Abstract
Objective:
Recent advances in neuroimaging techniques have enabled a better understanding of the neurobiological underpinnings of anorexia nervosa (AN). The aim of this paper was to summarise our current understanding of the neurobiology of AN.
Methods:
The literature was searched using the electronic databases PubMed and Google Scholar, and by additional hand searches through reference lists and specialist eating disorders journals. Relevant studies were included if they were written in English, only used human participants, had a specific AN group, used clinical populations of AN, group comparisons were reported for AN compared to healthy controls and not merely AN compared to other eating disorders or other psychiatric groups, and were not case studies.
Results:
The systematic review summarises a number of structural and functional brain differences which are reported in individuals with AN, including differences in neurotransmitter function, regional cerebral blood flow, glucose metabolism, volumetrics and the blood oxygen level dependent response.
Conclusion:
Several structural and functional differences have been reported in AN, some of which reverse and others which persist following weight restoration. These findings have important implications for our understanding of the neurobiological underpinnings of AN, and further research in this field may provide new direction for the development of more effective treatments.
Introduction
Anorexia nervosa (AN) is a serious psychiatric condition with a 12-month prevalence rate of 0.4% among females, and approximately one-tenth of that among males (American Psychiatric Association, 2013). The crude mortality rate of individuals admitted into US university hospitals with AN is approximately 5% per decade (American Psychiatric Association, 2013), among the highest mortality rate of any psychiatric disorder (Harris and Barraclough, 1998; Sullivan, 1995). Furthermore, AN is associated with exceptionally high relapse rates (Eckert et al., 1995; Löwe et al., 2001; Norring and Sohlberg, 1993; Strober et al., 1997; Zipfel et al., 2000). A major contributing factor for the high rates of morbidity and mortality experienced by individuals with this condition is that the cause or causes of the illness are not clear, and although treatment modalities such as cognitive behaviour therapy and family therapy have emerging evidence for efficacy, many patients remain under- or unresponsive. With increasing advances in technology, particularly with the development of sophisticated neuroimaging techniques, we are able to gain a better understanding of the neurobiological underpinnings of this condition.
A large number of structural neuroimaging studies have been undertaken in AN since the mid-1980s, and with the emergence of more advanced neuroimaging techniques, the number of functional imaging studies in AN has grown rapidly. Therefore, the aim of this systematic review was to summarise our current understanding of the neurobiology of AN. Specifically, differences in neurotransmitter function, regional cerebral blood flow, glucose metabolism, volumetrics and the blood oxygen level dependent (BOLD) response in AN will be reviewed.
Methods
The literature was searched using the electronic databases PubMed and Google Scholar, and by additional hand searches through reference lists and specialist eating disorders journals. Journals were searched from 1980 to July 2013. Search key terms included anorexia nervosa coupled with one or more of the following: neurobiology, neuronal, neurotransmitters, serotonin, dopamine, neuropeptides, SPECT, PET, MRI, fMRI, functional, structural, neuroimaging. The search resulted in an unmanageable number of hits (N > 100,000) and so only relevant studies meeting the eligibility criteria were included. For studies to be included they had to meet the following criteria: written in English; used only human participants; had a specific AN group and not combined with other eating disorder patients; group comparisons were reported for AN compared to healthy controls, not merely AN compared to other eating disorders or psychiatric groups; used clinical populations and not non-clinical populations with AN symptoms; not case studies.
Results and discussion
Our search strategy yielded a total of 29 and 81 publications meeting the above eligibility criteria reporting structural and functional brain imaging in AN, respectively, all of which are included in this review. Of the 29 studies reporting structural data in AN, the three earliest studies utilised computerised tomography (CT), while the remaining studies utilised magnetic resonance imaging (MRI). Of the 81 functional imaging studies in AN, 15 investigated differences in neuronal systems: one utilising blood samples, five using lumbar puncture techniques, one utilising single-photon emission tomography (SPECT) and eight employing positron emission tomography (PET). Fifteen of the 81 functional imaging studies reviewed utilised SPECT, seven employed PET to investigate glucose metabolism, and 44 of the studies reviewed utilised fMRI to study functional brain differences in AN. A summary of the results of these studies are presented in Tables 1 to 4.
Structural brain differences in anorexia nervosa utilising magnetic resonance imaging.
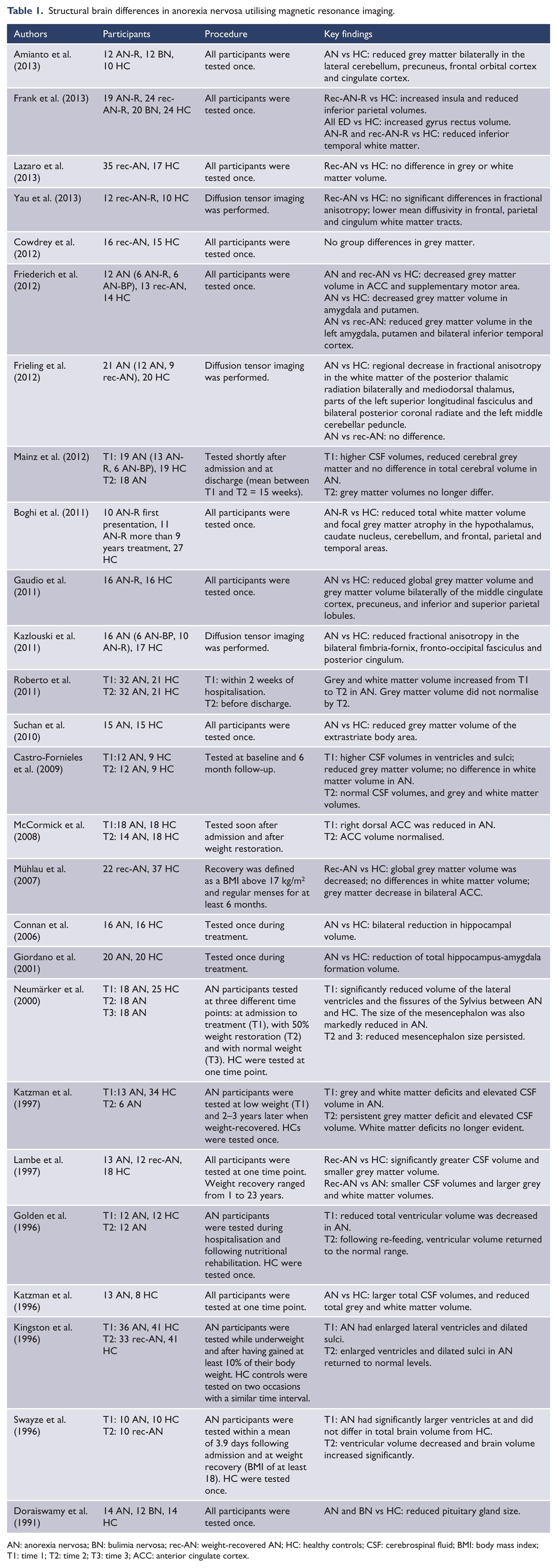
AN: anorexia nervosa; BN: bulimia nervosa; rec-AN: weight-recovered AN; HC: healthy controls; CSF: cerebrospinal fluid; BMI: body mass index; T1: time 1; T2: time 2; T3: time 3; ACC: anterior cingulate cortex.
Structural brain differences in anorexia nervosa utilising computed axial tomography.

AN: anorexia nervosa; rec-AN: weight-recovered AN; HC: healthy controls; CSF: cerebrospinal fluid; T1: time 1; T2: time 2; T3: time 3.
Functional brain differences in AN utilising functional magnetic resonance imaging.
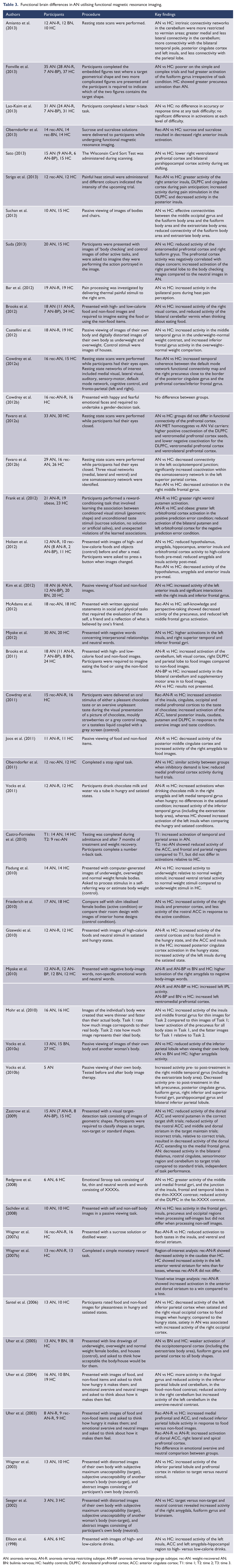
AN: anorexia nervosa; AN-R: anorexia nervosa restricting subtype; AN-BP: anorexia nervosa binge-purge subtype; rec-AN: weight-recovered AN; BN: bulimia nervosa; HC: healthy controls; DLPFC: dorsolateral prefrontal cortex; ACC: anterior cingulate cortex; T1: time 1; T2: time 2; T3: time 3.
Functional brain differences in anorexia nervosa utilising modalities other than functional magnetic resonance imaging.
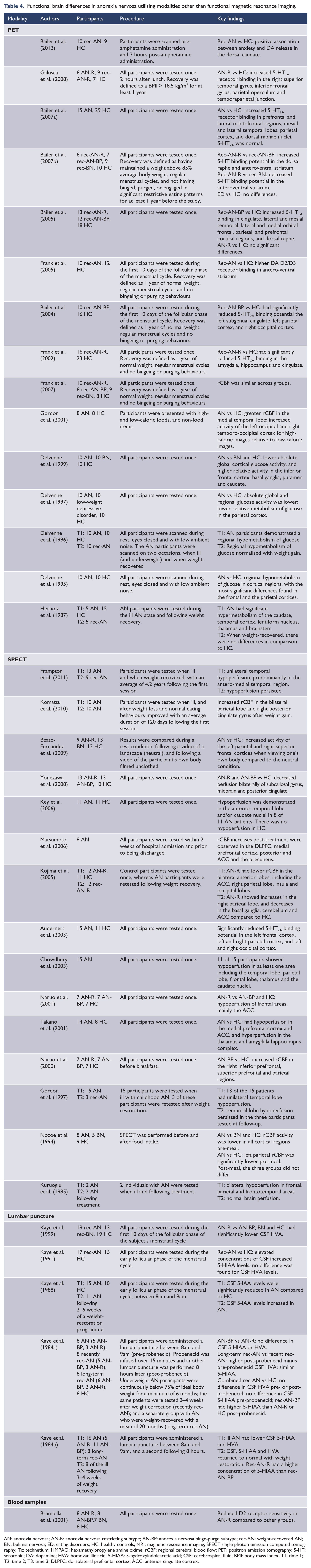
AN: anorexia nervosa; AN-R: anorexia nervosa restricting subtype; AN-BP: anorexia nervosa binge-purge subtype; rec-AN: weight-recovered AN; BN: bulimia nervosa; ED: eating disorders; HC: healthy controls; MRI: magnetic resonance imaging; SPECT:single photon emission computed tomography; Tc: technetium; HMPAO: hexamethylpropylene amine oxime; rCBF: regional cerebral blood flow; PET: positron emission tomography; 5-HT: serotonin; DA: dopamine; HVA: homovanillic acid; 5-HIAA: 5-hydroxyindoleacetic acid; CSF: cerebrospinal fluid; BMI: body mass index; T1: time 1; T2: time 2; T3: time 3; DLPFC: dorsolateral prefrontal cortex; ACC: anterior cingulate cortrex.
Structural brain differences
Structural changes are frequently observed in the brains of individuals with AN, and are generally thought to reflect the effects of malnutrition and starvation. The illness is associated with enlargement of the cortical sulci and ventricles (Artmann et al., 1985; Dolan et al., 1988; Kingston et al., 1996; Swayze et al., 1996), and enlargement of the inter-hemispheric fissure (Artmann et al., 1985). Cerebral dystrophic changes have been found to correlate with weight loss, and the reversal of these changes has also been found to correlate with the normalisation of body weight (Artmann et al., 1985; Golden et al., 1996; Kingston et al., 1996; Swayze et al., 1996). However, one study found no significant change in ventricular size, but a significant degree of sulcal widening after patients had attained normal body weight (Dolan et al., 1988). A more recent study found no differences in ventricular size of AN patients, but enlarged external cerebral spinal fluid (CSF) spaces when compared to controls (Palazidou et al., 1990). Individuals with AN have been reported to have larger total CSF volumes in the ventricles and sulci, and significantly reduced grey and white matter volumes (Boghi et al., 2011; Castro-Fornieles et al., 2009; Gaudio et al., 2011; Katzman et al., 1996, 1997; Lambe et al., 1997; Mainz et al., 2012; Mühlau et al., 2007; Roberto et al., 2011). Differences in white matter volume have not been consistently found (Castro-Fornieles et al., 2009; Mainz et al., 2012; Mühlau et al., 2007). Follow-up studies in weight-recovered patients have reported elevated CSF volumes and persistent grey but not white matter deficits (Katzman et al., 1997; Lambe et al., 1997; Roberto et al., 2011). However, two studies have reported normalisation of grey matter and CSF volume in weight-recovered AN patients (Castro-Fornieles et al., 2009; Cowdrey et al., 2012a; Mainz et al., 2012). Furthermore, a recent study by Lázaro et al. (2013) found no difference in grey or white matter volume between weight-recovered AN patients who were at 85% of the their expected body mass index (BMI) for at least one month, and a group of healthy controls.
Specific brain structures have also been found to differ between AN patients and controls, including reduced size of the pituitary gland (Doraiswamy et al., 1991), and a reduction of total hippocampus-amygdala formation volume (Giordano et al., 2001). Individuals with AN have also been found to have decreased grey matter in the anterior cingulate cortex (ACC) (Mühlau et al., 2007), and significantly reduced ACC volume whose degree of normalisation during treatment is related to outcome (McCormick et al., 2008). A more recent study has also reported a reduction of grey matter volume in both individuals currently ill with AN and weight-recovered patients (Friederich et al., 2012). This study also reported reduced grey matter volumes of the amygdala and putamen in ill individuals, and the supplementary motor area in both weight-recovered patients and patients with a current diagnosis. Furthermore, in comparison to weight-recovered individuals, individuals ill with AN were found to have reduced grey matter volumes of the left amygdala and putamen, and the bilateral inferior temporal cortex. Frank et al. (2013) on the other hand reported increased grey matter volume of the gyrus rectus and decreased insula volume compared to healthy controls. This study also reported reduced grey matter volume of the caudate and putamen and reduced inferior parietal white matter volume in weight-recovered AN, as well as reduced temporal white matter volume in both weight-recovered and ill AN. Other recent studies have also found decreased grey matter volumes in ill AN patients of the extrastriate body area (Suchan et al., 2010), and the middle cingulate cortex, precuneus, and inferior and superior parietal cortex (Gaudio et al., 2011). A recent study by Amianto et al. (2013) reported decreased grey matter in the lateral cerebellum, precuneus and frontal orbital and cingulate cortices. Furthermore, a study by Boghi et al. (2011) found reduced grey matter of the frontal and parietal cortices, caudate nucleus, hypothalamus and cerebellum. Additionally, in this study the AN group was divided into two subgroups based on illness duration: those who had a diagnosis of more than nine years, and those who were being treated at first presentation of the illness. The group with shorter disease duration were found to have more evident hypothalamic atrophy, whereas the cerebellum was more affected in the group with longer disease duration, in comparison to healthy controls. Additionally, individuals with AN have been found to have reduced hippocampal volumes in comparison to healthy controls (Connan et al., 2006), and the size of the mesencephalon has also been found to be markedly reduced in ill AN patients, which remains reduced in weight-restored individuals (Neumärker et al., 2000).
Several recent studies have also utilised diffusion tensor imaging (DTI) to assess white matter pathways in AN. Kazlouski et al. (2011) reported poorer white matter integrity in the bilateral fimbira-fornix, fronto-occipital fasciculus and posterior cingulum in AN, irrespective of AN subgroup. The AN group also showed higher mean diffusivity in these areas, in addition to frontal, parietal, temporal and occipital areas. However, in a more recent study by Yau et al. (2013), the authors reported lower mean diffusivity in frontal, parietal and cingulum white matter tracts in weight-recovered AN, unlike the study by Kazlouski et al. (2011) who found higher mean diffusivity in these areas. Yau et al. (2013) reported poorer white matter integrity in weight-recovered AN who had a history of more severe illness. Frieling et al. (2012), on the other hand, investigated white matter integrity in a group of individuals with a current diagnosis of AN combined with individuals weight-recovered from the illness, and a sample of healthy participants. This study reported regional decreases in white matter of the posterior thalamic radiation bilaterally and the mediodorsal thalamus in the combined AN group. Decreases were also reported in the left superior longitudinal fasciculus, bilateral posterior coronal radiate and left middle cerebellar peduncle. Additionally, no differences were found between the two subgroups of AN, currently ill or weight-recovered.
Functional differences
Neuronal systems
A neuronal system of particular interest in relation to the neurobiology of AN is the serotonin (5-HT) system. Research in AN has mainly focused on the main metabolite of 5-HT, 5-hydroxyindoleacetic acid (5-HIAA), and the 5-HT receptors 5-HT1A and 5-HT2A. Individuals with AN in the ill state have significantly lower CSF basal concentrations of the 5-HT metabolite 5-HIAA (Kaye et al., 1984b, 1988). However, those recovered from AN have significantly increased 5-HIAA levels in comparison to healthy controls (Kaye et al., 1991), particularly those recovered from the binge-eating purging subtype of AN (AN-BP) (Kaye et al., 1984a).
Recent neuroimaging studies have investigated 5-HT binding differences in the brain. In a study undertaken by Bailer and colleagues (2007b), the investigators reported that ill AN individuals had increased 5-HT1A binding in the subgenual, mesial temporal, orbital frontal and raphae brain regions, and the prefrontal, lateral temporal, ACC and parietal regions. Similar findings have been reported for 5-HT1A binding in ill participants (Bailer et al., 2007b; Galusca et al., 2008), whereas recovered AN sufferers have been reported to have diminished binding potential for 5-HT2A and increased binding potential of 5-HT1A. Audenaert et al. (2003) reported significantly reduced 5-HT2A binding in the left frontal cortex as well as the left and right parietal and occipital cortices of individuals with AN in comparison to control participants. Bailer et al. (2005), on the other hand, reported increased 5-HT1A binding potential of the dorsal raphe, and the cingulate, lateral and medial temporal, lateral and medial orbitofrontal, parietal and prefrontal cortices in weight-recovered AN. Studies investigating the specific subtypes of AN found that weight-restored restricting subtype AN (AN-R) participants had reduced 5-HT2A receptor activity in the subgenual and pregenual cingulate cortex, and mesial temporal and parietal cortical areas (Frank et al., 2002), whereas reduced 5-HT2A receptor activity in the subgenual cingulate, and the mesial temporal, lateral temporal, parietal and occipital cortices has been reported for recovered AN-BP participants, in comparison to controls (Bailer et al., 2004). A further study, however, found no difference in 5-HT binding potential between AN subgroups and controls, but increased 5-HT binding potential in the dorsal raphe and anteroventral striatum in recovered AN-R compared to recovered AN-BP participants; and decreased 5-HT binding potential in the anteroventral striatum in AN-R compared to recovered individuals with BN (Bailer et al., 2007a).
A neurotransmitter system largely related to the serotonergic system which has also been implicated in the aetiology and course of AN is the dopaminergic system. Homovanillic acid (HVA), the major metabolite of dopamine (DA) in humans, has been found to be decreased in the CSF of individuals with AN in the ill state (Kaye et al., 1984b). Two related studies have reported that HVA returns to normal levels following weight restoration (Kaye et al., 1984a, 1984b). However, a more recent study undertaken by the same investigators examining the HVA levels in individuals recovered from AN utilising more stringent criteria than purely weight restoration (including over one year of normal weight, regular menstrual cycles, no restricting eating patterns and no bingeing or purging) found that individuals recovered from AN-R had significantly lower CSF HVA than AN-BP, bulimia nervosa (BN) participants and healthy controls (Kaye et al., 1999).
In related work, Frank et al. (2005) used positron emission tomography (PET) imaging with radiogland [11C]raclopride to assess DA D2/D3 receptor function in recovered AN individuals. The investigators reported significantly higher [11C]raclopride binding potential in the antero-ventral striatum than control participants, providing support for the possibility that AN is associated with decreased intrasynaptic DA concentration or increased D2/D3 receptor density; the authors suggest this may contribute to the disturbed reward processing exhibited in AN. Another study using an amphetamine challenge and PET found that recovered AN participants exhibited a positive association between endogenous DA release and anxiety in the dorsal caudate, possibly explaining why food-related DA release produces anxiety in AN but is pleasurable in healthy individuals (Bailer et al., 2012).
Additionally, AN has been associated with altered D2 receptor sensitivity demonstrated by significantly reduced growth hormone response to growth hormone releasing hormone administered with apomorphine, a selective D1 and D2 receptor agonist (Brambilla et al., 2001). A study indirectly investigating dopaminergic function in AN also revealed a deficit in this group (Lawrence et al., 2003). Participants were administered a task which involved the learning of a series of two-alternative force-choice visual discriminations. This is analogous to tasks that activate DA neurons in primates and is sensitive to neurotransmission, including L-dopa treatment in Parkinson’s disease. The AN group was found to show deficits in learning during the early stages of the task when DA activity should be at a maximum, providing indirect evidence for altered DA neurotransmission in AN.
Neuropeptides have also been of interest in AN as they are involved in the regulation of feeding behaviours. Abnormalities in the ill state of AN have been found for a variety of neuropeptides including opioid peptides, oxytocin, neuropeptide-Y and leptin, although the differences observed appear to normalise after weight restoration, suggesting a state rather than trait effect (for a review see Bailer and Kaye, 2003).
Regional cerebral blood flow
Functional neuroimaging is relatively recent technology which has allowed the indirect measurement of brain activity. Early functional studies utilised single-photon emission computed tomography (SPECT) and showed hypo- and hyperperfusion at rest in a number of brain areas in the ill state of AN. Kuruoglu et al. (1985) reported hypoperfusion in frontal, parietal and frontotemporal areas, which normalised following weight restoration. Another study reported unilateral temporal lobe hypoperfusion which persisted in a subgroup who had a follow-up scan following weight restoration (Gordon et al., 1997). A more recent study by Chowdhury et al. (2003) revealed hypoperfusion in the temporal, parietal and frontal lobes, thalamus, and caudate nuclei. Other recent studies found hypoperfusion in the medial prefrontal cortex, the ACC (Takano et al., 2001), the posterior cingulate gyrus, the subcallosal gyrus, the midbrain (Yonezawa et al., 2008), the anterior temporal lobe and caudate nuclei (Key et al., 2006), and hyperperfusion in the thalamus and the amygdala-hippocampus complex (Takano et al., 2001). Hypoperfusion specific to AN-R has also been found in frontal areas, mainly the ACC (Naruo et al., 2001), and hyperperfusion specific to AN-BP in the right hemisphere, including inferior and superior prefrontal, and parietal regions (Naruo et al., 2000). Changes in regional cerebral blood flow (rCBF) at rest have been reported by some to normalise following weight restoration and remission (Kuruoglu et al., 1985), though others have found persistent changes (Frampton et al., 2011). One study reported that, in comparison with healthy controls, individuals with AN-R prior to treatment had lower cerebral blood flow in the ACC, right parietal lobe, insula and occipital lobes. Following treatment and weight gain, normalisation in a number of brain areas occurred, but decreased rCBF persisted in the ACC (Kojima et al., 2005). Another study by Matsumoto et al. (2006) found AN patients had increased rCBF post-treatment relative to pre-treatment in the dorsolateral prefrontal cortex (DLPFC), medial prefrontal cortex, anterior and posterior cingulate and precuneus. A more recent study by Komatsu et al. (2010), however, reported increased rCBF in the right posterior cingulate gyrus and bilateral parietal lobe following weight recovery. Differences in rCBF have also been reported in AN relative to different conditions. Prior to eating a meal, AN participants have been found to show significantly decreased rCBF in the left parietal cortex in comparison to controls. Post-meal, however, AN participants were found to not differ in rCBF relative to healthy controls or BN patients (Nozoe et al., 1995). Individuals with AN have also been found to show differences in rCBF when viewing stimuli of their own bodies. In a study by Beato-Fernández et al. (2009), healthy individuals, patients with BN and patients with AN viewed videos of themselves and landscapes. When viewing videos of one’s own body, increased rCBF of the left parietal and right superior frontal cortices was found in AN, whereas healthy controls showed decreased activation in these areas, suggesting a possible dysfunction in somatosensory integration in AN.
Glucose metabolism
Findings similar to those reported with the use of SPECT have also been found with PET. Individuals with AN have been shown to display global hypometabolism as well as relative hypometabolism of glucose in cortical regions, most significantly in the frontal and parietal cortices (Delvenne et al., 1995, 1997), which have been found to normalise with weight gain (Delvenne et al., 1996). The same authors also reported hypermetabolism in the inferior frontal cortex and basal ganglia when compared to controls, as well as increased glucose metabolism in the caudate and putamen when compared to participants with BN (Delvenne et al., 1999); whereas AN individuals in remission have been shown to display normal glucose metabolism in the brain (Delvenne et al., 1996; Frank et al., 2007; Herholz et al., 1987). Furthermore, individuals with AN have been shown to have greater regional cerebral blood flow within the bilateral medial temporal lobe in comparison to healthy controls (Gordon et al., 2001), an area heavily involved in memory processes (Squire and Zola-Morgan, 1991) and an area which has also been associated with increased blood flow in schizophrenia (Friston et al., 1992). Additionally, the study by Gordon et al. (2001) reported increased occipital and temporo-occipital cortex activity of AN patients relative to controls when viewing images of high- compared to low-calorie foods. These areas are related to the processing of visual information and increased activity of related visual processing areas has been found in individuals with specific phobias when presented with phobic stimuli (Wik et al., 1993).
Blood oxygen level dependent (BOLD) response
Changes in neural activity in response to different conditions that were initially explored with PET have typically been explored with functional magnetic resonance imaging (fMRI) since the technology became widely available. In the first fMRI study in AN, Ellison et al. (1998) presented individuals with images of high- and low-calorie drinks. The authors reported that the group of AN participants showed increased activity of the left insula, ACC and amygdala-hippocampal region in response to high- versus low-calorie drinks, relative to controls. In response to images of food and non-food items on the other hand, Joos et al. (2011) found increased right amygdala activity and decreased posterior middle cingulate cortex activity in AN-R compared to controls, whereas Kim et al. (2012) reported increased left anterior insula activity in AN, and significant interactions between the right insula and inferior frontal gyrus. However, it must be noted that the studies by Kim et al. (2012) and Joos et al. (2011) involved passive viewing of images. In a study undertaken by Brooks et al. (2011), the investigators asked participants to imagine they were eating the food in the images presented to them and were using the objects in the control images. Increased activity of the cerebellum, left visual cortex, right DLPFC and parietal lobe was reported in AN-R to food compared to non-food items, and in the bilateral cerebellum and supplementary motor area in AN-BP. However, this study only reported within-group comparisons for AN participants and did not present between-group comparisons with healthy controls. Utilising the same paradigm, a more recent study by this group, which reported results compared to healthy individuals, increased right visual cortex activity and reduced bilateral cerebellar vermis activity was reported in AN when thinking about eating food (Brooks et al., 2012). When the investigators examined AN-R and AN-BP separately in comparison to healthy controls, increased right visual cortex and DLPFC activity, and reduced left cerebellar vermis and right insular activity was found when thinking about eating food in AN-R. Similar results were reported for the AN-BP group, although no increase in right DLPFC activity was found. When comparing the two AN subgroups to one another, the AN-R group showed increased activity of the visual cortex, left parahippocampal gyrus and left ACC, suggesting a possible difference in the processing of food-related information between the two AN subgroups.
BOLD activity differences in response to images of food have also been found between patients currently ill with AN and weight-recovered patients. Holsen et al. (2012) presented ill AN, weight-recovered AN and healthy controls with images of food and non-food items before and after a meal. Pre-meal, the two patient groups showed reduced hypothalamus, amygdala and anterior insula activity when viewing images of high-calorie foods compared to controls. Additionally, the ill AN group also showed increased hippocampus and orbitofrontal cortex activity. Post-meal, the group with active illness persisted to show reduced amygdala and anterior insula activity, whereas the weight-recovered individuals did not. Other studies assessing AN participants in a hungry and satiated state have reported increased posterior cingulate (Gizewski et al., 2010), and inferior parietal and visual cortex activity (Santel et al., 2006) in a hungry state when viewing food images. When satiated, increased activity of the left insula to food images has been reported in AN in comparison to controls (Gizewski et al., 2010). In relation to a hungry state, however, increased occipital cortex activity has been reported in AN when viewing food images (Santel et al., 2006). Different neural responses in AN when hungry and satiated have also been investigated with the use of oral stimuli. In a task where participants were required to drink chocolate milk or water through a tube while undergoing fMRI, the group of AN-R participants showed increased right amygdala and left medial temporal gyrus activity relative to controls when drinking the flavoured milk (Vocks et al., 2011). Although groups did not differ in responses to the chocolate milk in the satiated condition, when comparing the hungry to the satiated condition the AN group displayed increased inferior temporal gyrus activity, which included the extrastriate body area, whereas the control group showed increased left insula activity. In a study by Wagner et al. (2007a), on the other hand, where participants were administered either a sucrose solution or water, the investigators reported reduced activation to both stimulus types in the insula, and ventral and dorsal striatum in AN. A similar finding was reported by Oberndorfer et al. (2013), who reported reduced right insula activity in AN to both sucrose and sucralose in a group of weight-recovered AN participants relative to healthy controls. Furthermore, differences in striatum and insula activity have also been reported in a study by Cowdrey et al. (2011). In this study the investigators administered a pleasant chocolate taste or an aversive unpleasant taste coupled with the presentation of chocolate, mouldy strawberries or a grey control image, or a grey screen coupled with a tasteless liquid which acted as a control condition, to healthy individuals and individuals weight-recovered from AN-R. The weight-recovered AN group was found to show increased ventral striatum and cingulate cortex activity to the taste of chocolate, and increased activity of the cingulate, medial prefrontal and occipital cortices in response to the sight of chocolate. When presented with the mouldy strawberry picture, however, no group differences were apparent, although increased activation of a number of areas was evident when the oral stimulus was administered alone or with the corresponding aversive image, including the ACC, DLPFC, lateral posterior insula, putamen and caudate.
Furthermore, in a series of studies undertaken by Uher and colleagues (Uher et al., 2003, 2004), the investigators presented participants with images of food and non-food items and asked them to think about how hungry the images made them; and emotional aversive and neutral images which required participants to think about how the images made them feel. In the food/non-food contrast, individuals with AN demonstrated more activity of the lingual gyrus and lower activity of the inferior parietal lobule and cerebellum when compared to controls (Uher et al., 2004); whereas individuals weight-recovered from AN showed increased prefrontal and ACC activity, and reduced inferior parietal activity in the same contrast compared to healthy individuals (Uher et al., 2003). Relative to individuals with a current diagnosis of AN-R, the weight-recovered group were found to show heightened activation of the dorsal ACC and the prefrontal cortex. In this earlier study, group differences were specific to food stimuli and no difference in activation was apparent for the emotional aversive stimuli for either AN-R or weight-recovered AN compared to controls. In the later study, however, which contained a larger sample size, AN participants were found to show reduced right cerebellar activity but increased left cerebellar activity when viewing the emotional aversive images compared to healthy individuals. Individuals with AN have also been found to show differences in brain activation when presented with negative words related to interpersonal relationships. Specifically, AN has been associated with increased left insula, and right superior temporal and inferior frontal gyri activation, relative to healthy controls (Miyake et al., 2012). In an earlier study, the same group investigated neural responses to negative body-image words (Miyake et al., 2010). AN-R was associated with more activity of the right amygdala and inferior parietal lobule, whereas AN-BP was associated with higher activity of the amygdala and left ventromedial prefrontal cortex, relative to a healthy comparison group. Responses to negative body-image words have also been investigated in AN, utilising an Emotional Stroop task consisting of fat, thin and neutral words, and words comprised of XXXXs (Redgrave et al., 2008). This study revealed increased activation at the junction of the left insula, and frontal and temporal lobes, and the left middle and medial frontal gyri to thin versus XXXX words in AN compared to controls; whereas the fat versus XXXX contrast revealed lower activity of the DLPFC and parietal areas in comparison to controls. These findings suggest that individuals with AN process positive and negative body image words differently to healthy individuals, with increased activation to thin words and decreased activation to fat words in areas related to executive function.
Furthermore, body image in AN has also been explored by examining responses to images of human bodies. A recent study involving passive viewing of body images and images of chairs, which acted as control condition, aimed to investigate the connectivity within the core network for body processing (Suchan et al., 2013). The findings of the study revealed a different network in AN during the processing of human bodies. In healthy controls, effective connectivity was found between the middle occipital gyrus and extrastriate body area, and the extrastriate body area and fusiform gyrus. In AN, however, effective connectivity was evident between the middle occipital gyrus and fusiform body area, and the fusiform body area and the extrastriate body area. Furthermore, reduced connectivity between the fusiform body area and extrastriate body area was found in the group of AN patients. Differences in the extrastriate body area and fusiform body area function between AN and healthy individuals have also been reported in a study involving the presentation of underweight, overweight and normal weight female bodies. Uher et al. (2005) reported reduced activation of the occipitotemporal cortex (including the extrastriate body area), the fusiform gyrus and the parietal cortex in a group of AN participants to all body shapes, relative to both BN and healthy controls. This task required participants to think about how acceptable the body or house (control image) would be for them. In a similar study utilising stimuli of computer-generated nude females depicting underweight, overweight and normal body weights, participants were asked to process the stimuli during two conditions: one in which they were required to imagine how it would feel to be the body shape presented, and the other where they needed to estimate the body weight of the stimuli, which acted as the control condition (Fladung et al., 2010). Greater activity of the ventral striatum to normal weight compared to underweight stimuli was evident in healthy controls, whereas individuals with AN were found to show greater activity to underweight stimuli in the same contrast. However, groups were not found to differ in response to overweight stimuli. In another study, Friederich et al. (2010) presented participants with images from magazines of either slim idealised female bodies or interior home designs in which participants were asked to compare their body shape or room design with the presented image. Group comparisons revealed that in response to the body images, individuals with AN showed decreased activity of the rostral ACC and greater activity of the right insula and premotor cortex. In a recent study by Suda et al. (2013), the investigators presented individuals with AN and healthy individuals with images of ‘body checking’, such as images of measuring leg width with a measuring tape and pinching skin folds, and neutral active images such as using a computer or writing. The authors reported increased right parietal cortex activity in the AN group to the body checking images compared to the neutral images, and lower activity of the anteromedial prefrontal cortex and right fusiform gyrus relative to controls.
Rather than displaying images of other people’s bodies, several studies have focused on responses to images of one’s own body. Sachdev et al. (2008) found that in comparison to a group of healthy participants, AN participants showed less activity of the frontal gyri, insula, precuneus and occipital regions when processing own body images, but did not differ when processing non-self body images. Vocks et al. (2010a), on the other hand, reported higher amygdala activity in AN compared to BN and controls when viewing another woman’s body, and reduced inferior parietal lobule activity when viewing own body images compared to controls alone. In a related study, the same authors presented AN patients with own body stimuli before and after undergoing body image therapy and found increased pre- to post-treatment activity in the right middle temporal gyrus (including the extrastriate body area); whereas decreased pre- to post-treatment activity in the left precuneus, posterior cingulate gyrus, fusiform gyrus, and right inferior and superior frontal gyri, parahippocampal gyrus, and bilateral inferior parietal lobule was reported in AN (Vocks et al., 2010b). Furthermore, other studies have not only presented individuals with images of their own body, but also distorted images of their own body. Castellini et al. (2013) asked participants to passively view images of their own body undistorted, their own body digitally distorted, and control of images of houses. The authors reported that in comparison to healthy controls, individuals with AN showed increased inferior frontal gyrus activity when presented with overweight stimuli relative to normal weight stimuli, and greater activity of the middle temporal gyrus in the underweight–normal weight contrast. In another study with similarly distorted images, participants were asked to either rate how much the image corresponded to their real body, or how much it represented their ideal body (Mohr et al., 2010). In the ideal body condition, increased activity of the insula and middle frontal gyrus was found for thin images in comparison to thin images of the real body condition. Additionally, the AN group were found to show decreased precuneus activity for all body sizes in the real body condition compared to controls, and to the heavier images in the real body condition relative to the ideal body condition in comparison to healthy individuals. Furthermore, an early fMRI study in AN undertaken by Seeger et al. (2002) presented distorted own body images of maximum subjective unacceptability to the participant, subjective unacceptability of another woman’s body and abstract images consisting of the participant’s own body. This study reported that AN participants showed increased activity of the right amygdala, fusiform gyrus and brainstem to distorted own body images compared to distorted images of other bodies and the abstract images. However, this study was only comprised of three individuals with AN and three comparison participants. In a follow-on study by the same group of investigators, the same task was performed in a larger sample and was found to lead to increased inferior parietal lobule and prefrontal cortex activity in the same comparison mentioned above (Wagner et al., 2003).
Although the great majority of fMRI studies have presented individuals with disorder-relevant stimuli such as bodies and food, other aspects of the disorder have also been investigated. A number of cognitive tasks have been performed while undergoing fMRI, including tasks of inhibition, working memory, visuospatial processing, cognitive flexibility and reward. During the completion of a stop-signal task, groups of AN participants and healthy controls demonstrated similar neural activity when the inhibitory demand was low. However, when the inhibitory demand was high, individuals with AN showed decreased prefrontal cortex activity in comparison to controls (Oberndorfer et al., 2011). However, during a number n-back task, a task assessing working memory, individuals with AN were reported to show increased activation of temporal and parietal areas compared to controls. A subgroup of the same AN participants were retested following seven months of treatment and weight restoration and were found to show reduced activity in the ACC, and frontal and parietal regions compared to the results from the original test session. At follow-up, the AN patients no longer differed from the control group (Castro-Fornieles et al., 2010). However, in a more recent study by Lao-Kaim et al. (2013), the authors reported no difference in performance or brain activity between groups of AN participants and healthy controls on a letter n-back task, suggesting that verbal working memory may be intact in AN. During a visuospatial processing task, on the other hand, in which participants were required to identify a target shape within more complicated figures, the AN group not only performed poorer on the task but also showed greater fusiform gyrus activity, whereas controls showed greater precuneus activity (Fonville et al., 2013). These findings suggest that unlike healthy individuals who utilise visuospatial search strategies to complete such tasks, individuals with AN may utilise strategies related to object recognition. During a behavioural response shifting task, on the other hand, where participants were required to respond to and classify shapes as targets, non-targets or standard shapes, Zastrow et al. (2009) described that individuals with AN showed decreased bilateral thalamus, rostral cingulate, sensorimotor region and cerebellum activity to target trials, independent of performance. The AN group also showed reduced activity of the dorsal ACC and ventral putamen in the correct shift target trials, and reduced activity in the rostral ACC and dorsal and middle striatum in the correct maintain target trials, relative to controls. Incorrect trials relative to correct trials, on the other hand, were associated with lower activity of the dorsal ACC, which extended to the medial frontal gyrus in AN. Together, the findings by Zastrow et al. (2009) indicate that during tasks of behavioural response shifting, individuals with AN may have altered fronto-striatal-thalamic function. Additionally, during the Wisconsin Card Sort Test, a task of behavioural response shifting and cognitive flexibility, individuals with AN are also found to show differences in neural response. In a study by Sato et al. (2013), the authors reported poorer performance on the task in AN, and lower right ventrolateral prefrontal cortex and bilateral parahippocampal cortex activity relative to healthy controls.
Differences in responses to stimuli related to reward have also been recently investigated in AN. In a study by Frank et al. (2012), participants performed a reward-conditioning task where they not only learned specific associations, but they would receive or be deprived of one of the two association stimuli, therefore causing an unexpected violation of these learned associations. When an unexpected stimulus was received, AN participants had greater right ventral putamen activation compared to controls, and greater left orbitofrontal cortex activity in comparison to a sample of obese participants and the healthy control group combined. When a stimulus was unexpectedly deprived, AN participants showed reduced activation of the bilateral putamen and left orbitofrontal cortex compared to the two comparison groups. Additionally, in a study presenting a simple monetary reward task to individuals weight-recovered from AN and a healthy comparison group, the healthy controls were found to show increased left anterior ventral striatum activity for wins than for losses, whereas the patient group did not differ and had similar responses to both conditions, suggesting a possible difficulty in differentiating positive and negative feedback in AN (Wagner et al., 2007b).
A range of other BOLD studies have been undertaken in AN. One study aiming to assess emotion processing in AN presented individuals with happy and fearful human faces (Cowdrey et al., 2012b). The study revealed no difference in neural response between groups. However, this result may be due to the fact that appropriate comparisons between conditions could not be made as a neutral control condition was not presented. A study investigating the processing of identity in AN, on the other hand, found reduced precuneus dorsal ACC and left middle frontal gyrus activity when presented with stimuli related to self-knowledge and perspective taking (McAdams and Krawczyk, 2012). Two recent studies have also looked at pain perception in AN. In the earlier study, Bär et al. (2012) administered a painful thermal stimulus to the right arm of participants and found that AN participants had an increased heat pain threshold and increased right pons activity in comparison to controls. The later study undertaken by Strigo et al. (2013) investigated pain processing and anticipation in weight-recovered AN. The authors reported increased DLPFC and decreased posterior insula activity during pain stimulation in the AN group. During pain anticipation, however, the AN group displayed greater right anterior insula, DLPFC and cingulate cortex activity anticipation, relative to controls, suggesting a potentially intensified stress response.
Recently, studies have begun to investigate functional connectivity in individuals with AN. During resting state scans of individuals weight-recovered from AN, increased temporal coherence has been reported between the default mode network functional connectivity map and the right precuneus and the DLPFC/inferior frontal gyrus (Cowdrey et al., 2012a). Decreased connectivity in the right middle frontal gyrus has also been reported for weight-recovered AN, and decreased connectivity of the left occipitotemporal junction within the ventral visual network (Favaro et al., 2013). Favaro et al. (2013) also reported increased connectivity in the left superior parietal cortex within the somatosensory network for individuals with AN. In another study, the same authors described increased prefrontal functional connectivity in individuals with AN who were Met-Met carriers of the COMT gene, compared to AN patients who were Val carriers of the gene (Favaro et al., 2012). In a more recent study by Amianto et al. (2013), the investigators reported intrinsic connectivity networks in the cerebellum in AN which were more restricted to vermian areas, and showed greater medial and less lateral connectivity in the cerebellum relative to healthy individuals. The AN group also demonstrated less cerebellar connectivity with the parietal lobe, but more cerebellar connectivity with the posterior cingulate cortex, bilateral temporal pole and insula. Furthermore, the anterior insula showed more connectivity in AN, in contrast to individuals with BN who showed hyperconnectivity with the posterior portion of the insula.
Conclusion
The objective of this review was to provide a summary of the current literature on the neurobiology of AN. It is apparent that structural and functional changes occur during the ill state of AN, some of which have been found to reverse with weight restoration, and others which persist following weight recovery. A number of structural brain differences have been reported in AN. The enlargement of cortical sulci and ventricles is commonly reported, as are deficits in grey and white matter volumes. Although reduced grey matter volumes are frequently found to persist in weight recovery, the findings regarding white matter volumes in both weight-recovered and ill AN patients remain inconsistent. Recent DTI studies have suggested poorer white matter integrity in a number of brain areas in AN, although a limited number of studies utilising this technique have thus far been undertaken. Grey matter deficits in specific brain areas have also been reported, particularly areas of the limbic system including the amygdala, hippocampus and cingulate cortex, areas heavily involved in emotion (LeDoux, 2000). Reduced grey matter volumes have also frequently been reported in the putamen, a structure of the basal ganglia involved in learning, as well as the regulation of DA (Packard and Knowlton, 2002).
Deficits in DA function have been reported in AN, specifically, reduced levels of CSF HVA and altered DA receptor function. DA plays an important role in eating behaviours, motivation, reinforcing behaviour and reward (Bassareo and Di Chiara, 1999; Holroyd and Coles, 2002; Phillips et al., 2008; Roitman et al., 2004; Schultz et al., 1997; Volkow et al., 2003; Wise, 2004); behaviours which are also found to be disturbed in AN (e.g. Fladung et al., 2010; Scheurink et al., 2010; Wagner et al., 2007b; Watson et al., 2010). Similarly, deficits in 5-HT function have also been reported in AN, namely, increased 5-HT1A and decreased 5-HT2A binding in ill and weight-recovered AN, and decreased CSF 5-HIAA in ill patients but increased levels in weight-recovered patients. 5-HT plays a role in a number of symptoms and behaviours which are evident in the condition, including obsessional behaviours, anxiety, impulse control, inhibition, attention and mood (Buhot, 1997; Higley and Linnoila, 1997; Lucki, 1998; Soubrie, 1986). It is also involved in regulating feeding behaviour such as playing an inhibitory role in eating, regulating meal size and controlling eating rate (Blundell, 1984; Leibowitz and Alexander, 1998; Simansky, 1995).
SPECT studies have revealed that individuals with AN show hypoperfusion in several brain areas, although whether these rCBF differences persist into weight recovery remains unclear. Similarly, PET studies have demonstrated global hypometabolism in individuals ill with AN, but this change in glucose metabolism is no longer evident in weight-recovered individuals. Functional differences in the brains of these individuals have however tended to be investigated with more sophisticated neuroimaging techniques such as fMRI.
Recent fMRI studies have provided us with a better understanding of the areas of the brain potentially responsible for the onset or maintenance of the illness. The function of the insular and cingulate cortices, in particular, have been regularly reported to differ in AN. The cingulate cortex is particularly involved in motivation, goal-directed behaviour and emotional processes (Devinsky et al., 1995); whereas the insula also plays a key role in emotional processes (Jones et al., 2010), particularly in the processing of disgust (Wicker et al., 2003), an emotion which AN patients have been reported to have particular difficulty recognising (Pollatos et al., 2008).
Due to inconsistencies across study procedures, findings are not always consistent and can be difficult to interpret. A major issue in all AN research is that it remains unclear whether changes occur as a result of the condition and are due to the effects of starvation, or whether they are innate differences that contribute to the development of the illness. This is a very challenging issue to address as it would be difficult, if not impossible, to test individuals before the onset of illness symptoms. This is one of the reasons why recovered AN patients are often researched. However, the findings observed at recovery may be state effects, reflecting damage due to starvation, rather than trait effects of AN. Additionally, the length of time in which patients are weight-recovered varies between studies as a standard minimum timeframe for what constitutes someone who is in recovery does not exist. Furthermore, patients with varying illness severity and duration are often included in the same analysis. Although particularly difficult to control due to small patient numbers and often limited information regarding illness duration, more systematic recruitment procedures would provide a better understanding of how these factors may contribute to neurobiological findings. Furthermore, if information is gathered about illness duration and severity, correlational analyses could be performed with neurobiological data.
The results of this review suggest a number of structural and functional deficits in the brains of individuals with AN as revealed with the use of a number of neuroimaging modalities. It is important to note that some modalities were not included in this review, such as electroencephalography (EEG), as they were beyond the scope of this paper. However, a number of recent papers have suggested EEG and event-related potential differences in AN (e.g. Hatch et al., 2010, 2011; for a review see Jáuregui-Lobera, 2012), and further studies utilising more sophisticated neuroimaging techniques such as magnetoencephalography will provide further information regarding differences in the time course of activity in AN.
The findings of this review have important implications for the treatment of AN, however, more systematic research with larger sample sizes is required. Of great benefit would also be research which utilises tasks that are known to employ the areas which are disturbed in AN, such as emotion processing tasks which employ areas such as the insula and limbic system, as well as saccadic eye movement tasks which involve the insula, cingulate cortex, basal ganglia, DLPFC and striatum, among other areas. With further investigation into the neurobiology of AN, a better understanding of the mechanisms involved in the development and maintenance of the illness will be established which will have the potential to provide new direction for the development of effective treatments.
Footnotes
Acknowledgements
The authors would like to thank Larry Abel, PhD, for his contribution to the manuscript.
Funding
This research was supported by a grant received from the Jack Brockhoff Foundation (3410), a donation from the Dick and Pip Smith Foundation, and an Australian Postgraduate Award.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
