Abstract
Objective:
Cortical and subcortical gray matter abnormalities have been reported in individuals at high genetic risk for bipolar disorder, but the findings are inconsistent. The aim of this study was to review the available literature to identify common findings that could represent brain structural vulnerability factors for bipolar disorder and to discuss challenges for the advancement of the field.
Method:
A systematic search was conducted using the PubMed database to identify all original articles investigating cortical or subcortical gray matter abnormalities in first-degree relatives of bipolar disorder patients.
Results:
Very few findings were replicated, with the exception of larger insular cortex volumes in adult first-degree relatives and larger right inferior frontal gyrus in offspring of probands with bipolar disorder, both when compared with healthy controls. Isolated findings included decreased gray matter density in the left thalamus, decreased gray matter volumes in the left hippocampus and parahippocampal gyrus, and thicker right hippocampus in unaffected first-degree relatives. Genetic liability for bipolar disorder was associated with gray matter volumes in regions of the anterior cingulate cortex, ventral striatum, medial frontal gyrus, right precentral gyrus, right insular cortex, and medial orbital gyrus. Some studies found no evidence for gray matter abnormalities in first-degree relatives of bipolar disorder patients.
Conclusions:
Possible reasons for the discrepancies of findings across studies include small samples sizes, small effect size of susceptibility genes, the phenotypic heterogeneity of bipolar disorder, and the possible confounding effect of other Axis I psychopathologies among the relatives of patients. Future multisite, prospective, large studies with more homogeneous samples would be a key strategy to advance the field. The ultimate benefit would be an understanding of how to use brain imaging tools to identify individuals at increased risk for bipolar disorder and develop preventive strategies for that population.
Introduction
Bipolar disorder (BD) is a severe, recurrent, and highly disabling psychiatric disorder which affects 1–3% of the world population (Belmaker et al., 2004). Family, twin, and adoption studies consistently show that BD is highly heritable (Kieseppä et al., 2004; McGuffin et al., 2003; Winokur et al., 1995). Twin studies show that environmental factors (including childhood traumatic experiences) represent 20% of the risk of developing BD for identical twins and 70–80% for fraternal twins (Kieseppä et al., 2004; McGuffin et al., 2003). The risk of developing BD probably represents the combined effects of several susceptibility genes and their interactions with environmental risk factors, such as a dysfunctional parent-child relationship, parental loss during childhood, the occurrence of stressful life events, and drug abuse (Mortensen et al., 2003; Tsuchiya et al., 2003).
Evidence in the last 20 years suggests that structural, neurochemical, and functional abnormalities in some brain regions that pertain to two interrelated circuits, a limbic-thalamic-cortical and a limbic-striatal-pallidal-thalamic circuit, are responsible for the symptomatic manifestations of BD (Soares and Mann, 1997; Strakowski et al., 2005; Strakowski et al., 2012). Regarding structural abnormalities, some findings associated with BD have been replicated, including lateral ventricular enlargement (Arnone et al., 2009; Hallahan et al., 2011; Kempton et al., 2008; McDonald et al., 2004), whole brain and prefrontal lobe volume reductions and increased globus pallidus (Arnone et al., 2009), and gray matter (GM) volume reductions in the anterior cingulate cortex and anterior insular cortex (Bora et al., 2010; Ellison-Wright and Bullmore, 2010). However, across studies, many discrepancies in abnormal findings also exist. The sources of discrepancies include the clinical characteristics of the disease that may impact brain structure and that differ among patients, such as developmental stage, illness duration, medication effects of lithium or antipsychotics, or phenotypic illness heterogeneity (Arnone et al., 2009; Bora et al., 2010; Ellison-Wright and Bullmore, 2010; Hallahan et al., 2011; Houenou et al., 2011; Kempton et al., 2008). Therefore, it remains unknown which brain structural abnormalities (if any) exist before symptom onset and whether the abnormalities represent potential vulnerability factors for the disease.
One way to investigate vulnerability factors for highly heritable diseases is to study individuals at high genetic risk for the disorder, such as first-degree relatives. The study of first-degree relatives of patients with BD allows the identification and investigation of endophenotypes for this disorder. The endophenotype is an intermediate phenotype for BD, internal and not visible to the naked eye (or, in the case of BD, not evident at the clinical evaluation) (Hasler et al., 2006). Because it is free of the characteristics that are inherent to the clinical expression of the disease, the endophenotype is an elementary and pure neurobiological abnormality that has the potential to clarify which neurobiological abnormalities in BD are a direct consequence of susceptibility genes and which are due to neurodegeneration or insults originated from affective states, alcohol or drug misuse, or psychiatric medication exposure (Hasler et al., 2006). The identification of structural brain abnormalities in this population may help define structural brain endophenotypes for the disorder, which in turn may help understand the mechanisms of vulnerability for or protection against the disease and, ultimately, may help develop primary and secondary mechanisms of prevention for the disease.
In this article, we have reviewed structural neuroimaging studies of populations at high genetic risk for BD, in an attempt to identify common findings of regional structural abnormalities that may correspond to a vulnerability factor for the disease. Considering the findings as a whole, we have critically appraised the literature, discussing pitfalls and challenges for the advancement of research in this topic.
Methods
A literature search was conducted using the PubMed database (www.ncbi.nlm.nih.gov/pubmed; National Center for Biotechnology Information, US National Library of Medicine, Bethesda, Maryland, USA) to identify relevant research articles related to BD that were published on or before February 2013. Search terms included: bipolar disorder; magnetic resonance imaging; healthy relatives; healthy siblings; at risk; and unaffected relatives. We included only articles that were published in the English language, reported original data, studied first- or second-degree relatives (including co-twins, siblings, parents, or offspring) of BD patients, included only unaffected or healthy relatives, and included a healthy control (HC) group for comparison. Relevant papers discovered by reading the reference lists of identified articles were included in a few instances. The present article focuses on comparisons between relatives and HCs. Studies without a HC group are discussed separately if they examined genetic susceptibility for BD in first-degree relatives of BD patients using correlational analysis between brain structural variables, such as GM volume or density and measures of genetic liability for BD.
Results
Twenty-four studies investigated structural GM abnormalities in unaffected co-twins, siblings, or first-degree relatives of BD patients using different methodologies, including region-of-interest (ROI) manual tracing, voxel-based morphometry (VBM), semiautomatic segmentation, or regional shape anomaly. In ROI manual tracing, the specific brain region (e.g. the hippocampus) is manually delineated (traced) by a blinded rater based on anatomical references, and the volume (in cm3) within the traced figure is calculated (Giuliani et al., 2005). The VBM method is a fully automated method that surveys the whole brain, looking for differences in density or volume of GM or white matter (WM) between groups (Ashburner and Friston, 2000). For reasons of space and because the remaining methods are less popular than ROI and VBM, the reader may refer directly to the specific articles for details about semiautomatic methods, cortical mapping, and regional shape anomaly or thickness. The articles in this review are discussed separately by imaging analysis methodology.
Of the 24 identified studies, 10 used ROI manual tracing to compare first-degree relatives of BD patients with HCs. These studies reported increased left caudate nucleus volumes in a small sample of unaffected co-twins of BD patients compared with HCs (Noga et al., 2001), and larger caudate nucleus volumes in the healthy offspring of BD parents compared with the healthy offspring of healthy parents (Hajek et al., 2009). One study found enlarged left amygdala volumes and decreased right hippocampal volumes in 10 healthy relatives from the same family of a BD type I patient when compared with 19 HCs (Boccardi et al., 2010). On the other hand, no differences were found in hemispheric GM (Kieseppä et al., 2002; Kieseppä et al., 2003), regional frontal and temporal GM (Kieseppä et al., 2003), subgenual anterior cingulate cortex (Hajek et al., 2008a; Hajek et al., 2010), amygdala/hippocampus complex (Noga et al., 2001), amygdala (Singh et al., 2008), hippocampus, thalamus (Singh et al., 2008), striatum (Hajek et al., 2009; Singh et al., 2008), putamen (Hajek et al., 2009; Noga et al., 2001), and pituitary volumes (Hajek et al., 2008b; Takahashi et al., 2010) of first-degree relatives of BD patients compared with HCs. Details of these studies, including sample size, clinical characteristics, and main findings, are shown in Table 1.
ROI manual tracing studies of GM abnormalities in relatives of BD patients.
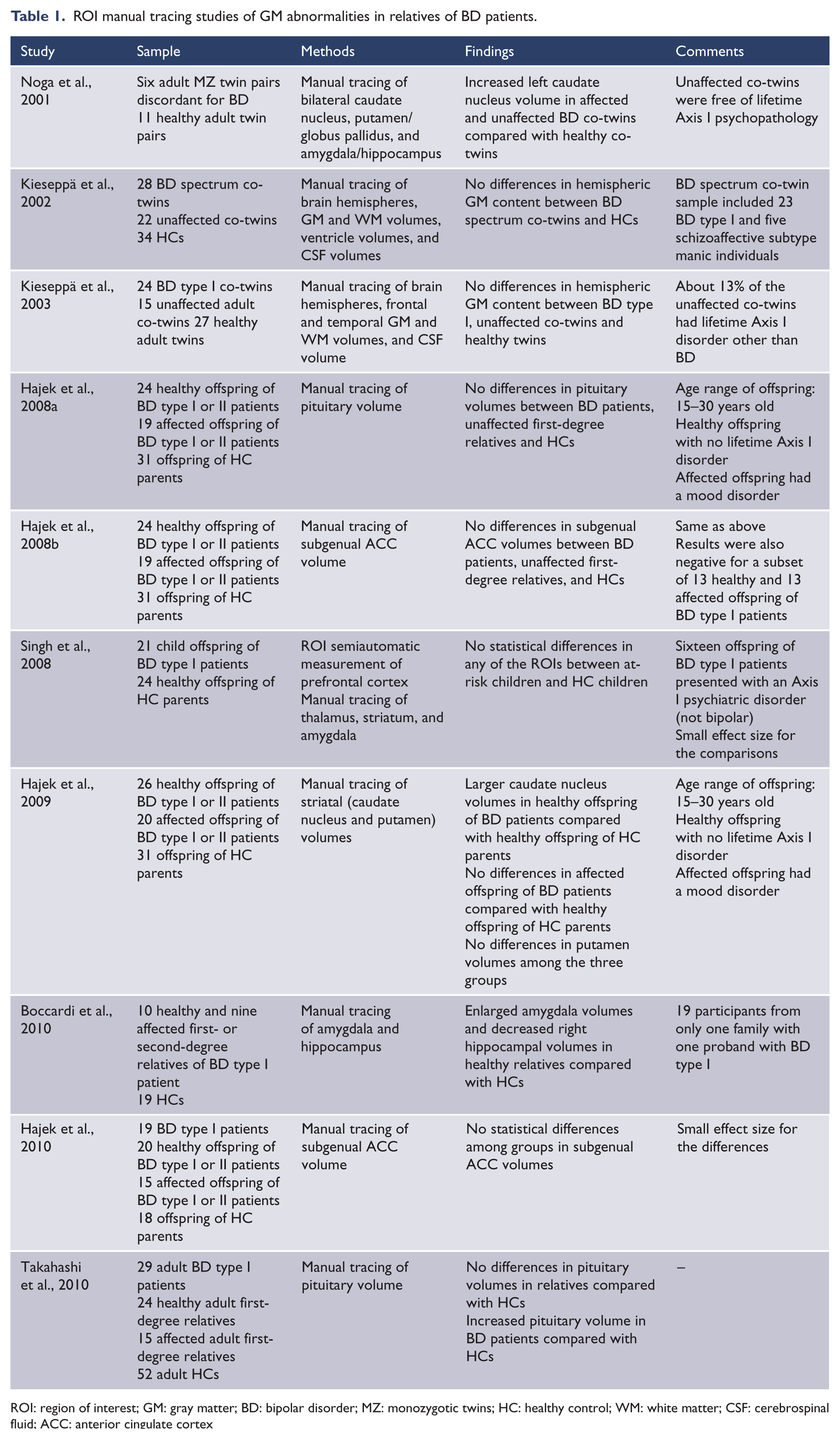
ROI: region of interest; GM: gray matter; BD: bipolar disorder; MZ: monozygotic twins; HC: healthy control; WM: white matter; CSF: cerebrospinal fluid; ACC: anterior cingulate cortex
Five VBM studies compared GM abnormalities in individuals at risk for BD using the voxelwise approach. These studies found decreased GM density in the left anterior thalamus and caudate nucleus (McIntosh et al., 2004), larger volumes of the left insular cortex (Kempton et al., 2009; Matsuo et al., 2012) and left cerebellum (Kempton et al., 2009) in first-degree relatives of BD patients compared with HCs, and reduced GM volumes of the left parahippocampal gyrus and left hippocampus of the healthy offspring of BD parents compared with the healthy offspring of healthy parents (Ladoucer et al., 2008). Another recent study reported larger GM volumes in the right inferior frontal gyrus of the healthy offspring of BD patients compared with HCs in two unrelated samples (Hajek et al., 2013). Details of these studies, including sample size, clinical characteristics, and main findings, are displayed in Table 2.
VBM studies of GM abnormalities in relatives of BD patients.
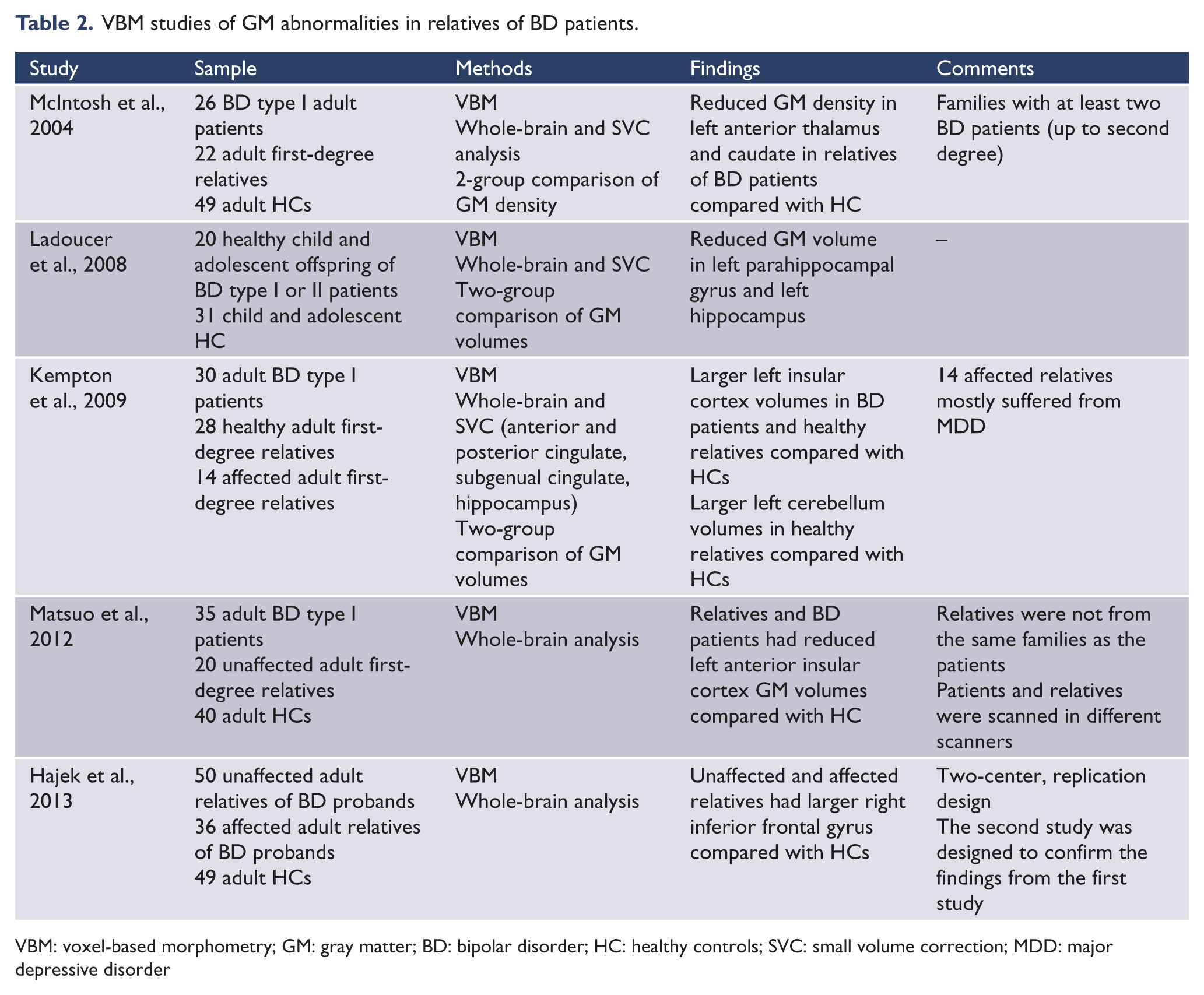
VBM: voxel-based morphometry; GM: gray matter; BD: bipolar disorder; HC: healthy controls; SVC: small volume correction; MDD: major depressive disorder
A few other studies used different methods to investigate cortical or subcortical GM abnormalities in relatives of BD patients, including stereological examination, semiautomatic segmentation, and cortical mapping. One study found no difference in gross cerebral and ventricular volumes and hippocampal volumes between relatives of BD patients and HCs (McDonald et al., 2006). Two studies investigated the shape and thickness of the hippocampus. One found no differences in hippocampal shape anomaly between first-degree relatives of BD and HCs (Connor et al., 2004). The other study, using a twin-pair design, found that unaffected co-twins of BD patients had thickened right hippocampal subregions (cornu ammonis and anterior subiculum) compared with HCs (Van Erp et al., 2012). Details of these studies, including sample size, clinical characteristics, and main findings, are shown in Table 3.
Structural studies of GM abnormalities in relatives of BD patient: Other methods.
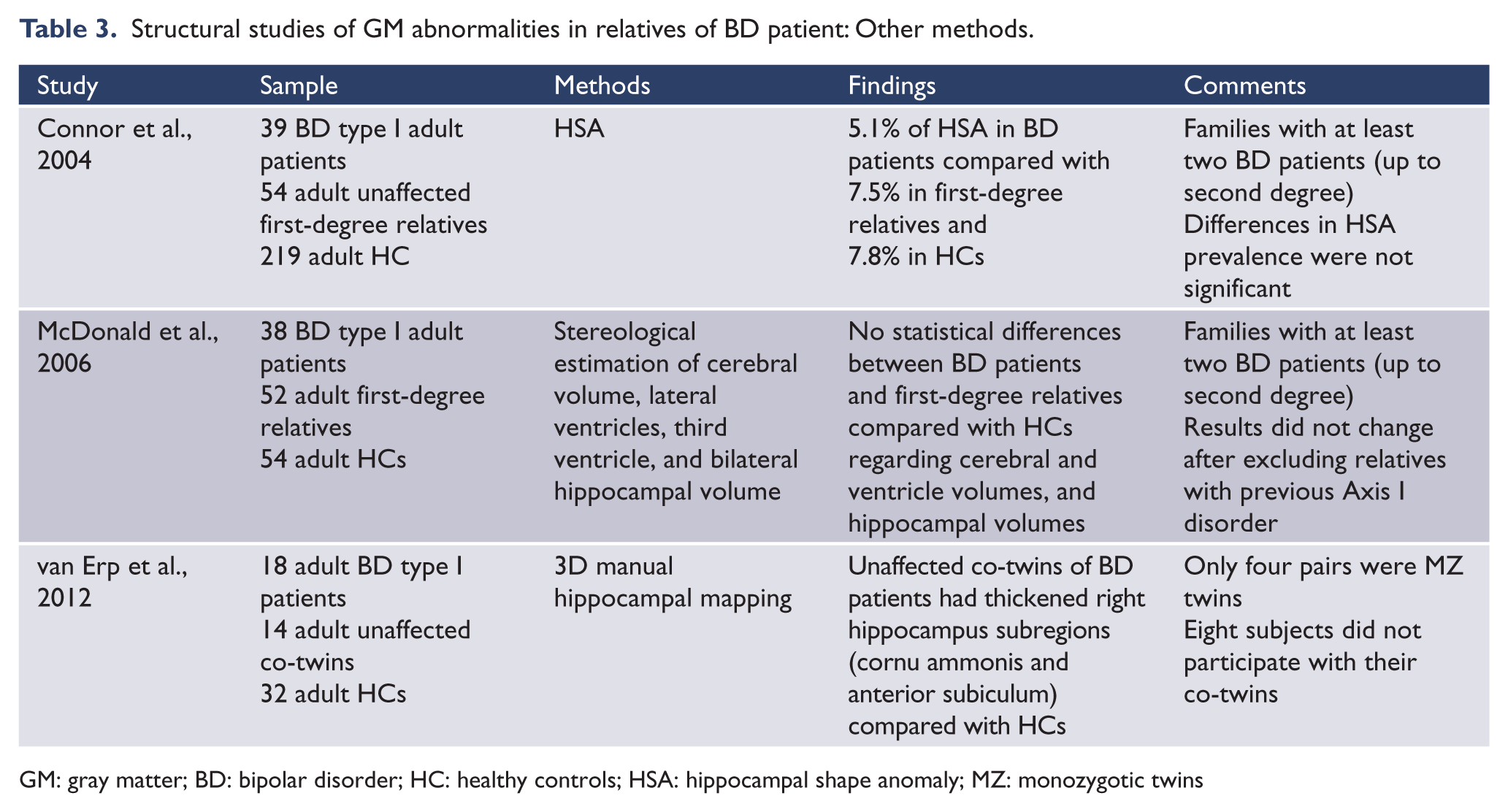
GM: gray matter; BD: bipolar disorder; HC: healthy controls; HSA: hippocampal shape anomaly; MZ: monozygotic twins
Finally, the relationship between genetic liability for BD and brain structure was investigated in five studies. In two studies, a genetic liability variable for BD was created to simulate the likely variation in genetic risk across individuals, including the number of BD patients within a family and the degree of relatedness between the scored individual and the index patient (McDonald et al., 2004; McIntosh et al., 2006). In three twin studies, the genetic liability score was calculated taking into consideration the heritability and prevalence of BD (Hulshoff Pol et al., 2012; Van der Schot, 2009, 2010). In two studies, the genetic liability score for BD was negatively correlated with GM volumes in the right anterior cingulate cortex and ventral striatum (McDonald et al., 2004), and with 30–39% lower GM density in the right medial frontal gyrus, right precentral gyrus, and right insular cortex (Van der Schot et al., 2010). The genetic liability for BD was positively correlated with GM density in the right medial orbital gyrus (Van der Schot et al., 2010). Conversely, one VBM study found no correlation between genetic liability scores for BD and GM volumes (McIntosh et al., 2006). Two twin-pair studies using segmentation methods found that higher genetic liability for BD was not associated with decreased GM volumes (Hulshoff Pol et al., 2012; Van der Schot et al., 2009). On the other hand, in one of these studies, cortical mapping analysis showed that higher genetic liability scores for BD was associated with thinner right and left hippocampus, thinner right orbitofrontal cortex, and thicker temporoparietal and left superior motor cortices (Hulshoff Pol et al., 2012). Details of these studies, including sample size, clinical characteristics, and main findings, are shown in Table 4.
Studies of the correlations between GM abnormalities and genetic liability for BD.
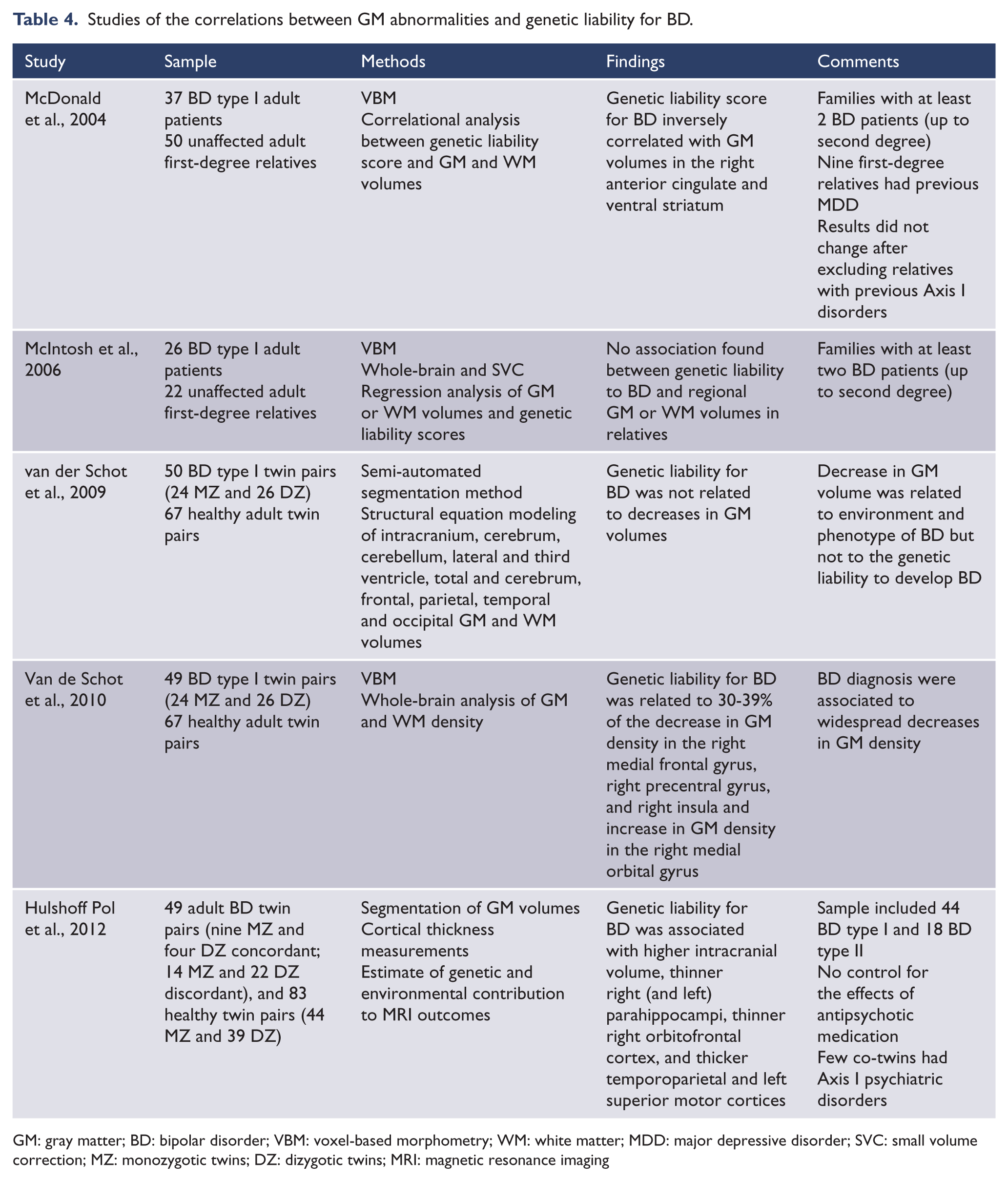
GM: gray matter; BD: bipolar disorder; VBM: voxel-based morphometry; WM: white matter; MDD: major depressive disorder; SVC: small volume correction; MZ: monozygotic twins; DZ: dizygotic twins; MRI: magnetic resonance imaging
Discussion
Twenty-four neuroimaging studies of structural abnormalities in individuals at increased genetic risk for developing BD were reviewed, focusing on cortical or subcortical GM abnormalities. Studies were grouped according to the methodology used to investigate GM differences between relatives and HCs. The most frequently used technique was ROI manual tracing, in which the investigation focuses on one hypothesis-driven brain region. Although time-consuming, ROI manual tracing is considered by many to be the gold standard of structural neuroimaging studies (Giuliani et al., 2005; Lyoo et al., 2004). Two preliminary findings are worthy of mention. First, Noga et al. (2001) found greater left caudate nucleus volumes in a small sample of unaffected co-twins of BD patients compared with HCs, and Hajek et al. (2009) found greater bilateral caudate nucleus volumes in the healthy offspring of BD parents compared to the healthy offspring of healthy parents. However, these results did not survive controlling for non-independence of observations in multiple subjects recruited within the same families; therefore, they were reported as negative. The remaining ROI studies reviewed here showed that first-degree relatives of BD patients do not differ from HCs on cortical GM regions, such as in the hemispheres, frontal and temporal lobes, or subcortical structures, including the subgenual anterior cingulate cortex, amygdala-hippocampus complex, amygdala, hippocampus, thalamus, striatum, and putamen volumes (Hajek et al, 2008a; Hajek et al., 2008b; Hajek et al., 2009; Hajek et al., 2010; Kieseppä et al., 2002; Kieseppä et al., 2003; Noga et al., 2001; Singh et al., 2008; Takahashi et al., 2010).
It is interesting to note that the VBM studies, which allow a survey of the whole brain, and, therefore, may capture regional differences in GM volume or density that are not evident in the ROI approach, yielded different results. McIntosh et al. (2004) reported abnormal GM density in the left caudate nucleus of adult first-degree relatives of BD patients, although in a different direction (decreased volume) than Noga et al. (2001) (increased volume). Two studies reported increased volume of the insular cortex in adult first-degree relatives of BD patients (Kempton et al., 2009; Matsuo et al., 2012), which is among the few replicated findings in the literature. Another interesting replicated finding was reported within the same study, in which the investigators performed the same experiment in two unrelated samples and found larger GM volumes in the right inferior frontal gyrus of the healthy offspring of BD parents compared with HCs (Hajek et al., 2013). Finally, there was an isolated finding of decreased GM density in the left anterior thalamus (McIntosh et al., 2004), and reduced GM volumes in the left parahippocampal gyrus and left hippocampus of the healthy offspring of BD patients compared with the healthy offspring of healthy parents (Ladoucer et al., 2008).
Anomalies in the shape and thickness of the hippocampus were examined in very few studies (Connor et al., 2004; Van Erp et al., 2012). Van Erp et al. (2012) reported increased thickness in the subregions of the right hippocampus in unaffected co-twins of BD patients compared with control twins, which was interpreted by the authors as a possible indication of neuroprotection against mood instability, given the observation that lithium use was also associated with increased thickness in the same area in BD co-twins compared with control twins.
Some studies investigated the associations between genetic susceptibility to BD (using a genetic liability scale as a proxy measure of genetic susceptibility) and brain structure. This approach does not directly investigate the existence of abnormal regional brain structures, but investigates whether a higher genetic susceptibility to BD engenders effects on brain structure (regardless of whether the structure is abnormal over a certain threshold or simply compared with a normal comparison standard). These studies showed that as the genetic risk for BD increased among first-degree relatives, smaller GM volumes were found in the right anterior cingulate cortex and ventral striatum, lower GM densities were found in the right medial frontal gyrus, right precentral gyrus, and right insular cortex, and decreased cortical thickness was found in the right and left hippocampus and right orbitofrontal cortex (Hulshoff Pol et al., 2012; McDonald et al., 2004; Van der Schot et al., 2010). Higher GM densities in the right medial orbital gyrus and increased cortical thickness in the temporoparietal and left superior motor cortices were also associated with higher genetic risk for BD (Hulshoff Pol et al., 2012; Van der Schot et al., 2010). However, it is noteworthy that one study did not find any correlations between genetic liability for BD and GM increases or decreases (McIntosh et al., 2006).
This review shows that despite the substantial literature, very few structural brain abnormalities were consistently found in relatives of patients with BD. At first glance, this may suggest that findings are spurious and that lack of consistency is due to several reasons, including the small sample sizes contributing to type I errors. However, it is important to note that some of the few replicated findings pertain to brain structures that are part of the fronto-limbic network postulated to be abnormal in BD, such as the insular cortex and the inferior frontal gyrus (Soares and Mann, 1997; Strakowski et al., 2005; Strakowski et al., 2012). Therefore, abnormal structure in these brain areas could represent potential brain structural endophenotypes for BD. However, strictly speaking, this assumption challenges the concept of endophenotype. To be considered an endophenotype for a psychiatric disorder, any neurobiological abnormality would have to meet certain criteria, including: evidence of being associated with the disease in the population; being heritable; being state-independent; cosegregating within families; and being found in unaffected family members at a higher rate than in the general population (Gottesman and Gould, 2003). For the first criterion (being associated with the disease), decreased insular cortex volumes in BD patients has been found consistently in meta-analyses of VBM studies (Bora et al., 2010; Ellison-Wright and Bullmore, 2010), although the inferior frontal gyrus findings have been more inconsistent (Blumberg et al., 2006; Lyoo et al., 2004; Nugent et al., 2006). Therefore, with the possible exception of the insular cortex, very few, if any, of the neuroimaging abnormalities in the relatives of BD patients presented in this review has been proven so far to meet all the criteria to be considered an endophenotype candidate.
One point to consider when interpreting the findings from this review is that the genetic effect sizes for endophenotypes of psychiatric disorders are no larger than those for phenotypes of psychiatric disorders (clinically manifested diseases) (Flint and Munafò, 2007), and the genetic architecture of psychiatric disorders has been shown to be much more complex than previously thought (Flint and Munafò, 2007; Maher, 2008). The study of high genetic risk for BD involves the proper identification of individuals with a recognized increased genetic risk for the disease, such as first-degree relatives, and comparison to the general population (Smoller and Finn, 2003). There is some evidence that the degree of relatedness and the number of patients within the family may impact the risk for transmission of BD (Birmaher et al., 2009; Smoller and Finn, 2003). It is also known that the heritability of BD most likely involves a multifactorial transmission pattern, and hundreds or thousands of genes with small effects act together to predispose the person to develop BD (Barnett and Smoller, 2009; Schulze, 2010). When sampling first-degree relatives of BD, one includes individuals that share only some of the putative genes for BD, and therefore, the effect sizes of these few genes on brain structure or brain function may be so small that they are nearly undetectable by current neuroimaging techniques using small samples. This may be an important limitation in the ability of neuroimaging studies to find associations of any structural brain abnormality with the diseased and with their relatives, and may only be overcome by studies using larger samples.
An alternative to improve the ability to identify the effect of susceptibility genes for BD on the brain structure of individuals at high genetic risk for the disorder is the study of monozygotic twins. Monozygotic twins share nearly 100% of their genes with their co-twins. So, if the BD patient inherited the susceptibility genes for BD, their monozygotic twin also inherited them. However, the twin studies presented in this review have identified very few GM structural abnormalities in the unaffected co-twins of BD patients, including increased left caudate nucleus densities and thickened hippocampus, and decreased left hemispheric WM volume (Noga et al., 2001; Van Erp et al., 2012). Some studies that included dizygotic twins reported negative correlations between genetic liability score and GM density in the medial frontal gyrus, precentral gyrus, and insular cortex, positive correlations in the orbital gyrus (Van de Schot et al., 2010), decreased thickness of the hippocampus and orbitofrontal cortex, or increased thickness of the temporoparietal and superior motor cortices (Hulshoff Pol et al., 2012). These findings may be the most striking and compelling evidence that genetic transmission of BD may result in vulnerable brain areas in fronto-limbic networks that subserve cognitive and emotional regulation. However, it should also be noted that one twin-pair study found no evidence of decreased GM volumes in the unaffected co-twins of BD patients (Van de Schot et al., 2009). An ideal study would be a prospective neuroimaging study of discordant twin pairs to attempt to disentangle the neurobiological pathways that go from the same genetic background in twins to a fully expressed disease in one and an aborted disease process in another.
Another aspect that may underlie the lack of consistency across findings is the clinical heterogeneity of BD (i.e. BD type I vs. type II, psychotic vs. nonpsychotic, and the presence of diverse psychiatric comorbidities). Most of the studies in this review included BD type I patients, but some also included BD type II or schizoaffective-type manic patients (Hulshoff et al., 2012; Kieseppä et al., 2002; Ladoucer et al., 2008), and several also included different Axis I comorbidities in the BD probands. Some evidence suggests that the clinical characteristics of BD, such as BD subtype, presence of lifetime psychotic symptoms, or presence of Axis I comorbidities may be associated with differences in brain structure (Ha et al., 2009; Nery et al., 2011; Stanfield et al., 2009; Tost et al., 2010). The clinical heterogeneity of BD is even more important in the study of endophenotypes, as several clinical studies have reported that the clinical aspects of BD, including BD subtype, psychotic features, and comorbid alcohol use disorders, have familial aggregation and, therefore, may themselves be an expression of specific genetic susceptibilities (Hua et al., 2011; Saunders et al., 2008; Schulze et al., 2006). It is important to note that the clinical heterogeneity of BD has been identified as an important confounder in neuroimaging studies of BD (Strakowski et al., 2012). Therefore, a narrower definition of the disease (e.g. including only BD type I without Axis I comorbidities in probands and their relatives) may improve the detection of brain structural abnormalities in BD patients and in their relatives.
Another potential source of discrepancy between studies that should be considered is whether additional Axis I psychiatric disorders exist among the unaffected relatives of BD patients. Only a few of the studies reported here included only the healthy relatives of BD patients (Hajek et al., 2008a; Hajek et al., 2009; Noga et al., 2001). The importance of this issue is that the inclusion of relatives of BD patients that present with another Axis I psychiatric disorder may add a confounding variable, i.e. the effect of that specific Axis I psychiatric disorder on brain structure or function. For instance, first-degree relatives of BD patients are also at increased risk to present major depressive disorder (unipolar depression) compared to the general population (Smoller and Finn, 2003). It would not be possible to differentiate brain abnormalities in unipolar relatives of BD patients due to the genetic susceptibility to BD from those due to their major depressive disorder (Kempton et al., 2011). On the other hand, including only the healthy relatives of BD patients may include only those relatives who either did not inherit enough genes to show any detectable abnormality or those who inherited or developed protective mechanisms to overcome any inheritable genetic vulnerabilities. In this sense, it is tempting to consider that observations of increased caudate nucleus, insular cortex, or inferior frontal gyrus volumes, or increased thickness of the hippocampus found in the unaffected relatives, offspring, or co-twins of BD patients by some of the studies reviewed here (Hajek et al., 2013; Noga et al., 2001; Van Erp et al., 2012) represent protective mechanisms related to neuroplasticity and cellular resilience, as increased GM volume in magnetic resonance imaging studies has been interpreted as an effect of compensatory changes related to cellular plasticity, as opposed to decreased GM as a correlate of cell or tissue loss or atrophy (Kapczinski et al., 2008; Lyoo et al., 2010; Moore et al., 2009; Sassi et al., 2002).
Is it possible that the genetic risk for BD influences the development of vulnerable or weaker brain areas or circuits in individuals that carry the putative pool of genes that causes BD? In the human cerebral cortex, GM is made up essentially of neurons and glial cells, in a ratio between 1.2 and 1.6, with glial cells outnumbering neurons (Herculano-Houzel, 2009; Pelvig et al., 2008). Neurotrophins may play an important role in brain cell development, as important neurotrophins (including brain-derived neurotrophic factor) are associated with the formation of new neurons or glial cells in animal models, and are decreased in the manic and depressive phases of BD (Cunha et al., 2006; Duman and Monteggia, 2006; Huang and Reichardt, 2001; Machado-Vieira et al., 2007; Schmidt and Duman, 2010). To date, several association and genome-wide association studies have identified few genes and polymorphisms that might be related to BD, including: brain-derived neurotrophic factor (BDNF); D-amino acid oxidase activator (DAOA); disrupted in schizophrenia 1 (DISC1); GRIA4 glutamate receptor, ionotropic, AMPA 4 (GRIA4); solute carrier family 6 (neurotransmitter transporter, serotonin), member 4 (SLC6A4); tryptophan hydroxylase 2 (TPH2); diacylglycerol kinase, eta (DGKH); calcium channel, voltage-dependent, L type, alpha 1C subunit (CACNA1C); and ankyrin 3, node of Ranvier (ankyrin G) (ANK3) (Barnett and Smoller, 2009; Schulze, 2010). Some of these genes participate in pathways of cell formation, renovation, or death, such as BDNF, or the myelin basic protein (MBP) gene, which encodes a protein that is a major constituent of the myelin sheath of Schwann cells in the central nervous system, and the glycogen synthase kinase 3 beta (GSK3B) gene, which encodes the glycogen synthase kinase 3 (GSK3) protein, a second messenger involved in intracellular pathways related to cell apoptosis, neuronal survival, and axonal outgrowth (Le-Niculescu et al., 2009; Schloessler et al., 2008). Therefore, it is possible that a malfunctioning gene or polymorphism may induce abnormal brain development of a brain region, which may later on translate abnormally. In fact, a recent study of gene expression in the postmortem brains of BD patients showed upregulation of genes related to neuronal development, differentiation, and protection (Chen et al., 2012). The authors hypothesized that the etiology of major psychosis could be related to developmental dysfunction in the cerebral cortex. This finding adds to the evidence that the pathophysiology of BD relies on the abnormal development of brain areas and consequent brain function.
Studies of individuals at high genetic risk for BD who have not developed the disease are also limited by another important confounder. It has been hypothesized that the environment plays a significant role in facilitating or attenuating gene expression (Bagot and Meaney, 2010; Feinberg, 2007). Though gene expression does influence brain structure, an epigenetic influence by the environment is also possible. For instance, an interesting study using diffusion tensor imaging in 705 twins aged 12–29 found that the heritability of WM integrity, as measured by fractional anisotropy, is higher in adolescence and lower in adulthood (Chiang et al., 2011). This finding suggests that, after adolescence and during adulthood, environmental influences outweigh genetic influences on brain structure and function. Therefore, exposure to the environment, including learning, education, life experiences, diet, exercise, and smoking, are all postulated to dominate and influence axonal fiber networks as one progresses into adulthood (Chiang et al., 2011). Obviously, these assumptions and the interactions between environmental factors and the genetic expression of BD influencing brain structure in individuals at high risk for the disease are still to be tested.
Another aspect of environment vs. gene interplay is that at least for certain aspects of brain function, such as cognitive aptitude and intelligence quotient (IQ), the genetic influence varies with specific environmental experiences. For instance, the heritability of IQ is higher among families with higher socioeconomic status and lower in families near or below poverty levels (Harden et al., 2007; Turkheimer et al., 2003). Based on these findings, and for some behavioral traits that are advantageous, such as IQ, it has been hypothesized that the genetic potential of an individual might be maximized if adverse environmental factors are reduced as much as possible (Turkheimer et al., 2003). Translating these findings to BD, is it possible that the genetic potential of an individual to express BD might be minimized if adverse environmental factors are also reduced? If so, the possible influence of environmental factors on brain structure or function in individuals at high risk for BD is an important area for future research efforts to increase resilience strategies for such individuals.
This article reviewed the current literature on abnormal brain structure in unaffected relatives of BD patients. Figure 1 shows the brain areas in which abnormal structural findings have been reported in this population. Despite a considerable literature, very few findings have been replicated, with the exception of a larger insular cortex and right inferior frontal gyrus in individuals at increased risk to develop BD. Possible reasons for the discrepant findings across studies were discussed, including the small effect size of susceptibility genes, the phenotypic heterogeneity of BD, and the possible confounding effect of other Axis I psychopathologies among relatives of patients. The study of individuals at risk for BD is a fascinating area for research. A key strategy to advance the field will be to mount multisite studies that are able to recruit large numbers of subjects, allowing the analysis of more homogeneous samples, and more importantly, in a prospective fashion. The ultimate benefit of such research would be to understand how to identify individuals at increased risk of developing BD using brain imaging tools and how to prevent the development of the disorder in those at risk.
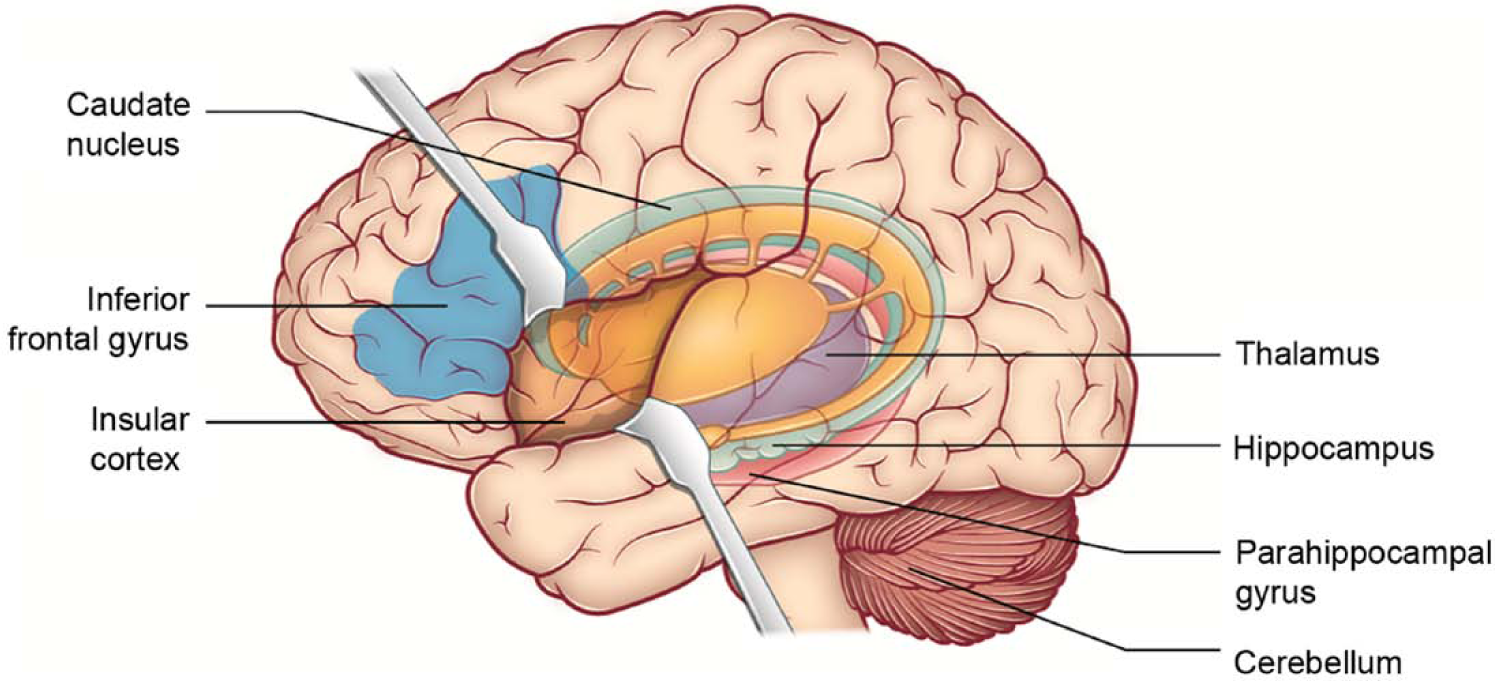
Abnormal brain areas in unaffected relatives of bipolar disorder patients. The figure shows the location of abnormal brain structures in first-degree unaffected relatives of bipolar disorder patients, as reported by neuroimaging studies. For reasons of clarity, the specific laterality of each finding is not shown. Findings include: larger left insular cortex volumes; larger inferior frontal gyrus; larger left caudate nucleus volumes; larger left cerebellum; decreased gray matter density in the left thalamus; decreased gray matter volumes in the left hippocampus and parahippocampal gyrus; and thicker right hippocampus in unaffected first-degree relatives of bipolar disorder patients compared with healthy controls.
Footnotes
Funding
This study was partly supported by a Conselho Nacional de Pesquisa (CNPq) grant # 478466/2009, Conselho de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) grant # 2632/09-8, and from a generous private donation from the family of Thompson Motta [to the Bipolar Disorder Program (PROMAN)].
Declaration of conflicting interest
Dr Nery held a temporary position of medical advisor in Eli Lilly from July 2012 to May 2013. Dr Monkul is an employee and company shareholder of Eli Lilly. The authors declare no conflict of interests within the scope of this article.
