Abstract
Objective:
The aim of this study was to examine the relationship between obsessive–compulsive symptoms (OCS) and substance use in patients with a non-affective psychotic disorder.
Method:
The data were derived from the Genetic Risk and Outcome of Psychosis study. Using the Yale–Brown Obsessive Compulsive Scale, three groups of in- and outpatients with non-affective psychotic disorder (76.6% male, mean age 27.7 years, mean duration of illness 4.5 years) were distinguished: patients without OCS (N = 777), patients with mild OCS (N = 143) and patients with more severe OCS (N = 85). These three groups were compared using various substance use variables, including quantitative substance use variables and severity of substance use [Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV) misuse disorders].
Results:
We found no statistically significant differences in smoking and other substance use variables between the three patients groups according to the severity of OCS.
Conclusions:
Our large study sample and detailed comparison of substance use rates strongly adds to the evidence that schizophrenia patients with OCS do not differ in prevalence of substance use compared to patients without OCS. This suggests that in clinical practice, enquiring into (problematic) substance use is relevant in both schizophrenia patients with co-morbid OCS and patients without OCS.
Introduction
Obsessive–compulsive symptoms (OCS) and obsessive–compulsive disorder (OCD) are common in patients with schizophrenia and related disorders, with prevalence rates ranging from 7.8 to 55% for OCS (Berman et al., 1998), and a mean prevalesnce rate of 12.1% for OCD (Achim et al., 2011). This is considerably higher than the prevalence of OCS and OCD in the general population in The Netherlands, which is estimated at 5.8 and 0.9% respectively (De Bruijn et al., 2010). The higher prevalence of OCS/OCD in schizophrenia suggests a relationship between the two disorders.
Prevalence rates of substance use disorders (SUDs; including nicotine use) are higher in patients with schizophrenia compared to the general population (Buckley, 1998; De Leon et al., 2002; Dixon et al., 1991; McCreadie, 2002; Mueser et al., 1990; Roick et al., 2007; Zammit et al., 2003). A co-morbid prevalence rate of 47.0% for abuse or dependence of substances has been found in schizophrenia patients in the Epidemiologic Catchment Area Study (Regier et al., 1990).
Results concerning the prevalence of SUDs in OCD patients compared to the general population are conflicting. For example, Denys et al. (2004) found a current prevalence of 4.3% of SUDs in 420 Dutch OCD patients receiving treatment, compared to 5.8% in the Dutch general population (Bijl et al., 1998). Furthermore, Bejerot and Humble (1999) found that smoking rates were lower in OCD patients, compared to smoking rates in the general population (14.5% vs. 25.4%). The authors discussed several possible explanations for this finding. For example, smoking may be less pleasant in patients with OCD, because smoking elevates the attention span and increases the awareness of obsession in OCD, thereby reinforcing the symptoms. They also suggested that symptoms linked to OCD (e.g. fear of bodily harm and diseases) may keep subjects from smoking, and substance use may deteriorate OCS by further overactivating the frontal cortex, thus keeping patients from smoking.
In contrast to the findings of Bejerot and Humble (1999), analysis on data derived from the large Netherlands Mental Health Survey and Incidence Study (NEMESIS) revealed that lifetime prevalence of SUDs is higher in people with OCD (40.1%), compared to people without OCD (17.8%; De Bruijn et al., 2010). A more extensive analysis on the same NEMESIS data showed that the lifetime and 12-month odds of being diagnosed with a SUD in subjects with OCD were significantly higher than the odds of SUD in people without a psychiatric disorder (Blom et al., 2011). Lifetime prevalence rates for any SUD in OCD compared to people without OCD were respectively 54.6 and 26.6 % in men, and 36.1 and 13.1% in women.
In summary, OCS occur relatively frequently in schizophrenia; schizophrenia patients show elevated rates of substance use as compared to the general population; and prevalence rates of substance use in OCD patients compared to the general population vary.
It would be interesting to know what the substance use rates are in schizophrenia patients with OCS compared to substance use rates in schizophrenia patients without OCS. One study found smoking rates to be significantly lower in schizophrenia patients with co-morbid OCD compared to those without OCD (De Haan et al., 2013). However, two studies that compared smoking rates between schizophrenia patients with and without co-morbid OCS found no significant differences (Dome et al., 2006; Fawzi et al., 2007). Additionally, three studies (De Haan et al., 2013; Poyurovsky et al., 2003; Puyorovsky et al., 2008) found that substance abuse (other than nicotine) rates did not differ between schizophrenia patients with and without OCD.
In summary, relatively few and small studies reported on the prevalence rates of substance use in patient with both schizophrenia and OCS. In these studies, comparisons were limited to smoking rates and overall substance use rates. Therefore, the aim of the present study was to investigate the relationship between OCS and substance use in patients with schizophrenia in a more extensive and detailed manner. We hypothesized that schizophrenia patients with co-morbid OCS have lower substance use rates than schizophrenia patients without OCS, owing to the deteriorating effect of substance use on OC symptoms by further overactivating the frontal cortex (Bejerot and Humble, 1999). This may keep schizophrenia patients with co-morbid OCS from substance use.
Methods
Participants
Patients took part in the Genetic Risk and Outcome of Psychosis (GROUP) study, a multi-site longitudinal cohort study in The Netherlands and Belgium that focuses on vulnerability and resilience factors for variation in expression and course of treatment-seeking patients with non-affective psychotic disorders (for details, see Korver et al., 2012). Inclusion criteria for patients were: (1) age between 16 and 50 years; (2) a diagnosis of non-affective psychotic disorder according to the Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV; American Psychiatric Association, 1994); (3) onset of contact with psychiatric caregivers for psychosis less than 10 years before study entrance; and (4) good command of the Dutch language. In selected representative geographical areas in The Netherlands, patients were identified through clinicians working in regional or academic psychosis centres whose caseloads were screened for the inclusion criteria. In addition, all consecutive patients presenting at these services – either as out- or inpatients – were recruited for the study. Both men and women were recruited. All participants gave written informed consent after complete description of the study. The study was approved by the human subject review boards of all four participating academic centres.
Measures
Clinical measures
The Yale–Brown Obsessive Compulsive Scale (Y-BOCS; Goodman et al., 1989a; Goodman et al., 1989b) was used to measure the severity of OCS over the previous week. The Y-BOCS consists of 13 items: the preliminary item evaluates the occurrence of obsessions and compulsions, and when OCS is present, the interview continues with five items that examine the severity of obsessions and five items that examine the severity of compulsions. In a psychometric analysis, Boyette et al. (2011) found the same underlying two-factor structure as that proposed by Goodman et al. (1989a, 1989b), in a study sample of patients with schizophrenia and co-morbid OCS. The produced factors showed good reliability and strong correlations with the Y-BOCS total score.
In the present study, patients were categorized into three groups according to their level of OCS; one group without obsessions or compulsions, one group with OCS but Y-BOCS total scores between 1 and 15 (mild OCS), and one group with total Y-BOCS scores that equalled or exceeded 16 (severe OCS). We used the threshold of 16 because two other studies on the relationship between smoking and OCS in schizophrenia also used this threshold (Dome et al., 2006; Fawzi et al., 2007). Furthermore, this threshold is typically used for inclusion in drug trials, and typical scores for patients with OCD are in the 16–30 range (Blacker, 2009).
The severity of positive and negative symptoms in patients was rated with the Positive and Negative Syndrome Scale with mean scores for positive, negative and general symptoms (Kay et al., 1987). Duration of illness was calculated by using the time (years) since the onset of the first psychotic episode until the date of testing.
Substance use measures
Use of nicotine, alcohol, cannabis and hard drugs (stimulants, opiates, hallucinogens, cocaine and ecstasy) was assessed with the Composite International Diagnostic Interview (World Health Organization, 1994), sections B, J and L. Nicotine use was defined as daily use of cigarettes for at least one month in the past 12 months. Alcohol use in the past year was defined as having consumed more than 12 alcoholic drinks in the past 12 months. Heavy alcohol use in the past year was defined as having consumed more than 21 alcoholic units per week. Diagnosis of cannabis or hard-drug abuse or dependency at present state (= in the past year) and lifetime was made according to the DSM-IV (APA, 1994). To have objective data about the recent use of cannabis, urinalysis for the presence of tetrahydrocannabinol, amphetamines and cocaine was carried out using immunoassays with cut-offs of 50 ng/ml, 1000 ng/ ml and 300 ng/ml, respectively.
The total score of the Obsessive Compulsive Drug Use Scale was used as a measure for cannabis craving in the past week (Dekker et al., 2012).
Statistical analysis
Box plots and histograms were used to check whether continuous data were normally distributed (confirmed with the Kolmogorov–Smirnov test) and for identifying potential outliers. Two potential outliers concerning consumption of alcohol units per week were excluded for analysis. The chi-square test for independence was used to determine whether two categorical variables were related to each other. A one-way, between-groups analysis of variance was conducted to explore the differences between the three groups in one dependent (continuous) variable. The Kruskal–Wallis test was conducted to explore the differences between the three groups in one dependent variable, in case data were not normally distributed. In case of a statistically significant Kruskal–Wallis test, post-hoc comparisons with Mann–Whitney tests were used, with alpha set at 0.017 (0.05/3).
Statistical analyses were performed using SPSS version 16.0 for Windows (SPSS Inc., Chicago, Illinois, USA).
Results
Sample characteristics
The demographic and clinical variables are summarized in Table 1. Of 1005 patients in whom the Y-BOCS was assessed, 777 patients did not have OCS, 143 patients had OCS with a total Y-BOCS score between 1 and 15 (mean 11.0, SD 2.5) and 85 patients had a total Y-BOCS score that equalled or exceeded 16 (mean 19.4, SD 3.6); 76.6% of the population were male. There were no differences in the proportion of males across the three groups according to OCS. The mean age of participants was 27.7 years (SD 8.1), with no differences between the groups according to OCS. The mean duration of illness was 4.5 years (SD 4.1). Patients with a total Y-BOCS score between 1 and 15 had a longer duration of illness than patients without OCS.
Clinical and substance use variables in patients with a total Y-BOCS score 1–15 and ≥ 16, and patients without OCS.
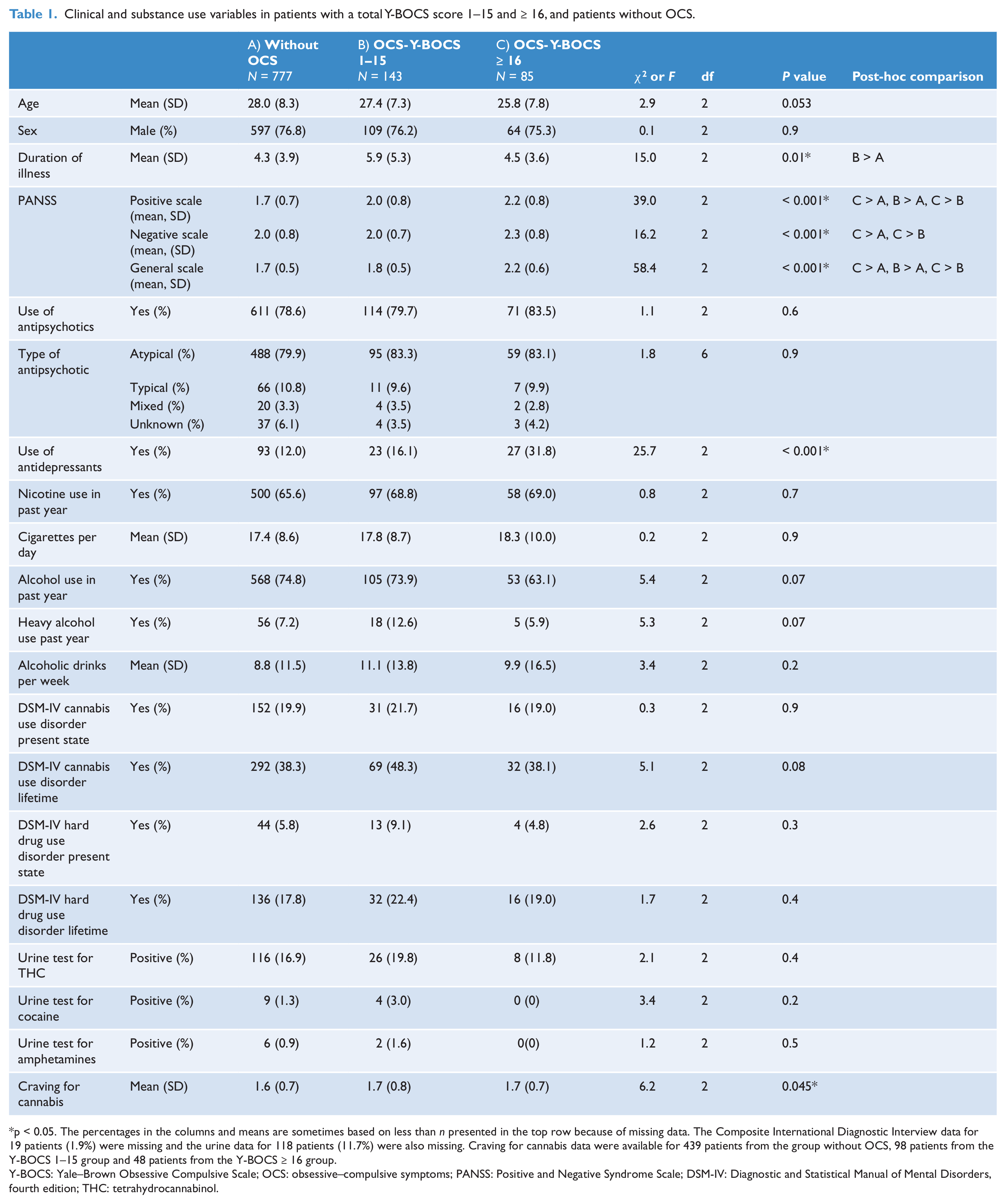
p < 0.05. The percentages in the columns and means are sometimes based on less than n presented in the top row because of missing data. The Composite International Diagnostic Interview data for 19 patients (1.9%) were missing and the urine data for 118 patients (11.7%) were also missing. Craving for cannabis data were available for 439 patients from the group without OCS, 98 patients from the Y-BOCS 1–15 group and 48 patients from the Y-BOCS ≥ 16 group.
Y-BOCS: Yale–Brown Obsessive Compulsive Scale; OCS: obsessive–compulsive symptoms; PANSS: Positive and Negative Syndrome Scale; DSM-IV: Diagnostic and Statistical Manual of Mental Disorders, fourth edition; THC: tetrahydrocannabinol.
DSM-IV diagnoses were: schizophrenia (66.2%), schizoaffective disorder (11.1%), schizophreniform disorder (5.5%), psychotic disorder not otherwise specified (10.2%) and other psychotic disorders (7.0%). The distribution of DSM-IV diagnoses across the three groups according to the level of OCS (no OCS, mild OCS, severe OCS) was not statistically different (χ2 = 9.7, p = 0.3).
Substance use characteristics
Substance use variables are summarized in Table 1. There were no significant differences between the three patients groups on smoking and other substance use variables, except for a statistically different score for cannabis craving in the past week (Kruskal–Wallis test: χ2 = 6.2, p = 0.045). However, post-hoc comparisons with Mann–Whitney tests did not survive the Bonferroni correction. Furthermore, there was a trend for the severe OCS group to be less likely to have used alcohol in the last 12 months compared to the other groups (no OCS or mild OCS). Lastly, there was a trend for the mild OCS group to be more likely to have a lifetime diagnosis of a cannabis use disorder use (p = 0.08) and to use alcohol heavily (p = 0.07), compared to the other groups (no OCS or Y-BOCS ≥ 16).
Discussion
In a large sample of patients with schizophrenia or related disorders we found no significant differences in substance use variables between patients with mild OCS (Y-BOCS score 1–15), patients with more severe OCS (Y-BOCS score ≥ 16) and patients without OCS. We found a trend for patients with more severe OCS to be less likely to have used alcohol in the last 12 months. On the contrary, there was a trend for patients with mild OCS to be more likely to use alcohol heavily and to have a lifetime diagnosis of cannabis use disorder. As these differences were all small and non-significant, we suggest that they are not clinically relevant.
We found no evidence in favour of our hypothesis that schizophrenia patients with co-morbid OCS have lower substance use rates than schizophrenia patients without OCS. Also, while OCD in the general population is clinically significantly associated with substance use, our data strongly suggest that this association is not mimicked in a population of schizophrenia patients. Taken together, our results strongly suggest that the presence of co-morbid OCS is not correlated with the use of nicotine and other substances in patients suffering from non-affective psychotic illness.
In agreement with our results, previous studies did not find differences in the prevalence rates of nicotine and substance use in schizophrenia patients with or without OCS (Dome et al., 2006; Fawzi et al., 2007; Poyurovsky et al., 2003; Puyorovsky et al., 2008). However, De Haan and colleagues (2013) did find smoking rates to be lower in schizophrenia patients who suffered from co-morbid OCD, compared to those without OCD. Given the fact that in this study there was also no difference in distribution of other substance use between groups, the finding of lower smoking rates in patients with OCD may be a chance finding. This interpretation is supported by the fact that lower prevalence of smoking in the co-morbid OCS group is not replicated in other studies, or in the current large study with detailed and comprehensive assessment of substance use.
Limitations
We acknowledge several limitations. First, the study was cross-sectional. As both the course of OCS/OCD and the course of substance use is variable during the course of schizophrenia (De Haan et al., 2013; Foti et al., 2010), it would be interesting to know whether the relationship between OCS and substance use varies over time. A longitudinal research perspective is needed to explore this relationship. Secondly, there were missing data concerning some substance variables, such as urine data and data on craving for cannabis (see the footnote to Table 1). However, we do believe that the percentage of missing data was relatively low and consequently enough data were available for a valid and reliable analysis in this study. Thirdly, we used the Y-BOCS as a measurement for the severity of OCS, but we made no formal diagnosis of OCD according to the DSM-IV (APA, 1994). By making a cut-off of the total Y-BOCS score of ≥ 16, we created a subgroup of severe OCS, which we believe will capture a large part of those patients with OCD. However, research is needed on the sensitivity and specificity of this cut-off on the Y-BOCS in patients with non-affective psychotic disorder, because these data are lacking. Lastly, subtypes of OCS (e.g. contamination/washing, harming/checking) were not assessed. It would be interesting to explore whether there is a relationship between types of OCS and alcohol, nicotine and other substance use.
Conclusion
Our large study sample and more detailed comparison of substance use rates strongly adds to the evidence that schizophrenia patients with OCS do not differ in the prevalence of substance use compared to patients without OCS. As substance use is highly prevalent in schizophrenia patients, it is important and relevant to enquire into it in both patients with co-morbid OCS and in patients without OCS.
Footnotes
Acknowledgements
The GROUP investigators are: René S Kahn and Wiepke Cahn, Department of Psychiatry, Rudolf Magnus Institute of Neuroscience, University Medical Center Utrecht, The Netherlands; Don H Linszen and Lieuwe de Haan, Academic Medical Centre University of Amsterdam, Department of Psychiatry, Amsterdam The Netherlands; Jim van Os, Lydia Krabbendam and Inez Myin-Germeys, Maastricht University Medical Centre, South Limburg Mental Health Research and Teaching Network, Maastricht The Netherlands; Durk Wiersma and Richard Bruggeman, University Medical Center Groningen, Department of Psychiatry, University of Groningen The Netherlands.
Funding
We are grateful for the generosity of time and effort by the patients and their families, healthy subjects and all researchers who make this GROUP project possible.
The infrastructure for the GROUP study is funded through the Geestkracht programme of the Dutch Health Research Council (ZON-MW, grant number 10-000-1002) and matching funds from participating universities and mental health-care organisations (Amsterdam: Academic Psychiatric Centre of the Academic Medical Centre and the mental health institutions: GGZ Ingeest, Arkin, Dijk en Duin, Rivierduinen, Erasmus Medical Centre, GGZ Noord Holland Noord; Maastricht: Maastricht University Medical Centre and the mental health institutions: GGZ Eindhoven, GGZ Midden-Brabant, GGZ Oost-Brabant, GGZ Noord- Midden Limburg, Mondriaan Zorggroep, Prins Clauscentrum Sittard, RIAGG Roermond, Universitair Centrum Sint-Jozef Kortenberg, CAPRI University of Antwerp, PC Ziekeren Sint-Truiden, PZ Sancta Maria Sint-Truiden, GGZ Overpelt, OPZ Rekem; Groningen: University Medical Center Groningen and the mental health institutions: Lentis, GGZ Friesland,GGZ Drenthe, Dimence, Mediant, GGZ De Grote Rivieren and Parnassia psycho-medical centre (The Hague); and Utrecht: University Medical Centre Utrecht and the mental health institutions Altrecht, Symfora, Meerkanten, Riagg Amersfoort, en Delta).
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
