPSRT/SSC), baseline cognitive domain score, age, gender, bipolar subtype (BD II and BD NOS were grouped together), substance use disorder (present or absent), co-morbid anxiety disorder (present or absent), change in LIFE depression/mania/total (baseline to follow-up), change in MADRS and YMRS (baseline to follow-up), change in the MADRS sleep score and number of previous episodes of depression/mania. The possible influence of being on medication was examined by dividing medication into (a) lithium, (b) antipsychotics, (c) sodium valproate and (d) antidepressants. Sodium valproate was analysed separately because most patients on an anticonvulsant mood stabiliser were on this and it was considered that lamotrigine and carbamazepine were significantly different in likely cognitive profile. In order to create a single variable which reflected being treated with these medications for the majority of the 18 months, we divided patients into those who had been on the medication in question for 8 of 10 time points (time points were baseline and every 2 months thereafter). This therefore included mainly patients who were on the medication throughout the study period or those who were started on the medication within 4 months of commencing in the study.
Results
Sample characteristics
One hundred patients were randomised, with 49 patients randomised to IPSRT and 51 to SSC. Seventy-eight patients (IPSRT [n = 35], SSC [n = 43]) completed both baseline and follow-up cognitive testing sessions. No significant differences between groups were found on demographic or clinical characteristics in those who completed follow-up testing (see Table 2).
Baseline demographic and clinical characteristics of the sample.
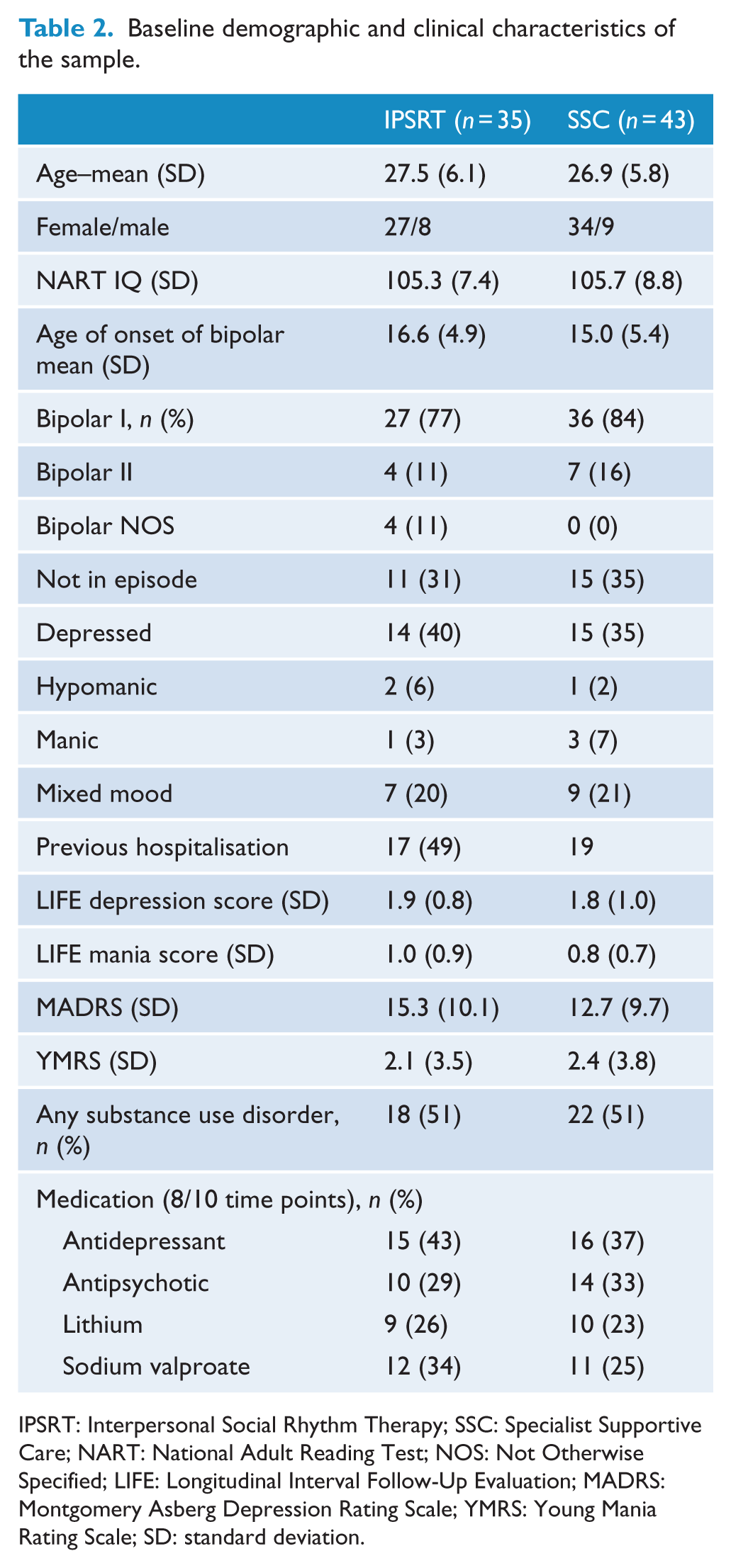
IPSRT: Interpersonal Social Rhythm Therapy; SSC: Specialist Supportive Care; NART: National Adult Reading Test; NOS: Not Otherwise Specified; LIFE: Longitudinal Interval Follow-Up Evaluation; MADRS: Montgomery Asberg Depression Rating Scale; YMRS: Young Mania Rating Scale; SD: standard deviation.
One hundred patients entered the study of whom three did not have baseline cognitive testing. A further 19 patients did not complete follow-up cognitive testing. Significant differences in demographic and clinical characteristics were found between patients who completed follow-up cognitive testing (n = 78) compared with those who did not (n = 22). Non-completers were significantly younger (mean = 24.2, standard deviation (SD) = 5.5 vs mean = 27.1, SD = 5.8, t = 2.1, p = 0.04) and had earlier age of onset (mean = 13.3, SD = 3.2 vs mean = 15.8, SD = 5.2, t = 2.1, p = 0.04). Scores on the National Adult Reading Test (NART), an estimate of verbal IQ, was significantly lower in non-completers (mean = 100.4, SD = 7.8 vs mean = 105.6, SD = 8.1, t = 2.6, p = 0.01). Comparing the 19 patients who had baseline cognitive testing but not follow-up cognitive testing, with the 78 who had both, there were no differences in any of the domains or in global cognition at baseline. Other clinical and demographic characteristics were not significantly different between groups.
Medication type, based on what patients were taking for the majority of the study (see Method section), is reported in Table 2.
Change in cognitive function
Figure 1 shows change in cognitive function from baseline to follow-up, expressed as a Z-score. Global Cognitive Composite, Executive Function and Psychomotor Speed improved significantly from baseline to 18-month follow-up. There was no significant change in Verbal or Visuospatial Learning and Memory.
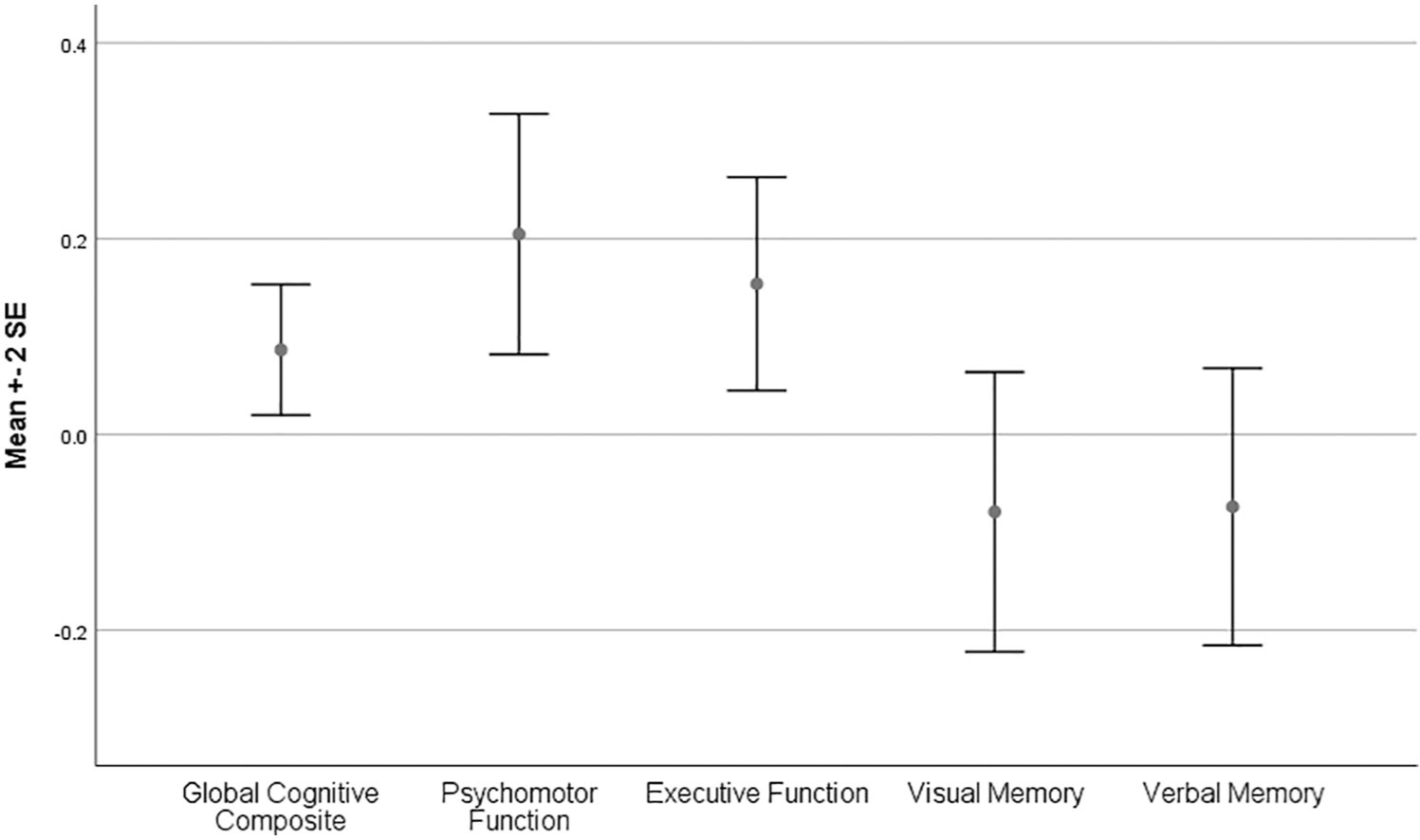
Change in global cognitive composite and individual domains between baseline and 18 months.
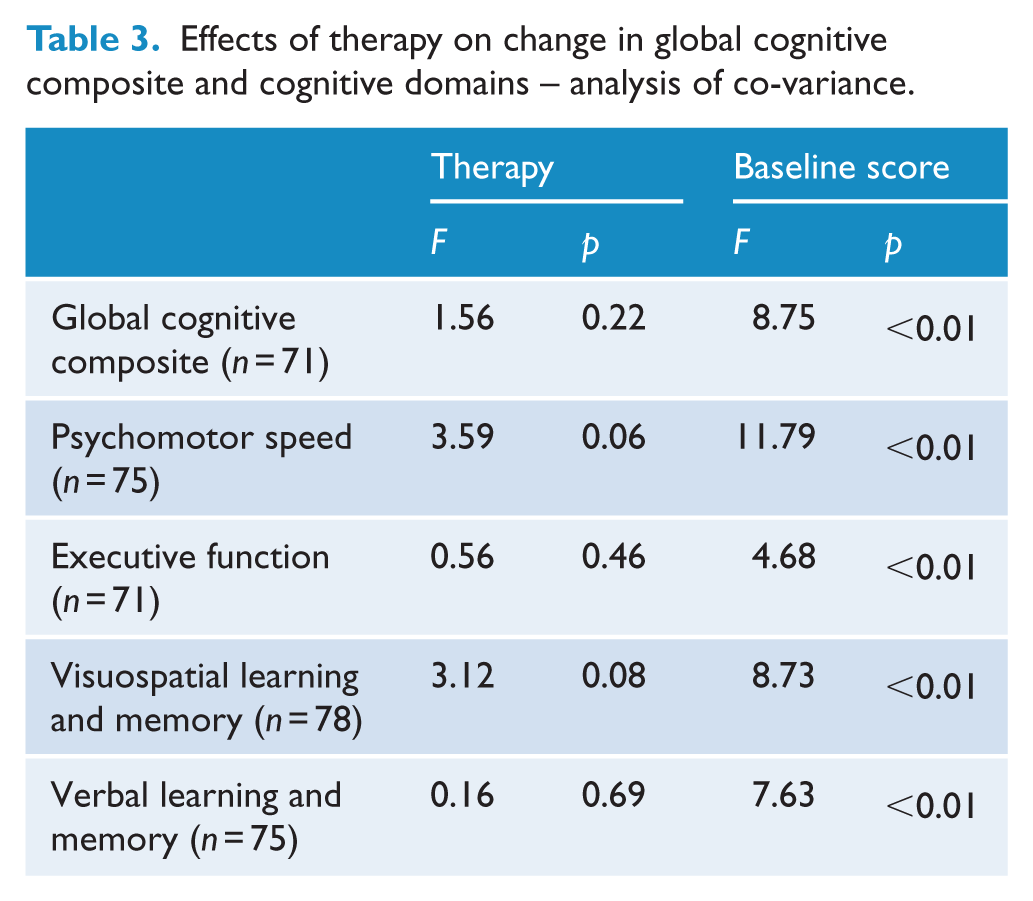
There was no difference in change in Global Cognitive Composite, or individual domains, between the groups receiving IPSRT and SSC when baseline function on the equivalent domain was taken into account. Table 3 shows the results examining the effect of therapy type on change in cognitive function with baseline cognitive scores as covariates. In each case, baseline score was a significant covariate with a lower score at baseline being associated with greater change.
Effects of therapy on change in global cognitive composite and cognitive domains – analysis of co-variance.
Planned regression analysis
In correlation analysis and t-tests, none of the hypothesised variables except for baseline score on the corresponding cognitive domain were associated with change in cognitive function (p > 0.1 for any of the domains and Global Cognitive Composite). Therefore, a further regression analysis was not conducted. The effects of baseline score are seen in the ANCOVA (Table 3).
Discussion
This study examined cognitive change over 18 months in an RCT comparing IPSRT and SSC in young people with BD. Both groups underwent intensive psychological therapy and regular psychiatric review with optimisation of pharmacotherapy. Patients were generally unwell at entry into the study, with approximately 70% in an episode. Overall, the significant finding is that global cognitive function improved significantly in the overall group as did two of the domains (executive function and psychomotor function). In the 78 of 100 patients who completed cognitive testing at baseline and follow-up, the main findings were as follows:
There was a statistically significant improvement in the Global Cognitive Composite and in the domains of Psychomotor Speed and Executive Function between baseline and 18 months across the whole sample.
There was no difference between IPSRT and SSC in change in cognitive function – either in the Global Cognitive Composite, or any of the four pre-defined cognitive domains.
For the Global Cognitive Composite, and all four cognitive domains, baseline performance relative to the rest of the group correlated with change – those performing least well at baseline showed the greatest positive change in that domain over 18 months.
No other clinical variables examined related significantly to change in cognitive function.
The significant improvement in overall cognitive function (Global Cognitive Composite) and in two individual domains (Psychomotor Speed and Executive Function) in this young, relatively unwell group of patients, over the 18-month period of stabilisation is important. In young people with BD, three previous studies have found no change in cognitive function (Bombin et al., 2013; Leany, 2010; Yucel et al., 2007) over 1 or 2 years. However, lack of power is likely to have been an issue in all three studies, with between 12 and 29 patients in each. Three studies have previously shown an improvement in aspects of cognitive function in younger patients (Lee et al., 2015; Lera-Miguel et al., 2015; Torres et al., 2014). Torres et al. (2014) found improvements over 1 year in 42 first-episode patients presenting in a stable state with mild depressive symptoms. Lera-Miguel et al. (2015) also found improvements over 2 years in adolescents who were euthymic at presentation. By virtue of having recruited patients who were not necessarily stable at baseline, and who then underwent treatment to attempt to achieve mood stability, the most comparable study is that of Lee et al. (2015), which also showed improvement in aspects of cognitive function over a mean follow-up interval of 20.6 months in 61 patients with BD. In terms of domain change, in contrast to the current study, Lee et al. reported improvement in verbal learning, but not psychomotor speed (as measured by Trail Making Test–Part A). Differences in this study compared with Lee et al. include a higher mean age (26.5 years compared with 22.8 years), an earlier age of onset (15 years compared with 22 years) and a longer period between onset and inclusion in the study and cognitive testing (11 years compared with 1 year). That the patients in the current study had an earlier age of onset and a long period of illness between onset and testing may account for many of the differences. What is important is that in both studies, aspects of cognitive function improved and that in the current study global cognitive function was improved. This indicates that cognitive decline is not necessarily a feature of BD and indeed stabilisation can have broad benefits on cognition even in a relatively unwell group.
Studies suggest impairment in cognitive function prior to the onset of BD (Bora and Ozerdem, 2017b), at first presentation (Lee et al., 2014) and during the illness course even during prospectively verified euthymia (see Porter et al., 2015 for review). There is the further suggestion that episodes of illness may cause progressive decline in cognitive function (López-Jaramillo et al., 2010). All of this evidence may lead to a degree of negativity regarding the prognosis for cognitive function in patients with BD. However, a recent meta-analysis suggested that overall, longitudinal studies have not suggested progressive decline in cognitive function in BD (Bora and Ozerdem, 2017b). This study, and also that of Lee et al. (2015), suggests that cognitive performance improves with an intensive intervention early in the course of the disorder. This study specifically indicates that those with worse cognitive function can improve. This gives rise to a degree of therapeutic optimism. Neither Lee et al. (2015) nor this study specifically targeted cognitive function. Doing this could potentially result in even greater gains (Miskowiak et al., 2016a, 2017).
The finding of a correlation between baseline performance and change in cognitive function has been demonstrated previously in a number of studies (Bonnin et al., 2016; Miskowiak et al., 2016b; Yatham et al., 2017). This has led to the recommendation by the International Society of Bipolar Disorder (Ghaemi et al., 2008) cognition taskforce that screening for impairment should be used to enrich samples in clinical trials aimed at enhancing cognitive function (Miskowiak et al., 2017). In addition, the taskforce has made recommendations for screening in clinical practice, so that patients can be identified and interventions provided at an early stage to improve cognitive function (Miskowiak et al., 2018).
The analysis of factors associated with change in cognitive function over 18 months showed no significant predictors apart from baseline function in the corresponding cognitive domain. The failure of this analysis to find significant predictors of cognitive change is perhaps not surprising given the relatively low number of patients and the complexity of the clinical group, issues which we note further among the limitations of the study. Despite the lower numbers, this is one of the largest follow-up studies particularly at an early stage of the illness. A way forward in increasing ability to determine predictors of cognitive change is to pool data between studies in different centres (Burdick et al., 2019).
Originally, we hypothesised that IPSRT would have a greater effect in reducing depressive symptoms than SSC and that this reduction in symptoms would positively impact on cognitive function. In a previous study, IPSRT when compared with a psychoeducational and supportive approach (similar to that used in this study) resulted in a more rapid improvement in occupational functioning, which could have been related to improved cognitive function, although this was not measured (Frank et al., 2008). A more recent study suggested that sleep disturbance has a significant effect on cognitive function in BD (Bradley et al., 2017). Hence, we hypothesised that the more explicit focus of IPSRT on regularising circadian rhythms could have resulted in a differential effect on cognitive function. However, while the primary outcome of the RCT, burden of depressive symptoms, reduced significantly during the 18 months of treatment, this did not differ between groups (Inder et al., 2015). Therefore, it was not surprising that there was no difference between the therapies in change in overall cognitive function or on any of the cognitive domains. Lack of difference in mood outcomes has been discussed previously and probably relates primarily to the very active treatments, both psychotherapeutic and medication management, which both groups including the putative control group (SSC) received (Inder et al., 2015). IPSRT is hypothesised to have a specific beneficial effect on sleep regularity. In this case, however, there was no difference in change in the MADRS sleep item between therapies. Neither was there a correlation between change in the MADRS sleep item and change in any of the domains of cognitive function. The MADRS sleep item may, however, be relatively insensitive in this situation and of course only refers to 1 week prior to interview. A specific sleep inventory would have been a better way of measuring this factor.
A strength of the study was the assessment of depressive and manic symptoms over time, giving an assessment of overall symptom load, which might be expected to affect change in cognitive function. If symptom burden was high throughout the study, we would hypothesise that there would be little change or a worsening in cognitive function. In fact, while symptom burden declined significantly, and cognitive function improved significantly, we could show no direct correlation between the two.
Limitations
It should be noted that there is the risk of chance findings particularly given that other outcomes of the study have previously been examined. To counter this, we reduced the degree of exploratory analysis by examining the data with an overall Global Cognitive Composite measure and only four pre-defined cognitive domains. It is important to note that the domains were defined a priori and no analyses were conducted on any other possible arrangement of variables. The domain-based analysis, however, has limitations. Most of the variables in the analysis involve a number of different cognitive processes and could be alternately classified depending on the model being used. Furthermore, a hierarchical model of organisation of cognitive processes may be an alternative way of viewing domain structure with deficits in, for example, psychomotor speed, underpinning all other cognitive deficits (Antila et al., 2011). This would not be captured in our domain-based model.
Second, recruiting a relatively impaired group of patients necessarily involves testing in various mood states. This constitutes a potential weakness of the study since mood state may be a primary factor affecting cognitive function. While we have attempted to account for this by examining the effect of current mood state in our regression analysis, this type of statistical correction is imperfect. We have previously reviewed the evidence regarding the direct relationship between mood state and aspects of cognitive function. Generally, there is some evidence of a relationship between severity of depression and cognitive deficit (Porter et al., 2015). This is somewhat inconsistent but the inconsistency may occur at least, in part, because most individual studies recruit patients with a relatively small range of severities. The only attempts to examine this issue directly in BD involve the examination of the effects of low levels of depression in euthymic patients (Bourne et al., 2013). These have generally shown a relationship between depressive symptoms and only a few of many cognitive measures. We are therefore unclear regarding the likely effects of having tested in different mood states and whether we have adequately addressed this issue in our analysis. This issue could also have been mitigated to some extent by testing at more frequent intervals. However, in the context of unwell patients undergoing intensive treatment, we decided that more frequent testing would be unduly burdensome.
Third, we did not recruit a healthy control group with whom to compare performance at each time point. This means that we cannot be certain to what degree the patients were actually impaired at baseline. However, most other studies have demonstrated impairment in relatively similar groups. In the study of Lee et al. (2015), which did test a control group, patients with BD performed at a level intermediate between patients with depression and schizophrenia on most tests and less well than the control group. In a meta-analysis (Lee et al., 2012) of patients with first episode BD, deficits in all domains were found compared with control participants. We believe, therefore, that in a group of this degree of severity (50% had been hospitalised and 70% were in episode), it is highly likely that there were group mean differences from healthy controls. Regarding the percentage of patients who were likely to have been significantly impaired, subgroup analyses (Burdick et al., 2014) and classifications based on deviation from norms (Douglas et al., 2018), suggest that 40–60% would be impaired.
Fourth, a related issue is that the degree of improvement from baseline to follow-up is based on the change and the standard deviation of the patient group scores at baseline. Had a control group been tested, a more conventional way of calculating change scores would be based on the standard deviation of the control group scores at baseline, yielding an effect size change which is more readily interpreted. Furthermore, it is not absolutely clear whether improvement in cognitive function can be attributed to practice or familiarity effects. This is mitigated to some extent by the use of parallel forms of the tests and by the fact that testing sessions were 18 months apart. Furthermore, it was those patients with poorer cognitive performance at baseline who improved most and we hypothesise that these individuals were less able to form strategies with practice and to remember the structure of tests.
Fifth, medication treatment was heterogeneous between patients and also changed in many patients during the course of the study. The variables used to code medication were therefore set according to what was being taken for the majority of the 18 months. This does not, however, capture more subtle inter-individual differences. For example, there may be a very significant difference between a patient who is on lithium and stops taking this after 13 months and a patient who begins taking lithium after 4 months and is still on this at 18 months. However, any attempt to capture these differences would result in multiple variables and possibly multiple post hoc tests – analyses which are not supported by the relatively small sample size. The heterogeneity of the sample in this regard reflects the relatively unstable illness suffered in this sample, which is also early in the course of treatment. Related to this, the size of the study only allows the analysis of the effects of a limited number of variables. The course of BD over 18 months is extremely complex with many different contributing factors. This variability is difficult to capture and the study had power to analyse only a few of these. This issue is likely only to be overcome by pooling of data from multiple international studies (Burdick et al., 2019).
Sixth, the study did not have a treatment as usual group, and therefore, the improvement in cognitive function seen in both groups may not relate to the intensive treatment and may occur with treatment as usual, which often does not involve specific psychotherapy. In order to investigate this a ‘treatment as usual’ arm would have had to be added to the study. However, our experience has been that including in the randomisation an arm with no specific additional treatment has limited recruitment and therefore the generalisability of the results.
Seventh (and finally), we note that this is a group of patients who agreed to take part in a relatively lengthy RCT. This potentially limits the generalisability of the results.
Conclusion
In this young and relatively seriously unwell group of patients with BD, cognitive function improved significantly during 18 months of intensive treatment with pharmacotherapy and psychological therapy. This is in line with the only previous study examining change in cognitive function in BD at a very early stage (Lee et al., 2012). The only consistent predictor of the degree of improvement was cognitive function at baseline, with those who had worse cognitive function at baseline improving more. This represents a positive outcome of stabilisation for these patients and a potential target for further intervention.
Footnotes
Acknowledgements
The authors acknowledge the input of Andrea Bartram, Research Co-ordinator, and Robyn Abbott, Research Nurse.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: R.J.P. has received support for travel to meetings from Lundbeck and Servier and uses Software for research at no cost supplied by SBTpro.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by a grant from the Health Research Council of New Zealand.
