Abstract
Background:
First described in 1955, night eating syndrome refers to an abnormal eating behavior clinically defined by the presence of evening hyperphagia (>25% of daily caloric intake) and/or nocturnal awaking with food ingestion occurring ⩾ 2 times per week.
Aims:
Although the syndrome is frequently comorbid with obesity, metabolic and psychiatric disorders, its etiopathogenesis, diagnosis, assessment and treatment still remain not fully understood.
Methods:
This review was conducted according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines; PubMed database was searched until 31 October 2020, using the key terms: ‘Night Eating Syndrome’ AND ‘complications’ OR ‘diagnosis’ OR ‘drug therapy’ OR ‘epidemiology’ OR ‘etiology’ OR ‘physiology’ OR ‘physiopathology’ OR ‘psychology’ OR ‘therapy’.
Results:
From a total of 239 citations, 120 studies assessing night eating syndrome met the inclusion criteria to be included in the review.
Conclusion:
The inclusion of night eating syndrome into the Diagnostic and Statistical Manual of Mental Disorders-5 ‘Other Specified Feeding or Eating Disorders’ category should drive the attention of clinician and researchers toward this syndrome that is still defined by evolving diagnostic criteria. The correct identification and assessment of NES could facilitate the detection and the diagnosis of this disorder, whose bio-psycho-social roots support its multifactorial nature. The significant rates of comorbid illnesses associated with NES and the overlapping symptoms with other eating disorders require a focused clinical attention. Treatment options for night eating syndrome include both pharmacological (selective serotonin reuptake inhibitors, topiramate and melatonergic drugs) and non-pharmachological approaches; the combination of such strategies within a multidisciplinary approach should be addressed in future, well-sized and long-term studies.
Introduction
Night eating syndrome (NES) is a new clinical syndrome described in the ‘Other Specified Feeding or Eating Disorders’ (OSFED) section of the Diagnostic and Statistical Manual of Mental Disorders, 5th edition—DSM-5—(American Psychiatric Association, 2013). It is mainly characterized by morning anorexia, evening hyperphagia (the consumption of > 25% of total daily food intake after the evening meal [EH]) and nocturnal ingestions (>2/week; NI) in subjects who concomitantly exhibit sleep and mood alterations. The syndrome is thought to be a combination of a sleep disorder, a mood disorder (Lundgren et al., 2008a, 2012a) and altered eating behaviors, such as emotional eating, food addiction (Baldofski et al., 2015) and delayed circadian food intake (Herpertz-Dahlmann, 2009). Stunkard et al. (1955) were the first to describe this syndrome as a stress-related eating disorder in obese outpatients; further studies focused on NES among obese patients (Cerú-Björk et al., 2001; Gluck et al., 2001; Stunkard et al., 1996), since the disorder is closely associated with overweight and obesity, higher general psychopathology levels (Cleator et al., 2012; Gallant et al., 2012), negative affects (Colles et al., 2007; Sevincer et al., 2016; Striegel-Moore et al., 2010) and low self-esteem (Gluck et al., 2001; Striegel-Moore et al., 2010). Moreover, negative health outcomes, such as substance use disorder (Lundgren et al., 2006b), and functional impairment (Sevincer et al., 2016; Striegel-Moore et al., 2010; Stunkard and Allison, 2003) have been described. In 2008, during the First Night Eating Symposium, diagnostic criteria were discussed, and specific reference parameters were established: a diagnosis of NES is considered if there is a consumption of at least 25% of daily caloric intake after the evening meal and/or evening awakenings with ingestions at least twice per week; furthermore, the subject must be aware of his nocturnal ingestions and must experience associated distress or impairment in functioning, with signs and symptoms lasting for at least 3 months (Allison et al., 2010b).
The present review is aimed at summarizing the available evidence from the literature on this clinical entity that, for its multiple physical, metabolic and psychopathological implications deserves more clinical awareness and further insight on its etiopathogenesis and potential treatment strategies.
Methods
Search processes
This review was conducted according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines (Moher, 2009). PubMed database was searched until 31 October 2020, using the following key terms: ‘Night Eating Syndrome’ AND ‘complications’ OR ‘diagnosis’ OR ‘drug therapy’ OR ‘epidemiology’ OR ‘etiology’ OR ‘physiology’ OR ‘physiopathology’ OR ‘psychology’ OR ‘therapy’. Additional articles have been identified by detecting similar articles and articles with titles containing our search terms. Articles have been selected by title and abstract; the entire article was read if title/abstract concerned the NES. References of the selected articles were also examined in order to identify additional studies that met the inclusion criteria.
Study selection
Articles were included in the review according to the following inclusion criteria: English language, publication in peer reviewed journals, articles about studies performed on humans on NES. Articles were excluded by title, abstract or full text for irrelevance to the topic in question. Further exclusion criteria were articles not written in English language, unpublished dissertations and theses, and other non-peer-reviewed material.
Data extraction
Two authors (L.C., G.T.) performed the initial search, independently reviewed and selected the references based on the inclusion and exclusion criteria. The results were subsequently re-evaluated by the auditors and the salient results were shown. After having discarded duplicate articles, data derived from our research of articles included study author names, publication dates, study designs (i.e. open-label uncontrolled and randomized controlled trial), sample (case and control group), main efficacy results (response and remission rate, mean change in outcome measure).
Results
Study characteristics
Figure 1 summarizes the flow chart of articles selected for the review. The search of PubMed database provided a total of 177 citations; 62 additional studies that met the criteria for inclusion were identified by checking the references of selected papers. After adjusting for duplicates, 198 records were screened. Of these, 79 studies were excluded because were irrelevant to the topic. After the screening, 119 studies assessing NES met the inclusion criteria and were included in the systematic review.
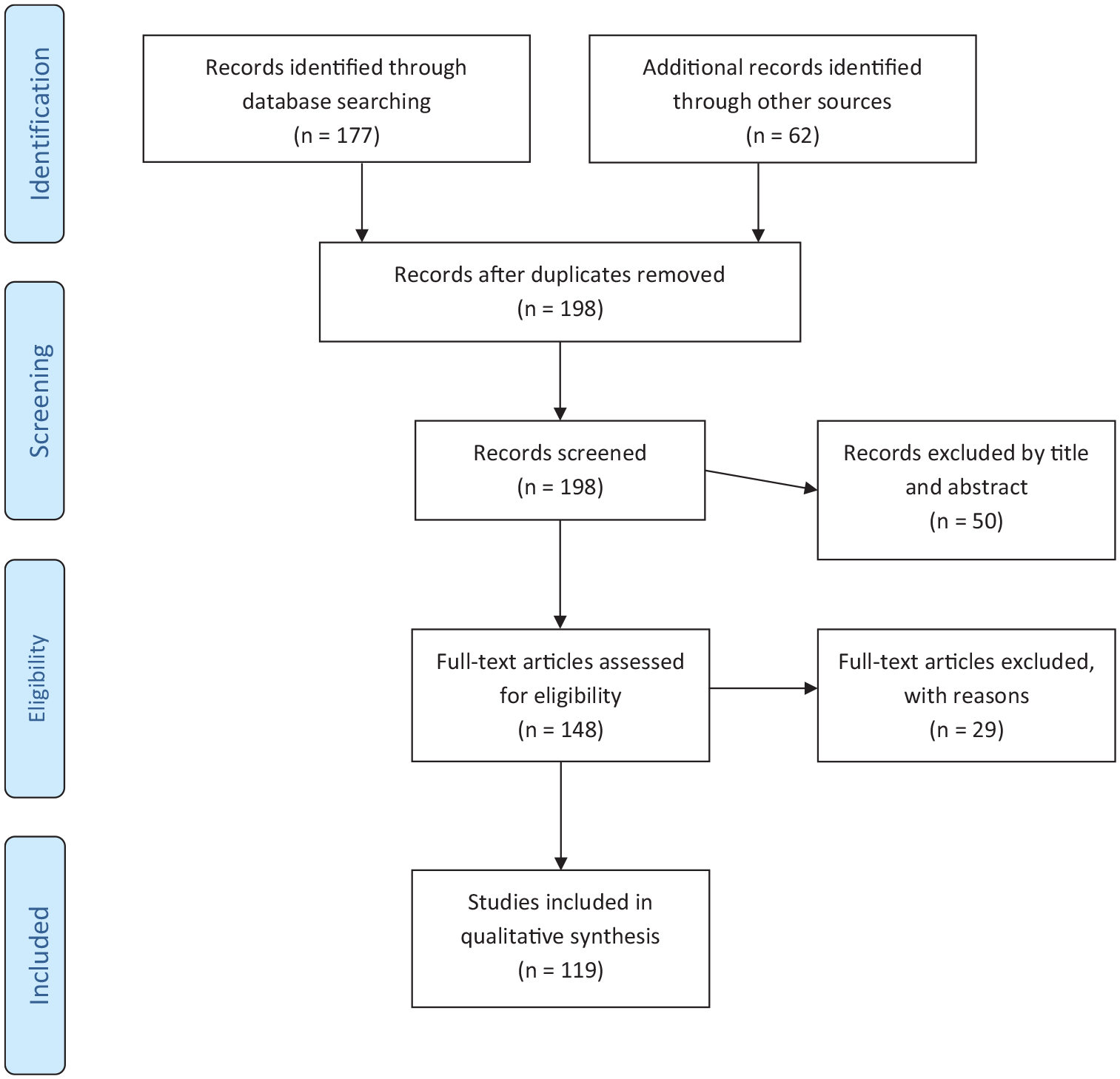
Flow diagram of the literature selection process.
Diagnosis, clinical features and assessment measures
According to the First International Night Eating Symposium held in April 2008, in Minneapolis, aimed at modifying previous diagnostic criteria used since the very first description of the syndrome (Stunkard et al., 1955), NES is described as a circadian disorder characterized by a delayed pattern of food intake, and it is diagnosed if one or both of the Allison et al’.s (2010b) core criteria are met (Table 1).
Proposed research diagnostic criteria for NES (Allison et al., 2010b).

Criterion A, concerning the consumption of at least 25% of daily caloric intake after dinner (evening hyperphagia) is unavoidably required for diagnosing the syndrome. Interpreting ‘after dinner’ can be controversial: in several studies, 5 p.m. has been set as the reference time for dinner, thus the first meal consumed after 5 p.m. has been considered after dinner. In the absence of the main meal, the first snack consumed during this time has been considered the evening meal; moreover, if no food is consumed by 8 p.m., any food consumed after that time will be considered ‘after dinner’ (Allison et al., 2010b). However, there is the need to introduce culture-specific norms and other variables in this fixed definition since in many countries (i.e.: Mediterranean cultures) the evening meal is about 8 p.m.; also, dinner time is often delayed according to lifestyle and work schedules. In these cases, if no other criterion is met, a diagnosis of NES is not justified.
Birketvedt et al. (1999) drew up the provisional criteria of NES without specifying the number of episodes of nocturnal ingestions for diagnosing the disorder, whereas other researchers (Lundgren et al., 2006a, 2008c; O’Reardon et al., 2005; Stunkard et al., 2006) considered three nocturnal ingestions per week in different reports. The NES Working Group proposed the frequency of two nocturnal ingestions per week, although other studies are necessary to establish if the diagnostic threshold should be at 3, 2 or 1 nocturnal ingestions per week. The criterion B, concerning the level of awareness, is crucial in the differential diagnosis between NES and sleep-related eating disorder (SRED) patients. The formers are conscious of their behavior and remember their ingestions, whereas SRED patients generally eat in a sleepwalking state, ingesting non-food or strange objects and usually they do not recall the eating episodes.
Criterion C includes the five core descriptors of NES and at least three of them should be present for diagnosing the disorder. The descriptor C1 refers to morning anorexia, which refers to lack of appetite in the morning or skipping breakfast most days of the week (Gluck et al., 2001), such condition is common for NES subjects, but it is not indispensable for the diagnosis. The descriptor C2 consists in a strong desire/urge to eat after dinner and before falling asleep and/or upon awakening at night from sleep. The amount of the ingested food is not excessive, usually subjects crave for specific foods, and anxiety and agitation remit after the nocturnal feeding (Allison et al., 2004). The descriptor C3 is focused on sleep disruption: sleep onset and maintenance insomnia. Although it is not always present, a person may wake up several times during the night and eat, but may falling asleep without problems (Tzischinsky and Latzer, 2004). On the other hand, an individual may eat continually in the evening in order to fall asleep and then may not have awakenings until morning. The descriptor C4 refers to cognition and concerns the belief that it is necessary to eat in order to falling asleep; in several Authors’ opinion, this descriptor is central for differentiating NES from binge eating/obesity, since NES patients, differently from binge eaters and obese people, hold the belief that good sleep is dependent on night eating which is capable to relieve their anxiety and/or insomnia (Vinai et al., 2014). The presence of depressed or lowered mood (C5) in NES patients is supported by different studies that evidenced a worsening of mood with the progression of the day (Allison et al., 2005b; Birketvedt et al., 1999; de Zwaan et al., 2006; Stunkard et al., 1955; Tzischinsky and Latzer, 2004). However, depressed mood has not been consistently found in the NES clinical population, thus it has been considered as a possible descriptor (Allison et al., 2007b). Distress, such as feelings of shame, guilt, and fear of weight gain related to nocturnal eating, impairment in functioning, mainly caused by daytime sleepiness or tiredness, and duration, established at 3 months, are defined by Criteria D and E, whereas Criterion F excludes other potential psychiatric/medical disorders or side effects of medications that can provoke NES disorder. Starting from the basis of the proposed criteria, it seems plausible to identify different subtypes of the disorder corresponding to diverse clinical phenotypes. A recent polysomnographic study on 20 outpatients (13 women and 7 men) supported the existence of at least two different subtypes of NES: the Evening Hyperphagia (EH) and the Nocturnal Ingestions (NI) subgroups (Loddo et al., 2019). The EH patients used to eat between dinner and sleep onset, although they could also eat during the night, whereas the NI subjects ate exclusively during an awakening from sleep. The two clinical typologies also differed in video-polysomnographic (VPSG) features, with EH pattern showing the characteristics of a sleep-onset and sleep-maintenance insomnia, and the NI pattern those of an arousal disorder associated with goal-directed and compulsive behaviors with small latency from awakenings. Furthermore, NI patients were younger, with a shorter disease duration, and not always obese. In both subgroups sleep macrostructure, stages and phases were substantially preserved.
The severity of NES can be assessed through a questionnaire, the Night Eating Questionnaire—NEQ (Allison et al., 2008). The first, unpublished version of the NEQ consisted of nine items with a 4-point Likert-type scale. The items measured psychological and behavioral symptoms on NES: morning anorexia, evening hyperphagia, initial insomnia, mid-phase insomnia, nocturnal ingestions and mood. Over the years, the accumulated knowledge on NES (Gluck et al., 2001: 38; Marshall et al., 2004: 116; Napolitano et al., 2001; O’Reardon et al., 2004a) has led to the revision of the scale, whose items were converted to a 5-point scale; moreover, six additional items were also provided, one of them intentionally added to assess the degree of awareness of nocturnal eating episodes, a differential feature between NES and SRED (Schenck and Mahowald, 1994). The actual version of NEQ is a 14-item questionnaire that assesses morning hunger and timing of first food consumption (2 items), food craving and control over eating behavior before bedtime (2 items) and during nocturnal awakenings (2 items), amount of food consumed after dinner (1 item), initial insomnia (1 item), frequency of nocturnal awakenings and ingestion of food (3 items), mood disturbance (2 items) and awareness of nocturnal eating episodes (1 item). A score of 25 or higher is suggestive of NES with a positive predictive value (PPV) of 40.7%; a score of 30 or higher is a strong indicator of NES, with a PPV of 72.7%. One of the limitations of the questionnaire was the lack of items aimed at assessing the degree of the perceived distress caused by night eating, and the degree of daily functional impairment. For this reason, other 2 items were added at the end of the questionnaire, although they are not included in the final score (0–52).
Another assessment tool is the Night Eating Syndrome History and Inventory (NESHI; Lundgren et al., 2012b), a semi-structured interview assessing a typical 24-hours food intake. The interview includes NEQ items but also gathers more information on average weekly frequency of nocturnal ingestions, and investigates sleeping routine, mood symptoms and life stressors, weight and diet history, medical history about NES symptoms and previous treatment strategies for the same disorder (O’Reardon et al., 2004a). The Night Eating Diagnostic Questionnaire (NEDQ; Lundgren et al., 2012b) is a self-report instrument assessing the presence and frequency of night eating-related symptoms and behaviors and providing scores that classify subjects as ‘Non-NE’ or ‘NES’ and, when NES is present, it can be further defined in terms of severity as ‘mild’, ‘moderate’ and ‘full’ NES. The Night Eating Symptom Scale (NESS; Lundgren et al., 2012b) also derives from the NEQ, with several differences. Items are not measured on a 0–4 Likert-type scale ratings, since each item assessing the frequency of awakenings and nocturnal ingestions requires a specific number. Furthermore, the time frame to assess symptoms focuses on symptoms in the previous week; this feature offers the possibility of evaluating symptoms changes after treatment trials.
Prevalence and epidemiology
NES equally affects men and women (Aronoff et al., 2001; Colles et al., 2007) as well as in individuals of all age group or ethnicity (Striegel-Moore et al., 2006). Estimates of the prevalence of NES varies across different studies and samples, ranging from 0.5% to 1.5% in the general population up to 55% in patients undergoing bariatric surgery for severe obesity (Adami et al., 1999, 2006; Colles and Dixon, 2006; de Zwaan et al., 2015;; Gallant et al., 2012; Greeno et al., 1995; Hsu et al., 1996; Powers et al., 1999; Rand et al., 1997; Rand and Kuldau, 1986; Striegel-Moore et al., 2005). Regarding general population, Striegel-Moore et al. (2006) found that evening hyperphagia was higher among young adults than any other age group. In college and university students, the most studied populations, evening hyperphagia was related with high stress (Cooley et al., 2007), sleep problems (Lund et al., 2010) and irregular eating (Delinsky and Wilson, 2008), with rates ranging from 2.8% to 9.5% (He et al., 2018; Sevincer et al., 2016). When the proposed diagnostic criteria (Allison et al., 2010b) were used, the prevalence of NES in university samples was 5.7% (Nolan and Geliebter, 2012); in this study, after controlling for the presence of Binge Eating (BE), that affects approximately one third of NES individuals, the prevalence of ‘pure’ NES fell to 2.9%, an almost doubled percentage compared with the prevalence rate of 1.3% found in a population-based young adult sample evaluated by the same diagnostic criteria and assessment methods (Fischer et al., 2012). These data suggest that NES may be more common in student populations. In overweight and obese populations, including patients attending obesity clinics, the prevalence was 6–16% (Calugi et al., 2005; Cerú-Björk et al., 2001; Gluck et al., 2001; Kucukgoncu et al., 2015; McCuen-Wurst et al., 2018; Stunkard et al., 1996; Vander Wal, 2012). To date, several studies evaluated the prevalence of NES in psychiatric outpatients, reporting rates ranging from 12.3% to 22.4% (Saraçlı et al., 2015); prevalence rates of 5.8% and up to 52% were found in patients with sleep and eating disorders, respectively (Lundgren et al., 2011; Manni et al., 1997). The high variability in NES prevalence rates found in general and clinical populations (Table 2) probably reflects differences in the conceptual definitions of the syndrome, in the diagnostic criteria applied, with studies that examined only several specific NES dimensions, such as evening hyperphagia, and others that looked at the full syndrome as defined by the proposed diagnostic criteria; accordingly, further research is needed for reaching more accurate estimates on NES distribution, incidence and prevalence.
Epidemiology of NES.
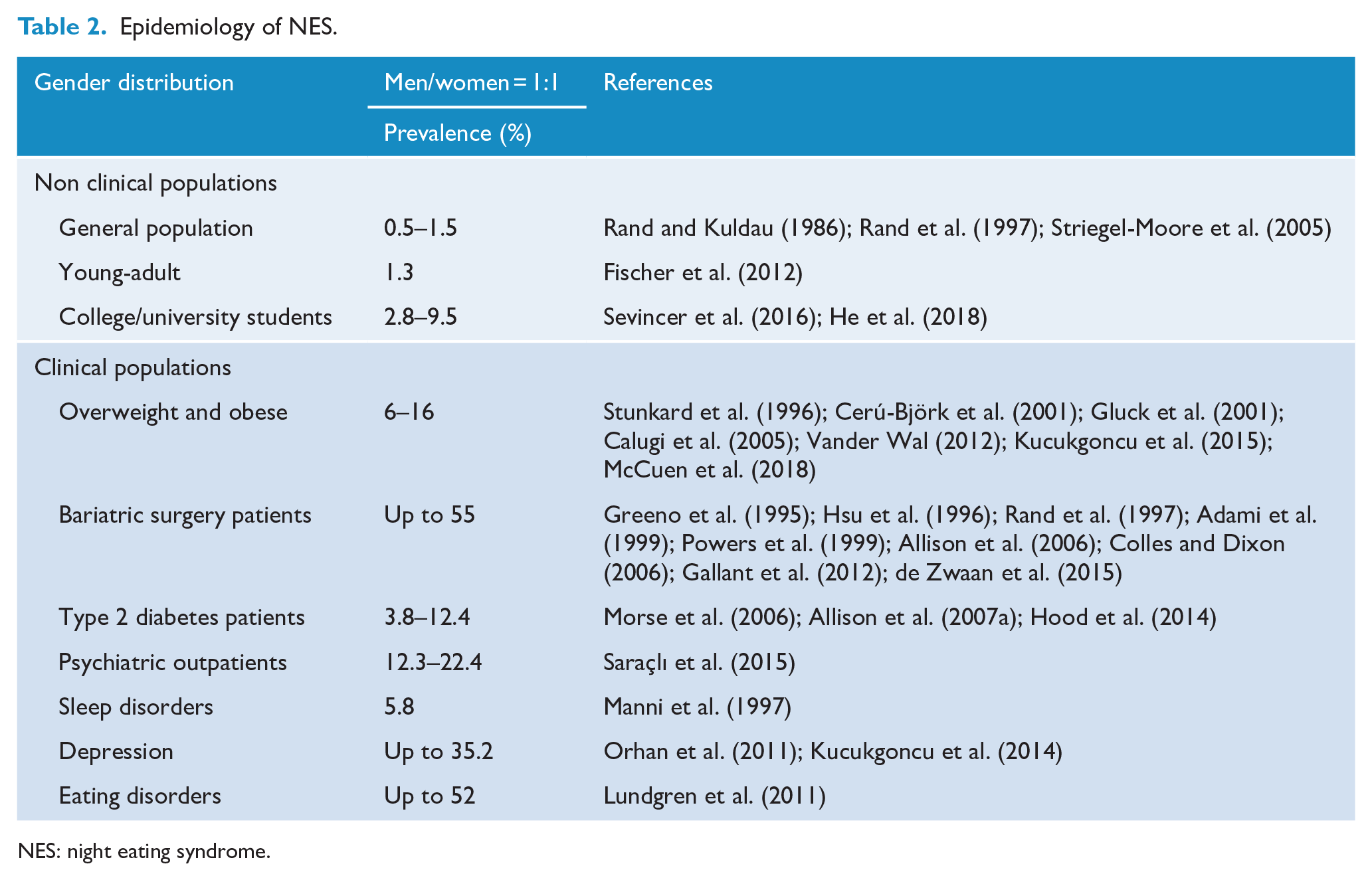
NES: night eating syndrome.
Risk factors
The etiology of NES is presumably multifactorial, with biological, psychological and social factors influencing the development and maintenance of the syndrome (Pollack and Lundgren, 2014). Table 3 summarizes the main theories about the predisposing and risk factors associated with the onset of NES.
Risk factors for NES.

NES: Night eating syndrome; SERT: serotonin transporter; CRH: corticotropin releasing hormone.
There is limited evidence on the role of genetic factors, as highlighted by few studies; not different from obesity, night eating behaviors aggregate in families: the first-degree relatives of NES patients were 4.9 times more likely than the first-degree relatives of unaffected controls to meet criteria for NES (Lundgren et al., 2006a). Data from the Swedish Twin Register at the Karolinska Institute, Stockholm on a total sample of 11,604 same-sex twins has examined shared and unique genetic and environmental influences comparing binge eating (prevalence in females = 5.7% vs prevalence in males = 0.7%) and night eating (reported in 3.4% of females and 4.3% of males) disorders. Results showed that heritability estimates for NES were moderate for both genders and lower than that observed with binge eating; moreover, the genetic correlation of 0.66 found in the study disclosed the substantial overlap in those genetic factors influencing the liability to both disorders (Root et al., 2010). It has also been suggested that genetic factors related with NES may be included in a genetic pool contributing in conferring a generic vulnerability to stress; this is supported by the observation that NES onset is often coincident with stressful periods, and by the confirmation of higher cortisol levels in night eaters compared with controls (Birketvedt et al., 1999).
Regarding other biological factors, several researches focused on the serotonergic system, starting by the clinical observation that symptoms of NES decreased after the administration of sertraline and other selective serotonin reuptake inhibitors (SSRIs), that presumably act by restoring a more regular circadian rhythm of food intake through effects on the suprachiasmatic nucleus that receives receive serotonergic inputs from the dorsal raphe nuclei of the brain stem (O’Reardon et al., 2004b; Stunkard et al., 2006). A disrupted serotonin system influences the synchronization of circadian rhythms of sleep and eating; accordingly, the administration of SSRIs to NES people resulted in restoring serotonin availability, and in re-establishing a normal circadian rhythm of food intake (Stunkard et al., 2009b). Beyond serotonin availability, the potential role of the SERotonin Transporter (SERT) in NES has been investigated in single photon emission computed tomography (SPECT) studies; results showed that NES patients had higher levels of SERT uptake in the mid-brain, right temporal lobe and left temporal lobe than patients with major depressive disorder (Lundgren et al., 2009) and higher mid-brain SERT uptake than healthy controls (Lundgren et al., 2008b). Based on those results, it was supposed that decreased post-synaptic serotonin availability in night eaters was caused by higher SERT levels.
Several imaging studies about obesity and eating behaviors showed an altered brain functioning in key brain regions associated with taste, reward sensitivity, self-regulation and inhibition and sleep. It is possible that similar processes are involved in the development and/or maintenance of NES. Particularly, NES subjects are lacking in cognitive and behavioral self-regulation and show an increased reward response to food cues in the evening and at night involving the same neural pathways of obesity and sleep disruption (Pollack and Lundgren, 2014). To the best of our knowledge, only one preliminary, functional magnetic resonance imaging (fMRI) study (Lundgren et al., 2013) has investigated brain activation in response to food cues in obese subjects with and without evening hyperphagia (EH), one of the core symptoms of NES, showing different patterns of brain activation between groups. The EH subjects had decreased activation pre- to post-meal for food images of the right inferior frontal gyrus, an area involved in inhibitory processing, and increased activation pre- to post-meal when viewing neutral images, whereas the control group showed an opposite pattern. No group differences were found in limbic regions. Based on these results, it has been hypothesized that people with EH have a reduced mid-day inhibitory processing in response to food cues, thus less motivation to eat at this time of day, compared to controls (Lundgren et al., 2013). Other studies investigated circadian neuroendocrine patterns of subjects with NES. Differences in plasma melatonin, plasma cortisol, plasma insulin, blood glucose and plasma leptin levels between NES subjects and controls have been demonstrated (Birketvedt et al., 1999). Among night eaters, plasma melatonin levels at night (from 10 PM to 6 AM) and rise in leptine levels (12 AM to 6 AM) were lower (p < 0.001), whereas plasma cortisol levels (from 8 AM to 2 AM) were higher than those of the control subjects (p = 0.002); finally, the preprandial and postprandial blood glucose and plasma insulin levels did not differ between night eaters and controls. The reduction of the nocturnal rise in leptin and melatonin may be caused by the increased levels of corticotropin releasing hormone (CRH) (Heiman et al., 1997; Kellner et al., 1997; Stratakos, 1995). This finding supports the association between NES, stressful events and heightened cortisol levels. However, such endocrinological profiles have not been thoroughly found; Allison et al. (2005a) did not find differences between NES patients and controls in leptin, melatonin or cortisol levels, whereas insulin (p < 0.001) and glucose levels (p = 0.07) were higher and ghrelin levels, measured in the early morning hours, were significantly lower among NES subjects than in controls (p = 0.003). According to the results of this study focused on appetite-regulating hormones, the Authors concluded that NES subjects have different neuroendocrine profiles from controls as a consequence of increased nocturnal eating. Within the research field of neuroendocrine studies on NES, to the best of our knowledge, there is only one recent study that has investigated the possible influence of pregnancy on NES and the relationship among NES and metabolic variations; pregnant women showed an increased proneness to night eating, which was associated with augmented insulin resistance. Thus, pregnancy can lead to modifications in eating attitudes even in women without previous eating disorders (Deniz et al., 2019).
Although few studies have investigated patients’ subjective experiences (Lundgren et al., 2012a), it should be recognized that several emotional and environmental factors leading to psychological distress can significantly influence the strong urge toward night eating experienced by NES subjects (He et al., 2018; Sevincer et al., 2016; Striegel-Moore et al., 2010). Moreover, during night binging, the tendency to choose foods with a high caloric amount could be merely explained by the feeling of comfort provided by this category of foods during either temporary and enduring states of emotional distress, such as boredom, anxiety, depression, and low self-esteem (Gluck et al., 2001); within this framework, the onset of NES may be a detector of psychological distress. Regarding traumatic issues, childhood maltreatment has been related with both eating disorders and obesity; BED and NES subjects were more likely to have an history of early trauma when compared with non-treatment-seeking overweight and obese controls, with a slight difference across groups, since emotional neglect was prevalent in BED subjects, whereas the NES group reported higher rates of physical neglect (Allison et al., 2007b).
Taken together, genetics, pharmacologic, neuroimaging, neuroendocrine and psychological data offer scientific support to the bio-psycho-social model of NES, providing a pathophysiological framework based on the interaction of genetic and epigenetic factors, including stress and psychosocial triggers.
Comorbidity: obesity and metabolic disorders
Generally, NES implies weight gain as a result of the excessive caloric intake at night. Thus, the syndrome is considered a risk factor for overweight and body mass index (BMI) increase, potentially contributing to obesity development, maintaining and worsening (Cleator et al., 2012; Gallant et al., 2012; Gluck et al., 2017; Meule et al., 2014; Morse et al., 2006). Beyond overweight and obesity, NES alone or comorbid with BED resulted associated with Type II Diabetes; when present, NES is related with poorer glycemic control, increased glycemic variability, and with diabetes-related complications, although studies on long-term outcomes are still lacking (Abbott et al., 2018; Gallant et al., 2014; Hood et al., 2014). Although not all subjects with a NES diagnosis are obese or overweight, the syndrome is more common in the obese population, with rates of prevalence going from 6% to 16% (Kucukgoncu et al., 2015; McCuen-Wurst et al., 2018; Vander Wal, 2012). When present, NES symptoms usually preceded obesity in 40–52% of patients (de Zwaan et al., 2006; Lundgren et al., 2006a; Marshall et al., 2004; Tholin et al., 2009). A longitudinal study covering a period of about three years showed that obese subjects who engaged in excessive evening and nocturnal food intake gained more weight than those who did not engage in night-time eating (6.2 vs 1.7 kg) (Gluck et al., 2008). Nocturnal ingestion of food has also been associated with a greater 6 year weight gain (4.3 kg) in obese women who consumed food during nighttime compared with obese women who did not (Andersen et al., 2004).
Findings concerning the relationship between NES and BMI provided conflicting results (McCuen-Wurst et al., 2018), with several studies reporting the absence of significant relationships between NES and BMI (He et al., 2018; Nolan and Geliebter, 2012; Striegel-Moore et al., 2006), whereas other studies supported the association between NES and higher BMI (Colles et al., 2007; Gallant et al., 2014, 2015; Meule et al., 2014; Uher et al., 2013). It has been hypothesized that the mixed findings on the relationship between NES and BMI are probably due to the use of different measurement methods and to the involvement of several moderators that have not been systemically investigated in the available studies, such as different age ranges, emotional and/or social eating, socioeconomic status, symptom variability, and sub-clinical NES forms (Bruzas and Allison, 2019). Regarding night eating severity and BMI, NES was positively associated with higher BMI in those aged between 31 and 60 years, but not in younger or older participants in whom this association tended to disappear (Meule et al., 2014). A longitudinal association between night-eating symptoms and anthropometric changes was documented in parents from the QUALITY (Québec Adipose and Lifestyle InvesTigation in Youth, Lambert et al., 2011) family study, a prospective cohort investigation of the course and risk factors for childhood obesity (Gallant et al., 2015). Among severely obese parents, NES severity, as documented by a high NEQ score, was linked to a lower percent change in BMI, whereas a high NEQ score was associated with a higher percent change in BMI in the nonobese parents. Moreover, in non-obese individuals a high NEQ score was linked to a larger percent change in BMI, whereas morning anorexia and nocturnal ingestions were associated respectively with an increase in weight change and BMI.
Independently from the presence of overweight or obesity, few results are available on the influence of NES on losing weight during obesity treatments based on lifestyle interventions. Gluck et al. (2001) reported that night eaters were characterized by a poorer weight loss outcome than non-night eaters. In contrast, another study outlined that diagnosing NES did not influence weight loss outcome (Dalle Grave et al., 2011). The discrepancy in the results is probably due to the different types of intervention: in the study by Gluck et al. (2001), dietary intervention consisted of a daily 900 kcal liquid diet supervised by weekly sessions with a nutritionist, whereas in the second study a mild caloric restrictive diet, daily exercises and psychoeducational group sessions have been provided (Dalle Grave et al., 2011). However, a large percentage of NES patients reduced their nocturnal ingestions after significant lifestyle modifications (Dalle Grave et al., 2011; de Zwaan et al., 2015). Further research suggested no association between NES and weight loss (Colles and Dixon, 2006). Among obese individuals with type 2 diabetes, NES is more common than BED, with rates of prevalence ranging approximately 3.8 to 12.4% versus 1.4% (Allison et al., 2007a; Hood et al., 2014; Morse et al., 2006). However, such prevalence estimates were lower than those previously reported in other studies on obese diabetic population (Gluck et al., 2001; Herpertz et al., 2000; Mannucci et al., 2002). In obese diabetic subjects with NES, changes in BMI began earlier in life than in obese individuals without an eating disorder, and the former patients also developed type 2 diabetes at a younger age (Allison et al., 2007a). Although evidence of the influence of NES on glycemic control is heterogeneous, individuals with diabetes and night eating symptoms were more likely to have diabetes-related complications and poorer glycemic control (Marshall et al., 2004; Morse et al., 2006); Hood et al. (2014), designating hemoglobin A1c as a measure of glycemic control, found an association between night eating symptoms and poorer glycemic control (HbA1c levels > 7%). However, since NES patients are at high risk of obesity, poorer glycemic control might be due to the influence of obesity rather than night eating symptoms (Calugi et al., 2005).
Further research showed that up to 55% of people undergoing bariatric surgery showed symptoms of NES (Adami et al., 1999; Allison et al., 2006; Colles and Dixon, 2006; de Zwaan et al., 2015; Gallant et al., 2012; Greeno et al., 1995; Hsu et al., 1996; Powers et al., 1999; Rand et al., 1997; Rand and Kuldau, 1986; Striegel-Moore et al., 2005); moreover, bariatric surgery has shown to be effective in reducing NES symptoms (Gallant et al., 2012). Particularly, improvement of night eating symptoms was mainly observed in patients with pre-operative depressive symptoms, as documented by NEQ scores (16.03 ± 7.73 vs 12.80 ± 7.76; p = 0.01) (Ferreira-Pinto et al., 2017).
Comorbidity: psychiatric disorders
In general, patients with eating disorders usually have high rates of comorbidity with other psychiatric disorders; furthermore, eating disorders tend to occur during stressful periods or negative life events.
The prevalence of psychiatric disorders among NES patients has been evaluated in several studies (Allison et al., 2005b; Calugi et al., 2009; Gluck et al., 2001): patients with NES exhibit significant comorbid depressive symptoms, negative affects (Colles et al., 2007; Sevincer et al., 2016; Striegel-Moore et al., 2010), major depressive disorder, anxiety disorders, substance abuse, and functional impairment (Lundgren et al., 2006b, 2010; Pawlow et al., 2003; Striegel-Moore et al., 2008, 2010; Stunkard and Allison, 2003). On the other hand, less is known about prevalence rates and NES features in patients with psychiatric disorders. The estimated prevalence of NES ranged from 12.4% in psychiatric outpatients to 25% in overweight or obese patients with schizophrenia, schizoaffective disorder, bipolar disorder and depression (Kucukgoncu and Bestepe, 2014; Lundgren et al., 2006b; Palmese et al., 2013; Saraçlı et al., 2015); NES was more common in patients with depression, with prevalence rates up to 35.2% (Kucukgoncu et al., 2014; Orhan et al., 2011). The discrepancy between rates of prevalence reported in different studies may probably be attributed to the wide range of diagnostic criteria and to the different features of the study samples. Certainly, depressed patients complaining about abnormal eating behaviors and/or experiencing weight fluctuations should be evaluated for NES. A study conducted on obese veterans showed that those who screened positive for NES were significantly more likely to have comorbid depression, sleep disorders, and post-traumatic stress disorder (PTSD) (Dorflinger et al., 2017). In those patients, night eating was associated with other atypical eating behaviors such as emotional eating and binge eating; moreover, even among those who screened negative for NES, many exhibited dysfunctional eating-related behaviors, such as lack of control over eating at night, delayed morning intake and nocturnal craving for food. Overall, these findings suggest that, in this special population, night eating may represent a coping strategy for dealing with hyperarousal, a core feature of PTSD.
Although NES appears to be an eating disorder in its own right, subjects diagnosed with NES are more likely to have a comorbid eating disorder, whose prevalence ranges from 5% to 44% (Allison et al., 2007b; Colles et al., 2007; Vander Wal, 2012). The high comorbidity rates and the significant symptomatic overlap between NES and other eating disorders led several authors to consider NES as a subtype of other eating disorders rather than an autonomous clinical entity (Kucukgoncu et al., 2015). Nevertheless, NES differs from other eating disorders for the peculiar eating habits, the number of calories consumed after dinner, the absence of compensatory behavior and for the strict relationship with insomnia (Vander Wal, 2012). It has been estimated that about 15–20% of NES patients also presented BED (Allison et al., 2005b, 2006); this subgroup of patients was characterized by higher levels of psychopathology and more weight gain (Kucukgoncu et al., 2015). In addition, the comorbidity among NES, anorexia nervosa (AN) and bulimia nervosa (BN) has been described. In a recent study, NES was detected in 10.3% of patients with AN, in 34.9% of patients with BN and in 51.7% of patients with BED (Tu et al., 2019). When compared with patients with only NES, the BN-only group exhibited similar levels of depression, sleep disturbances, disordered eating and functional impairment; except for evening hyperphagia and nocturnal eating, patients with NES alone scarcely differed from those with BN alone (Tu et al., 2019). The comorbid group had more episodes of evening hyperphagia, nocturnal ingestions and weight concerns than the BN-only groups. In general, BN is statistically more likely than AN to be associated with NES features, especially for evening hyperphagia (Lundgren et al., 2011). Another disorder that shares with NES the night eating features associated with sleep disturbances is SRED, characterized by recurrent episodes of involuntary eating and drinking during sleep. Although SRED and NES are both associated with other sleep disturbances, eating disorders and obesity (Winkelman, 2006), the differential diagnosis between the two disorders relies on the level of consciousness, since SRED is characterized by the unawareness of the eating episodes, possibly leading to the consumption of inedible or toxic substances, along with dangerous behaviors performed while in pursuit of food, and by the frequent comorbidity with other sleep disorders, such as sleepwalking and restless legs syndrome (Vinai et al., 2012).
NES is frequently comorbid with sleep disturbances. Patients with NES usually experience difficulties in sleep onset and maintenance (de Zwaan et al., 2003; Rogers et al., 2006); greater sleep latency, reduced sleep duration, poorer sleep quality and daytime sleepiness are common features, and in several cases sleep disorders and insomnia may precede NES (Hood et al., 2014). It has also been documented that increased severity of night eating symptoms may have an additive influence on sleep quality, and that depressed patients with NES exhibited more sleep disturbances than depressed patients without NES symptoms (Striegel-Moore et al., 2008). Regarding substance abuse, 30.6% of the patients with NES met criteria for a lifetime substance use disorder, and one study designed smoking as one of the determinants of NES (Kucukgoncu et al., 2014).
Treatment options for NES
Several treatment strategies have been evaluated in the literature for the therapy of NES including pharmacological approaches, psychotherapy and bariatric surgery, when NES is comorbid with severe obesity (Shoar et al., 2019). Nevertheless, there is still a paucity of studies focused on NES treatment, and the limited amount of available evidence derives from clinical trials with insufficient study power and long-term follow-up.
Pharmacological treatment
Serotonergic system, the main regulator of a wide variety of physiological functions and behaviors such as eating, sleep and mood is thought to be intrinsically involved in NES; mainly, a decrease in serotonin levels would lead to the alteration of circadian rhythms and to a high risk of evening hyperphagia and nocturnal eating (Allison et al., 2005a; Pinto et al., 2016; Stunkard et al., 2009a). Moreover, as previously reported, in night eaters higher levels of SERT in the temporal lobe have been documented (Lundgren et al., 2008b); this finding supports the presence of dysfunctions in postsynaptic serotonergic transmission, that contributes to changes in circadian rhythms and appetite (Stunkard et al., 2009b). Based on the involvement of the serotonergic system in NES, pharmacological interventions mostly focused on the use of antidepressants medications (Kucukgoncu et al., 2015; O’Reardon et al., 2005). The efficacy of SSRIs for the treatment of NES has been evaluated in two randomized, placebo-controlled trials, two open-label studies and one case series. Sertraline has proven more effective than placebo in improving NES symptoms and quality of life, and in reducing the number of nocturnal ingestions (81% vs 14%), awakenings (74% vs 14%) and evening hyperphagia (68% vs 29%) (O’Reardon et al., 2006); response rates achieved by NES patients were 67–71% (O’Reardon et al., 2004a, 2006) with remission rates of 29% (O’Reardon et al., 2004a). In an open-label, long-distance treatment study on 50 NES patients (78% women), sertraline was effective in improving NES symptoms, mood and quality of life (Stunkard et al., 2006). A significant weight loss in sertraline-treated patients was also observed in the cited studies. A case series showed that both paroxetine and fluvoxamine resulted effective in reducing episodes of night eating (Miyaoka et al., 2003). Escitalopram has also been tested with discordant results (Allison et al., 2013; Vander Wal et al., 2012). Findings from the randomized, placebo-controlled trial by Vander Wal et al. (2012) showed that escitalopram was not statistically superior to placebo in reducing NES symptoms, although there was a favorable trend for escitalopram on the primary and secondary outcome measures, as well as on weight, with effect sizes ranging from small to moderate. Conversely, in the 12-week open-label trial (Allison et al., 2013), escitalopram significantly improved night eating symptoms such as the amount of caloric intake consumed after dinner, and number of nocturnal ingestions per week; furthermore, 58% of participants were in symptomatic remission at the end of treatment. Beyond SSRIs, other drugs have been used for the treatment of NES. The efficacy of topiramate, a gamma-aminobutyric acid (GABA) agonist and glutamatergic antagonist, has been evaluated in one case series and two case reports (Cooper-Kazaz, 2012; Tucker et al., 2004; Winkelman, 2003). Under topiramate treatment, a reduction in nocturnal awakenings and evening food intake with sleep improvement and weight loss has been observed in 4 patients who did not respond to previous pharmacotherapy and psychological interventions (Winkelman, 2003). Other authors described a case of an obese woman with nocturnal awakenings and eating episodes that resulted reduced under topiramate treatment (Tucker et al., 2004). Topiramate added to ongoing venlafaxine treatment was effective in improving both depressive and night eating symptoms after 6 weeks in a 40-year-old woman with comorbid NES and PTSD (Cooper-Kazaz, 2012). The mechanism by which topiramate has proven effective in NES is probably attributable to its peculiar adverse events profile, including anorexigenic effect and high somnolence that promotes sleep. However, the available evidence comes from small-sized case series and case reports, and other well-known side effects of topiramate, such as cognitive and language impairment, must be considered before the prescription. Previous studies showed a decrease of the nocturnal rise in plasma of melatonin levels in patients with NES (Birketvedt et al., 1999); accordingly, agomelatine, a selective melatonin agonist and a weak serotonin 5-HT2C receptor antagonist has been considered as an option treatment for NES for its normalizing outcome on the sleep-wake cycle besides the effect on depression and anxiety symptoms (Kucukgoncu et al., 2015). Agomelatine was administered to depressed patients with NES resulting in a global improvement of symptomatology (Milano et al., 2013a,b).
Even though the collective evidence regarding the administration of pharmacological therapy is still sparse, the summarized results from published efficacy trials on pharmacological treatment of NES (Table 4) suggest that SSRIs, particularly sertraline and escitalopram can be effective treatments, whereas more research is needed to delineate the place in therapy of topiramate and melatonergic agents, since the available evidence derives from uncontrolled studies with small sample sizes. Further randomized controlled studies are crucial for orientating prescription toward evidence-based psychopharmacology. Currently, there are no shared guidelines on the choice of treatment and its duration, although it is recommended to administer a pharmacological treatment for no fewer than 8 weeks in order to determine its efficacy on NES; if beneficial, treatment should be continued for at least 1 year (O’Reardon et al., 2005).
Published efficacy trials on pharmacological treatment of NES.
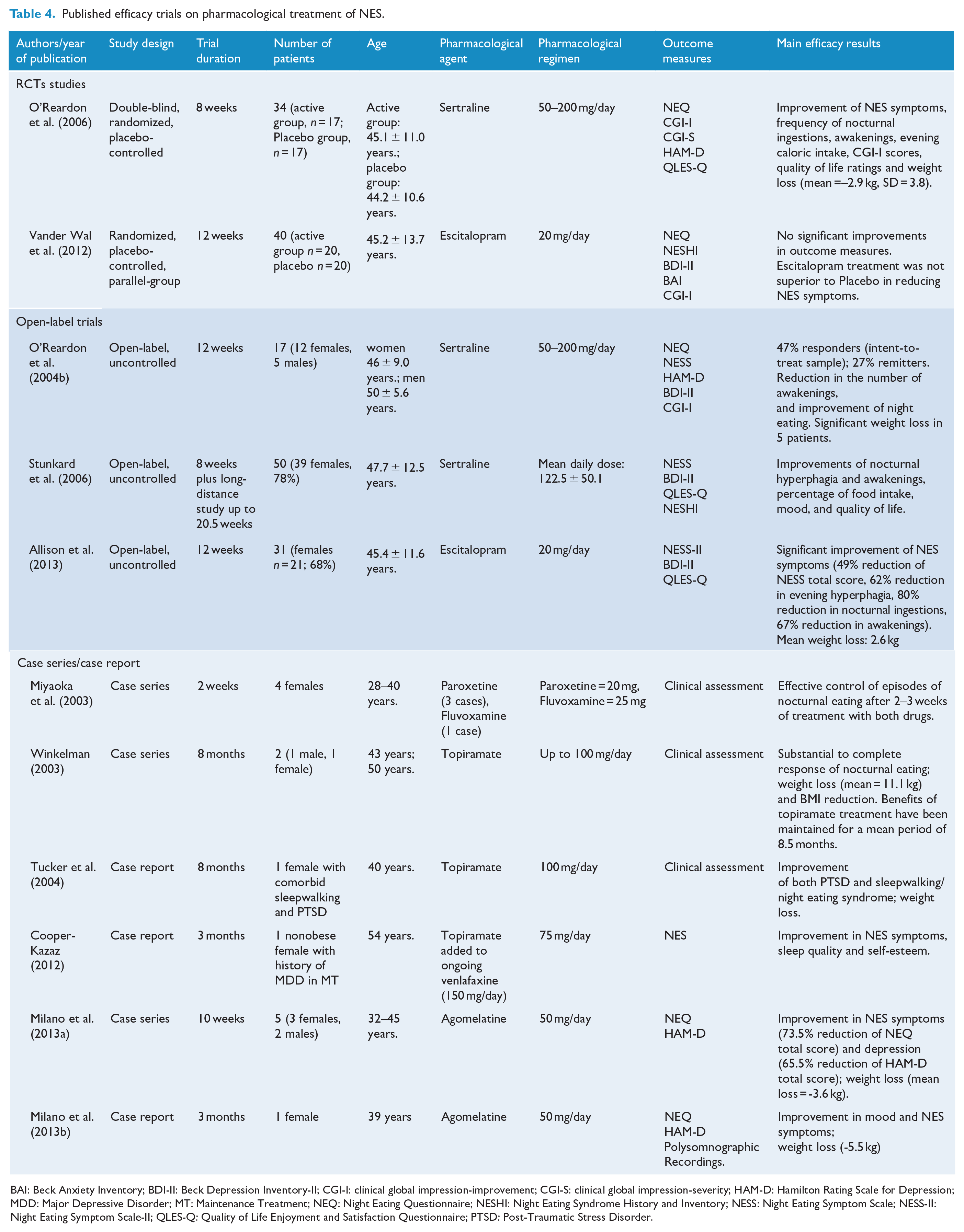
BAI: Beck Anxiety Inventory; BDI-II: Beck Depression Inventory-II; CGI-I: clinical global impression-improvement; CGI-S: clinical global impression-severity; HAM-D: Hamilton Rating Scale for Depression; MDD: Major Depressive Disorder; MT: Maintenance Treatment; NEQ: Night Eating Questionnaire; NESHI: Night Eating Syndrome History and Inventory; NESS: Night Eating Symptom Scale; NESS-II: Night Eating Symptom Scale-II; QLES-Q: Quality of Life Enjoyment and Satisfaction Questionnaire; PTSD: Post-Traumatic Stress Disorder.
Non-pharmacological treatment
In an effort to find alternative treatments for NES, non-pharmacological options have been widely tested (Table 5). Bright light therapy has been shown to increase the availability of postsynaptic serotonin and it has been evaluated in three case studies, resulting in a significant reduction of mood disturbances and NES symptoms (Friedman et al., 2002, 2004; McCune and Lundgren, 2015). In the first case, phototherapy was added to paroxetine in a 51-year-old woman with NES and comorbid depression. A remission of depressive and NES symptoms was evidenced after 14 daily sessions of bright light therapy. Despite this, once the phototherapy was suspended, NES symptoms returned, and a further period of phototherapy treatment has been necessary to achieve remission (Friedman et al., 2002). The same research group reported that a non-obese patient with NES and comorbid depression treated with daily 30 minutes phototherapy obtained improvements in depression and NES features after 2 weeks (Friedman et al., 2004).
Published efficacy trials on non-pharmacological treatment of NES.
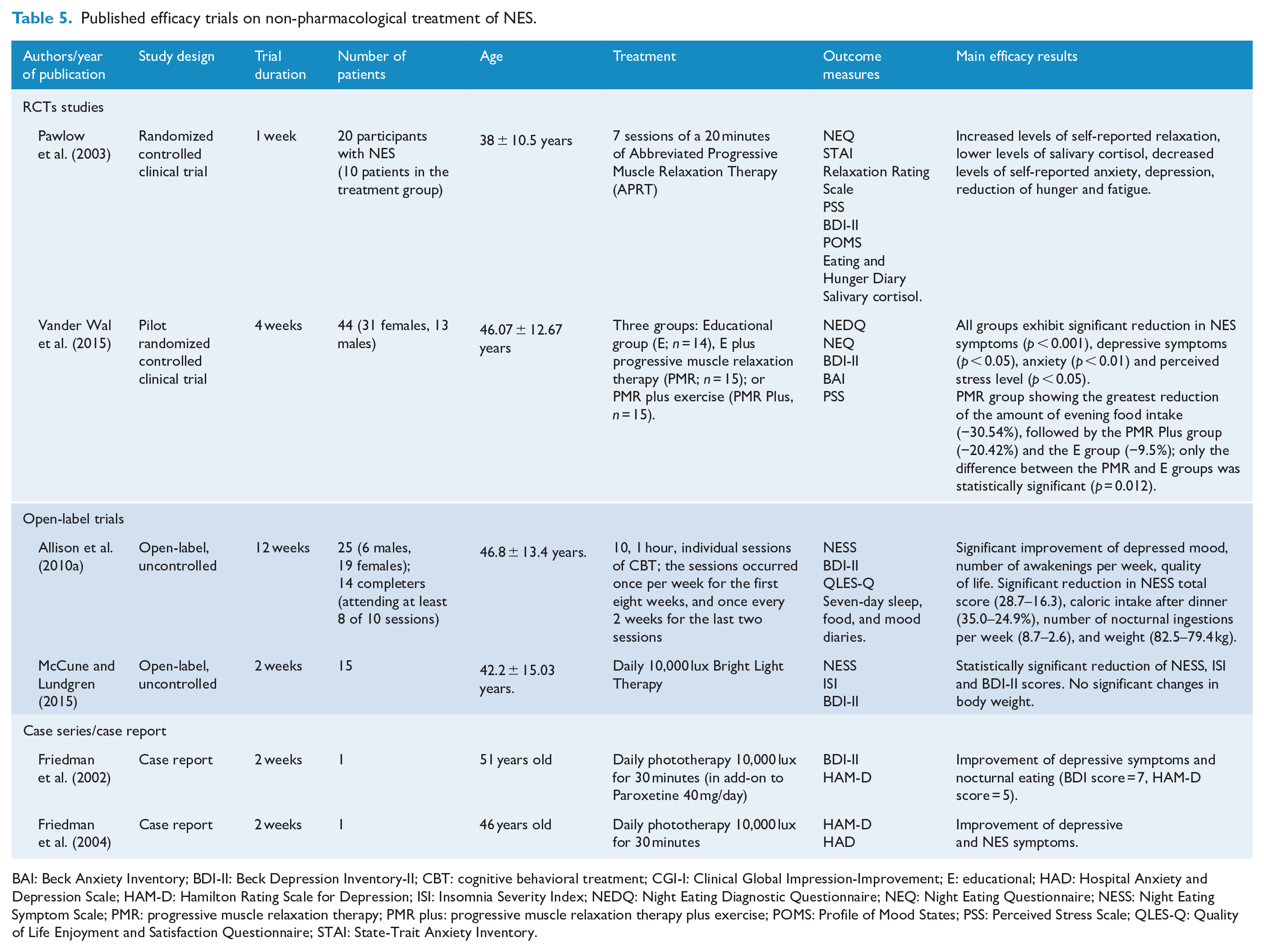
BAI: Beck Anxiety Inventory; BDI-II: Beck Depression Inventory-II; CBT: cognitive behavioral treatment; CGI-I: Clinical Global Impression-Improvement; E: educational; HAD: Hospital Anxiety and Depression Scale; HAM-D: Hamilton Rating Scale for Depression; ISI: Insomnia Severity Index; NEDQ: Night Eating Diagnostic Questionnaire; NEQ: Night Eating Questionnaire; NESS: Night Eating Symptom Scale; PMR: progressive muscle relaxation therapy; PMR plus: progressive muscle relaxation therapy plus exercise; POMS: Profile of Mood States; PSS: Perceived Stress Scale; QLES-Q: Quality of Life Enjoyment and Satisfaction Questionnaire; STAI: State-Trait Anxiety Inventory.
According to its effect on mood, stress, and hunger, progressive muscle relaxation (PMR) has also been studied as alternative treatment for NES (Pawlow et al., 2003; Vander Wal et al., 2015). The first study focused on the utility of relaxation training on stress and mood, and its impact on night eating symptoms (Pawlow et al., 2003). A total of 10 individuals with NES, randomly assigned to an abbreviated progressive muscle relaxation therapy (APRT) performed a 20 minutes session of muscle relaxation exercises for a week. At the end of the protocol, the treatment subgroup showed significant improvements in depression, anxiety and stress as well as a reduction in night eating behaviors (Pawlow et al., 2003). In addition, in a subsequent study, a group of 44 individuals with NES was randomized to an educational group, an educational group plus PMR, and PMR plus exercise (Vander Wal et al., 2015). Participants across all groups showed significant reductions of NES symptoms as well as an improvement of mood disturbances and anxiety levels. These results are encouraging, and prompt the interest toward this treatment strategy that deserves further research to better understand the beneficial role of exercise in NES.
NES is largely associated with cognitive distortions linked to specific food cravings and the belief that one is unable to sleep unless eating beforehand (Vinai et al., 2014). Cognitive behavioral therapy (CBT) has proven effective in reducing NES symptoms, such as evening caloric intake, awakenings and nocturnal ingestions (Allison et al., 2010a). Specifically tailored for patients with NES, an intensive model of CBT combining psychoeducation, healthy nutrition, sleep hygiene rules, exercises, and relaxation strategies was evaluated in a sample of 25 patients. The results obtained from this pilot trial were comparable to those of the published randomized controlled trials with sertraline (O’Reardon et al., 2004b, 2006), although no data are available on the stability of improvement in the follow-up; long-term studies on larger samples are needed to fully understand the therapeutic potential of this combination strategy (Allison et al., 2010a). Overall, given the promising results, future randomized controlled trials on larger samples and comparison studies of CBT with other treatment strategies are required.
Conclusions and future perspectives
The inclusion of NES into the DSM-5 ‘Other Specified Feeding or Eating Disorders’ category should drive the attention of clinician and researchers toward this syndrome that is still defined by evolving diagnostic criteria. The correct identification and assessment of NES through a focused clinical attention, and the use of screening questionnaires could facilitate the detection and the diagnosis of this disorder, often submerged by feelings of guilt and shame. NES is burdened by complex associations with obesity, endocrine and metabolic alterations, and with mood, anxiety and sleep disorders; accordingly, mainly in the obese population, the diagnosis of NES deserves more attention and research in order to define whether specific treatments other than lifestyle modification are needed. The bio-psycho-social model of NES suggests the multifactorial contributors to the syndrome by highlighting the role of serotonin imbalance, circadian disruptions, stress, poor nighttime satiety and negative emotions. Within this framework, and taking into account the significant amount of the psychopathological comorbidities associated with NES, further research should better explore the role of depression in NES, with the aim of investigating whether nocturnal eating is only an indicator of depression or whether depression is a risk factor for the onset of NES or a consequence of the syndrome (Calugi et al., 2009). Complementary research is also necessary to define the clinical significance of the overlapping symptoms between NES and other eating disorders and to determine if patients with both NES and other night eating disorders require more intensive treatment approaches.
NES should be specifically suspected and carefully investigated when the clinician encounters weight loss failure in obese people despite the adherence to dietary regimens and lifestyle changes, weight changes in depressed and/or anxious patients, and in all those clinical conditions that are frequently comorbid with NES, such as sleep disorders and parasomnias, diabetes, restless legs syndrome, and obstructive sleep apnea. On the other hand, the impact of diagnostic failure of NES on the course of the medical comorbidities, psychological functioning and quality of life in affected patients does not appear to have been completely recognized.
Treatment options for NES include both pharmacological (SSRIs, topiramate and melatonergic drugs) and non-pharmachological approaches, including psychotherapy, behavioral measures, exercise and relaxation techniques, and phototherapy; the combination of such strategies within a multidisciplinary approach should be addressed in future, well-sized, and long-term studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
