Abstract
Background:
Elderly bipolar disorder (BD) and behavioural variant of frontotemporal dementia (bvFTD) may exhibit similar symptoms and both disorders are characterized by selective abnormalities in cortical and subcortical regions that are associated with cognitive and emotional impairments. We aimed to investigate common and distinct neural substrates of BD and bvFTD by coupling, for the first time, magnetic resonance imaging (MRI) and positron emission tomography (PET) techniques.
Methods:
3-Tesla MRI and 18 fluorodeoxyglucose–PET scans were acquired for 16 elderly BD patients, 23 bvFTD patients with mild cognitive impairments and 68 healthy controls (48 for PET and 20 for MRI analyses).
Results:
BD and bvFTD patients exhibit a different localization of grey matter reductions in the lateral prefrontal cortex, with the first group showing grey matter decrease in the ventrolateral prefrontal cortex and the latter group showing grey matter reductions in the dorsolateral prefrontal cortex as well as unique grey matter and metabolic alterations within the orbitofrontal cortex. The bvFTD group also displayed unique volumetric shrinkage in regions within the temporo-parietal network together with greater metabolic impairments within the temporal cortex and more extensive volumetric and metabolic abnormalities within the limbic lobe. Finally, while the BD group showed greater grey matter volumes in caudate nucleus, bvFTD subjects displayed lower metabolism.
Conclusion:
This MRI-PET study explored, for the first time to the best of our knowledge, structural and functional abnormalities in bvFTD and elderly BD patients, with the final aim of identifying the specific biological signature of these disorders, which might have important implications not only in prevention but also in differential diagnosis and treatment.
Keywords
Introduction
The behavioural variant of frontotemporal dementia (bvFTD) is characterized by a predominant behavioural alteration mainly due to progressive degeneration of the orbitomesial surface of the frontal lobe and the anterior temporal cortex (Rascovsky et al., 2011). Notably, psychiatric conditions, such as bipolar disorder (BD), must be excluded in order to diagnose bvFTD (Rascovsky et al., 2011). In this regard, Woolley et al. (2011) showed that patients with bvFTD had higher rates of previous psychiatric diagnoses (50.7%) compared to other neurodegenerative disorders and this was more evident in those bvFTD patients with younger age, higher education and family history of psychiatric diseases. Furthermore, affective symptoms observed in these patients, such as apathy, social withdrawal, disinhibition, inappropriate behaviours and impulsivity, could be ascribed to BD in depressive or hypomanic phases (Rascovsky et al., 2011). In addition, psychotic symptoms, which may lead the clinicians towards a diagnosis of a psychiatric disease (e.g. BD type I and schizophrenia), might also be present in bvFTD patients. Indeed, some authors showed that delusions, hallucinations and suspiciousness could reach a prevalence of 22.7% in bvFTD patients, with a higher prevalence of negative psychotic symptoms (95.5%) (Gossink et al., 2017). Furthermore, cognitive impairments in executive functions, verbal memory and emotional processing may affect both bvFTD (Rascovsky et al., 2011) and BD (Martino et al., 2013). With regard to cerebral abnormalities, bvFTD patients showed atrophy in both cortical regions, especially within frontal, parietal and temporal cortices, and subcortical ones, mainly in caudate and putamen (Rohrer, 2012), coupled with frontal hypometabolism and frontotemporal hypoperfusion (Vitali et al., 2008). Similarly, the key grey matter (GM) abnormalities associated with BD were located in prefrontal cortex (PFC), orbitofrontal cortex (OFC), anterior cingulate cortex (ACC) and temporo-limbic structures (Hibar et al., 2017), whereas metabolic alterations were detected in cortico-limbic regions (Altamura et al., 2017). Nonetheless, only one recent study has directly compared those two clinical populations, demonstrating greater cognitive deficits and GM volume reductions, especially in frontal, temporal and parietal regions, in bvFTD compared to elderly BD patients (Baez et al., 2017).
In this context, the aim of this study was to explore, for the first time to the best of our knowledge, common and distinct GM and metabolic deficits in bvFTD and elderly BD by coupling structural magnetic resonance imaging (MRI) and positron emission tomography (PET) techniques. We hypothesized that bvFTD and elderly BD patients will show similar alterations in regions within the fronto-temporo-limbic network compared to healthy controls (HC), with bvFTD patients having, though, wider alterations compared to elderly BD.
Methods
Participants
MRI and PET scans for 16 elderly BD patients and 23 bvFTD patients with mild cognitive impairments were acquired. BD patients were recruited at the psychiatric inward of the University Policlinico Hospital of Milan, Italy. Elderly BD patients fulfilled the diagnostic criteria of the Diagnostic and Statistical Manual of Mental Disorders, 4th edition, text revision (DSM-IV-TR; American Psychiatric Association, 2000), based on the Structured Clinical Interview for Diagnosis (SCID-I; First et al., 2002a, 2002b). Clinical symptoms were evaluated with the Hamilton Depressive Rating Scale (HDRS; Hamilton, 1960) and the Young Mania Rating Scale (YMRS; Young et al., 1978). All elderly BD patients were in euthymic phase at the time of the scanning (HDRS < 10 and YMRS < 12) and only 3 elderly BD patients had a history of past psychotic symptoms. Exclusion criteria for all elderly BD patients were a diagnosis of mental retardation, any current major medical or neurological illness, a history of traumatic head injury with loss of consciousness and any other Axis-I and Axis-II disorders.
The bvFTD patients were recruited at the Alzheimer Unit of the Fondazione Ca’ Granda, IRCCS Ospedale Maggiore Policlinico, University of Milan. All patients underwent a general and neurological examination, a formal neurocognitive assessment (including Mini–Mental State Examination (MMSE), Trail Making Test, digit span test, Corsi’s spatial test, Clock Drawing Test, Rey complex figure copy, Free and Cued Selective Reminding Test, verbal fluency test and Boston Naming Test) and a structural neuroimaging examination with a brain computed tomography (CT), a MRI and, whenever needed, a PET scan. In addition, a detailed past medical history and blood samples were acquired. The presence of significant vascular brain damage (Hachinski Ischemic Score < 4) and comorbidities were excluded. The diagnosis of probable bvFTD was made according to current criteria (for details, see Rascovsky et al., 2011). The bvFTD patients employed in the MRI and PET analysis only partially overlap (9 bvFTD patients out of 23). Nonetheless, all bvFTD patients had similar age, gender ratio and a mild cognitive impairment, which ranges from an MMSE score of 24.4 ± 0.5 (mean ± standard deviation) for the MRI group to a score of 27 ± 1.8 for the PET group. Moreover, while all bvFTD patients in the PET group had a score of 0.5 in the Clinical Dementia Rating Scale (CDR), 6 out of 21 patients in the MRI group had a higher score (CDR = 1).
At the time of the scan, all the patients were on stable pharmacological treatment with different psychotropic compounds. Specifically, within the group of elderly BD patients, 9 were taking antidepressants (Venlafaxine up to 150 mg per day, Mirtazapine up to 30 mg per day, Sertraline up to 100 mg per day, Citalopram up to 20 mg per day, Clomipramine up to 75 mg per day and Trazodone up to 300 mg per day), 9 were taking mood stabilizers (Carbolithium up to 1050 mg per day, Valproate up to 1000 mg per day and Lamotrigine up to 200 mg per day), 8 were taking atypical antipsychotics (Aripiprazole up to 15 mg per day, Quetiapine up to 400 mg per day and Asenapine up to 20 mg per day) and 2 were taking typical antipsychotics (Haloperidol up to 3 mg per day).
Within the group of bvFTD patients employed for the MRI analysis, 3 were taking antidepressants (Citalopram up to 30 mg per day and Fluoxetine up to 20 mg per day), 1 was taking atypical antipsychotics (Quetiapine up to 50 mg per day), 1 was taking typical antipsychotics (Haloperidol up to 2.2 mg per day), 1 was taking levodopa and the decarboxylase inhibitor benserazide (Madopar up to 1000 mg per day) and 2 were taking acetylcholinesterase inhibitor (Rivastigmine up 9.5 mg per day). Within the group of bvFTD used in the PET study, 7 were taking antidepressants (Citalopram up to 20 mg per day, Paroxetine up to 30 mg per day and Amitriptyline up to 10 mg per day), 6 were taking atypical antipsychotics (Quetiapine up to 50 mg per day), 2 were taking typical antipsychotics (Haloperidol up to 0.7 mg per day and Promazine up to 10 mg per day), 1 was taking decarboxylase inhibitor benserazide (Madopar up to 1000 mg per day) and 2 were taking acetylcholinesterase inhibitors (Rivastigmine up 9.5 mg per day). Refer to Table 1 for a more detailed socio-demographic, clinical and cognitive description of the sample.
Socio-demographic, clinical and cognitive characteristics of the sample.
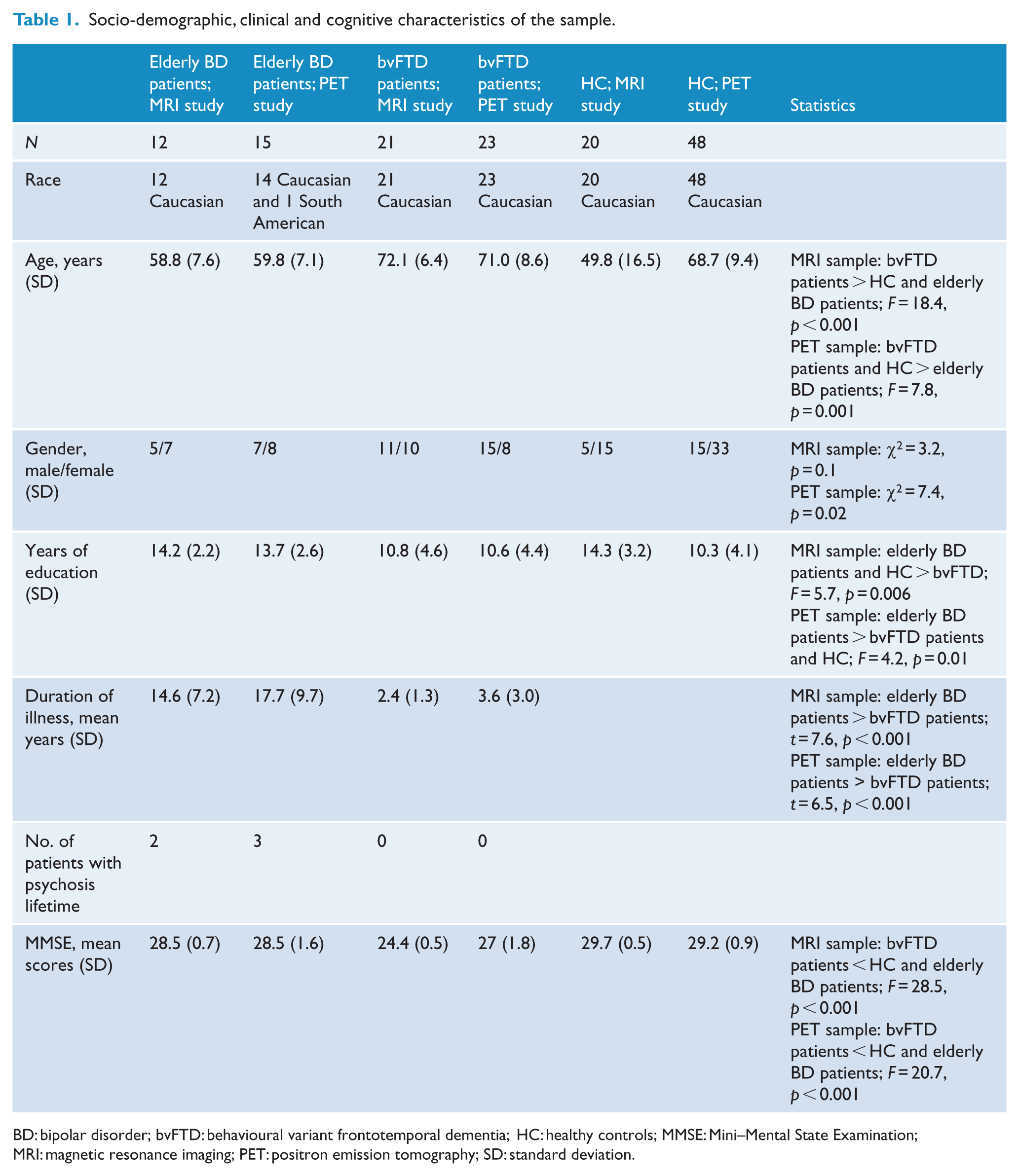
BD: bipolar disorder; bvFTD: behavioural variant frontotemporal dementia; HC: healthy controls; MMSE: Mini–Mental State Examination; MRI: magnetic resonance imaging; PET: positron emission tomography; SD: standard deviation.
Finally, we included a group of HC based on the absence of a personal or family lifetime history of any psychiatric disorder. Two independent samples of 20 HC for the MRI and 48 HC for PET were included for the analyses. The general cognitive status of all participants was also assessed using the MMSE (Folstein et al., 1975). All controls had a MMSE ⩾ 28/30. The study was approved by the local ethical committee. Informed consent was obtained for all participants. This clinical study was conducted complying with the principles of the Declaration of Helsinki (version Seoul, 2008).
MRI
Acquisition parameters
MR images were acquired using a 3-Tesla Philips Achieva MRI scanner. All the participants reclined in a supine position on the bed of the scanner and a radio frequency (RF) coil (Bruker NMR Instruments Inc., Fremont, CA) was placed over their head. Earplugs and headphones were provided to block background noise. Following a three-plane gradient echo scan for alignment and localization, a shim procedure was performed to generate a homogeneous, constant magnetic field. A total of 185 contiguous 1 mm sagittal slices extending superiorly from the inferior aspect of the cerebellum to encompass most of the brain were selected from a sagittal localizer scan. A high-resolution, T1-weighted, three-dimensional (3D) brain scan was then obtained (repetition time [TR] = 9.7, echo time [TE] = 4.5 ms, field of view [FOV] = 25.6 × 25.6 × 18.5 cm3, flip angle = 8°).
PET
Acquisition parameters
PET scans were obtained with a Biograph Truepoint 64 PET/CT scanner (Siemens, Erlangen, Germany) at the Nuclear Medicine Department of Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico in Milan. All patients underwent 18 fluorodeoxyglucose (FDG)-PET scanning at rest after intravenous injection of 170 MBq. Each acquisition included a CT transmission scan of the head (50 mAs lasting 16 s) followed by a 3D static emission of 15 min using a Biograph Truepoint 64 PET/CT scanner (v). PET sections were reconstructed in the form of transaxial images of 128 × 128 pixels of 2 mm, using an iterative algorithm, ordered subset expectation maximization, corrected for scatter and for attenuation using density coefficients derived from the low-dose CT scan of the head obtained with the same scanner, with the proprietary software. The resolution of the PET system was 4–5 mm full width at half maximum (FWHM).
Neuroimaging data analysis
Pre-processing
Different pre-processing steps were carried out for the voxel-based morphometry (VBM) and the PET analysis using statistical parametric mapping (SPM12; www.fil.ion.ucl.ac.uk/spm/software/spm12/) implemented in MATLAB R2017a (MathWorks Inc., Natick, MA). For VBM analyses, all T1-weighted images were segmented according to GM, white matter and cerebrospinal fluid, bone, soft tissue and air/background. Second, the DARTEL (Diffeomorphic Anatomical Registration Through Exponentiated Lie algebra) tools were then used to determine the nonlinear deformations for registering the GM and white matter images of all subjects. Finally, the resulting images were spatially normalized into the Montreal Neurological Institute (MNI) space and smoothed with an isotropic Gaussian kernel of 6 mm FWHM. For PET analyses, all FDG-PET images were subjected to affine and nonlinear spatial normalization into the MNI space with SPM12 using the dementia-optimized brain FDG-PET template as described by Della Rosa et al. (2014). Finally, all normalized PET images were smoothed with an 8-mm isotropic Gaussian filter to blur individual variations in gyral anatomy and to increase the signal-to-noise ratio.
Statistical analyses
First, to investigate the presence of differences between the groups on demographic and clinical variables, we performed a chi-square test for categorical variables and analysis of variance (ANOVA) or two sample t tests for quantitative variables (Table 1). Second, for both the VBM and PET analyses, we performed a one-way ANOVA with nuisance covariates of age and gender. The volumetric differences among subjects were considered by proportional scaling for the total intracranial volume (ICV) and the metabolic differences across subjects and scans were removed by scaling each image to have an arbitrary level of 50 with the threshold masking relative to the default value 0.8. Inference on the GM volumes and metabolic differences between groups was made using double-sided t-tests with a threshold of p < 0.001 and a minimum cluster size of k > 150 for both the analyses (Tables 2 and 3). Stereotactic coordinates of the peak maxima of the suprathreshold clusters were converted from the MNI spatial array (www.mni.mcgill.ca) to that of Talairach and Tournoux (1988).
VBM results: Brain regions showing significant gray matter volume alterations between the three groups of subjects (p < 0.001, cluster size k > 150).
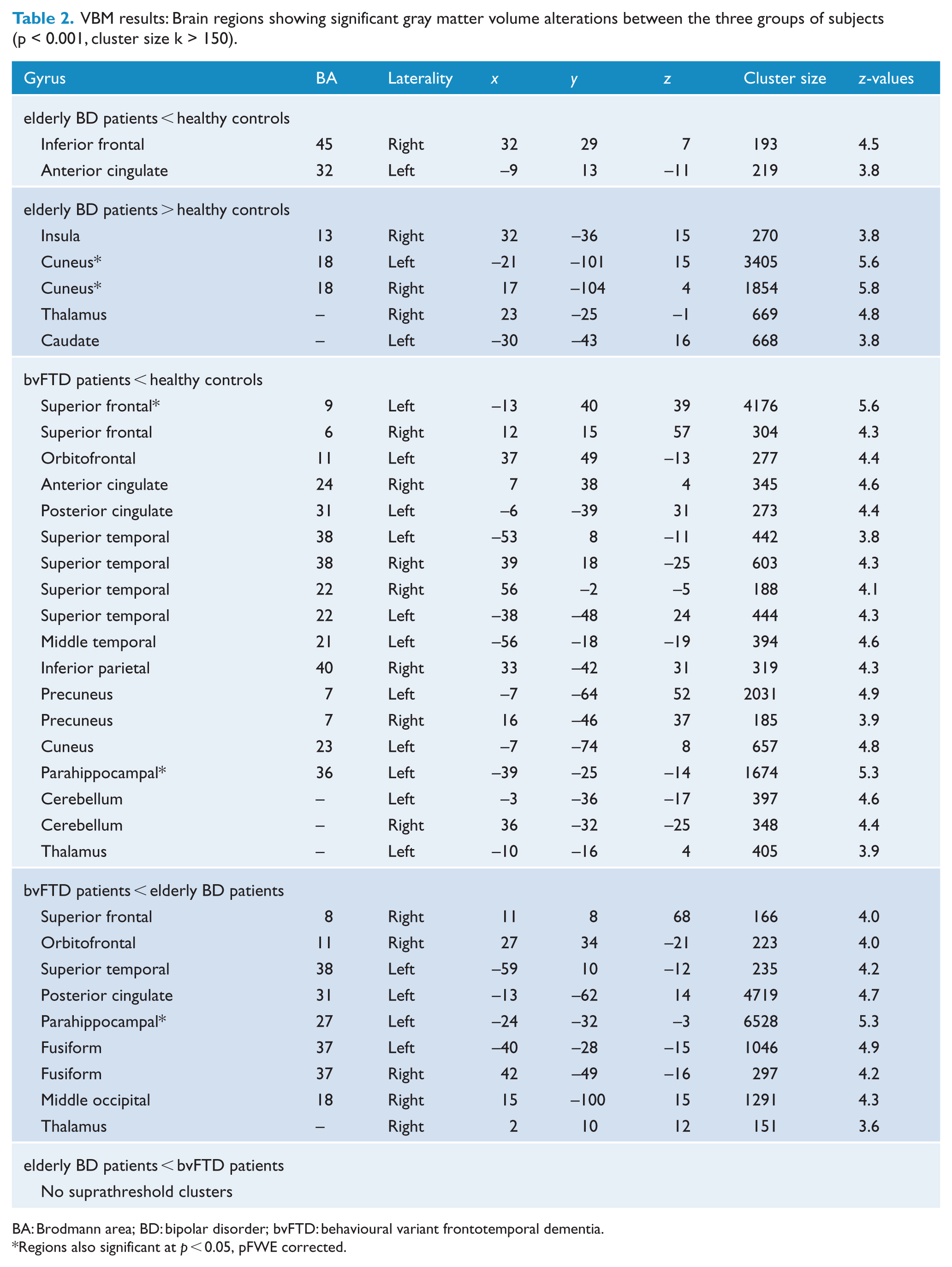
BA: Brodmann area; BD: bipolar disorder; bvFTD: behavioural variant frontotemporal dementia.
Regions also significant at p < 0.05, pFWE corrected.
PET results: Brain regions showing significant metabolic alterations between the three groups of subjects (p < 0.001, cluster size k > 150).
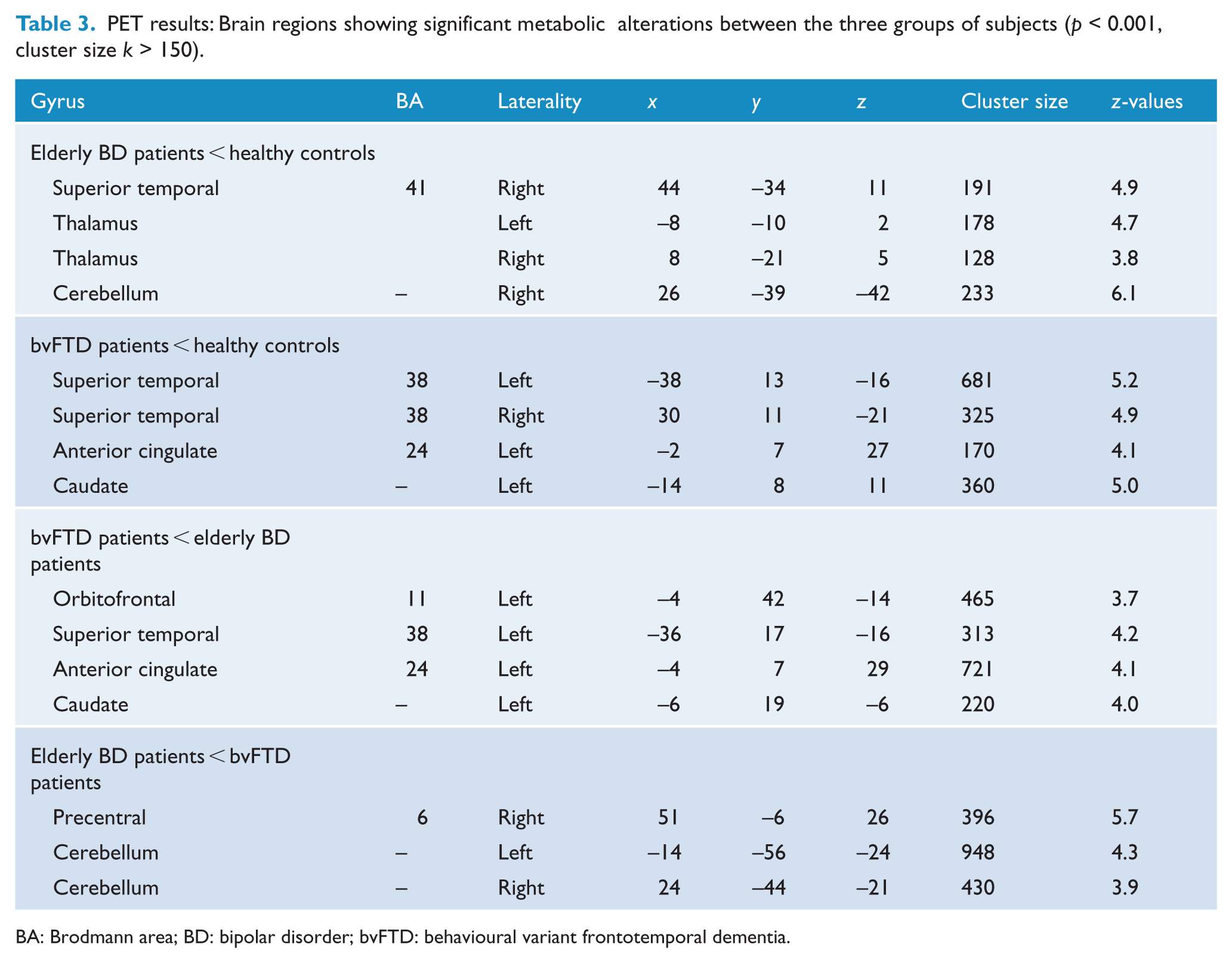
BA: Brodmann area; BD: bipolar disorder; bvFTD: behavioural variant frontotemporal dementia.
Results
A total of 16 elderly BD patients, 23 bvFTD patients and 68 HC (48 for PET and 20 for MRI analyses) were enrolled in this study. However, after the quality check, five elderly BD (four for the MRI and one for the PET analysis) and two bvFTD patients (only for the MRI analysis) were excluded from the analyses. Therefore, the MRI analysis was performed on 12 elderly BD patients and 21 bvFTD patients, whereas the PET analysis was carried out on 15 elderly BD patients and 23 bvFTD patients.
Analysis of socio-demographic and clinical variables
For both the MRI and PET analyses, we observed significant group differences in terms of age. Specifically, in the groups considered for the MRI analysis, we found that bvFTD patients had higher mean age compared to both elderly BD patients and HC (F = 18.4, p = 0.001). Similarly, in the groups employed in the PET study, we found that both bvFTD patients and HC showed higher mean age compared to elderly BD patients (F = 7.8, p = 0.001). Moreover, only for the PET study, a significant gender difference was observed between the three groups (χ2 = 7.4, p = 0.02). Finally, as expected, we observed that bvFTD patients had lower MMSE scores compared to both elderly BD patients and HC, as well as lower duration of illness compared to elderly BD patients in both the MRI and PET investigations (p < 0.001).
Analysis of MRI data
Group comparisons
Elderly BD patients vs HC
Compared to HC, elderly BD patients showed abnormally reduced GM volumes in right inferior (BA45) frontal gyrus (BA45) and left ACC (BA32). In addition, BD patients showed increased GM volumes in right insula, right thalamus, left caudate and cuneus bilaterally (BA18) (p < 0.001, cluster size k > 150). Notably, the clusters within the cuneus survived a more stringent threshold of p < 0.05 peak family-wise error (pFWE) corrected (Table 2; Figure 1).
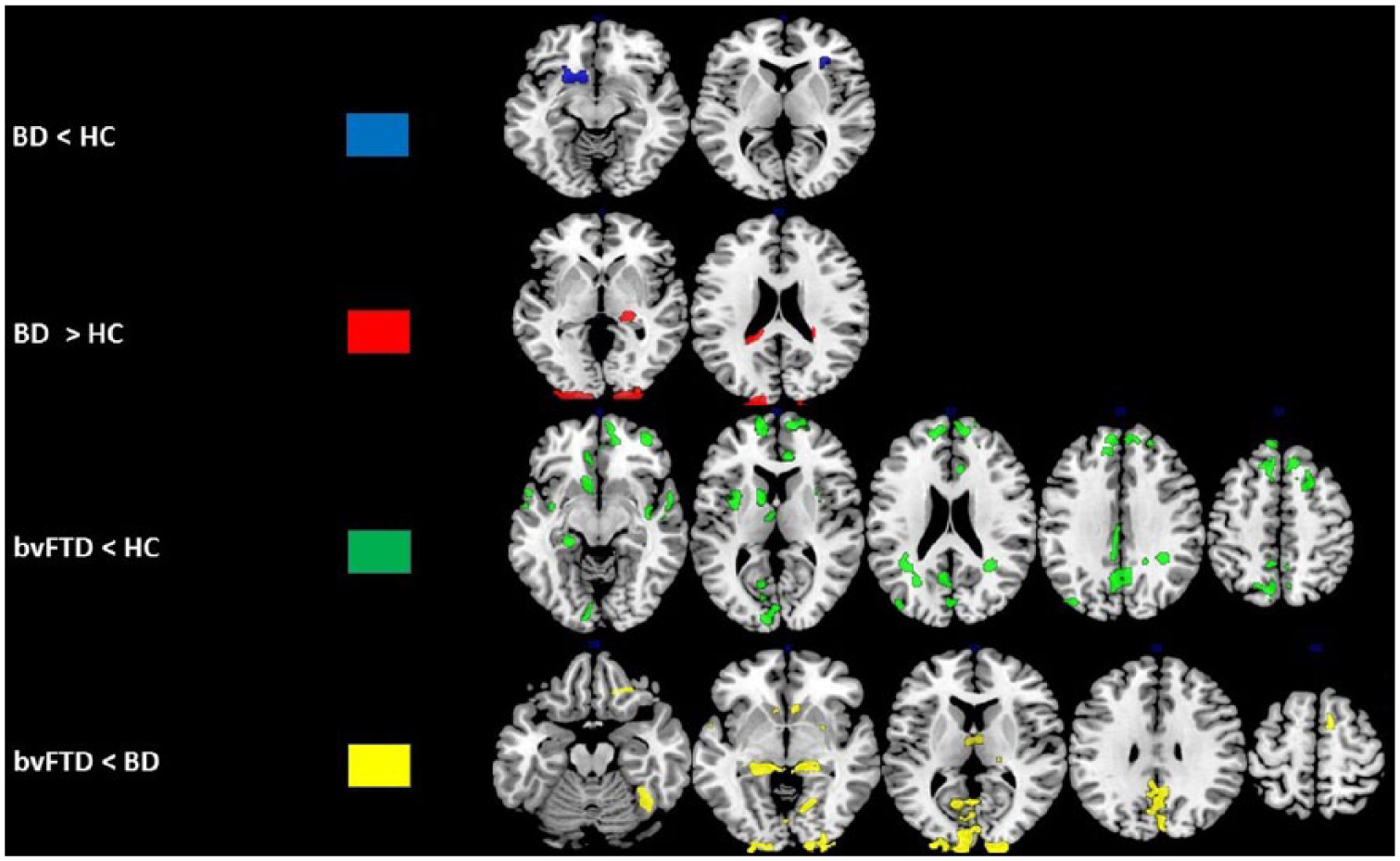
Brain regions showing GM reductions in elderly bipolar disorder (BD) and behavioural variant frontotemporal dementia (bvFTD) compared to healthy controls (HC) (p < 0.001, cluster size k > 150).
bvFTD patients vs HC
Compared to HC, bvFTD patients showed abnormally reduced GM volumes in superior frontal gyrus bilaterally (BA9/BA6), left OFC (BA11), left posterior (BA31) and right anterior (BA24) cingulate cortex, superior temporal gyrus bilaterally (BA38/BA22), left middle temporal gyrus (BA21), right inferior parietal lobule (BA40), precuneus bilaterally (BA7), left cuneus (BA23), left parahippocampal gyrus (BA36), left thalamus and cerebellum bilaterally (p < 0.001, cluster size k > 150). Notably, the clusters within the left superior frontal gyrus (BA9) and left parahippocampal gyrus survived a more stringent threshold of p < 0.05 pFWE corrected.
bvFTD patients vs elderly BD patients
Compared to elderly BD patients, bvFTD patients showed abnormally reduced GM volumes in right superior frontal gyrus (BA8), right OFC (BA11), left superior temporal gyrus (BA38), left posterior cingulate cortex (PCC; BA31), left parahippocampal gyrus (BA27), fusiform gyrus bilaterally (BA37), right thalamus and right middle occipital gyrus (BA18) (p < 0.001, cluster size k > 150). Notably, the cluster within left parahippocampal gyrus (BA27) survived a more stringent threshold of p < 0.05 pFWE corrected. In contrast, no significant GM volume reductions were observed in elderly BD patients compared to bvFTD patients.
Analysis of PET data
Group comparisons
Elderly BD patients vs HC
Elderly BD patients had abnormally decreased GM metabolism in right superior temporal gyrus (BA41), thalamus bilaterally and right cerebellum (p < 0.001, cluster size k > 150) (Table 3; Figure 2).
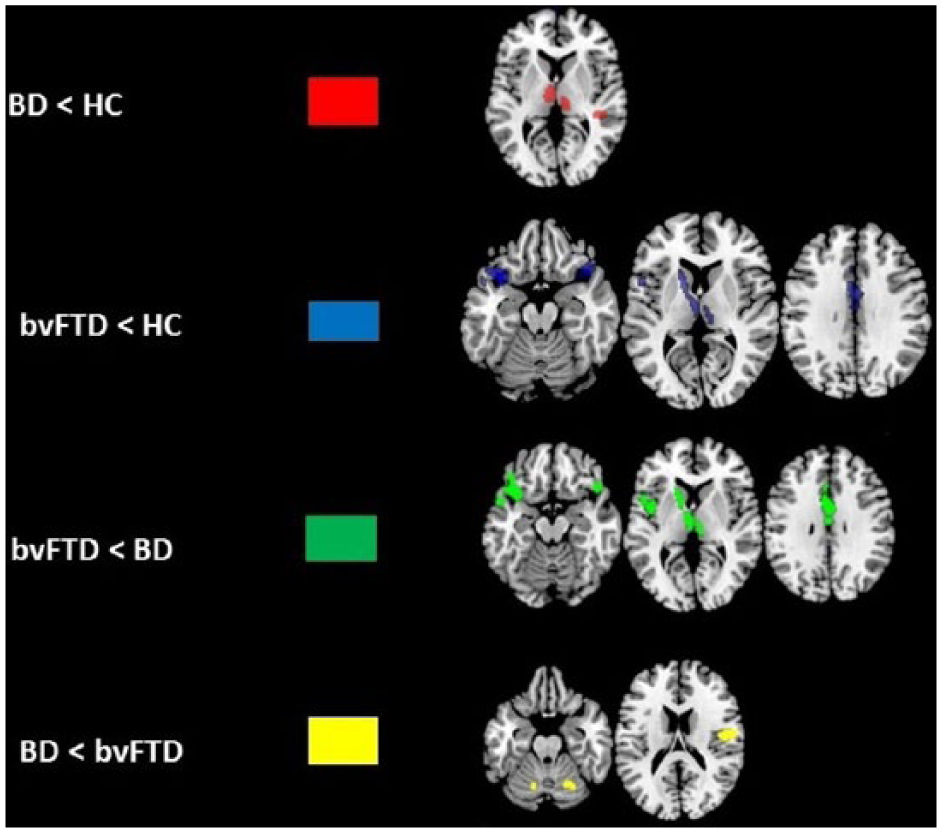
Analysis of variance maps showing metabolic reductions in elderly bipolar disorder (BD) and behavioral variant frontotemporal dementia (bvFTD) compared to healthy controls (HC) (p < 0.001, cluster size k > 150).
bvFTD patients vs HC
Compared to HC, bvFTD patients had abnormally decreased GM metabolism in superior temporal gyrus bilaterally (BA38), left ACC (BA24) and left caudate (p < 0.001, cluster size k > 150).
bvFTD patients vs elderly BD patients
Compared to elderly BD patients, bvFTD patients had abnormally decreased metabolism in left OFC (BA11), left superior temporal gyrus (BA38), left ACC (BA32) and left caudate (p < 0.001, cluster size k > 150).
Elderly BD patients vs bvFTD patients
Compared to bvFTD patients, elderly BD patients showed decreased metabolism in right precentral gyrus (BA6) and cerebellum bilaterally (p < 0.001, cluster size k > 150).
Discussion
To our knowledge, this is the first study to explore both GM and metabolic alterations in elderly BD and bvFTD patients by coupling MRI and PET techniques. Common and distinct GM and metabolic alterations have been found in elderly BD and bvFTD patients. First, the two groups exhibited different localization of GM reductions in the lateral PFC, with elderly BD patients showing greater atrophy within ventrolateral prefrontal cortex (VLPFC) and bvFTD patients within dorsolateral prefrontal cortex (DLPFC). In addition, bvFTD subjects showed volumetric and metabolic reductions within OFC. Second, the bvFTD group displayed unique volumetric shrinkage in regions within the temporo-parietal network together with greater metabolic impairments within the temporal cortex. Third, more extensive volumetric and metabolic abnormalities were found in bvFTD patients in limbic areas, especially in parahippocampal and cingulate regions. Fourth, a common involvement of caudate nucleus was detected in both disorders, with elderly BD group showing greater GM volumes, whereas bvFTD patients displaying lower metabolism.
Different localization of GM and metabolic alterations in PFC between elderly BD and bvFTD
We found that while BD patients showed decreased GM volumes in VLPFC, bvFTD patients displayed wider GM reductions in DLPFC and OFC, compared to both HC and elderly BD patients. Moreover, bvFTD patients showed metabolic reductions within OFC compared to elderly BD patients. Interestingly, our results are partially in line with a recent study carried out by Baez et al. (2017), which reported GM volume reductions in PFC in bvFTD patients compared to both HC and elderly BD patients. In general, our findings are not surprising since previous studies consistently found PFC GM reductions, coupled with deficits in executive functions, in both BD (Hanford et al., 2016) and bvFTD (Raust et al., 2014) patients.
However, the distinct localization of GM reductions within the PFC between elderly elderly BD and bvFTD may reflect the presence of different cognitive and behavioural dysfunctions characterizing these disorders.
Indeed, the VLPFC is involved in goal-directed behaviour and in the integration of emotional processing (Sakagami and Pan, 2007) and atrophy in this structure has been consistently reported in BD (Selvaraj et al., 2012), also in a large collaborative study (Hibar et al., 2017). Moreover, functional MRI (fMRI) studies found that BD patients showed deactivation of VLPFC while performing incentive decision-making tasks (Frangou et al., 2008) and during facial affect processing (Delvecchio et al., 2013) compared to HC, ultimately suggesting that VLPFC deficits might explain the presence of disinhibited and socially inadequate behaviours and greater impulsivity in these subjects.
With regard to bvFTD patients, our results are in line with previous independent MRI studies showing a tardive DLPFC shrinkage in bvFTD with mild cognitive impairments (Bertoux et al., 2015) and reductions in OFC in bvFTD patients (Berti et al., 2011). Specifically, the DLPFC holds a primary role in executive functions, including working memory and selective attention (Barbey et al., 2013), whereas OFC is a key region involved in reward expectation, social processing and behavioural inhibition (Jonker et al., 2015), all abilities often found altered in bvFTD patients (Viskontas et al., 2007).
In conclusion, all together these results suggest that GM alterations are localized in different regions within the PFC in elderly BD and bvFTD subjects. Specifically, while the disinhibited behavioural and emotional symptoms in elderly BD could be mainly ascribable to VLPFC alterations, behavioural inhibition and planning impairments in bvFTD could be related to wider prefrontal alterations.
Deficits in the temporo-parietal network: a common or unique biological marker of bvFTD and elderly BD?
Unique GM reductions in middle and superior temporal gyri as well as in inferior parietal gyrus and precuneus, coupled with hypometabolism in superior temporal gyrus, were observed in bvFTD compared to HC. In addition, bvFTD subjects displayed GM volume reductions in superior temporal gyrus and fusiform gyrus as well as metabolic deficits in superior temporal gyrus compared to elderly BD patients. Interestingly, decreased metabolism in superior temporal gyrus, although less extensive, was also observed in elderly BD patients compared to HC.
Also in this case, our findings are partially in line with independent MRI studies reporting temporal atrophy and hypometabolism (Tosun et al., 2012) and parietal GM degeneration, especially in precuneus (Morbelli et al., 2016), in bvFTD. Moreover, the study carried out by Baez et al. (2017), which explored neuropsychological and neurobiological differences between bvFTD and BD patients, demonstrated that decreased GM volumes in superior temporal gyrus in bvFTD patients correlated with impairments in executive functions. However, these correlations were not observed in BD patients, which in turn seemed to be less compromised in terms of cognitive processing. Furthermore, the extensive deficits in temporal cortex observed in our group of bvFTD patients might explain the behavioural changes characterizing this disease. Indeed, this cortex plays a key role in the prefronto-amygdalar pathway, which regulates the social cognition processes (Matsukawa and Murai, 2013), often found impaired in bvFTD with maladaptive social interactions (Piguet et al., 2011).
Notably, metabolic deficits in superior temporal cortex observed in our group of elderly BD patients have been previously observed in two studies from our research group (Altamura et al., 2017; Caletti et al., 2017), especially in patients with more severe illness and lower cognitive insight (Caletti et al., 2017). The presence of a shared alteration in this structure between elderly BD and bvFTD patients is not surprising especially because this area contains the primary auditory cortex and Wernicke’s area, two important regions implicated in speech processing and recognition of sounds, abilities often found altered in both bvFTD (Clark et al., 2017) and BD (Zenisek et al., 2015) patients. Moreover, it has also been reported that both disorders share linguistic deficits, with bvFTD patients showing reduced narrative organization manifested as tangential speech, limited story comprehension, and impaired expression of abstract words and propositional speech (Cousins et al., 2016), whereas BD patients having mostly syntactic and lexical-semantic impairments (Radanovic et al., 2013).
With regard to the parietal cortex, the presence of unique parietal abnormalities in bvFTD may be related to a greater cognitive impairment observed in this disorder. Indeed, the parietal cortex has been reported to play a major role in various cognitive domains, such as episodic memory, language, counting function and theory of mind (Cabeza et al., 2012). Interestingly, parietal volumetric alterations have been found among specific sub-populations of bvFTD patients (Risacher and Saykin, 2013), with Baez et al. (2017) also showing parietal GM volume reductions in bvFTD in association with specific theory of mind deficits.
In conclusion, these evidence suggest that extensive temporal-parietal alterations seem to characterize only our group of bvFTD patients, which may in turn explain the extensive cognitive deficits often observed in this disorder. Instead, metabolic alterations of the superior temporal gyrus could represent a common neurobiological substrate characterizing both elderly BD and bvFTD patients, with the latter, though, showing more extensive and profound alterations.
Greater volumetric and metabolic alterations within the limbic lobe in bvFTD
The bvFTD group showed extensive GM and metabolic reductions in parahippocampal gyrus and cingulate cortex compared to both HC and elderly BD patients. Specifically for bvFTD, parahippocampal alterations have been previously detected in these groups of patients (Seeley et al., 2008). This structure, as part of the limbic system, detains a major role in both cognitive functioning, especially topographical memory and spatial configural learning, and emotional regulation (RajMohan and Mohandas, 2007). Notably, GM volume reduction in parahippocampus was also found by Baez et al. (2017), who linked the atrophy in this structure with lower verbal inhibitory control and poorer affective theory of mind functioning observed in bvFTD subjects.
Moreover, a common GM deficit in ACC was observed in both bvFTD and elderly BD patients, with bvFTD patients also showing lower metabolism in this structure compared to both HC and elderly BD patients. The ACC is considered a part of the limbic lobe; it is richly connected with frontal areas thus playing a significant role in specific cognitive functions, including attention, impulse control and emotional regulation (Bush et al., 2000). Interestingly, alterations within ACC regions have been associated with dysfunctions of social behaviour and affective control in bvFTD (Seeley et al., 2008) and with emotional dysregulation and vulnerability to psychosis in BD (Lim et al., 2013).
Finally, bvFTD patients showed unique GM alterations in PCC compared to both HC and elderly BD patients. Although no consensus has been reached yet regarding its role in cognition, it has been reported that PCC is involved in various functions, ranging from preserving vigilance and attentional states to emotional processing and goal-directed behaviour (Spreng et al., 2010). However, the role of PCC in bvFTD is far to be conclusive. Indeed, the few MRI studies in bvFTD suggested that the atrophy in this region seems to be absent at earlier stages of bvFTD and to mainly occur in later stages of disease progression (Tan et al., 2013).
In conclusion, although the presence of a partial overlap of limbic deficits between bvFTD and elderly BD patients, the former group showed a more extensive alteration in limbic regions suggesting a greater cognitive, social and emotional impairment in this disorder.
Divergent alterations in caudate between bvFTD and elderly BD patients
Our results showed the presence of alterations in caudate in both disorders, with elderly BD patients showing increased GM volumes compared to HC and bvFTD patients having reduced metabolism compared to both HC and elderly BD patients. The importance of caudate in both BD and bvFTD is given to its specific role in goal-directed behaviour, evaluation of actions and emotional regulation (Graff-Radford et al., 2017).
Interestingly, caudate enlargement has been previously described in psychiatric disorders as being related to the medication effects on D2-subtype dopaminergic receptor, which are abundant in basal ganglia structures (Van Erp et al., 2016). Specifically for BD, the increased GM in caudate observed in our group of elderly BD patients is in line with previous studies (Maller et al., 2014), but not all (Hibar et al., 2017). Therefore, all together these results suggest that the caudate enlargement found in our group of elderly BD could be a consequence of ongoing use of antipsychotic drugs, which are predominantly antagonists of the D2-subtype dopaminergic receptor.
With regard to bvFTD patients, deficit in caudate may indicate a greater dysfunction of emotional-cognitive integrative network (Raczka et al., 2010) in these patients, since anatomically this structure is widely connected with several areas within this network, including the PCC, parahippocampus, superior/inferior temporal gyrus, inferior occipital gyrus and thalamus (Robinson et al., 2012), all regions found altered in bvFTD patients in this study.
Limitations
This study should be considered in light of some limitations. First, the small sample size employed in our study might have limited the statistical power of our analyses. However, our sample size was similar to the only MRI study directly comparing bvFTD and BD patients (Baez et al., 2017). Second, only for the PET study, significant gender differences were observed. However, gender was included as controlling variable in the PET analysis. Third, both BD and bvFTD patients were taking medications, therefore it is not possible to exclude the influence of pharmacological treatments on cerebral volumetric and metabolic alterations. Finally, we did not investigate cognitive functioning in both groups and therefore we could not directly correlate cerebral alterations with specific cognitive performances.
Conclusion
The close link between BD and bvFTD has been previously described since these disorders share similar symptomatology and cognitive dysfunctions which, in some cases, may entail misdiagnosis. For this reason, the approach to directly compare cerebral alterations between neurodegenerative and psychiatric disorders may provide a better understanding of neural circuits underlying BD and bvFTD with significant implications for prevention, diagnosis and treatment.
From our study emerged that (a) common PFC deficits seem to characterize both elderly BD and bvFTD patients, with, though, specific differences in terms of localization, VLPFC for elderly BD and DLPFC for bvFTD, (b) bvFTD patients showed more extensive brain alterations in limbic regions compared to elderly BD and (c) bvFTD patients hold unique volumetric and metabolic reductions in regions within the temporo-parietal network.
Therefore, our findings provide important neurobiological evidence to discriminate between bvFTD and elderly BD as distinct clinical entities and could help clinicians to make a more accurate differential diagnosis. This could be particularly relevant in case of psychotic and affective symptoms in elderly with no previous history of BD. In this regard, further studies enrolling late-onset BD patients are needed in order to clarify the presence of common cerebral abnormalities pattern in comparison with bvFTD subjects. Moreover, a discrimination between BD patients with and without cognitive impairment is warranted to better link cerebral anomalies with cognitive functioning.
Footnotes
Acknowledgements
Giuseppe Delvecchio and Gian Mario Mandolini contributed equally to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by grants from the Ministry of Health to P.B. (RF-2011-02352308) and from the Cariplo Foundation to P.B. and C.M.M. (grant 2016:0908).
