Abstract
Objective:
While mindfulness-based cognitive therapy is effective in reducing depressive relapse/recurrence, relatively little is known about its health economic properties. We describe the health economic properties of mindfulness-based cognitive therapy in relation to its impact on depressive relapse/recurrence over 2 years of follow-up.
Method:
Non-depressed adults with a history of three or more major depressive episodes were randomised to mindfulness-based cognitive therapy + depressive relapse active monitoring (n = 101) or control (depressive relapse active monitoring alone) (n = 102) and followed up for 2 years. Structured self-report instruments for service use and absenteeism provided cost data items for health economic analyses. Treatment utility, expressed as disability-adjusted life years, was calculated by adjusting the number of days an individual was depressed by the relevant International Classification of Diseases 12-month severity of depression disability weight from the Global Burden of Disease 2010. Intention-to-treat analysis assessed the incremental cost–utility ratios of the interventions across mental health care, all of health-care and whole-of-society perspectives. Per protocol and site of usual care subgroup analyses were also conducted. Probabilistic uncertainty analysis was completed using cost–utility acceptability curves.
Results:
Mindfulness-based cognitive therapy participants had significantly less major depressive episode days compared to controls, as supported by the differential distributions of major depressive episode days (modelled as Poisson, p < 0.001). Average major depressive episode days were consistently less in the mindfulness-based cognitive therapy group compared to controls, e.g., 31 and 55 days, respectively. From a whole-of-society perspective, analyses of patients receiving usual care from all sectors of the health-care system demonstrated dominance (reduced costs, demonstrable health gains). From a mental health-care perspective, the incremental gain per disability-adjusted life year for mindfulness-based cognitive therapy was AUD83,744 net benefit, with an overall annual cost saving of AUD143,511 for people in specialist care.
Conclusion:
Mindfulness-based cognitive therapy demonstrated very good health economic properties lending weight to the consideration of mindfulness-based cognitive therapy provision as a good buy within health-care delivery.
Keywords
Introduction
Major depressive disorder (MDD) is a major public health problem with increasing proportional contribution to the Burden of Disease even in countries with well-developed mental health-care services (Bromet et al., 2011). MDD often recurs, so indicated preventive interventions targeting people with confirmed vulnerability based on history of repeated major depressive episodes (MDEs) have potential to appreciably reduce population prevalence, associated disease burden and influence of a range of health-care and societal costs (Patten, 2007). Mindfulness-based cognitive therapy (MBCT) (Segal et al., 2002) is a group-based programme that, as an intervention indicated for people with three or more previous MDEs, has convincingly demonstrated efficacy in reducing relapse/recurrence rates of MDD in controlled trials (Chiesa and Serretti, 2011; Piet and Hougaard, 2011). While the Royal Australian and New Zealand College of Psychiatrists Clinical Practice Guidelines and the United Kingdom’s National Institute of Health and Clinical Excellence (NICE) Guidelines include MBCT for prevention of relapse/recurrence of multiple recurring MDD (Malhi et al., 2015; NICE, 2009), the adoption of MBCT as a complex intervention in health care will be influenced not only by effectiveness findings but also by health economic and other considerations (Demarzo et al., 2015). The relatively sparse economic evidence on MBCT to date is mainly based on work comparing MBCT and maintenance antidepressant medication tapering support (MBCT-TS) with continuation of maintenance antidepressant medication (m-ADM) (Kuyken et al., 2008, 2015). Choice of comparison group in MBCT studies is important (Edwards et al., 2015), and in the Australian context at least, this body of work may not represent how MBCT is commonly used in practice; available Australian and New Zealand clinical practice guidelines promote, for people with histories of three or more MDEs, m-ADM for 3 years or more (Malhi et al., 2015). Published work in Australia has demonstrated that MBCT has effect in the context of taking m-ADM (Meadows et al., 2014), so, in clinical practice, it may be often the case that MBCT would become an advised additional rather than alternative treatment to m-ADM (Kuyken et al., 2008, 2015). Hence, there is value in exploring the health economic properties of adding MBCT to routine care as actually delivered, whether this includes m-AMD or not, for people with three or more prior MDEs. This will inform decision making for policy-makers as to whether the addition of MBCT represents a ‘best buy’.
The Depression Awareness Recovery Effectiveness (DARE) study (Shawyer et al., 2012) was a large Australian randomised controlled trial (RCT) which examined the effectiveness of MBCT over 2 years of follow-up (Meadows et al., 2014). The study was sufficiently large to provide information on predefined subgroups including site of usual care being primary or specialist care (Meadows et al., 2014) and designed to examine health economic properties (Shawyer et al., 2012). The main results showed that the average number of days in MDEs over 2 years was 65 for MBCT participants and 112 for controls, significantly different with repeated-measures analysis of variance (ANOVA), F(1, 164) = 4.56, p = 0.03. Compared to controls, proportionally fewer MBCT participants relapsed in both year 1 and year 2 (odds ratio = 0.45, p < 0.05). Kaplan–Meier survival analysis for time to first depressive episode was non-significant, although trends favoured the MBCT group. Subgroup analyses showed that MBCT was most effective for people receiving specialist care and for people taking antidepressant/mood stabiliser medication.
Using data from DARE, this paper reports on the health economic properties of MBCT. This is done by considering cost-effectiveness and cost–utility through the 2 years of available data from the perspectives of mental health care alone, whole of health care and whole of society. We also report findings from subgroups defined by site of usual care (primary or specialist care) because these are important referral pathways for service design planning.
Method
Design
The DARE study design (Shawyer et al., 2012) was a prospective, pragmatic, multi-site, individually randomised controlled assessor-blind clinical trial based in Victoria, Australia. MBCT was delivered in line with published guidelines and with fidelity monitoring via video-taping (Segal et al., 2002). All participants, whether receiving MBCT or not, participated in ‘Depression Relapse Active Monitoring’ (DRAM), a supported self-monitoring intervention. Usual treatment was not otherwise directly altered by trial participation: participants remained free to use all health services including medication. Treatment and outcome data were collected over 26 months. The test condition was MBCT plus DRAM, now referred to as MBCT. The control condition was DRAM only, now referred to as the control. The multi-site study was approved by the Alfred Hospital, Barwon Health, Monash University, Peninsula Health, Monash Health and The Melbourne Clinic ethics committees. Written informed consent was obtained from all participants (Australian and New Zealand Clinical Trials Registry [ANZCTR], 2007). The specific health economic research question was, ‘How cost-effective is MBCT for non-depressed adults with a history of three or more MDEs in the Australian health-care setting, as compared with an active control group?’
Recruitment, selection and enrolment
Details about recruitment are found in the protocol paper (Shawyer et al., 2012). In summary, the study included people who met Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) (American Psychiatric Association [APA], 2000) criteria for ⩾3 MDEs in the context of a DSM-IV diagnosis of MDD (Recurrent) or Bipolar Disorder I or II, were aged between 18 and 75 years and were able to speak and read English fluently. Exclusions assessed at interview were current episode of MDD, current symptoms of a psychotic disorder, current eating disorder or obsessive–compulsive disorder, current alcohol or drug dependency other than tobacco, current benzodiazepine intake of more than 20 mg diazepam equivalent and inability to give informed consent. Other exclusions were a past diagnosis of a psychotic disorder where the treating clinician believed MBCT might be contraindicated, organic mental disorder or pervasive developmental delay, and current borderline or antisocial personality disorder. A telephone screening procedure preceded a face-to-face interview at which relevant modules from the Composite International Diagnostic Instrument 2.1 Auto Lifetime version (CIDI-LT) were administered.
Interventions
The health economic analysis in this trial compared costs associated with those who received the MBCT intervention to costs of control participants. The interventions are described in detail elsewhere (Shawyer et al., 2012). In summary, MBCT involved eight 2-hour group-training sessions delivered weekly and 3-monthly optional ‘booster sessions’. Four or more MBCT sessions were considered a minimal treatment dosage. All participants (MBCT group and controls) participated in DRAM, which involved monthly supported self-monitoring using the Patient Health Questionnaire-2 and -9 (Kroenke et al., 2001, 2003).
Sample size and randomisation
The sample size calculation for clinical outcomes indicated desired recruitment of 204 participants (Shawyer et al., 2012). Random assignment to treatment or control was conducted independently using a minimisation routine (Evans et al., 2001). Stratification variables included sex, diagnosis (bipolar disorder: yes/no), medication status (taking antidepressants/mood stabilisers: yes/no) and site of usual mental health care in the past 12 months (primary care: consultations with a general practitioner only; specialist care: consultations with a general practitioner and/or specialist). Including site of usual mental health care as a stratification factor allowed an examination of primary and specialist care subgroups separately, done to potentially inform future mental health-care planning.
One participant randomised to the control group committed suicide during the study. It seems a reasonable conjecture that a suicide represents a relapse in MDD. The loss of this individual’s whole remaining lifetime productivity constituted a clear outlier and was removed in favour of the control condition.
Cost measures
The clinical delivery costs of MBCT and DRAM were calculated directly using Medicare Benefit Schedule (MBS) clinician fees. DRAM direct costs were estimated using MBS item 80020 (Group Psychological Therapy – clinical psychologist) and based on the cost of a 1-hour group induction and an assumed monthly cost of 8 hours of self-monitoring support per 100 participants, in line with study experience. MBCT costs were based on a 1-hour initial consultation by a clinical psychologist (MBS 80000) and up to eight 2-hour group sessions (MBS 80020) with eight participants. DRAM intervention costs are assumed to be the same for all participants since this treatment, following a mandatory induction session, was delivered to all participants at fixed cost. In contrast, realised MBCT costs were dependent on the number of sessions attended by each participant.
National health-care unit costs were calculated for prescription medications and hospital, community health and social service contacts. Productivity losses were imputed using the human capital approach by multiplying reported days off work due to mental illness with an individual’s estimated salary.
Service use data, including overnight stays in hospital and health-care consultations, were collected with an adapted form of the instrumentation from the 2007 Australian National Survey of Mental Health and Wellbeing (Australian Bureau of Statistics [ABS], 2008) with additional details on health-care contacts. Prescription medication data collected through 3-monthly questionnaires provided information about medications used including name, frequency of use and dose. Package costs were collected from the Australian Monthly Index of Medical Specialties (MIMS) online database and medications were classified as ‘mental health–related pharmaceuticals’ (antidepressants, herbal nervous system preparations, antianxiety agents, antipsychotic agents, sedatives and hypnotics) and ‘all other pharmaceuticals’. Pharmaceutical Benefits Scheme (PBS) pricing was used with private retail pricing entered when PBS prices were unavailable. These were matched to self-reported individual dosage, adherence rates and typical package sizes to calculate weekly participant-specific medication costs. Where there was missing prescription medication data, we assumed changes to prescriptions and associated costs would be gradual and replaced missing values with estimated costs given by the mean of the adjacent observed data for the same individual.
Hospital Admissions Costs included a weighted average of daily hospital admission costs by admission type sourced from the Australian Refined Diagnosis Related Groups (AR-DRG5.2) (Australian Government Department of Health, 2012) weighted by the number of admission days in each admission sub-type. Practitioner consultation costs were sourced from the MBS. Additional participant-reported out-of-pocket expenses were also included.
Where participants had a paid occupation, these were allocated to Australian and New Zealand Standard Classification of Occupations (ANZSCO) Major Group classifications and matched with ABS earnings data. Participant days away from work were then used to calculate productivity losses related to absenteeism using the human capital approach. Where participants did not have a paid occupation but engaged in unpaid economic activity such as home duties or volunteering, we used the survey-measured days out of role alongside ANZSCO labourer earnings to estimate the value of unpaid economic activity. The impact of presenteeism was not measured.
Unit costs not available from the resources above were collected from national literature, statistical surveys and other similar sources detailed in the Australian unit cost literature (Mihalopoulos et al., 2005).
The resultant cost data are presented in 2009 first quarter Australian dollars, with costs of each participant discounted by the appropriate ABS (2013) health group index and ordinary consumer price index (CPI) indexed to individuals’ date of commencement. A yearly discount rate of 3% is used.
Clinical outcome measure
Days in MDEs was assessed using the relevant World Health Organization CIDI question, following administration of the CIDI 2.1 12-month version depression module to diagnose MDE(s) in the previous 12 months. Treatment utility was expressed as disability-adjusted life years (DALYs). The DALY is a measure of participant-specific relative quality and quantity of life lived and was calculated by adjusting the number of days in an MDE over the previous 12 months by the relevant International Classification of Disease (ICD-10) 12-month severity of depression disability weight from the Global Burden of Disease 2010 (Vos et al., 2005a, 2005b). The use of the DALY measure allows comparisons with studies from the Assessing Cost-Effectiveness–Mental Health (ACE-MH) framework (e.g. Vos et al., 2005b) which forms the basis for the broadest comparative analyses of health interventions for the Australian population to date.
Procedure
The costs associated with each participant were estimated based on both medical and non-medical factors that affect health, and the health economic analysis followed general health economic principles (Glied and Smith, 2013). Cost measures were recorded at face-to-face interviews conducted at baseline and at the 14- and 26-month follow-up. Days in MDEs were recorded at the 14- and 26-month follow-up. Self-report instruments administered at baseline, end of treatment and at 3-monthly intervals thereafter included medications taken and absenteeism from usual daily duties. See immediately below and Shawyer et al. (2012) for further details.
Statistical analysis
Days in MDEs during each of the two 12-month study periods were determined for each participant, and the distributions of MDE days for the two groups compared using zero-inflated Poisson and negative binomial models.
Cost-effectiveness analysis (intention to treat [ITT]) was carried out for three perspectives: mental health-care costs, total health-care costs and whole-of-society costs over the full 2-year study period. Costs in the mental health-care perspective included physician, hospital and pharmaceutical costs related to the person’s mental health, while the total health-care perspective represented these same cost categories pertaining to both physical and mental health. The whole-of-society perspective included total health-care costs and cost to society from the individual being unable to attend to their usual economic activities due to illness. Per protocol analysis (PPA) were also undertaken for those attending four or more sessions, while separate analyses were conducted for site-of-usual-care subgroups.
Cost–utility was evaluated by relating the cost per participant receiving each of the two treatments to their relative effectiveness in terms of the primary clinical outcome (DALYs). The cost–utility of the MBCT treatment was assessed by the incremental cost–utility ratio (ICUR) calculated as the difference in mean costs divided by the difference in mean-DALYs between the two treatment groups accrued over the study period (Fenwick and Byford, 2005). Evaluated costs and outcomes were aggregated at three perspectives: mental health care, health care, and societal level.
We present in graphical format the whole of mental health cost–utility plane of the bootstrapped mean differences in costs and DALY scores between study groups: x-axis (utility differential) and y-axis (cost differential). The data points were created by drawing with replacement 1000 bootstrapped samples of the same size as the original data set (Campbell and Torgerson, 1999). The axes divide the graph into four quadrants that represent the four possible outcomes for MBCT treatment compared to the control. The quadrant representing ‘more effective and less costly’ is deemed dominant and represents the best outcome where a treatment is both cheaper and more effective than its comparator (Glied and Smith, 2013). In contrast, the worst outcome would be the quadrant representing ‘less effective and more costly’. Figure 1 shows the four quadrants and an explanation for the outcome in each.
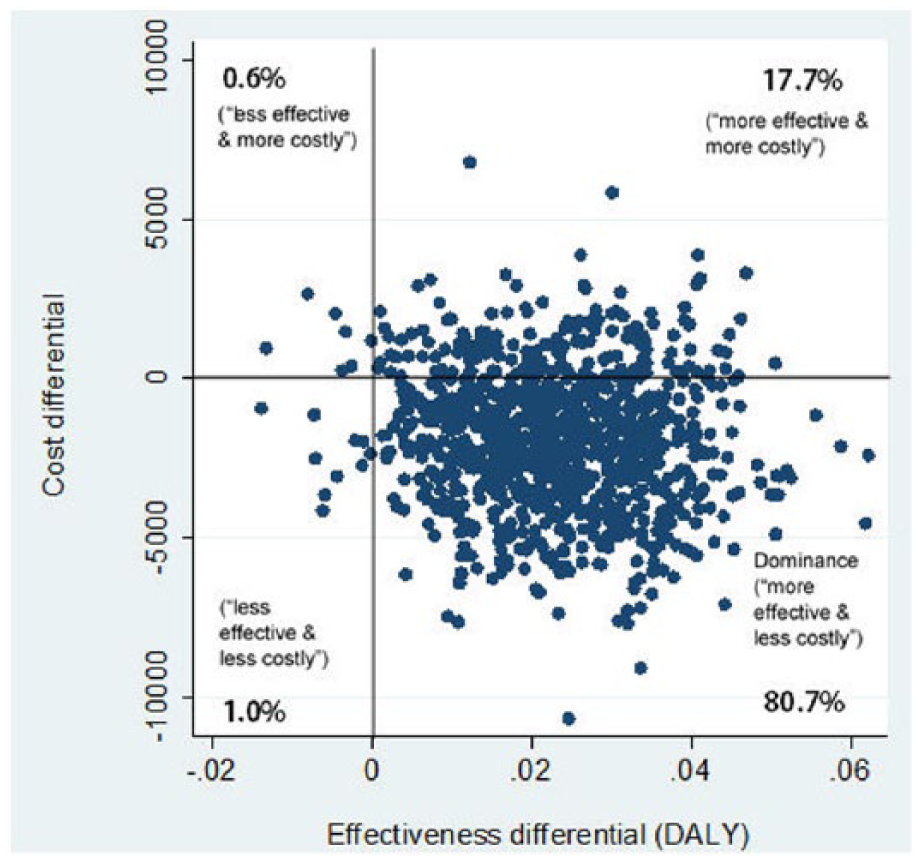
Cost–utility plane for mental health services perspective (intention to treat). The x-axis (effectiveness differential) and y-axis (cost differential) divide the graph into four quadrants that represent the four possible outcomes for MBCT treatment compared to the control. The percentage of bootstrap iterations in each of the four quadrants is shown.
A probabilistic sensitivity analysis was conducted by way of a set of cost–utility acceptability curves (CUACs) for the intervention compared with its alternative, using the bootstrapped data. The x-axis shows a range of values for the ceiling ratio and the y-axis indicates the probability that the data are consistent with a true cost–utility ratio falling below these ceiling amounts. These curves represent the uncertainty around estimates of average costs and effects resulting from sampling variation in terms of the maximum cost–utility ratio a decision maker would consider acceptable (Fenwick and Byford, 2005). When all points are in the optimal quadrant of ‘more effective and less costly’, the curve runs flat at 100% representing a dominant intervention. Likewise, when all points are in the most suboptimal quadrant of ‘less effective and more costly’, the curve runs flat at 0% and represents a dominated intervention. When observations feature in the other two quadrants, the CUAC shows the probability that the incremental cost–utility is below a particular threshold.
Missing data
In the main results paper (Meadows et al., 2014), sensitivity analyses showed that the missing data did not change the overall result with respect to days in MDEs. The cost–utility assessment in this study uses days in MDEs as the main unit for estimating overall costs, so the missing data are unlikely to largely influence the results. However, we explore this further by performing a missing data analysis for missing-at-random (MAR) properties. MAR was examined using univariate logistic regression of missing data outcome as binary (yes/no) and factors of test group, gender, age, site of usual care, antidepressant use and diagnosis of bipolar disorder. Values of p > 0.1 indicated MAR. ITT primary analyses were done using all available data but only if missing data were MAR and less than 12%. An overall missing data percentage of less than 12% was determined to be acceptable in this pragmatic trial.
Results
Participants and summary of clinical results
Details of study participants have been reported in an earlier paper (Meadows et al., 2014) according to CONSORT (CONsolidated Standards of Reporting Trials) guidelines (Zwarenstein et al., 2008) and are summarised here. Of the 482 applicant inquiries and referrals, 380 underwent phone screening and 326 appeared eligible to participate. A total of 250 participants proceeded to face-to-face intake assessment of whom 45 were excluded based on previously stated criteria and 1 declined further participation. The remaining 204 participants were randomised into the trial, and of these, 1 participant randomised to the MBCT group was later withdrawn when further information indicated a problem with eligibility. The final sample size was 203 including 102 and 101 individuals randomised to control and treatment groups, respectively. There were 203 participants with baseline data; however, some participants did not attend key study appointments at 14 and 26 months: failing to attend at 14 months were 20 individuals (12 in MBCT; 8 in control) and at 26 months were 25 individuals (12 in MBCT; 13 in control). This produced an overall attrition rate of 10.8%. Missing data were unrelated to test group, gender, age, diagnosis of bipolar disorder, or antidepressant use. The ITT primary analyses were done using all available data since missing data were MAR and less than 12%.
The average numbers of days in MDEs per year are shown in Table 1; average MDE days were consistently less in the MBCT group compared to controls in the overall sample (ITT) and all subgroups. The distribution of days was significantly different between the two groups (count data [days] modelled using zero-inflated Poisson and negative binomial distributions) indicating less days in MDE for the MBCT group compared to the control group. These results are consistent with the primary outcomes of the trial (Meadows et al., 2014).
Average days in major depressive episodes (MDEs) per year over 2 years for MBCT and control groups (intention to treat and per protocol).
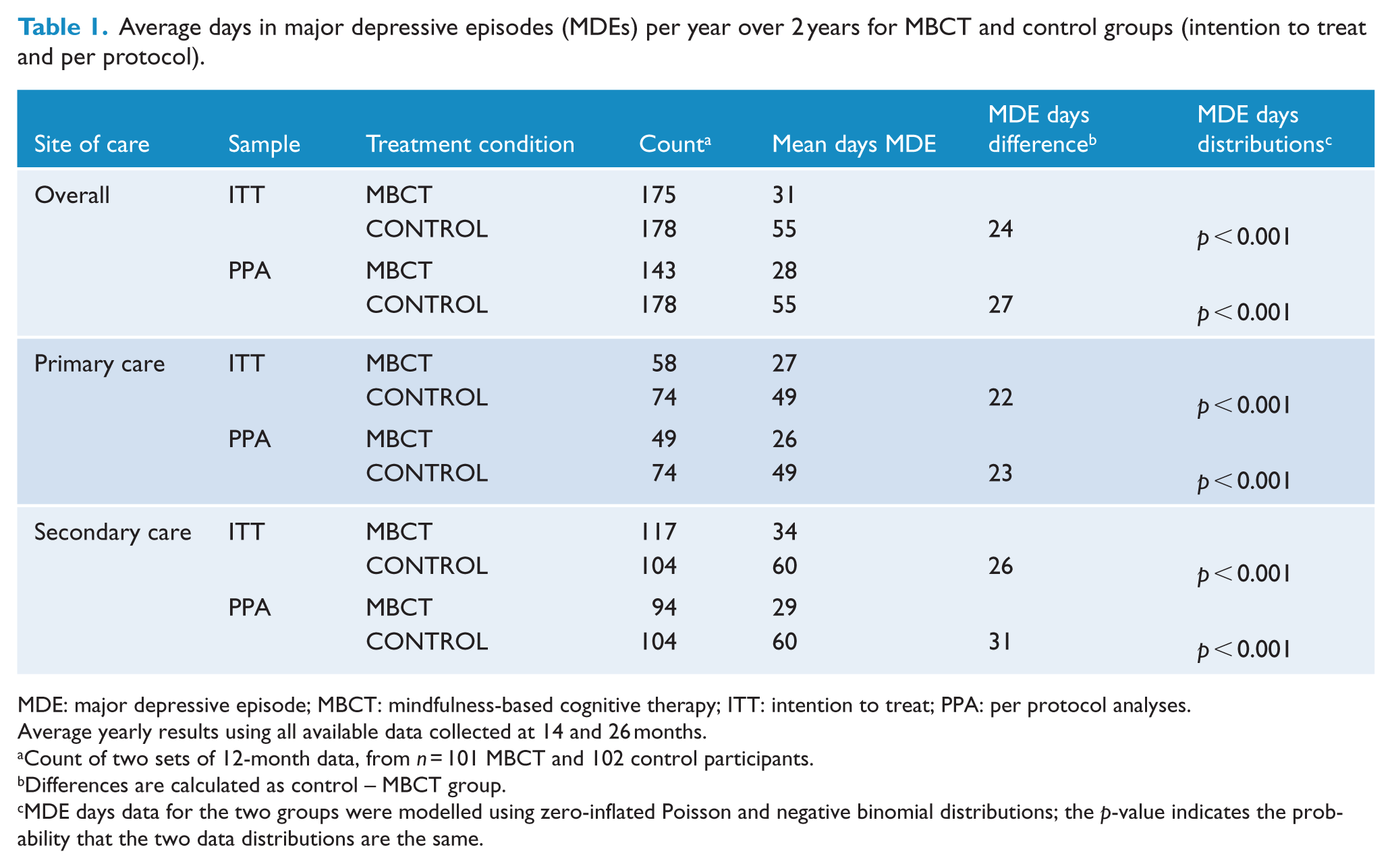
MDE: major depressive episode; MBCT: mindfulness-based cognitive therapy; ITT: intention to treat; PPA: per protocol analyses.
Average yearly results using all available data collected at 14 and 26 months.
Count of two sets of 12-month data, from n = 101 MBCT and 102 control participants.
Differences are calculated as control – MBCT group.
MDE days data for the two groups were modelled using zero-inflated Poisson and negative binomial distributions; the p-value indicates the probability that the two data distributions are the same.
Cost-effectiveness
The mean yearly costs for both groups are shown in Table 2, including the three perspectives: mental health-care costs, total health-care costs and whole-of-society costs. Median costs are also provided due to skewed data. A negative mean difference indicates a cost saving in favour of the MBCT intervention. Table 2 highlights cost saving in favour of the MBCT intervention in all three perspectives and all subgroup analyses, with one exception. The exception was the primary care analysis within the total health-care costs perspective only, which showed greater yearly costs in the MBCT group compared to controls.
Average yearly costs over 2 years for MBCT and control groups (intention to treat and per protocol).
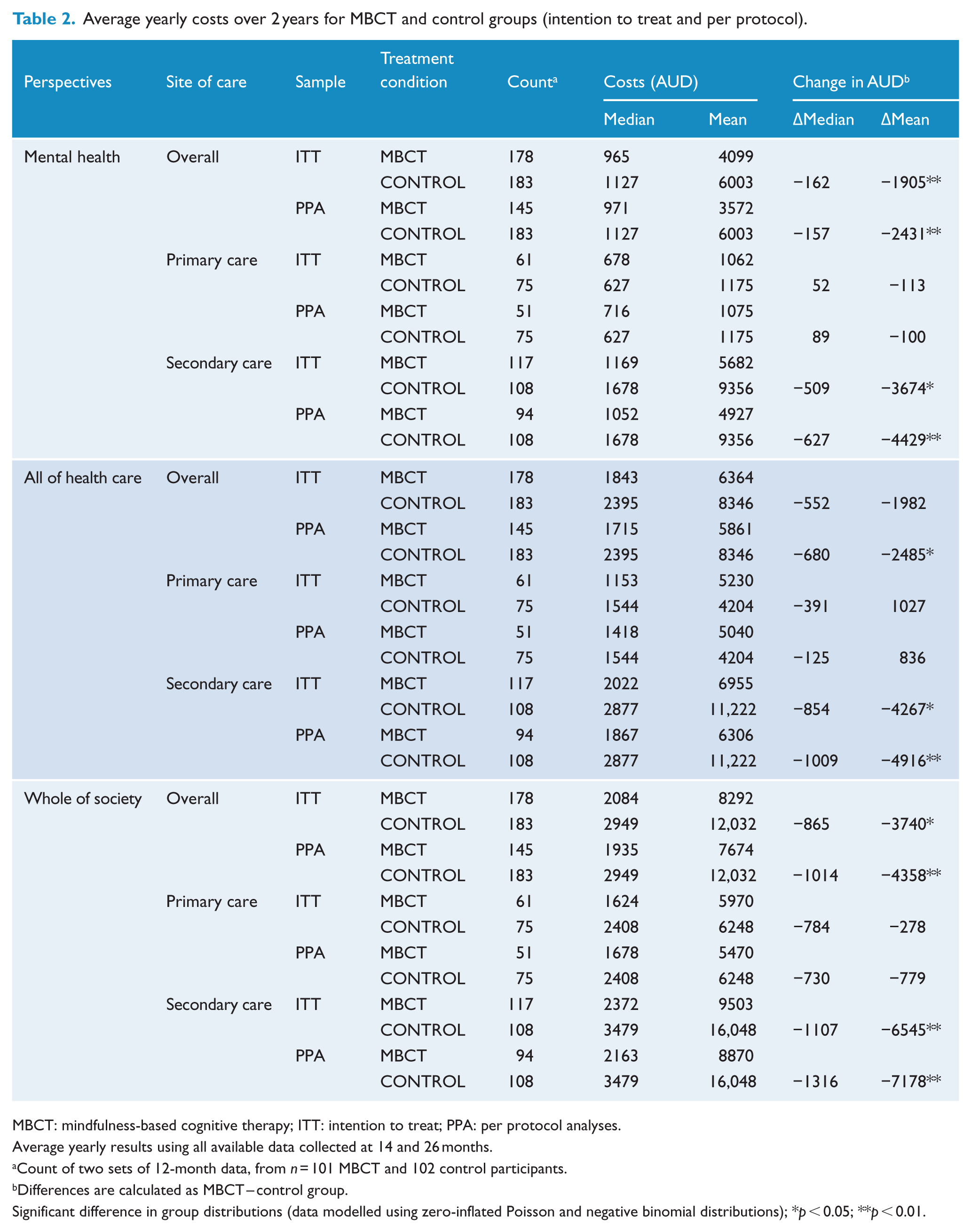
MBCT: mindfulness-based cognitive therapy; ITT: intention to treat; PPA: per protocol analyses.
Average yearly results using all available data collected at 14 and 26 months.
Count of two sets of 12-month data, from n = 101 MBCT and 102 control participants.
Differences are calculated as MBCT – control group.
Significant difference in group distributions (data modelled using zero-inflated Poisson and negative binomial distributions); *p < 0.05; **p < 0.01.
Table 2 shows that for the ITT analysis the average yearly mental health costs were AUD4099 per person randomised to MBCT compared to AUD6003 in the control group. For the PPA analysis, the average yearly mental health costs were AUD3572 per person randomised to MBCT compared to AUD6003 in the control group.
The average yearly cost-effectiveness and cost–utility derived measures per person are shown in Table 3, and are displayed by mental health-care costs, total health-care costs and whole-of-society costs. An incremental cost value (in AUD) to avert a day of major depression in one person by intervening with MBCT is shown, and the majority of these values are negative representing savings (less costs) achieved by intervening compared to the control. The average yearly DALY values are shown. The incremental cost values to intervene and avert a DALY per person are shown. Again, the majority of these values are negative representing yearly savings (less costs) achieved by intervening and averting a DALY compared to the control. The exception was the primary care analyses within the total health-care costs perspective, which showed greater yearly costs for MBCT compared to control.
Average yearly cost-effectiveness and cost–utility for MBCT and control groups (intention to treat and per protocol).
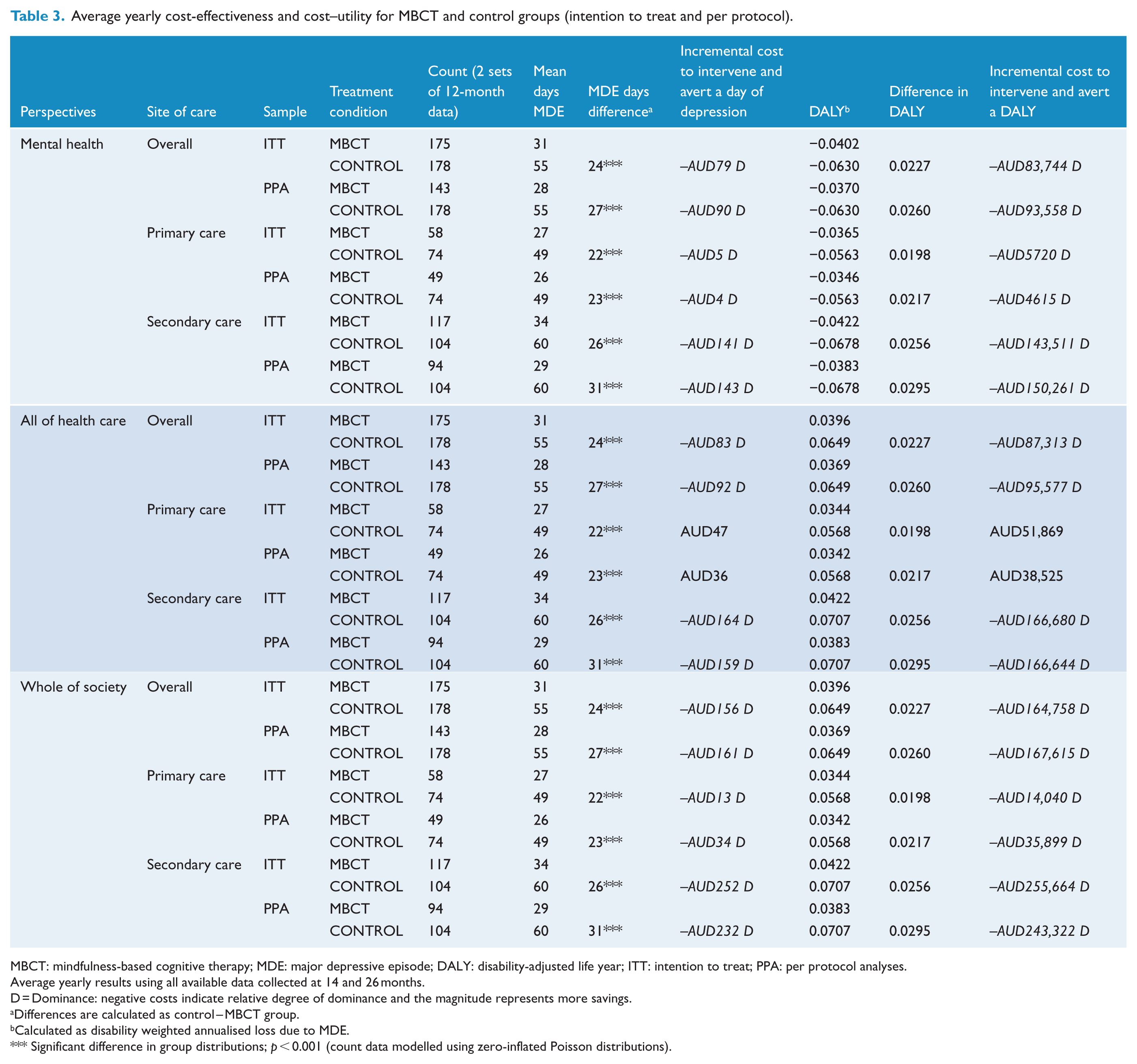
MBCT: mindfulness-based cognitive therapy; MDE: major depressive episode; DALY: disability-adjusted life year; ITT: intention to treat; PPA: per protocol analyses.
Average yearly results using all available data collected at 14 and 26 months.
D = Dominance: negative costs indicate relative degree of dominance and the magnitude represents more savings.
Differences are calculated as control – MBCT group.
Calculated as disability weighted annualised loss due to MDE.
Significant difference in group distributions; p < 0.001 (count data modelled using zero-inflated Poisson distributions).
Table 3 presents a tiered comparison of costs and DALYs. In the ITT analysis, when compared to the control condition, the mental health costs for MBCT produced a savings of AUD83,744 per person each year by averting a DALY and intervening with MBCT. In the PPA analysis, when compared to the control, the mental health savings were AUD93,558 per person each year by averting a DALY and intervening with MBCT.
Figure 1 shows the ITT cost–utility assessment for the mental health services perspective. The majority of the data points are in the ‘dominant’ quadrant, indicating that MBCT is more effective and less costly compared to the control.
Figure 2 shows the ITT cost–utility assessment results for the mental health services perspective, separated into primary care and specialist settings. This suggests that MBCT delivered to participants from specialist care is more cost-effective and largely dominant compared to participants from primary care.
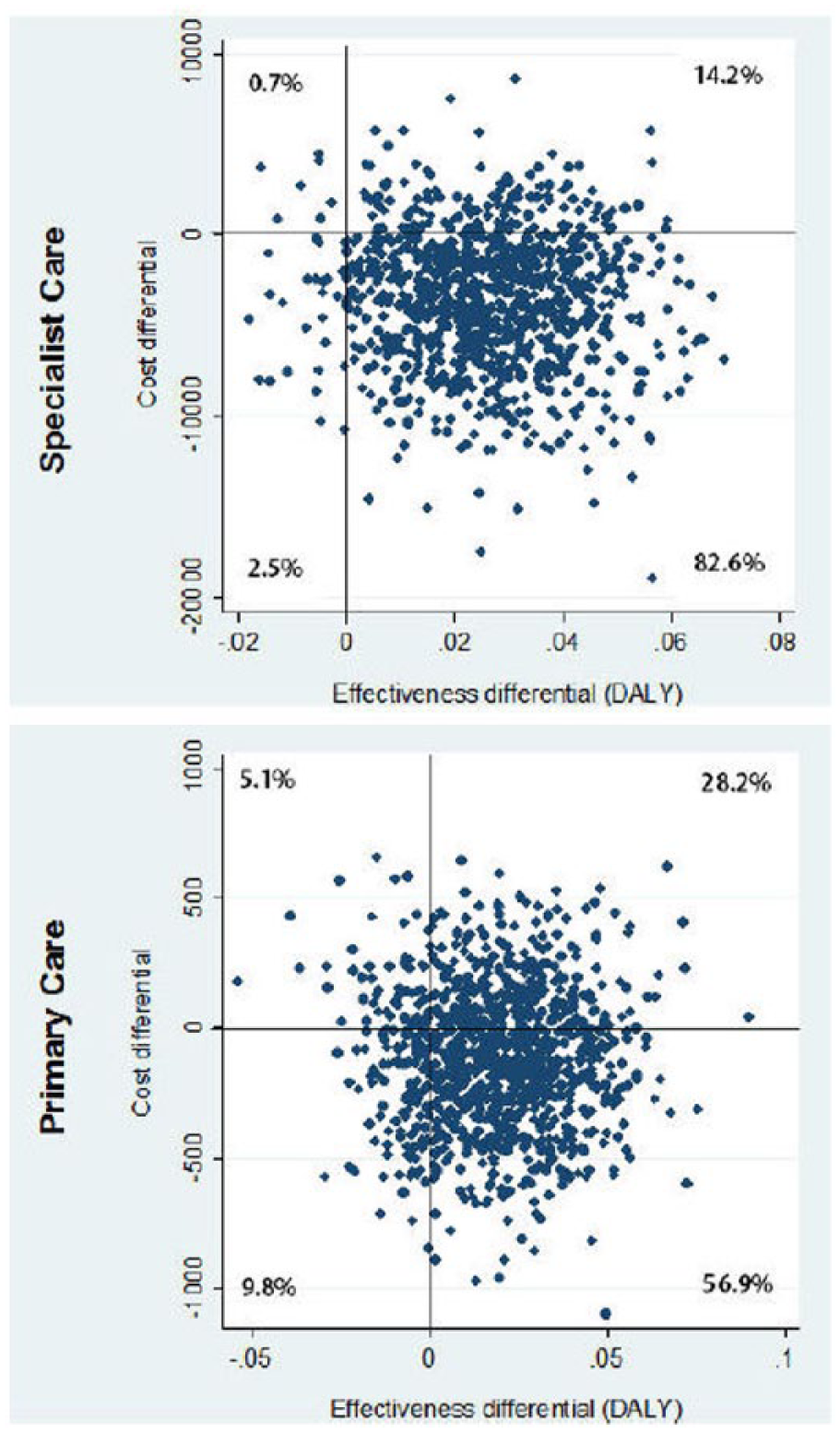
Cost–utility planes for specialist care and primary care (mental health-care perspective, intention to treat). The percentage of bootstrap iterations in each of the four quadrants is shown.
Sensitivity analysis
Figure 3 shows the ITT CUACs for the mental health perspective. It presents a probabilistic sensitivity analysis conducted using a set of CUACs for the intervention. The probability of MBCT treatment being less costly than the control (i.e. the probability of it being cost-effective when the decision maker is unwilling to pay anything additional for an extra point increase in DALY) is 81% from the mental health budget perspective, 62% at primary care level and 83% at specialist care level.
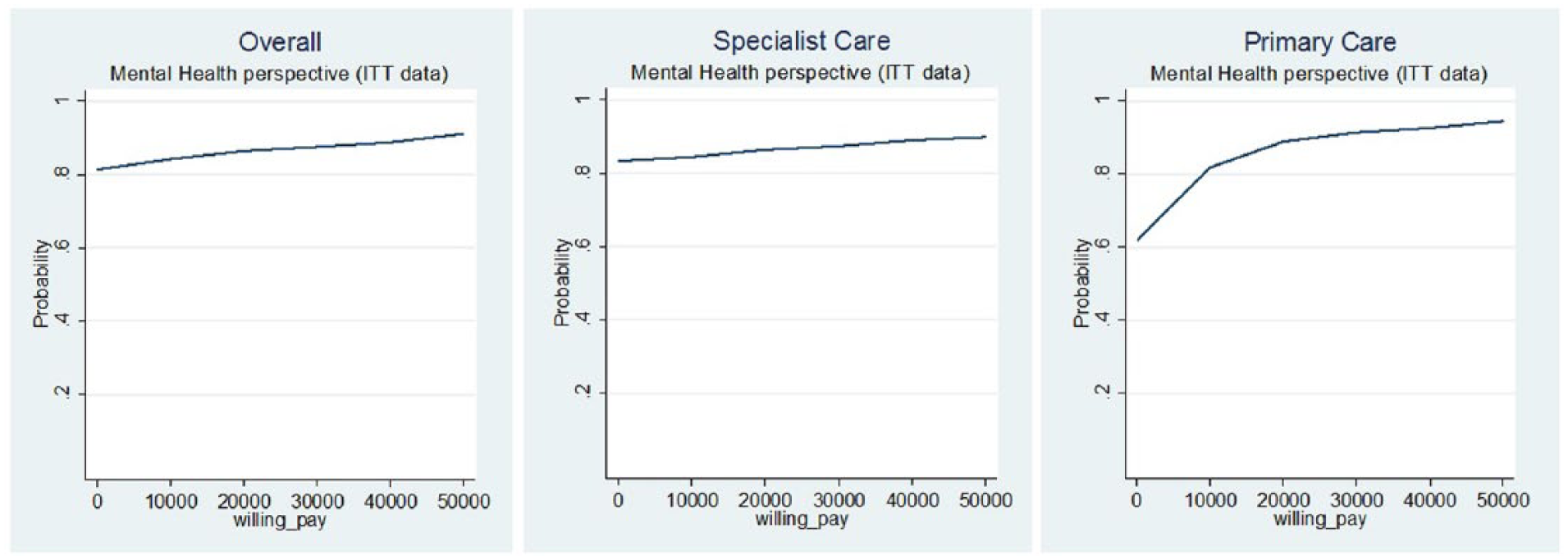
Cost–utility acceptability curves for overall, specialist care and primary care (mental health-care perspective, intention to treat). The x-axis represents the willingness to pay and y-axis represents the probability that the intervention will be effective.
Discussion
Summary of findings
This study provides evidence that MBCT as a treatment for preventing MDD has very good health economic properties. The results lend weight to the active consideration of MBCT provision to people with recurrent depression as a very good buy by commissioners, service managers, lead clinicians, and others involved in considering the profile of services available.
Taking a mental health-care or whole-of-society perspective, the intervention consistently demonstrates dominance in health economic terms. Taking a utilitarian moral position, we might reasonably conclude that, as a selective preventative intervention (Gordon, 1983), MBCT be targeted to people with a history of three or more MDEs and that all eligible participants be encouraged to participate. It is, however, often the case that commissioners or managers need to make decisions based on a narrower budgetary perspective. In this context, it is potentially of significance that the central estimate of costs within the mental health-care system as ascertained in this study indicates that introducing MBCT into a service profile is likely to lead to an overall reduction in service costs. The ITT CUACs for the mental health perspective demonstrated that the probability of MBCT treatment being less costly than the control is 81% from the mental health budget perspective, 62% at the primary care level and 83% at the specialist care level. This exceeds the 50% decision rule (Claxton et al., 2005), consistent with maximising expected health gain from limited resources. Furthermore, for the Australian context, Vos et al. (2010) have suggested that interventions which improve health at a cost of less than AUD10,000 per DALY are ‘very cost-effective’; those between AUD10,000 and AUD50,000 per DALY are ‘cost-effective’ and those greater than AUD50,000 per DALY are ‘not cost-effective’. The CUAC plots how the probability of MBCT being cost-effective rises with increasing acceptability threshold values. If society’s maximum willingness to pay is AUD10,000, the probability of MBCT being ‘very cost-effective’ is 83% from the mental health perspective (82% primary care and 81% specialist care). At a figure of AUD50,000, the simulated probability of MBCT being ‘cost-effective’ is 91% (93% primary care and 89% specialist care). Where enrolled participants had received specialist mental health care, the cost of the MBCT is more than offset by gains in hospital mental health services and ambulatory care provision.
Considerations for service delivery
For many mental health services, making the transition to including MBCT in the range of available treatments presents multiple challenges (Demarzo et al., 2015). Commonly, mental health-care services are not optimally configured for the delivery of group interventions. Very often, the legitimate needs for the care of people with acute illnesses will understandably be placed above preventive activities. Furthermore, in the case of MBCT, we are considering an intervention that requires a significant time investment in developing a new set of clinical skills. In fact, the skill requirements go beyond narrowly clinical ones because there is also personal time investment from MBCT instructors adopting and sustaining at least some degree of mindfulness practice. For services to effectively implement MBCT, some investment will also need to be made in actively recruiting people who may be currently in remission. There are a numerous ways this could be done. These include analogues of the approach detailed in this study, that is, advertising directly through services or the media; an alternative or complimentary strategy would be to identify people during acute-phase treatment and then set up a recall system for contact some months later.
MBCT broadly falls with the paradigm for Cognitive Behaviour Therapy, which is already billed by certain practitioners under the MBS in Australia. Psychological services funded by MBS have recently been proven to be inequitable, particularly those having high co-payments paid by consumers (Meadows et al., 2015). It is probable that reducing any co-payment will make MBCT delivery more equitable. More generally, MBS group therapy items across allied health and psychiatry are not well suited to MBCT. Adding a new MBS item specific to MBCT, including increased remuneration to reflect the 2-hour sessions, could facilitate increased provision and equity of delivery. The impact and equitable delivery of MBCT could be further increased by having available an individual format for delivery, fully evaluated with health economic analysis. MBCT was originally designed as a group rather than individual therapy to increase its potential cost-effectiveness (Teasdale et al., 2000). However, treatment savings may be offset by potential reductions in effectiveness since it is common for participants to miss several sessions, a problem likely to be exacerbated in low socio-economic settings (Meadows et al., 2014; Mein et al., 2012). With no redundancy in the programme, this means there will be reduced benefit commensurate with the number of sessions missed. MBCT has been studied only as a group intervention and this delivery format limits its dissemination potential, especially within MBS where, in fiscal year 2014–2015, of 4.2 million ‘Better Access’ psychology services, 0.8% were for group therapy (Medicarestatistics.humanservices.gov.au, 2015).
Comparison with other studies
It is difficult to compare health economic outcomes from one study to another due to non-standardisation of methodologies and comparison groups (Edwards et al., 2015). While imperfect, such comparisons are of important value. The benefit forgone in engaging in one intervention over another, the opportunity costs of the intervention is a crucial consideration in maximising individual’s wellbeing as well as efficiently deploying limited resources society-wide. In Britain, Kuyken et al. (2008) found that MBCT was no more effective or cost-effective than m-ADM. Their societal cost-effectiveness analysis suggested that the additional cost of MBCT may be justified in terms of improvements in the proportion of patients who relapse, but only if the willingness to pay for such improvements is AUD1000 or above. In contrast, this study with a different comparison group found MBCT to be cost-effective in all but the primary care subgroup even at an AUD0 willingness to pay. A Dutch study in a primary care setting by Van Ravesteijn et al. (2013) found MBCT applied to patients with medically unexplained symptoms resulted in an ICUR of €56,000 per quality-adjusted life year (QALY). Like in this study, they observed that MBCT appears to cause a shift in the use of health-care resources with increased mental health-care costs and decreased hospital care costs under the MBCT treatment.
Strengths and limitations
The 2-year follow-up and larger sample size in this study have advantages compared to some other studies in that it allows for at least some short-term effects to become apparent. However, lifetime health economic outcomes are unclear. It is hypothesised that the effects of MBCT can diminish once the initial treatment period is over, but also that these effects can be maintained with regular booster sessions. Maintenance may require repeated return to intensive practice while increased experience may reduce the required investment over time. MBCT requires a significant upfront time investment by the participant as well as health-care professionals. While during the group participation phase there is 40 minutes per day self-engagement in treatment, the time cost in such activities needed to maintain the benefit of MBCT is unknown. This time cost is not straightforward to assess since while it may simply be considered an opportunity cost against time devoted to labour, such an opportunity cost may be mediated by the individual’s leisure and lifestyle preferences. However, once MBCT techniques are learned, the continuing burden on the health-care system is minimal and average costs per DALY per year are likely to decline over the longer term even with the provision of regular booster sessions. As a further point, this analysis does not incorporate losses due to diminished efficiency at work as well as of unpaid work and domestic tasks. While the distributional characteristics of long-run outcomes in this post-treatment period are uncertain, there remains a strong case for assuming increased cost-effectiveness for MBCT as treatment for MDD over time.
Conclusion
Clinical leaders, service managers, insurers and commissioners responsible for determining what options are available for populations within their care need to decide proportionally how much of the resource to allocate to an intervention such as MBCT. In this consideration, health economic analyses such as that laid out in this paper can have an important part to play. In Australia, it has been suggested that preventive intervention should be judged according to a series of cost thresholds that identify levels of cost-effectiveness. Within the context of these definitions, the findings of this study would suggest that MBCT is highly cost-effective and a desirable option among the range of preventive interventions.
Footnotes
Acknowledgements
We gratefully acknowledge the regular infrastructure and financial contributions from Monash Health towards this research centre. We would also like to acknowledge the support of the associate investigators and mental health services within the following organisations: Alfred Health (Professor Jayashri Kulkarni), Barwon Health (Professor Michael Berk), Peninsula Health (Associate Professor Richard Newton; Dr Sean Jesperson), Monash Health (Associate Professor Saji Damodaran) and Healthscope. Most of all, we would like to express our gratitude to our participants who made the trial possible. This work was undertaken at Southern Synergy, Monash University, Australia.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by a grant from the National Health and Medical Research Council of Australia (Grant 436897).
