Abstract
Objective:
While mindfulness-based cognitive therapy (MBCT) has demonstrated efficacy in reducing depressive relapse/recurrence over 12–18 months, questions remain around effectiveness, longer-term outcomes, and suitability in combination with medication. The aim of this study was to investigate within a pragmatic study design the effectiveness of MBCT on depressive relapse/recurrence over 2 years of follow-up.
Method:
This was a prospective, multi-site, single-blind trial based in Melbourne and the regional city of Geelong, Australia. Non-depressed adults with a history of three or more episodes of depression were randomised to MBCT + depression relapse active monitoring (DRAM) (n=101) or control (DRAM alone) (n=102). Randomisation was stratified by medication (prescribed antidepressants and/or mood stabilisers: yes/no), site of usual care (primary or specialist), diagnosis (bipolar disorder: yes/no) and sex. Relapse/recurrence of major depression was assessed over 2 years using the Composite International Diagnostic Interview 2.1.
Results:
The average number of days with major depression was 65 for MBCT participants and 112 for controls, significant with repeated-measures ANOVA (F(1, 164)=4.56, p=0.03). Proportionally fewer MBCT participants relapsed in both year 1 and year 2 compared to controls (odds ratio 0.45, p<0.05). Kaplan-Meier survival analysis for time to first depressive episode was non-significant, although trends favouring the MBCT group were suggested. Subgroup analyses supported the effectiveness of MBCT for people receiving usual care in a specialist setting and for people taking antidepressant/mood stabiliser medication.
Conclusions:
This work in a pragmatic design with an active control condition supports the effectiveness of MBCT in something closer to implementation in routine practice than has been studied hitherto. As expected in this translational research design, observed effects were less strong than in some previous efficacy studies but appreciable and significant differences in outcome were detected. MBCT is most clearly demonstrated as effective for people receiving specialist care and seems to work well combined with antidepressants.
Keywords
Introduction
Many people who have had major depressive episodes (MDEs) experience multiple recurrences. Indicated tertiary prevention (Mrazek and Haggerty, 1994) for those at most risk may have a substantial impact on individuals and on population health (Patten and Meadows, 2009). To this end, mindfulness-based cognitive therapy (MBCT) (Segal et al., 2013) integrates aspects of cognitive behaviour therapy (CBT) with mindfulness training (Kabat-Zinn, 1990) in an instructional group setting. High-level evidence supports the efficacy of MBCT compared to treatment as usual (TAU) in reducing relapse/recurrence, with an equivalent outcome obtained from maintenance antidepressant medication (m-ADM) over 12–18 months of follow-up (Chiesa and Serretti, 2011; Piet and Hougaard, 2011). The United Kingdom’s National Institute of Health and Clinical Excellence (NICE) guidelines for preventing depressive recurrence include the recommendation of MBCT where people have experienced more than two prior MDEs (>2MDEs) (National Collaborating Centre for Mental Health, 2009).
There is now a need for later phase translational research work with MBCT (Lean et al., 2008; Shawyer et al., 2012; Zwarenstein et al., 2008) following existing efficacy studies with effectiveness studies and addressing specific translational research issues such as the following:
Typically, meditation experience prior to entering instructor training has been required, making a possible barrier to widespread implementation. Studies with practitioners who developed the necessary skills during training would be valuable.
Recruitment sites should include diverse settings and feature longer follow-up.
For many people with >2MDEs, long-term m-ADM is recommended (National Collaborating Centre for Mental Health, 2009); trials of MBCT to date have typically been structured to consider MBCT as an alternative to m-ADM rather than a complement, so evidence of effectiveness in the context of m-ADM is important.
Existing studies with TAU-only controls could usefully be supplemented by work with more active control conditions, reducing the risk of bias resulting from resentful demoralisation in control group participants.
The indication of MBCT for >2MDE episodes was based on subgroup analyses from early studies; planned subgroup analyses from large-scale trials could further the understanding of who might benefit most from the treatment.
Inclusion of people who have had >2MDEs, even if their primary affective disorder was other than major depressive disorder (MDD), would further contribute to the understanding of effectiveness.
This large-scale, multi-site, single-blind, randomised controlled trial aimed to examine the effectiveness of MBCT in varied settings, delivered by practitioners trained in a readily implementable model and provided to people who have >2MDEs regardless of affective diagnosis and antidepressant regimen over a 2-year follow-up period. All participants received some active treatment or support. The primary hypothesis was that MBCT delivered within a pragmatic design study could reduce the experience of MDEs compared to controls. Secondary hypotheses included that MBCT would be more effective in people taking medication. This first results paper reports on primary outcome measures (days in MDEs, proportion of relapse/recurrence, and time to relapse/recurrence) and key subgroup analyses (site of usual care and medication status).
Method
Design and settings
The Depression Awareness Recovery Effectiveness (DARE) study protocol, detailed elsewhere (Shawyer et al., 2012), involved recruitment from urban and regional contexts in Victoria, Australia (Melbourne and Geelong). Recruitment areas varied in socio-economic status (SES); the largest site, Greater Dandenong, has Victoria’s highest level of socio-economic disadvantage (Australian Bureau of Statistics, 2013).
The study, conducted in compliance with the Helsinki Declaration, was approved by the Alfred Hospital, Barwon Health, Monash University, Peninsula Health, Monash Health, and The Melbourne Clinic ethics committees. Written informed consent was obtained from all participants.
Recruitment and participants
Recruitment, between May 2007 and January 2009, was through private and public mental health services and local media notices. Eligibility criteria were assessed sequentially: (1) a referral form for treating clinicians included key selection criteria; (2) structured telephone screening used selected depression questions from the Composite International Diagnostic Interview 2.1 − Lifetime version (CIDI 2.1 LT) (Andrews and Peters, 1998; Wittchen, 1994; Wittchen and Essau, 1993); and (3) an intake interview used sections of the CIDI 2.1 LT.
Enrolled participants met DSM-IV criteria for >2MDEs with a Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) diagnosis of MDD (recurrent) or bipolar disorder (BD) I or II (assessed from 1 to 3 above), were aged between 18 and 75 years (1–3), and able to speak and read English fluently (1–3). Diagnostic exclusions included current MDE (1–3); current symptoms of a psychotic disorder or a past diagnosis of a psychotic disorder where the treating clinician believes the therapy may be contraindicated (1); organic mental disorder or pervasive developmental delay (1); current eating disorder or obsessive-compulsive disorder (1, 3); current borderline or antisocial personality disorder (1); current alcohol or drug dependency other than tobacco (1, 3); current benzodiazepine intake of more than 20 mg diazepam equivalent (1, 3); and inability to give informed consent (3).
Treatment conditions
All participants received a supported self-monitoring intervention: depression relapse active monitoring (DRAM). Half were randomised to receive DRAM alone (control group) while half received MBCT + DRAM (MBCT group). Control participants were offered MBCT following conclusion of data collection. Participants continued usual treatment during the trial.
MBCT
After an initial individual orientation session, the MBCT program was delivered by an instructor in eight weekly 2-hour group training sessions involving up to 10 participants. As per previous trials, four sessions was considered the minimal treatment dose. Sessions incorporated mindfulness practices and CBT-based exercises (Segal et al., 2002b). Homework included formal daily meditation practices and exercises for the development of everyday mindful awareness. In 3-monthly ‘booster sessions’, optional for MBCT participants, an experienced MBCT practitioner led mindfulness practices over a 5-hour period.
MBCT sessions were video-recorded with the camera field including the instructor only. Supervision of instructors with review of the recording occurred weekly for the first half of the course, then every 1–2 weeks as needed. Treatment fidelity was assessed for 25% of sessions: one early (sessions 1–4) and one late (sessions 4–8) recorded session was randomly selected from each of the 11 MBCT groups by a researcher external to the project. Fidelity was rated using the Mindfulness-Based Cognitive Therapy Adherence Scale (MBCT-AS; Segal et al., 2002a). The MBCT-AS has 17 items assessing instructor behaviours specific to MBCT, including practices shared with CBT (Segal et al., 2002a). Item scores range from 0 to 2 (0: no evidence for item; 1: slight evidence; 2: definite evidence). The scale has been found to be reliable in distinguishing MBCT from pure CBT (Segal et al., 2002a). Fidelity ratings were undertaken by an independent clinical psychologist with training and experience in MBCT.
MBCT instructor training
GM trained in MBCT with ZS, then led development of the DARE MBCT instructor training program with advice from ZS (Shawyer et al., 2012). Participation in a slightly modified MBCT group preceded a 4-day residential course with intensive training and coaching in MBCT theory and technique. For entrants unfamiliar with mindfulness practices before entry to training, instructor training supported development of a personal mindfulness practice. Following the formal program, instructors engaged in supervised practice either by shared delivery of MBCT with a more experienced instructor and/or with supervision based on review of video-recorded sessions. By the time of the interventions for DARE, all instructors had had a regular mindfulness practice for at least 1 year but not necessarily much longer than this. GM (a psychiatrist) was an instructor and supervised the others (five psychologists and a social worker). Well after this study was designed, good practice guidelines have been published in the UK (UK Mindfulness-Based Teacher Trainer Network, 2011). Mostly, study practitioners would have met these criteria, though often only by recent attainment and in one case not fully. Generally, the level of training of study practitioners is in alignment with what might be achieved pragmatically in the development of an MBCT program after about 2 years.
Depression Relapse Active Monitoring (DRAM)
DRAM was designed as an alternative to TAU-only control with considerations including seeking to equalise treatment expectancy across treatment conditions and attenuate the risk of resentful demoralisation in participants allocated to the control group. DRAM comprised training on self-management of depression and supported monthly self-monitoring using the Patient Health Questionnaire-2 (PHQ-2; Kroenke et al., 2003) and PHQ-9 (Kroenke et al., 2001) (for further details see Shawyer et al., 2012).
Randomisation
Enrolled participants were randomised independently by a statistician. To allow for the group-based nature of MBCT, a minimisation routine (Evans et al., 2001) balanced intervention allocations within 11 cohorts of 18–21 participants. To guard against confounding, and allow testing of secondary hypotheses, randomisation was stratified by medication (currently taking antidepressants and/or mood stabilisers: yes/no), site of usual care (primary care or specialist clinic), diagnosis (BD: yes/no) and sex.
Outcome measures
Relapse/recurrence of MDD
The primary outcome variables of days in MDEs, proportion of relapse/recurrence, and time to relapse/recurrence were assessed across each cohort 14 and 26 months following commencement of MBCT in that cohort. The CIDI 2.1 12-month version depression module was used to assess relapse/recurrence, with questions from the World Health Organization (WHO) CIDI added to collect the month that the first/only MDE commenced and the total days of MDEs over the previous 12 months. Time to relapse/recurrence was measured in months from the start of therapy.
PHQ data (Shawyer et al., 2012) is not reported due to a high missing-data rate – less than half the sample having complete data.
Patient expectations
The Credibility/Expectancy Questionnaire (CEQ; Devilly and Borkovec, 2000) is a well-validated scale assessing therapy credibility and client expectancy for improvement. CEQ instructions were modified to be appropriate for participants in either treatment condition. On the advice of the author, scoring was standardised across items. The CEQ was administered to all participants at the fourth and eighth week of MBCT for their cohort.
Intake and follow-up interviews were conducted by research assistants with relevant health science qualifications who had received additional training on research interviewing including the CIDI. Considerable efforts were made to ensure that rater blindness was maintained at follow-up (Shawyer et al., 2012).
Sample size
Our power calculation for n=204 (Shawyer et al., 2012) assumed: recurrence rates in the treated group of 22% and the control group of 42%; dropout rate of 20% over 2 years; power of 0.8 and α of 0.05. Where stratification and interaction tests support subgroup analyses for a Type I error, positive findings will be reported; we will not seek to draw inferences from negative subgroup statistical testing as power is relatively low.
Statistical analysis
Previous studies of MBCT have used survival analysis and proportion relapse/recurrence as key dependent variables and for comparability we replicated these analyses. However, where MDEs for participants in different groups may have differing numbers, severity and duration, relying solely on these dependent variables may be misleading – so in the study protocol we described a potentially more sensitive primary outcome measure as days in MDEs.
Intention to treat (ITT) analysis was the primary analytic framework with per protocol (PP) analyses also undertaken for those attending four or more sessions. Preliminary analyses were conducted to examine if stratifying variables included in randomisation moderated treatment outcomes; subgroup analyses were conducted where there were positive interaction tests. Alpha was set at p<0.05 for analyses of main effects and p<0.10 for significant treatment-subgroup interactions (Lu et al., 2005).
A repeated-measures analysis of variance (ANOVA) was used to examine the dependent variable of days in MDEs (transformed) and repeated-measures analysis of covariance (ANCOVA) used to test whether treatment effects differed between subgroups (treatment–subgroup interaction test).
Chi-squared tests were used to examine the dependent variable of proportion relapse/recurrence with logistic regression used to test for treatment–subgroup interactions. Kaplan–Meier survival analysis was used to examine the dependent variable of time to relapse/recurrence of depression, and Cox proportional hazards regression used to test for treatment–subgroup interactions. Individuals who experienced no relapse or recurrence were considered as censored for the survival analyses.
Missing data were handled using casewise deletion based on availability of complete data for individual variables. In consideration of the possibility of data missing not at random (MNAR) rather than missing at random (MAR) (Héraud-Bousquet et al., 2012), we followed recommendations to examine the robustness of results to the possibility of data MNAR (Carpenter et al., 2007). Therefore, we performed sensitivity analyses for key positive findings for which, where data were missing, data with a poorer differential treatment outcome were inserted.
Results
Participants
Figure 1 shows participant flow through the trial. Of 482 initial inquires and referrals, 380 undertook telephone screening and 326 met preliminary screening criteria. Of these, 250 proceeded to the intake assessment providing written informed consent. There were 45 excluded candidates, with reasons as follows: no diagnosis of MDD or BD (n=7), not having >2MDEs (n=27), current MDE (n=3), presence of other psychiatric disorder such as eating disorder, OCD or alcohol dependency (n=8). One individual declined participation, having undertaken similar therapy to MBCT with his psychologist. Of 204 participants proceeding to randomisation, one individual randomised to MBCT was withdrawn as further assessment indicated she was ineligible (current MDE). The final sample size was therefore 203 (101 in the MBCT group, 102 in the control group).
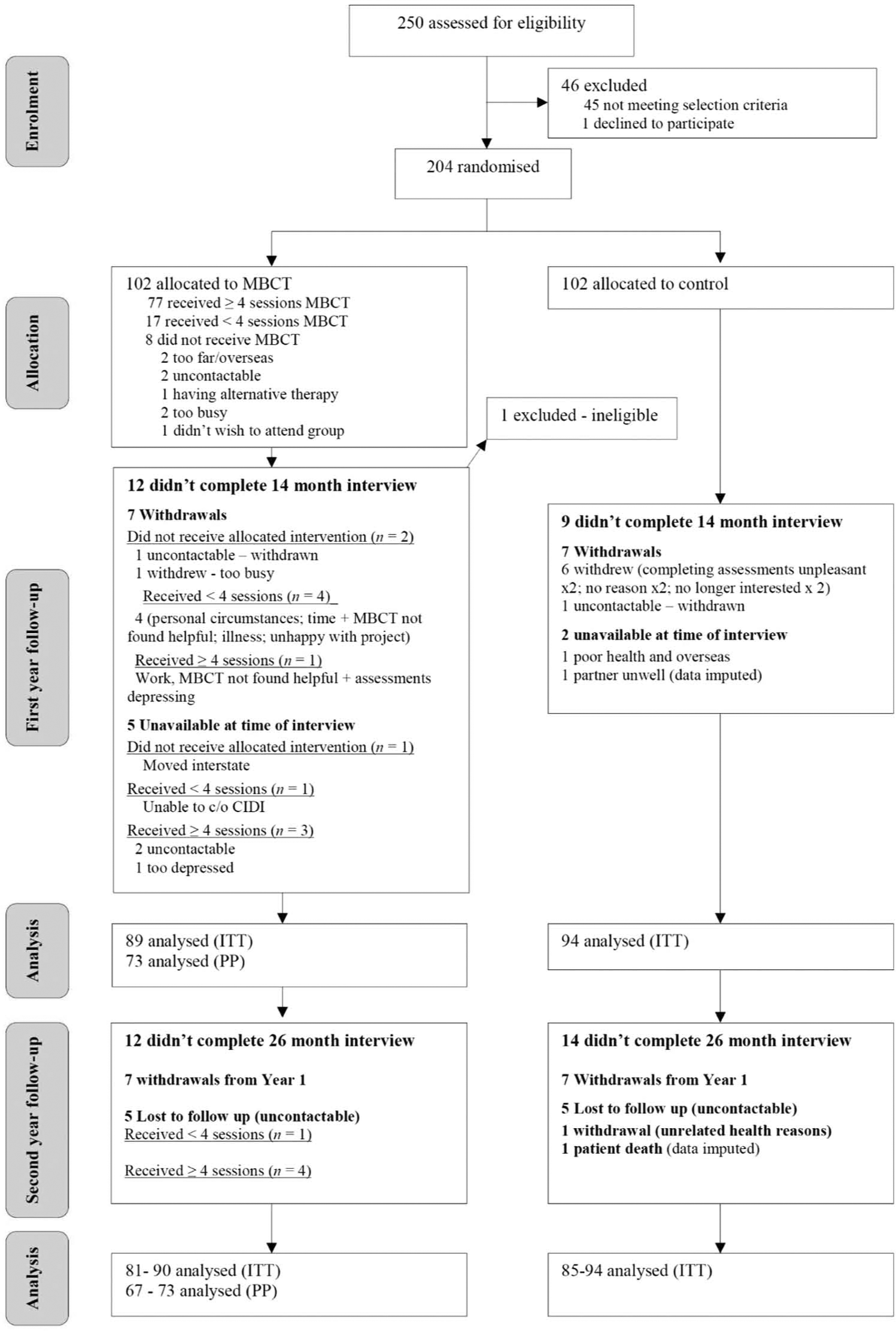
Study design and flow of participants through trial.
Baseline characteristics for MBCT and control groups are provided in Table 1. The sample was predominantly females of middle years with long histories of recurrent MDD. Twenty-one enrolled participants had BD and more than two prior MDEs, all BD I. There were no statistical differences on baseline clinical or demographic characteristics between the 25 participants who attended fewer than four MBCT sessions and the 77 participants who attended four or more sessions.
Demographic and clinical characteristics of MBCT and control groups at baseline.
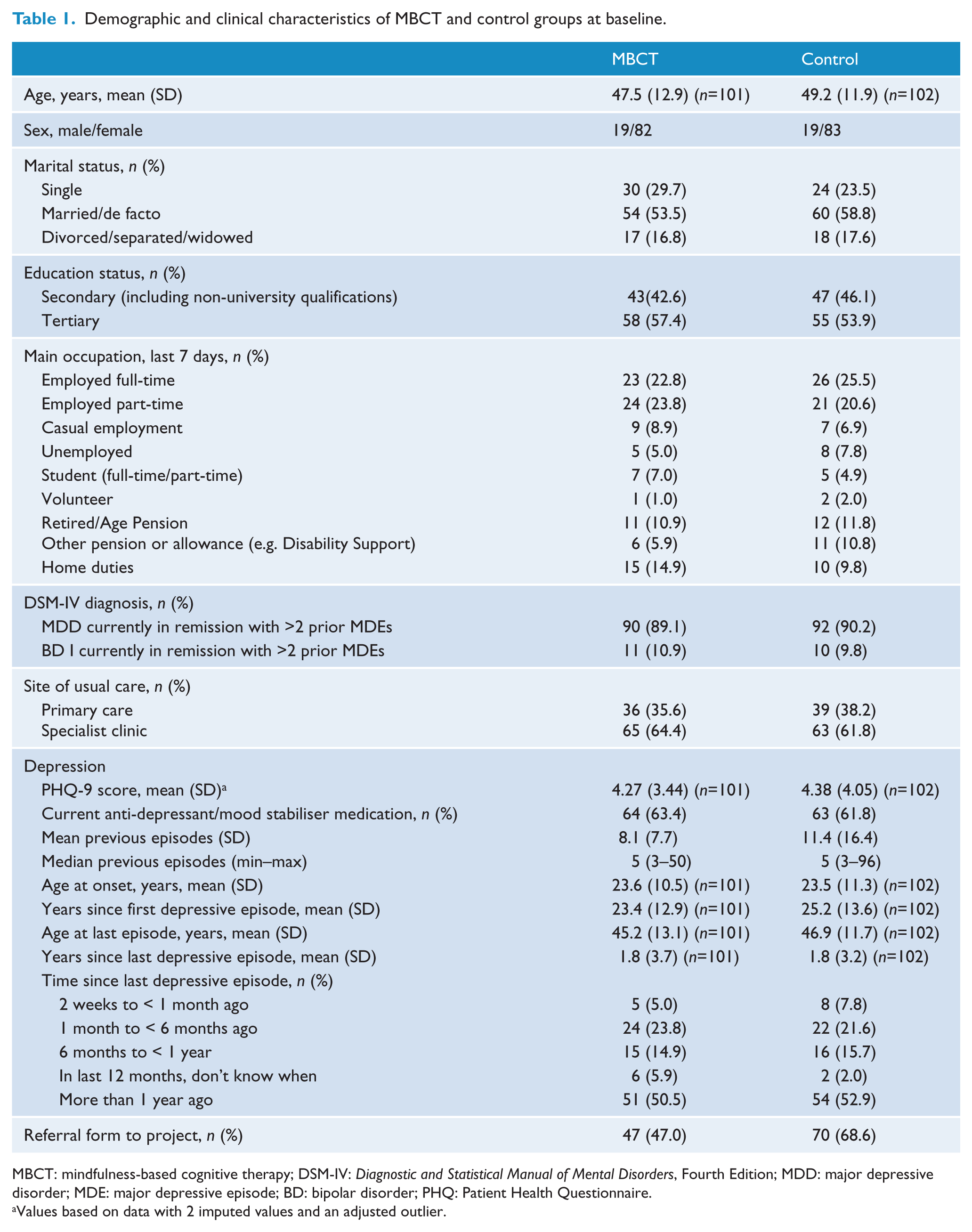
MBCT: mindfulness-based cognitive therapy; DSM-IV: Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition; MDD: major depressive disorder; MDE: major depressive episode; BD: bipolar disorder; PHQ: Patient Health Questionnaire.
Values based on data with 2 imputed values and an adjusted outlier.
Data collection quality
Reliability
Reliability checking of CIDI administration relied on three video-recorded role-played interviews rated by assessors. Rating of all three interviews by 20 of the 24 assessors during the follow-up period showed 100% agreement on current MDE and one discrepancy for recency of last episode.
Blindness
Rater-blindness to intervention was maintained for 94% of assessment interviews; raters’ selection of the treatment condition was not above statistical expectation based on chance.
Treatment expectation
MBCT and control participants were compared on treatment credibility and expectation at 4 weeks using t-tests. At 36.2 (SD: 8.2, n=87), the mean CEQ score in the MBCT group was not significantly higher than that of the control group at 33.5 (SD: 12.3, n=94), t(163.1) = 1.73; Cohen’s d=0.22 (small effect size), p=0.09. The baseline CEQ score and treatment–subgroup interactions had no influence on key dependent variables (e.g. days in MDE interactions on ITT and PP repeated-measures ANCOVAs – ITT: F(2, 148) = 1.38, p=0.26; PP: F(2, 138) = 1.16, p=0.32). Given that treatment expectation was not significantly different, we did not include this as a covariate in the main analyses.
MBCT adherence
The mean fidelity rating for the 22 MBCT sessions rated using the MBCT-AS was 1.84 (SD: 0.17), indicating very good treatment adherence, comparable to that reported by Segal et al. (2010).
Preliminary subgroup analyses
Preliminary tests showed no evidence that treatment effects were moderated by diagnostic and sex stratification variables for any of the outcome variables, so these were not included in further analyses.
Outcome analyses
Days in MDEs
Table 2 shows the results regarding days in MDEs. Over the 2-year follow-up period, the MBCT group experienced an average of 47 fewer days in MDEs than the control group (65 vs 112 days) for the ITT analysis and 54 fewer days for the PP analysis (58 vs 112 days). Repeated-measures ANOVAs, using days in MDE as log transformation to improve normality of distribution, showed that both these differences were significant at p<0.05 (p=0.03, 0.02).
Days in major depressive episode(s) for years 1 and 2 for MBCT and control groups (intention to treat and per protocol) with per-protocol medication subgroup analyses.
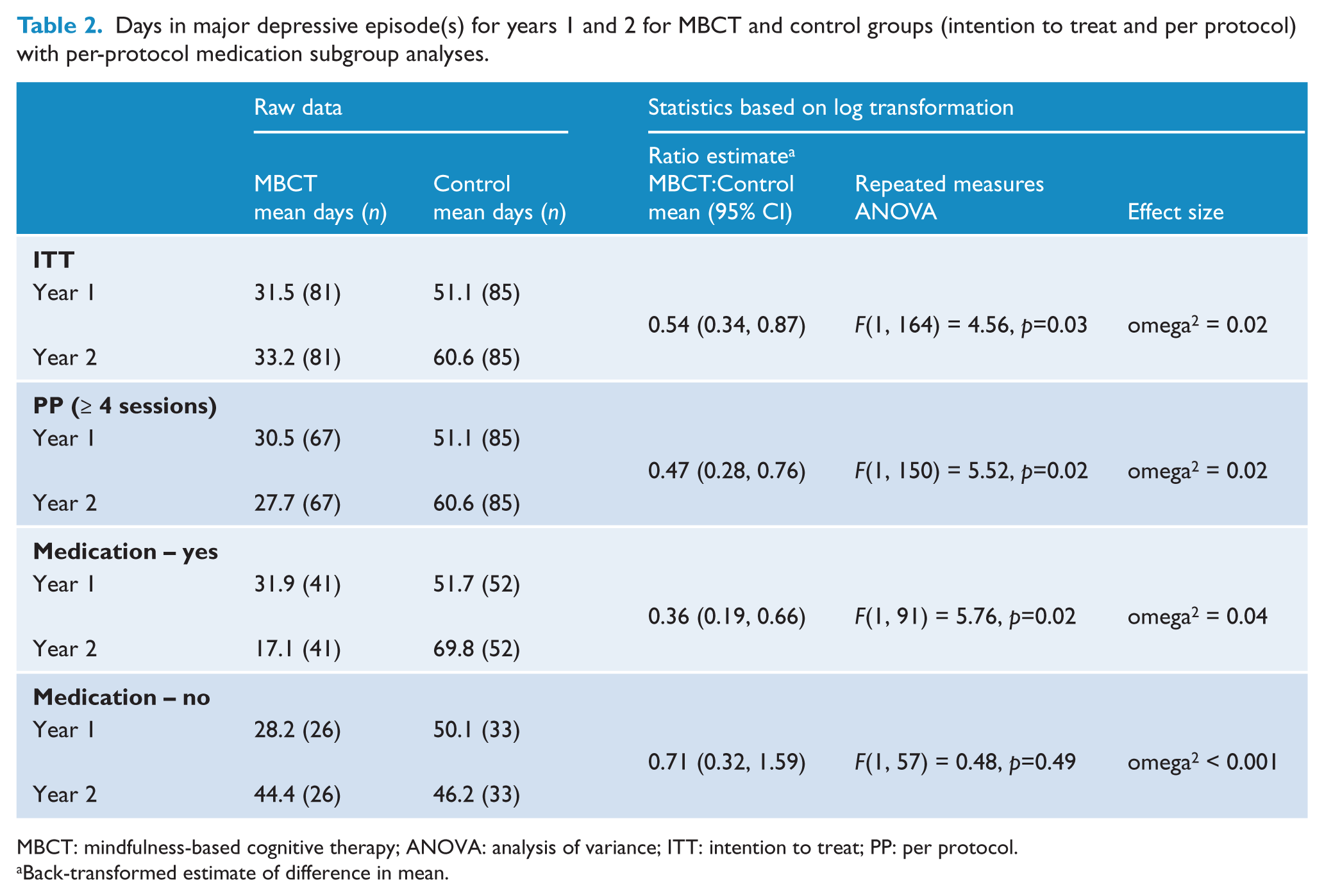
MBCT: mindfulness-based cognitive therapy; ANOVA: analysis of variance; ITT: intention to treat; PP: per protocol.
Back-transformed estimate of difference in mean.
For site of usual care, the treatment–subgroup interaction did not reach significance on both the ITT and PP repeated-measures ANCOVAs and so we did not proceed with subgroup analyses for this variable. For medication, the treatment–subgroup interaction did not reach significance on the ITT repeated-measures ANCOVA, F(2, 163) = 2.14, p=0.12. For the PP analysis, the three-way interaction reached significance, F(1, 148) = 2.91, p=0.09. Separate repeated measures ANOVAs conducted for those on or not on medication showed that treatment group differences over the 2 years were evident only for those taking medication (see Table 2). For this subgroup, the MBCT group experienced an average of 73 fewer days in MDEs (49 vs 122) than the control group over the 2-year follow-up period.
Relapse/recurrence: odds ratios (ORs) and number needed to treat (NNT)
As shown in Table 3, ITT analyses show odds ratios favouring the MBCT group in year 1 and year 2, though this is of borderline statistical significance. Findings regarding relapses in both year 1 and year 2 were more strongly favourable and significant. PP analyses were generally more favourable again.
Proportion relapsing, number needed to treat and odds ratios over the first, second and 2-year follow-up periods for MBCT and control groups (intention to treat and per protocol).
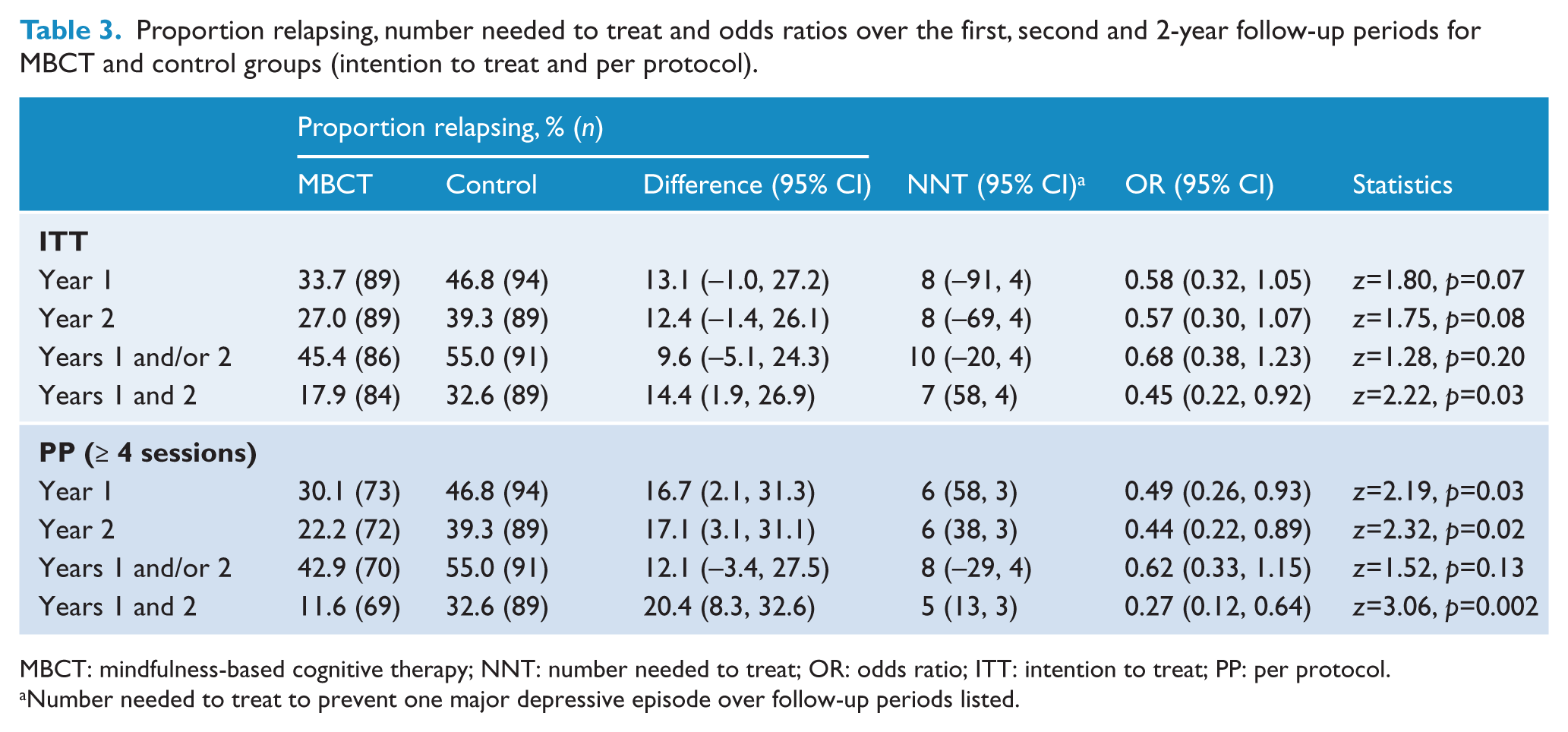
MBCT: mindfulness-based cognitive therapy; NNT: number needed to treat; OR: odds ratio; ITT: intention to treat; PP: per protocol.
Number needed to treat to prevent one major depressive episode over follow-up periods listed.
Results of a logistic regression analysis supported the presence of a site of usual care–treatment group interaction for relapse/recurrence over years 1 and/or 2 in the ITT sample, OR = 0.33 (95% CI: 0.10, 1.13), p=0.08, the PP ≥ 4 sessions sample, OR = 0.30 (95% CI: 0.08, 1.12), p=0.07, and for medication for the PP sample, OR = 0.30 (95% CI: 0.08, 1.09), p=0.07 but not for medication for the ITT sample, OR = 0.48 (95% CI: 0.14, 1.64), p=0.24. Table 4 shows the ITT and PP subgroup results for site of usual care and the PP results for medication. In the context of specialist care, there was a 20% absolute risk reduction (ARR) in relapse/recurrence rates in the MBCT group (41% vs 61%), greater with PP analyses (38% vs 61%), and for PP analyses for people also taking medication an ARR of 24% (33% vs 57%). Hence, and noting the context of limited power for subgroup analyses, there was no definite evidence of benefit from MBCT for participants in primary care while those from specialist care showed a significant reduction in relapse/recurrence rates. Those on medication at baseline and who attended at least four sessions of MBCT showed a significantly greater reduction in relapse/recurrence compared to controls.
Proportion relapsing, number needed to treat and odds ratios over the 2-year follow-up period for MBCT and control groups by site of usual care (intention to treat and per protocol) and medication subgroups (per protocol).
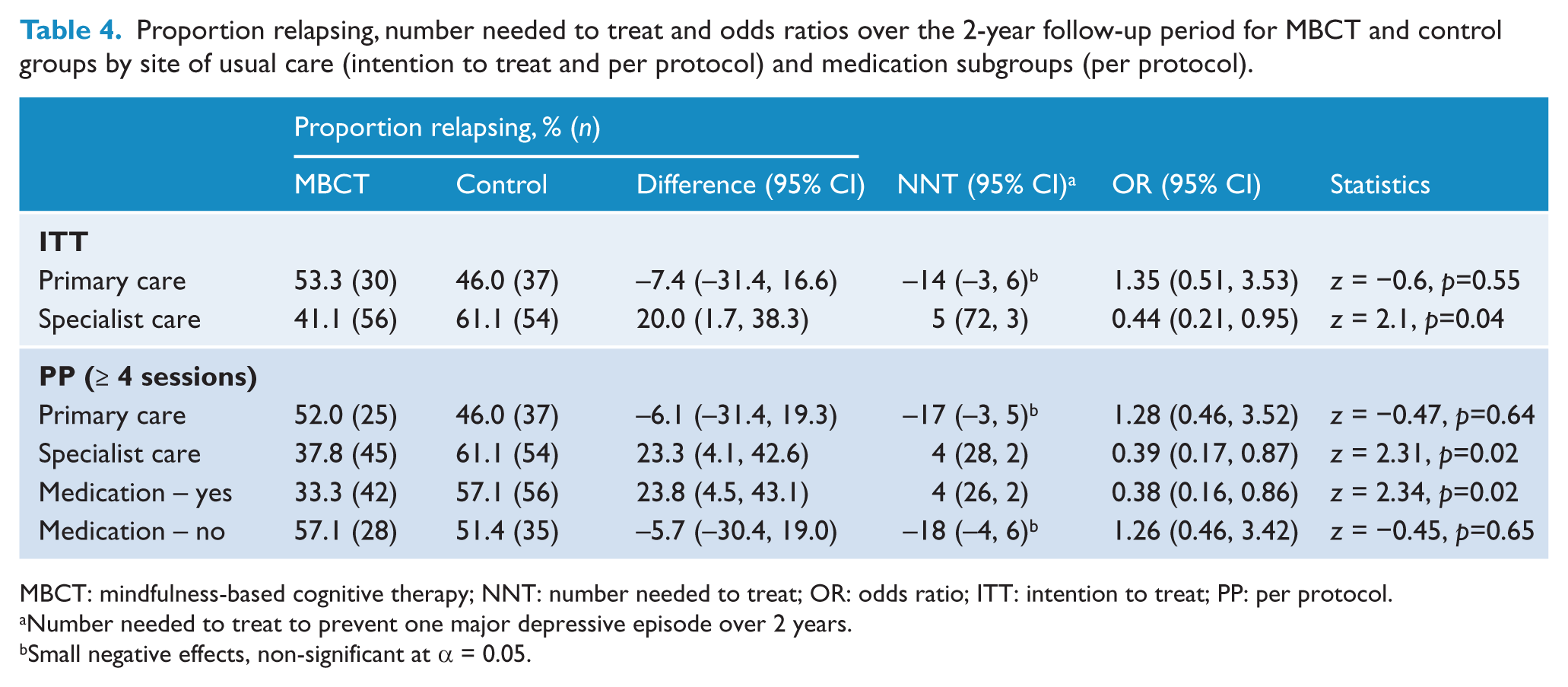
MBCT: mindfulness-based cognitive therapy; NNT: number needed to treat; OR: odds ratio; ITT: intention to treat; PP: per protocol.
Number needed to treat to prevent one major depressive episode over 2 years.
Small negative effects, non-significant at α = 0.05.
Time to relapse/recurrence
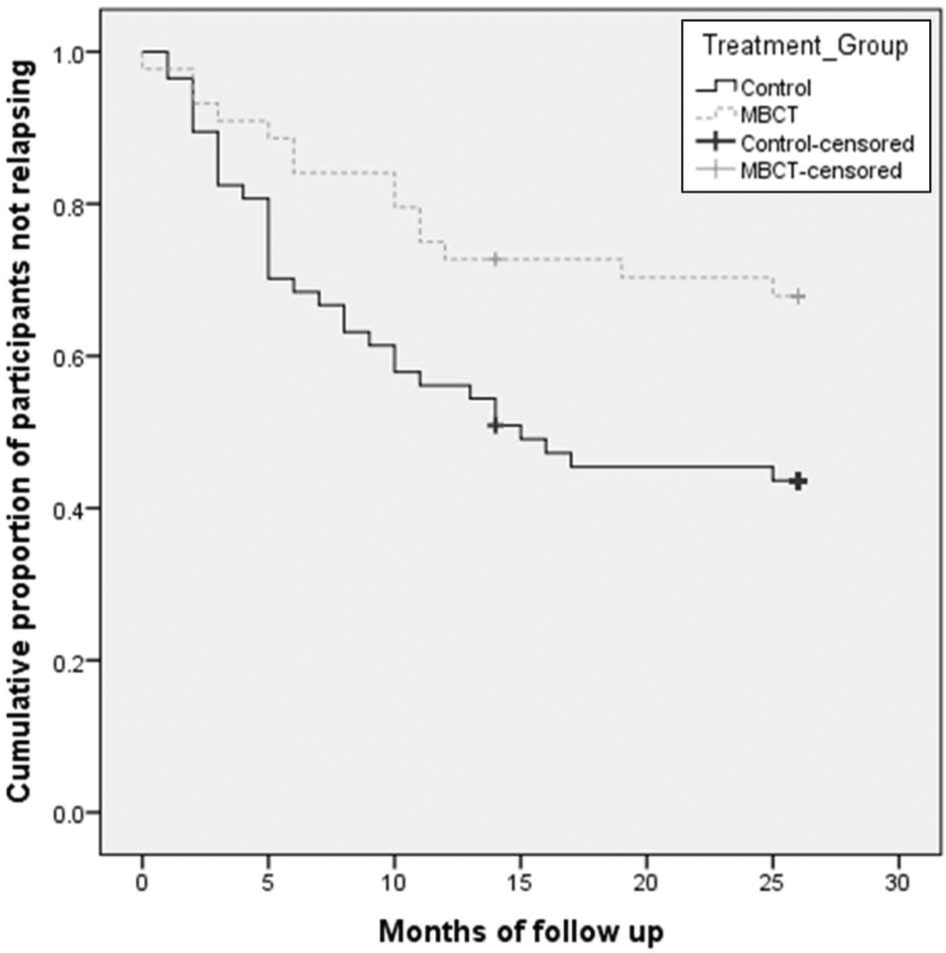
Based on data collected at the 14- and 26-month follow-up CIDI interviews, time to relapse or recurrence (in months) over the 26-month follow-up period was compared between MBCT and control groups using Kaplan–Meier survival analysis. The DRAM PHQ protocol (Shawyer et al., 2012) was used to provide data for survival analyses when needed if the CIDI was positive for MDE but duration data were missing or invalid (n=6) and in one case of apparent suicide in the control group. The results of these analyses for both ITT and PP are presented in Figure 2 and Table 5 and show a non-significant trend favouring the MBCT group. This borderline effect weakens over time, as indicated by the stronger results on the Breslow test, which gives greater weight to early relapses/recurrences compared to the log-rank test, which weights data equally over time.
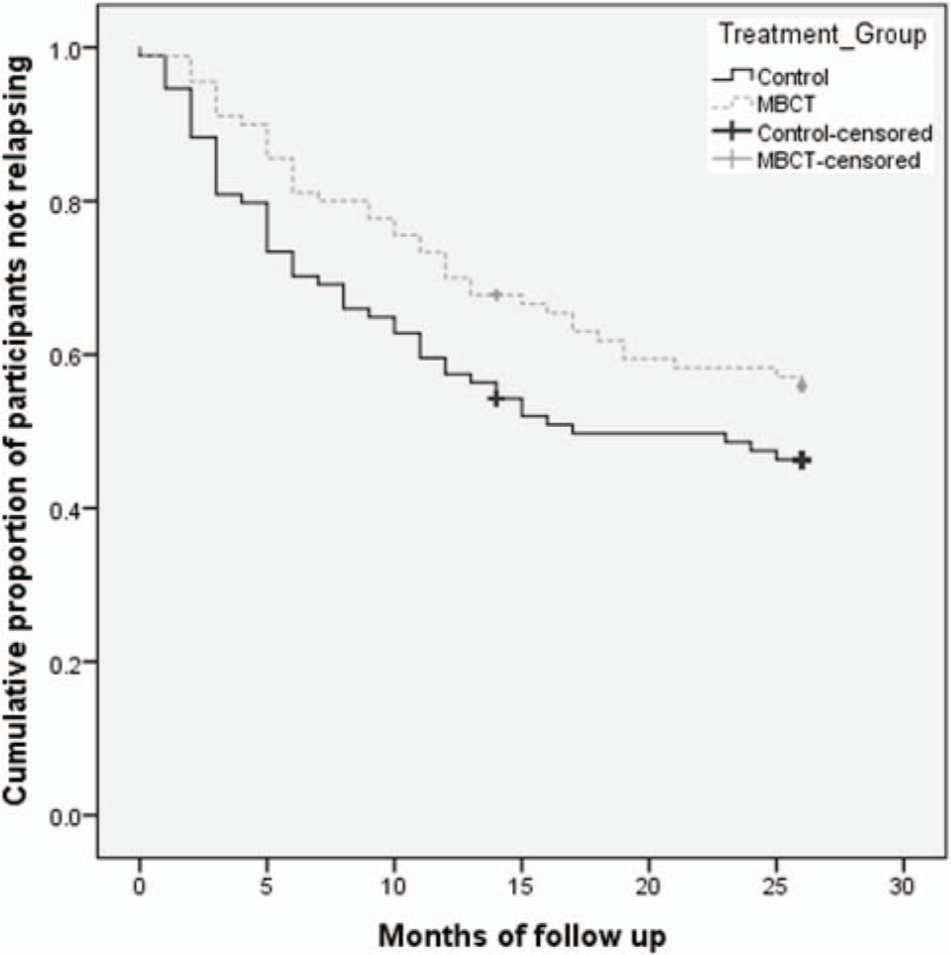
Survival (non-relapse/recurrence) curves comparing relapse/recurrence to major depression for MBCT and control groups over a 26-month follow-up period (intention to treat).
Mean time (months) to relapse/recurrence of depression for MBCT and control groups over 2-year follow-up (intention to treat and per protocol) and subgroup analyses.
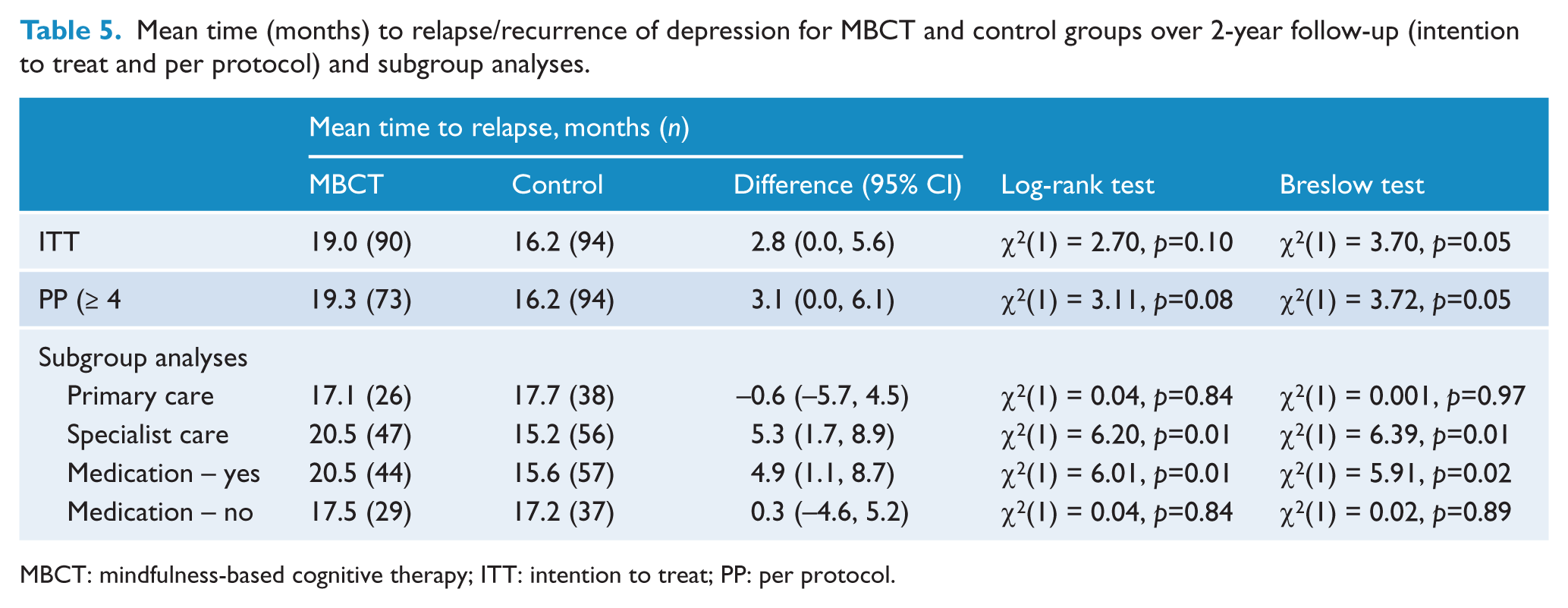
MBCT: mindfulness-based cognitive therapy; ITT: intention to treat; PP: per protocol.
To examine whether the effect on treatment group was moderated by stratifying the variables medication and site of usual care, a Cox regression was conducted including, separately, each of these variables and its interaction with the treatment group together with treatment group. For the ITT analyses, treatment group did not interact significantly with either site of usual care (Wald(1) = 2.55, p=0.11, OR: 0.49 (95% CI: 0.21, 1.17)) or medication (Wald(1) = 1.34, p=0.25, OR: 0.60 (95% CI: 0.26, 1.42)). For the PP analyses, treatment group did interact significantly with site of usual care (Wald(1) = 2.82, p=0.09, OR: 0.45 (95% CI: 0.18, 1.14)) and medication (Wald(1) = 3.12, p=0.08, OR: 0.44 (95% CI: 0.17, 1.10)), here supporting further subgroup analyses.
Separate PP Kaplan–Meier survival analyses were conducted for primary and specialist care data and medication yes/no data. As shown in Figures 3 and 4 and Table 5, participants in the MBCT group who were in specialist care or who were on medication had a significantly longer time to relapse/recurrence compared to controls. There were no significant group differences for PP participants in primary care and for those not taking medication.
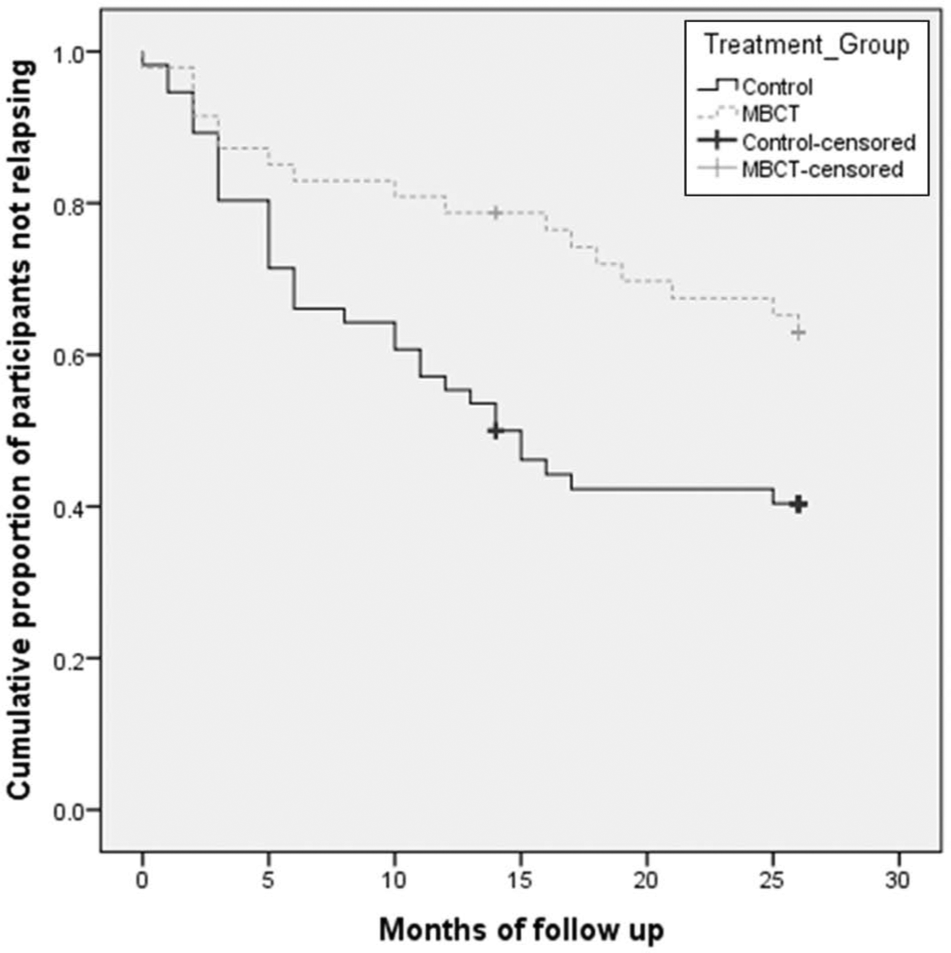
Survival (non-relapse/recurrence) curves comparing relapse/recurrence to major depression for MBCT and control groups in specialist care over a 26-month follow-up period (per-protocol sample ≥ 4 sessions).
Survival (non-relapse/recurrence) curves comparing relapse/recurrence to major depression for MBCT and control groups for participants on antidepressants at baseline over a 26-month follow-up period (per-protocol sample ≥ 4 sessions).
Sensitivity analyses and consideration of study design effect for key positive findings
Missing data were unrelated to treatment condition in either year 1 or year 2. Sensitivity analyses focused on key positive findings; scenarios chosen assumed no treatment effect in missing data and relapse properties typical of the control group (i.e. all missing values were replaced with a 50% relapse rate and 50 days in MDE per year). Given this scenario, analyses for proportion relapse all still showed significance (p<0.05). For days in MDEs, while ITT sensitivity analysis still showed a ratio difference of 0.63 (95% CI: 0.33, 1.20), this was no longer significant; per protocol analyses all remained significant.
Finally, subgroup analyses for the key positive findings showed no evidence that treatment effects were influenced by site of recruitment or cohort.
Discussion
Key findings
MBCT delivered within this pragmatic design was associated with reduced time in MDEs as measured here compared to the active comparison group. Fewer people receiving MBCT had relapses across both years of follow-up. A trend favouring the MBCT group for increased time to relapse was evident although non-significant at p<0.05. Subgroup analyses provided support for effectiveness of MBCT for people taking antidepressant medication and for people receiving usual care in a specialist setting. The subgroup analysis for primary care was not adequately powered as a base for firm conclusions but findings were less encouraging there.
Comparisons with previous efficacy studies
Relapse/recurrence rates in the MBCT group for this study were typical of previous efficacy studies. Meta-analysis has found this to be 32% (Chiesa and Serretti, 2011); the 1-year relapse/recurrence rate in this study was 33.7%. In this study, 1-year relapse/recurrence in the control group (47%) was lower than in much previous work where control relapse/recurrence rates have ranged between 60% and 78% (Godfrin and van Heeringen, 2010; Kuyken et al., 2008; Ma and Teasdale, 2004; Segal et al., 2010; Teasdale et al., 2000). One Swiss study reporting low recurrence rate in controls was published by Bondolfi and colleagues (2010) who proposed as an explanation the high availability of mental health care in Switzerland; a treatment signal for MBCT is more difficult to detect against such a background. Might this be the case here? Clinicians within the Australian mental health system may be acutely aware of the limitations of the system in Australia. However there is evidence that mental health treatment in primary care has advanced over the past decade along with initiatives including Better Access and Better Outcomes and in Victoria, the setting for the current study, Primary Mental Health Teams. In this context there are signs of possible narrowing of the treatment gap (Meadows and Burgess, 2009) and improvements in quality of care delivered (Meadows and Bobevski, 2011).
We also should consider the possible effect of DRAM. This intervention was constructed towards relative equalising of treatment expectation, something reasonably well achieved with only a small and non-significant effect size between the two treatment conditions. Based on principles of chronic disease management (Bodenheimer et al., 2002), DRAM could be expected to have some potency in decreasing relapse/recurrence rates and duration over an unmodified TAU control (Ludman et al., 2003). This design, considered compared to a TAU comparison (Bondolfi et al., 2010; Godfrin and van Heeringen, 2010; Orsillo et al., 2004; Teasdale et al., 2000), could increase the risk of Type II error but should not be a source of Type I error.
In the context of this effectiveness/translational research design (Shawyer et al., 2012), an appreciable proportion of people did not complete the MBCT course. In Australia, as elsewhere, affective disorders have been demonstrated to be more prevalent within low SES regions (Hudson, 2005; Meadows et al., 2002). However, people in low SES regions are less likely to participate in research (Furler et al., 2012; Watt, 2002) possibly limiting generalisability of trial outcomes for such areas. This trial included low SES regions to recruit within and deliver MBCT programs. Higher attrition is expected in low SES groups due at least in part to disadvantage-related social and financial stress (Mein et al., 2012). In this study setting and design we might reasonably expect higher attrition than in a pure efficacy study but it suggests that adherence may be a challenge to be addressed in routine delivery of MBCT.
Subgroups
Findings for the PP analyses as more favourable compared with ITT are not surprising. In the MBCT course, each session has a carefully sequenced place in providing opportunities to develop protective skills. This study would support clinical guidance for people considering MBCT to consider their motivation and to be clear that it is possible to attend all sessions. It could be of value to adapt the therapy better to cope with the situation where people have to miss one or two sessions. While group effects may well be important for MBCT, it could usefully be explored whether individual delivery might achieve comparable results.
The positive finding for people receiving specialist care is not an issue examined before now. Here the NNT of 5 from the ITT analysis (Table 4) suggests MBCT is an effective treatment in this setting (Citrome and Ketter, 2013). The subgroup comparison reported here as elsewhere in this paper is justified both on the fact that this was an initial stratification variable and based on tests of interaction. While MBCT has major components that are orientated towards mindfulness-based responses to rumination, it also includes a strong element of a relapse-signature and relapse-drill. It is possible that this is a significant part of the action of the approach in this setting, and that relapse-drills activated in the context of specialist care are more potent by being able to include contributions of more skilled providers. Alternative explanations might include a difference in assertiveness and motivation to participate in treatment in general on the part of the patients, these being perhaps typically greater among people who have negotiated their way into specialist care. These issues could be examined in future research.
This is also the first study to demonstrate an apparent adjunctive role of MBCT along with m-ADM, using a naturalistic follow-up. Most other studies have instead examined MBCT as a putative alternative to m-ADM (Bondolfi et al., 2010; Kuyken et al., 2008; Orsillo et al., 2004; Teasdale et al., 2000). From the point of view of prescribing psychiatrists, patients generally regarded as best eligible for MBCT, that is people who have at least had three prior episodes of major depression, are also commonly those for whom m-ADM is recommended by many guidelines. Selecting people with at least three prior episodes of depression, then ceasing medication while offering MBCT as an alternative may not be something that psychiatrists wishing to abide by guidelines would advocate, indeed such an approach may be seen as unsafe. This study offers some reassurance that MBCT can safely and effectively be administered in combination with m-ADM. At very least, m-ADM is certainly not an contra-indication to MBCT where this can otherwise be considered, and this study would align well with the existing NICE guidelines suggesting that MBCT be considered for people with at least three prior episodes of depression (National Collaborating Centre for Mental Health, 2009). The NICE prescribing guidelines would also suggest long term antidepressant prescribing (National Collaborating Centre for Mental Health, 2009). Based on this study’s findings, these two interventions appear to be well compatible.
Limitations
Assessments of MDEs were based on fully-structured diagnostic interviews which may be less accurate than clinician-administered diagnostic interviews (Kessler et al., 2008) and assessment of the depressive outcome variables was based on retrospective reporting over the previous 12 months. However, while possibly less precise than some alternatives, this measurement approach is not necessarily a source of systematic bias to the findings. We note that while PP analyses are seen as useful within relevant CONSORT guidelines (Zwarenstein et al., 2008), as noted by Hollis and Campbell, (1999) due caution must be taken when evaluating subgroups defined by participant responses post randomisation.
Conclusion
These findings add to evidence supporting the effectiveness of MBCT. In this study, MBCT effects on some variables were not as large as in some previous effectiveness studies (Chiesa and Serretti, 2011; Piet and Hougaard, 2011), although not all (Bondolfi et al., 2010). The combination of a more active control condition than in much previous work with the pragmatic setting where effect sizes will likely be smaller than in efficacy studies (Patsopoulos, 2011) makes this unsurprising. An important aspect of this work is that practitioners were recruited from among practising mental health clinicians and trained using a program that is not excessively demanding. More expert instructors may have achieved greater effects but this study lends weight to the proposition that MBCT is an intervention that can practically and with appreciable clinical benefit be included as an offering in the broad scale development of mental health services. The findings provide information on subgroups that appear to benefit most from MBCT. Within this study, MBCT had a more clearly evident and substantial effect in people in receipt of specialist care. Of practical clinical significance is the finding here that MBCT seems to work well in combination with antidepressant therapy. The position implied in NICE guidelines that MBCT is a desirable therapy to have broadly accessible therefore is supported, and this paper would lend weight to the importance of initiatives that can be seen to increase the supply of clinician therapists able to deliver the intervention and to promote the extent to which it is widely available. The findings also clearly provide encouragement to clinicians that co-treatment with MBCT and medication is likely to be an effective and useful option for many people with extensive histories of recurrent depression.
Footnotes
Acknowledgements
We would like to express our thanks for the support of the associate investigators and mental health services within the following organisations: Alfred Health (Professor Jayashri Kulkarni), Barwon Health (Professor Michael Berk), Peninsula Health (Associate Professor Richard Newton; Dr Sean Jesperson), Monash Health (Associate Professor Saji Damodaran) and Healthscope. Phuong-Tu Prowse conducted the MBCT-AS ratings. We also gratefully acknowledge the regular infrastructure and financial contributions from Monash Health toward this research centre. Most of all we would like to express our gratitude to our study participants who made the trial possible.
Funding
This research was supported by a grant from the National Health and Medical Research Council of Australia (grant number 436897).
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
